1. Introduction
The incidence of gynecological tumors in China is
increasing annually, accounting for 15-20% of female malignant
tumors; its overall incidence is on the rise (1) and has become the third most lethal
disease among female tumors (2).
For patients with early-stage gynecological tumors, standard
treatment can notably improve prognosis. Nevertheless, for
recurrent, metastatic and drug-resistant patients, there is often a
lack of effective therapeutic measures in the early stage of
disease. Besides, research into the mechanisms contributing to the
poor prognosis of tumor therapy remains insufficient. Therefore,
discovering new biomarkers and therapeutic targets is the top
priority for curing gynecological tumors.
Complement component 1 Q subcomponent-binding
protein (C1QBP), a multifunctional protein, is upregulated in a
variety of tumor types (3-5).
C1QBP plays a vital role in both normal physiological processes and
the development of various tumors such as cervical, ovarian and
endometrial cancer, particularly by regulating critical biological
processes such as proliferation, migration and invasion (3-7).
However, multiple questions still remain concerning the specific
mechanism or the role of C1QBP in gynecological tumors and its
potential as a therapeutic target. In view of these findings, the
present review summarizes the research progress on C1QBP in
gynecological tumors in recent years, explores its mechanism of
action in tumorigenesis and development and assesses its potential
as a biomarker and therapeutic target, with the aim of providing a
theoretical basis for subsequent research. A search for articles
was conducted using databases such as Web of Science (https://www.webofscience.com/), PubMed (https://pubmed.ncbi.nlm.nih.gov/) and CNKI
(https://www.cnki.net/). The key words included
‘gynecological tumors’, ‘ovarian cancer’, ‘endometrial cancer’,
‘cervical cancer’, ‘mitochondria’, ‘C1QBP protein’, ‘tumor
pathogenesis’, ‘clinical’ and ‘non-clinical’. Literature published
in Chinese and English were focused on the reduce information
overload and improve research efficiency. Additionally, literature
within a 16-year range (2010-2016) was focused on, but earlier
literature was included if the content was particularly relevant to
the topic. To the best of our knowledge, the present review is the
first to provide a detailed description of the role of C1QBP in
gynecological tumors and its related mechanisms and to summarize
the retrospective research results in recent years.
2. Structure and function of C1QBP
C1QBP was initially isolated from human Burkitt
lymphoma cell membranes (8).
C1QBP, also known as p32 or gC1qR, is a multifunctional and
evolutionarily conserved protein predominantly localized in the
mitochondrial matrix. The C1QBP gene is located on chromosome 17
and p53 can bind to the promoter region of the C1QBP gene to
influence its transcription (9,10).
The C1QBP precursor protein consists of 282 amino acids and
eventually forms a mature protein of 209 amino acids (8) (Fig.
1). C1QBP is a donut-shaped, acidic protein located in the
mitochondrial matrix. Crystal structure analysis reveals that it
folds into a characteristic trimeric architecture, with each
monomer consisting of seven antiparallel β-strands (β1-β7), in
which a highly distorted antiparallel β-fold is formed where the
β1 strand is positioned almost perpendicular to
β7 (11). This β-fold
consists of an N-terminal α-helix and two C-terminal α-helices,
whose distinctive features add to the stability of the structure
(11). The high negative charge
density and asymmetric charge distribution result in functional
roles on both sides of the protein that are asymmetric, which is
essential for protein-protein interactions and ligand binding
(11,12).
C1QBP is widely expressed in several subcellular
compartments, including the nucleus, Golgi, endoplasmic reticulum
and mitochondria. C1QBP fulfills specific biological functions
according to the distribution states in different microenvironments
(13). For example, C1QBP in the
nucleus participates in the regulation of RNA metabolism and a
study has reported that C1QBP distributed in the Golgi network
participates in cellular mitosis (14). C1QBP also has an important role in
the morphology and structure of the endoplasmic reticulum under
normal cellular conditions. For instance, a lack of C1QBP leads to
fragmentation of the endoplasmic reticulum structure, dissociation
of ribosomes and ultimately the inability to synthesize proteins,
lipids and other substances in normal cells (15). In addition, when localized in
mitochondria, C1QBP is involved in mitochondrial quality control
and metabolism (16), maintenance
of oxidative phosphorylation (OXPHOS) (12) and the synthesis of mitochondrial
proteins (8).
C1QBP also participates in the regulation of immune
cells. Gotoh et al (17)
reported that C1QBP supports the maturation of dendritic cells,
which induces primary immune responses. C1QBP is closely related to
the regulation of humoral immunity. After complement activation,
the generated fragments, such as C3d, covalently bind to the
surface of the activating antigen involving a pathogen and immune
complex (18). B cells recognize
C3d through their complement receptor 2 and obtain a strong
co-stimulatory signal, enhancing the immunogenicity of the antigen.
C1QBP indirectly controls this key process by regulating the
complement activation front (12).
By binding to C1q in its soluble form, C1QBP inhibits classical
pathway activation and subsequent C3d production. Conversely, when
expressed on the cell surface under pathological conditions, C1QBP
can directly activate the classical complement pathway, leading to
enhanced C3d deposition and modulation of B cell co-stimulatory
signals (12). Furthermore, C1QBP
mediates the clearance of complement component 1q (C1q)-coated
immune complexes and apoptotic cells by phagocytes. The absence of
C1q or the presence of anti-C1q antibodies can lead to the
deposition of immune complexes in tissues such as the kidneys,
triggering autoimmune diseases such as lupus nephritis, which is a
pathological form of humoral immunity (19). Additionally, C1QBP can promote the
coagulation process by inhibiting fibrin polymerization (20). C1QBP is involved in the regulation
of nuclear function; it not only regulates apoptosis but also
regulates the splicing process through the interaction of the
function and/or localization of different splicing factors
(21,22). C1QBP also promotes the occurrence
of microbial infection. Staphylococcus aureus protein A
contributes to pathogenesis by interacting with the host complement
receptor C1QBP (23). Hepatitis C
virus, human immunodeficiency virus, herpes simplex virion type 1
and Epstein-Barr virus can bind to C1QBP on the surface of T
lymphocytes and inhibit the proliferation of T cells, thus exerting
immunosuppressive effects (12,24,25).
Plasmodium falciparum infected red blood cells can not only
use C1QBP as a receptor to bind to human endothelial cells but also
bind to C1QBP on platelets to form clumps, thereby mediating
vascular endothelial cell proliferation. These two factors are the
key mechanisms of malaria pathogenesis (26). In summary, C1QBP is related to
humoral immunity, coagulation and fibrin polymerization as well as
microbial infection, which is summarized in Fig. 2.
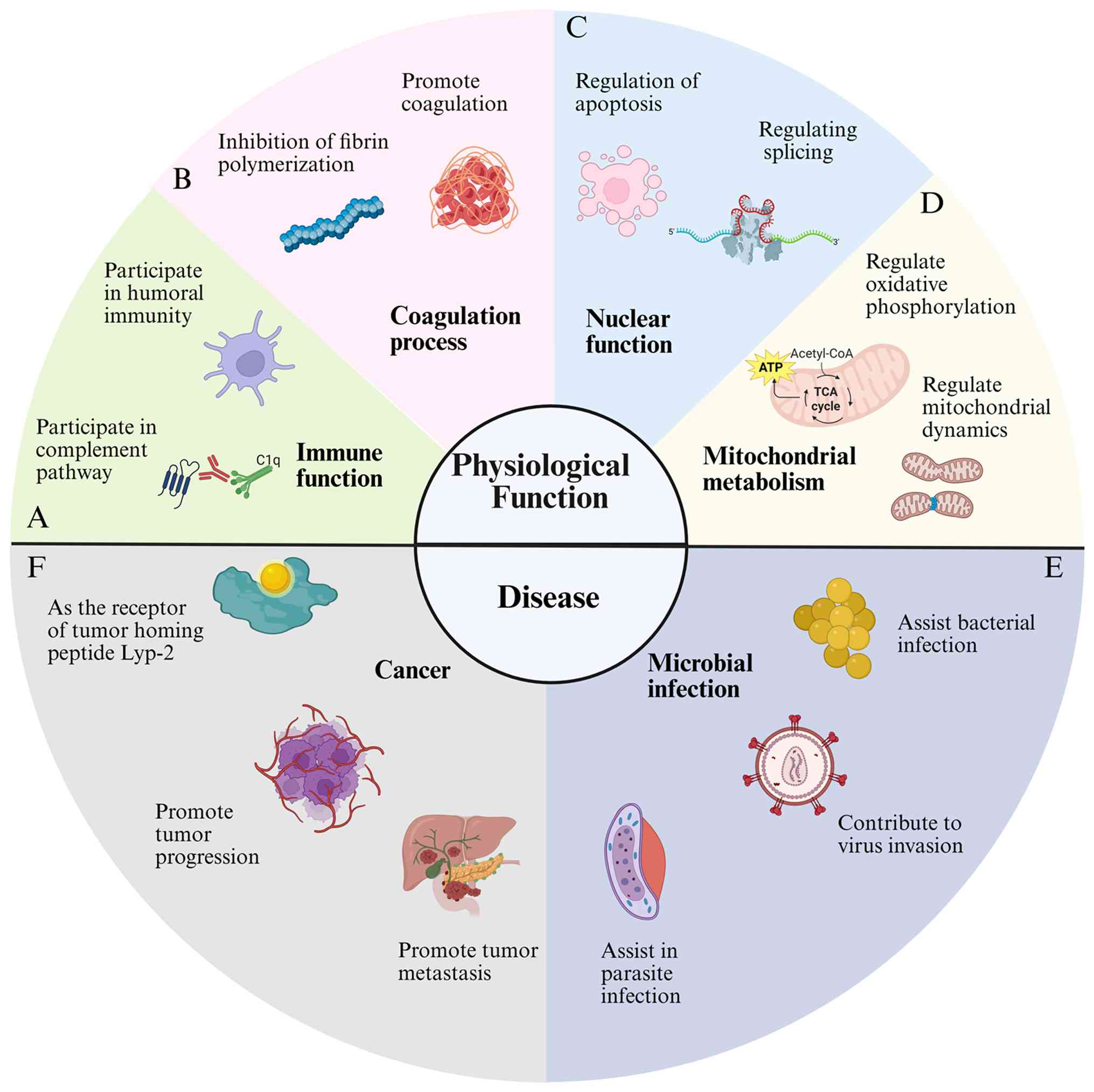 | Figure 2C1QBP can interact with a number of
ligands with different properties and positions. (A) C1QBP is
involved in the regulation of immune function. C1q is the initial
molecule of the classical complement pathway, which can recognize
immune complexes and initiate the classical pathway of the
complement system (12). (B) C1QBP
can promote the coagulation process by inhibiting fibrin
polymerization (20). (C) C1QBP is
involved in the regulation of nuclear function; it can not only be
used as a regulator of apoptosis, but also regulates the splicing
process through the interaction with various splicing factors and
modulating their function (8). (D)
C1QBP is involved in the regulation of mitochondrial metabolism.
