Introduction
Recent advances in immunotherapy, particularly
checkpoint inhibitors (CPIs), have transformed the treatment
paradigm for patients with metastatic non-small cell lung cancer
(mNSCLC) and metastatic renal cell carcinoma (mRCC). Nivolumab is a
human immunoglobulin G4 monoclonal antibody, which binds to the
programmed death-1 (PD-1) receptor and blocks its interaction with
its ligands, programmed death-ligand (PD-L)1 and PD-L2,
potentiating T-cell responses (1).
The United States Food and Drug Administration (FDA) initially
approved nivolumab monotherapy in 2015 for patients with mNSCLC who
develop progressive disease (PD) on or after platinum-based
chemotherapy. In 2020, nivolumab received FDA approval as
first-line treatment of patients with mNSCLC (without EGFR or
anaplastic lymphoma kinase genomic aberrations) in combination with
ipilimumab, an anti-cytotoxic T-lymphocyte antigen-4 antibody, for
patients expressing PD-L1 >1%; or for all comers, regardless of
PD-L1 expression, in combination with two cycles of platinum
doublet chemotherapy. For patients with mRCC, nivolumab has been
FDA-approved in the second-line setting after prior anti-angiogenic
therapy and in treatment-naive intermediate- to poor-risk patients
in combination with ipilimumab.
Nivolumab is effective and well-tolerated in
patients with mNSCLC and mRCC; however, the response rates with
monotherapy remain low (~20%) and most patients eventually develop
PD. Therefore, immunotherapy trials are currently focusing on
immunotherapy combinations to enhance therapeutic efficacy. Recent
clinical trials have shown improvement in clinical outcomes by
combining CPIs with other CPIs or chemotherapeutic agents (2,3).
However, the clinical application of these combinations may be
limited by prohibitive cost and overlapping additional
toxicities.
In the phase III CheckMate 067 trial for patients
with advanced melanoma, the combined administration of nivolumab
and ipilimumab demonstrated improvement in progression-free
survival (PFS) and overall survival (OS) when compared to
monotherapy with either drug alone; however, the combination was
associated with grade 3-4 toxicities in 59% of the patients
(3). CheckMate 214, a phase III
trial for patients with mRCC, demonstrated improved OS for patients
with intermediate- to poor-risk mRCC when compared to sunitinib.
However, the combination was associated with grade 3-4 toxicities
in 46% of the patients and toxicity-related treatment
discontinuation in 22% of the patients.
Radiotherapy (RT) is an important cancer treatment
modality, which is indicated in ~52% of all patients with cancer
(4). RT has limited side effects
and is highly cost-effective, accounting for only 5% of the total
cost of cancer care, making it an attractive modality for using in
combination with other systemic therapies (5). RT may augment the immune response
achieved with the use of CPIs, either through direct cytotoxic
antitumour effect or by modifying the tumour microenvironment to
promote an antitumour immune response (6). The antitumour effects of RT have also
been observed outside the radiation field, referred to as the
‘abscopal effect’ (7).
The aim of the present retrospective study was to
assess the safety and efficacy of nivolumab and concurrent RT for
patients with mNSCLC and mRCC with oligometastatic PD on nivolumab
monotherapy.
Patients and methods
Patients
A total of consecutive 96 patients who received
nivolumab at the Department of Oncology, Prince Sultan Military
Medical City (Riyadh, Saudi Arabia) between November 2016 and April
2018 were retrospectively identified. The inclusion criteria for
this study were patients who developed oligometastatic PD during
nivolumab treatment and were subsequently treated with RT, with
nivolumab continued beyond disease progression. A total of 22
patients met the criteria, of whom 12 had mNSCLC and 10 had mRCC.
For the study population, a comprehensive chart review was
completed. Clinical data and RT details were collected from our
electronic medical records, as well as the RT planning system (ARIA
OIS; Varian Medical Systems, Inc.). Patient demographic data and
radiological and pathological details were obtained, in addition to
RT indications, treatment sites, techniques and fractionations.
