Introduction
Intrahepatic cholangiocarcinoma (ICC) is an
aggressive disease representing the second most common liver
malignancy (1). The incidence of
ICC is increasing worlwide (2).
Surgery is considered a potential option with an overall 5-year
survival of about 25-30%. (3,4).
Gemcitabine-based chemotherapy (CT) represents the standard therapy
in unresectable or metastatic biliary cancers with reported median
overall survival (OS) of approximately 12 months (5,6)
Particularly, in case of unresectable ICC, both CT or concurrent
chemoradiation (CRT) (7,8) are treatment options.
Currently, different local therapies are available
for inoperable ICC. The role of trans-arterial chemo-embolization
(TACE) has been investigated (9). A
meta-analysis on TACE in ICC treatment has been recently published,
demonstrating a survival benefit compared to standard systemic
therapies, with a good toxicity profile (10) Similarly, radiofrequency ablation
resulted an effective treatment option in unresectable ICC. In
fact, it was investigated in a systematic review and meta-analysis,
reported 82 and 47% 1-year and 3-year survival rates, respectively
(11).
Stereotactic body radiation therapy (SBRT) is a
modern radiotherapy technique delivering ablative radiation doses
while sparing the surrounding normal tissues. Despite surrounding
organs at risk (OAR) and particularly liver radiosensitivity, SBRT
has been considered as a possible therapeutic option in inoperable
ICC. Two systematic reviews explored the role of SBRT in
cholangiocarcinoma, but systematic reviews specifically addressed
to ICC are lacking (12,13). However, the latter have different
characteristics compared to tumors in other sites of the biliary
tract, both in terms of routes of spread and clinical management.
Therefore, even considering the scarce availability of clear
guidelines in this setting, we considered it important to analyze
the role of this technique (effective in other gastrointestinal
neoplasms, such as HCC and pancreatic carcinomas) in the specific
field of intrahepatic biliary carcinomas. The aim of this analysis
is to present a systematic review of the available literature in
the setting of SBRT in ICC.
Materials and methods
Inclusion criteria
We included in this analysis prospective and
retrospective studies reporting OS after SBRT in inoperable ICC.
Only English language written papers were considered. Only studies
enrolling at least 10 patients with inoperable, non-metastatic,
primary, or recurrent ICC were considered. Papers including
hepatocellular carcinoma (HCC) patients were considered only if
results on subjects with ICC were reported separately. Studies
regarding SBRT planned and performed with any technique and device
were included in the analysis even if preceded or followed by
systemic treatments or other local therapies.
Studies on adjuvant SBRT after radical surgery were
excluded. Analyses on preclinical models, planning studies, study
protocols, case reports, letters and commentaries, editorials,
systematic reviews, and meta-analyses were excluded. Primary
endpoint of the analysis was OS. Secondary endpoints were local
control (LC), progression-free survival (PFS), and treatment
related toxicity.
Literature search strategy
A systematic search was performed on PubMed database
on June 1st 2020 including papers published from the earliest date
until 31st May 2020. The following search strategy was used:
(‘cholangiocarcinoma’ OR ‘cholangiocarcinomas’ OR (‘intrahepatic’
AND ‘cholangiocarcinoma’) OR ‘intrahepatic cholangiocarcinoma’) AND
(radiosurgery OR ‘stereotactic body radiotherapy’ OR ‘stereotactic
body radiation therapy’ OR ‘stereotactic body radiosurgery’ OR
‘stereotactic radiotherapy’ OR ‘stereotactic radiation therapy’ OR
‘stereotactic radiosurgery’ OR stereotaxis OR stereotaxic OR sbrt
OR sbrs OR sabr OR ‘stereotactic ablative’). The analysis was based
on the PRISMA methodology (14).
From the first search on PubMed, title, abstracts,
and keywords were independently analyzed by two authors (ACC, FB).
Any disagreement was solved by the senior author (RA). A second
search based on previous reviews and meta-analyses was performed by
two other authors (SB, AA). Full-text evaluation based on the
above-mentioned inclusion criteria was independently carried out by
the latter two authors. Data on tumor, treatments characteristic,
and outcomes were separately collected by two authors (SB and FB),
with the supervision of AA. Any disagreement was resolved by a
consensus-based discussion with the 2 senior authors (SiC and MB).
Draft correction and critical revision were subsequently performed
by all the author, as expressed below.
