Introduction
High-grade gliomas (HGGs), encompassing WHO grade
III-IV malignant brain tumors, are characterized by aggressive
growth and poor prognosis, with a median survival of ~12-15 months
despite multimodal therapy (1).
The current standard of care involves maximal safe surgical
resection followed by radiotherapy and chemotherapy. The extent of
resection (EOR) is a critical prognostic factor, as more complete
tumor removal is consistently associated with longer overall
survival (OS) and progression-free survival (PFS) in patients with
HGGs (1,2).
Achieving gross total resection (GTR) under
conventional white-light microscopy remains challenging due to the
highly infiltrative nature of gliomas and the difficulty in
distinguishing tumor tissue from normal brain parenchyma at the
resection margins. To address this limitation, fluorescence-guided
surgery (FGS) has emerged as an important adjunct, enhancing
intraoperative visualization of tumor tissue and thereby improving
the likelihood of achieving maximal resection (3-5).
Two fluorescent agents are most widely used in HGG
surgery: 5-aminolevulinic acid (5-ALA) and sodium fluorescein (SF).
5-ALA is a prodrug metabolized within tumor cells to protoporphyrin
IX (PpIX), a fluorescent molecule that selectively accumulates in
malignant glioma cells and emits a characteristic red-violet
fluorescence under blue-light excitation (1,6,7).
Oral administration of 5-ALA (20 mg/kg) 3-4 h prior to surgery has
been approved for malignant glioma resection in adults, following a
pivotal randomized trial that demonstrated significantly higher GTR
rates with 5-ALA guidance compared with white-light resection
(1,8).
By contrast, SF is an intravenous fluorescent dye
that extravasates through areas of blood-brain barrier (BBB)
disruption. When activated by 460-500 nm blue light and observed
through a 540-690 nm yellow filter, fluorescein highlights tumor
tissue as green-yellow fluorescence within the surgical field
(1). Although SF has been used
sporadically in neurosurgery for decades, its adoption has
increased markedly in recent years due to the development of
dedicated microscope filters (for example, YELLOW-560 filter) and
its favorable safety profile at low doses (3-5 mg/kg) (1). Unlike 5-ALA, SF is not tumor-cell
specific, as it marks all areas of BBB breakdown. However, it
offers practical advantages, including bigger range of indications
and more practical administration (1).
Both 5-ALA and SF aim to maximize tumor removal
while minimizing neurological morbidity. Importantly, 5-ALA is
supported by Level I evidence and regulatory approval, whereas SF
remains an off-label but increasingly popular alternative with an
increasing body of supportive clinical data (1).
The present systematic review compares 5-ALA and SF
in HGG surgery, evaluating their relative effectiveness in
maximizing resection, impact on patient outcomes, safety profiles,
cost considerations, and practical advantages or limitations. In
addition, the present meta-analysis directly compares the clinical
outcomes associated with these two modalities, providing an updated
and comprehensive synthesis of current evidence to inform
neurosurgical practice.
Materials and methods
For the present meta-analysis and literature review,
a systematic literature search was performed to identify clinical
studies evaluating 5-ALA-guided resection and fluorescein-guided
surgery (FGS) in HGGs (9). The
search strategy and study selection process is detailed in the
PRISMA flow diagram and protocol. Two investigators (GF, a
neurosurgeon and GC, a gastric cancer surgeon) individually
extracted information from the enclosed articles using the
epidemiology principles of meta-analysis. The search strategy
retrieved 501 titles and abstracts [PubMed (https://pubmed.ncbi.nlm.nih.gov/) :305; Embase
(https://www.embase.com) :146; Cochrane Library
(https://www.cochranelibrary.com/) :24;
Scopus (https://www.scopus.com/) :26]. Eligible
studies included randomized controlled trials (RCTs), prospective
observational studies, retrospective cohorts, and relevant
meta-analyses. Both comparative and single-arm studies were
considered, provided they reported outcomes in adult patients with
HGGs (primarily glioblastomas and anaplastic gliomas) undergoing
resection with either 5-ALA or fluorescein guidance.
The search strategy used the following key words:
‘5-ALA AND HGGs’, ‘5-ALA AND Fluorescein Guided Resection’,
‘Fluorescein Guided Resection AND HGGs’, and ‘5-ALA AND Fluorescein
Guided Resection AND HGGs’. The final search was conducted on June
18, 2025. Only full-text articles in English were included.
Non-English language papers and studies focusing on low-grade
gliomas were excluded. In addition, articles with unclear/confusing
results or articles with less than 3 included cases, were excluded.
To minimize publication bias, the final aim was to collect a
homogeneous pool of manuscripts and thus studies were included only
if they compared outcomes between the two modalities or provided
sufficient outcome data for analysis. Articles focused solely on
technical aspects or novel imaging adjuncts were considered for
contextual discussion if they contained data on resection rates or
clinical outcomes.
A total of 41 articles met the eligibility criteria:
36 studies were included in the qualitative review (1-36),
and 5 comparative studies were included in the quantitative
synthesis (meta-analysis) (37-41)
(Fig. 1). Eligible studies
reported at least one of the following outcomes: EOR, GTR rate,
survival outcomes (PFS and OS), adverse events, or measures of
fluorescence effectiveness (for example, sensitivity/specificity of
tumor visualization, cost-effectiveness analyses).
Data were systematically extracted from each study,
including: First author, year of publication, total sample size,
patient demographics (age, sex), tumor type, median PFS, OS, pre-
and postoperative Karnofsky Performance Status (KPS ≥70 within 30
days of surgery), and postoperative neurological deficits.
Neurological deficits were defined as newly developed impairments
such as a ≥2-point reduction in the Glasgow Coma Scale (GCS),
hemiparesis, or aphasia (Table
SI).
Risk of bias was assessed using the Cochrane
Collaboration tool for randomized studies (42) and the Newcastle-Ottawa Scale (NOS)
for non-randomized studies (43)
(Table I). Statistical analyses
were conducted using Review Manager (RevMan 5.3, Nordic Cochrane
Centre, Cochrane Collaboration, Copenhagen, Denmark). Odds ratios
(ORs) with 95% confidence intervals (CIs) were calculated for
dichotomous outcomes. An OR <1 indicated a favorable outcome for
the 5-ALA group. Statistically significant threshold was set at
P<0.05. Heterogeneity among studies was assessed using the
Chi-square test and quantified with the I² statistic, with
P<0.05 or I² >50% indicating significant heterogeneity.
