Introduction
Breast cancer (BC) is the most common malignancy and
the second cause of cancer-related deaths among women worldwide
(1). By 2040, over 3 million of BC
new cases and 1 million of related deaths are annually predicted as
a result of multiple and interconnected factors, including: global
population growth, unhealthy lifestyles, genetic predispositions
and environmental exposures, among others (2-4).
Taking into account tumor load and molecular markers, the
therapeutic approach involves a multidisciplinary setting where
standard management is lined with targeted and immune therapies
(5). Despite the constant
therapeutic advances, the 5-year survival rate for advanced BC
patients is not greater than 30%, making prevention and early
screening crucial for successful recovery (6). Mammography, ultrasonography and
magnetic resonance imaging are considered the primary tools for
detecting the presence of BC. Nevertheless, circulating biomarkers
can offer an attractive alternative for BC screening because of
their low cost and noninvasive nature (7). Several biomarkers have been explored
over the years, and some of them have been approved in BC, such as
carcinoembryonic antigen (CEA), carbohydrate antigen 15-3 (CA15-3),
and carbohydrate antigen 27-29 (CA27-29) (8). Regrettably, these markers have shown
low diagnostic specificity and sensitivity, while, conversely, they
provide useful insights for monitoring disease recurrence or
progression, following response to treatment, and even determining
targetable mutations to direct therapy (9-11).
Hence, there is still a need to discover non-invasive,
straightforward and low-risk biomarkers for the screening,
diagnosis and management of BC.
Monocyte distribution width (MDW) is a novel
hematological parameter provided along with the complete blood
count (CBC) by the last generation of DxH hematology analyzers
(Beckman Coulter, Inc.) (12). As
a direct measure of monocyte volume, MDW reflects the degree of
similarity or dissimilarity (anisocytosis) within the cell
population. Changes in MDW are emerging as an early marker of
innate immune activation in different pathological conditions,
including sepsis, viral infections, and other inflammatory-related
diseases (13-16).
The clinical usage of MDW is currently restricted to sepsis, where
its values correlate with the severity of this life-threatening
condition (17,18).
A recent study reported alterations in MDW across
different stages of chronic liver disease, from hepatitis B to
hepatocellular carcinoma, passing through cirrhosis (19). Dynamic upward changes in MDW levels
were detected in HBeAg-positive patients, while a positive
association was observed with the pathological progression of
cirrhosis. Notably, MDW was positively correlated with occurrence
and development of hepatocellular carcinoma, providing the first
evidence of its role in malignant disorders.
In the light of the aforementioned state-of-the-art,
herein we assessed MDW as a potential hematological parameter in
BC. Using a retrospective approach, a comparative analysis was
carried out between control and BC samples for both MDW and
absolute monocyte number (MONO#). Additionally, MDW was also
examined with respect to the histological stratification of BC
patients.
Materials and methods
Study population and baseline
characteristics
A total of fifty-six BC patients admitted at Betania
Hospital (Naples, Italy) between October 2023 and February 2024
were retrospectively analyzed in this study at the time of
diagnosis. The following exclusion criteria were applied to select
the enrolled population: i) male; ii) under 18 years of age; iii)
stage IV (metastatic); iv) presurgical treatment such as
neoadjuvant chemotherapy. The baseline data required for this study
was extrapolated from the medical records, and include sex, age,
histological appearance, grade, lymph nodes involvement,
lymphovascular invasion and molecular typing (Table I). Similarly, fifty-six whole blood
samples matching the following stated exclusion criteria were
selected as control: i) male; ii) under 18 years of age; iii)
cancer; iv) infection; v) sepsis; iv) systemic inflammatory
response syndrome. Only control subjects who matched BC
participants in age were selected for this study. This study was
approved by the local Ethics Review Board ‘Campania 1’ (Reference
Number 19/24 OSS), and conducted in accordance with the local
legislation and institutional requirements.
 | Table ICharacteristics of patients with
breast cancer. |
Table I
Characteristics of patients with
breast cancer.
| Factors | No. (%) |
|---|
| Age | |
|
<66.2 | 26 (46.43) |
|
≥66.2 | 30 (53.57) |
| Histology | |
|
Invasive
ductal carcinoma | 46 (82.14) |
|
Invasive
lobular carcinoma | 6 (10.71) |
|
Others | 4 (7.14) |
| Surgery | |
|
Mastectomy | 19 (33.93) |
|
Lumpectomy | 35 (62.50) |
|
Unknown | 2 (3.57) |
| Node | |
|
N0 | 29 (51.79) |
|
N1-3 | 12 (21.43) |
|
NX | 7 (12.50) |
|
Unknown | 8 (14.29) |
| Histological
grade | |
|
I-II | 32 (57.14) |
|
III | 18 (32.14) |
|
Unknown | 6 (10.71) |
| LV invasion | |
|
Positive | 36 (64.29) |
|
Negative | 3 (5.36) |
|
Unknown | 17 (30.36) |
| ER status | |
|
Positive | 46 (82.14) |
|
Negative | 6 (10.71) |
|
Unknown | 4 (7.14) |
| PR status | |
|
Positive | 35 (62.50) |
|
Negative | 15 (26.79) |
|
Unknown | 6 (10.71) |
| HER2 status | |
|
Positive | 22 (39.29) |
|
Negative | 26 (46.43) |
|
Unknown | 8 (14.29) |
| Molecular
subtype | |
|
Luminal
A | 20 (35.71) |
|
Luminal
B | 22 (39.29) |
|
HER2-enriched | 3 (5.36) |
|
TNBC | 3 (5.36) |
|
Unknown | 8 (14.29) |
Blood analysis and data
collection
A complete blood count was performed for both BC
patients and controls by DxH900 hematology analyzers (Beckman
Coulter, Inc., Brea, California) in compliance with the
manufacturer's guidelines. MDW and MONO# data were retrospectively
collected and evaluated for statistical purposes in this study.
Statistical analysis
Experimental data were statistically analyzed using
GraphPad Prism Software (Version 8.0.2). Box and Whisker Plot were
chosen to graphically display the whole data distribution (from the
lowest to the highest values passing through median and quartiles).
