Introduction
A non-negligible number of patients are diagnosed in
the advanced stages of gastric cancer despite advances in mass
screening methods for this malignanacy. Therefore, novel biomarkers
for the early detection of gastric cancer are urgently required to
improve clinical outcomes. Serum autoantibodies against
tumor-associated antigens (TAAs) have been reportedly found in
patients with gastric cancer, including P53(1), Muc1(2), c-myc (3), and Survivin (4). Moreover, autoantibodies could be
detected in the early stages of cancers (5,6).
Because of their easy accessibility and stability, autoantibodies
against TAAs could serve as novel screening biomarkers.
WT1 was originally isolated as a tumor
suppressor gene responsible for Wilms' tumor, a kidney neoplasm of
the childhood, (7). This gene is
overexpressed in various types of cancers such as leukemia
(8), lung (9), colorectal (10), gastric cancer (11), and glioblastoma (12). Furthermore, its gene product is
highly immunogenic (13) and
proves to be a promising target molecule for cancer immunotherapy
(14-17).
We found that IgM and IgG antibodies against WT1 whole protein are
produced in patients with hematopoietic malignancies and lung
cancer (17-19).
The dominant subtypes of WT1 IgG antibody were the Th1 type IgG1
and IgG3(18) and its production
requires helper T cells for IgG class switching; therefore, WT1 IgG
production indicates the activation of Th1 helper T cells in these
patients. We previously reported that the production of IgG
antibody against WT1 whole protein was associated with prolonged
disease-free survival in patients with tumor resected non-small
cell lung cancers (19). In
addition, in our therapeutic cancer vaccine trial with WT1-235
peptide for patients with recurrent glioblastoma, combining the
production of WT1-235 IgG antibody and positive delayed-type
hypersensitivity to the WT1-235 peptide was a better prognostic
marker for long-term overall survival than either parameter alone
(20). Associating WT1 IgG
production with favorable prognosis supports the idea that IgG
antibody against WT1 epitope or epitopes could act a biomarker
indicating, at least, a part of WT1-specific antitumor cellular
immune responses.
Compared with studies on IgG antibodies, fewer
studies have analyzed the production of IgM antibodies in
tumor-associated antigens. However, IgM antibodies are the first
responders in the humoral immune system and do not depend on helper
T lymphocytes for their production (21). Furthermore, the IgM receptor FcμR
is also expressed on T and NK cells in addition to B cells in
humans (22), and IgM itself may
be involved in the regulation of cellular immune responses.
Therefore, IgM antibodies against TAAs could be a biomarker that
can indicate the antigen-specific tumor recognition of the host
immune system in patients with cancer.
In the present study, we explored WT1 epitopes to
identify highly antigenic epitopes in WT1 protein and identified an
18 a.a.-long WT1-271 epitope. We examined the serum positivity of
WT1-271 IgG and IgM antibodies and further analyzed the association
between the production of WT1-271 IgG and IgM antibodies with
clinicopathological character and prognosis in 98 patients with
surgically treated gastric cancer. The positivity of serum WT1-271
IgM antibody was compared with that of conventional gastric cancer
tumor markers CEA and CA19-9.
Materials and methods
Collection of serum samples
This study was approved by the Ethics Committee of
the Toho University Omori hospital (no. A19033) and Osaka
University Hospital Ethical Committee (#13110-10). We collected. a
total of 97 serum samples from patients with gastric cancer who
provided a written informed consent. All patients underwent radical
surgery between January 2014 and July 2017, with 58 cases of distal
gastrectomy, 5 of proximal gastrectomy, and 35 cases of total
gastrectomy, at the Toho University Omori Hospital. All patients
did not undergo neoadjuvant chemotherapy. The pathological stage of
each patient as per the Japanese Classification of Gastric
Carcinoma (23) was as follows: 52
cases with stage I, 19 with stage II, 17 with stage III, and 9 with
stage IV. All patients were followed up until December 2019 or
death. We examined the clinicopathological features and prognosis
of gastric cancer in each patient.
Peptides
We focused on IgG, which, at 21 days, has the
longest half-life among all the isotypes of WT1 antibody. The
production of IgG antibody against each WT1 peptide was analyzed
against the WT1 peptide library. Next, we searched for WT1 peptide
IgG antibodies that may correlate with the WT1 whole protein IgG
levels. Thereafter, we analyzed the WT1 peptide antibodies that may
correlate with tumor WT1 expression levels.
WT1 antigen epitopes were explored from the 18
mer-length WT1 overlapping peptide library to establish a simple
enzyme-linked immunosorbent assay (ELISA) system using a WT1
peptide as a capture antigen and a tool for analyzing humoral
immune responses to WT1. Notably, an 18 mer-length WT1 overlapping
peptide library covering the whole WT1 protein was synthesized at
PH Japan Co. Ltd. (Hiroshima, Japan) with a purity of >75%. The
amino acid sequence of the WT1-271 peptide was
YESDNHTTPILCGAQYRI.
ELISA
ELISA was performed as previously reported (20) with minor modifications. The
peptides (0.2 µg/well) were covalently linked to a 96-well plate
using the Peptide Coating kit (Takara, Shiga, Japan) as per the
manufacturer's instructions. Plates were blocked using Blocking One
(Nacalai Tesque, Kyoto, Japan) that had been diluted with distilled
water (1:5) for 2 h at room temperature and washed with 0.05% TBST.
The sera were diluted at 1:100 using the blocking buffer of the
Peptide Coating kit. Then, 100 µl diluted sera was added to each
well and incubated overnight at 4˚C. After washing with
Tris-buffered saline containing Tween-20 (TBST), horseradish
peroxidase (HRP)-conjugated rabbit anti-human IgG (cat. no.
309-035-003; Jackson ImmunoResearch Europe, Ltd.) or HRP-conjugated
goat anti-human IgM antibody (cat. no. A80-100P; Bethyl
Laboratories, Inc.), diluted at 1:2,000 in TBST, was added to each
well and incubated at room temperature for 2 h. After washing with
TBST, the corresponding third antibody, HRP-conjugated goat
anti-rabbit IgG (cat. no. ab6721; Abcam) or HRP-conjugated rabbit
anti-goat IgG antibody (cat. no. 546; MBL International Co.),
diluted to 1:5,000 in TBST, was added to each well and incubated at
room temperature for 2 h. Bound WT1 epitope-specific IgG or IgM
antibodies were colorimetrically detected using the
3,3',5,5'-tetramethylbenzidine substrate (KPL, Inc.). Absorbance
was measured at 450 nm using a microplate reader (MULTISKAN FC;
Thermo Fisher Scientific, Inc.).
Statistical analyses
Mann-Whitney U test or Chi-squared test was used to
compare the unpaired groups. Fisher's exact test was used to
evaluate the differences in the distribution of two variables.
Kruskal-Wallis test (Mann-Whitney U test with applied Bonferroni's
correction) was used to examine the corresponding differences among
three variables. Logistic regression analysis was used to analyze
the clinicopathological data to evaluate the association with serum
WT1 antibody levels. Survival curves were calculated using the
Kaplan-Meier method and compared using the log-rank test.
Significant predictors were assessed using Cox proportional hazards
model with multivariate analysis. EZR software (version 1.41) was
used for all data analyses (24).
P<0.05 was considered to indicate a statistically significant
difference.