Random-effects models were applied in the presence of significant
heterogeneity, while fixed-effects models were used otherwise.
 | Table INewcastle-Ottawa Scale (NOS) quality
assessment of final article pool. |
Table I
Newcastle-Ottawa Scale (NOS) quality
assessment of final article pool.
| | | Newcastle-Ottawa
Scale | |
|---|
| First author/s,
year | Study design | Selection | Comparability | Exposure | Total scores | (Refs.) |
|---|
| Eljamel et
al, 2008 | Retrospective | 3 | 3 | 3 | 9 | (39) |
| Hansen et
al, 2019 | Retrospective | 3 | 3 | 3 | 9 | (37) |
| Baig Mirza et
al, 2021 | Retrospective | 3 | 2 | 2 | 7 | (40) |
| Baig Mirza et
al, 2022 | Retrospective | 3 | 3 | 3 | 9 | (41) |
| Zeppa et al,
2022 | Retrospective | 3 | 3 | 2 | 8 | (38) |
Results
The final set comprised 36 studies. These included
two phase III RCTs evaluating 5-ALA-guided resection versus
conventional microsurgery (1,11);
One multicenter randomized trial of FGS versus placebo (dark-light)
control (12); several prospective
single-arm studies of fluorescence guidance, including a Phase II
trial of fluorescein in glioblastoma (12) and multiple Phase I-II studies of
5-ALA in various clinical contexts (10,13-15);
a number of retrospective comparative series (16,28,29,37,39);
one case-control study (14); as
well as systematic reviews (11,15,17,18,26,27)
and meta-analyses (11) focused on
intraoperative imaging modalities.
The evidence base for 5-ALA is supported by
large-scale trials, notably the pivotal study by Stummer et
al (19), the recent French
RESECT trial (11), and
multicenter long-term experiences (11,12).
By contrast, the evidence for fluorescein is more limited,
primarily deriving from smaller RCTs (12), institutional series, and more
recent comparative analyses. Additionally, studies investigating
these agents in combination with other intraoperative technologies
(for example, MRI, ultrasound) or in other tumor types (such as
brain metastases and pediatric tumors) were included to provide
contextual insights into their broader clinical applicability
(1). The characteristics of all
included studies are summarized in Table SI.
5-ALA studies
The pivotal RCTs by Stummer et al (19) and Picart et al (11) established the efficacy of 5-ALA in
improving resection rates in newly diagnosed glioblastomas
(1). Numerous retrospective
multicenter cohorts and single-institution series further
demonstrated that 5-ALA guidance significantly increases the
likelihood of GTR without compromising patient safety (1). Several additional investigations
explored correlations between intraoperative 5-ALA fluorescence,
tumor pathology, and patient outcomes, reinforcing its role as a
reliable intraoperative tool (11).
Fluorescein (SF) studies
Early evidence of fluorescein's utility was provided
by small institutional series and a pilot double-blind trial
(12), which demonstrated improved
resection rates when using a dedicated yellow filter. A subsequent
multicenter Phase II trial [(‘FLUOGLIO’, Acerbi et al,
(12)] in 2014 reported complete
resection rates of 82-83% in glioblastoma surgery. More recently, a
large retrospective controlled study of 347 patients compared
fluorescein-guided surgery with white-light resection, showing
superior EOR and improved survival in the fluorescein group
(13). Consistent findings across
multiple single-center series, several synthesized in a 2021
meta-analysis, further confirmed that FGS is a safe and effective
technique for maximizing tumor resection in HGGs (14).
Meta-analysis-comparative studies
Direct head-to-head comparisons between 5-ALA and
fluorescein-guided surgery are limited. From an initial pool of 511
studies, 112 were considered potentially eligible (Fig. 1). After applying all exclusion
criteria, 41 studies remained. Further exclusion of studies
focusing exclusively on one modality reduced the final dataset to 5
comparative studies (37-41).
Across these 5 studies, the mean patient age was 57.9 years. The
pooled cohort consisted of 732 patients with HGGs, of whom 503
(68.7%) underwent 5-ALA-guided resection (Group A) and 229 (31.3%)
underwent fluorescein-guided resection (Group B) (Table II).
 | Table IIPooled effect estimates of 5-ALA vs.
fluorescein-guided resection in high-grade gliomas. |
Table II
Pooled effect estimates of 5-ALA vs.
fluorescein-guided resection in high-grade gliomas.
| | | Number of
events | Overall effect | Heterogeneity |
|---|
| Parameter | Included Trials,
n=5 | 5-ALA | FGS | Effect
estimate | 95% CI | P-value | I2
(%) | P-value |
|---|
| Postoperative | 5 | 70/503 | 73/229 | 0.88 | (0.17-4.57) | 0.88 | 90 | <0.00001 |
| Neurological
Deficit | | (9.5%) | (9.9%) | | | | | |
| Postoperative | 5 | Mean ± SD | 49.46 | (-2.21-101.13) | 0.06 | 100 | <0.00001 |
| KPS≥70 | | 92.8±95.0 | 43.4±25.3 | | | | | |
| OS | 5 | 26.2±15.4 | 17.5±5.8 | 4.05 | (1.33-6.77) | <0.05 | 98 | <0.00001 |
Postoperative neurological
deficit
All five studies provided data on neurological
outcomes (37-41).