Data distribution was assessed by Anderson-Darling test for both
#MONO and MDW. Welch's t-test or Brown-Forsythe and Welch ANOVA
were performed to estimate the mean differences between two or more
than two pairs of experimental groups when the normality test was
passed, respectively. Games-Howell correction was used for multiple
comparisons as a post hoc test. Conversely, Mann-Whitney
non-parametric test was applied for non-Gaussian distribution
datasets. All tests were two tailed, and a P-value of less than
0.05 was considered statistically significant. The area under the
curve (AUC) for each of the receiver operating characteristic (ROC)
curve is annotated with the 95% confidence interval (CI) by
Wilson/Brown. False Discovery Rate (FDR) was calculated by
two-stage linear step-up procedure of Benjamini, Krieger and
Yekutieli, by setting a Q-value of 0.05 as significant.
Results
Pathological features of BC
patients
This study included fifty-six women with BC
undergoing either mastectomy (33.93%) or lumpectomy (62.50%).
Histologically, the most common subtype was the invasive ductal
carcinoma (IDC) (46 out of 56, equal to 82.14%), followed by the
invasive lobular carcinoma (ILC) (6 out of 56, 10.71%) and others
(4 out of 56, 7.14%). This latter group enclosed all the BC
patients that could not be included in the previous two subtypes,
such as in-situ and sarcoma. Lymph node spreading assessment
displayed a percentage equal to 51.79% (29 out of 56) for the N0
cluster, 21.43% (12 out of 56) for N1-3 and 12.50% (7 out of 56)
for NX. The most frequently occurring grade was grade I-II (32 out
of 56, 57.14%), while 18 patients showed a higher grading (32.14%).
Regarding the molecular typing, 82.14% (46 out of 56), 62.50% (35
out of 56), 39.29% (22 out of 56) of the cases were classified as
estrogen receptor (ER), progesterone receptor (PR) and human
epidermal growth factor receptor 2 (HER2) positive, respectively.
Luminal A (35.71%) and Luminal B (39.29) were the most frequently
occurring molecular subtype, followed by HER2-enriched (5.36%) and
triple-negative breast cancer (TNBC) (5.36%) (Table I).
Breast cancer patients exhibit higher
MDW than controls
To assess potential differences in monocyte number
and volume between BC patients and controls, we retrospectively
analyzed MONO# and MDW data from the complete blood count obtained
by DxH900 hematology analyzers (Beckman Coulter, Inc., Brea,
California).
Initially, a normality test was carried out to
define #MONO and MDW distribution in either control or BC patients.
Anderson-Darling statistic indicated a Gaussian distribution for
both control (P=0.0633) and BC (P=0.2753) group with respect to
MDW. Conversely, #MONO did not pass normality test in control
(P=0.0013) as well as BC (P<0.0001) samples.
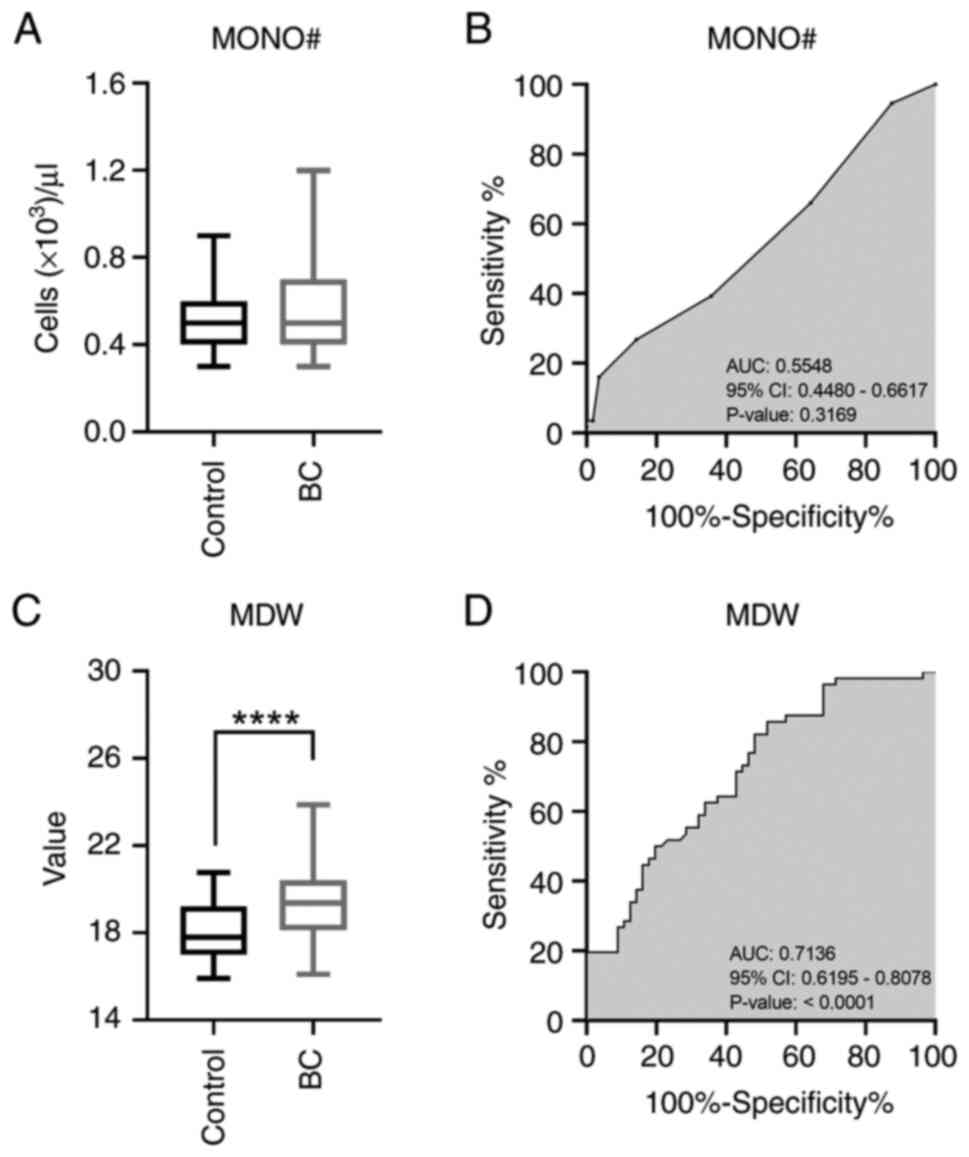
MONO# displayed a median of
0.555x103±0.186 per microliter in the BC patients, while
a value of 0.507x103±0.136 per microliter was observed
in controls (P=0.3068 by Mann-Whitney test; Q 0.1611) (Fig. 1A). Interestingly, MDW showed a
value of 19.41±1.66 in BC patients and 18.14±1.41 in control group
(P<0.0001 by Welch's t-test; Q 0.0001) (Fig. 1C). ROC curve analyses provided an
AUC of 0.5548 (95% CI: 0.4480-0.6617) for MONO#, while for MDW the
same value stood at 0.7136 (95% CI: 0.6195-0.8078) (Fig. 1B and D).