Results
Identification of an antigenic epitope
WT1-271
First, we searched for IgG antibodies that bind to
WT1-derived peptides (WT1 peptide IgG antibodies) whose serum
levels correlated with the serum WT1 protein IgG levels in patients
with intestinal malignancies. For this purpose, we used four sera
samples with high levels of WT1 protein IgG and two samples with
low levels of WT1 protein IgG. Of the seven IgG antibodies that
bound to their corresponding WT1 peptide that were identified,
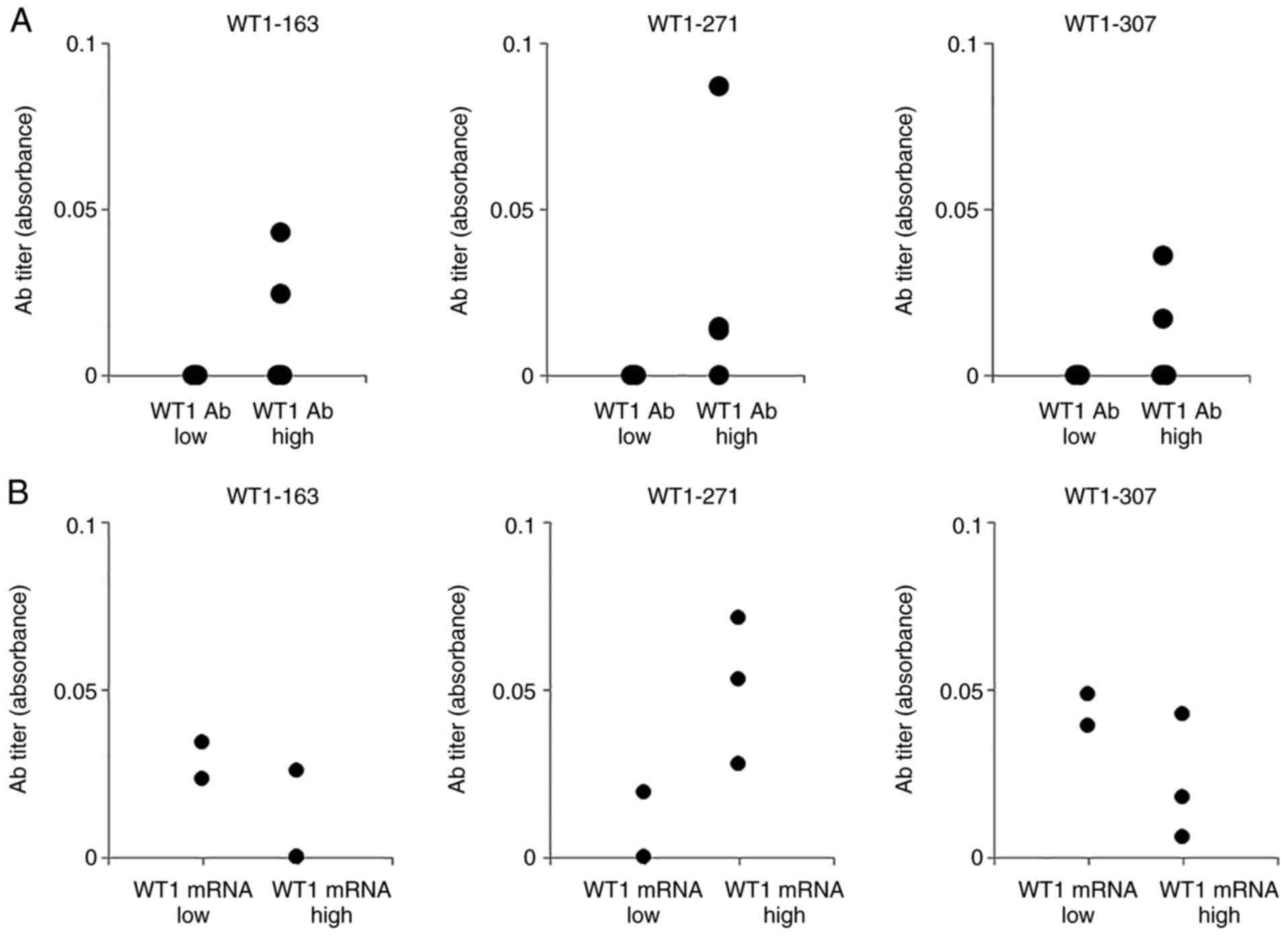
WT1-271 peptide (271-288 a.a.) IgG antibody was identified as a WT1
peptide IgG antibody whose serum levels may correlate with WT1 mRNA
expression levels in peripheral blood using serum samples taken
from five patients with hematological malignancies (Fig. 1). These results may indicate the
potential of using WT1-271 IgM antibody levels as a detection
marker for gastric cancer (Fig.
1).
Increased production of WT1-271 IgM
antibodies in patients with gastric cancer
Production of IgM and IgG antibodies against WT1-271
epitope was investigated in 39 healthy individuals and 97 patients
with gastric cancer to analyze immune recognition of the WT1
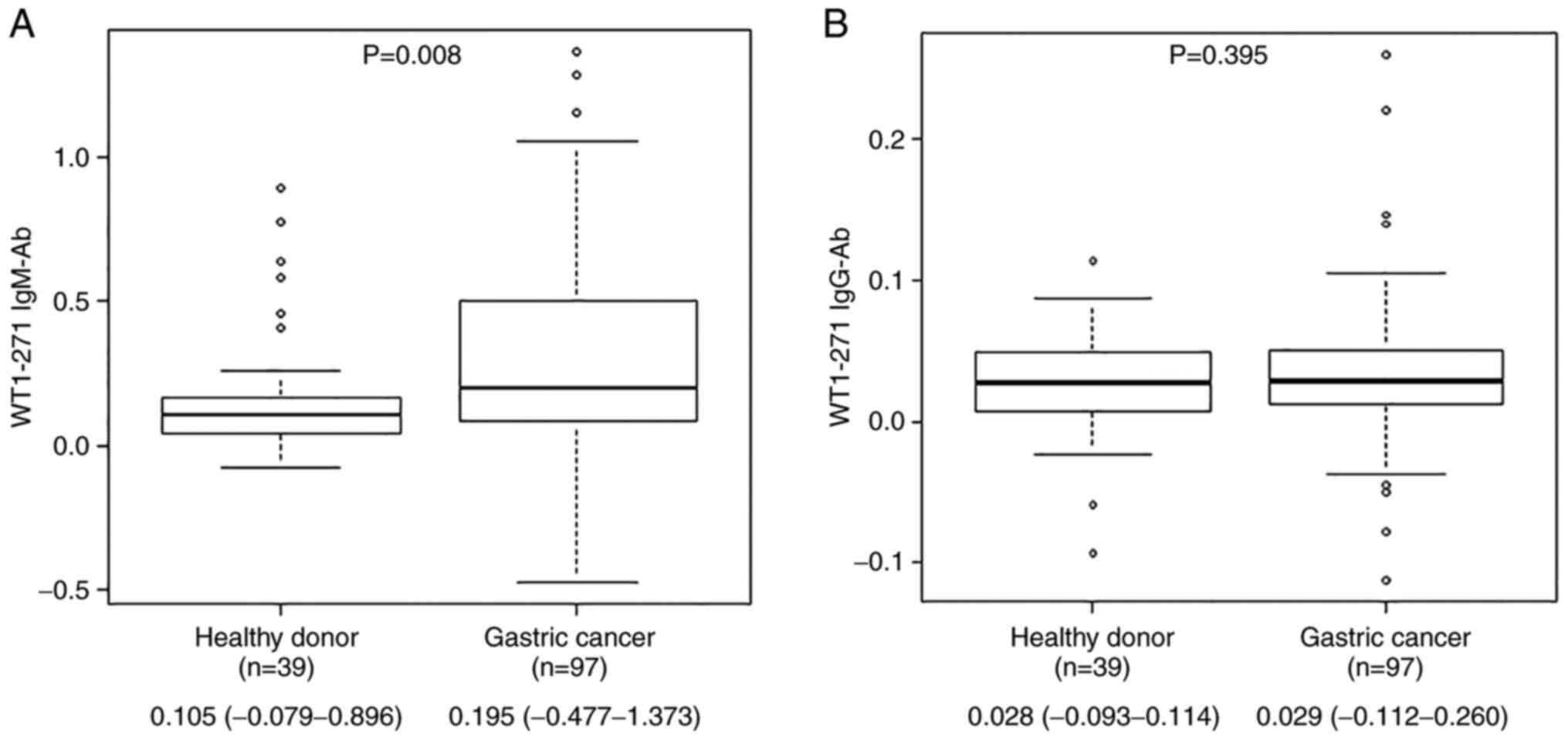
antigen by the host immune system. The median (range) antibody
levels of WT1-271 IgM were 0.105 (-0.079 to 0.896) for healthy
individuals and 0.195 (-0.477 to 1.373) for patients with gastric
cancer, whereas those for IgG were 0.028 (-0.093 to 0.114) for
healthy individuals and 0.029 (-0.112 to 0.260) for patients with
gastric cancer. WT1-271 IgM levels in patients with gastric cancer
were significantly higher than those in healthy individuals as per
the Mann-Whitney U test results (Fig. 2).