C1QBP plays a vital role in the biosynthesis of proteins encoded by
the mitochondrial genome, especially in maintaining oxidative
stress (20). C1QBP has also been
found to affect mitochondrial metabolism and kinetic processes in
recent years (16). (E) C1QBP
promotes the occurrence of microbial infection. The process of
Staphylococcus aureus protein A recognizing platelet C1QBP
is key to the pathogenesis of S. aureus (23). HCV, HIV and other virions can bind
to C1QBP on the surface of T lymphocytes and inhibit the
proliferation of T cells, thus exerting immunosuppressive effects
(24). Plasmodium
falciparum infected red blood cells can not only use C1QBP as a
receptor to bind to human endothelial cells but also bind to C1QBP
on platelets to form clumps, thereby mediating vascular endothelial
cell proliferation. These two factors are the key mechanisms of
malaria pathogenesis (1). (F)
C1QBP is involved in the occurrence, progression, metastasis and
prognosis of cancer and can also be used as a molecular target for
cancer treatment (32,103). TCA cycle, tricarboxylic acid
cycle; C1QBP, complement component 1 Q subcomponent-binding
protein; HVC, hepatitis C virus; HIV, human immunodeficiency
virus. |
As aforementioned, the unique structural features of
C1QBP allow it to interact with several different ligands and
regulate their functions. At present, the main ligands identified
are C1q, calreticulin, hyaluronic acid (HA), the splicing factor
ASF/SF2 and CD44(27). C1QBP was
initially identified as a receptor for C1q in the study by
Ghebrehiwet et al (12),
and initiates organismal immunity by activating the classical
complement pathway. The binding of C1QBP to calreticulin is related
to the adhesion and spreading of endothelial cells (28). Furthermore, the binding of C1QBP to
HA promotes the adhesion and de-adhesion of cells (29). The binding of C1QBP to ASF/SF2
blocks ASF/SF2 phosphorylation and inhibits its binding to
pre-messenger RNA, thereby inhibiting its role as an initiator of
pre-spliceosome formation (30).
Besides, the interaction of C1QBP with CD44v6 in lipid rafts, a
cholesterol-rich and sphingomyelin-rich microstructural domain of
plasma membranes, promotes the phosphorylation of insulin-like
growth factor 1 receptor, thereby activating downstream PI3K and
MAPK signaling pathways that mediate the metastatic potential of
pancreatic cancer cells (31). In
summary, C1QBP is involved in regulating gene expression, splicing,
mitochondrial metabolism and cell signaling and plays a vital role
in tumor development.
The mitochondrial localization of C1QBP is
associated with multiple biological processes, including
mitochondrial morphology regulation, metabolic regulation, cell
fate determination and disease (32). Knockdown of C1QBP leads to abnormal
mitochondrial morphology and ultrastructural damage, indicating its
key role in maintaining the dynamic balance of the mitochondrial
network (15). C1QBP is a key
regulator of Y-box binding protein 1 (YBX1), enhancing the
expression of respiratory complexes, ATP production and reactive
oxygen species (ROS) generation through promoting the translation
of mitochondrial gene-encoded proteins (33). The mitochondrial dysfunction of
C1QBP is also closely related to cancer, including gynecological
cancer (Table I).
 | Table ISummary of the clinicopathological
factors associated with C1QBP expression. |
Table I
Summary of the clinicopathological
factors associated with C1QBP expression.
| First author,
year | Tumor type | Sample size | Testing method | Relevant indicators
related to C1QBP | (Refs.) |
|---|
| Zhang et al,
2017 | Cervical
cancer | 30 cervical
intra-epithelial neoplasia and 118 CC specimens compared with 10
normal cervical specimens |
Immunohistochemistry | FIGO stage, poor
histological grade, large tumor volume, lymphatic vessel
interstitial infiltration, deep interstitial infiltration, lymph
node metastasis and disease-free survival | (3) |
| Yu et al,
2013 | Ovarian cancer | 161 patients with
epithelial ovarian cancer |
Immunohistochemistry and western
blotting | FIGO stage,
peritoneal dissemination, lymph node metastasis, degree of tissue
differentiation and residual tumor size | (4) |
| Zhao et al,
2015 | Endometrial
cancer | 188 endometrial
cancer specimens, 43 benign endometrial lesion specimens and 41
normal endometrium specimens |
Immunohistochemistry and western
blotting | FIGO stage, high
histological grade, deep myometrial infiltration, lymphatic vessel
interstitial infiltration, lymph node metastasis and recurrence,
disease-free survival and overall survival | (5) |
3. Role of C1QBP in cancer
C1OBP is highly expressed in epithelial tumors of
the breast, lung, liver, prostate and colon, as well as in squamous
and basal cell carcinoma (34-39).
The expression level of C1QBP is also increased in inflammatory
lesions and cells with a high cell division rate (32). The data accumulated so far indicate
that C1QBP is upregulated in most cancer cells and it not only
participates in shaping the inflammatory tumor microenvironment,
but also regulates mitochondrial metabolism and dynamics,
angiogenesis, metastasis and cell proliferation (40). Moreover, C1QBP enhances
anti-apoptotic ability and induces resistance to cisplatin
(41). The key regulatory roles of
C1QBP in different physiological and pathological processes are
summarized in Fig. 2.
Tumor migration and invasion
Cell migration is the hallmark of cancer, regulating
the anchorage-independent growth and invasiveness of tumor cells
(42). C1QBP plays a crucial role
in the growth, survival, metastasis and invasion of tumor cells.
The expression level of C1QBP is associated with the stage, grade,
size and clinical prognosis of the tumor (3-5).
In breast cancer, the upregulation of C1QBP is significantly
associated with distant metastasis, higher TNM stage, larger tumor
volume, axillary lymph node metastasis and poor prognosis (43,44).
Furthermore, when C1QBP is absent, the proliferation, migration and
invasion of liver cancer cells is reduced, while cell apoptosis
increases (45). C1QBP induces
tumor cell migration and tumor growth by interacting with integrin
and activating matrix metalloproteinase-2 through a nuclear
factor-κB-dependent mechanism (32,46).
C1QBP interacts with YBX1, inhibiting the activation of YBX1 by
altering its phosphorylation and nuclear transport (47). This inhibition of YBX1 leads to the
suppression of its activity in renal cell carcinoma cells,
potentially preventing the migration and invasion of renal cell
carcinoma (48). The inhibition of
YBX1 might involve altering the matrix metalloprotease 9 signaling
pathway (48). Besides, C1QBP
promotes melanoma progression and metastasis by targeting
epithelial mesenchymal transition markers, the Akt/protein kinase B
pathway and the tumor microenvironment (49,50).
Additionally, the enhanced migration, invasion and proliferation of
oral squamous cell carcinoma cells in vitro is dependent on C1QBP
(51). In conclusion, C1QBP
regulates tumor growth and invasion through multiple pathways
(40). In cancer cells, C1QBP
further enhances OXPHOS activity by stabilizing proteins such as
PA28y, thereby supporting the energy and biosynthesis required for
tumor growth, and high expression of C1QBP promotes malignant
progression by regulating cell proliferation, migration, apoptosis
and differentiation, whereas its inhibition can induce apoptosis
and enhance glycolysis (52).
Mitochondrial metabolism and
dynamics
Studies have shown that the absence of the C1QBP
gene activates the overlapping activity with m-AAA protease,
thereby cleaving optic atrophy 1, leading to mitochondrial division
and swelling, accompanied by altered expression of Mitofusin1 and
Mitofusin2 (53,54). C1QBP is also crucial for
maintaining metabolic homeostasis and stress responses; it supports
cell survival during nutrient deprivation by facilitating
autophagic flux and preserving mitochondrial OXPHOS function,
processes vital for adaptation to starvation (55). The tumor occurrence mediated by
C1QBP is closely related to the maintenance of mitochondrial
metabolism. Reducing the expression of C1OBP in human cancer cells
significantly alters their metabolic patterns, reprogramming
metabolism from OXPHOS to glycolysis (56,57).
The absence of C1QBP leads to a reduced ability of tumors to form
within the body, which may be due to a decrease in the synthesis of
OXPHOS proteins encoded by mitochondrial DNA. Glutamine is an
important substrate for mitochondrial metabolism. The higher the
degree of tumor progression, the greater the amount of glutamine
oxidation in the body, reflecting an increased demand for
mitochondrial anaplerosis to fuel the tricarboxylic acid (TCA)
cycle. This heightened state of glutamine metabolism stands in
stark contrast to the metabolic state observed upon C1QBP loss,
where impaired OXPHOS and a shift toward glycolysis suggest an
inability to effectively utilize such mitochondrial substrates
(58). Myc upregulates the
transcription of C1QBP, thereby regulating glutamine metabolism
(45). In malignant brain cancer,
a high level of Myc is accompanied by an increase in C1QBP
expression (59,60). Research indicates the
proto-oncogene c-Myc directly regulates C1QBP expression at the
transcriptional level. First identified in cholangiocarcinoma, this
mechanism involves c-Myc binding to the C1QBP promoter to activate
its transcription, subsequently driving tumor proliferation
(45). The mitochondrial
dysfunction of C1QBP is closely related to cancer progression. For
example, in triple-negative breast cancer (TNBC), targeting C1QBP
can induce mitochondrial damage and tumor regression; its
expression level is related to the prognosis of patients, making it
a potential target for cancer treatment (41). Studies have shown that C1QBP is
highly expressed in gynecological tumor tissues and may be used as
a biomarker to predict the progression and prognosis of gynecologic
tumors (3-5).
However, few studies have investigated the specific mechanism of
C1QBP in gynecological tumors, and the role of C1QBP in these tumor
types requires further experimental and clinical validation.
Furthermore, C1QBP has been demonstrated to be
associated with mitochondrial-dependent tumor cell apoptosis.
Knockdown of C1QBP enhances mitochondrial fragmentation,
accompanied by the loss of detectable levels of mitochondrial
fusion intermediates Mitofusin1/2(53). Proteasome activator and C1QBP are
co-localized in mitochondria and promote mitochondrial fusion by
upregulating optic atrophy 1, Mitofusin1/2, respiratory complex
expression, OXPHOS, ATP production and ROS generation. Mitochondria
are highly dynamic organelles that constantly undergo division and
fusion (61,62).
Immunoregulation
C1QBP is related to the immune process during the
occurrence and development of tumors, such as the regulation of T
cells and macrophages, as well as the proliferation and invasion of
tumors (52,58,59).
Studies have shown that C1QBP also participates in regulating the
differentiation of effector CD8+ T cells, thereby
influencing their antiviral and antitumor immune responses
(56,63,64).
In immune cells, the absence of C1QBP intrinsically inhibits
effector CD8+ T cell differentiation by impairing
mitochondrial respiratory capacity and increasing sensitivity to
defective OXPHOS, leading to metabolic reprogramming, epigenetic
disorders and a subsequent disruption of the antitumor immune
response (56). Knocking out the
C1QBP protein can regulate T cells by impairing mitochondrial
fitness, leading to the accumulation of ROS and the loss of
mitochondrial membrane potential, thereby compromising
mitochondrial health and function (56). The absence of C1QBP weakens the
infiltration of T cells into tumors and exacerbates the exhaustion
of tumor-infiltrating T lymphocytes (65,66).
Furthermore, C1QBP is also related to the regulation
of macrophages (6). The
vitronectin secreted by tumor cells interacts with C1QBP located on
the surface of tumor-associated macrophages, inhibiting the
phagocytic activity of tumor cells and causing the macrophages to
transform into the M2 subtype in the tumor microenvironment
(67). Mechanistically, the
vitronectin-C1QBP axis promotes Shp1 uptake induced by
FcyRIIIA/CD16, thereby reducing the phosphorylation of Syk
(67). This reduction in Syk
phosphorylation impairs downstream phagocytic signaling, ultimately
inhibiting the phagocytic activity of macrophages against tumor
cells. Tumor cell-derived vitronectin (Vin) suppresses phagocytosis
through its interaction with C1QBP on the macrophage surface
(63). Moreover, the combination
of Vin knockdown and the anti-CD47 antibody effectively enhances
the phagocytic activity and infiltration of macrophages, thereby
reducing tumor growth in vivo. Further studies have shown
that C1QBP may promote the polarization of macrophages from
macrophages to M2 and M3 types (67,68).
Mitochondrial function not only affects the occurrence, progression
and distant metastasis of tumors, but also regulates the
maturation, differentiation and long-term maintenance of immune
cells.
Angiogenesis
The secreted form of C1QBP can upregulate the
expression of the inducible bradykinin receptor I through autocrine
action, thereby exacerbating vascular leakage and opening channels
within the cells, allowing tumor cells to escape and metastasize to
distant sites for metastatic invasion (6,69).
Proliferating tumor cells rely on angiogenesis to maintain
survival, development and escape to secondary sites for metastatic
invasion. Multiple studies have confirmed that the hypoxic tumor
microenvironment plays a role in promoting angiogenesis (27,70-72).
