Introduction
Gliomas (grade IV) are the most malignant tumors and
still remain a lethal brain cancer with a short overall survival
(OS) time. Standard treatments, including surgical resection,
chemotherapy with temozolomide (TEMO), radiotherapy, and new
therapeutic strategies that appeared to be promising did not
prolong patients' lives nor did they reduce cancer recurrence
rates. Genetic, epigenetic, environmental heterogeneity and cell
plasticity, which are specific to each patient, are responsible for
the resistance to treatment of gliomas (1). Therefore, there is a great effort to
propose new therapeutic lines or to design new drugs for glioma
therapy. Immunotherapeutic strategies are emerging as a promising
avenue that might offer new hope for the treatment of gliomas.
Research that has been conducted in recent years has
shed a light on a new direction related to an attractive strategy
called ‘drug repurposingʼ, which includes broadening the clinical
indications of some drugs to use in therapy for other diseases
(2,3). Among the current candidates for
glioma adjuvant therapy are antidepressants, which have been used
in other clinical treatments for numerous years. Their potential
anticancer properties, which are beyond their antidepressant
effect, have been revealed. Their well-understood safety profile
and cellular effects, including as an
anti-inflammatory/immunomodulatory for glial cells, antioxidant
activity, influence on neural plasticity and transcriptional
factors, receptor action, and modulatory effect on trophic factors
were assessed. Additionally, the possibility of them reaching a
high concentration in the brain due to their ability to cross
through the blood brain barrier, which is occasionally impossible
for typical anticancer drugs, renders antidepressants an attractive
tool in the fight against brain cancer (3-7).
Furthermore, depression is a common psychological
disorder among patients with glioma (8); therefore, finding antidepressants
with well-documented antitumor effects would be a promising
adjuvant strategy for treating glioma. However, this issue requires
further investigation because the results of the experimental
studies that have been conducted in various models are
contradictory. Some results have also suggested that
antidepressants may also promote cancer progression (9).
Amitriptyline (AMI), a tricyclic antidepressant,
appears to be particularly interesting due to its high
neurobiological activity and effectiveness in therapy not only for
depression but also for chronic pain and neuropathic pain in
oncology patients (10). However,
at present, it cannot be clearly stated whether AMI inhibits or
supports the progress of cancer. The majority of preclinical in
vitro studies have been focused on the effects of AMI alone on
glioma cells, whereas, in clinics, patients with glioma are
subjected to both polypharmacotherapy and integrated treatment.
Therefore, experimental models should reflect not only the hypoxia
that is typical for the glioma environment, but the impact of any
interactions between the therapeutic strategies on glioma cells
should also be studied.
Gliomas are immunologically ‘coldʼ brain cancers
that activate various mechanisms in order to escape from immune
surveillance (11). The pathway
that is mediated by programmed cell death ligand 1 (PD-L1) and
programmed death receptor 1 (PD-1) includes the major negative
regulatory molecules at the immune checkpoint axis and the main
players in immunotherapy (12).
PD-L1 is also a marker of the activation of the natural killer
cells that infiltrate head and neck cancers (13). Its expression is found in 85% of
newly diagnosed and 73% of patients with recurrent glioma.
Moreover, it has also been shown that PD-L1 is even more
overexpressed in certain patients with glioma compared with the
others (14). PD-L1 acts like a
pro-cancer factor, stimulates cellular proliferation, and later
tumor progression. Its overexpression makes the prognosis worse,
and as a result, correlates with a short OS time (15). Therefore, inhibiting the PD-L1
pathway could have significant therapeutic value and has been
hailed as a promising therapeutic strategy to activate antitumor
immunity.
In the present study, the influence of AMI alone or
its co-administration with TEMO (the first-line cytostatic in
therapy of glioma) on rat C6 glioma cells that had or had not been
exposed to radiation for the first time were investigated. The
viability and proliferation of cells and PD-L1 expression were
assessed. Moreover, morphological changes in the cell cultures were
determined using microscopic analyses.