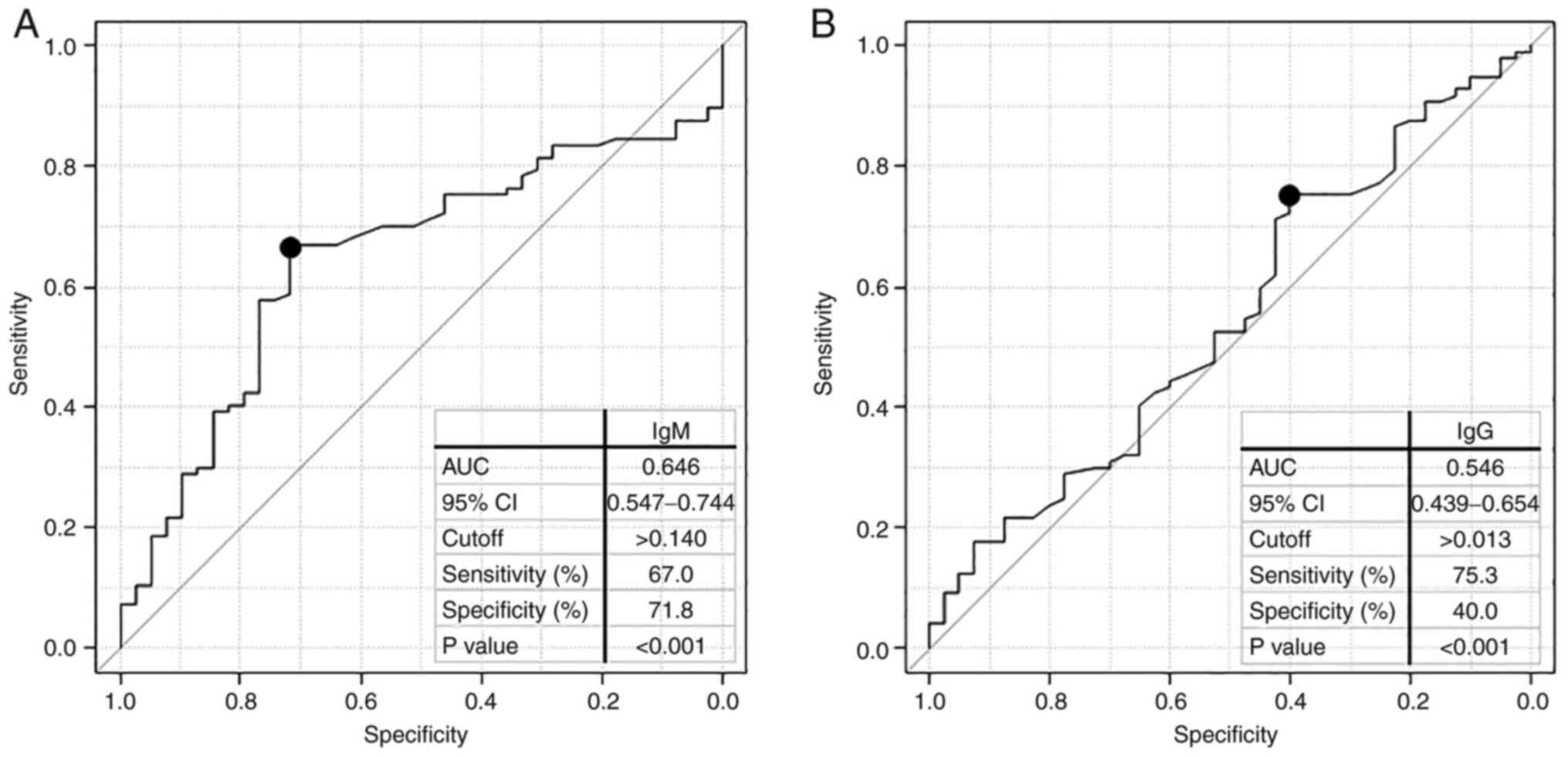
We performed a receiver operating characteristic
(ROC) curve analysis to evaluate the sensitivity and specificity of
healthy individuals and patients with gastric cancer (Fig. 3), and the results revealed that the
area under the curve (AUC) value was <0.6 for WT1-271 IgG but
>0.6 for WT1-271 IgM antibodies (Fig. 3A,B). When the cutoff value for IgM
was considered as 0.140, the sensitivity and specificity of serum
WT1 antibody for patients with gastric cancer were 67.0 and 71.8%,
respectively. Next, we created a 2x2 contingency table for patients
with gastric cancer and healthy individuals to present statistical
evaluation. The positive predictive value was 85.5% and negative
predictive value was 46.7% (Table
I).
 | Table ICorrelation between WT1-271 IgM
antibody positive and negative cases in healthy donors and patients
with gastric cancer. |
Table I
Correlation between WT1-271 IgM
antibody positive and negative cases in healthy donors and patients
with gastric cancer.