The expression level of C1QBP is higher in the hypoxic regions of
tumors, indicating that the tumor microenvironment is associated
with the expression level and function of C1QBP in cancer (34). Additionally, hypoxia induces an
increase in the C1QBP expression level in TNBC cells through the
hypoxia-inducible factor α transcription factor (69,73).
The neutralizing effect of anti-C1QBP antibodies on C1QBP located
on the cell surface also inhibits angiogenesis, as this anti-C1QBP
antibody prevents the activation of receptor tyrosine kinase
stimulated by growth factors, the formation of pseudopodia, cell
migration and the formation of capillary tubes (74).
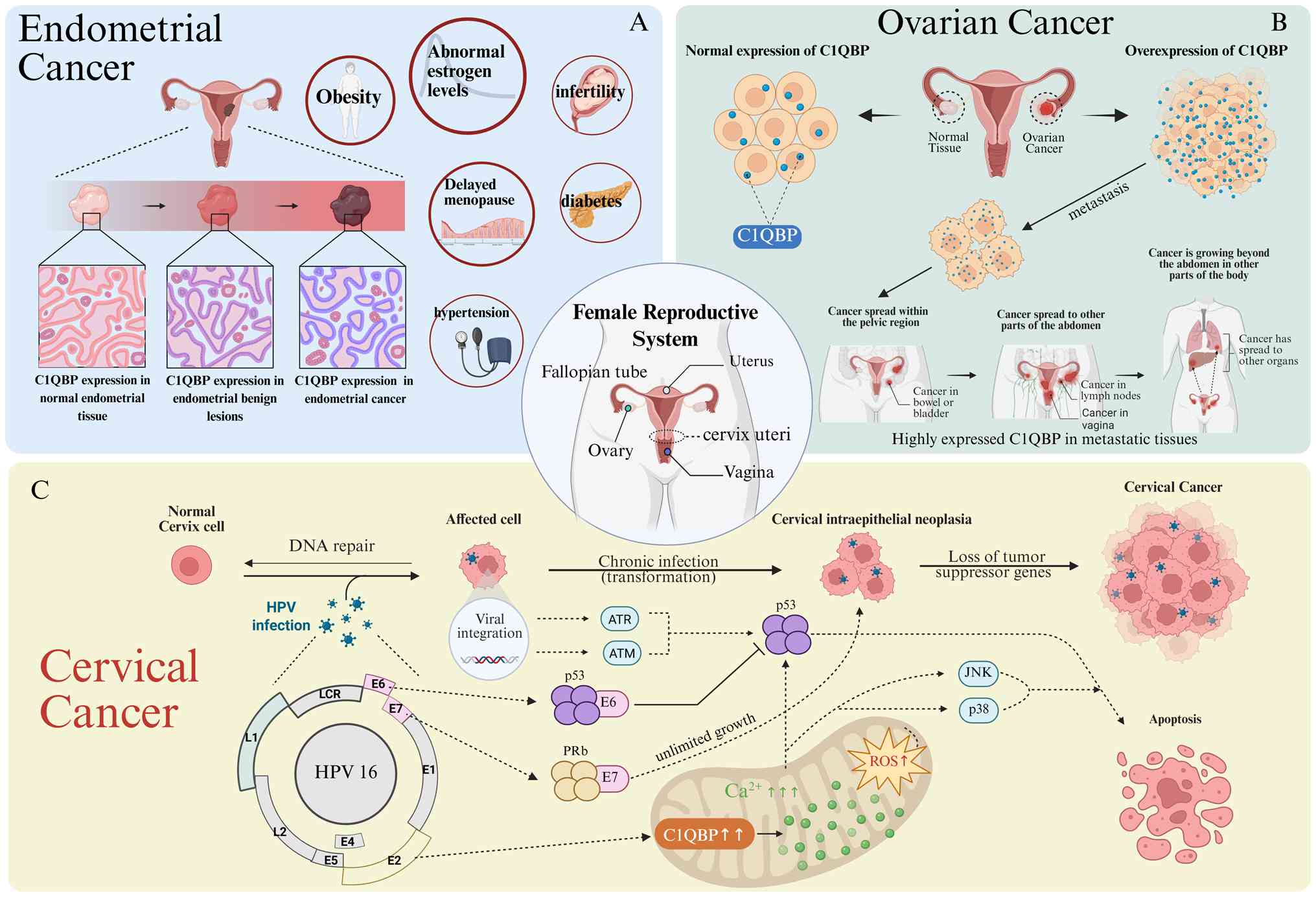
4. C1QBP and gynecological tumors
C1QBP and cervical cancer
The incidence of cervical cancer has declined
slightly over the past decade, but it remains the second most
common cause of cancer-related death among women in developing
countries (75). The primary
causative agent of cervical cancer is oncogenic human
papillomavirus (HPV), of which HPV 16 and HPV 18 are predominant
(76). Identifying genes involved
in cervical carcinogenesis and progression may help elucidate the
underlying molecular mechanisms and provide valuable therapeutic
targets.
Current research progress on cervical cancer and
C1QBP has conflicting results. Some studies have shown that C1QBP
is associated with the promotion of cervical cancer development,
while others have indicated that C1QBP is related to the inhibition
of cervical cancer. Zhang et al (3) first reported that C1QBP is
upregulated in cervical tumor tissues and that the degree of
elevated expression is associated with advanced international
Federation of Gynecology and Obstetrics (FIGO) stage (P=0.001),
poor histological grade (P=0.013), large tumor volume (P=0.025),
lymphatic vessel interstitial infiltration (P=0.024), deep
interstitial infiltration (P=0.001) and lymph node metastasis
(P=0.023). This study also demonstrated that elevated C1QBP
expression is an independent predictor of disease-free survival
(P=0.007) in patients with cervical cancer via multivariate Cox
regression analysis. Due to insufficient sample size, more
experimental results should be obtained with larger sample sizes in
future to obtain more representative conclusions.
Conversely, another study has shown that, compared
with normal cervical tissue, the expression of C1QBP protein in
human cervical squamous cell carcinoma tissue is significantly
decreased (3). C1QBP is associated
with the apoptosis, survival, migration and proliferation of
cervical squamous cell carcinoma cells (3). Relevant pathways have also been
explored in existing studies. After transfection of C33a cells with
the C1QBP overexpression vector, the apoptosis rate and the
expression of p38 mitogen-activated protein kinase (p38 MAPK)
significantly increased. Moreover, when C1QBP was overexpressed,
the changes in the survival rate, migration ability and
proliferation ability of C33a cells could be eliminated with the
p38 MAPK inhibitor SB202190(77).
Chen et al (78) reported
that when C1QBP is overexpressed, Ca2+ overload in
mitochondria, increased ROS and decreased mitochondrial membrane
potential lead to reduced cell viability, which leads to the
induction of apoptosis, confirming the involvement of C1QBP in the
p53-dependent apoptotic pathway. Moreover, the involvement of C1QBP
in inducing apoptosis in cervical cancer cells via another pathway,
p38 MAPK/JNK, was also identified in a study by Gao et al
(79). Itahana and Zhang (80) and Liu et al (81) also reported that apoptosis was
increased when C1QBP was overexpressed in cervical tumor tissues.
These data indicate that C1QBP inhibits the survival, migration and
proliferation of cervical squamous cell carcinoma cells through the
p38 MAPK signaling pathway (79).
A persistent infection of high-risk HPV type has
been correlated with the development of cervical cancer (78). The HPV 16 genome is composed of six
regulatory proteins, composed of E1, E2, E4, E5, E6 and E7, which
regulate the life cycle, gene expression and the function of
cervical cancer. HPV 16 E2 induces the apoptosis of tumor cells and
inhibits tumor growth in cervical cancer by increasing the
expression of C1QBP. Compared with non-cancerous cervical samples,
the expression levels of the HPV-16 E2 and C1QBP genes were
significantly decreased in human cervical squamous cell carcinoma
samples (78). In C33a and SiHa
cells transfected with a vector encoding HPV-16 E2, the expression
of the C1QBP gene significantly increased, which was accompanied by
mitochondrial dysfunction and the upregulation of apoptosis;
transfecting with C1QBP small interfering RNA (siRNA) could limit
these phenomena. These data support a mechanism in which C1QBP
plays an important role in the HPV-16 E2-induced apoptosis of human
cervical squamous cell carcinoma cells through a
mitochondrial-dependent pathway (78). The C33a and SiHa cells transfected
with the HPV16 E2 vector also showed a significant increase in the
activation of p38 MAPK/JNK. Moreover, the changes in the survival
rate, migration ability and proliferation ability of C33a cells
after HPV 16 E2 transfection were eliminated after treatment with
the p38 MAPK inhibitor SB203580 and the JNK inhibitor SP600125.
These data support the mechanism that HPV16 E2 induces cell
apoptosis by silencing the C1QBP gene or inhibiting the p38
MAPK/JNK signaling pathway in cervical squamous cell carcinoma
(78). Another study revealed that
HPV E2 could induce apoptosis in cervical cancer cells through the
upregulation of C1QBP (81).
The E6 and E7 proteins encoded by high-risk HPV
types are also associated with the function of C1QBP in cervical
cancer, and their functions are different from those of the
aforementioned E2 type. Under the influence of the E6 and E7
proteins encoded by high-risk HPV, C1QBP promotes the immune escape
of cervical cancer cells (79).
Gao et al (79) examined
C1QBP protein levels in C-33A cells transfected with or without the
HPV-16 E6 and E7 oncogenes and reported that C1QBP protein
expression was significantly lower in HPV-16 E6- and E7-treated
C-33A cells than in untreated C-33A cells. The excessive expression
of C1QBP in cells induces apoptosis, mediated by the activation of
caspase-3 and the onset of mitochondrial dysfunction. Cells
transfected with the GFP-C1QBP vector showed increased expression
of C1QBP protein and a gradual increase in ROS production.
Additionally, the increase in ROS production and Ca2+
influx in mitochondria led to the loss of mitochondrial
transmembrane potential. Notably, when C1QBP was overexpressed in
C-33A cells and the cells were treated with metformin, cell
apoptosis was significantly inhibited, which may be due to the
protection of mitochondrial function by metformin (79). These data suggest that C1QBP may
play an important role in the immune escape of cervical cancer
triggered by HPV-16 E6 and E7, depending on its expression level
and cellular subcellular localization. Specifically, E6 and E7
downregulate C1QBP expression, thereby relieving its pro-apoptotic
effect, which is mediated by caspase-3 activation, ROS production
and mitochondrial dysfunction. This suppression of C1QBP-dependent
apoptosis allows HPV-infected cells to evade immune clearance and
persist, contributing to cervical carcinogenesis. Rangsee et
al (13) reported that HPV-E7
can also inhibit C1QBP by increasing C1QBP promoter methylation in
HPV-positive cervical cancer cell lines and decreasing its protein
abundance, which ultimately promotes cervical carcinogenesis.
HPV-16 is closely associated with the occurrence of 50% of cervical
cancer cases (79).
In summary, C1QBP is associated with the apoptosis,
survival, migration and proliferation of cervical squamous cell
carcinoma cells. There is a close link between HPV-related proteins
and C1QBP. C1QBP has context-dependent conflicting roles in
HPV-associated cervical cancer, functioning as an anti-tumorigenic
factor in the presence of HPV E2 and as a pro-tumorigenic factor in
the presence of high-risk HPV E6 and E7 oncogenes. These findings
provide new ideas for the identification of new therapeutic targets
for the treatment of human cervical cancer. However, the specific
mechanism involved is still unclear and needs to be further
verified by a number of basic experiments. Besides, although
previous cervical cancer-related studies have analyzed cervical
cancer samples, the statistical power of stratified analyses, such
as different histological types or clinical stages, may be
insufficient, which leads to the results being susceptible to
confounding factors and difficulties verifying their universality.