These results indicate no significant difference in
MONO# between BC and control groups, while, conversely, MDW levels
were higher in BC patients than in controls.
MDW levels correlate with invasive
ductal carcinoma expressing estrogen and progesterone
receptors
Focusing on the MDW parameter, we subsequently
analyzed its performance across the different BC histopathological
features. Histological appearance, ER, PR and HER2 data were
obtained from biopsy reports and subsequently examined in relation
to the MDW levels.
Compared to the control group (18.14±1.41), MDW
values of 18.77±2.07 (P=0.8815; Q 0.8696), 19.53±1.76 (P=0.0002, Q
0.0011), and 19.84±2.43 (P=0.5824; Q 0.8697) were recorded in ILC,
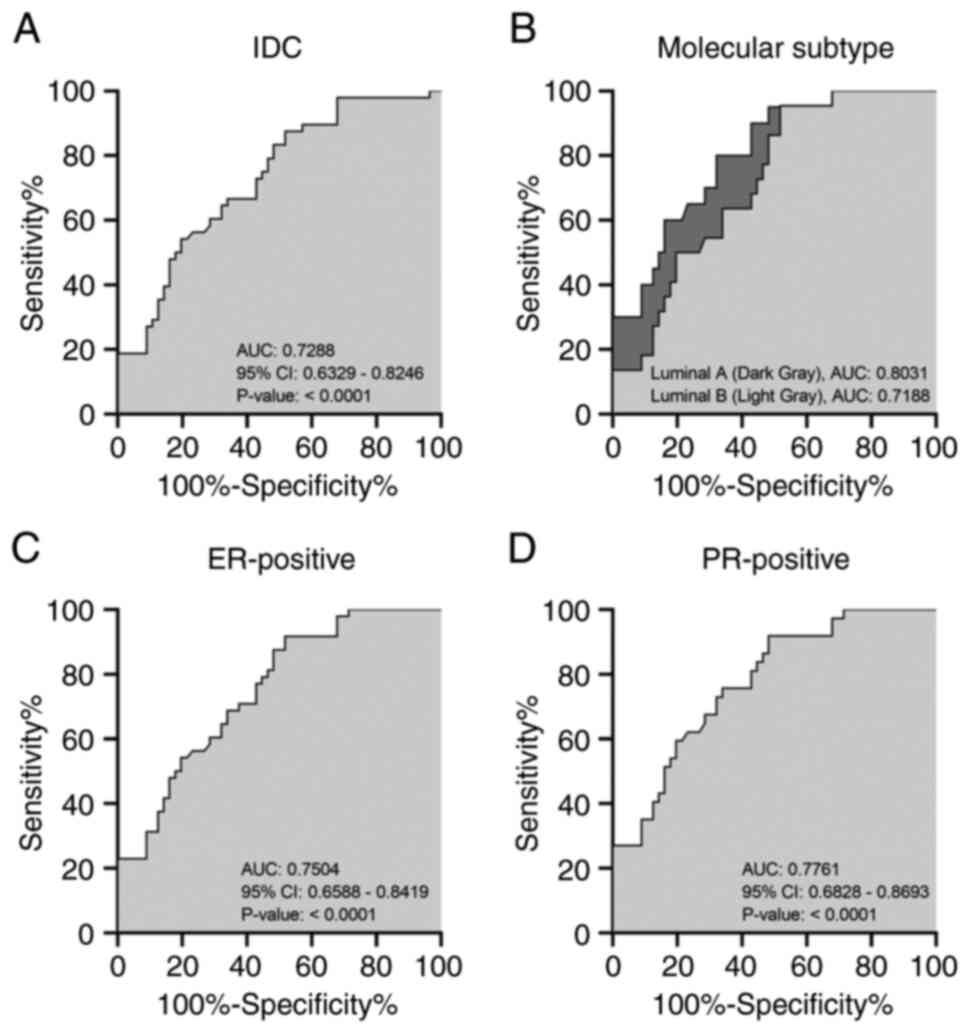
IDC and others histological subtype, correspondingly (Fig. 2A). As the only significant subset,
ROC curve revealed an AUC value of 0.7288 (95% CI: 0.6329-0.8246)
for IDC (Fig. 3A).