Treatment
Nivolumab treatment was administered intravenously
as per the standard protocol (https://www.accessdata.fda.gov/drugsatfda_docs/label/2021/125554s090lbl.pdf),
at a dose of either 240 mg every 2 weeks or 480 mg every 4 weeks.
All patients were assessed by the clinician prior to each nivolumab
administration.
RT was delivered as per standard departmental
protocols for stereotactic or conformal treatment. RT was
administered with a linear accelerator and delivered as conformal
RT, or intensity-modulated RT, as clinically appropriate.
Stereotactic body RT (SBRT) was delivered with linear accelerator
whereas Gamma Knife (Elekta Instrument AB) was used for
stereotactic radiosurgery (SRS). Fractionated RT was generally
administered as a palliative hypofractionation schedule determined
according to the discretion of treating physician. The
fractionation schedules for conformal external beam RT included 20
Gy/5 fractions (fr), 30 Gy/10 fr, 25 Gy/5 fr, 20 Gy/4 fr, 16 Gy/4
fr and 8 Gy/1 fr. The fractionation schedules for stereotactic RT
included 48 Gy/4 fr, 40 Gy/10 fr, 40 Gy/4 fr, 34 Gy/4 fr and 22
Gy/1 fr.
Response assessment
Objective clinical response [complete response (CR),
partial response (PR), stable disease (SD) or PD] was evaluated
using CT, MRI or 18F-fluorodeoxyglucose positron
emission tomography-CT scans.
The modified Response Evaluation Criteria In Solid
Tumours criteria, version 1.1, were adopted for the assessment of
response at target lesions receiving RT (8). Response was defined as follows: CR,
complete disappearance of the target lesion; PR, at least 30%
decrease in the longest diameter of the target lesion; PD, >20%
increase in the longest diameter of the target lesion; and SD,
neither sufficient shrinkage to qualify for PR, nor sufficient
increase to qualify for PD. Patients considered as CR, PR or SD
should not have unequivocal PD in the non-target lesions. Adverse
events and laboratory abnormalities were evaluated using the
National Cancer Institute Common Toxicity Criteria (version 3.0) of
the National Cancer Institute (9).
Results
Patient characteristics and adverse
events
A total of 22 patients received multiple courses of
RT. The median age of the patients was 59 years (range, 39-72
years) and the male:female ratio was 2.7:1. Stereotactic and
conformal RT was delivered to 5 and 34 sites, respectively. The
treatment sites included the following: Bone (n=15), lung (n=9),
brain (n=8), adrenal gland (n=2), renal bed (n=2), skin (n=1),
ethmoid sinus (n=1) and scalp (n=1). The gap between RT and
nivolumab did not exceed 2 weeks for all patients. No patients
developed any grade 3-4 toxicities. Grade 2 pneumonitis was noted
in 3 patients receiving lung RT. Other RT-related side effects
included grade 2 lethargy (n=2), grade 1 diarrhoea (n=3) and grade
1 skin erythema (n=3). The most common fractionation, used in 54%
of the courses, was 20 Gy/5 fr.
Response
Of the 39 treatment sites, PR and CR were noted at
25 (64%) and 3 (8%) sites, respectively. SD was noted at 6 sites
(15%) and PD was noted at 5 sites (13%). Overall, 72% of the sites
irradiated exhibited an excellent response to the concurrent
treatment. The efficacy of RT to the bone sites was assessed by
comparing the pre-treatment pain intensity scores using an
11-categorical point scale (0=lack of pain and 10=worst pain
imaginable) to the 6-8-week follow-up pain score assessment. Pain
response was defined as a reduction in pain scores by ≥2 points
compared to baseline. The median baseline pain score was 7 (range,
5-10) and pain response was observed in 9 of the 15 treatment sites
(60%). The median time spent on nivolumab from the date of the
first fraction of RT was 4.5 months (range, 1.5-29 months) for
patients with mNSCLC and 5 months (range, 1-38.5 months) for
patients with mRCC.