Among patients treated with 5-ALA, 70 of 503 (9.5%) developed new
neurological deficits, compared with 73 of 229 (9.9%) in the
fluorescein group (Table II). The
pooled analysis demonstrated a statistically significant difference
favoring 5-ALA [(OR=0.88; 95% CI (0.17-4.57); P<0.05)]. However,
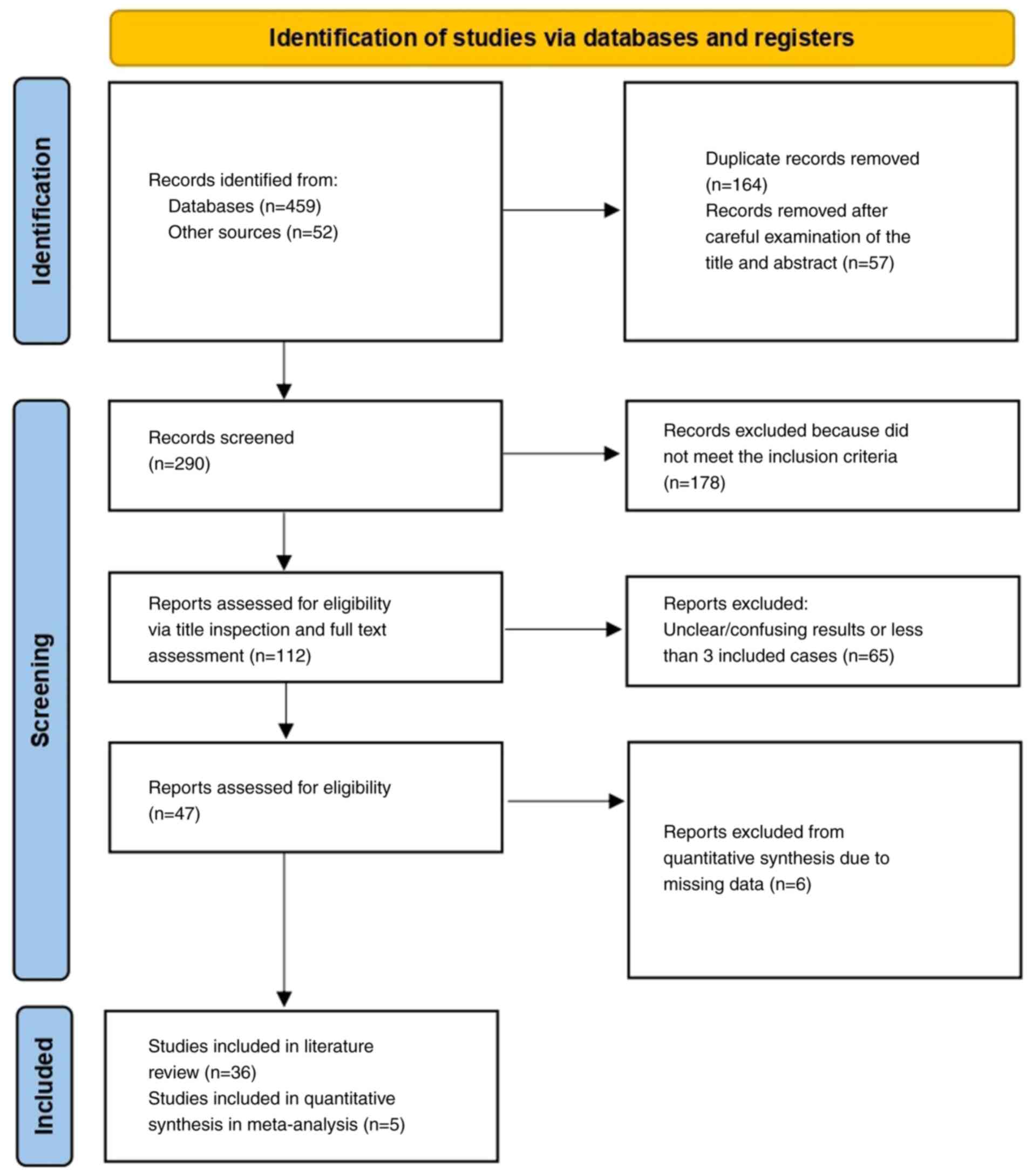
heterogeneity was substantial [(I²=90%; P=0.88) (Fig. 2A)], and funnel plot analysis
indicated publication bias (Fig.
2B).
![(A) Forest plot for postoperative
neurological deficit, with the results demonstrated a statistically
significant difference between groups [OR: 0.88, 95% CI:
(0.17,4.57), P<0.00001], but with heterogeneity (P=0.88,
I2=90%). (B) Funnel plots for Postoperative Neurological
deficit in the two groups with heterogeneity (P=0.88,
I2=90%). I2, percentage of total variation
across studies that is due to heterogeneity rather than chance; CI,
confidence interval; OR, odds ratio.](/article_images/mco/24/3/mco-24-03-02927-g01.jpg) | Figure 2(A) Forest plot for postoperative
neurological deficit, with the results demonstrated a statistically
significant difference between groups [OR: 0.88, 95% CI:
(0.17,4.57), P<0.00001], but with heterogeneity (P=0.88,
I2=90%). (B) Funnel plots for Postoperative Neurological
deficit in the two groups with heterogeneity (P=0.88,
I2=90%). I2, percentage of total variation
across studies that is due to heterogeneity rather than chance; CI,
confidence interval; OR, odds ratio. |
Postoperative KPS ≥70
Information on postoperative KPS ≥70 within 30 days
was reported in all five studies (37-41).
The pooled results suggested a statistically significant difference
between the two groups [OR=49.46; 95% CI (-2.21-101.13);
P<0.05], though heterogeneity was extremely high (I²=100%,
P=0.06) (Fig. 3A). Funnel plot
inspection again revealed evidence of publication bias (Fig. 3B) (Table II).
OS
Data on OS were available from all five studies
(37-41).
The pooled analysis showed a significant difference in favor of
5-ALA [OR=4.05; 95% CI (1.33-6.77); P<0.05]. Nonetheless,
heterogeneity was substantial (I²=98%; P<0.05) (Fig. 4A), and the funnel plot demonstrated
publication bias (Fig. 4B;
Table II).
Extent of resection and GTR rate
Both 5-ALA and fluorescein significantly increase
the extent of tumor resection in HGGs compared with conventional
white-light microsurgery. The pivotal 2006 ALA-Glioma Study by
Stummer et al (19)
demonstrated that 5-ALA guidance nearly doubled the rate of
complete resection of contrast-enhancing tumor (65% with 5-ALA vs.
36% with white-light, P<0.0001). This finding has been
consistently validated in subsequent studies. The multicenter
RESECT trial reported GTR rates of 79.1% with 5-ALA versus 47.8% in
controls (P=0.0002) (11).
For fluorescein-guided surgery, reported GTR rates
are comparably high. A prospective phase II trial in glioblastoma
achieved complete resection in 82.6% of patients (12), and a meta-analysis confirmed that
fluorescein significantly improves GTR rates compared with
non-fluorescent surgery (12,14,16).
