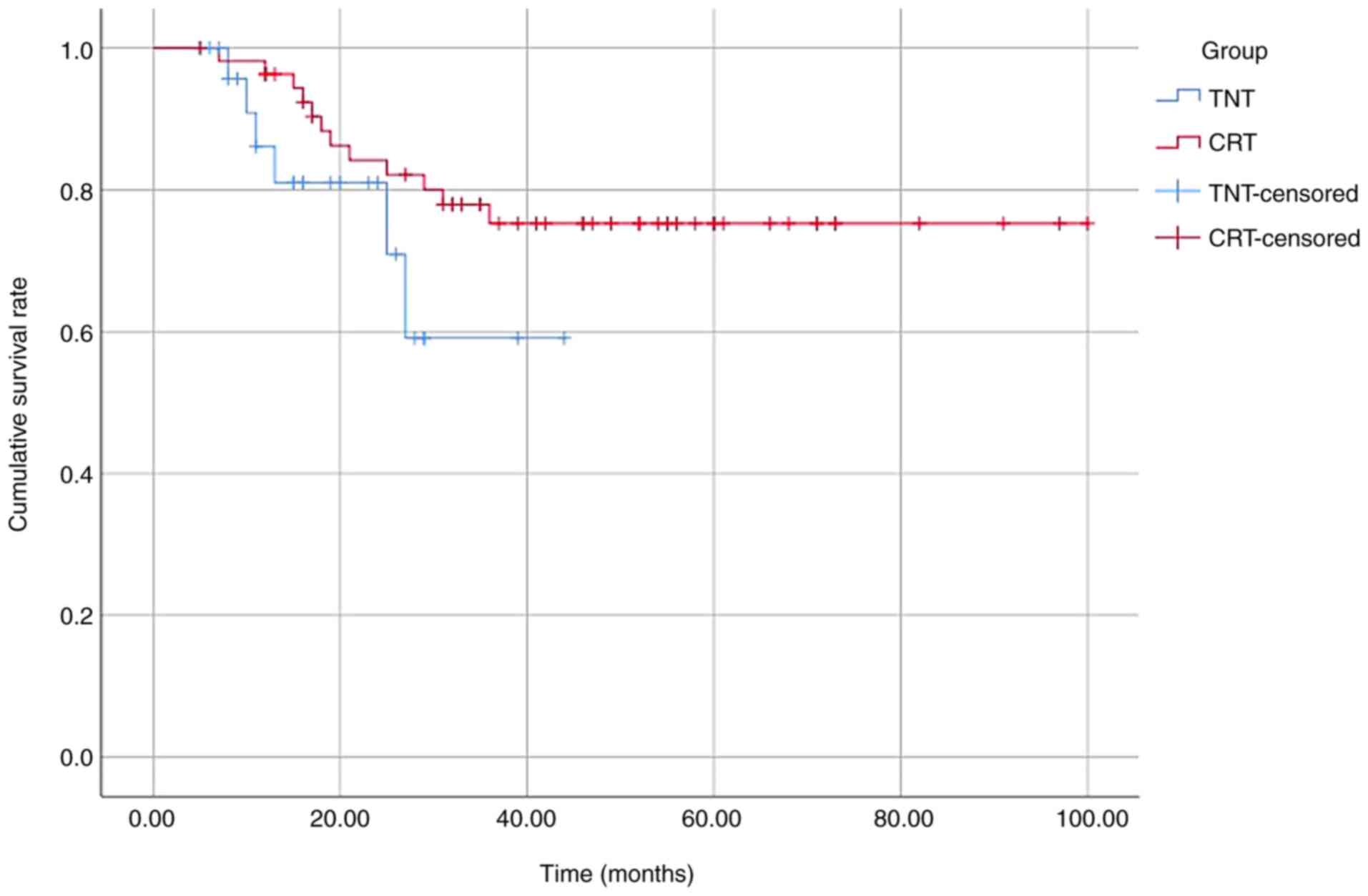
|
1
|
Sauer R, Becker H, Hohenberger W, Rödel C,
Wittekind C, Fietkau R, Martus P, Tschmelitsch J, Hager E, Hess CF,
et al: Preoperative vs. postoperative chemoradiotherapy for rectal
cancer. N Engl J Med. 351:1731–1740. 2004.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Ruo L and Guillem JG: Major 20th-century
advancements in the management of rectal cancer. Dis Colon Rectum.