Discussion
The rationale for combining RT and CPIs is that RT
may lead to polyclonal T-cell infiltration and expansion at the
treatment site and, in addition, CPIs block the PD-1/PD-L1 axis
generating a systemic T-cell response (10). Dovedi et al (11) demonstrated the synergistic
potential of RT and anti-PD-1 treatment in their study using dual
tumour-bearing mouse models. In that study, it was observed that RT
alone was highly effective for disease control at the irradiated
site, but there were no abscopal responses. However, when RT was
delivered concurrently with an anti-PD-1 monoclonal antibody, there
was regression of both the irradiated and out-of-field tumours,
with >70% of mice undergoing complete responses suggesting a
synergistic potential for this combination. Deng et al
(12) demonstrated that the
combination of RT and PD-L1 inhibitors exerted a notably more
significant effect on tumour growth in mouse models compared with
either treatment alone. Twyman-Saint Victor et al (13) observed marked tumour regression in
a subset of patients with metastatic melanoma treated with an
anti-CTLA4 antibody; they also noted that resistance was common,
but could be overcome with the addition of RT and anti-PD-L1
treatment. The authors subsequently demonstrated that RT along with
dual checkpoint blockade may activate non-redundant immune
mechanisms and reverse T-cell exhaustion, leading to improved
efficacy. Shaverdian et al (14) assessed the disease control and
pulmonary toxicity in the phase I Keynote-001 trial for patients
with NSLC who previously received RT prior to pembrolizumab. Of the
98 patients who received pembrolizumab on that trial, 43% had
received prior RT. There was a significant improvement in PFS
[HR=0.56 (95% CI: 0.34-0.91), P=0.019] and OS [HR=0.58 (95% CI:
0.36-0.94); P=0.026] for patients treated with pembrolizumab and RT
compared with pembrolizumab alone, with an acceptable toxicity
profile. In their retrospective study, Samstein et al
(15) demonstrated an improvement
in OS with concurrent CPIs and RT, particularly when CPI therapy
was initiated at least 1 month prior to RT.
NIVES, a phase II multicentre trial, is the first
prospective trial of nivolumab plus SBRT in mRCC. At a median
follow-up of 15 months, the study showed acceptable safety, but the
primary endpoint of improving response rate to 40% was not met. The
objective response rate (ORR) in an intent-to-treat analysis was
17.4% (12 of 68 patients) and included one case of CR. An
additional 28 patients (40.6%) had SD. The estimated median OS was
22 months, and the 12-month survival rate was 73.4% (16).
However, a higher ORR was observed with the use of
dual checkpoint inhibition in the RADVAX trial. RADVAX, a small,
multi-institution, single-arm phase II study assessed the safety
and efficacy of SBRT in combination with nivolumab + ipilimumab in
patients with mRCC (15). Hammers
et al (17) reported that
the ORR was 56% during a median follow-up of 24 months for the 25
patients. SBRT was administered to 1-2 metastatic sites at a dose
of 50 Gy/5 fr between the first and second cycles of
nivolumab-ipilimumab. The median PFS was 8.21 months and the 1-year
PFS rate was 36%. Grade 3-4 treatment-related adverse events were
noted in 36% of the patients, with radiation pneumonitis reported
in 2 patients. The authors concluded that the combination was
feasible and associated with an acceptable safety profile.
Despite the significant clinical benefits of
combining RT with chemotherapy, however, chemoradiotherapy
protocols are often associated with increased incidence and
severity of side effects (18).
The unique toxicity profile of immunotherapy, which is distinct
from that of conventional chemotherapeutic agents, together with
the technological advances in modern RT, including highly
conformal, intensity-modulated and stereotactic techniques combined
with high-precision image guidance, are expected to improve the
tolerability of the combination and the therapeutic ratio (19). Von Reibnitz et al (20) retrospectively evaluated the
toxicity profile of patients receiving immune CPIs and thoracic RT
and concluded that concurrent immunotherapy and thoracic RT may be
safe. The ETOP NICOLAS, is an ongoing phase II trial evaluating the
safety and efficacy of nivolumab combined with radical concurrent
chemoradiotherapy (66 Gy/33 fr) in stage III NSCLC (21). Interim analysis after the initial
21 patients completed at the 3-month post-RT follow-up has
demonstrated no grade ≥3 pneumonitis. The results of efficacy
analysis from that study are still awaited. Tables I and II outline selected ongoing clinical
trials investigating the combination of nivolumab and RT in
patients with mRCC or mNSCLC.