In a large retrospective study, Schebesch et al (13) showed that fluorescein guidance led
to significantly higher GTR rates and greater mean extent of
resection than white-light microsurgery.
Direct comparisons between the two modalities
suggest broadly equivalent resection outcomes. In the largest
comparative study to date, Hansen et al (37) found no significant difference in
median extent of resection between 5-ALA-guided and
fluorescein-guided groups (96.9 vs. 97.4%; P=0.46) (10). Similarly, the proportion of
patients with only microscopic residual tumor (<0.175 cm³) did
not differ significantly (29.5 vs. 36.2%; P=0.39) (37). These results suggest that in
experienced hands, fluorescein can achieve resection completeness
comparable to 5-ALA. Importantly, both techniques outperform
conventional microsurgery: A network meta-analysis ranked
fluorescence guidance (5-ALA or fluorescein) and intraoperative MRI
as superior to white-light surgery for maximizing GTR in HGGs
(15). In summary, both agents
reliably enable high EOR, with GTR rates frequently exceeding 80%,
whereas white-light resection typically achieves ≤50% (1,12).
Survival outcomes
Maximal resection is a well-established determinant
of survival in glioblastoma, and the rationale for fluorescence
guidance is to increase GTR rates and thereby improve PFS and OS.
The original 5-ALA trial demonstrated a significant improvement in
6-month PFS (41 vs. 21% with white-light surgery) (1), though no significant OS difference
was observed, likely due to crossover and subsequent adjuvant
therapies. Similarly, the RESECT trial did not show a significant
OS benefit (24-month OS: ~30% with 5-ALA vs. 37% in controls,
P=0.89), nor a significant PFS difference (11). Notably, in RESECT, both arms
achieved higher-than-expected PFS rates (~69-70%), and multivariate
analysis identified GTR, not the choice of fluorophore, as the
independent predictor of survival (11).
For fluorescein, most evidence derives from
non-randomized studies. Schebesch et al (13) reported that fluorescein-guided
resection was associated with significantly longer median PFS and
OS compared with white-light surgery, with fluorescein use emerging
as an independent prognostic factor in multivariate analysis. A
network meta-analysis by Naik et al (15) similarly found that fluorescein
guidance was associated with improved OS versus no fluorescence,
whereas pooled data for 5-ALA did not show a statistically
significant OS advantage. However, this apparent difference must be
interpreted cautiously, as the fluorescein data come predominantly
from observational cohorts subject to selection bias.
Head-to-head comparisons again suggest broadly
similar outcomes. In the study by Hansen et al (37), median OS was longer in the
fluorescein group (19.7 months) compared with the 5-ALA group (14.8
months), though this difference did not reach statistical
significance (P=0.06). PFS was marginally higher with fluorescein
(9.2 vs. 8.7 months, P=0.03) (37), but the retrospective design limits
firm conclusions.
Taken together, current evidence does not support a
definitive survival advantage of one fluorophore over the other.
Both 5-ALA and fluorescein contribute to improved PFS and OS
indirectly, insofar as they enable higher rates of complete
resection. Importantly, neither technique has been associated with
increased neurological morbidity; instead, both are intended to
facilitate maximal safe resection, which remains the key
determinant of long-term outcomes (11).
Discussion
Both 5-ALA and fluorescein have demonstrated clear
benefits in facilitating maximal resection of HGGs. The present
meta-analysis indicated that 5-ALA was associated with improved
outcomes in terms of postoperative neurological deficits,
postoperative KPS ≥70, and OS compared with fluorescein. However,
significant heterogeneity and evidence of publication bias mitigate
the strength of these findings, precluding definitive conclusions
regarding superiority of one technique over the other.
5-ALA's principal advantage lies in its tumor
specificity. After metabolic conversion to protoporphyrin IX, 5-ALA
accumulates predominantly in high-grade tumor cells, producing a
highly specific fluorescent signal that correlates with tumor cell
density and offers near-perfect positive predictive value (1,19,34).
Its efficacy is supported by the highest level of evidence,
including a Phase III randomized trial demonstrating superior
resection rates versus white-light surgery (1), and its regulatory approval
facilitates clinical adoption. In addition, 5-ALA fluorescence can
extend beyond MRI-visible contrast enhancement and, in some
settings, enables adjunctive photodynamic therapy (22,23,33,35).
Limitations include preoperative dosing requirements, need for
blue-light optics, transient photosensitivity, restricted utility
in lower-grade gliomas and high cost (1,11,30-32).
Fluorescein, by contrast, is inexpensive, easy to
administer intravenously at the start of surgery, and widely
available (1,7,32).
It does not require extensive preoperative preparation, has minimal
side effects, and allows surgery under near-normal illumination,
which preserves visualization of vasculature and facilitates
uninterrupted workflow (1,31). Fluorescein is also versatile, as it
highlights any contrast-enhancing lesion, including metastases and
pediatric tumors (1). Its
disadvantages are mainly related to limited tumor specificity, as
it accumulates in any area of BBB disruption, raising the
possibility of false positives in cases of radiation necrosis or
inflammation (17,18,20).
It may also fail to detect infiltrative tumor beyond enhancing
margins, where 5-ALA may still fluoresce (20). Practical considerations include the
need for a dedicated yellow filter and occasional difficulties with
color contrast in bloody fields (20,31).
An important aspect of comparing 5-ALA and
fluorescein is the safety profile of each agent and its impact on
surgical morbidity. Both agents have been shown to be very safe in
patients with HGG, with low rates of drug-specific adverse events
and no increase in permanent neurological deficit rates
attributable to their use (11,19).
5-ALA safety
Oral 5-ALA is generally well tolerated. In the 5-ALA
arm of the RESECT trial (88 patients), adverse events related to
5-ALA were rare: Only 4.6% of patients experienced transient
photosensitivity reactions and 1.1% had transient elevated liver
enzymes (‘hepatic cytolysis’) (19). No significant differences in
postoperative Karnofsky performance or neurological deficits were
observed between 5-ALA and control groups indicating that
fluorescence guidance did not lead to higher neurological risk
(11,19). However, 5-ALA does have known side
effects and practical precautions. Because it can cause cutaneous
photosensitivity for ~24 h, treated patients must avoid direct
bright light exposure post-operatively (11). Some patients report mild nausea or
hypotension related to 5-ALA ingestion, and very rarely allergic
reactions have been noted (though none was severe in reported
trials). Overall, the incidence of serious adverse reactions to
5-ALA is low, and no long-term toxicities have been identified with
its intermittent use in surgery.