42:563–578. 1999.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Maas M, Nelemans PJ, Valentini V, Crane
CH, Capirci C, Rödel C, Nash GM, Kuo LJ, Glynne-Jones R,
García-Aguilar J, et al: Adjuvant chemotherapy in rectal cancer:
Defining subgroups who may benefit after neoadjuvant chemoradiation
and resection: A pooled analysis of 3,313 patients. Int J Cancer.
137:212–220. 2015.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Biagi JJ, Raphael MJ, Mackillop WJ, Kong
W, King WD and Booth CM: Association between time to initiation of
adjuvant chemotherapy and survival in colorectal cancer: A
systematic review and meta-analysis. JAMA. 305:2335–2342.
2011.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Breugom AJ, Swets M, Bosset JF, Collette
L, Sainato A, Cionini L, Glynne-Jones R, Counsell N, Bastiaannet E,
van den Broek CB, et al: Adjuvant chemotherapy after preoperative
(chemo)radiotherapy and surgery for patients with rectal cancer: A
systematic review and meta-analysis of individual patient data.
Lancet Oncol. 16:200–207. 2015.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Bujko K, Glimelius B, Valentini V,
Michalski W and Spalek M: Postoperative chemotherapy in patients
with rectal cancer receiving preoperative radio(chemo)therapy: A
meta-analysis of randomized trials comparing surgery ± a
fluoropyrimidine and surgery + a fluoropyrimidine ± oxaliplatin.
Eur J Surg Oncol. 41:713–723. 2015.PubMed/NCBI View Article : Google Scholar
|
|
7
|
van Gijn W, Marijnen CA, Nagtegaal ID,
Kranenbarg EM, Putter H, Wiggers T, Rutten HJ, Påhlman L, Glimelius
B and van de Velde CJ: Dutch Colorectal Cancer Group. Preoperative
radiotherapy combined with total mesorectal excision for resectable
rectal cancer: 12-year follow-up of the multicentre, randomised
controlled TME trial. Lancet Oncol. 12:575–582. 2011.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Kapiteijn E, Marijnen CA, Nagtegaal ID,
Putter H, Steup WH, Wiggers T, Rutten HJ, Pahlman L, Glimelius B,
van Krieken JH, et al: Preoperative radiotherapy combined with
total mesorectal excision for resectable rectal cancer. N Engl J
Med. 345:638–646. 2001.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Hartley A, Ho KF, McConkey C and Geh JI:
Pathological complete response following pre-operative
chemoradiotherapy in rectal cancer: Analysis of phase II/III
trials. Br J Radiol. 78:934–938. 2005.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Cotte E, Passot G, Decullier E, Maurice C,
Glehen O, François Y, Lorchel F, Chapet O and Gerard JP: Pathologic
response, when increased by longer interval, is a marker but not
the cause of good prognosis in rectal cancer: 17-year follow-up of
the Lyon R90-01 randomized trial. Int J Radiat Oncol Biol Phys.
94:544–553. 2016.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Park IJ, You YN, Agarwal A, Skibber JM,
Rodriguez-Bigas MA, Eng C, Feig BW, Das P, Krishnan S, Crane CH, et
al: Neoadjuvant treatment response as an early response indicator
for patients with rectal cancer. J Clin Oncol. 30:1770–1776.
2012.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Pretzsch E, Bösch F, Neumann J, Ganschow
P, Bazhin A, Guba M, Werner J and Angele M: Mechanisms of
metastasis in colorectal cancer and metastatic organotropism:
Hematogenous vs. peritoneal spread. J Oncol.
2019(7407190)2019.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Sauer R, Liersch T, Merkel S, Fietkau R,
Hohenberger W, Hess C, Becker H, Raab HR, Villanueva MT, Witzigmann
H, et al: Preoperative vs. postoperative chemoradiotherapy for
locally advanced rectal cancer: Results of the German
CAO/ARO/AIO-94 randomized phase III trial after a median follow-up
of 11 years. J Clin Oncol. 30:1926–1933. 2012.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Hospers G, Bahadoer RR, Dijkstra EA, Etten
BV, Marijnen C, Putter H, Meershoek-Klein Kranenbarg E, Roodvoets
AG, Nagtegaal ID, Regina GH Beets-Tan, et al: Short-course
radiotherapy followed by chemotherapy before TME in locally
advanced rectal cancer: The randomized RAPIDO trial. J Clin Oncol.
38 (Suppl 15)(S4006)2020.
|
|
15
|
Bahadoer RR, Dijkstra EA, van Etten B,
Marijnen CAM, Putter H, Kranenbarg EM, Roodvoets AGH, Nagtegaal ID,
Beets-Tan RGH, Blomqvist LK, et al: Short-course radiotherapy
followed by chemotherapy before total mesorectal excision (TME) vs.
preoperative chemoradiotherapy, TME, and optional adjuvant
chemotherapy in locally advanced rectal cancer (RAPIDO): A
randomised, open-label, phase 3 trial. Lancet Oncol. 22:29–42.