 | Table IOngoing phase I-II clinical trials
investigating the combination of nivolumab and radiotherapy in
patients with mRCC or mNSCLC. |
Table I
Ongoing phase I-II clinical trials
investigating the combination of nivolumab and radiotherapy in
patients with mRCC or mNSCLC.
| Clinical trials | Phase | Indication | Treatment arms | Number of
patients | Primary outcome | Expected trial
completion date |
|---|
| NCT03044626 | II | Metastatic
non-squamous NSCLC | Arm 1: Nivolumab 240
mg q2w + 20 Gy/5 fractions to one metastatic site Arm 2: Nivolumab
240 mg q2w | 130 | Objective response
rate | November 2021 |
| NCT02696993 | I/II | NSCLC with brain
metastases | Nivolumab +
ipilimumab + whole brain radiotherapy or stereotactic
radiosurgery | 80 | Maximum tolerated
dose nivolumab | December 2020 |
| NCT02781506 | II | Metastatic clear cell
RCC | Nivolumab alone: IV,
3 mg/kg q2w + stereotactic ablative radiotherapy (1-3 fractions for
1-3 lesions) | 35 | Response rate | December 2022 |
| NCT03149159 | II | mRCC | Nivolumab 3 mg/kg +
ipilimumab 1 mg/kg + 30 Gy/5 fractions | | Withdrawn | |
| NCT03223155 | I | Stage 4 small cell
lung cancer | Nivolumab +
ipilumumab + sterotactic body radiotherapy concurrent vs.
sequential | 80 | Serious adverse
events | December 2020 |
 | Table IIOngoing phase III clinical trials
investigating the combination of nivolumab and radiotherapy. |
Table II
Ongoing phase III clinical trials
investigating the combination of nivolumab and radiotherapy.
| Clinical trials | Indication | Treatment arms | Number of
patients | Endpoints | Estimated completion
date |
|---|
| NCT03349710 | Squamous cell
carcinoma of the head and neck | Cohort 1: nivolumab +
RT vs. cetuximab + RT Cohort 2: Nivolumab + cisplatin + cisplatin +
RT vs. cisplatin + RT | 1,046 | Event-free
survival | November 2022 |
| NCT02768558 (RTOG
3505) | Stage III
unresectable non-small cell lung cancer | Cisplatin etoposide +
60 Gy concurrent RT followed by ± nivolumab 240 mg q2w x 1
year | 660 | Overall and
progression-free survival | October 2024 |
In the present retrospective study, all patients
received hypofractionated RT concurrently with nivolumab. Different
hypofractionation protocols were used according to the tumour site,
clinical indications and technique of irradiation. The use of
hypofractionated RT in this context is based on the findings of
several preclinical studies, which suggested a higher immunogenic
effect when higher dose per fraction is delivered (22,23).