Results
Search result
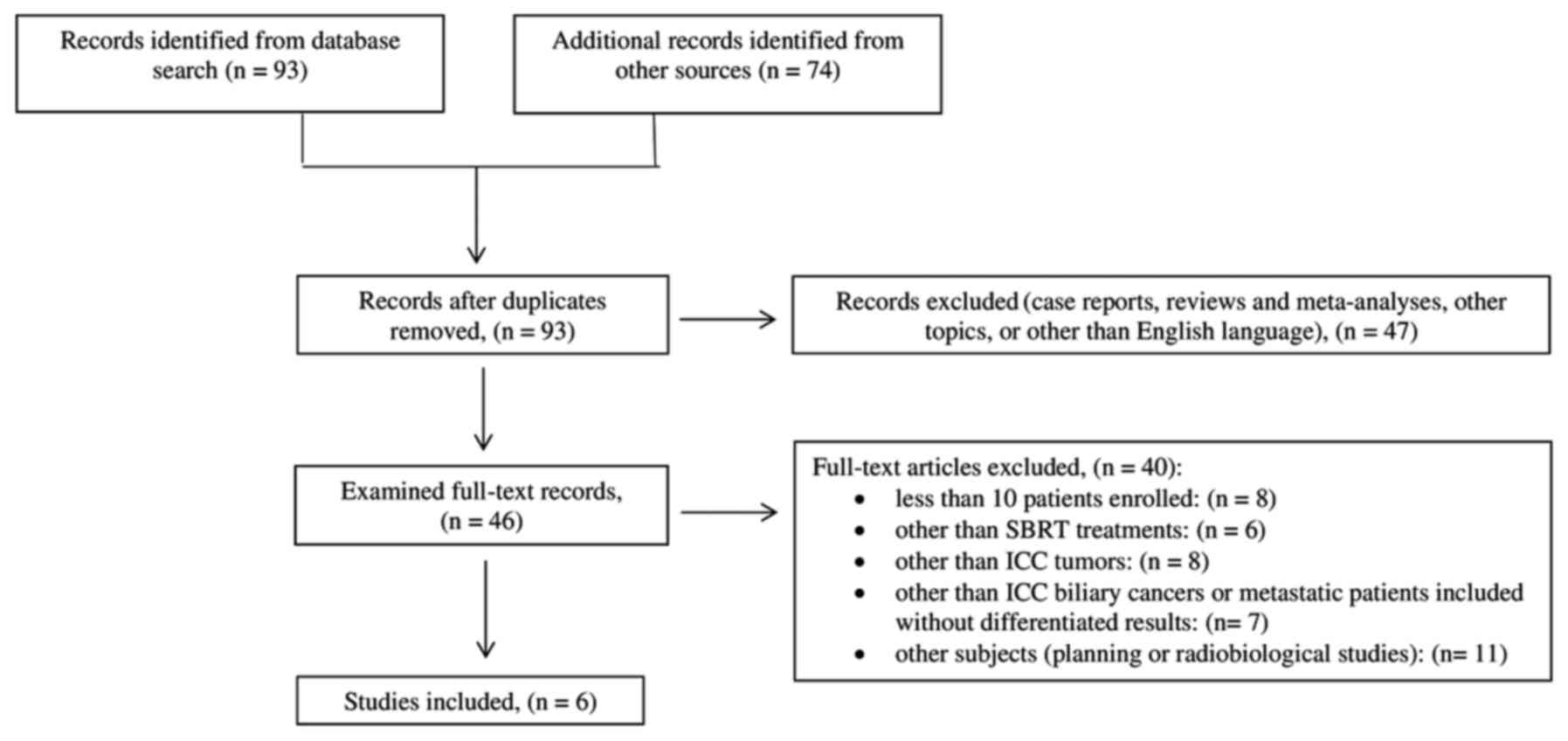
Ninety-three papers were identified from PubMed.
Other 74 papers were identified by checking the references list.
After duplicates removal, based on a first screening of titles and
abstracts, 47 records were excluded. Forty-six papers were
subjected to full-text evaluation and 40 were excluded for the
reasons described in Fig. 1. Only 6
papers met our inclusion criteria. The flowchart of papers
selection is detailed in Fig.
1.
Literature review
Shen et al (15) published the results of a prospective
analysis on 28 patients with locally advanced, non-metastatic ICC.
All patients underwent SBRT (median prescribed dose: 45 Gy),
delivered in three to five fractions using a robotic linear
accelerator. Median OS and PFS were 15 and 11 months, respectively.
A total of 15 patients (53.7%) manifested grade ≥3 acute toxicity.
A higher overall response rate was recorded in patients receiving a
biological effective dose (BED) >100 Gy (52.4 vs. 0.0%,
P=0.023).
Weiner et al (16) published the results of a phase I/II
trial on SBRT in patients with unresectable HCC or ICC. Twelve
patients had ICC and two had a bi-phenotypic disease. Median dose
was 55 Gy delivered in 5 fractions. With a median follow-up of 8.8
months, median and 1-year OS were 13.2 months and 51.0%,
respectively. Seventeen patients (65.4%) showed grade ≥3 acute
toxicity in the whole population (HCC and ICC patients).
Klein et al (17) reported a prospective assessment of
quality of life in patients with liver tumors treated with SBRT
(24-60 Gy in 6 fractions). Physical and functional items were
recorded during and after treatment. Median OS was 12.1 months in
the ICC sub-group (20 patients).
Jung et al (18) retrospectively analyzed 58 patients
with primary or recurrent intra- or extra-hepatic
cholangiocarcinoma treated with SBRT. Thirty-three patients had
ICC. Considering the whole population included in the analysis, 91%
(53/58) of patient received SBRT alone (median dose: 45 Gy in 3-5
fractions) while five patients (9%) received external beam
radiotherapy plus SBRT boost (15 to 18 Gy in a single fraction).