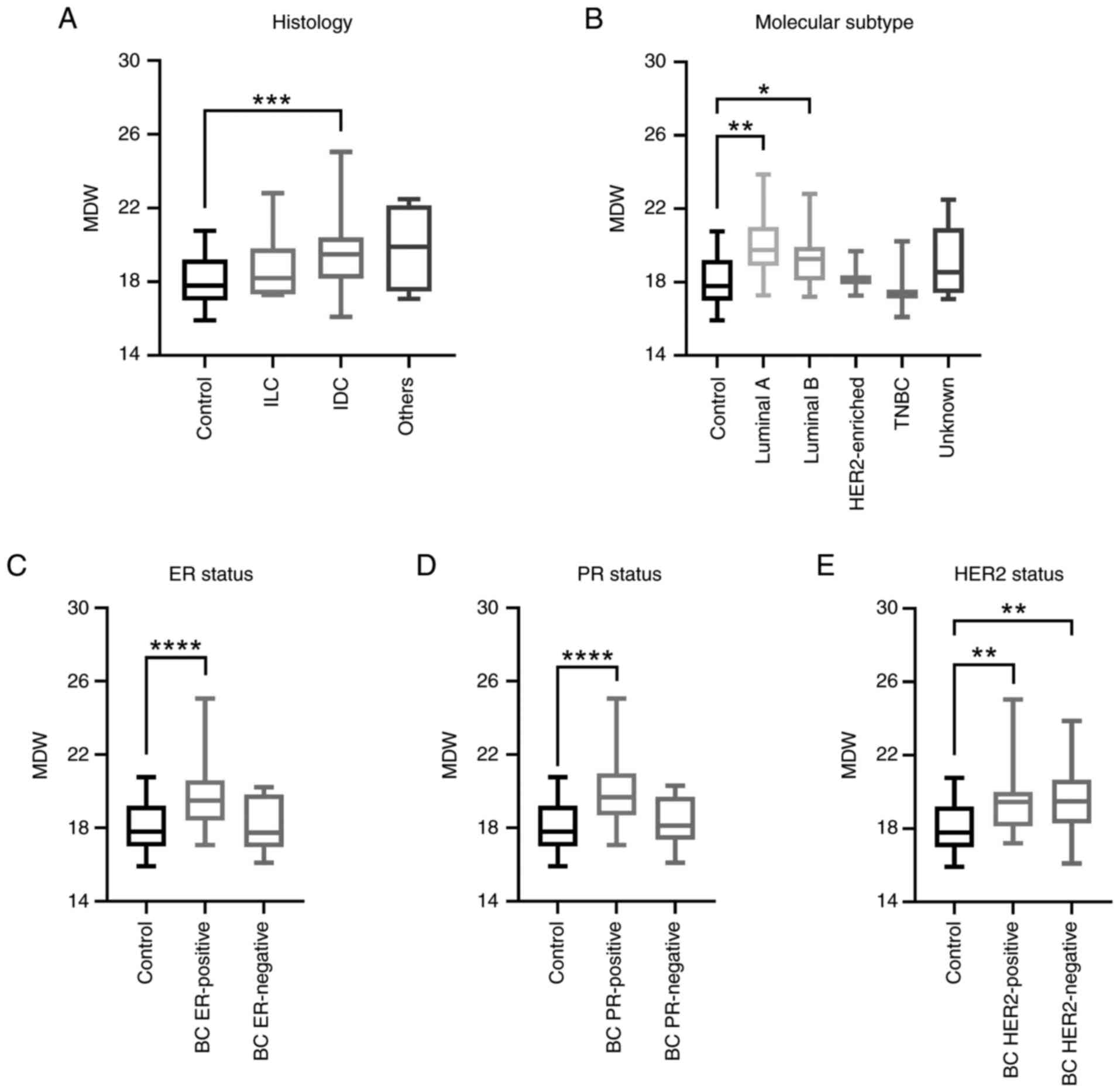 | Figure 2Investigation of the MDW distribution
in patients with BC with different histopathological features. Box
and whisker plots displaying MDW levels in controls and patients
with BC categorized by (A) histologic, (B) molecular subtype, (C)
ER, (D) PR or (E) HER2 status. *P<0.05,
**P<0.01, ***P<0.001,
****P<0.0001 (Brown-Forsythe and Welch ANOVA). BC,
breast cancer; ER, estrogen receptor; IDC, invasive ductal
carcinoma; ILC, invasive lobular carcinoma; MDW, monocyte
distribution width; PR, progesterone receptor; TNBC,
triple-negative breast cancer. |
Moving to the hormone-related receptors, BC patients
showed MDW levels of 19.68±1.76 (P<0.0001; Q 0.0002) when ER was
expressed and 18.12±1.57 (P=0.9996; Q 0.6997) in the absence of the
latter (Fig. 2C). Remarkably, the
AUC obtained from ER-positive ROC curve stood at 0.7504 (95% CI:
0.6588-0.8419) (Fig. 3C).
Analogues results were also obtained concerning the PR status
because the expression of this receptor was associated with higher
MDW (19.90±1.80; P<0.0001; Q 0.0001) with respect to the
negative subset (18.35±1.23; P=0.8372; Q 0.2930) (Fig. 2D). ROC analysis indicated good
specificity and sensitivity in predicting the PR status by MDW
(AUC: 0.7761; 95% CI: 0.6828-0.8693) (Fig. 3D). Concerning HER2, MDW values were
19.51±1.87 (P=0.0077; Q 0.0040) in HER2-positive and 19.55±1.79
(P=0.0030; Q 0.0032) in HER2-negative BC patients (Fig. 2E).
Starting from the ER, PR and HER2 status, the
molecular subtype was subsequently addressed in relation to the MDW
levels. Luminal A (20.02±1.64) and TNBC (17.89±1.64) exhibited the
highest and lowest MDW values, correspondingly (Fig. 2B). Intermediate measures were
detected for Luminal B (19.31±1.45) and HER2-enriched (18.36±1.23)
instead (Fig. 2B). Statistical
analysis also revealed significant differences in mean by comparing
both Luminal A (P=0.0010; Q 0.0042) and Luminal B (P=0.0275; Q
0.0578) with controls. Remarkably, ROC curves displayed and AUC of
0.8031 (95% CI: 0.6897-0.9076) and 0.7188 (95% CI: 0.6013-0.8332)
in these two subsets (Fig.
3B).
Overall, these findings propose a possible
association between MDW and certain histopathological features in
BC. Specifically, higher MDW levels were typically found in IDC
expressing ERs and PRs. Elevated MDW values were also noted in BC
patients presenting either Luminal A or B subtype.
MDW exhibits higher values in BC
patients presenting grade III, lymph nodes involvement, and
lymphovascular invasion
The prognostic significance of histologic grading
has extensively been studied in BC, representing one of two
benchmarks for the outcome prediction along with disease stage
(20). The TNM system defines
disease stage taking into account tumor size, involvement of nearby
lymph nodes and dissemination to other parts of the body (21). More recently, additional prognostic
factors, such as the lymphovascular invasion (LV), have been
included to provide a more comprehensive assessment on BC
progression (22,23).
Using the biopsy reports, we later evaluated the MDW
levels across the different BC grades and stages, as well as LV
invasion. MDW showed value of 19.32±1.64 (P=0.0028; Q 0.0026) in
grade I-II and 19.88±2.00 (P=0.0050; Q 0.0026) in grade III, with
respect to 18.14±1.41 as median in controls (Fig. 4A). Using the N category as one of
hallmarks of the disease stage, 19.35±2.01 (P=0.0234; Q 0.0143) and
19.69±1.63 (P=0.0273; Q 0.0143) were the MDW average obtained in N0
and N1-3 clusters, correspondingly (Fig. 4B). Higher MDW values were also
recorded in LV-positive patients (19.62±1.63; P<0.0001; Q
0.0001), while no significant changes were documented in
LV-negative with respect to control group (17.93±0.19; P=0.6056; Q
0.2120) (Fig. 4C).