Materials and methods
Model in vitro
Experiments have been conducted on a C6 glioma cell
line that is considered to be a safe and popular glioma model in
the literature providing a favorable simulation of glioblastoma
multiforme in human. C6 is a glial cell line that was isolated from
the brain of a rat with glioma. The glial tumor was induced by
N-nitrosomethyl urea after a series of alternate culture and animal
passages. Genetically, C6 cells overexpress the same genes that are
expressed in human gliomas. This cell line can be used in
neuroscience and toxicology research.
C6 rat glioma cells (cat. no. 92090409;
MilliporeSigma) were cultured in DMEM with 10% FBS (cat. no.
SLM-241) as a monolayer in cell culture bottles and hypoxia
conditions (2.5% oxygen, 5% CO2; Hypoxylab, AnimaLab).
After achieving 90% confluence, glioma cells were trypsinized and
passaged. On the second day, the medium was replenished with a
fresh portion containing AMI [Amitriptyline hydrochloride, powder,
≥98% (TLC; cat. no. A8404-10G; MilliporeSigma); 10 µM and/or TEMO
(99% HPLC, powder, cat. no. T2577-25 mg; 1 mM MilliporeSigma. The
cells were exposed to the studied drugs for 72 h. In order to
compare the effect of radiation and its interaction with the
studied drugs, C6 glioma cells were exposed to radiation [single
exposure, 10 Gy; Glioma (rad)] or were not exposed (glioma
(non-rad). Next, functional tests (viability, mortality, cell
proliferation) and microscopy observations of glioma cells
(morphology and colony forming) were conducted. Finally, the PD-L1
expression was determined in each experimental group.
Radiation
The samples were irradiated with 6 MV energy and a
dose rate of 600 on a Clinac 600 CD accelerator from Varian Medical
Systems. The Clinac accelerator is a type of linear accelerator
used in radiotherapy to treat lesions, both cancer and
non-cancerous conditions, by delivering precise doses of ionizing
radiation directly to the tumor or affected area. These devices
utilize high-energy X-rays, and the Clinac 600 CD is equipped with
a multileaf collimator to shape the radiation beam, allowing for