The clinical significance of C1QBP in cervical cancer is mostly
based on data from single centers, lacking independent multi-center
cohort verification. Moreover, the application of functional
experiments in cervical cancer cell lines is limited, which
restricts the ability of causal inference. At present, experiments
have been largely limited to a few cell lines, such as C-33A and
SiHa, making it difficult to generalize the conclusions to cervical
cancer of all HPV subtypes and genetic backgrounds. Besides, the
inconsistent results of C1QBP in cervical cancer research may be
caused by small sample size, different population characteristics,
inconsistent tissue preservation methods, differences in detection
methods, data collection biases, short follow-up time or
insufficient control of confounding factors. At present, there are
relatively few studies examining the mechanistic pathways and
clinical research between C1QBP and cervical cancer; therefore,
further research and discussion are needed.
C1QBP and ovarian cancer
Among the gynecological malignancies, ovarian cancer
is the most common cause of death (82). Despite patients receiving advanced
tumor cytoreduction and chemotherapy, 5-year survival rates still
remain low. The use of multiple biomarkers is an accurate method
for preoperatively predicting the risk of ovarian cancer in
patients with metastases. Therefore, testing for new biomarkers may
improve the sensitivity and specificity of predicting disease.
Yu et al (4)
reported that C1QBP upregulation in epithelial ovarian cancer was
associated with FIGO stage (P=0.0074), peritoneal dissemination
(P<0.0001), lymph node metastasis (P<0.0001), degree of
tissue differentiation (P<0.0001) and residual tumor size
(P=0.0013). They collected paraffin-embedded specimens from 161
patients with epithelial ovarian cancer (EOC) and found that C1QBP
was lowly expressed in benign ovarian tissues via western blot
analysis. Low levels of C1QBP expression were also detected in the
tissues of patients without peritoneal and lymph node metastasis;
however, high levels were detected in the tissues of patients with
peritoneal and lymph node metastasis. The same results were
confirmed by immunohistochemical staining. Moreover, within the
same patients, the intensity and frequency of C1QBP staining
increased from the primary lesion to peritoneal dissemination and
lymph node metastasis. Therefore, it was noted that elevated C1QBP
expression may be a new indicator of lymph node and peritoneal
metastasis in patients with EOC. Besides, regarding the differences
in expression and function of C1QBP in different pathological
subtypes such as serous ovarian cancer and endometrioid ovarian
cancer, current research is limited and there are no published data
for reference. This is also a promising research direction.
In a study by Yu et al (83), C1QBP was found to be only weakly to
non-expressed in all normal ovarian tissues, highly expressed in
plasmacytoid ovarian cancer tissues, more highly expressed in
poorly differentiated plasmacytoid ovarian cancer tissues than in
moderately differentiated ovarian cancers (P=0.0182) and
significantly elevated in patients with lymph node metastasis
(P=0.0021). Univariate analysis revealed that C1QBP upregulation
was a significant predictor of poor progression-free survival and
overall survival (83). Due to
limitations such as insufficient sample size and the regional
nature of sample collection, the reliability of these findings
needs to be further validated in larger, multi-center cohorts.
Duan et al (84) have designed a novel C1QBP probe
based on 99mTc-radiolabeled siRNA, which can non-invasively detect
C1QBP expression in vivo, thus enabling the early diagnosis
of ovarian cancer. Hunt et al (85) also reported that C1QBP can be
inhibited by the intraperitoneal injection of linTT1 peptide-guided
proapoptotic nanoparticles to treat peritoneal metastatic cancer
caused by ovarian cancer dissemination.
Moreover, C1QBP also has a role in the drug
resistance of ovarian cancer cells. Yu et al (4) reported that the expression of C1QBP
was increased in cisplatin-resistant ovarian cancer and that the
staining intensity and frequency in chemotherapy-resistant ovarian
tumor tissues were significantly greater than that in
chemotherapy-sensitive ovarian tumor tissues.
In conclusion, C1QBP is closely related to lymph and
lesion metastasis in ovarian cancer, which is a research direction
that can be further explored in the future. Further studies are
needed to confirm whether the C1QBP protein has the potential to
serve as a predictive marker for the clinical progression of
ovarian cancer alone or in combination with other markers. In
addition, the detailed mechanism by which increased C1QBP
expression leads to ovarian cancer metastasis still remains unclear
and should be further explored in future basic studies and clinical
trials.
C1QBP and endometrial cancer
The incidence of endometrial cancer has been
increasing worldwide in recent year (86). In total, 80% of endometrial cancer
cases are estrogen-related and the pathological type is
predominantly endometrioid, with early-stage patients tending to
have an improved prognosis and patients with advanced, recurrent or
high-grade lesions having a poor prognosis (87,88).
However, few therapeutic agents are available to achieve durable
remission in patients with recurrent or advanced disease.
Therefore, developing novel molecular biomarkers that may be
involved in the molecular mechanisms of endometrial carcinogenesis
and progression is imperative.
Rubinstein et al (89) examined C1QBP expression in
endometrial cancer via immunohistochemical staining and reported
that no significant differential expression of C1QBP was detected
compared with non-malignant tissues of the same histology. However,
another study noted that, compared with normal endometrium, C1QBP
was more highly expressed in endometrial cancer and endometrial
benign lesions and that the expression of C1QBP in endometrial
carcinoma was significantly greater than that in endometrial benign
lesions. In the study, high C1QBP expression was significantly
associated with advanced International Federation of Gynecology and
Obstetrics stage (P=0.019), high histological grade (P<0.001),
deep myometrial infiltration (P=0.013), lymphatic vessel
interstitial infiltration (P=0.010), lymph node metastasis
(P=0.015) and recurrence (P=0.009). The results of the multivariate
Cox regression analysis revealed that the C1QBP expression status
was an independent prognostic factor for disease-free survival
(P=0.022) and overall survival (P=0.025) in patients with
endometrial cancer (5). Due to the
limitations of the sample size and its single-region nature,
further research is needed to reach a more definite conclusion. The
research on C1QBP in endometrial cancer may have a relatively small
sample size and insufficient statistical power, which limits the
generalizability of the results. Moreover, if the data comes from a
single medical institution, it may not be representative of the
heterogeneity of a multi-center population. The results may not be
representative of other populations with different environmental
exposures. Therefore, large-scale, multi-center studies with
diverse geographic cohorts are warranted to validate the
independent prognostic value of C1QBP in endometrial cancer.
Furthermore, since the patient groups were formed based on
historical data rather than random allocation, the baseline
characteristics, such as age, molecular subtype and treatment
regimen, may be unbalanced, which could potentially confound the
effect estimation. Comorbidities, treatment intensity or hormone
receptor status may lead to the association between C1QBP and the
outcome being partially attributed to unmeasured factors.
There are similar pathological and physiological
connections among obesity, diabetes and endometrial cancer. The
occurrence of endometrial cancer is related to factors such as
hormonal level changes, metabolic abnormalities and chronic
inflammation. Obese women have higher levels of estrogen in their
bodies, which may stimulate the abnormal proliferation of
endometrial cancer cells (85).
Endometrial cancer also has an association with hypertension,
mainly related to the shared metabolic abnormalities such as
obesity and insulin resistance (90-92).
The long-term chronic inflammation and hormone imbalance in the
body of patients with hypertension may indirectly increase the risk
of endometrial cancer, but the specific mechanism still needs
further research (93). Moreover,
women who experience menopause later in life have a relatively
higher risk of endometrial cancer (94). The mechanism of endometrial cancer
and C1QBP protein is still poorly understood at present. It is well
established that estrogen and metabolic disorders are risk factors
for endometrial cancer, and that C1QBP plays a central role in
mitochondrial metabolism and tumorigenesis (95,96).
Moreover, indirect evidence suggests that estrogen can regulate
C1q, the primary ligand of C1QBP (97-99).
Estrogen signaling and metabolic disorders such as obesity and
diabetes may be associated with the potential role of C1QBP in the
occurrence of endometrial cancer; however, there are no direct
experimental data to support this conclusion at present and it
should be regarded as a knowledge gap. Diabetes is an important
risk factor for endometrial cancer in women and it has a negative
impact on the survival rate and prognosis of patients. Molecular
mechanisms such as the insulin-like growth factor family, oxidative
stress and inflammatory cytokines have been considered to explain
this relationship (100). C1QBP
is involved in the regulation of OXPHOS and glucose oxidation
(16). The absence of C1QBP is
associated with resistance to age-related and diet-induced obesity
(101). C1QBP promotes lipid
biosynthesis by regulating fatty acid-induced endoplasmic reticulum
stress (16,101). C1QBP interacts with the
endoplasmic reticulum-anchored enzyme mannosyl-oligosaccharide
glucosidase I (MOGS) (100,102). We hypothesize that the C1QBP-MOGS
interaction represents a novel mitochondria-endoplasmic reticulum
coordination mechanism, enabling cells to synchronize energy
production (via mitochondrial OXPHOS) with protein synthesis and
folding (via endoplasmic reticulum glycosylation). This
coordination is particularly critical in rapidly proliferating
endometrial cancer cells, which require both robust energy supply
from mitochondria and extensive protein production from the
endoplasmic reticulum. The findings highlight important directions
for future research aimed at elucidating the precise molecular
mechanisms by which C1QBP exerts its diverse biological functions.
Few studies have explored the relationship between endometrial
cancer and C1QBP and numerous basic studies are needed to further
demonstrate the association between C1QBP and endometrial cancer
and its specific molecular mechanisms.
5. Clinical applications of C1QBP in
cancer
C1QBP, as a key protein highly expressed in various
types of cancer, has demonstrated potential in clinical
applications as a diagnostic biomarker, prognostic predictor and
therapeutic target. C1QBP is involved in the occurrence,
progression, metastasis and prognosis of cancer and can also be
used as a molecular target for cancer treatment (32,103). C1QBP is upregulated in various
tumor tissues and its high expression level is significantly
associated with poor prognosis in patients. For instance, in oral
squamous cell carcinoma, the expression levels of C1QBP and PA28y
are positively correlated and their high co-expression indicates a
poor prognosis (104).
Additionally, the mRNA expression level of C1QBP is significantly
higher in TNBC tissues than normal tissues and its high expression
is associated with a shortened disease-free survival period
(105). Moreover, a C1QBP
expression change may serve as an indicator of disease progression.
For instance, in pancreatic cancer, the level of the exosomal
CD44v6/C1QB complex can predict the risk of liver metastasis
(40). Furthermore, C1QBP can also
serve as an indicator for peritoneal and lymph node metastasis in
EOC. In a previous study, among the 89 patients whose primary tumor
immunohistochemical staining showed high expression levels of
C1QBP, 95.5% had peritoneal metastasis and 48.3% had lymph node
metastasis (12). Univariate and
multivariate logistic regression analyses revealed that
upregulation of C1QBP was associated with peritoneal dissemination
and lymph node metastasis in EOC.
C1QBP plays a crucial role in DNA damage repair. For
instance, in breast cancer, C1QBP regulates homologous combination
repair by stabilizing the MRE11 protein (41). C1QBP inhibition can enhance the
efficacy of chemotherapy, a mechanism that provides a new approach
for overcoming drug resistance. For example, combining a C1QBP
inhibitor with DNA damage drugs may improve the therapeutic effect
(106). Additionally, the role of
C1QBP in metabolic regulation, such as mitochondrial OXPHOS,
suggests that its targeted therapy may be applicable to tumors that
are metabolically dependent. C1QBP, frequently upregulated in
cancer, is secreted into the tumor microenvironment where it drives
metastatic progression via the complement and kinin systems.
Furthermore, C1QBP participates in a novel immune checkpoint axis;
cancer cell-surface C1QBP directly interacts with cytotoxic T cells
to suppress their activity, analogous to the established programmed
cell death ligand 1 and programmed cell death protein 1 pathway
(6).
Due to limitations such as insufficient sample size
and the regional nature of sample collection, subsequent
researchers can improve the experimental conclusion by increasing
the sample size. According to existing research, the high
immunohistochemical expression of C1QBP can serve as an indicator
of poor prognosis for ovarian, cervical and endometrial cancer
(3-5).