With a median follow-up of 10 months, 1-year OS and PFS were 45 and
26%, respectively. Two patients from the whole population
experienced grade 3 acute toxicity. During follow-up, four patients
showed grade 4 late toxicity and two patients with cholangitis and
bile duct stenosis required hospitalization. Considering only
patients with ICC, median OS was 10 months and actuarial 1- and
2-year OS were 39.0 and 18.0%, respectively.
Sebastian et al (19) retrospectively analyzed 141 patients
with inoperable non-metastatic ICC treated with SBRT (27 patients),
or TARE (60 patients), or CRT (54 patients). With a median follow
up of 17 months, median OS was 48 months in the SBRT group, while
median OS in TARE and CRT groups were 20 and 14 months,
respectively. At multivariate analysis, after propensity weighting
adjustment, SBRT was correlated with higher OS compared to CRT
(P<0.0001) and TARE (P<0.019).
Kozak et al (20) retrospectively collected data on 40
patients treated with SBRT for unresectable intrahepatic (25
patients) or hilar (15 patients) cholangiocarcinoma. For the entire
group of patients, 25% were previously treated with CT, 20.0% with
surgery, 7.5% with radiofrequency ablation, and 12.5% with TACE.
Median dose was 40 Gy (range: 26-50) in five fractions. In ICC
patients, with a median follow-up of 18 months, median OS was 23
months while in-field local failure and regional failure rates were
40 and 8%, respectively. Considering the whole group, both acute
and late grade ≥3 hepatobiliary toxicity were recorded in 17
patients.
Analysis of selected studies
Six studies were included in this analysis reporting
data on 145 patients. Four studies were retrospective (15,18-20)
one was prospective (17), and one
was a phase I/II trial (16).
Patients were treated between 2003 and 2017. Studies'
characteristics are shown in Table
I.
 | Table ICharacteristics of the studies
included in the systematic review. |
Table I
Characteristics of the studies
included in the systematic review.
| Author, year | Study design | Accrual period | No. of
patients | Median follow-up
(range), months | ICC features,
% | Treatment before
SBRT, % | (Refs.) |
|---|
| Shen et al,
2017 | Retrospective | 2009-2012 | 28 | 16.0
(3.0-42.0) | Peripheral lesions:
78.6, Central lesions: 21.4 | TACE: 28.6 | (15) |
| | | | | | Solitary lesions:
71.4 | | |
| | | | | | Multiple lesions:
28.6 | | |
| | | | | | AJCC stage: II
21.4, III | | |
| | | | | | 67.9, IVA 10.7 | | |
| Weiner et
al, 2016 | Phase I-II | 2012-2014 | 12 | 8.8 (0.3-33.0) | NRS | CHT: 66.7 | (16) |
| Klein et al,
2015 | Prospective | 2003-2011 | 20 | NR | NR | NR | (17) |
| | | | | | | | |
| Jung et al,
2014 | Retrospective | 2005-2013 | 33 | 10.0
(1.0-97.0) | NRS | NRS | (18) |
| Sebastian et
al, 2019 | Retrospective | 2004-2014 | 27 | 17.0 (NR) | AJCC stage: T1
59.3; T2 25.9; T3 11.1; T4 3.7 | CHT: 40.7 | (19) |
| Kozak et al,
2020 | Retrospective | 2003-2017 | 25 | 18.0 (NR) | Positive nodes:
15.0 | CHT: 24.0 | (20) |
| | | | | | | Surgery: 20.0 | |
| | | | | | | | |
| | | | | | | RFA: 7.5 | |
| | | | | | | TACE: 12.5 | |
Median follow-up was reported in five studies
(15,16,18-20)
and ranged between 8.8 and 18.0 months (median: 16 months). In two
studies (15,19) ICC characteristics were described in
detail. In one report, treated lesions were classified as solitary
or multiple and as central or peripheric (15). The same paper reported the disease
stage as follows: Stage II: 21.4%, stage III: 67.9%, and stage IVA:
10.7% (15). Another paper reported
the tumor stages as follows: T1: 59.3%, T2:
25.9%, T3: 11.1%, and T4: 3.7% (19). In one study a 15.0% rate of patients
with nodal involvement was reported (20).
Most patients included in this analysis were
previously treated with local or systemic therapies. In one trial
28.6% of patients were previously treated with TACE (15). Two studies enrolled patients
previously treated with CT in 66.7% (16) and 40.7% (19) of cases, respectively. Another paper
reported on patients who received several previous therapies: CT
(24.0%), surgery (20.0%), radiofrequency ablation (7.5%), and TACE
(12.5%) (20).
Table II
synthesizes SBRT planning and delivery characteristics. In three
studies, the planning target volume (PTV) was defined as the gross
tumor volume (GTV) plus 5 mm margin (15-17),
while in two studies, the PTV was defined as the internal target
volume (ITV) plus 2-4 mm margin (18,20).
Three studies reported details on dose prescription (15,16,18).