2021.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Becker K, Mueller JD, Schulmacher C, Ott
K, Fink U, Busch R, Böttcher K, Siewert JR and Höfler H:
Histomorphology and grading of regression in gastric carcinoma
treated with neoadjuvant chemotherapy. Cancer. 98:1521–1530.
2003.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Delibegovic S: Introduction to total
mesorectal excision. Med Arch. 71:434–438. 2017.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Bosset JF, Calais G, Mineur L, Maingon P,
Stojanovic-Rundic S, Bensadoun RJ, Bardet E, Beny A, Ollier JC,
Bolla M, et al: Fluorouracil-based adjuvant chemotherapy after
preoperative chemoradiotherapy in rectal cancer: Long-term results
of the EORTC 22921 randomised study. Lancet Oncol. 15:184–190.
2014.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Sainato A, Cernusco Luna Nunzia V,
Valentini V, De Paoli A, Maurizi ER, Lupattelli M, Aristei C,
Vidali C, Conti M, Galardi A, et al: No benefit of adjuvant
Fluorouracil Leucovorin chemotherapy after neoadjuvant
chemoradiotherapy in locally advanced cancer of the rectum (LARC):
Long term results of a randomized trial (I-CNR-RT). Radiother
Oncol. 113:223–229. 2014.PubMed/NCBI View Article : Google Scholar
|
|
20
|
Schmoll HJ, Haustermans K, Price TJ,
Nordlinger B, Hofheinz R, Daisne JF, Janssens J, Brenner B, Schmidt
P, Reinel H, et al: Preoperative chemoradiotherapy and
postoperative chemotherapy with capecitabine +/-oxaliplatin in
locally advanced rectal cancer: Final results of PETACC-6. J Clin
Oncol. 36 (Suppl 15)(S3500)2018.
|
|
21
|
Rödel C, Graeven U, Fietkau R, Hohenberger
W, Hothorn T, Arnold D, Hofheinz RD, Ghadimi M, Wolff HA,
Lang-Welzenbach M, et al: Oxaliplatin added to fluorouracil-based
preoperative chemoradiotherapy and postoperative chemotherapy of
locally advanced rectal cancer (the German CAO/ARO/AIO-04 study):
Final results of the multicentre, open-label, randomised, phase 3
trial. Lancet Oncol. 16:979–989. 2015.PubMed/NCBI View Article : Google Scholar
|
|
22
|
Mari GM, Maggioni D, Crippa J, Costanzi
ATM, Scotti MA, Giardini V, Garancini M, Cocozza E, Borroni G,
Benzoni I, et al: Compliance to adjuvant chemotherapy of patients
who underwent surgery for rectal cancer: Report from a
Multi-institutional Research Network. World J Surg. 43:2544–2551.
2019.PubMed/NCBI View Article : Google Scholar
|
|
23
|
Marco MR, Zhou L, Patil S, Marcet JE,
Varma MG, Oommen S, Cataldo PA, Hunt SR, Kumar A, Herzig DO, et al:
Consolidation mFOLFOX6 chemotherapy after chemoradiotherapy
improves survival in patients with locally advanced rectal cancer:
Final results of a multicenter phase II trial. Dis Colon Rectum.
61:1146–1155. 2018.PubMed/NCBI View Article : Google Scholar
|
|
24
|
Garcia-Aguilar J, Chow OS, Smith DD,
Marcet JE, Cataldo PA, Varma MG, Kumar AS, Oommen S, Coutsoftides
T, Hunt SR, et al: Effect of adding mFOLFOX6 after neoadjuvant
chemoradiation in locally advanced rectal cancer: A multicentre,
phase 2 trial. Lancet Oncol. 16:957–966. 2015.PubMed/NCBI View Article : Google Scholar
|
|
25
|
Ngan SY, Burmeister B, Fisher RJ, Solomon
M, Goldstein D, Joseph D, Ackland SP, Schache D, McClure B,
McLachlan SA, et al: Randomized trial of short-course radiotherapy
vs. long-course chemoradiation comparing rates of local recurrence
in patients with T3 rectal cancer: Trans-Tasman Radiation Oncology
Group trial 01.04. J Clin Oncol. 30:3827–3833. 2012.PubMed/NCBI View Article : Google Scholar
|
|
26
|
Erlandsson J, Holm T, Pettersson D,
Berglund Å, Cedermark B, Radu C, Johansson H, Machado M, Hjern F,
Hallböök O, et al: Optimal fractionation of preoperative
radiotherapy and timing to surgery for rectal cancer (Stockholm
III): A multicentre, randomised, non-blinded, phase 3,
non-inferiority trial. Lancet Oncol. 18:336–346. 2017.PubMed/NCBI View Article : Google Scholar
|
|
27
|
Ciseł B, Pietrzak L, Michalski W, Wyrwicz
L, Rutkowski A, Kosakowska E, Cencelewicz A, Spałek M, Polkowski W,
Jankiewicz M, et al: Long-course preoperative chemoradiation vs.