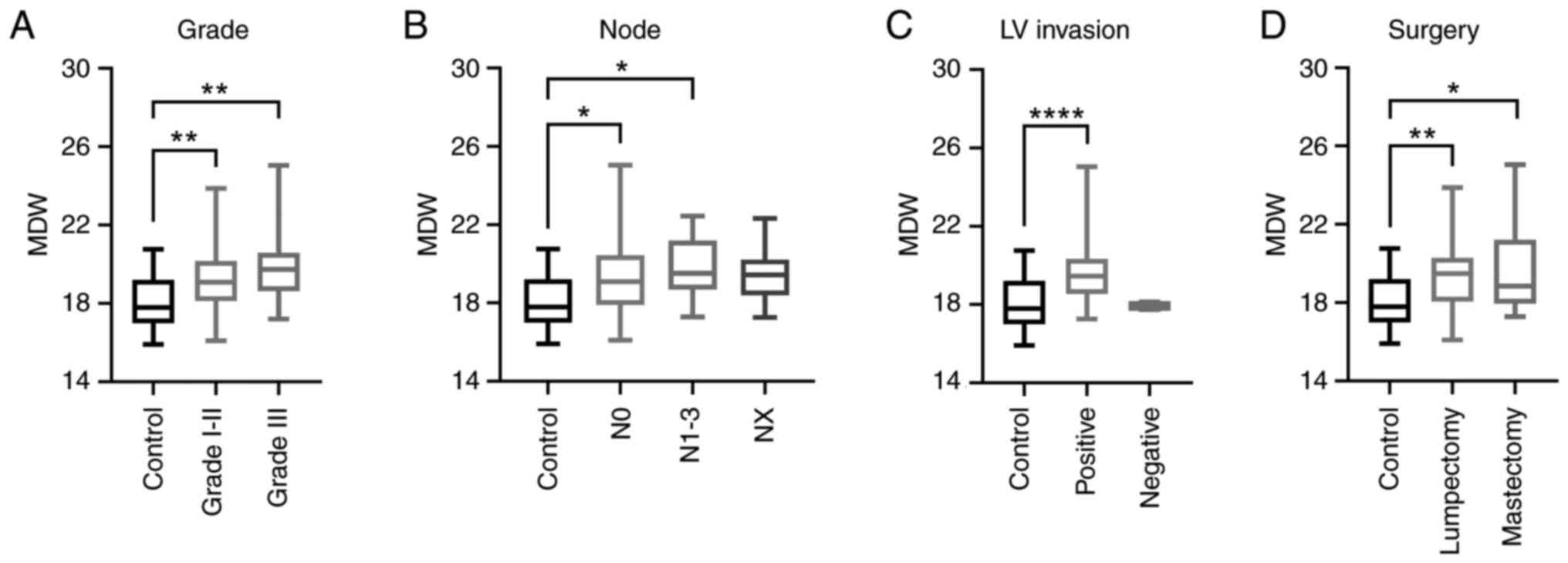 | Figure 4Assessment of the MDW with respect to
grade, lymph node involvement, LV invasion, and surgery in patients
with BC. Box and whisker plots outlining MDW distribution in
controls and patients with BC grouped by (A) grade, (B) lymph node
involvement, (C) LV invasion and (D) surgery.
*P<0.05, **P<0.01,
****P<0.0001 (Brown-Forsythe and Welch ANOVA). BC,
breast cancer; LV, lymphovascular; MDW, monocyte distribution
width. |
Taken together, these findings suggest that MDW
levels could correlate with the progression of both grades and
stages in BC. The elevated MDW levels detected in LV-positive BC
patients, as well in those undergoing mastectomy (19.55±2.11;
P=0.0262; Q 0.0138) rather than lumpectomy (19.37±1.70; P=0.0016; Q
0.0017) (Fig. 4D), support its
possible correlation with the BC severity.
Discussion
The prognostic assessment of BC has undergone a
profound change over the years, moving this pathological condition
from a life-threatening to a potentially curable disease. What made
it all possible was the persistent advances achieved in therapeutic
field, along with the implementation of screening programs, which
have ensured an early detection of BC (24,25).
Nevertheless, there is a significant number of BC patients still
progressing to the advanced metastatic stage due to tumor
recurrence and drug resistance (26,27).
Additionally, early detection remains intricate for certain BC
subtypes like the TNBC (28,29).
Therefore, identifying non-invasive, straightforward and low-risk
biomarkers is still pertinent and timely for early detection of
BC.
As a novel hematological parameter, MDW has recently
been recognized as an effective diagnostic and prognostic biomarker
in different pathological conditions, including sepsis, viral
infections and other inflammatory-related diseases (13-15,30).
The growing interest in MDW is further supported by
the increasing number of available studies over time. Not
surprisingly, PubMed database reveals that 483 out of 539
MDW-related items have only been released in the last ten years.
Although the vast majority of these findings focus on infectious
and inflammatory-related illnesses, additional medical applications
are beginning to emerge, particularly in malignant disorders. On
that note, a recent study reported alterations in MDW across
different stages of chronic liver diseases, demonstrating for the
first time a positive correlation between MDW levels and occurrence
and development of hepatocellular carcinoma (19).
Herein, we observed no difference in MONO# between
BC patients and control group, while conversely, MDW was higher in
BC patients than in controls. Although our results suggest that MDW
could serve as a potential marker in BC diagnosis, the obtained AUC
values highlight a ‘fair leaning towards good’ performance of this
test in distinguishing the presence or absence of the disease.
Nevertheless, as widely known, labelling systems for AUC are quite
arbitrary since strong discriminatory ability is not sufficient to
claim a positive effect in clinical practice. The primary purpose
of AUC values should be to compare discriminative aptitude of
different biomarkers (31). Future
investigations should aim to conduct AUC comparative analysis
between MDW and other hematological parameters currently employed
for BC diagnosis. After all, a combination of different biomarkers
has already been recommended to provide a reliable diagnosis in
cancer, and thus MDW could be included on the list to further
enhance BC detection and stratification (8,32).
With respect to this latter concern, we observed a
positive association between MDW levels and some specific BC
features, such as histologic subtype and molecular typing.
Specifically, higher MDW values were detected in IDC but not in
ILC, as well as in BC expressing both ERs and PRs. Conversely,
elevated MDW was recorded in both HER2-positive and HER2-negative
BC patients compared with controls.
The hypothesis that MDW levels could be related to
the ER and PR expression was also suggested by the subtype
analysis. Not coincidentally, higher MDW values were detected in
Luminal A/B but not in HER2-enriched/TNBC, which do not express or
exhibit very low levels of hormone receptors.
The possibility of using a simple blood count test
to obtain relevant BC histological features could offer a rapid and
non-invasive option to the well-recognized biopsy procedure.