For patients with mRCC or mNSCLC with PD on nivolumab therapy there
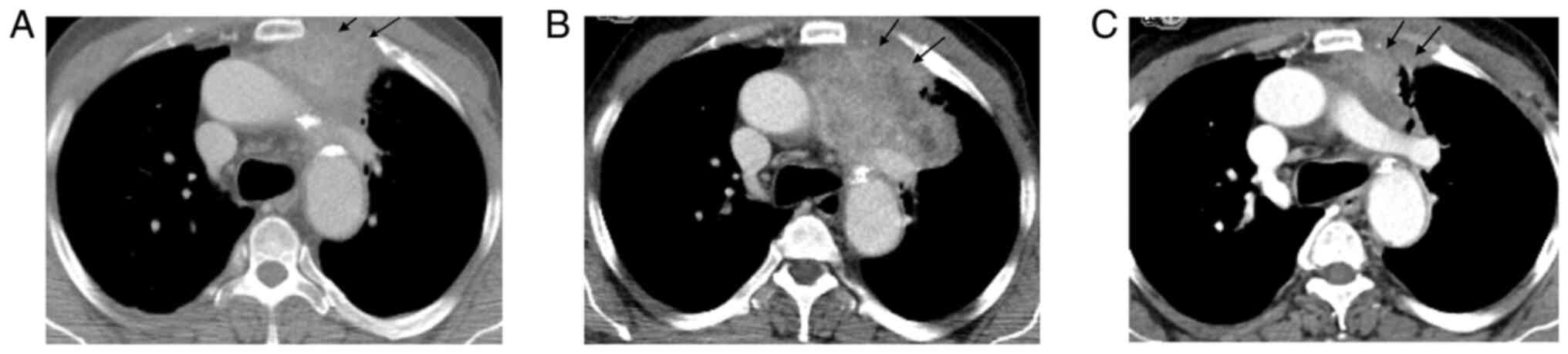
are limited treatment options and no standard guidelines. Figs. 1 and 2 illustrated the case of a 62-year-old
male patient with mNSCLC who was treated with nivolumab following
disease progression on platinum-based chemotherapy. As the patient
developed oligometastatic PD in the lung primary (Fig. 1A and B) following 8 weeks of nivolumab therapy,
he was treated with palliative RT, which resulted in PR (Fig. 1C). The patient subsequently
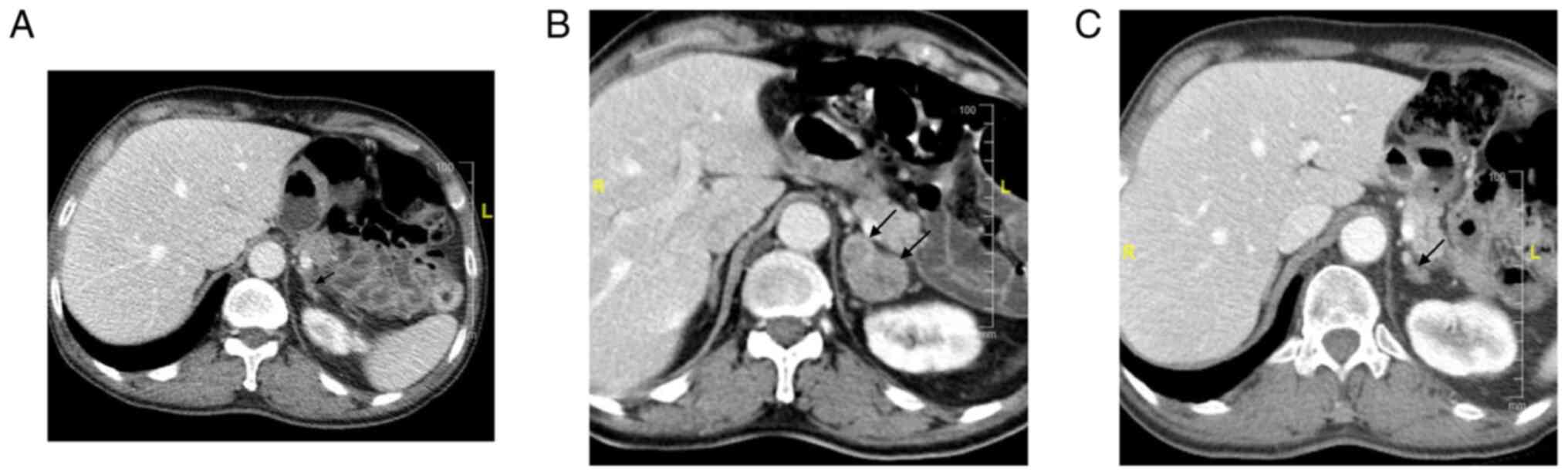
developed oligometastatic PD in the adrenal metastasis (Fig. 2A and B) and was treated with SBRT with further
PR (Fig. 2C).
