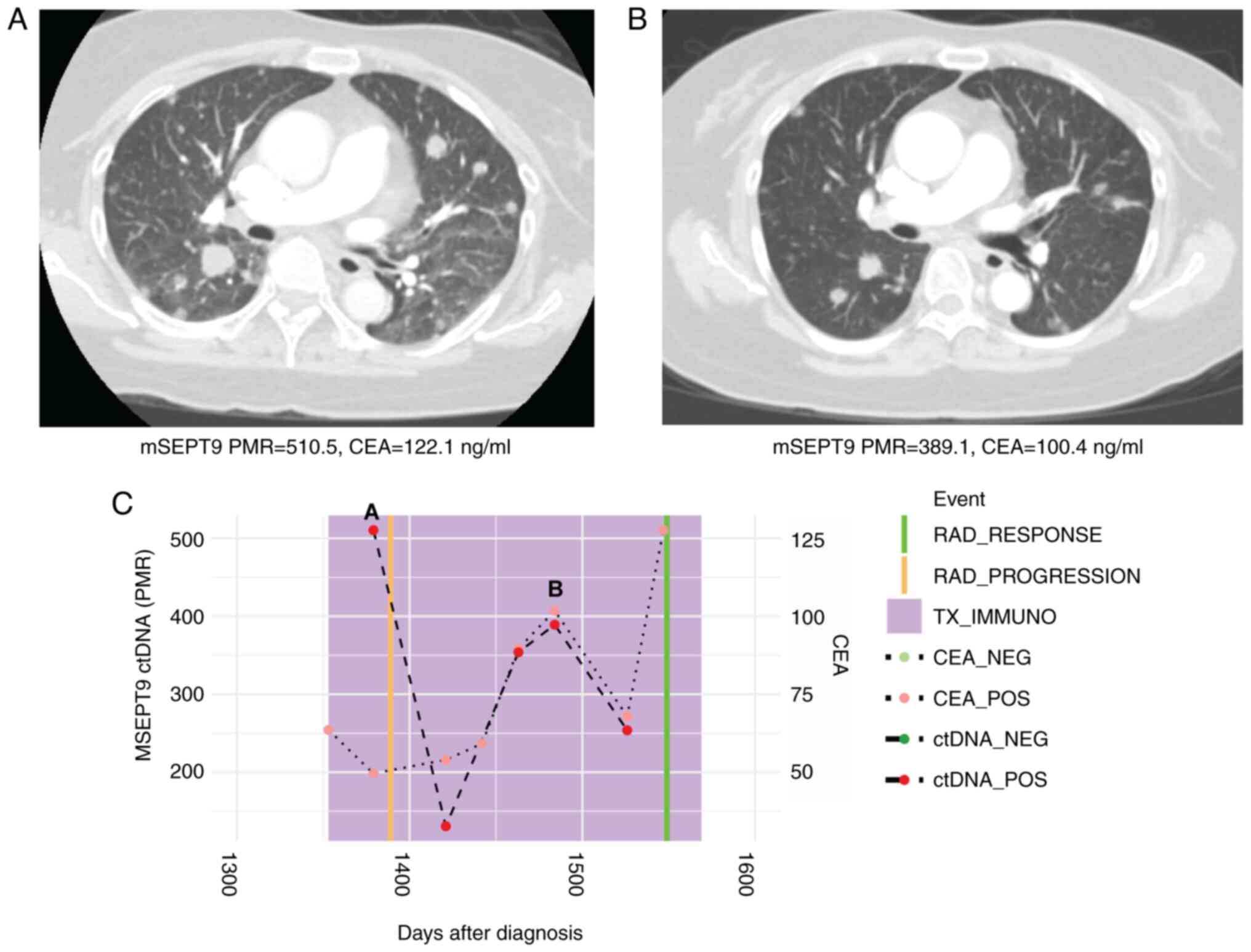
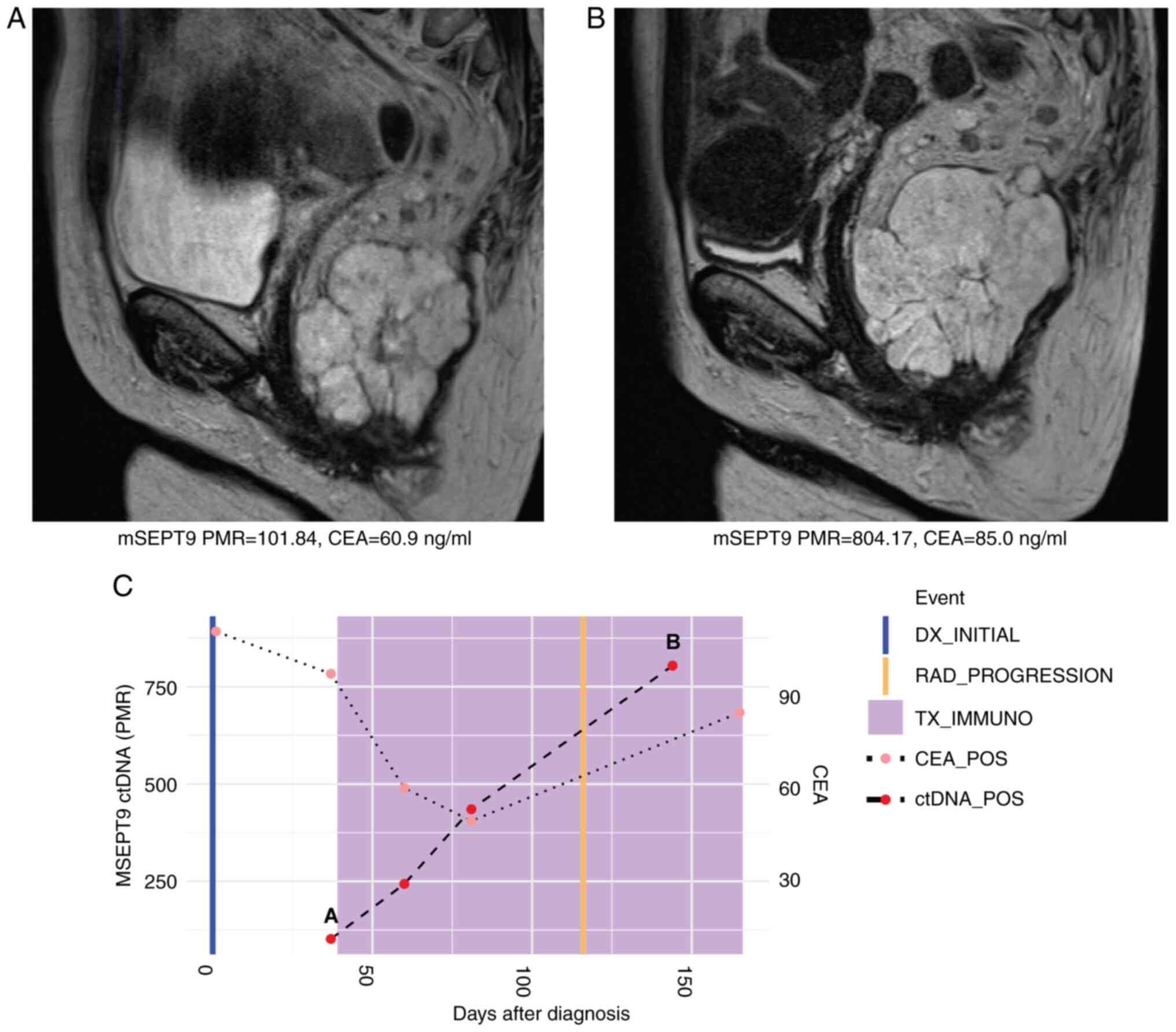
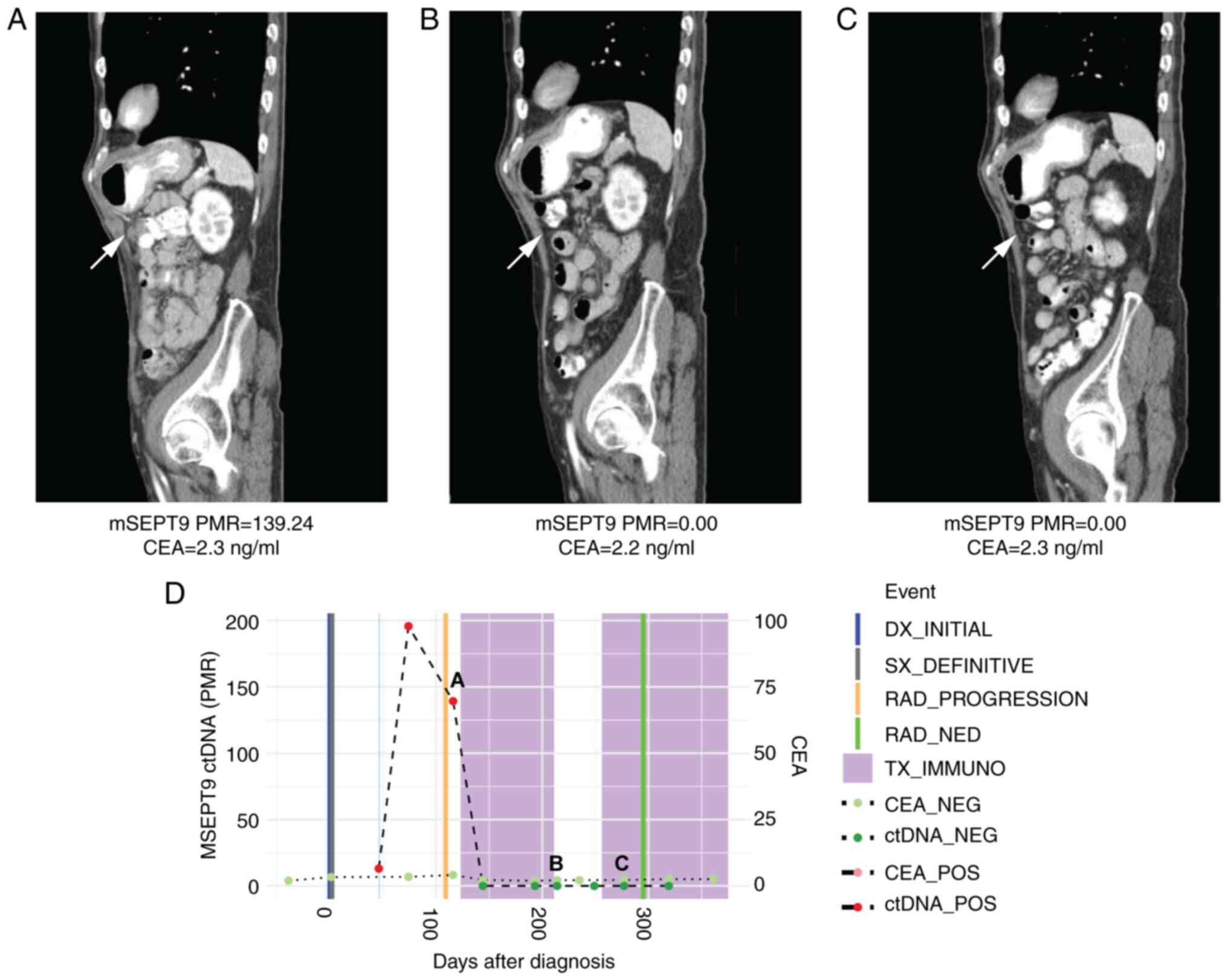
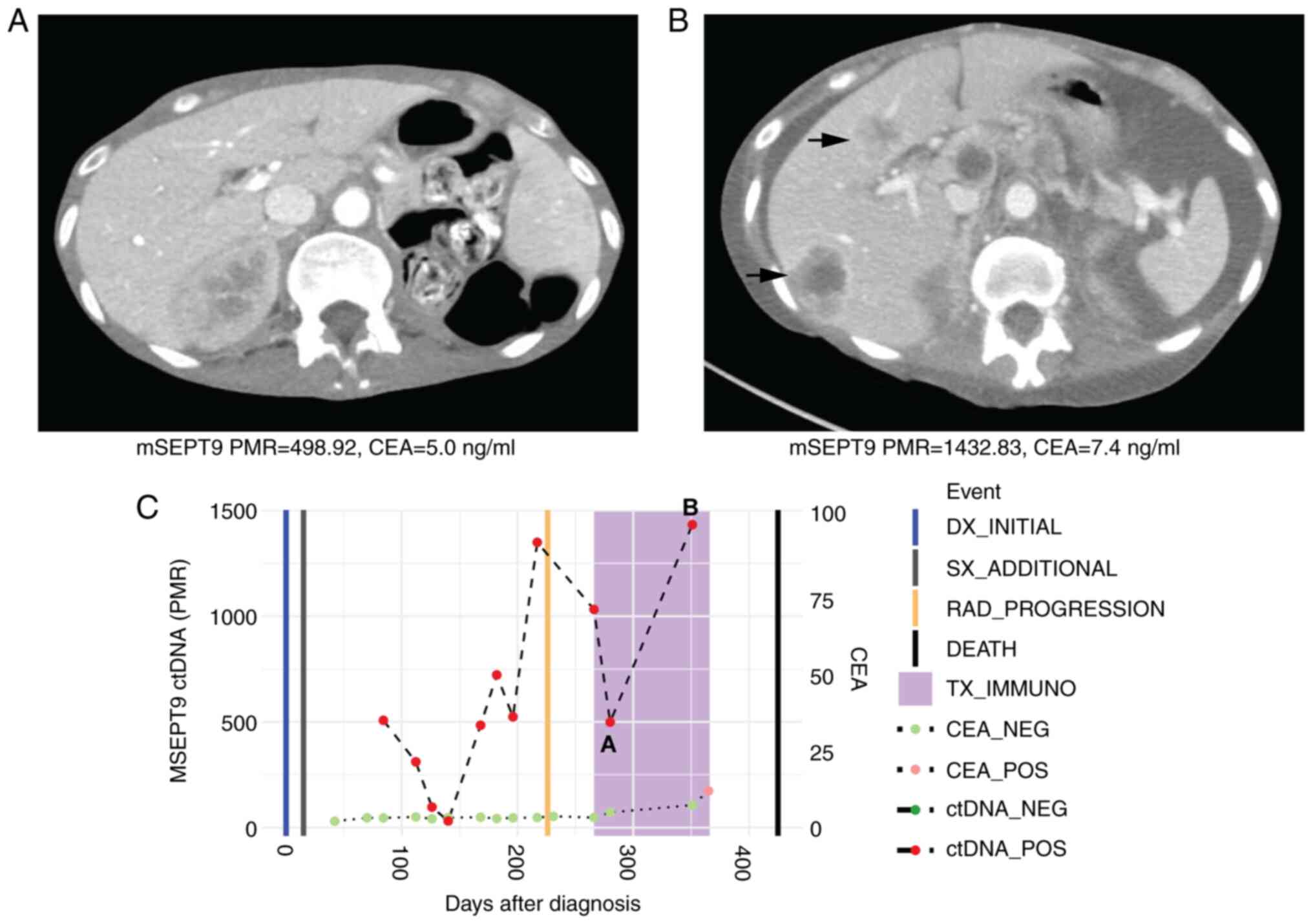
|
1
|
André T, Shiu KK, Kim TW, Jensen BV,
Jensen LH, Punt C, Smith D, Garcia-Carbonero R, Benavides M, Gibbs
P, et al: Pembrolizumab in microsatellite-instability-high advanced
colorectal cancer. N Engl J Med. 383:2207–2218. 2020.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Marcus L, Lemery SJ, Keegan P and Pazdur
R: FDA Approval summary: Pembrolizumab for the treatment of
microsatellite instability-high solid tumors. Clin Cancer Res.