The 95% isodose line had to cover 95% of the PTV in 2 studies
(15,16), while in one study the 92-99% isodose
line had to cover 95% of PTV (18).
 | Table IITreatment planning and dose
delivery. |
Table II
Treatment planning and dose
delivery.
| Author, year | Target
definition | Median GTV (range),
ml | Dose
prescription | Median dose,
Gy/fractions | Dose range, Gy | Median
BED10Gy (range) | (Refs.) |
|---|
| Shen et al,
2017 | PTV: GTV + 5
mm | NR | 100% isodose line
to cover | 45/3-5 | 36-54 | 85.5
(72.0-124.8) | (15) |
| | | | GTV 95% isodose
line to cover PTV | | | | |
| Weiner et
al, 2016 | PTV: GTV + 5
mm | 107.0
(16.9-625.9)a | 95% isodose line to
cover 95% PTV | 55/5 | 40-55 | 99.0
(72.0-115.5) | (16) |
| Klein et al,
2015 | PTV: GTV + 5
mm | NR | NR | NR/6 | 24-60a | NRS | (17) |
| Jung et al,
2014 | PTV: ITV + 2-4
mm | 40.0
(5.0-1287.0) | 70-80% isodose line
(Cyber Knife) 92-99% isodose line to cover 95% PTV (Rapid Arc) | 45/3-5b EBRT + SBRT boost, 16/1 | 30-60b EBRT + SBRT boost, 15-18 | 86.0
(48.0-150.0) | (18) |
| Sebastian et
al, 2019 | NR | NR | NR | 45/5 | 40-50 | NR | (19) |
| Kozak et al,
2020 | PTV: ITV + 2-3
mma | NR | NR | 40/5 | 26-50a | NR | (20) |
Median prescribed dose was reported in five studies
(15,16,18-20),
ranging between 45 and 55 Gy (median 45 Gy), delivered in 3-5
fractions. The biological equivalent dose (BED) was reported in 3
studies and ranged between 85.5 and 99.0 Gy (median: 86.0 Gy)
(15,16,18).
Only one study reported actuarial LC data, with 85
and 71.0% 1- and 2-year LC rates, respectively (18). Moreover, one study reported 40 and
8% local and regional failure crude rates. Respectively (20). Two papers reported 1-year and median
PFS. One-year PFS was 50% (15) and
68% (16) while median PFS was
11(15) and 24.7 months (16).
Actuarial OS was reported in three studies with
1-year rates ranging between 39 and 57% (median value 51%)
(15,16,18).
Furthermore, two-year OS rates were 32% (15) and 18% (18) in two studies. Median OS was reported
in all papers and ranged between 10 and 48 months (median: 14
months) (15-20).
Toxicity was generally not described in detail. One paper reported
a broad range of grade ≥3 gastrointestinal and hematological
toxicities while three papers reported toxicity related to the
entire cohort (16,18,20).
Two of these papers (16,18) reported grade ≥3 acute toxicity in
3.4% (18) and 65.4% (16) of patients and grade ≥3 late toxicity
in 6.9% (18) and 69.2% (16) of patients, respectively. Another
paper reported grade ≥3 acute and late hepatobiliary toxicity, both
recorded in 42.5% of cases (20).
Only two papers reported tumor response (15,16).
The first one, according to the RECIST criteria, reported 10.7,
35.7, 42.9 and 10.7% rates of complete response, partial response,
stable disease, and progressive disease, respectively (15). In the other paper, according to
European Association for the Study of the Liver criteria, 33.3% of
patients had a radiographic response and 8.3% had a complete
radiographic response (16).
Table III summarizes the
treatment outcomes.
 | Table IIISurvival outcomes. |
Table III
Survival outcomes.
| Author, year | LC rate,% | PSF rate, % | Median PFS time,
months | OS rate, % | Median OS time,
months | Toxicity
criteria | Acute toxicity,
grade ≥3, % (n) | Late toxicity,
grade ≥3, % (n) | Tumour response,
% | (Refs.) |
|---|
| Shen et al,
2017 | NR | 1-year: 50.0
2-year: 21.4 | 11.0 | 1-year: 57.1
2-year: 32.1 | 15.0 | CTCAE v.4.0 | Nausea: 3.6(1)
Anorexia: 7.1(2) Vomiting: 3.6(1) Gastric ulcer: 3.6(1) Altered
hepatic values: 25.0(7) Haematological toxicity: 10.8(3) | NR | Complete response:
10.7 Partial response: 35.7 Stable disease: 42.9 Progressive
disease: 10.7 (RECIST criteria) | (15) |
| Weiner et
al, 2016 | NRS | 1-year: 68.0 | 24.7 | 1-year: 51.0 | 13.2 | CTCAE v.4.0 |
65.4(17)a |
69.2(18)a | Radiographic
response: 33.3 Complete radiographic response: 8.3 (EASL
criteria) | (16) |
| Klein et al,
2015 | NR | NR | NR | NR | 12.1 | NR | NR | NR | NR | (17) |
| Jung et al,
2014 | 1-year: 85.0
2-year: 71.0 | NR | NR | 1-year: 39.0
2-year: 18.0 | 10.0 | CTCAE v.4.0 | 3.4(2)a | 6.9(4)a | NR | (18) |
| Sebastian et
al, 2019 | NR | NR | NR | NR | 48.0 | NR | NR | NR | NR | (19) |
| Kozak et al,
2020 | Local failures:
40.0 Regional failures: 8.0 | NR | NR | NR | 23.0 | NR | Hepatobiliary:
42.5(17)a
Non-hepatobiliary: 2.5(1)a | Hepatobiliary:
42.5(17)a
Non-hepatobiliary: 2.5(1)a | NR | (20) |
Discussion
To the best of our knowledge, this is the first
systematic review on SBRT in the treatment of inoperable,
non-metastatic, ICC.