Measuring the level of C1QBP in serum or abdominal fluid may be
used for early diagnosis or recurrence monitoring. PDBAG1, a
peptide derived from the precursor protein of glycerol-3-phosphate
dehydrogenase 1, can directly bind to C1QBP and induce its
ubiquitin-dependent degradation (43). In the TNBC model, PDBAG1
significantly inhibits tumor growth and induces regression
(100). The LyP-1 peptide can
specifically bind to C1QBP on the surface of tumor cells and be
used for drug delivery (107).
Additionally, the green tea polyphenol epigallocatechin gallate can
bind to C1QBP and inhibit tumor growth (58). In addition, as a research target,
anti-C1QBP monoclonal antibodies are currently being developed to
block its tumor-promoting functions. C1QBP inhibitors combined with
chemotherapy, such as paclitaxel and cisplatin, may reverse drug
resistance. Studies have demonstrated the feasibility of the
chimeric antigen receptor (CAR)-T cell immunotherapy approach in
glioblastoma (108-110).
C1QBP is highly expressed on the surface of tumor cells and can be
used as a target antigen for immunotherapy, such as CAR-T and
peptide vaccines. At present, there are no direct CAR-T products
targeting C1QBP that have been approved. However, given its central
role in T-cell metabolism, enhancing the function of C1QBP or
maintaining mitochondrial health may become a strategy to overcome
drug resistance. Besides, at present, no C1QBP-based diagnostic or
therapeutic applications have entered clinical trials.
C1QBP is expressed in multiple intracellular
locations such as mitochondria, cytoplasm and cell membranes, and
its structural characteristics provide binding sites for small
molecule or peptide drugs. Regarding tumor specificity, C1QBP is
highly expressed in various tumor types, such as TNBC, oral
squamous cell carcinoma and acute myeloid leukemia, but is
expressed at a lower level in normal tissues (34,65,104). For instance, the mRNA level of
C1QBP in TNBC is significantly higher than that in adjacent normal
tissues, and its high expression is associated with poor patient
prognosis (34). This differential
expression may support the selectivity toxicity of targeted
therapy. Regarding the safety and toxicity of C1QBP drugs, current
studies show that targeting C1QBP has acceptable safety in animal
models (43,100). For example, PDBAG1 inhibits the
growth of TNBC tumors without reporting significant toxicity
(43). However, C1QBP participates
in basic metabolic functions such as mitochondrial homeostasis in
normal cells and the potential toxicity needs further evaluation.
The overall safety of C1QBP-targeted drugs, involving preclinical
toxicity, clinical safety and the therapeutic window, still need to
be verified through preclinical and clinical trials.
6. Summary and outlook
The present review summarized the research progress
of C1QBP in cervical, ovarian and endometrial cancer (Fig. 3). Early gynecological tumors often
have no notable symptoms or signs, but serum tumor markers, such as
the now familiar biomarkers CA125 and HE4, are elevated to varying
degrees (111,112). Tumor-related markers have
important guiding significance for the early screening of
gynecological tumors, adjuvant diagnosis, localization and
diagnosis, therapeutic efficacy evaluation, prognosis assessment
and targeted therapies. Numerous studies have demonstrated the
potential of targeting C1QBP to treat various tumor types (6,43,65,82,113). C1QBP affects the proliferation
and metastasis of gynecological tumors and has a certain impact on
drug resistance. However, the future of C1QBP in clinical use as a
diagnostic and prognostic marker for gynecological tumors still
needs further basic experiments and clinical trials for
verification.
Research on the role of C1QBP in gynecological
tumors is still in its initial stage and there are several
unresolved controversies and major research gaps. Firstly, there
are disputes regarding the expression and function of C1QBP in
gynecological tumors. For instance, in cervical cancer, the
expression level of the homologous subunit of C1QBP is related to
tumor progression, but the specific mechanism is still unclear and
the evidence is limited. Additionally, the expression pattern and
clinical significance of C1QBP in other gynecological tumors, such
as ovarian and cervical cancer, have not been systematically
studied. Different studies have differing opinions on whether C1QBP
can serve as an independent prognostic marker. For example, in
TNBC, high expression of C1QBP is associated with poor prognosis,
but similar validation is lacking in gynecological tumors.
Secondly, the molecular mechanism of C1QBP in gynecological tumors
has not been elucidated. Studies have shown that C1QBP promotes
tumor progression by regulating mitochondrial function, but the
applicability of this mechanism in gynecological tumors is unknown.
Therapeutic strategies targeting C1QBP face challenges. Although a
peptide inhibitor, PDBAG1, has been found to inhibit tumors by
inducing C1QBP degradation in breast cancer, the feasibility of
this strategy in gynecological tumors is unknown and there is
currently a lack of validation of the targeted effect in
gynecological tumor-specific models. Moreover, the limitations of
current research methods and data restrict progress. Current
evidence mostly originates from retrospective cohorts or public
databases, with small sample sizes and a lack of multicenter
validation.
C1QBP may promote viral replication by inhibiting
immune responses. For example, both hepatitis C virus (HCV) and
human immunodeficiency virus can inhibit or destroy T cells via
C1QBP, thus suppressing the body's immune response (84). The known causative virus of
cervical cancer is HPV, and it is not clear whether there is a
pathogenic effect between HPV and C1QBP similar to that between HIV
and HCV with C1QBP. HPV-associated proteins influence cervical
cancer development by affecting C1QBP. However, the exact mechanism
needs to be further explored. C1QBP maintains OXPHOS and its
absence increases glycolysis while impairing cell proliferation
in vitro and tumor growth in vivo (114). In addition, mitochondria seem to
play an intermediary role between C1QBP and gynecological tumors
and it has been shown that C1QBP affects gynecological tumor
development by regulating mitochondrial biogenesis and metabolic
processes (16). Thus, the use of
mitochondrion-targeted anticancer drugs mediated by C1QBP may
constitute a novel strategy for treating gynecological tumors in
the future. C1QBP may also be a potential molecular marker for
diagnosing gynecological tumors and a new indicator for predicting
the prognosis of this disease. Although C1QBP is highly expressed
in most tumor tissues, whether it can be used as a predictive
marker for the clinical progression of gynecologic tumors, either
alone or in combination with other markers, needs to be confirmed
by further studies.
C1QBP is a multifunctional protein that plays a
crucial role in mitochondrial function, cellular metabolism and
tumor progression. In the field of gynecological tumors, the
research on C1QBP is still in its early stages. Future studies on
C1QBP should focus on its molecular mechanisms and diagnostic and
therapeutic potential while combining multidisciplinary techniques
and cross-disciplinary collaborations to promote the translational
application of C1QBP in gynecological oncology. These studies will
not only help elucidate the pathogenesis of gynecological tumors in
detail but may also provide an essential basis for the development
of new diagnostic methods and therapeutic strategies. The main
research focus in the future should be concentrated on the
following aspects: i) Mechanism exploration: Deeply analyze the
regulatory mechanisms of C1QBP in mitochondrial dynamics such as
fusion and fission balance, OXPHOS activity and metabolic pathways
in gynecological tumor cells, especially its role in chemotherapy
resistance and the tumor microenvironment; ii) clinical relevance
analysis: Through databases such as The Cancer Genome Atlas and
clinical cohorts, the correlation between the expression level of
C1QBP and the prognosis of patients with gynecological tumors (such
as in terms of disease-free survival) should be clarified and its
potential as a biomarker should be evaluated; and iii) targeted
strategy development: Explore molecular tools for direct targeting
of C1QBP, such as peptide inhibitors or small molecule compounds,
and draw on the strategy of PDBAG1 in TNBC research, which inhibits
tumors by inducing the degradation of C1QBP, to optimize their
applicability in gynecological tumor models. The transformation
from basic research to clinical application needs to be carried out
step-by-step: i) Based on the structural characteristics of C1QBP,
such as the N-terminal functional domain, specific inhibitors can
be discovered through high-throughput screening or computational
simulation, and their killing effects on gynecological tumor cell
lines and organoid models can be evaluated; ii) combining
C1QBP-targeted drugs with existing therapies such as chemotherapy,
poly ADP-ribose polymerase inhibitors or immunotherapy, to overcome
drug resistance and improve efficacy, for example, by inhibiting
mitochondrial function to enhance oxidative stress-induced cancer
cell death; and iii) early clinical trials should be prioritized in
gynecological tumors with active mitochondrial metabolism, such as
ovarian cancer, while establishing biomarker detection methods to
screen potential patient groups that may benefit.
Acknowledgements
Not applicable.
Funding
Funding: This work was supported by the Sichuan Science and
Technology Program, China (grant no. 2022ZHYZ0012).
Availability of data and materials
Not applicable.
Authors' contributions
HZ contributed to conceptualization, writing the
original draft, reviewing and editing the manuscript and
visualization by designing and creating the mechanistic figures. JF
contributed to reviewing and editing the manuscript and
supervision. XZ, BL, TG and FF contributed to reviewing the
manuscript and supervision. JL contributed to reviewing the
manuscript, resources, supervision, funding acquisition and project
administration. PZ contributed to editing the manuscript,
resources, supervision and funding acquisition. All authors read
and approved the final version of the manuscript. Data
authentication is not applicable.
Ethics approval and consent to
participate
Not applicable.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Sung H, Ferlay J, Siegel RL, Laversanne M,
Soerjomataram I, Jemal A and Bray F: Global cancer statistics 2020:
GLOBOCAN estimates of incidence and mortality worldwide for 36
cancers in 185 countries. CA Cancer J Clin. 71:209–249.
2021.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Qiu H, Cao S and Xu R: Cancer incidence,
mortality, and burden in China: A time-trend analysis and
comparison with the United States and United Kingdom based on the
global epidemiological data released in 2020. Cancer Commun (Lond).
41:1037–1048. 2021.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Zhang M, Li N, Liang Y, Liu J, Zhou Y and
Liu C: Hyaluronic acid binding protein 1 overexpression is an
indicator for disease-free survival in cervical cancer. Int J Clin
Oncol. 22:347–352. 2017.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Yu H, Liu Q, Xin T, Xing L, Dong G, Jiang
Q, Lv Y, Song X, Teng C, Huang D, et al: Elevated expression of
hyaluronic acid binding protein 1 (HABP1)/P32/C1QBP is a novel
indicator for lymph node and peritoneal metastasis of epithelial
ovarian cancer patients. Tumour Biol. 34:3981–3987. 2013.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Zhao J, Liu T, Yu G and Wang J:
Overexpression of HABP1 correlated with clinicopathological
characteristics and unfavorable prognosis in endometrial cancer.
Tumour Biol. 36:1299–1306. 2015.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Ghebrehiwet B, Zaniewski M, Fernandez A,
DiGiovanni M, Reyes TN, Ji P, Savitt AG, Williams JL, Seeliger MA
and Peerschke EIB: The C1q and gC1qR axis as a novel checkpoint
inhibitor in cancer. Front Immunol. 15(1351656)2024.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Wang Q, Chai D, Sobhani N, Sun N, Neeli P,
Zheng J and Tian H: C1QBP regulates mitochondrial plasticity to
impact tumor progression and antitumor immune response. Front
Physiol. 13(1012112)2022.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Ghebrehiwet B, Lim BL, Peerschke EI and
Reid KB: Isolation, cDNA cloning, and overexpression of a 33-kD
cell surface glycoprotein that binds to the globular ‘heads’ of
C1q. J Exp Med. 179:1809–1821. 1994.PubMed/NCBI View Article : Google Scholar
|
|
9
|
George SA, Kotapalli V, Ramaswamy P, Kumar
R, Gowrishankar S, Uppin SG and Bashyam MD: Novel oncogenic
transcriptional targets of mutant p53 in esophageal squamous cell
carcinoma. J Cell Biochem. 125(e30534)2024.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Guo N, Weremowicz S, Lynch N, Lim BL,
Schwaeble W, Peerschke EI, Morton CC, Reid KB, Ghebrehiwet B and
Sastry KN: Assignment of C1QBP encoding the C1q globular domain
binding protein (gC1q-R) to human chromosome 17 band p13.3 by in
situ hybridization. Cytogenet Cell Genet. 77:283–284.