| Correlation | Gastric cancer | Healthy donor | Total cases |
|---|
| Positive | 65 | 11 | 76 |
| Negative | 32 | 28 | 60 |
Association between serum WT1-271 IgM
and IgG antibody levels with clinical stages
We focused on the pathological characteristics of 97
surgical cases of patients with gastric cancer and examined their
stages (23). Stages IA and IB
were integrated as I; stages IIA and IIB were integrated as II; and
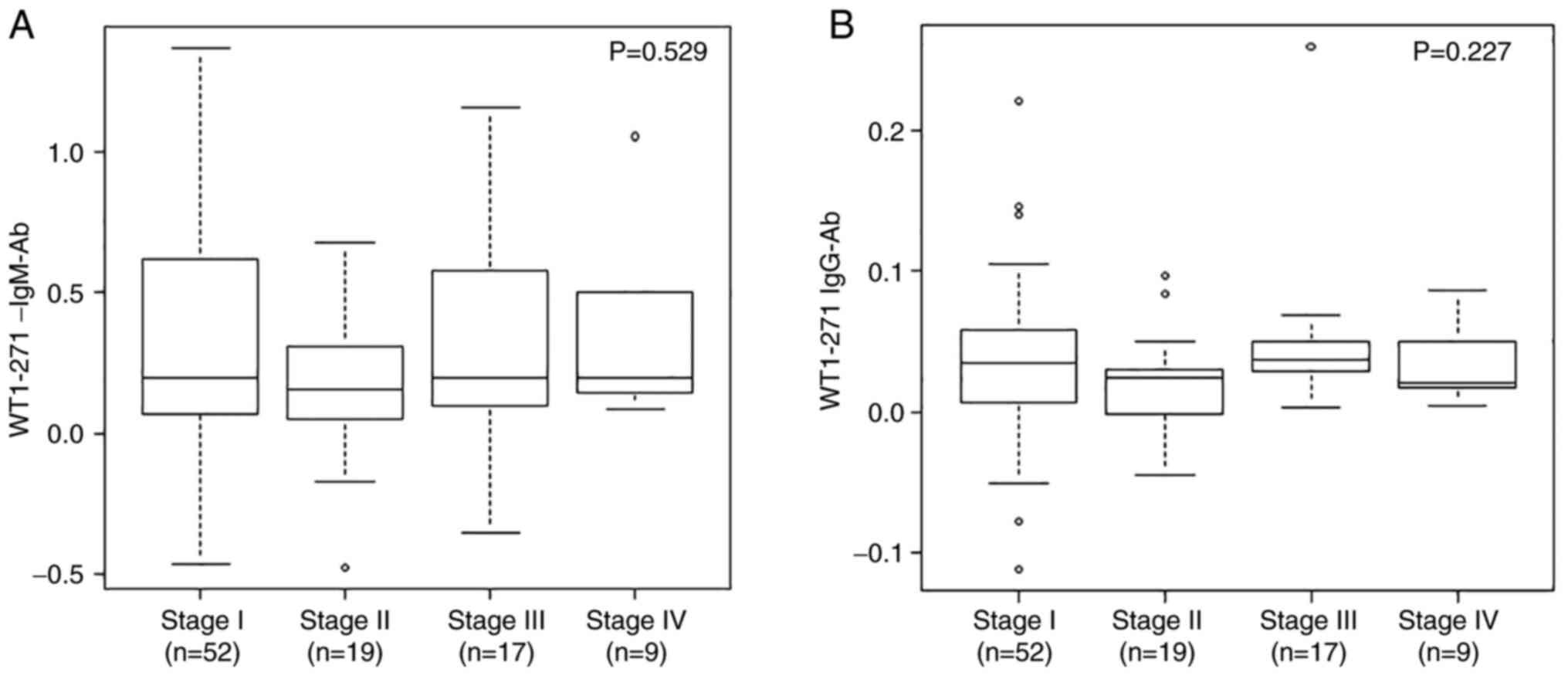
stages IIIA, IIIB, and IIIC were integrated as III. The mean ±
standard deviation serum WT1 IgM antibody levels for stages I
(n=52), II (n=19), III (n=17), and IV (n=9) were 0.327 ± 0.397,
0.178 ± 0.275, 0.309 ± 0.366, and 0.353 ± 0.308, respectively
(Fig. 4). WT1 IgG antibody levels
for stages I, II, III, and IV were 0.037 ± 0.053, 0.020 ± 0.035,
0.049 ± 0.057, and 0.035 ± 0.029, respectively. Based on the
Kruskal-Wallis test results, there was no significant association
between the clinical stage and WT1-271 antibody levels. In terms of
the pathological type, 57 cases were differentiated and 40 cases
were poorly differentiated. Recurrence or liver, lymphatic node,
bone and peritoneal metastases were observed in 19 patients. The
remaining 78 patients showed no recurrence or metastasis.
Association between serum WT1-271 IgM
antibody levels and clinicopathological factors
We examined the association of WT1-271 IgM levels
with clinicopathological factors using Fisher's exact probability
test and logistic regression analysis using the cutoff level as
0.140 for the elevation of serum IgM level. According to the
univariate analysis results, no significant difference was found in
terms of gender, age, tumor depth (T1 vs. T2-4), lymph node (LN)
metastasis and white blood cell counts, and CEA and CA19-9 levels
(Table II, left panel). However,
logistic regression analysis revealed that WT1-271 IgM positivity
was significantly associated with earlier T stage (T1 vs. T2-4;
P=0.049) but tended to be associated with advanced N stage (N0 vs.
N1; P=0.125) (Table II, right
panel). Table III shows WT1-271
IgM positive rates in combination to the T and N stages. In both T1
and T2-4 stages, the WT1-271 IgM positive rate was higher in N1
stage than in N0.
 | Table IIComparison of serum wilms tumor 1 IgM
levels according to the clinicopathological characteristics of the
patients with gastric cancer. |
Table II
Comparison of serum wilms tumor 1 IgM
levels according to the clinicopathological characteristics of the
patients with gastric cancer.
| | Fisher's exact
probability test | Logistic regression
analysis |
|---|
| Variables | 271-IgM level
<0.140 | 271-IgM level
≥0.140 | P-value | Odds ratio | 95% CI | P-value |
|---|
| Sex | | | >0.999 | | | |
|
Male | 24 | 47 | | | | |
|
Female | 8 | 18 | | | | |
| Age | | | 0.329 | | | |
|
>65 | 21 | 50 | | | | |
|
≤65 | 11 | 15 | | | | |
| Tumor depth | | | 0.518 | 2.994 | 0.112-0.997 | 0.049 |
|
T2-T4 | 18 | 31 | | | | |
|
T1 | 14 | 34 | | | | |
| Lymph node
metastasis | | | 0.370 | 2.500 | 0.775-8.100 | 0.125 |
|
N1 | 9 | 25 | | | | |
|
N0 | 23 | 40 | | | | |
| WBC (/µl) | | | >0.999 | | | |
|
>8,000 | 4 | 9 | | | | |
|
≤8,000 | 28 | 56 | | | | |
| CEA (ng/ml) | | | 0.472 | | | |
|
>5.0 | 4 | 5 | | | | |
|
≤5.0 | 28 | 60 | | | | |
| CA19-9 (U/ml) | | | 0.095 | 6.520 | 0.752-56.60 | 0.089 |
|
>37.0 | 1 | 10 | | | | |
|
≤37.0 | 31 | 55 | | | | |
 | Table IIIWilms tumor 1-271 IgM positive rates
in a combination of pathological T and N stages. |
Table III
Wilms tumor 1-271 IgM positive rates
in a combination of pathological T and N stages.