ICC is considered as a rare malignancy in the
western world and the management of locally advanced disease is
still debated. CT is the standard therapy for advanced ICC
(5,6) but the prognosis is still poor. SBRT
allows the delivery of ablative doses to the tumor with minimal
irradiation of the surrounding tissues. For this reason, some
researchers have proposed SBRT as a possible therapeutic option in
biliary tract cancer (12,13,21-23).
Furthermore, the intrahepatic site of these tumours, therefore
within an organ with parallel functional organization, generally
far from serial organs unlike extrahepatic biliary tumours, makes
ICCs particularly suitable to be treated with SBRT.
Our study has obvious limitations. Firstly, the
selection of papers led to the evaluation of only 6 studies. More
evidence is available on SBRT in HCC. However, we selected only
papers on ICC due to the higher liver radiation sensitivity of HCC
patients. In fact, the latter often present an impaired liver
function or a cirrhotic status that can lead to higher complication
rate. In those cases, a more conservative approach in volume
definition or dose delivery may be needed.
The majority of these papers had a retrospective
design (15,18-20).
Moreover, the number of patients enrolled in the various studies
was limited (range of patients enrolled, 12-33 patients).
Furthermore, several data were lacking thus limiting the
possibility to compare the results. Information on the disease
stage (15,19) and treatments prior to SBRT (15,16,19,20)
was present only in a few of the papers. Finally, the case series
were very heterogeneous in terms of patient characteristics,
treatment planning and delivery methods, total dose, fractionation,
and results evaluation. The latter issue severely limits the
possibility of an inherent comparison between the analyzed studies
and among SBRT and other treatments results.
However, there are some worth noting considerations
from our review. The best median OS (48 months) was reported in the
study of Sebastian et al (19). This result can be justified by the
favorable tumor stage (cT1-2 in 85.2% of patients).
Therefore, this data suggests that OS can be particularly favorable
in patients with less extensive ICC. In fact, 5/37 patients
undergoing SBRT had >5 years OS.
From the comparison between the case series with the
highest median SBRT dose (55 Gy) (16) and those with the lowest median dose
(40 Gy) (20), it can be observed
that median OS was 13.2 months in the former and 23 months in the
latter. This comparison seems to suggest the lack of a positive
correlation between delivered dose and patients outcome. However,
this result contrasts with a study on 79 patients with inoperable
ICC where a significant relationship between delivered dose and LC
and OS was recorded (24). This
difference can be explained by the use of 15-30 fractions in most
patients in the latter study.
In terms of toxicity, an analytical description of
acute toxicity has been reported only in the paper by Shen et
al (15). Considering
gastrointestinal toxicity, two out of 28 enrolled patients had
anorexia, one nausea, one vomiting, and one a gastric ulcer
(15). This finding seems to
suggest a reasonable tolerability of SBRT in this setting,
particularly in terms of low gastrointestinal toxicity rates.
Three papers reported the overall percentage of
patients with grade ≥3 acute toxicity, with a wide range of
incidence (3.4-65.4%) (16,18,20).
It can be observed that the highest acute toxicity rate was
observed in the series with the highest median delivered dose
(16). Furthermore, in the series
with the worst acute toxicity, the median GTV volume was 107.0 ml
(16) while in the series with the
lower acute toxicity rate, the median PTV volume was 40.0 ml
(18). Comparing late toxicity, the
rates recorded in the two series were 69.2% (16) and 6.9% (18) respectively. Even within the limits
of a comparison between different series, these data would suggest
a negative impact on toxicity of higher doses and larger irradiated
volumes.
Further considerations can be made by comparing the
results of our review with those of other treatments of locally
advanced ICC. In particular, our analysis showed that ICC patients
undergoing SBRT had a median OS of 14.1 months (range, 10-48
months). However, it should be noted that SBRT was frequently used
as a salvage treatment, since 28.6-66.7% of patients received
previous systemic or local treatments (15,16,19,20).
This may have affected the results recorded in our analysis.
CT is a standard treatment of advanced ICC. In a
series of 26 patients with unresectable ICC, median OS was 18.4
months, slightly higher compared to our analysis (25). However, it should be considered
that, in that study, CT was used as an up-front treatment while
SBRT was often used as a salvage therapy in our analysis. That same
publication reported 30.8 months median OS (P<0.001) in another
group of patients undergoing CT plus intrahepatic arterial infusion
(25). This data suggests that
combinations of systemic and local therapies may improve outcome in
patients with advanced ICC.