5x5 Gy and consolidation chemotherapy for clinical T4 and fixed
clinical T3 rectal cancer: Long-term results of the randomized
Polish II study. Ann Oncol. 30:1298–1303. 2019.PubMed/NCBI View Article : Google Scholar
|
|
28
|
Fokas E, Allgäuer M, Polat B, Klautke G,
Grabenbauer GG, Fietkau R, Kuhnt T, Staib L, Brunner T, Grosu AL,
et al: Randomized phase II trial of chemoradiotherapy plus
induction or consolidation chemotherapy as total neoadjuvant
therapy for locally advanced rectal cancer: CAO/ARO/AIO-12. J Clin
Oncol. 37:3212–3222. 2019.PubMed/NCBI View Article : Google Scholar
|
|
29
|
Manthravadi S, Sun W, Saeed A, Baranda JC
and Kasi A: Total neoadjuvant therapy compared with standard
therapy in locally advanced rectal cancer: A systematic review and
meta-analysis. J Clin Oncol. 37 (Suppl 4)(S709)2019.
|
|
30
|
Chapman W Jr, Roxburgh C, Makhdoom B, Roy
A, Youssef FF, Brady P, Olsen J, Kim H, Pedersen K, Mutch H, et al:
Rectal cancer downstaging is significantly improved with different
regimens of total neoadjuvant therapy. Int J Radiation Oncol Biol
Phys. 102 (Suppl):S65–S66. 2018.
|
|
31
|
Garcia-Aguilar J, Patil S, Kim JK, Yuval
JB, Thompson H, Verheij F, Lee M and Saltz LB: on behalf of the
OPRA Consortium. Preliminary results of the organ preservation of
rectal adenocarcinoma (OPRA) trial. J Clin Oncol. 38 (Suppl
15)(S4008)2020.
|
|
32
|
Shui L, Yang X, Li J, Yi C, Sun Q and Zhu
H: Gut microbiome as a potential factor for modulating resistance
to cancer immunotherapy. Front Immunol. 10(2989)2020.PubMed/NCBI View Article : Google Scholar
|
|
33
|
Zhang MX, Li XB, Guan BJ, Guan GX, Lin XY,
Wu XD, Chi P and Xu BH: Dose escalation of preoperative
short-course radiotherapy followed by neoadjuvant chemotherapy in
locally advanced rectal cancer: Protocol for an open-label,
single-centre, phase I clinical trial. BMJ Open.
9(e025944)2019.PubMed/NCBI View Article : Google Scholar
|
|
34
|
Yuki S, Bando H, Tsukada Y, Inamori K,
Komatsu Y, Homma S, Uemura M, Kato T, Kotani D, Fukuoka S, et al:
Short-term results of VOLTAGE-A: Nivolumab monotherapy and
subsequent radical surgery following preoperative chemoradiotherapy
in patients with microsatellite stable and microsatellite
instability-high locally advanced rectal cancer. J Clin Oncol. 38
(Suppl 15)(4100)2020.
|
|
35
|
Capdevila J, Declara IM, Martinez MCR,
Maurel J, Hernando J, Alonso V, Graña Suárez B, Plazas JG, Losa F,
Vera R, et al: Phase II study of durvalumab plus total neoadjuvant
therapy (TNT) in locally advanced rectal cancer: The GEMCAD-1703
DUREC trial. J Clin Oncol. 38 (Suppl 15)(TPS4122)2020.
|
|
36
|
Shamseddine A, Zeidan Y, Khalifeh IM,
Kattan JG, Turfa R, Mukherji D, Naji Temraz S, Jamali F, Hani Shaib
Y, Soweid A, et al: Short-course radiation followed by mFOLFOX-6
plus avelumab for locally advanced rectal adenocarcinoma. J Clin
Oncol. 38 (Suppl 4)(S139)2020.PubMed/NCBI View Article : Google Scholar
|