| Pathological
stage | Negative | Positive | Total | Positive rate
(%) |
|---|
| T1N0 | 14 | 31 | 45 | 68.9 |
| T1N1 | 0 | 3 | 3 | 100.0 |
| T2-4N0 | 9 | 9 | 18 | 50.0 |
| T2-4N1 | 9 | 22 | 31 | 71.0 |
Association between serum WT1-271 IgM
antibody levels and overall survival
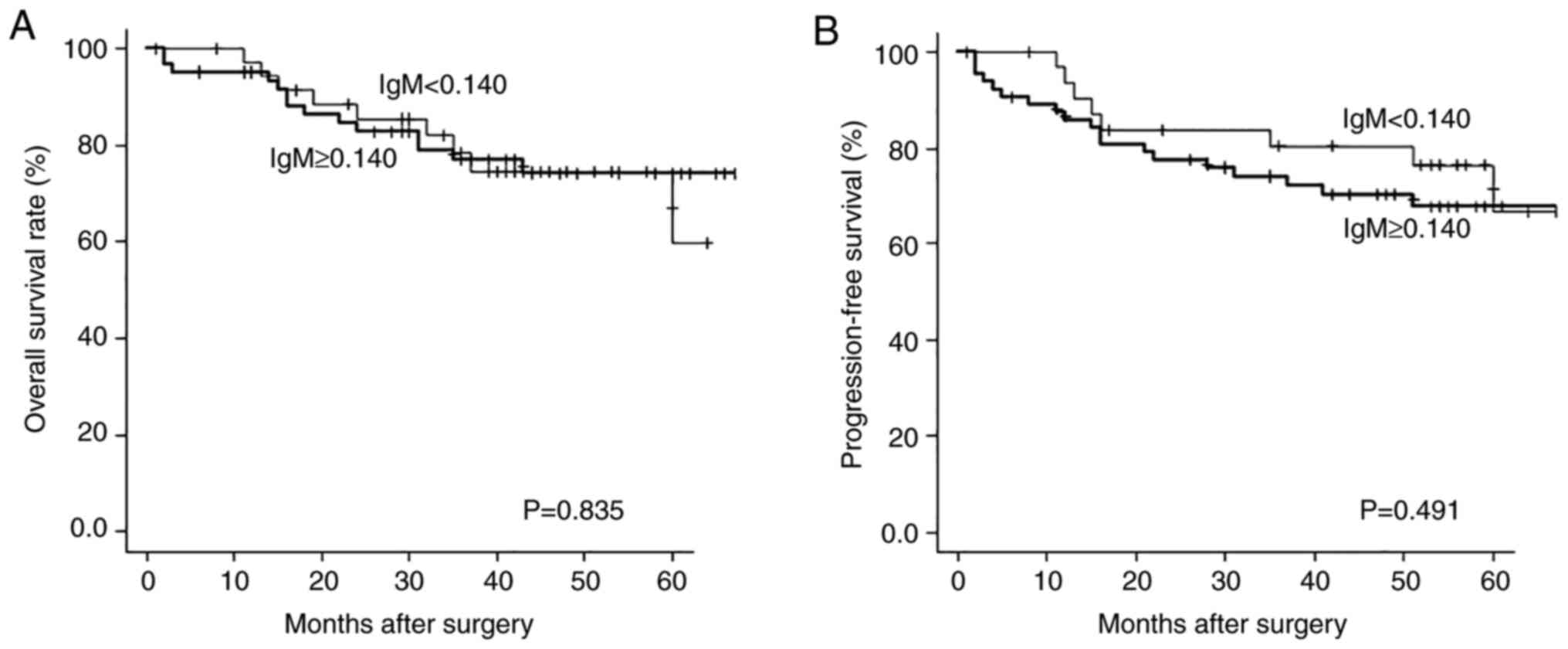
Using the log-rank test, we investigated the
association between serum WT1-271 IgM levels and clinical outcome
in patients with gastric cancer. Based on the cutoff level of
0.140, we compared the overall survival and progression-free
survival between the two groups of patients with high and low
WT1-271 IgM levels. At 60 months after surgery, the P-values for
overall survival and progression-free survival were 0.835 and
0.491. However, similar to overall survival, progression-free
survival tended to be shorter in the high WT1-271 IgM group for up
to 57 months. No significant difference was found between the two
groups (Fig. 5A,B). However,
analysis at 6 months after surgical resection revealed that
although the difference was not significant, the immunological
status represented by higher serum WT1-271 IgM levels might have an
unfavorable impact on the clinical outcome (Fig. S1).
Univariate analysis revealed that tumor depth, LN
metastasis, and CEA and CA19-9 levels indicated significantly worse
overall survival on the log-rank test. However, WT1-271 IgM levels
was not significantly associated with overall survival of patients
with gastric cancer (Table IV,
left panel).
 | Table IVUnivariate and multivariate analysis
of risk factors for the overall survival of patients with gastric
cancer. |
Table IV
Univariate and multivariate analysis
of risk factors for the overall survival of patients with gastric
cancer.
| | Multivariate
analysis |
|---|
| Variable | Univariate analysis
P-valuea | Hazard ratio | 95% CI |
P-valueb |
|---|
| Male vs.
female | 0.158 | | | |
| Age >65 vs. ≤65
years | 0.068 | | | |
| Tumor depth T1 vs.
T2-4 | <0.001 | 4.945 | 1.776-13.770 | 0.002 |
| Lymph node
metastasis N-vs. N+ | <0.001 | | | |
| WBC (/µl) >8,000
vs. ≤8,000 | 0.073 | | | |
| CEA (ng/ml) >5.0
vs. ≤5.0 | <0.001 | 4.713 | 1.664-13.350 | 0.004 |
| CA19-9 (U/ml)
>37 vs. ≤37 | 0.007 | | | |
| WT1 IgM ≥0.140 vs.
<0.140 | 0.835 | 1.700 | 0.672-4.299 | 0.262 |
Multivariate analysis indicated that tumor, depth,
and CEA levels were independent prognostic factors in the Cox
proportional hazard model. However, no significant association was
found between serum WT1 IgM levels and overall survival in patients
with gastric cancer (Table IV,
right panel).
WT1-271 IgM antibody levels as a
detection marker for gastric cancer
We compared the detection ability of WT1-271 IgM
antibodies for gastric cancer with that of the currently available
tumor markers CEA and CA19-9 (Table
V). In patients with gastric cancer, positive rates of CEA and
CA19-9 were 9.3 and 11.3%, respectively. Using chi-squared test
WT1-271 IgM antibodies showed a distinctively higher positive rate
of 67.0% in patients with gastric cancer (P<0.001). Of the 52
patients with clinical stage I disease, WT1-271 IgM antibody was
detected in 35 patients (67.3%), whereas either CEA or CA19-9 was
detected in only 1 by one (1.9%) (Table V). Even in stage II, III and IV,
higher positive rate was shown in WT1-271 IgM antibodies (66.7%)
than CEA (17.8%) or CA19-9 (22.2%). These results may show the
potential of WT1-271 IgM antibody levels as a detection marker for
gastric cancer.
 | Table VRelationship of WT1-271 IgM antibody
positivity and each tumor marker according to gastric cancer
stage. |
Table V
Relationship of WT1-271 IgM antibody
positivity and each tumor marker according to gastric cancer
stage.