Concurrent CRT is another therapeutic option for
advanced non-metastatic cholangiocarcinoma, and proper target
volume definition could achieve good results (26). Median OS was 13.5 months in a
retrospective multicentric study on 76 patients with unresectable
biliary cancer (8). Moreover, the
results on 27 patients with advanced cholangiocarcinoma who
underwent CRT +/- brachytherapy boost were reported in another
study (27). Median OS was similar
(14 months) and the toxicity grade ≥3 rate was 37%. Although
separate data for ICC patients were not reported in these studies,
the results seem to suggest that outcomes after SBRT are similar to
those of CRT, with a clear benefit in terms of shorter treatment
duration for SBRT.
Trans-catheter arterial chemoembolization is another
treatment for biliary tract cancer. A meta-analysis on unresectable
cholangiocarcinoma reported 13.4 months median OS from the time of
first treatment. Severe toxicity was recorded in 18.9% of patients
with 0.7% rate of 30-days mortality rate (10). Again, there is no clear difference
with the results of our analysis on SBRT.
Similarly, a meta-analysis of 12 studies on
radio-embolization with 99-Ittrium microsphere reported
15.5 months median weighted OS. Also this result is similar to the
one of our analysis on SBRT. Toxicity was mainly represented by
fatigue (33%), pain (28%) and nausea (25%) (28).
Finally, some recent studies evaluated the potential
role of proton beam radiation therapy in cholangiocarcinoma. In a
retrospective study on unresectable or locally recurrent ICC, 34
patients were treated with photon-based radiotherapy while 32
patients were treated with protons. Two-year OS was 49 and 65%,
respectively. At multivariate analysis, compared to photon-based
radiotherapy, there was a trend for improved OS with protons (HR:
0.50; p: 0.05) (29). In another
retrospective analysis (30) on 30
patients with unresectable cholangiocarcinoma treated with
proton-therapy (median dose: 72.6 cobalt Gy equivalent) median OS
was 19.3 months. However, 10% of patients developed grade ≥3
gastric/duodenal ulcers. This irradiation modality deserves further
study in the ICC setting, based on the theoretical advantages of
proton therapy in terms of dose distribution. However, the
currently available results do not allow a comparison with
SBRT.
In conclusion, although other options are available
in this setting, preliminary literature data on SBRT in ICC seems
to suggest that this treatment could be considered as a possible
therapeutic option.
Further studies are needed in this field. These
analyzes could be aimed to the: i) assessment of efficacy and
tolerability of SBRT in ICC by large and prospective trials; ii)
definition of optimal dose, fractionation and GTV to PTV margins in
this setting; iii) comparison between SBRT and other ICC local
treatments; considering the palliative aim of advanced ICC
therapies, these studies should include an accurate assessment of
treatments impact on quality of life; iv) feasibility and efficacy
of combined modality treatments including SBRT (for example: SBRT
combined with systemic therapies; SBRT as a boost after concurrent
chemoradiation).
Acknowledgements
The abstract was presented as an electronic poster
at the virtual XXX Congresso Nazionale Associazione Italiana
Radioterapia Oncologia on 9-12th December 2020.
Funding
Availability of data and materials
The datasets used and/or analyzed during the current
study are available from the corresponding author on reasonable
request.
Authors' contributions
SB, MB, GMac, GMat, GB, LS, FD and AGM conceived and
organized the study. FB, AC, SB, AA and RA selected the papers and
acquired the data. SB, FB, MB, SCa, SCi and AA analysed and
interpreted the data. AC, SB, MB, RA, FD, SCi and AGM drafted the
manuscript. SCa, FD and SCi assessed the authenticity of the raw
data. SCa, FD, GB, AGM, GMat and GMac critically revised the
manuscript for important intellectual content. SB, AGM, MB, FD and
SCi discussed and reviewed the latest changes of the manuscript.
All authors read and approved the final manuscript.
Ethics approval and consent to
participate
Not applicable.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Shaib YH, Davila JA, McGlynn K and
El-Serag HB: Rising incidence of intrahepatic cholangiocarcinoma in
the United States: A true increase? J Hepatol. 40:472–477.
2004.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Saha SK, Zhu AX, Fuchs CS and Brooks GA:
Forty-year trends in cholangiocarcinoma incidence in the U.S.:
Intrahepatic disease on the rise. Oncologist. 21:594–599.
2016.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Endo I, Gonen M, Yopp AC, Dalal KM, Zhou
Q, Klimstra D, D'Angelica M, dematteo RP, Fong Y, Schwartz L, et
al: Intrahepatic cholangiocarcinoma: Rising frequency, improved
survival, and determinants of outcome after resection. Ann Surg.
248:84–96. 2008.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Chou FF, Sheen-Chen SM, Chen YS, Chen MC
and Chen CL: Surgical treatment of cholangiocarcinoma.