Fluorescein safety
Modern fluorescein-guided neurosurgery typically
uses a low-dose regimen (3-5 mg/kg IV) at induction of anesthesia,
which has an excellent safety profile. Across numerous series
totaling hundreds of patients, there have been no significant
adverse reactions attributed to fluorescein at this dosage
(12). For instance, in the
FLUOGLIO trial (46 patients), no fluorescein-related toxic events
were recorded (12). Similarly,
Martinez et al (44)
reported no complications in 47 patients; importantly, they did not
observe any cases of anaphylaxis, hypotension, or end-organ
toxicity (14). Older literature
on fluorescein (using higher doses of ~20 mg/kg without filtered
microscopy) noted rare side effects such as transient skin
discoloration, nausea, or very infrequently anaphylactic shock, but
such issues have not been reported with the current low-dose,
filtered technique. As with 5-ALA, fluorescein guidance has not
been associated with increased neurological deficits, in fact, one
study noted that the fluorescein-guided group had a slightly
shorter operative time and trend toward fewer complications than
controls (16). A practical point
is that fluorescein will turn the patient's urine bright yellow for
~1 day as it is excreted, but this is harmless.
In terms of surgical safety, both methods are
intended to improve the surgeon's ability to resect tumor while
avoiding eloquent areas, and neither has shown a detriment to
functional outcomes. The field of fluorescence-guided neurosurgery
is advancing rapidly, with several emerging technologies poised to
expand its utility. One promising area is next-generation
fluorophores, particularly near-infrared (NIR) agents operating at
700-1,000 nm, which allow deeper tissue penetration and reduced
background autofluorescence, enabling visualization of subcortical
tumor (23-25).
Another frontier is tumor-targeted molecular probes. Unlike 5-ALA
and fluorescein, which mark metabolic activity or BBB disruption,
newer probes aim at specific tumor markers. For example, an
EGFR-targeted probe (cetuximab-IRDye800) has been tested in
EGFR-positive glioblastomas (21,36),
while antibodies and peptides against IL13Rα2 and integrins are
under early investigation. Dual-modality agents, labeled for both
PET and NIR fluorescence, have also been developed, such as
GRPR-targeting probes that guide resection of otherwise
non-enhancing gliomas (24).
Combined modalities may further enhance resection
safety. Integration with intraoperative MRI (26) or quantitative fluorescence analysis
has shown promise; for instance, Valdés et al (33) demonstrated that spectroscopic
measurement of protoporphyrin can identify residual tumor below
visual detection thresholds (27,28).
Theranostic approaches are also being explored, including
nanoparticles capable of dual imaging and therapy, with some
NIR-responsive platforms combining tumor visualization with drug
delivery or photothermal ablation (39).
There were also systematic reviews and network
meta-analyses using three-way comparison of three modalities
(intraoperative MRI with 5 ALA or FGS) leading to greater rates of
GTR in HGGs (6,15). However, the present meta-analysis
focusing on correct comparative studies may constitute valuable
research in this field, as more clearly explicit the real
usefulness of these two modalities (5-ALA and FGS) separately.
The present systematic review has several
limitations. First, direct head-to-head trials comparing 5-ALA and
fluorescein remain scarce. Most included studies differed in
patient populations, tumor grades and outcome definitions,
resulting in significant heterogeneity and limiting quantitative
synthesis. Numerous fluorescein studies were small, single-arm
series without control groups, raising the risk of publication bias
and overestimation of benefit (17,18,20).
Blinding is inherently impossible in this field, introducing
observer bias, and surgeon behavior may be influenced by the
presence of fluorescence itself.
Second, available data were limited, particularly
regarding long-term survival with fluorescein, which is largely
inferred from indirect evidence. Most studies emphasized short-term
endpoints such as extent of resection or early postoperative
outcomes, rather than durable oncological control. Unpublished
data, conference abstracts and non-English studies were also
excluded, which may omit emerging trial results.
Third, methodological quality varied. While 5-ALA
has strong Phase III trial evidence (1), most fluorescein studies remain
observational with intermediate to high risk of bias. Although it
was attempted to focus on objective MRI-confirmed resection rates,
the lack of uniform methodology across studies restricts
comparability. Also, according to the fifth edition of the WHO
Classification of Tumors of the Central Nervous System, published
in 2021, new tumor types and subtypes are introduced, some based on
novel diagnostic technologies such as DNA methylome profiling, and
these may be helpful for the management of HGMs (45). In addition, gliomas exhibit a broad
range of molecular alterations, including gene mutations,
amplifications and deletions, that influence tumor development and
patient prognosis. Key biomarkers include IDH1/2 mutations, as well
as alterations in TP53, ATRX, PTEN and EGFR. Additional important
features include 1p/19q codeletion, TERT promoter mutations and
CDKN2A/B deletions. Thus, further analysis and data are needed for
more accurate results regarding HGMs prognosis (45). Last but not at least, another
limitation was that both 5-ALA-guided resection and other FGS
methods have limited sensitivity and specificity, including the
false-positive and false-negative rates (46).
Despite these limitations, current evidence
consistently supports the role of both 5-ALA and fluorescein in
enhancing the extent of glioma resection. Each technique has
distinct advantages and trade-offs, and both can be applied safely
in routine neurosurgical practice. Ongoing comparative trials,
standardized outcome reporting, and cost-effectiveness analyses
will be crucial to clarify their relative roles and optimize
fluorescence-guided strategies in malignant glioma surgery.
In conclusion, in HGG surgery, fluorescence guidance
has become an indispensable strategy to maximize tumor resection.
5-ALA and fluorescein each have well-demonstrated benefits in this
regard. 5-ALA offers tumor-selective visualization and a robust
evidence base, whereas fluorescein provides versatility and
cost-effectiveness with comparable resection outcomes.
Neurosurgeons should be familiar with both modalities and their
respective pros and cons. In practice, usage may be tailored to the
clinical scenario and resource setting. Ongoing innovations promise
to further refine fluorescence-guided resection, but at present,
5-ALA and fluorescein remain the two cornerstone options that have
measurably improved surgical outcomes for patients with malignant
gliomas. The ultimate goal is to achieve the maximal safe resection
for every patient, a goal that, thanks to these fluorescence
techniques, is more attainable than ever before.