Although MDW will never replace biopsy, its assessment could
accelerate the therapeutic decision-making processes.
If supported by future data, MDW could also serve as
a conceivable predictive/prognostic marker in BC. The obtained data
propose, indeed, the existence of a positive correlation between
MDW and the progression of both BC grades and stages. Elevated MDW
levels were also detected in LV-positive BC patients, reinforcing
the possible association with the severity of BC.
Tumor microenvironment (TME) has revealed an active
involvement of host immune cells in BC (33). TAMs constitute the most prominent
immune infiltrated component of TME in BC, reaching almost 50% of
non-cancer cells in some cases (34). They are usually recruited from
circulating monocytes, which differentiate in situ via a
CCL2-CCR2 chemokine signaling pathway (35,36).
M1 and M2 identify two distinct TAM subtypes with competing views
on tumor growth and progression, as well as on TME inflammation
(33,37). Nevertheless, M1/M2 distinction is
somewhat ambiguous because intermediate states have also been
recognized in TME (38). Assuming
a correlation with TAMs recruitment and differentiation, MDW
fluctuations could intuitively reflect remodeling in TME. However,
further studies are needed to confirm the mechanisms linking MDW to
TAMs in BC, as well as in other kinds of cancer.
However, it is also necessary to mention some
limitations of the study. Among the others, special attention
should be paid to the sample size of the proposed investigation, as
well as to the stated conclusions. Pilot studies constitute a
useful analytical approach to test both feasibility and
acceptability of proposed trials (39). Due to its preliminary feature,
pilot study does not rely on power calculations to determine the
appropriate sample size. However, different rules of thumb have
been proposed for addressing this issue (40). Typically, a sample size ranging
from 12 to 35 per group is applied as regards the continuous
outcomes. In accordance with this statement, the International
Standard Randomized Controlled Trial Number (ISRCTN) registry
showed a median sample size of 30 (Interquartile Range 20-43)
across the 592 continuous outcome studies carried out between 2013
and 2020(41). Whilst rules of
thumb offer valuable guidance, the optimal sample size should be
determined by considering the main purpose of the pilot study. A
higher number is demanded to estimate differences among the
experimental groups in a pilot approach for instance. Benchmark
metric indicates that a sample size between 60 and 100 might be
required to assess event rates in an intervention group (42). Despite the selected sample number
is slightly below the proposed range, it broadly covers the 9% of
the main planned trial, as suggested by Cocks and Torgerson for
pilot study (43).
By gathering both the Italian National Statistics
Institute (ISTAT) and the Italian Association of Medical Oncology
(AIOM) data, the prevalence of BC has been estimated to 3.07% in
2024 (44,45). Applying a confidence interval (CI)
equal to 95%, large scale trials should enroll a number of patients
between 1941 and 125 to achieve a precision ranging from 0.77 to
2.99%, namely below its prevalence as recommended when the
occurrence is not higher than 10% (46). In light of these considerations,
the number of samples enrolled in this study confers a degree of
precision equal to 4.51%. Exactly the restricted number of enrolled
patients, as well as the resulting precision, requires a certain
caution in interpreting the study results. Whilst deviations are
statistically significant across the experimental groups (as
supported by both P-value and Q-value), overestimation or
underestimation cannot be excluded precisely because of the limited
number of samples.
Moreover, the impact of confounding variables has
not been addressed in our analysis, and thus further caution is
required in drawing the appropriate conclusions. A very recent
study has examined comorbidity affecting MDW pattern in
non-infectious (control) group (47). Aside from increasing in cancer
patients, MDW elevation was detected in some chronic diseases, such
as diabetes, cirrhosis and immune suppression (47). Whilst these findings emphasize the
relevance of confounding factors in MDW interpretation, identifying
potential biases remains intricate even just at physiological
level. Reference interval is still being defined for MDW, and thus
changes by either age or gender are not yet understood for instance
(48).
In conclusion, we demonstrated for the first time
that MDW can serve as a potential hematological biomarker in BC.
Higher MDW levels were typically observed in BC patients presenting
IDC subtype and expressing either ERs or PRs. A positive
correlation between MDW and BC severity was also suggested based on
grade, lymph nodes involvement and LV-invasion. However,
considering the exploratory pilot nature of the study, large-scale
trials like multi-center prospective studies are mandatory to
increase statistical robusteness, generalizability, and
reproducibility of MDW in BC.
Acknowledgements
Not applicable.
Funding
Funding: The present study was funded by the University of
Campania ‘Luigi Vanvitelli’, iRESCUE project.
Availability of data and materials
The data generated in the present study may be
requested from the corresponding author.
Authors' contributions
LS and SN were involved in the conceptualization and
design of the study. RN, GC and RC collected data. GN and RG
curated the data, and confirmed the authenticity of all the raw
data. RG also implemented the research methodology. GN handled
preparation, creation and presentation of the published data. AR,
SK and GP performed statistical analyses. MC and AL contributed to
data interpretation. LS wrote the original draft. MC, AL and SN
reviewed and edited the manuscript. SN supervised the study. LS
acquired funding. All authors have accepted responsibility for the
entire content of this manuscript and approved its submission. All
authors read and approved the final version of the manuscript.
Ethics approval and consent to
participate
The present study complied with all relevant
national regulations and institutional policies, and was performed
in accordance with the tenets of the Helsinki Declaration. The
study approval was granted by the local Ethics Review Board
‘Campania 1’ located at the National Cancer Institute ‘Fondazione
G. Pascale’ of Naples (Naples, Italy; approval no. 19/24 OSS).
Written informed consent was obtained from all individuals included
in the present study.
Patient consent for publication
The patients provided written informed consent for
the publication of any data and/or accompanying images.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Dizon DS and Kamal AH: Cancer statistics
2024: All hands on deck. CA Cancer J Clin. 74:8–9. 2024.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Arnold M, Morgan E, Rumgay H, Mafra A,
Singh D, Laversanne M, Vignat J, Gralow JR, Cardoso F, Siesling S
and Soerjomataram I: Current and future burden of breast cancer:
Global statistics for 2020 and 2040. Breast. 66:15–23.
2022.PubMed/NCBI View Article : Google Scholar
|
|
3
|
The Lancet Oncology: Curbing the climb in
cancer incidence. Lancet Oncol. 25(529)2024.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Xiong X, Zheng LW, Ding Y, Chen YF, Cai
YW, Wang LP, Huang L, Liu CC, Shao ZM and Yu KD: Breast cancer:
Pathogenesis and treatments. Signal Transduct Target Ther.