In the present study, the role of adding concurrent
RT for patients developing oligometastatic PD on nivolumab therapy
was explored, and excellent responses were demonstrated. In
accordance with other retrospective studies, the results of the
present study demonstrated good tolerance for the combination of
nivolumab and RT, without grades 3-4 toxicities (24,25).
Treatment beyond progression is currently not the standard of care
for patients receiving nivolumab. Radiological assessment of
response and treatment beyond progression on immunotherapy may
represent the most robust tools for assessment of response in this
indication of oligometastatic PD on nivolumab (26,27).
In the present study, the addition of RT and continuation of
nivolumab beyond progression appear to be feasible for patients
with oligometastatic PD.
One of the limitations of the present study is its
retrospective nature, in addition to the limited number of cases
and short follow-up interval time. The heterogeneity of the
included cases in terms of primary diagnosis, RT technique and
fractionation also added to these limitations. A prospective study
with a larger patient population and adequate follow-up is required
to further elucidate these issues. However, clinical discretion
should be used in the meantime while recommending this combination
of nivolumab and RT, keeping in view the emerging data and the
potential benefits.
In conclusion, the results achieved with the
combination of nivolumab and RT appear promising in several
retrospective studies. In the present study for patients with mRCC
or mNSCLC with oligometastatic PD during nivolumab therapy, the
addition of RT appears to initiate a response and prolong the time
on nivolumab treatment. The combination of nivolumab and RT appears
to be well-tolerated, with response rates exceeding published
studies of nivolumab monotherapy.
The optimal strategy for integrate RT into the
immune treatment pathway still remains to be determined. Several
questions regarding optimal RT dose and fractionation, treatment
technique, timing and safety still remain unanswered. A plethora of
clinical trials are currently investigating these RT-immune
interactions (Tables I and
II), however, several challenges
remain to be addressed to maximize the efficacy of this promising
combination.
Acknowledgements
Not applicable.
Funding
No funding was received.
Availability of data and materials
All the data relevant to the present study are
available from the corresponding author on reasonable request.
Authors' contributions
JA: Primary author, conception and design,
preparation and review of the manuscript; AF: Literature review,
patient follow-up, preparation and review of the manuscript; AMA
and MAl: Literature review and data collection; EM, AA and MAb:
Case analysis, preparation and critical review of the manuscript;
HA: Literature review, data interpretation, review of imaging and
response assessment; SH and JG: Expert opinion for management of
toxicities, case review, radiotherapy guidance, literature review,
conception, supervision and review of the manuscript. JA and AF
have seen the raw data and can confirm their authenticity. All the
authors have read and approved the final manuscript.
Ethics approval and consent to
participate
Ethics approval has been obtained from the
Institutional Review Board of the Prince Sultan Military Medical
City HP-01-R079, approval no. 1490 on 18/03/2021.
Patient consent for publication
Not applicable.
Competing interests
Dr Jawaher Ansari, Dr Abdulaziz Alhamad and
Professor Syed Hussain have received lecture fees from Bristol
Myers Squibb. All the other authors declare that they have no
competing interests.
References
|
1
|
Brahmer JR, Drake CG, Wollner I, Powderly
JD, Picus J, Sharfman WH, Stankevich E, Pons A, Salay TM, McMiller
TL, et al: Phase I study of single-agent anti-programmed death-1
(MDX-1106) in refractory solid tumors: Safety, clinical activity,
pharmacodynamics, and immunologic correlates. J Clin Oncol.
28:3167–3175. 2010.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Gandhi L, Rodríguez-Abreu D, Gadgeel S,
Esteban E, Felip E, De Angelis F, Domine M, Clingan P, Hochmair MJ,
Powell SF, et al: KEYNOTE-189 investigators: Pembrolizumab plus
chemotherapy in metastatic non-small-cell lung cancer. N Engl J
Med. 378:2078–2092. 2018.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Larkin J, Chiarion-Sileni V, Gonzalez R,
Grob JJ, Cowey CL, Lao CD, Schadendorf D, Dummer R, Smylie M,
Rutkowski P, et al: Combined nivolumab and ipilimumab or
monotherapy in untreated melanoma. N Engl J Med. 373:23–34.