Hepatogastroenterology. 44:760–765. 1997.PubMed/NCBI
|
|
5
|
Valle JW, Wasan H, Johnson P, Jones E,
Dixon L, Swindell R, Baka S, Maraveyas A, Corrie P, Falk S, et al:
Gemcitabine alone or in combination with cisplatin in patients with
advanced or metastatic cholangiocarcinomas or other biliary tract
tumours: A multicentre randomised phase II study- the UK ABC-01
study. Br J Cancer. 101:621–627. 2009.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Okusaka T, Nakachi K, Fukutomi A, Mizuno
N, Ohkawa S, Funakoshi A, Nagino M, Kondo S, Nagaoka S, Funai J, et
al: Gemcitabine alone or in combination with cisplatin in patients
with biliary tract cancer: A comparative multicentre study in
Japan. Br J Cancer. 103:469–474. 2010.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Bisello S, Buwenge M, Zamagni A, Deodato
F, Macchia G, Alessandra A, Cammelli S, Mattiucci GC, Cellini F and
Morganti AG: Chemoradiation in unresectable biliary tract cancer: A
systematic review. Hepatol Pancreat Sci 2018.
|
|
8
|
Bisello S, Buwenge M, Palloni A, Autorino
R, Cellini F, Macchia G, Deodato F, Cilla S, Brandi G, Tagliaferri
L, et al: Radiotherapy or chemoradiation in unresectable biliary
cancer: A retrospective study. Anticancer Res. 39:3095–3100.
2019.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Zechlinski JJ and Rilling WS:
Transarterial therapies for the treatment of intrahepatic
cholangiocarcinoma. Semin Intervent Radiol. 30:21–27.
2013.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Ray CE Jr, Edwards A, Smith MT, Leong S,
Kondo K, Gipson M, Rochon PJ, Gupta R, Messersmith W, Purcell T and
Durham J: Metaanalysis of survival, complications, and imaging
response following chemotherapy-based transarterial therapy in
patients with unresectable intrahepatic cholangiocarcinoma. J Vasc
Interv Radiol. 24:1218–1226. 2013.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Han K, Ko HK, Kim KW, Won HJ, Shin YM and
Kim PN: Radiofrequency ablation in the treatment of unresectable
intrahepatic cholangiocarcinoma: Systematic review and
meta-analysis. J Vasc Interv Radiol. 26:943–948. 2015.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Frakulli R, Buwenge M, Macchia G, Cammelli
S, Deodato F, Cilla S, Cellini F, Mattiucci GC, Bisello S, Brandi
G, et al: Stereotactic body radiation therapy in
cholangiocarcinoma: A systematic review. Br J Radiol.
92(20180688)2019.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Lee J, Yoon WS, Koom WS and Rim CH:
Efficacy of stereotactic body radiotherapy for unresectable or
recurrent cholangiocarcinoma: A meta-analysis and systematic
review. Strahlenther Onkol. 195:93–102. 2019.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Hutton B, Salanti G, Caldwell DM, Chaimani
A, Schmid CH, Cameron C, Ioannidis JP, Straus S, Thorlund K, Jansen
JP, et al: The PRISMA extension statement for reporting of
systematic reviews incorporating network meta-analyses of health
care interventions: Checklist and explanations. Ann Intern Med.
162:777–784. 2015.PubMed/NCBI View
Article : Google Scholar
|
|
15
|
Shen ZT, Zhou H, Li AM, Li B, Shen JS and
Zhu XX: Clinical outcomes and prognostic factors of stereotactic
body radiation therapy for intrahepatic cholangiocarcinoma.
Oncotarget. 8:93541–93550. 2017.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Weiner AA, Olsen J, Ma D, Dyk P, dewees T,
Myerson RJ and Parikh P: Stereotactic body radiotherapy for primary
hepatic malignancies-Report of a phase I/II institutional study.
Radiother Oncol. 121:79–85. 2016.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Klein J, Dawson LA, Jiang H, Kim J,
Dinniwell R, Brierley J, Wong R, Lockwood G and Ringash J:
Prospective longitudinal assessment of quality of life for liver
cancer patients treated with stereotactic body radiation therapy.
Int J Radiat Oncol Biol Phys. 93:16–25. 2015.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Jung DH, Kim MS, Cho CK, Yoo HJ, Jang WI,
Seo YS, Paik EK, Kim KB, Han CJ and Kim SB: Outcomes of
stereotactic body radiotherapy for unresectable primary or
recurrent cholangiocarcinoma. Radiat Oncol J. 32:163–169.
2014.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Sebastian NT, Tan Y, Miller ED, Williams
TM and Alexandra Diaz D: Stereotactic body radiation therapy is
associated with improved overall survival compared to
chemoradiation or radioembolization in the treatment of
unresectable intrahepatic cholangiocarcinoma. Clin Transl Radiat
Oncol. 19:66–71. 2019.PubMed/NCBI View Article : Google Scholar
|
|
20
|
Kozak MM, Toesca DAS, von Eyben R, Pollom
EL and Chang DT: Stereotactic body radiation therapy for
cholangiocarcinoma: Optimizing locoregional control with elective
nodal irradiation. Adv Radiat Oncol. 5:77–84. 2019.PubMed/NCBI View Article : Google Scholar
|
|
21
|
Mahadevan A, Dagoglu N, Mancias J, Raven
K, Khwaja K, Tseng JF, Ng K, Enzinger P, Miksad R, Bullock A and
Evenson A: Stereotactic body radiotherapy (SBRT) for intrahepatic
and Hilar cholangiocarcinoma. J Cancer. 6:1099–1104.