| Marker | Stage I (n=52), n
(%) | Stage II, III and
IV (n=45), n (%) | All stages (n=97),
n (%) |
|---|
| IgM | | | |
|
Negative | 17 (32.7) | 15 (33.3) | 32 (33.0) |
|
Positive | 35 (67.3) | 30 (66.7) | 65 (67.0) |
| CEA | | | |
|
Negative | 51 (98.1) | 37 (82.2) | 88 (90.7) |
|
Positive | 1 (1.9) | 8 (17.8) | 9 (9.3) |
| CA19-9 | | | |
|
Negative | 51 (98.1) | 35 (77.8) | 86 (88.7) |
|
Positive | 1 (1.9) | 10 (22.2) | 11 (11.3) |
| All negative | 17 (32.7) | 11 (24.4) | 28 (25.9) |
Discussion
In this study, we identified WT1-271, a 271-288-a.a.
long WT1 sequence, as an antigenic epitope for antibody production
and determined that, compared with healthy individuals, patients
with gastric cancer produced significantly higher levels of WT1-271
IgM but not IgG antibodies. A cutoff level of 0.14 was determined
for positive serum WT1-271 IgM antibodies based on the ROC analysis
with an AUC of 0.6. According to this cutoff level, 67% of the
patients with gastric cancer were scored as positive for WT1-271
IgM antibodies. Autoantibodies corresponded to an efficient
biological amplification of the antigens in tumors and are secreted
in the serum before the antigens can be detected (25). In contrast to the high positive
rate of WT1-271 IgM antibodies, currently available gastric cancer
tumor markers CEA and CA19-9 were positive in only 9.3 and 11.3% of
the patients, respectively, in this study. This higher positivity
of WT1-271 IgM antibodies is more evident in patients in clinical
stage I of the disease. CEA or CA19-9 was detected in only 1.9% of
the patients, whereas WT1-271 IgM antibody was detected in 67.3%.
Early detection is one of the most promising approaches to improve
clinical outcomes for patients with cancer. Considerable efforts
have been made to develop more sensitive and specific tests to
detect cancer with different combinations of autoantibodies
(6,26-28).
Because of its high sensitivity to early gastric cancers, serum
WT1-271 IgM antibody may be used as a detection marker in the
screening of gastric cancer in combination with autoantibodies,
especially in the early stages.
Two-thirds of the patients with gastric cancer were
positive for WT1-271 IgM antibody for the cutoff level determined
by the ROC analysis. Two reasons can explain this high positivity
of WT1-271 IgM antibody in patients with gastric cancer. First,
this can be explained by the high frequency of WT1 overexpression
in gastric cancer tumors as reported in our previous
immunohistochemical study, which indicated that tumor cells
overexpressed WT1 protein in 42.0% of patients with gastric cancer.
Moreover, WT1 overexpression was more frequent (65.2%) in well- or
moderately-differentiated gastric adenocarcinoma (11). Most patients included in the study
had well- or moderately-differentiated adenocarcinomas, and thus,
the majority of tumors overexpressed the WT1 protein. Second, the
high positivity of the WT1-271 IgM antibody can be due to its T
cell-independent production. In B-cell differentiation, the
Ig-constant regions can be changed from IgM to other Ig isotypes
through class-switch recombination. Because the class-switch into
IgG is dependent on helper T cells, insufficient helper T-cell
function may impair IgG production. In contrast, IgM antibodies are
produced independently of helper T cells (21).
The immune system recognizes nonself in the body,
and its components (immune cells and humoral factors) respond in an
orchestrated manner. Production of IgM and IgG antibodies against
the WT1-271 epitope may reflect the immune recognition and
responses to the tumor-associated antigen WT1 in patients with
gastric cancer. A significant increase in WT1-271 IgM in patients
with gastric cancer compared with healthy individuals represents
the recognition of the WT1-271 antigen by the immune system of the
patient. In this study, the elevated serum WT1-271 IgM levels
indicated two aspects of WT1 immune recognition in patients with
gastric cancer. First, serum WT1 271 IgM level was increased in
patients with T1N0 gastric cancer wherein the tumor remained within
the gastric submucosal layer without LN metastasis. This B cell
response in the very early stage of gastric cancer may support the
concept of immune surveillance, which proposes eliminating cancer
cells in the early stages of cancer development (29,30).
Second, in T1 and T2-4 stages, the WT1-271 IgM positive rate in the
N1 stage was higher than that in the N0. Reportedly, draining LNs
are active sites for B- and T-cell responses in early-stage breast
cancers. McDaniel et al (31) reported that draining LNs are a rich
source of tumor-reactive B cells. In addition, Gillmore et
al (32) found WT1-specific
CD8 CTLs in the draining LNs in stage I/II breast cancer. These
findings for breast cancer allow us to consider that the draining
LNs of gastric cancer are active sites of WT1-specific immune
responses, and metastasis of WT1-expressing tumor cells to the
draining LNs could have triggered WT1-271 IgM production.
In the present study, we identified the WT1-271 IgG
antibody as whose serum level correlated with serum WT1 protein IgG
levels in patients with intestinal malignancies. It may be
associated with WT1 mRNA expression levels in the peripheral blood
of patients with hematological malignancies. These indicate that
the WT1-271 antibodies are produced in response to an antigenic
overload of WT1 antigens from tumors. Although serum WT1-271 IgM
levels were elevated in most patients with gastric cancer, WT1-271
IgG levels were low and did not significantly differ from those in
healthy individuals. These results indicate a lack of robust
WT1-specific Th help in the patients (20). This is supported by the findings of
the present study that high serum WT1-271 IgM levels is associated
with unfavorable overall survival 6 months after the surgical
resection (Fig. S1), although
this was not statistically significant. Similar to overall
survival, progression-free survival tended to be shorter in the
high-WT1-271 IgM group for up to 57 months. Future studies are
required to examine whether WT1-271 IgM antibody could help predict
the prognosis of patients with gastric cancer.
Recently, several studies (33-36)
have reported the involvement of B cells in cellular immunity as
well as humoral immunity. B cells may play protumorigenic roles
through the secretion of immunosuppressive cytokines, such as IL-10
(33,34) and TGF-β (35). However, B cells may also have
antitumorigenic functions through IFN-γ secretion to enhance tumor
killing by NK cells and CTLs (34,36)
and even kill tumor cells directly via the Fas-FasL system
(35). A possible association
between WT1-271 IgM antibody and the unfavorable clinical outcome
could partly be a result of the pro-tumorigenic cellular functions
of WT1-271-specific B cells under the persistent antigenic overload
of WT1 in patients with WT1-expressing gastric cancer.
One-third of the healthy individuals in this study
were positive for WT1-271 IgM antibodies. One explanation for this
is the production of WT1-271 IgM due to the presence of latent
cancer. Autoantibodies against tumor-associated antigens can appear
in the early stages of cancer development and could be detected
months to years before the onset of clinical symptoms. Another
explanation for the high positive rate of WT1-271 IgM in healthy
individuals is that the WT1-271 IgM antibody is an IgM natural
autoantibody. Researchers have reported that natural autoantibodies
are spontaneously produced and can exist without antigen
stimulation (37-39).
Historically, natural autoantibodies have been primarily associated
with autoimmune diseases (37).
However, compelling evidence indicates that not all natural human
autoantibodies are pathogenic. Future studies are necessary to
elucidate the role of both WT1-271-specific B cells and their
producing WT1-271 autoantibodies in antitumor immunity of healthy
individuals.