1997.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Jiang J, Zhang Y, Krainer AR and Xu R:
Crystal structure of human p32, a doughnut-shaped acidic
mitochondrial matrix protein. Proc Natl Acad Sci USA. 96:3572–3577.
1999.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Ghebrehiwet B, Geisbrecht BV, Xu X, Savitt
AG and Peerschke EIB: The C1q receptors: Focus on gC1qR/p33 (C1qBP,
p32, HABP-1)1. Semin Immunol. 45(101338)2019.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Rangsee N, Yanatatsaneejit P, Pisitkun T,
Somparn P, Jintaridth P and Topanurak S: Host proteome linked to
HPV E7-mediated specific gene hypermethylation in cancer pathways.
Infect Agent Cancer. 15(7)2020.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Sengupta A, Banerjee B, Tyagi RK and Datta
K: Golgi localization and dynamics of hyaluronan binding protein 1.
Cell Res. 15:183–186. 2005.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Hu M, Crawford SA, Henstridge DC, Ng IH,
Boey EJ, Xu Y, Febbraio MA, Jans DA and Bogoyevitch MA: p32 protein
levels are integral to mitochondrial and endoplasmic reticulum
morphology, cell metabolism and survival. Biochem J. 453:381–391.
2013.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Fogal V, Richardson AD, Karmali PP,
Scheffler IE, Smith JW and Ruoslahti E: Mitochondrial p32 protein
is a critical regulator of tumor metabolism via maintenance of
oxidative phosphorylation. Mol Cell Biol. 30:1303–1318.
2023.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Gotoh K, Morisaki T, Setoyama D, Sasaki K,
Yagi M, Igami K, Mizuguchi S, Uchiumi T, Fukui Y and Kang D:
Mitochondrial p32/C1qbp is a critical regulator of dendritic cell
metabolism and maturation. Cell Rep. 25:1800–1815.e1804.
2018.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Dempsey PW, Allison ME, Akkaraju S,
Goodnow CC and Fearon DT: C3d of complement as a molecular
adjuvant: Bridging innate and acquired immunity. Science.
271:348–350. 1996.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Botto M, Dell'Agnola C, Bygrave AE,
Thompson EM, Cook HT, Petry F, Loos M, Pandolfi PP and Walport MJ:
Homozygous C1q deficiency causes glomerulonephritis associated with
multiple apoptotic bodies. Nat Genet. 19:56–59. 1998.PubMed/NCBI View Article : Google Scholar
|
|
20
|
Muta T, Kang D, Kitajima S, Fujiwara T and
Hamasaki N: p32 protein, a splicing factor 2-associated protein, is
localized in mitochondrial matrix and is functionally important in
maintaining oxidative phosphorylation. J Biol Chem.
272:24363–24370. 1997.PubMed/NCBI View Article : Google Scholar
|
|
21
|
Krainer A, Mayeda A, Kozak D and Binns G:
Functional expression of cloned human splicing factor SF2-homology
to RNA-binding proteins, U1 70K, and Drosophila splicing
regulators. Cell. 66:383–394. 1991.PubMed/NCBI View Article : Google Scholar
|
|
22
|
Honor B, Madsena P, Rasmusserf HH,
Vandekerckhove J, Celis JE and Leffers H: Cloning and expression of
a cDNA covering the complete coding region of the P32 subunit of
human pre-mRNA splicing factor SF2. Gene. 134:283–287.
1993.PubMed/NCBI View Article : Google Scholar
|
|
23
|
Peerschke EI, Petrovan RJ, Ghebrehiwet B
and Ruf W: Tissue factor pathway inhibitor-2 (TFPI-2) recognizes
the complement and kininogen binding protein gC1qR/p33 (gC1qR):
Implications for vascular inflammation. Thromb Haemost. 92:811–819.
2004.PubMed/NCBI View Article : Google Scholar
|
|
24
|
Xu L, Xiao N, Liu F, Ren H and Gu J:
Inhibition of RIG-I and MDA5-dependent antiviral response by gC1qR
at mitochondria. Proc Natl Acad Sci USA. 106:1530–1535.
2009.PubMed/NCBI View Article : Google Scholar
|
|
25
|
Song K, Wu Y, Fu B, Wang L, Hao W, Hua F,
Sun Y, Dorf ME and Li S: Leaked mitochondrial C1QBP inhibits
activation of the DNA sensor cGAS. J Immunol. 207:2155–2166.
2021.PubMed/NCBI View Article : Google Scholar
|
|
26
|
Biswas AK, Hafiz A, Banerjee B, Kim KS,
Datta K and Chitnis CE: Plasmodium falciparum uses gC1qR/HABP1/p32
as a receptor to bind to vascular endothelium and for
platelet-mediated clumping. PLoS Pathog. 3:1271–1280.
2007.PubMed/NCBI View Article : Google Scholar
|
|
27
|
Egusquiza-Alvarez CA and Robles-Flores M:
An approach to p32/gC1qR/HABP1: A multifunctional protein with an
essential role in cancer. J Cancer Res Clin Oncol. 148:1831–1854.
2022.PubMed/NCBI View Article : Google Scholar
|
|
28
|
Feng X, Tonnesen MG, Peerschke EI and
Ghebrehiwet B: Cooperation of C1q receptors and integrins in
C1q-mediated endothelial cell adhesion and spreading. J Immunol.
168:2441–2448. 2002.PubMed/NCBI View Article : Google Scholar
|
|
29
|
Deb TB and Datta K: Molecular cloning of
human fibroblast hyaluronic acid-binding protein confirms its
identity with P-32, a protein co-purified with splicing factor SF2.
Hyaluronic acid-binding protein as P-32 protein, co-purified with
splicing factor SF2. J Biol Chem. 271:2206–2212. 1996.PubMed/NCBI View Article : Google Scholar
|
|
30
|
Petersen-Mahrt SK, Estmer C, Ohrmalm C,
Matthews DA, Russell WC and Akusjärvi G: The splicing
factor-associated protein, p32, regulates RNA splicing by
inhibiting ASF/SF2 RNA binding and phosphorylation. EMBO J.
18:1014–1024. 1999.PubMed/NCBI View Article : Google Scholar
|
|
31
|
Shi H, Fang W, Liu M and Fu D: Complement
component 1, q subcomponent binding protein (C1QBP) in lipid rafts
mediates hepatic metastasis of pancreatic cancer by regulating
IGF-1/IGF-1R signaling. Int J Cancer. 141:1389–1401.
2017.PubMed/NCBI View Article : Google Scholar
|
|
32
|
Wang Z, Sun A, Yan A, Yao J, Huang H, Gao
Z, Han T, Gu J, Li N, Wu H and Li K: Circular RNA MTCL1 promotes
advanced laryngeal squamous cell carcinoma progression by
inhibiting C1QBP ubiquitin degradation and mediating beta-catenin
activation. Mol Cancer. 21(92)2022.PubMed/NCBI View Article : Google Scholar
|
|
33
|
Ma J, Ren C, Yang H, Zhao J, Wang F and
Wan Y: The expression pattern of p32 in sheep muscle and its role
in differentiation, cell proliferation, and apoptosis of myoblasts.
Int J Mol Sci. 20(5161)2019.PubMed/NCBI View Article : Google Scholar
|
|
34
|
Wu H, Chu Y, Sun S, Li G, Xu S, Zhang X,
Jiang Y, Gao S, Wang Q, Zhang J and Pang D: Hypoxia-mediated
complement 1q binding protein regulates metastasis and
chemoresistance in triple-negative breast cancer and modulates the
PKC-NF-κB-VCAM-1 signaling pathway. Front Cell Dev Biol.
9(607142)2021.PubMed/NCBI View Article : Google Scholar
|
|
35
|
Sünderhauf A, Hicken M, Schlichting H,
Skibbe K, Ragab M, Raschdorf A, Hirose M, Schäffler H, Bokemeyer A,
Bettenworth D, et al: Loss of Mucosal p32/gC1qR/HABP1 triggers
energy deficiency and impairs goblet cell differentiation in
ulcerative colitis. Cell Mol Gastroenterol Hepatol. 12:229–250.
2021.PubMed/NCBI View Article : Google Scholar
|
|
36
|
Kaul R, Saha P, Saradhi M, Prasad RL,
Chatterjee S, Ghosh I, Tyagi RK and Datta K: Overexpression of
hyaluronan-binding protein 1 (HABP1/p32/gC1qR) in HepG2 cells leads
to increased hyaluronan synthesis and cell proliferation by
up-regulation of cyclin D1 in AKT-dependent pathway. J Biol Chem.
287:19750–19764. 2012.PubMed/NCBI View Article : Google Scholar
|
|
37
|
Egusquiza-Alvarez CA, Moreno-Londoño AP,
Alvarado-Ortiz E, Ramos-Godínez MDP, Sarabia-Sánchez MA,
Castañeda-Patlán MC and Robles-Flores M: Inhibition of
multifunctional protein p32/C1QBP promotes cytostatic effects in
colon cancer cells by altering mitogenic signaling pathways and
promoting mitochondrial damage. Int J Mol Sci.
25(2712)2024.PubMed/NCBI View Article : Google Scholar
|
|
38
|
Krust B, El Khoury D, Nondier I,
Soundaramourty C and Hovanessian AG: Targeting surface nucleolin
with multivalent HB-19 and related Nucant pseudopeptides results in
distinct inhibitory mechanisms depending on the malignant tumor
cell type. BMC Cancer. 11(333)2011.PubMed/NCBI View Article : Google Scholar
|
|
39
|
Dembitzer FR, Kinoshita Y, Burstein D,
Phelps RG, Beasley MB, Garcia R, Harpaz N, Jaffer S, Thung SN,
Unger PD, et al: gC1qR expression in normal and pathologic human
tissues: differential expression in tissues of epithelial and
mesenchymal origin. J Histochem Cytochem. 60:467–474.
2012.PubMed/NCBI View Article : Google Scholar
|
|
40
|
Xie Z, Gao Y, Ho C, Li L, Jin C, Wang X,
Zou C, Mao Y, Wang X, Li Q, et al: Exosome-delivered CD44v6/C1QBP
complex drives pancreatic cancer liver metastasis by promoting
fibrotic liver microenvironment. Gut. 71:568–579. 2022.PubMed/NCBI View Article : Google Scholar
|
|
41
|
Bai Y, Wang W, Li S, Zhan J, Li H, Zhao M,
Zhou XA, Li S, Li X, Huo Y, et al: C1QBP promotes homologous
recombination by stabilizing MRE11 and controlling the assembly and
activation of MRE11/RAD50/NBS1 complex. Mol Cell.
75:1299–1314.e1296. 2019.PubMed/NCBI View Article : Google Scholar
|
|
42
|
Chastney MR, Kaivola J, Leppänen VM and
Ivaska J: The role and regulation of integrins in cell migration
and invasion. Nat Rev Mol Cell Biol. 26:147–167. 2025.PubMed/NCBI View Article : Google Scholar
|
|
43
|
Li X, Wu Y, Zhang M, Wang F, Yin H, Zhang
Y, Zhao S, Ma J, Lv M and Lu C: A new peptide inhibitor of C1QBP
exhibits potent anti-tumour activity against triple negative breast
cancer by impairing mitochondrial function and suppressing
homologous recombination repair. Clin Transl Med.
15(e70162)2025.PubMed/NCBI View Article : Google Scholar
|
|
44
|
Kerdidani D, Aerakis E, Verrou KM,
Angelidis I, Douka K, Maniou MA, Stamoulis P, Goudevenou K, Prados
A, Tzaferis C, et al: Lung tumor MHCII immunity depends on in situ
antigen presentation by fibroblasts. J Exp Med.
219(e20210815)2022.PubMed/NCBI View Article : Google Scholar
|
|
45
|
Cheng X, Wang M, Li W, Tian B, Zhang J,
Zhang C and Liu P: C1QBP forms a positive feedback loop with the
PAICS/FAK/C-MYC axis to promote cancer cell proliferation.