2015.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Delaney G, Jacob S, Featherstone C and
Barton M: The role of radiotherapy in cancer treatment: Estimating
optimal utilization from a review of evidence-based clinical
guidelines. Cancer. 104:1129–1137. 2005.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Ringborg U, Bergqvist D, Brorsson B,
Cavallin-Ståhl E, Ceberg J, Einhorn N, Frödin JE, Järhult J,
Lamnevik G, Lindholm C, et al: The swedish council on technology
assessment in health care (SBU) systematic overview of radiotherapy
for cancer including a prospective survey of radiotherapy practice
in Sweden 2001-summary and conclusions. Acta Oncol. 42:357–365.
2003.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Walle T, Martinez Monge R, Cerwenka A,
Ajona D, Melero I and Lecanda F: Radiation effects on antitumor
immune responses: Current perspectives and challenges. Ther Adv Med
Oncol. 10(1758834017742575)2018.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Demaria S, Ng B, Devitt ML, Babb JS,
Kawashima N, Liebes L and Formenti SC: Ionizing radiation
inhibition of distant untreated tumors (abscopal effect) is immune
mediated. Int J Radiat Oncol Biol Phys. 58:862–870. 2004.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Eisenhauer EA, Therasse P, Bogaerts J,
Schwartz LH, Sargent D, Ford R, Dancey J, Arbuck S, Gwyther S,
Mooney M, et al: New response evaluation criteria in solid tumours:
Revised RECIST guideline (version 1.1). Eur J Cancer. 45:228–247.
2009.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Trotti A, Colevas AD, Setser A and Basch
E: Patient-reported outcomes and the evolution of adverse event
reporting in oncology. J Clin Oncol. 25:5121–5127. 2007.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Colton M, Cheadle EJ, Honeychurch J and
Illidge TM: Reprogramming the tumour microenvironment by
radiotherapy: Implications for radiotherapy and immunotherapy
combinations. Radiat Oncol. 15(254)2020.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Dovedi SJ, Cheadle EJ, Popple AL, Poon E,
Morrow M, Stewart R, Yusko EC, Sanders CM, Vignali M, Emerson RO,
et al: Fractionated radiation therapy stimulates antitumor immunity
mediated by both resident and infiltrating polyclonal T-cell
populations when combined with PD-1 blockade. Clin Cancer Res.
23:5514–5526. 2017.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Deng L, Liang H, Burnette B, Beckett M,
Darga T, Weichselbaum RR and Fu YX: Irradiation and anti-PD-L1
treatment synergistically promote antitumor immunity in mice. J
Clin Invest. 124:687–695. 2014.PubMed/NCBI View
Article : Google Scholar
|
|
13
|
Twyman-Saint Victor C, Rech AJ, Maity A,
Rengan R, Pauken KE, Stelekati E, Benci JL, Xu B, Dada H, Odorizzi
PM, et al: Radiation and dual checkpoint blockade activate
non-redundant immune mechanisms in cancer. Nature. 520:373–377.
2015.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Shaverdian N, Lisberg AE, Bornazyan K,
Veruttipong D, Goldman JW, Formenti SC, Garon EB and Lee P:
Previous radiotherapy and the clinical activity and toxicity of
pembrolizumab in the treatment of non-small-cell lung cancer: A
secondary analysis of the KEYNOTE-001 phase 1 trial. Lancet Oncol.