Some limitations of the study is the unknown
expression of WT1 protein in the tumor cells and the lack of
samples from patients with benign diseases. Future studies are
warranted to demonstrate the diagnostic value of the WT1-271 IgM
antibody in gastric cancer.
We identified WT1-271, a representative highly
immunogenic epitope, among multiple epitopes in WT1. Preoperative
serum WT1-271 IgM antibody levels in patients with gastric cancer
were significantly higher than those in healthy individuals.
Although the serum WT1-271 IgM antibody was not associated with
clinicopathological factors, it could be used as a diagnostic
biomarker for gastric cancer.
Supplementary Material
Survival curve at 6 months after
surgical resection was performed by median. High serum Wilms tumor
1-271 IgM levels tended to associate with unfavorable clinical
outcome in the six months after the surgical resection. The P-value
was obtained using a Log-Rank test.
Acknowledgements
The authors would like to thank Ms. Seiko Otsuka,
Ms. Chiho Kusaka and Ms. Satoko Ishibashi (Toho University Graduate
School of Medicine) for preparing patient data.
Funding
Funding: The current study was supported by the Project for
Cancer Research and Therapeutic Evolution (P-CREATE) from the Japan
Agency for Medical Research and Development, AMED (grant no.
21cm0106403h0006) and Grants-in-Aid for Scientific Research
(KAKENHI) from the Japan Society for the Promotion of Science, JSPS
(grant no. 16K10520).
Availability of data and materials
All data generated or analyzed during this study are
included in this published article.
Authors' contributions
MI, YOj, HSh and HSu designed the study. MI, YOj and
HSh wrote the manuscript. YOj, MA, RI and SA performed biological
measurement. YOs, SY, TS, TN, MS, FS and KF collected patient
sample data. MI, YOj, HSh and TH analyzed data. HSu, HSh and KF
confirmed the authenticity of all the raw data. All authors read
and approved the final manuscript.
Ethics approval and consent to
participate
The present study was approved by the Ethics
Committee of the Toho University Omori hospital, Tokyo, Japan
(approval no. A19033). All patients provided written informed
consent.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Shimada H: p53 molecular approach to
diagnosis and treatment of esophageal squamous cell carcinoma. Ann
Gastroenterol Surg. 2:266–273. 2018.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Kurtenkov O, Klaamas K, Mensdorff-Pouilly
S, Miljukhina L, Shljapnikova L and Chuzmarov V: Humoral immune
response to MUC1 and to the Thomsen-Friedenreich (TF) glycotope in
patients with gastric cancer: Relation to survival. Acta Oncol.
46:316–323. 2007.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Koziol JA, Zhang JY, Casiano CA, Peng XX,
Shi FD, Feng AC, Chan EK and Tan EM: Recursive partitioning as an
approach to selection of immune markers for tumor diagnosis. Clin
Cancer Res. 9:5120–5126. 2003.PubMed/NCBI
|
|
4
|
Megliorino R, Shi FD, Peng XX, Wang X,
Chan EK, Tan EM and Zhang JY: Autoimmune response to anti-apoptotic
protein survivin and its association with antibodies to p53 and
c-myc in cancer detection. Cancer Detect Prev. 29:241–248.
2005.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Werner S, Chen H, Tao S and Brenner H:
Systematic review: Serum autoantibodies in the early detection of
gastric cancer. Int J Cancer. 136:2243–2252. 2015.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Hoshino I, Nagata M, Takiguchi N, Nabeya
Y, Ikeda A, Yokoi S, Kuwajima A, Tagawa M, Matsushita K, Satoshi Y
and Hideaki S: Panel of autoantibodies against multiple
tumor-associated antigens for detecting gastric cancer. Cancer Sci.
108:308–315. 2017.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Call KM, Glaser T, Ito CY, Buckler AJ,
Pelletier J, Haber DA, Rose EA, Kral A, Yeger H, Lewis WH, et al:
Isolation and characterization of a zinc finger polypeptide gene at
the human chromosome 11 Wilms' tumor locus. Cell. 60:509–520.
1990.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Inoue K, Ogawa H, Yamagami T, Soma T, Tani
Y, Tatekawa T, Oji Y, Tamaki H, Kyo T, Dohy H, et al: Long-term
follow-up of minimal residual disease in leukemia patients by
monitoring WT1 (Wilms tumor gene) expression levels. Blood.
88:2267–2278. 1996.PubMed/NCBI
|
|
9
|
Oji Y, Miyoshi S, Maeda H, Hayashi S,
Tamaki H, Nakatsuka S, Yao M, Takahashi E, Nakano Y, Hirabayashi H,
et al: Overexpression of the Wilms' tumor gene WT1 in de novo lung
cancers. Int J Cancer. 100:297–303. 2002.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Oji Y, Yamamoto H, Nomura M, Nakano Y,
Ikeba A, Nakatsuka S, Abeno S, Kiyotoh E, Jomgeow T, Sekimoto M, et
al: Overexpression of the Wilms' tumor gene WT1 in colorectal
adenocarcinoma. Cancer Sci. 94:712–717. 2003.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Nakatsuka S, Oji Y, Horiuchi T, Kanda T,
Kitagawa M, Takeuchi T, Kawano K, Kuwae Y, Yamauchi A, Okumura M,
et al: Immunohistochemical detection of WT1 protein in a variety of
cancer cells. Mod Pathol. 19:804–814. 2006.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Oji Y, Suzuki T, Nakano Y, Maruno M,
Nakatsuka S, Jomgeow T, Abeno S, Tatsumi N, Yokota A, Aoyagi S, et
al: Overexpression of the Wilms' tumor gene W T1 in primary
astrocytic tumors. Cancer Sci. 95:822–827. 2004.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Cheever MA, Allison JP, Ferris AS, Finn
OJ, Hastings BM, Hecht TT, Mellman I, Prindiville SA, Viner JL,
Weiner LM and Matrisian LM: The prioritization of cancer antigens:
A national cancer institute pilot project for the acceleration of
translational research. Clin Cancer Res. 15:5323–5337.
2009.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Oka Y, Tsuboi A, Taguchi T, Osaki T, Kyo
T, Nakajima H, Elisseeva OA, Oji Y, Kawakami M, Ikegame K, et al:
Induction of WT1 (Wilms' tumor gene)-specific cytotoxic T
lymphocytes by WT1 peptide vaccine and the resultant cancer
regression. Proc Natl Acad Sci USA. 101:13885–13890.
2004.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Keilholz U, Letsch A, Busse A, Asemissen
AM, Bauer S, Blau IW, Hofmann WK, Uharek L, Thiel E and
Scheibenbogen C: A clinical and immunologic phase 2 trial of Wilms
tumor gene product 1 (WT1) peptide vaccination in patients with AML
and MDS. Blood. 113:6541–6548. 2009.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Anguille S, Van de Velde AL, Smits EL, Van
Tendeloo VF, Juliusson G, Cools N, Nijs G, Stein B, Lion E, Van
Driessche A, et al: Dendritic cell vaccination as postremission
treatment to prevent or delay relapse in acute myeloid leukemia.
Blood. 130:1713–1721. 2017.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Elisseeva OA, Oka Y, Tsuboi A, Ogata K, Wu
F, Kim EH, Soma T, Tamaki H, Kawakami M, Oji Y, et al: Humoral
immune responses against Wilms tumor gene WT1 product in patients
with hematopoietic malignancies. Blood. 99:3272–3279.