10(49)2025.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Wang J and Wu SG: Breast cancer: An
overview of current therapeutic strategies, challenge, and
perspectives. Breast Cancer (Dove Med Press). 15:721–730.
2023.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Shin J, Kim JY, Oh JM, Lee JE, Kim SW, Nam
SJ, Park W, Park YH, Ahn JS and Im YH: Comprehensive clinical
characterization of decade-long survivors of metastatic breast
cancer. Cancers (Basel). 15(4720)2023.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Seale KN and Tkaczuk KHR: Circulating
biomarkers in breast cancer. Clin Breast Cancer. 22:e319–e331.
2022.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Zhou Y, Tao L, Qiu J, Xu J, Yang X, Zhang
Y, Tian X, Guan X, Cen X and Zhao Y: Tumor biomarkers for
diagnosis, prognosis and targeted therapy. Signal Transduct Target
Ther. 9(132)2024.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Moar K, Pant A, Saini V, Pandey M and
Maurya PK: Potential diagnostic and prognostic biomarkers for
breast cancer: A compiled review. Pathol Res Pract.
251(154893)2023.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Varzaru VB, Eftenoiu AE, Vlad DC, Vlad CS,
Moatar AE, Popescu R and Cobec IM: The influence of tumor-specific
markers in breast cancer on other blood parameters. Life (Basel).
14(458)2024.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Lopez-Gonzalez L, Sanchez Cendra A,
Sanchez Cendra C, Roberts Cervantes ED, Espinosa JC, Pekarek T,
Fraile-Martinez O, García-Montero C, Rodriguez-Slocker AM,
Jiménez-Álvarez L, et al: Exploring biomarkers in breast cancer:
Hallmarks of diagnosis, treatment, and follow-up in clinical
practice. Medicina (Kaunas). 60(168)2024.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Agnello L, Ciaccio AM, Vidali M,
Cortegiani A, Biundo G, Gambino CM, Scazzone C, Lo Sasso B and
Ciaccio M: Monocyte distribution width (MDW) in sepsis. Clin Chim
Acta. 548(117511)2023.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Ligi D, Lo Sasso B, Henry BM, Ciaccio M,
Lippi G, Plebani M and Mannello F: Deciphering the role of monocyte
and monocyte distribution width (MDW) in COVID-19: An updated
systematic review and meta-analysis. Clin Chem Lab Med. 61:960–973.
2023.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Malinovska A, Hernried B, Lin AD,
Badaki-Makun O, Fenstermacher K, Ervin AM, Ehrhardt S, Levin S and
Hinson JS: Monocyte distribution width as a diagnostic marker for
infection a systematic review and meta-analysis. Chest.
164:101–113. 2023.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Wu J, Li L and Luo J: Diagnostic and
prognostic value of monocyte distribution width in sepsis. J
Inflamm Res. 15:4107–4117. 2022.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Cusinato M, Sivayoham N and Planche T:
Sensitivity and specificity of monocyte distribution width (MDW) in
detecting patients with infection and sepsis in patients on sepsis
pathway in the emergency department. Infection. 51:715–727.
2022.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Agnello L, Ciaccio AM, Ben FD, Gambino CM,
Scazzone C, Giglia A, Biundo G, Cortegiani A, Sasso BL and Ciaccio
M: Clinical usefulness of presepsin and monocyte distribution width
(MDW) kinetic for predicting mortality in critically ill patients
in intensive care unit. Front Med (Lausanne).
11(1393843)2024.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Agnello L, Ciaccio AM, Del Ben F, Lo Sasso
B, Biundo G, Giglia A, Giglio RV, Cortegiani A, Gambino CM and
Ciaccio M: Monocyte distribution width (MDW) kinetic for monitoring
sepsis in intensive care unit. Diagnosis (Berl). 11:422–429.
2024.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Lin S, Yang X, Yang X, Tang M, Yao X, Ye
Y, Huang Q, Huang J, Li J, Yi Q, et al: Monocyte distribution width
as a promising biomarker for differential diagnosis of chronic
hepatitis, cirrhosis, and hepatocellular carcinoma. Front Immunol.
15(1406671)2024.PubMed/NCBI View Article : Google Scholar
|
|
20
|
Jaroensri R, Wulczyn E, Hegde N, Brown T,
Flament-Auvigne I, Tan F, Cai Y, Nagpal K, Rakha EA, Dabbs DJ, et
al: Deep learning models for histologic grading of breast cancer
and association with disease prognosis. NPJ Breast Cancer.
8(113)2022.PubMed/NCBI View Article : Google Scholar
|
|
21
|
Brierley J, O'Sullivan B, Asamura H, Byrd
D, Huang SH, Lee A, Piñeros M, Mason M, Moraes FY, Rösler W, et al:
Global consultation on cancer staging: Promoting consistent
understanding and use. Nat Rev Clin Oncol. 16:763–771.
2019.PubMed/NCBI View Article : Google Scholar
|
|
22
|
Houvenaeghel G, Cohen M, Classe JM, Reyal
F, Mazouni C, Chopin N, Martinez A, Daraï E, Coutant C, Colombo PE,
et al: Lymphovascular invasion has a significant prognostic impact
in patients with early breast cancer, results from a large,
national, multicenter, retrospective cohort study. ESMO Open.
6(100316)2021.PubMed/NCBI View Article : Google Scholar
|
|
23
|
Nishimura R, Osako T, Okumura Y, Nakano M,
Ohtsuka H, Fujisue M and Arima N: An evaluation of lymphovascular
invasion in relation to biology and prognosis according to subtypes
in invasive breast cancer. Oncol Lett. 24(245)2022.PubMed/NCBI View Article : Google Scholar
|
|
24
|
Emanuelle Pereira Santos V, Luiz de Franca
Neto P, Eda de Oliveira Isidio B, Henrique Bezerra Fontes P,
Andrêssa de Moura I, Isabel Santos Cruz B, Máyra Gois de Sousa M,
Luana Dos Santos D, de França São Marcos B, Sousa de Pinho S, et
al: An overview about biomarkers in breast cancer: Insights into
the diagnostic and prognostic significance. Clin Chim Acta.