Supplementary Material
Design and baseline characteristics of
the trials included in the present meta- analysis.
Acknowledgements
Not applicable.
Funding
Funding: No funding was received.
Availability of data and materials
Not applicable.
Authors' contributions
EK and GF conceptualized the study. EK, NF, GF, NT,
IK, VEG and DAS made a substantial contribution to data
interpretation and analysis and wrote and prepared the draft of the
manuscript. GF and NF analyzed the data and provided critical
revisions. All authors contributed to manuscript revision, read and
approved the final version of the manuscript.
Ethics approval and consent to
participate
Not applicable.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
Use of artificial intelligence tools
During the preparation of this work, artificial
intelligence tools were used to improve the readability and
language of the manuscript or to generate images, and subsequently,
the authors revised and edited the content produced by the
artificial intelligence tools as necessary, taking full
responsibility for the ultimate content of the present
manuscript.
References
|
1
|
Ahrens LC, Krabbenhøft MG, Hansen RW,
Mikic N, Pedersen CB, Poulsen FR and Korshoej AR: Effect of
5-aminolevulinic acid and sodium fluorescein on the extent of
resection in high-grade gliomas and brain metastasis. Cancers
(Basel). 14(617)2022.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Gandhi S, Meybodi AT, Belykh E, Cavallo C,
Zhao X, Syed MP, Moreira LB, Lawton MT, Nakaji P and Preul MC:
Survival outcomes among patients with high-grade glioma treated
with 5-aminolevulinic acid-guided surgery: A systematic review and
meta-analysis. Front Oncol. 9(620)2019.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Cordova JS, Gurbani SS, Holder CA, Olson
JJ, Schreibmann E, Shi R, Guo Y, Shu HK, Shim H and Hadjipanayis
CG: Semi-automated volumetric and morphological assessment of
glioblastoma resection with fluorescence-guided surgery. Mol
Imaging Biol. 18:454–462. 2016.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Richter JCO, Haj-Hosseini N, Hallbeck M
and Wårdell K: Combination of hand-held probe and microscopy for
fluorescence guided surgery in the brain tumor marginal zone.
Photodiagnosis Photodyn Ther. 18:185–192. 2017.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Patil CG, Walker DG, Miller DM, Butte P,
Morrison B, Kittle DS, Hansen SJ, Nufer KL, Byrnes-Blake KA, Yamada
M, et al: Phase 1 safety, pharmacokinetics, and fluorescence
imaging study of tozuleristide (BLZ-100) in adults with newly
diagnosed or recurrent gliomas. Neurosurgery. 85:E641–E649.
2019.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Valerio JE, de Jesús Aguirre Vera G,
Zumaeta J, Rea NS, Gomez MP, Mantilla-Farfan P, Valente L and
Alvarez-Pinzon AM: Comparative analysis of 5-ALA and fluorescent
techniques in high-grade glioma treatment. Biomedicines.
13(1161)2025.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Stummer W, Tonn JC, Mehdorn HM, Nestler U,
Franz K, Goetz C, Bink A and Pichlmeier U: ALA-Glioma Study Group.
Counterbalancing risks and gains from extended resections in
malignant glioma surgery: A supplemental analysis from the
randomized 5-aminolevulinic acid glioma resection study. Clinical
article. J Neurosurg. 114:613–623. 2011.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Lau D, Hervey-Jumper SL, Chang S, Molinaro
AM, McDermott MW, Phillips JJ and Berger MS: A prospective phase II
clinical trial of 5-aminolevulinic acid to assess the correlation
of intraoperative fluorescence intensity and degree of histologic
cellularity during resection of high-grade gliomas. J Neurosurg.
124:1300–1309. 2016.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Page MJ, McKenzie JE, Bossuyt PM, Boutron
I, Hoffmann TC, Mulrow CD, Shamseer L, Tetzlaff JM, Akl EA, Brennan
SE, et al: The PRISMA 2020 statement: An updated guideline for
reporting systematic reviews. BMJ. 372(n71)2021.PubMed/NCBI View
Article : Google Scholar
|
|
10
|
Stummer W, Nestler U, Stockhammer F, Krex
D, Kern BC, Mehdorn HM, Vince GH and Pichlmeier U: Favorable
outcome in the elderly cohort treated by concomitant temozolomide
radiochemotherapy in a multicentric phase II safety study of 5-ALA.
J Neurooncol. 103:361–370. 2011.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Picart T, Pallud J, Berthiller J, Dumot C,
Berhouma M, Ducray F, Armoiry X, Margier J, Guerre P, Varlet P, et
al: Use of 5-ALA fluorescence-guided surgery versus white-light
conventional microsurgery for the resection of newly diagnosed
glioblastomas (RESECT study): a French multicenter randomized phase
III study. J Neurosurg. 140:987–1000. 2023.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Acerbi F, Broggi M, Schebesch KM, Höhne J,
Cavallo C, De Laurentis C, Eoli M, Anghileri E, Servida M, Boffano
C, et al: Fluorescein-guided surgery for resection of high-grade
gliomas: A multicentric prospective phase II study (FLUOGLIO). Clin
Cancer Res. 24:52–61. 2018.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Schebesch KM, Höhne J, Rosengarth K, Noeva
E, Schmidt NO and Proescholdt M: Fluorescein-guided resection of
newly diagnosed high-grade glioma: Impact on extent of resection
and outcome. Brain Spine. 2(101690)2022.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Francaviglia N, Iacopino DG, Costantino G,
Villa A, Impallaria P, Meli F and Maugeri R: Fluorescein for
resection of high-grade gliomas: A safety study control in a single
center and review of the literature. Surg Neurol Int.
8(145)2017.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Naik A, Smith EJ, Barreau A, Nyaeme M,
Cramer SW, Najafali D, Krist DT, Arnold PM and Hassaneen W:
Comparison of fluorescein sodium, 5-ALA, and intraoperative MRI for
resection of high-grade gliomas: A systematic review and network
meta-analysis. J Clin Neurosci. 98:240–247. 2022.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Xi C, Jinli S, Jianyao M, Yan C, Huijuan
L, Zhongjie S, Zhangyu L, Liwei Z, Yukui L, Sifang C and Guowei T:
Fluorescein-guided surgery for high-grade glioma resection: A
five-year-long retrospective study at our institute. Front Oncol.