25:3753–3758. 2019.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Overman MJ, McDermott R, Leach JL, Lonardi
S, Lenz HJ, Morse MA, Desai J, Hill A, Axelson M, Moss RA, et al:
Nivolumab in patients with metastatic DNA mismatch repair-deficient
or microsatellite instability-high colorectal cancer (CheckMate
142): An open-label, multicentre, phase 2 study. Lancet Oncol.
18:1182–1191. 2017.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Overman MJ, Lonardi S, Wong KYM, Lenz HJ,
Gelsomino F, Aglietta M, Morse MA, Van Cutsem E, McDermott R, Hill
A, et al: Durable clinical benefit with nivolumab plus ipilimumab
in DNA mismatch repair-deficient/microsatellite instability-high
metastatic colorectal cancer. J Clin Oncol. 36:773–779.
2018.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Andre T, Berton D, Curigliano G, Ellard S,
Pérez JMT, Arkenau HT, Abdeddaim C, Moreno V, Guo W, Im M and
Starling N: Safety and efficacy of anti-PD-1 antibody dostarlimab
in patients (pts) with mismatch repair-deficient (dMMR) solid
cancers: Results from GARNET study. J Clin Oncol. 39 (Suppl
3)(S9)2021.
|
|
6
|
Cho M, Akiba C, Lau C, Smith D, Telatar M,
Afkhami M, Sentovich S, Melstrom K and Fakih M: Impact of RAS and
BRAF mutations on carcinoembryonic antigen production and pattern
of colorectal metastases. World J Gastrointest Oncol. 8:128–135.
2016.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Dasari A, Morris VK, Allegra CJ, Atreya C,
Benson AB III, Boland P, Chung K, Copur MS, Corcoran RB, Deming DA,
et al: ctDNA applications and integration in colorectal cancer: An
NCI colon and rectal-anal task forces whitepaper. Nat Rev Clin
Oncol. 17:757–770. 2020.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Hitchins MP, Vogelaar IP, Brennan K,
Haraldsdottir S, Zhou N, Martin B, Alvarez R, Yuan X, Kim S, Guindi
M, et al: Methylated SEPTIN9 plasma test for colorectal cancer
detection may be applicable to Lynch syndrome. BMJ Open
Gastroenterol. 6(e000299)2019.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Cabel L, Riva F, Servois V, Livartowski A,
Daniel C, Rampanou A, Lantz O, Romano E, Milder M, Buecher B, et
al: Circulating tumor DNA changes for early monitoring of anti-PD1
immunotherapy: A proof-of-concept study. Ann Oncol. 28:1996–2001.
2017.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Stein A, Simnica D, Schultheiß C, Scholz
R, Tintelnot J, Gökkurt E, von Wenserski L, Willscher E, Paschold
L, Sauer M, et al: PD-L1 targeting and subclonal immune escape
mediated by PD-L1 mutations in metastatic colorectal cancer. J
Immunother Cancer. 9(e002844)2021.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Wang C, Chevalier D, Saluja J, Sandhu J,
Lau C and Fakih M: Regorafenib and nivolumab or pembrolizumab
combination and circulating tumor DNA response assessment in
refractory microsatellite stable colorectal cancer. Oncologist.
25:e1188–e1194. 2020.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Gong J, Hendifar A, Gangi A, Zaghiyan K,
Atkins K, Nasseri Y, Murrell Z, Figueiredo JC, Salvy S, Haile R and
Hitchins M: Clinical applications of minimal residual disease
assessments by tumor-informed and tumor-uninformed circulating
tumor DNA in colorectal cancer. Cancers (Basel).
13(4547)2021.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Tie J, Cohen JD, Lo SN, Wang Y, Li L,
Christie M, Lee M, Wong R, Kosmider S, Skinner I, et al: Prognostic
significance of postsurgery circulating tumor DNA in nonmetastatic
colorectal cancer: Individual patient pooled analysis of three
cohort studies. Int J Cancer. 148:1014–1026. 2021.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Reinert T, Henriksen TV, Christensen E,
Sharma S, Salari R, Sethi H, Knudsen M, Nordentoft I, Wu HT, Tin
AS, et al: Analysis of plasma cell-free DNA by ultradeep sequencing
in patients with stages I to III colorectal cancer. JAMA Oncol.