Oncogene. 44:4363–4376. 2025.PubMed/NCBI View Article : Google Scholar
|
|
46
|
Prakash M, Kale S, Ghosh I, Kundu GC and
Datta K: Hyaluronan-binding protein 1 (HABP1/p32/gC1qR) induces
melanoma cell migration and tumor growth by NF-kappa B dependent
MMP-2 activation through integrin α(v)β(3) interaction. Cell
Signal. 23:1563–1577. 2011.PubMed/NCBI View Article : Google Scholar
|
|
47
|
Yue D, Wang Y, Sun Y, Niu Y and Chang C:
C1QBP regulates YBX1 to suppress the androgen receptor
(AR)-enhanced RCC cell invasion. Neoplasia. 19:135–144.
2017.PubMed/NCBI View Article : Google Scholar
|
|
48
|
Wang Y, Yue D, Xiao M, Qi C, Chen Y, Sun
D, Zhang N and Chen R: C1QBP negatively regulates the activation of
oncoprotein YBX1 in the renal cell carcinoma as revealed by
interactomics analysis. J Proteome Res. 14:804–813. 2015.PubMed/NCBI View Article : Google Scholar
|
|
49
|
Leucci E, Vendramin R, Spinazzi M,
Laurette P, Fiers M, Wouters J, Radaelli E, Eyckerman S, Leonelli
C, Vanderheyden K, et al: Melanoma addiction to the long non-coding
RNA SAMMSON. Nature. 531:518–522. 2016.PubMed/NCBI View Article : Google Scholar
|
|
50
|
Sinha S, Singh SK, Jangde N, Ray R and Rai
V: p32 promotes melanoma progression and metastasis by targeting
EMT markers, Akt/PKB pathway, and tumor microenvironment. Cell
Death Dis. 12(1012)2021.PubMed/NCBI View Article : Google Scholar
|
|
51
|
Qin X, Kang K, Zhu B, Shi Y and Bu S:
C1QBP drives M2 macrophage polarization via TRAF2-CCL2 to promote
oral squamous cell carcinoma progression. Int Dent J.
75(103938)2025.PubMed/NCBI View Article : Google Scholar
|
|
52
|
Zhang S, Wang C, Wang Y, Zhang H, Xu C,
Cheng Y, Yuan Y, Sha J, Guo X and Cui Y: A novel protein encoded by
circRsrc1 regulates mitochondrial ribosome assembly and translation
during spermatogenesis. BMC Biol. 21(94)2023.PubMed/NCBI View Article : Google Scholar
|
|
53
|
Becker YLC, Gagné JP, Julien AS, Lévesque
T, Allaeys I, Gougeard N, Rubio V, Boisvert FM, Jean D, Wagner E,
et al: Identification of Mitofusin 1 and complement component 1q
subcomponent binding protein as mitochondrial targets in systemic
lupus erythematosus. Arthritis Rheumatol. 74:1193–1203.
2022.PubMed/NCBI View Article : Google Scholar
|
|
54
|
Duan Y, Yang Y, Zhu R and Xiao M:
Endotoxin lipopolysaccharide challenge triggers gut microbiota
dysbiosis and host immune remodeling in the tiger grouper
Epinephelus fuscoguttatus. Comp Biochem Physiol C Toxicol
Pharmacol. 299(110364)2026.PubMed/NCBI View Article : Google Scholar
|
|
55
|
Wang SQ, Jin MJ, Guo ZK, Shen DR, Jin LN,
Cheng F, Zhao YR, Liu T, Li YC, Wang NY, et al: Trace
element-dictated exosome modules and self-adaptive dual-network
hydrogel orchestrate diabetic foot regeneration through
complement-mitochondria-autophagy circuitry. Mil Med Res.
12(71)2025.PubMed/NCBI View Article : Google Scholar
|
|
56
|
Zhai X, Liu K, Fang H, Zhang Q, Gao X, Liu
F, Zhou S, Wang X, Niu Y, Hong Y, et al: Mitochondrial C1qbp
promotes differentiation of effector CD8(+) T cells via
metabolic-epigenetic reprogramming. Sci Adv.
7(eabk0490)2021.PubMed/NCBI View Article : Google Scholar
|
|
57
|
Feichtinger RG, Oláhová M, Kishita Y,
Garone C, Kremer LS, Yagi M, Uchiumi T, Jourdain AA, Thompson K,
D'Souza AR, et al: Biallelic C1QBP mutations cause severe
Neonatal-, Childhood-, or later-onset cardiomyopathy associated
with combined respiratory-chain deficiencies. Am J Hum Genet.
101:525–538. 2017.PubMed/NCBI View Article : Google Scholar
|
|
58
|
Yenugonda V, Nomura N, Kouznetsova V,
Tsigelny I, Fogal V, Nurmemmedov E, Kesari S and Babic I: A novel
small molecule inhibitor of p32 mitochondrial protein overexpressed
in glioma. J Transl Med. 15(210)2017.PubMed/NCBI View Article : Google Scholar
|
|
59
|
Reef S, Shifman O, Oren M and Kimchi A:
The autophagic inducer smARF interacts with and is stabilized by
the mitochondrial p32 protein. Oncogene. 26:6677–6683.
2007.PubMed/NCBI View Article : Google Scholar
|
|
60
|
Bi Q, Nie J, Wu Q, Sun L, Zhu S, Bai J,
Liu Y, Huang F and Chai K: Integrative pan-cancer genomic analysis
highlights mitochondrial protein p32 as a potential therapeutic
target in Myc-driven tumorigenesis. Med Oncol.
42(60)2025.PubMed/NCBI View Article : Google Scholar
|
|
61
|
Wen X, Li W, Li S, Chen D, Zheng J, Wang
X, Zhang C, Liu Y, Ning Y, Jia R, et al: Longitudinal single-cell
RNA sequencing reveals a heterogeneous response of plasma cells to
colonic inflammation. Int J Biol Macromol.
294(139307)2025.PubMed/NCBI View Article : Google Scholar
|
|
62
|
Mizuguchi S, Gotoh K, Nakashima Y,
Setoyama D, Takata Y, Ohga S and Kang D: Mitochondrial reactive
oxygen species are essential for the development of psoriatic
inflammation. Front Immunol. 12(714897)2021.PubMed/NCBI View Article : Google Scholar
|
|
63
|
Yao ZQ, Prayther D, Trabue C, Dong ZP and
Moorman J: Differential regulation of SOCS-1 signalling in B and T
lymphocytes by hepatitis C virus core protein. Immunology.
125:197–207. 2008.PubMed/NCBI View Article : Google Scholar
|
|
64
|
Yao ZQ, Eisen-Vandervelde A, Waggoner SN,
Cale EM and Hahn YS: Direct binding of hepatitis C virus core to
gC1qR on CD4+ and CD8+ T cells leads to impaired activation of Lck
and Akt. J Virol. 78:6409–6419. 2004.PubMed/NCBI View Article : Google Scholar
|
|
65
|
Mumme H, Thomas BE, Bhasin SS, Krishnan U,
Dwivedi B, Perumalla P, Sarkar D, Ulukaya GB, Sabnis HS, Park SI,
et al: Single-cell analysis reveals altered tumor microenvironments
of relapse- and remission-associated pediatric acute myeloid
leukemia. Nat Commun. 14(6209)2023.PubMed/NCBI View Article : Google Scholar
|
|
66
|
Tian H, Wang G, Wang Q, Zhang B, Jiang G,
Li H, Chai D, Fang L, Wang M and Zheng J: Complement C1q binding
protein regulates T cells' mitochondrial fitness to affect their
survival, proliferation, and anti-tumor immune function. Cancer
Sci. 113:875–890. 2022.PubMed/NCBI View Article : Google Scholar
|
|
67
|
Zhang C, Liu Y, Jiang J, Chen C, Duan Z,
Su H, Wang S, Tian B, Shi Y, Xiang R and Luo Y: Targeting tumor
cell-to-macrophage communication by blocking Vtn-C1qbp interaction
inhibits tumor progression via enhancing macrophage phagocytosis.
Theranostics. 14:2757–2776. 2024.PubMed/NCBI View Article : Google Scholar
|
|
68
|
Wu X, Tang N, Zhao Q and Xiong J:
Spatiotemporal evolutionary process of osteosarcoma immune
microenvironment remodeling and C1QBP-driven drug resistance
deciphered through single-cell multi-dimensional analysis. Bioeng
Transl Med. 9(e10654)2024.PubMed/NCBI View Article : Google Scholar
|
|
69
|
Piltti KM, Lakatos A, Benavente-Perez F,
Elrachid ZH, Nava AA, Addonizio NT, Nguyen V, Zahedi A, Song WA,
Chen X, et al: C1q drives neural stem cell quiescence by regulating
cell cycle and metabolism through BAI1. Nat Commun.
16(11386)2025.PubMed/NCBI View Article : Google Scholar
|
|
70
|
Ghose S, Biswas S, Datta K and Tyagi RK:
Dynamic Hyaluronan drives liver endothelial cells towards
angiogenesis. BMC Cancer. 18(648)2018.PubMed/NCBI View Article : Google Scholar
|
|
71
|
Peerschke EI and Ghebrehiwet B: cC1qR/CR
and gC1qR/p33: Observations in cancer. Mol Immunol. 61:100–109.
2014.PubMed/NCBI View Article : Google Scholar
|
|
72
|
Dong X, Liu Z, Yu M, Yang X and Cai H:
Identification of the whole genome of alternative splicing and
RNA-binding proteins involved in nintedanib-induced apoptosis in
gastric cancer cells. PeerJ. 12(e18697)2024.PubMed/NCBI View Article : Google Scholar
|
|
73
|
Rousso-Noori L, Mastandrea I, Talmor S,
Waks T, Levin AG, Haugas M, Teesalu T, Alvarez-Vallina L, Eshhar Z
and Friedmann-Morvinski D: P32-specific CAR T cells with dual
antitumor and antiangiogenic therapeutic potential in gliomas. Nat
Commun. 12(3615)2021.PubMed/NCBI View Article : Google Scholar
|
|
74
|
Lei Y, Li X, Qin D, Zhang Y and Wang Y:
gC1qR: A new target for cancer immunotherapy. Front Immunol.
14(1095943)2023.PubMed/NCBI View Article : Google Scholar
|
|
75
|
Momenimovahed Z, Mazidimoradi A, Maroofi
P, Allahqoli L, Salehiniya H and Alkatout I: Global, regional and
national burden, incidence, and mortality of cervical cancer.
Cancer Rep (Hoboken). 6(e1756)2023.PubMed/NCBI View Article : Google Scholar
|
|
76
|
Burd EM: Human papillomavirus and cervical
cancer. Clin Microbiol Rev. 16:1–17. 2003.PubMed/NCBI View Article : Google Scholar
|
|
77
|
Gao LJ, Gu PQ, Zhao W, Ding WY, Zhao XQ,
Guo SY and Zhong TY: The role of globular heads of the C1q receptor
in HPV 16 E2-induced human cervical squamous carcinoma cell
apoptosis is associated with p38 MAPK/JNK activation. J Transl Med.
11(118)2013.PubMed/NCBI View Article : Google Scholar
|
|
78
|
Chen ZL, Su YJ, Zhang HL, Gu PQ and Gao
LJ: The role of the globular heads of the C1q receptor in HPV-16
E2-induced human cervical squamous carcinoma cell apoptosis via a
mitochondria-dependent pathway. J Transl Med.
12(286)2014.PubMed/NCBI View Article : Google Scholar
|
|
79
|
Gao LJ, Gu PQ, Fan WM, Liu Z, Qiu F, Peng
YZ and Guo XR: The role of gC1qR in regulating survival of human
papillomavirus 16 oncogene-transfected cervical cancer cells. Int J
Oncol. 39:1265–1272. 2011.PubMed/NCBI View Article : Google Scholar
|
|
80
|
Itahana K and Zhang Y: Mitochondrial p32
is a critical mediator of ARF-induced apoptosis. Cancer Cell.