13(1191470)2023.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Schupper AJ, Rao M, Mohammadi N, Baron R,
Lee JYK, Acerbi F and Hadjipanayis CG: Fluorescence-guided surgery:
A review on timing and use in brain tumor surgery. Front Neurol.
12(682151)2021.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Mansour HM, Shah S, Aguilar TM,
Abdul-Muqsith M, Gonzales-Portillo GS and Mehta AI: Enhancing
glioblastoma resection with NIR fluorescence imaging: A systematic
review. Cancers (Basel). 16(3984)2024.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Stummer W, Stepp H, Wiestler OD and
Pichlmeier U: Randomized, prospective double-blinded study
comparing 3 different doses of 5-aminolevulinic acid for
fluorescence-guided resections of malignant gliomas. Neurosurgery.
81:230–239. 2017.PubMed/NCBI View Article : Google Scholar
|
|
20
|
Della Puppa A, Ciccarino P, Lombardi G,
Rolma G, Cecchin D and Rossetto M: 5-Aminolevulinic acid
fluorescence in high grade glioma surgery: Surgical outcome,
intraoperative findings, and fluorescence patterns. Biomed Res Int.
2014(232561)2014.PubMed/NCBI View Article : Google Scholar
|
|
21
|
Cramer SW and Chen CC: Photodynamic
therapy for the treatment of glioblastoma. Front Surg.
6(81)2020.PubMed/NCBI View Article : Google Scholar
|
|
22
|
Molina ES and Brokinkel B: Letter to the
editor. Sodium fluorescein versus 5-aminolevulinic acid to
visualize high-grade gliomas. J Neurosurg. 133:1627–1630.
2020.PubMed/NCBI View Article : Google Scholar
|
|
23
|
Shi X, Zhang Z, Zhang Z, Cao C, Cheng Z,
Hu Z, Tian J and Ji N: Near-infrared window II fluorescence
image-guided surgery of high-grade gliomas prolongs the
progression-free survival of patients. IEEE Trans Biomed Eng.
69:1889–1900. 2022.PubMed/NCBI View Article : Google Scholar
|
|
24
|
Cesca BA, Martin KP, Caverzan MD, Oliveda
PM and Ibarra LE: State-of-the-art photodynamic therapy for
malignant gliomas: Innovations in photosensitizers and combined
therapeutic approaches. Explor Target Antitumor Ther.
6(1002303)2025.PubMed/NCBI View Article : Google Scholar
|
|
25
|
Tao T, Li G, Zhou K, Pan Q, Wu D, Lai L,
Gao M, Li S, Chen L, Han RPS, et al: Discovery of fatty acid
translocase CD36-targeting near-infrared fluorescent probe enables
visualization and imaging-guided surgery for glioma. Anal Chem.
97:3687–3695. 2025.PubMed/NCBI View Article : Google Scholar
|
|
26
|
Panciani PP, Fontanella M, Schatlo B,
Garbossa D, Agnoletti A, Ducati A and Lanotte M: Fluorescence and
image guided resection in high grade glioma. Clin Neurol Neurosurg.
114:37–41. 2012.PubMed/NCBI View Article : Google Scholar
|
|
27
|
Henderson F Jr, Belykh E, RaOS AD and
Schwartz TH: Qualitative head-to-head comparison of headlamp and
microscope for visualizing 5-ALA fluorescence during resection of
glioblastoma. Neurosurg Focus Video. 6(V7)2022.PubMed/NCBI View Article : Google Scholar
|
|
28
|
Chen L, Zhang J, Chi C, Che W, Dong G,
Wang J, Du Y, Wang R, Zhu Z, Tian J, et al: Lower-grade gliomas
surgery guided by GRPR-targeting PET/NIR dual-modality image probe:
A prospective and single-arm clinical trial. Theranostics.
14:819–829. 2024.PubMed/NCBI View Article : Google Scholar
|
|
29
|
Inogés S, Tejada S, de Cerio AL,
Pérez-Larraya JG, Espinós J, Idoate MA, Domínguez PD, de Eulate RG,
Aristu J, Bendandi M, et al: A phase II trial of autologous
dendritic cell vaccination and radiochemotherapy following
fluorescence-guided surgery in newly diagnosed glioblastoma
patients. J Transl Med. 15(104)2017.PubMed/NCBI View Article : Google Scholar
|
|
30
|
Chung IW and Eljamel S: Risk factors for
developing oral 5-aminolevulinic acid-induced side effects in
patients undergoing fluorescence guided resection. Photodiagnosis
Photodyn Ther. 10:362–367. 2013.PubMed/NCBI View Article : Google Scholar
|
|
31
|
Feigl GC, Ritz R, Moraes M, Klein J,
Ramina K, Gharabaghi A, Krischek B, Danz S, Bornemann A, Liebsch M
and Tatagiba MS: Resection of malignant brain tumors in eloquent
cortical areas: a new multimodal approach combining
5-aminolevulinic acid and intraoperative monitoring. J Neurosurg.
113:352–357. 2010.PubMed/NCBI View Article : Google Scholar
|
|
32
|
Roberts DW, Olson JD, Evans LT, Kolste KK,
Kanick SC, Fan X, Bravo JJ, Wilson BC, Leblond F, Marois M and
Paulsen KD: Red-light excitation of protoporphyrin IX fluorescence
for subsurface tumor detection. J Neurosurg. 128:1690–1697.
2018.PubMed/NCBI View Article : Google Scholar
|
|
33
|
Valdés PA, Leblond F, Kim A, Harris BT,
Wilson BC, Fan X, Tosteson TD, Hartov A, Ji S, Erkmen K, et al:
Quantitative fluorescence in intracranial tumor: Implications for
ALA-induced PpIX as an intraoperative biomarker. J Neurosurg.
115:11–17. 2011.PubMed/NCBI View Article : Google Scholar
|
|
34
|
Nabavi A, Thurm H, Zountsas B, Pietsch T,
Lanfermann H, Pichlmeier U and Mehdorn M: 5-ALA Recurrent Glioma
Study Group. Five-aminolevulinic acid for fluorescence-guided
resection of recurrent malignant gliomas: A phase II study.