2015.PubMed/NCBI View Article : Google Scholar
|
|
22
|
Brunner TB, Blanck O, Lewitzki V,
Abbasi-Senger N, Momm F, Riesterer O, Duma MN, Wachter S, Baus W,
Gerum S, et al: Stereotactic body radiotherapy dose and its impact
on local control and overall survival of patients for locally
advanced intrahepatic and extrahepatic cholangiocarcinoma.
Radiother Oncol. 132:42–47. 2019.PubMed/NCBI View Article : Google Scholar
|
|
23
|
Liu MY, Lo CH, Lin CS, Chao HL, Yang JF,
Lin KT, Fan CY, Su YF and Huang WY: Stereotactic ablative
radiotherapy for patients with unresectable or medically inoperable
cholangiocarcinoma. Tumori. 103:236–241. 2017.PubMed/NCBI View Article : Google Scholar
|
|
24
|
Tao R, Krishnan S, Bhosale PR, Javle MM,
Aloia TA, Shroff RT, Kaseb AO, Bishop AJ, Swanich CW, Koay EJ, et
al: Ablative radiotherapy doses lead to a substantial prolongation
of survival in patients with inoperable intrahepatic
cholangiocarcinoma: A retrospective dose response analysis. J Clin
Oncol. 34:219–226. 2016.PubMed/NCBI View Article : Google Scholar
|
|
25
|
Konstantinidis IT, Koerkamp BG, Do RK,
Gönen M, Fong Y, Allen PJ, D'Angelica MI, Kingham TP, Dematteo RP,
Klimstra DS, et al: Unresectable intrahepatic cholangiocarcinoma:
Systemic plus hepatic arterial infusion chemotherapy is associated
with longer survival in comparison with systemic chemotherapy
alone. Cancer. 122:758–765. 2016.PubMed/NCBI View Article : Google Scholar
|
|
26
|
Bisello S, Renzulli M, Buwenge M, Calculli
L, Sallustio G, Macchia G, Deodato F, Mattiucci G, Cammelli S,
Arcelli A, et al: An atlas for clinical target volume definition,
including elective nodal irradiation in definitive radiotherapy of
biliary cancer. Oncol Lett. 17:1784–1790. 2019.PubMed/NCBI View Article : Google Scholar
|
|
27
|
Autorino R, Mattiucci GC, Ardito F,
Balducci M, Deodato F, Macchia G, Mantini G, Perri V, Tringali A,
Gambacorta MA, et al: Radiochemotherapy with gemcitabine in
unresectable extrahepatic cholangiocarcinoma: Long-term results of
a phase II study. Anticancer Res. 36:737–740. 2016.PubMed/NCBI
|
|
28
|
Al-Adra DP, Gill RS, Axford SJ, Shi X,
Kneteman N and Liau SS: Treatment of unresectable intrahepatic
cholangiocarcinoma with yttrium-90 radioembolization: A systematic
review and pooled analysis. Eur J Surg Oncol. 41:120–127.
2015.PubMed/NCBI View Article : Google Scholar
|
|
29
|
Smart AC, Goyal L, Horick N, Petkovska N,
Zhu AX, Ferrone CR, Tanabe KK, Allen JN, Drapek LC, Qadan M, et al:
Hypofractionated radiation therapy for unresectable/locally
recurrent intrahepatic cholangiocarcinoma. Ann Surg Oncol.
27:1122–1129. 2020.PubMed/NCBI View Article : Google Scholar
|
|
30
|
Hung SP, Huang BS, Hsieh CE, Lee CH, Tsang
NM, Chang JT, Chen JS, Chou WC, Tseng JH and Hong JH: Clinical
outcomes of patients with unresectable cholangiocarcinoma treated
with proton beam therapy. Am J Clin Oncol Cancer Clin Trials.
43:180–186. 2020.PubMed/NCBI View Article : Google Scholar
|
|
31
|
Edge BS and Compton CC: American joint
committee on cancer: The 7th edition of the AJCC cancer staging
manual and the future of TNM. Ann Surg Oncol. 17:1471–1474.
2010.PubMed/NCBI View Article : Google Scholar
|
|
32
|
Trotti A, Colevas AD, Setser A, Rusch V,
Jaques D, Budach V, Langer C, Murphy B, Cumberlin R, Coleman CN and
Rubin P: CTCAE v3.0: Development of a comprehensive grading system
for the adverse effects of cancer treatment. Semin Radiat Oncol.
13:176–181. 2003.PubMed/NCBI View Article : Google Scholar
|
|
33
|
Bruix J, Sherman M, Llovet JM, Beaugrand
M, Lencioni R, Burroughs AK, Christensen E, Pagliaro L, Colombo M
and Rodés J: EASL Panel of Experts on HCC. Clinical management of
hepatocellular carcinoma. Conclusions of the Barcelona-2000 EASL
conference. European association for the study of the liver. J
Hepatol. 35:421–430. 2001.PubMed/NCBI View Article : Google Scholar
|
|
34
|
Schwartz LH, Litière S, de Vries E, Ford
R, Gwyther S, Mandrekar S, Shankar L, Bogaerts J, Chen A, Dancey J,
et al: RECIST 1.1-update and clarification: From the RECIST
committee. Eur J Cancer. 62:132–137. 2016.PubMed/NCBI View Article : Google Scholar
|