18:895–903. 2017.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Samstein R, Rimner A, Barker CA and Yamada
Y: Combined immune checkpoint blockade and radiation therapy:
timing and dose fractionation associated with greatest survival
duration among over 750 treated patients. Int J Radiat Oncol Biol
Phys. 99 (Suppl 2):S129–S130. 2017.
|
|
16
|
Masini C, Iotti C, De Giorgi U, Bellia RS,
Buti S, Salaroli F, Zampiva I, Mazzarotto R, Mucciarini C,
Baldessari C, et al: Nivolumab (NIVO) in combination with
stereotactic body radiotherapy (SBRT) in pretreated patients (pts)
with metastatic renal cell carcinoma (mRCC): First results of phase
II NIVES study. J Clin Oncol. 38 (6 Suppl)(S613)2020.
|
|
17
|
Hammers HJ, Vonmerveldt D, Ahn C, Nadal
RM, Drake CG, Folkert MR, Lain AM, Courtney KD, Brugarolas J, Song
DY, et al: Combination of dual immune checkpoint inhibition (ICI)
with stereotactic radiation (SBRT) in metastatic renal cell
carcinoma (mRCC) (RADVAX RCC). J Clin Oncol. 38 (Suppl 6):614.
2020.
|
|
18
|
Deutsch E, Chargari C, Galluzzi L and
Kroemer G: Optimising efficacy and reducing toxicity of anticancer
radioimmunotherapy. Lancet Oncol. 20:e452–e463. 2019.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Jagodinsky JC, Harari PM and Morris ZS:
The promise of combining radiation therapy with immunotherapy. Int
J Radiat Oncol Biol Phys. 108:6–16. 2020.PubMed/NCBI View Article : Google Scholar
|
|
20
|
Von Reibnitz D, Chaft JE, Wu AJ, Samstein
R, Hellmann MD, Plodkowski AJ, Zhang Z, Shi W, Dick-Godfrey R,
Panchoo KH, et al: Safety of combining thoracic radiation therapy
with concurrent versus sequential immune checkpoint inhibition. Adv
Radiat Oncol. 3:391–398. 2018.PubMed/NCBI View Article : Google Scholar
|
|
21
|
Peters S, Felip E, Dafni U, Belka C,
Guckenberger M, Irigoyen A, Nadal E, Becker A, Vees H, Pless M, et
al: Safety evaluation of nivolumab added concurrently to
radiotherapy in a standard first line chemo-radiotherapy regimen in
stage III non-small cell lung cancer-The ETOP NICOLAS trial. Lung
Cancer. 133:83–87. 2019.PubMed/NCBI View Article : Google Scholar
|
|
22
|
Chajon E, Castelli J, Marsiglia H and De
Crevoisier R: The synergistic effect of radiotherapy and
immunotherapy: A promising but not simple partnership. Crit Rev
Oncol Hematol. 111:124–132. 2017.PubMed/NCBI View Article : Google Scholar
|
|
23
|
Camphausen K, Moses MA, Ménard C, Sproull
M, Beecken WD, Folkman J and O'Reilly MS: Radiation abscopal
antitumor effect is mediated through p53. Cancer Res. 63:1990–1993.
2003.PubMed/NCBI
|
|
24
|
Amin NP, Zainib M, Parker SM, Agarwal M
and Mattes MD: Multicenter report on toxicities of concurrent
nivolumab and radiation therapy. Adv Radiat Oncol. 3:399–404.
2018.PubMed/NCBI View Article : Google Scholar
|
|
25
|
Bang A, Wilhite TJ, Pike LRG, Cagney DN,
Aizer AA, Taylor A, Spektor A, Krishnan M, Ott PA, Balboni TA, et
al: Multicenter evaluation of the tolerability of combined
treatment with PD-1 and CTLA-4 immune checkpoint inhibitors and
palliative radiation therapy. Int J Radiat Oncol Biol Phys.
98:344–351. 2017.PubMed/NCBI View Article : Google Scholar
|
|
26
|
Dromain C, Beigelman C, Pozzessere C,
Duran R and Digklia A: Imaging of tumour response to immunotherapy.
Eur Radiol Exp. 4(2)2020.PubMed/NCBI View Article : Google Scholar
|
|
27
|
Ge X, Zhang Z, Zhang S, Yuan F, Zhang F,
Yan X, Han X, Ma J, Wang L, Tao H, et al: Immunotherapy beyond
progression in patients with advanced non-small cell lung cancer.
Transl Lung Cancer Res. 9:2391–2400. 2020.PubMed/NCBI View Article : Google Scholar
|