13:542–553. 2008.PubMed/NCBI View Article : Google Scholar
|
|
81
|
Liu Z, Su YJ, Gu PQ, Ji ZY, Wang XG and
Gao LJ: The role of the globular heads of C1q receptor (gC1qR) gene
in regulating apoptosis of human cervical squamous cell carcinoma.
Cell Physiol Biochem. 30:1181–1190. 2012.PubMed/NCBI View Article : Google Scholar
|
|
82
|
Siegel RL, Miller KD, Fuchs HE and Jemel
A: Cancer statistics, 2021. CA Cancer J Clin. 71:7–33.
2021.PubMed/NCBI View Article : Google Scholar
|
|
83
|
Yu G and Wang J: Significance of
hyaluronan binding protein (HABP1/P32/gC1qR) expression in advanced
serous ovarian cancer patients. Exp Mol Pathol. 94:210–215.
2013.PubMed/NCBI View Article : Google Scholar
|
|
84
|
Duan C, Gao Y, Luan S, Guo S, Cao X, Xu P,
Fu P and Zhao C: Noninvasive evaluation of HABP1 expression with
99mTc-labeled small-interference RNA in ovarian cancer. Int J
Radiat Biol. 97:1569–1577. 2021.PubMed/NCBI View Article : Google Scholar
|
|
85
|
Hunt H, Simón-Gracia L, Tobi A, Kotamraju
VR, Sharma S, Nigul M, Sugahara KN, Ruoslahti E and Teesalu T:
Targeting of p32 in peritoneal carcinomatosis with intraperitoneal
linTT1 peptide-guided pro-apoptotic nanoparticles. J Control
Release. 260:142–153. 2017.PubMed/NCBI View Article : Google Scholar
|
|
86
|
Bryce C, Gazda R and Fuerst H: Endometrial
cancer: Rapid evidence review. Am Fam Physician. 111:526–531.
2025.PubMed/NCBI
|
|
87
|
Galani A, Stavros S, Moustakli E, Potiris
A, Zikopoulos A, Anagnostaki I, Zacharis K, Paraskevaidi M, Lyons
D, Maneta-Stavrakaki S, et al: Endometrial hyperplasia: Current
insights into epidemiology, risk factors, and clinical management.
Cancers (Basel), 2025, 18(1).
|
|
88
|
Zheng Y, Hu R, Yang F, Liu G, Peng T, Xie
L, Wu J, Hou L and Yuan R: Comparing survival outcomes between
surgical and non-surgical treatments in patients with early-onset
endometrial cancer and developing a nomogram to predict survival: A
study based on Eastern and Western data sets. World J Surg Oncol.
23(184)2025.PubMed/NCBI View Article : Google Scholar
|
|
89
|
Rubinstein DB, Stortchevoi A, Boosalis M,
Ashfaq R, Ghebrehiwet B, Peerschke EI, Calvo F and Guillaume T:
Receptor for the globular heads of C1q (gC1q-R, p33,
hyaluronan-binding protein) is preferentially expressed by
adenocarcinoma cells. Int J Cancer. 110:741–750. 2004.PubMed/NCBI View Article : Google Scholar
|
|
90
|
Yang M, Hu Y, Guo W, Yang F, Qi X and
Zheng Y: Causal relationship between components of metabolic
syndrome and endometrial carcinoma. Sichuan Da Xue Xue Bao Yi Xue
Ban. 55:1557–1566. 2024.PubMed/NCBI View Article : Google Scholar : (In Chinese).
|
|
91
|
Pérez-Martín AR, Castro-Eguiluz D,
Cetina-Pérez L, Velasco-Torres Y, Bahena-González A, Montes-Servín
E, González-Ibarra E, Espinosa-Romero R and Gallardo-Rincón D:
Impact of metabolic syndrome on the risk of endometrial cancer and
the role of lifestyle in prevention. Bosn J Basic Med Sci.
22:499–510. 2022.PubMed/NCBI View Article : Google Scholar
|
|
92
|
de Boer SM, Powell ME, Mileshkin L,
Katsaros D, Bessette P, Haie-Meder C, Ottevanger PB, Ledermann JA,
Khaw P, D'Amico R, et al: Adjuvant chemoradiotherapy versus
radiotherapy alone in women with high-risk endometrial cancer
(PORTEC-3): patterns of recurrence and post-hoc survival analysis
of a randomised phase 3 trial. Lancet Oncol. 20:1273–1285.
2019.PubMed/NCBI View Article : Google Scholar
|
|
93
|
Ryu KJ, Kim MS, Lee JY, Nam S, Jeong HG,
Kim T and Park H: Risk of endometrial polyps, hyperplasia,
carcinoma, and uterine cancer after tamoxifen treatment in
premenopausal women with breast cancer. JAMA Netw Open.
5(e2243951)2022.PubMed/NCBI View Article : Google Scholar
|
|
94
|
Katagiri R, Iwasaki M, Abe SK, Islam MR,
Rahman MS, Saito E, Merritt MA, Choi JY, Shin A, Sawada N, et al:
Reproductive factors and endometrial cancer risk among women. JAMA
Netw Open. 6(e2332296)2023.PubMed/NCBI View Article : Google Scholar
|
|
95
|
Liang J and Shang Y: Estrogen and cancer.
Annu Rev Physiol. 75:225–240. 2013.PubMed/NCBI View Article : Google Scholar
|
|
96
|
Hu Z, Tang M, Huang Y, Cai B, Sun X, Chen
G, Huang A, Li X, Shah AR, Jiang L, et al: SIRT7 facilitates
endometrial cancer progression by regulating PTEN stability in an
estrogen-dependent manner. Nat Commun. 16(2989)2025.PubMed/NCBI View Article : Google Scholar
|
|
97
|
Thakur SC, Kumar V, Ghosh I, Bharadwaj A
and Datta K: Appearance of hyaluronan binding protein 1 proprotein
in pachytene spermatocytes and round spermatids correlates with
spermatogenesis. J Androl. 27:604–610. 2006.PubMed/NCBI View Article : Google Scholar
|
|
98
|
Xiao S, Xu C and Jarvis JN: C1q-bearing
immune complexes induce IL-8 secretion in human umbilical vein
endothelial cells (HUVEC) through protein tyrosine kinase- and
mitogen-activated protein kinase-dependent mechanisms: evidence
that the 126 kD phagocytic C1q receptor mediates immune complex
activation of HUVEC. Clin Exp Immunol. 125:360–367. 2001.PubMed/NCBI View Article : Google Scholar
|
|
99
|
Dong M, Seemann F, Humble JL, Liang Y,
Peterson DR, Ye R, Ren H, Kim HS, Lee JS, Au DWT and Lam YW:
Modification of the plasma complement protein profile by exogenous
estrogens is indicative of a compromised immune competence in
marine medaka (Oryzias melastigma). Fish Shellfish Immunol.
70:260–269. 2017.PubMed/NCBI View Article : Google Scholar
|
|
100
|
Wu Y, Li X, Hou Y, Lv M and Zhao S:
Targeting the KRAS-p38-ATF4 axis enhances the therapeutic
sensitivity of the peptide PDBAG1 in ovarian cancer via stress
response modulation. Eur J Pharmacol. 1006(178128)2025.PubMed/NCBI View Article : Google Scholar
|
|
101
|
Liu Y, Leslie PL, Jin A, Itahana K, Graves
LM and Zhang Y: p32 heterozygosity protects against age- and
diet-induced obesity by increasing energy expenditure. Sci Rep.
7(5754)2017.PubMed/NCBI View Article : Google Scholar
|
|
102
|
Liu Y, Leslie PL, Jin A, Itahana K, Graves
LM and Zhang Y: p32 regulates ER stress and lipid homeostasis by
down-regulating GCS1 expression. FASEB J. 32:3892–3902.
2018.PubMed/NCBI View Article : Google Scholar
|
|
103
|
van den Heerik A, Horeweg N, de Boer SM,
Bosse T and Creutzberg CL: Adjuvant therapy for endometrial cancer
in the era of molecular classification: Radiotherapy,
chemoradiation and novel targets for therapy. Int J Gynecol Cancer.
31:594–604. 2021.PubMed/NCBI View Article : Google Scholar
|
|
104
|
Wang J, Shi Y, Wang Y, Shen Y, Liu H, Sun
S, Wang Y, Zhou X, Zhou Y, Zeng X, et al: PA28γ promotes the
malignant progression of tumor by elevating mitochondrial function
via C1QBP. Elife. 13(RP101244)2025.PubMed/NCBI View Article : Google Scholar
|
|
105
|
Yagi M, Uchiumi T, Takazaki S, Okuno B,
Nomura M, Yoshida S, Kanki T and Kang D: p32/gC1qR is indispensable
for fetal development and mitochondrial translation: Importance of
its RNA-binding ability. Nucleic Acids Res. 40:9717–9737.
2012.PubMed/NCBI View Article : Google Scholar
|
|
106
|
Wang Z, Chen J, Wu X, Ma D, Zhang X, Li R,
Han C, Liu H, Yin X, Du Q, et al: PCV2 targets cGAS to inhibit type
I interferon induction to promote other DNA virus infection. PLoS
Pathog. 17(e1009940)2021.PubMed/NCBI View Article : Google Scholar
|
|
107
|
Song N, Zhao L, Zhu M and Zhao J: Recent
progress in LyP-1-based strategies for targeted imaging and
therapy. Drug Deliv. 26:363–375. 2019.PubMed/NCBI View Article : Google Scholar
|
|
108
|
Jenkins MR and Drummond KJ: CAR T-cell
therapy for glioblastoma. N Engl J Med. 390:1329–1332.
2024.PubMed/NCBI View Article : Google Scholar
|
|
109
|
Bagley SJ, Logun M, Fraietta JA, Wang X,
Desai AS, Bagley LJ, Nabavizadeh A, Jarocha D, Martins R, Maloney
E, et al: Intrathecal bivalent CAR T cells targeting EGFR and
IL13Rα2 in recurrent glioblastoma: Phase 1 trial interim results.
Nat Med. 30:1320–1329. 2024.PubMed/NCBI View Article : Google Scholar
|
|
110
|
Choe JH, Watchmaker PB, Simic MS, Gilbert
RD, Li AW, Krasnow NA, Downey KM, Yu W, Carrera DA, Celli A, et al:
SynNotch-CAR T cells overcome challenges of specificity,
heterogeneity, and persistence in treating glioblastoma. Sci Transl
Med. 13(eabe7378)2021.PubMed/NCBI View Article : Google Scholar
|
|
111
|
Cai G, Huang F, Gao Y, Li X, Chi J, Xie J,
Zhou L, Feng Y, Huang H, Deng T, et al: Artificial
intelligence-based models enabling accurate diagnosis of ovarian
cancer using laboratory tests in China: A multicentre,
retrospective cohort study. Lancet Digit Health. 6:e176–e186.
2024.PubMed/NCBI View Article : Google Scholar
|
|
112
|
Njoku K, Barr CE, Sutton CJJ and Crosbie
EJ: Urine CA125 and HE4 for the triage of symptomatic women with
suspected endometrial cancer. Cancers (Basel).
14(3306)2022.PubMed/NCBI View Article : Google Scholar
|
|
113
|
Scully OJ, Shyamasundar S, Matsumoto K,
Dheen ST, Yip GW and Bay BH: C1QBP Mediates breast cancer cell
proliferation and growth via multiple potential signalling
pathways. Int J Mol Sci. 24(1343)2023.PubMed/NCBI View Article : Google Scholar
|
|
114
|
Sonveaux P, Végran F, Schroeder T, Wergin
MC, Verrax J, Rabbani ZN, De Saedeleer CJ, Kennedy KM, Diepart C,
Jordan B, et al: Targeting lactate-fueled respiration selectively
kills hypoxic tumor cells in mice. J Clin Invest. 118:3930–3942.
2008.PubMed/NCBI View Article : Google Scholar
|