Neurosurgery. 65:1070–1076. 2009.PubMed/NCBI View Article : Google Scholar
|
|
35
|
Zimmermann A, Ritsch-Marte M and Kostron
H: mTHPC-mediated photodynamic diagnosis of malignant brain tumors.
Photochem Photobiol. 74:611–616. 2001.PubMed/NCBI View Article : Google Scholar
|
|
36
|
Li C, Sullivan PZ, Cho S, Nasrallah MP,
Buch L, Chen HC and Lee JYK: Intraoperative molecular imaging with
second window indocyanine green facilitates confirmation of
contrast-enhancing tissue during intracranial stereotactic needle
biopsy: A case series. World Neurosurg. 126:e1211–e1218.
2019.PubMed/NCBI View Article : Google Scholar
|
|
37
|
Hansen RW, Pedersen CB, Halle B, Korshoej
AR, Schulz MK, Kristensen BW and Poulsen FR: Comparison of
5-aminolevulinic acid and sodium fluorescein for intraoperative
tumor visualization in patients with high-grade gliomas: A
single-center retrospective study. J Neurosurg. 133:1324–1331.
2019.PubMed/NCBI View Article : Google Scholar
|
|
38
|
Zeppa P, De Marco R, Monticelli M, Massara
A, Bianconi A, Di Perna G, Crasto SG, Cofano F, Melcarne A, Lanotte
MM and Garbossa D: Fluorescence-guided surgery in glioblastoma:
5-ALA, SF or both? Differences between fluorescent dyes in 99
consecutive cases. Brain Sci. 12(555)2022.PubMed/NCBI View Article : Google Scholar
|
|
39
|
Eljamel MS, Goodman C and OSeley H: ALA
and Photofrin fluorescence-guided resection and repetitive PDT in
glioblastoma multiforme: A single centre Phase III randomised
controlled trial. Lasers Med Sci. 23:361–367. 2008.PubMed/NCBI View Article : Google Scholar
|
|
40
|
Mirza AB, Christodoulides I, Lavrador JP,
Giamouriadis A, Vastani A, Boardman T, Ahmed R, Norman I, Murphy C,
Devi S, et al: 5-Aminolevulinic acid-guided resection improves the
overall survival of patients with glioblastoma-a comparative cohort
study of 343 patients. Neurooncol Adv. 3(vdab047)2021.PubMed/NCBI View Article : Google Scholar
|
|
41
|
Mirza AB, Lavrador JP, Christodoulides I,
Boardman TM, Vastani A, Al Banna Q, Ahmed R, Norman ICF, Murphy C,
Devi S, et al: 5-Aminolevulinic acid-guided resection in grade III
Tumors-A comparative cohort study. Oper Neurosurg (Hagerstown).
22:215–223. 2022.PubMed/NCBI View Article : Google Scholar
|
|
42
|
Sterne JAC, Savović J, Page MJ, Elbers RG,
Blencowe NS, Boutron I, Cates CJ, Cheng HY, Corbett MS, Eldridge
SM, et al: RoB 2: A revised tool for assessing risk of bias in
randomised trials. BMJ. 366(l4898)2019.PubMed/NCBI View Article : Google Scholar
|
|
43
|
Stang A: Critical evaluation of the
Newcastle-Ottawa scale for the assessment of the quality of
nonrandomized studies in meta-analyses. Eur J Epidemiol.
25:603–605. 2010.PubMed/NCBI View Article : Google Scholar
|
|
44
|
Martinez-Castellanos MA, Velez-Montoya R,
Price K, Henaine-Berra A, García-Aguirre G, Morales-Canton V and
Cernichiaro-Espinosa LA: Vascular changes on fluorescein
angiography of premature infants with low risk of retinopathy of
prematurity after high oxygen exposure. Int J Retina Vitreous.
3(2)2017.PubMed/NCBI View Article : Google Scholar
|
|
45
|
Louis DN, Perry A, Wesseling P, Brat DJ,
Cree IA, Figarella-Branger D, Hawkins C, Ng HK, Pfister SM,
Reifenberger G, et al: The 2021 WHO classification of tumors of the
central nervous system: A summary. Neuro Oncol. 23:1231–1251.
2021.PubMed/NCBI View Article : Google Scholar
|
|
46
|
Gautheron A, Bernstock JD, Picart T,
Guyotat J, Valdés PA and Montcel B: 5-ALA induced PpIX fluorescence
spectroscopy in neurosurgery: A review. Front Neurosci.
18(1310282)2024.PubMed/NCBI View Article : Google Scholar
|















![(A) Forest plot for postoperative
neurological deficit, with the results demonstrated a statistically
significant difference between groups [OR: 0.88, 95% CI:
(0.17,4.57), P<0.00001], but with heterogeneity (P=0.88,
I2=90%). (B) Funnel plots for Postoperative Neurological
deficit in the two groups with heterogeneity (P=0.88,
I2=90%). I2, percentage of total variation
across studies that is due to heterogeneity rather than chance; CI,
confidence interval; OR, odds ratio.](/article_images/mco/24/3/mco-24-03-02927-g01.jpg)
![(A) Forest plot for Postoperative KPS
≥70, with the results demonstrated a statistically significant
difference between groups [OR=49.46; 95% CI: (-2.21, 101.13);
P<0.00001] but with heterogeneity (P=0.06 and
I2=100%). (B) Funnel plots for Postoperative KPS≥70 in
the two groups with high heterogeneity (P=0.06 and
I2=100%). I2, percentage of total variation
across studies that is due to heterogeneity rather than chance; CI,
confidence interval; KPS, Karnofsky Performance Scale.](/article_images/mco/24/3/mco-24-03-02927-g02.jpg)
![(A) Forest plot for OS, with the
results demonstrated a statistically significant difference between
groups [OR=4.05; 95% CI: (1.33, 6.77); P<0.00001] but a high
heterogeneity was found (P<0.05 and I2=98%). (B)
Funnel plots for OS in the two groups with high heterogeneity
(P<0.05 and I2=98%). I2, percentage of
total variation across studies that is due to heterogeneity rather
than chance; CI, confidence interval; OS, overall survival.](/article_images/mco/24/3/mco-24-03-02927-g03.jpg)