567(120030)2025.PubMed/NCBI View Article : Google Scholar
|
|
25
|
Zhang H, Hussin H, Hoh CC, Cheong SH, Lee
WK and Yahaya BH: Big data in breast cancer: Towards precision
treatment. Digit Health. 10(20552076241293695)2024.PubMed/NCBI View Article : Google Scholar
|
|
26
|
Kinnel B, Singh SK, Oprea-Ilies G and
Singh R: Targeted therapy and mechanisms of drug resistance in
breast cancer. Cancers (Basel). 15(1320)2023.PubMed/NCBI View Article : Google Scholar
|
|
27
|
Pedersen RN, Esen BO, Mellemkjaer L,
Christiansen P, Ejlertsen B, Lash TL, Nørgaard M and Cronin-Fenton
D: The incidence of breast cancer recurrence 10-32 years after
primary diagnosis. J Natl Cancer Inst. 114:391–399. 2022.PubMed/NCBI View Article : Google Scholar
|
|
28
|
Adrada BE, Moseley TW, Kapoor MM, Scoggins
ME, Patel MM, Perez F, Nia ES, Khazai L, Arribas E, Rauch GM and
Guirguis MS: Triple-negative breast cancer: Histopathologic
features, genomics, and treatment. Radiographics.
43(e230034)2023.PubMed/NCBI View Article : Google Scholar
|
|
29
|
Manjunath M and Choudhary B:
Triple-negative breast cancer: A run-through of features,
classification and current therapies (Review). Oncol Lett.
22(512)2021.PubMed/NCBI View Article : Google Scholar
|
|
30
|
Liu Y, Wang R, Zhong S, Qian L and Wang D:
Monocyte distribution width as an early predictor of short-term
outcome in adult patients with sepsis. Clin Chem Lab Med.
62:562–571. 2023.PubMed/NCBI View Article : Google Scholar
|
|
31
|
de Hond AAH, Steyerberg EW and van Calster
B: Interpreting area under the receiver operating characteristic
curve. Lancet Digit Health. 4:e853–e855. 2022.PubMed/NCBI View Article : Google Scholar
|
|
32
|
Li D, Ju F, Wang H, Fan C, Jacob JC, Gul
S, Zaliani A, Wartmann T, Polidori MC, Bruns CJ and Zhao Y:
Combination of the biomarkers for aging and cancer?-Challenges and
current status. Transl Oncol. 38(101783)2023.PubMed/NCBI View Article : Google Scholar
|
|
33
|
Zhang Y, Zhong F and Liu L: Single-cell
transcriptional atlas of tumor-associated macrophages in breast
cancer. Breast Cancer Res. 26(129)2024.PubMed/NCBI View Article : Google Scholar
|
|
34
|
Huang XQ, Cao JS and Zu XY:
Tumor-associated macrophages: An important player in breast cancer
progression. Thorac Cancer. 13:269–276. 2022.PubMed/NCBI View Article : Google Scholar
|
|
35
|
Arwert EN, Harney AS, Entenberg D, Wang Y,
Sahai E, Pollard JW and Condeelis JS: A unidirectional transition
from migratory to perivascular macrophage is required for tumor
cell intravasation. Cell Rep. 23:1239–1248. 2018.PubMed/NCBI View Article : Google Scholar
|
|
36
|
Munir MT, Kay MK, Kang MH, Rahman MM,
Al-Harrasi A, Choudhury M, Moustaid-Moussa N, Hussain F and Rahman
SM: Tumor-Associated macrophages as multifaceted regulators of
breast tumor growth. Int J Mol Sci. 22(6526)2021.PubMed/NCBI View Article : Google Scholar
|
|
37
|
Pan Y, Yu Y, Wang X and Zhang T:
Tumor-associated macrophages in tumor immunity. Front Immunol.
11(583084)2020.PubMed/NCBI View Article : Google Scholar
|
|
38
|
Komohara Y, Kurotaki D, Tsukamoto H,
Miyasato Y, Yano H, Pan C, Yamamoto Y and Fujiwara Y: Involvement
of protumor macrophages in breast cancer progression and
characterization of macrophage phenotypes. Cancer Sci.
114:2220–2229. 2023.PubMed/NCBI View Article : Google Scholar
|
|
39
|
In J: Introduction of a pilot study.
Korean J Anesthesiol. 70:601–605. 2017.PubMed/NCBI View Article : Google Scholar
|
|
40
|
Kunselman AR: A brief overview of pilot
studies and their sample size justification. Fertil Steril.
121:899–901. 2024.PubMed/NCBI View Article : Google Scholar
|
|
41
|
Totton N, Lin J, Julious S, Chowdhury M
and Brand A: A review of sample sizes for UK pilot and feasibility
studies on the ISRCTN registry from 2013 to 2020. Pilot Feasibility
Stud. 9(188)2023.PubMed/NCBI View Article : Google Scholar
|
|
42
|
Teresi JA, Yu X, Stewart AL and Hays RD:
Guidelines for designing and evaluating feasibility pilot studies.
Med Care. 60:95–103. 2022.PubMed/NCBI View Article : Google Scholar
|
|
43
|
Cocks K and Torgerson DJ: Sample size
calculations for pilot randomized trials: A confidence interval
approach. J Clin Epidemiol. 66:197–201. 2013.PubMed/NCBI View Article : Google Scholar
|
|
44
|
Italian National Statistics Institute
(ISTAT). ISTAT, Rome, 2025. Accessed on July 3, 2025. https://www.istat.it/en/.
|
|
45
|
Italian Association of Medical Oncology
(AIOM). AIOM, Mila, 2025. Accessed on July 3, 2025. https://www.aiom.it/en/.
|
|
46
|
Naing L, Nordin RB, Abdul Rahman H and
Naing YT: Sample size calculation for prevalence studies using
Scalex and ScalaR calculators. BMC Med Res Methodol.
22(209)2022.PubMed/NCBI View Article : Google Scholar
|
|
47
|
Kralovcova M, Müller J, Hajsmanova Z,
Sigutova P, Bultasova L, Palatova J and Matejovic M: Understanding
the value of monocyte distribution width (MDW) in acutely ill
medical patients presenting to the emergency department: A
prospective single center evaluation. Sci Rep.
14(152555)2024.PubMed/NCBI View Article : Google Scholar
|
|
48
|
García-Álvarez A, Posada Franco Y,
Martinez-Novillo González M, Hernández Álvarez E, Serrano García I
and Sanz-Casla MT: Monocyte distribution width (MDW): Study of
reference values in blood donors. Clin Chem Lab Med. 62:1133–1137.
2024.PubMed/NCBI View Article : Google Scholar
|