2002.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Wu F, Oka Y, Tsuboi A, Elisseeva OA, Ogata
K, Nakajima H, Fujiki F, Masuda T, Murakami M, Yoshihara S, et al:
Th1-biased humoral immune responses against Wilms tumor gene WT1
product in the patients with hematopoietic malignancies. Leukemia.
19:268–274. 2005.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Oji Y, Kitamura Y, Kamino E, Kitano A,
Sawabata N, Inoue M, Mori M, Nakatsuka S, Sakaguchi N, Miyazaki K,
et al: WT1 IgG antibody for early detection of nonsmall cell lung
cancer and as its prognostic factor. Int J Cancer. 125:381–387.
2009.PubMed/NCBI View Article : Google Scholar
|
|
20
|
Oji Y, Hashimoto N, Tsuboi A, Murakami Y,
Iwai M, Kagawa N, Chiba Y, Izumoto S, Elisseeva O, Ichinohasama R,
et al: Association of WT1 IgG antibody against WT1 peptide with
prolonged survival in glioblastoma multiforme patients vaccinated
with WT1 peptide. Int J Cancer. 139:1391–1401. 2016.PubMed/NCBI View Article : Google Scholar
|
|
21
|
Jones K, Savulescu AF, Brombacher F and
Hadebe S: Immunoglobulin M in Health and Diseases: How Far Have We
Come and What Next? Front Immunol. 11(595535)2020.PubMed/NCBI View Article : Google Scholar
|
|
22
|
Kubagawa H, Oka S, Kubagawa Y, Torii I,
Takayama E, Kang DW, Gartland GL, Bertoli LF, Mori H, Takatsu H, et
al: Identity of the elusive IgMFc receptor (FcmuR) in humans. J Exp
Med. 206:2779–2793. 2009.PubMed/NCBI View Article : Google Scholar
|
|
23
|
Japanese Gastric Cancer Association.
Japanese classification of gastric carcinoma: 3rd English edition.
Gastric Cancer. 14:101–112. 2011.PubMed/NCBI View Article : Google Scholar
|
|
24
|
Kanda Y: Investigation of the freely
available easy-to-use software ‘EZR’ for medical statistics. Bone
Marrow Transplant. 48:452–458. 2013.PubMed/NCBI View Article : Google Scholar
|
|
25
|
Desmetz C, Mange A, Maudelonde T and
Solassol J: Autoantibody signatures: Progress and perspectives for
early cancer detection. J Cell Mol Med. 15:2013–2024.
2011.PubMed/NCBI View Article : Google Scholar
|
|
26
|
Meistere I, Werner S, Zayakin P, Siliņa K,
Rulle U, Pismennaja A, Šantare D, Kikuste I, Isajevs S, Leja M, et
al: The prevalence of cancer-associated autoantibodies in patients
with gastric cancer and progressive grades of premalignant lesions.
Cancer Epidemiol Biomarkers Prev. 26:1564–1574. 2017.PubMed/NCBI View Article : Google Scholar
|
|
27
|
Yang Q, Qin J, Sun G, Qiu C, Jiang D, Ye
H, Wang X, Dai L, Zhu J, Wang P and Zhang J: Discovery and
validation of serum autoantibodies against tumor-associated
antigens as biomarkers in gastric adenocarcinoma based on the
focused protein arrays. Clin Transl Gastroenterol.
12(e00284)2020.PubMed/NCBI View Article : Google Scholar
|
|
28
|
Zayakin P, Ancāns G, Siliņa K, Meistere I,
Kalniņa Z, Andrejeva D, Endzeliņš E, Ivanova L, Pismennaja A,
Ruskule A, et al: Tumor associated autoantibody signature for the
early detection of gastric cancer. Int J Cancer. 132:137–147.
2013.PubMed/NCBI View Article : Google Scholar
|
|
29
|
Shankaran V, Ikeda H, Bruce AT, White JM,
Swanson PE, Old LJ and Schreiber RD: IFNgamma and lymphocytes
prevent primary tumour development and shape tumour immunogenicity.
Nature. 410:1107–1111. 2001.PubMed/NCBI View
Article : Google Scholar
|
|
30
|
Schreiber RD, Old LJ and Smyth MJ: Cancer
immunoediting: Integrating immunity's roles in cancer suppression
and promotion. Science. 331:1565–1570. 2011.PubMed/NCBI View Article : Google Scholar
|
|
31
|
McDaniel JR, Pero SC, Voss WN, Shukla GS,
Sun Y, Schaetzle S, Lee CH, Horton AP, Harlow S, Gollihar J, et al:
Identification of tumor-reactive B cells and systemic IgG in breast
cancer based on clonal frequency in the sentinel lymph node. Cancer
Immunol Immunother. 67:29–738. 2018.PubMed/NCBI View Article : Google Scholar
|
|
32
|
Gillmore R, Xue SA, Holler A, Kaeda J,
Hadjiminas D, Healy V, Dina R, Parry SC, Bellantuono I, Ghani Y, et
al: Detection of Wilms' tumor antigen-specific CTL in
tumor-draining lymph nodes of patients with early breast cancer.
Clin Cancer Res. 12:34–42. 2006.PubMed/NCBI View Article : Google Scholar
|
|
33
|
Sarvaria A, Madrigal JA and Saudemont A: B
cell regulation in cancer and anti-tumor immunity. Cell Mol
Immunol. 14:662–674. 2017.PubMed/NCBI View Article : Google Scholar
|
|
34
|
Tao H, Lu L, Xia Y, Dai F, Wang Y, Bao Y,
Lundy SK, Ito F, Pan Q, Zhang X, et al: Antitumor effector B cells
directly kill tumor cells via the Fas/FasL pathway and are
regulated by IL-10. Eur J Immunol. 45:999–1009. 2015.PubMed/NCBI View Article : Google Scholar
|
|
35
|
Olkhanud PB, Damdinsuren B, Bodogai M,
Gress RE, Sen R, Wejksza K, Malchinkhuu E, Wersto RP and Biragyn A:
Tumor-evoked regulatory B cells promote breast cancer metastasis by
converting resting CD4+ T cells to T-regulatory cells. Cancer Res.
71:3505–3515. 2011.PubMed/NCBI View Article : Google Scholar
|
|
36
|
Schwartz M, Zhang Y and Rosenblatt JD: B
cell regulation of the anti-tumor response and role in
carcinogenesis. J Immunother Cancer. 4(40)2016.PubMed/NCBI View Article : Google Scholar
|
|
37
|
Fereidan-Esfahani M, Nayfeh T, Warrington
A, Howe CL and Rodriguez M: IgM Natural autoantibodies in
physiology and the treatment of disease. Methods Mol Biol.
1904:53–81. 2019.PubMed/NCBI View Article : Google Scholar
|
|
38
|
Meffre E and Salmon JE: Autoantibody
selection and production in early human life. J Clin Invest.
117:598–601. 2007.PubMed/NCBI View Article : Google Scholar
|
|
39
|
Elkon K and Casali P: Nature and functions
of autoantibodies. Nat Clin Pract Rheumatol. 4:491–498.
2008.PubMed/NCBI View Article : Google Scholar
|