5:1124–1131. 2019.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Tie J, Cohen JD, Wang Y, Christie M,
Simons K, Lee M, Wong R, Kosmider S, Ananda S, McKendrick J, et al:
Circulating tumor DNA analyses as markers of recurrence risk and
benefit of adjuvant therapy for stage III colon cancer. JAMA Oncol.
5:1710–1717. 2019.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Tie J, Wang Y, Tomasetti C, Li L, Springer
S, Kinde I, Silliman N, Tacey M, Wong HL, Christie M, et al:
Circulating tumor DNA analysis detects minimal residual disease and
predicts recurrence in patients with stage II colon cancer. Sci
Transl Med. 8(346ra92)2016.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Tie J, Kinde I, Wang Y, Wong HL, Roebert
J, Christie M, Tacey M, Wong R, Singh M, Karapetis CS, et al:
Circulating tumor DNA as an early marker of therapeutic response in
patients with metastatic colorectal cancer. Ann Oncol.
26:1715–1722. 2015.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Tarazona N, Gimeno-Valiente F, Gambardella
V, Zuñiga S, Rentero-Garrido P, Huerta M, Roselló S,
Martinez-Ciarpaglini C, Carbonell-Asins JA, Carrasco F, et al:
Targeted next-generation sequencing of circulating-tumor DNA for
tracking minimal residual disease in localized colon cancer. Ann
Oncol. 30:1804–1812. 2019.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Kotaka M, Shirasu H, Watanabe J, Yamazaki
K, Hirata K, Akazawa N, Matsuhashi N, Yokota M, Ikeda M, Kato K, et
al: Association of circulating tumor DNA dynamics with clinical
outcomes in the adjuvant setting for patients with colorectal
cancer from an observational GALAXY study in CIRCULATE-Japan. J
Clin Oncol. 40 (4 Suppl)(S9)2022.
|
|
20
|
Henriksen TV, Reinert T, Christensen E,
Sethi H, Birkenkamp-Demtröder K, Gögenur M, Gögenur I, Zimmermann
BG, Dyrskjøt L and Andersen CL: IMPROVE Study Group. The effect of
surgical trauma on circulating free DNA levels in cancer
patients-implications for studies of circulating tumor DNA. Mol
Oncol. 14:1670–1679. 2020.PubMed/NCBI View Article : Google Scholar
|
|
21
|
Moser T, Waldispuehl-Geigl J, Belic J,
Weber S, Zhou Q, Hasenleithner SO, Graf R, Terzic JA, Posch F, Sill
H, et al: On-treatment measurements of circulating tumor DNA during
FOLFOX therapy in patients with colorectal cancer. NPJ Precis
Oncol. 4(30)2020.PubMed/NCBI View Article : Google Scholar
|
|
22
|
Demisse R, Damle N, Kim E, Gong J, Fakih
M, Eng C, Oesterich L, McKenny M, Ji J, Liu J, et al: Neoadjuvant
immunotherapy-based systemic treatment in MMR-deficient or MSI-high
rectal cancer: Case series. J Natl Compr Canc Netw. 18:798–804.
2020.PubMed/NCBI View Article : Google Scholar
|
|
23
|
Chalabi M, Fanchi LF, Dijkstra KK, Van den
Berg JG, Aalbers AG, Sikorska K, Lopez-Yurda M, Grootscholten C,
Beets GL, Snaebjornsson P, et al: Neoadjuvant immunotherapy leads
to pathological responses in MMR-proficient and MMR-deficient
early-stage colon cancers. Nat Med. 26:566–576. 2020.PubMed/NCBI View Article : Google Scholar
|
|
24
|
Ma Y, Wang Q, Dong Q, Zhan L and Zhang J:
How to differentiate pseudoprogression from true progression in
cancer patients treated with immunotherapy. Am J Cancer Res.
9:1546–1553. 2019.PubMed/NCBI
|