more precise targeting and minimizing the radiation dose to healthy
tissue. All of the measurements were taken in triplicate.
Viability/mortality tests
To estimate the percentage of live and dead glioma
cells in each experimental group, an MTT test [as previously
described (16)] and an analysis
using Trypan blue dye in EVE (Cell automated counter; NanoEntek)
were performed. Each test for each experimental group was conducted
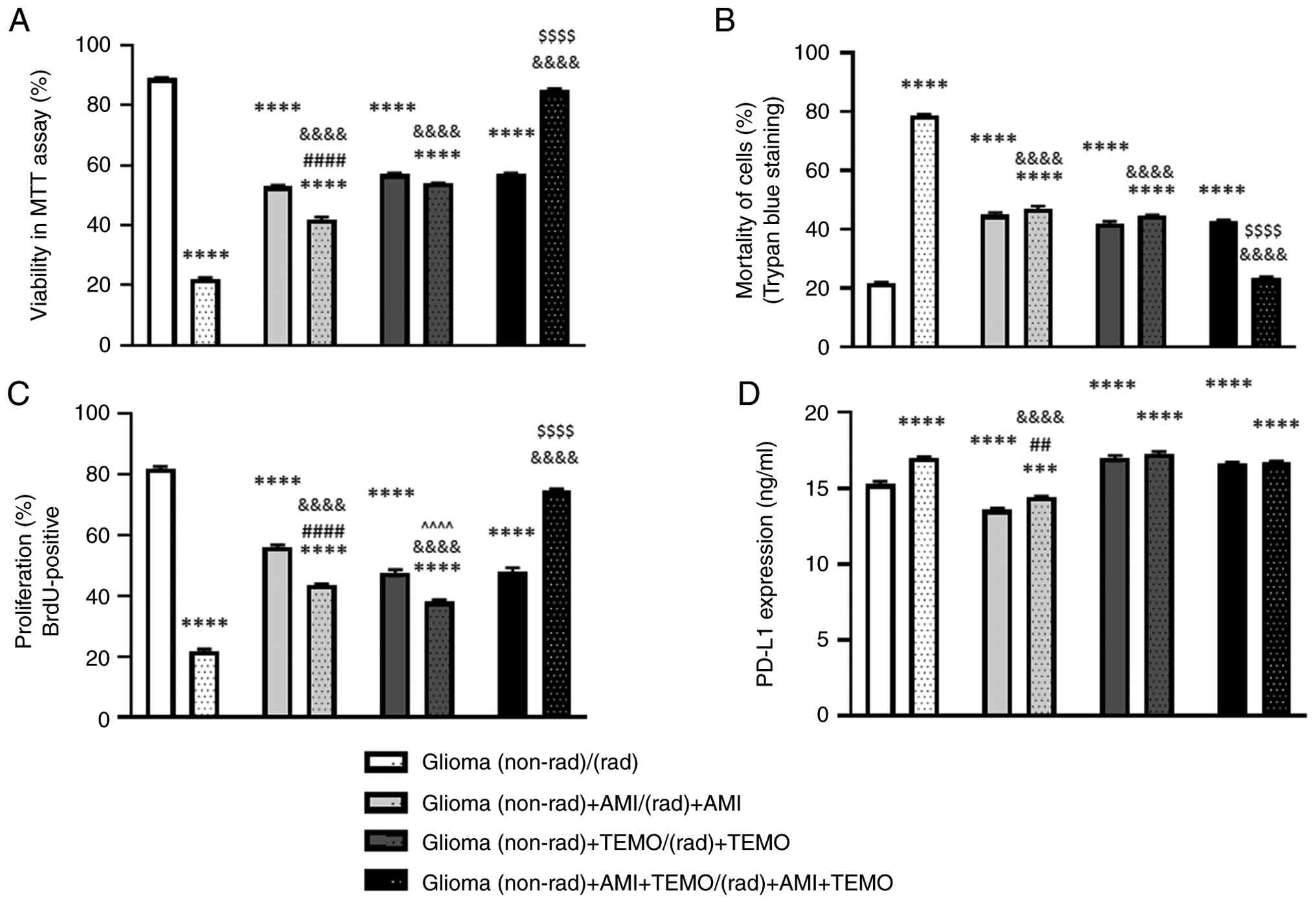
three times. The average results are presented in Fig. 1A and B.
Cell proliferation
The intensity of cell division was evaluated using
an immunoenzymatic assay kit based on a determination of the
bromodeoxyuridine (BrdU) level (Abcam), which is incorporated into
the DNA of dividing cells (cell density was 3x104
cells/ml). The determination was conducted according to the
manufacturer's instructions. Absorbance was measured in a Multiscan
RC microplate reader at 450 nm (Thermo Labsystems). (Fig. 1C). All of the measurements were
received in triplicate.
PD-L1 expression
The expression of PD-L1 was measured using a
commercially available immunoenzymatic assay (cat. no. E1629 Ra;
ACRO Biosystems) according to the manufacturer's protocol. The
optical density (OD) values were measured using an automated
microplate reader (Multiscan RC microplate reader (Labsystems) with
a wavelength of 450 nm as the reference wavelength. All of the
measurements were received in triplicate.
Microscopy observations
Daily observations of the growth dynamics,
morphology and dispersion of glioma cells of C6 lineage that had
been exposed to AMI and/or TEMO and /or radiation were performed
using a JuLi Stage (NanoEntek) microscope. The image analysis was
performed using a camera software (Image software NIS Element),
which is an integrated part of an Eclipse TS-100 microscope and
ImageJ software (Fiji version, 10.1038/nmeth.2019). Colony
formation assessment (minimum number of cells forming a colony was
50) was performed using crystal violet staining (in concentration
0.25% in 25% methanol, at room temperature for 20 min). Each
experimental group was analyzed in 10 fields of view.
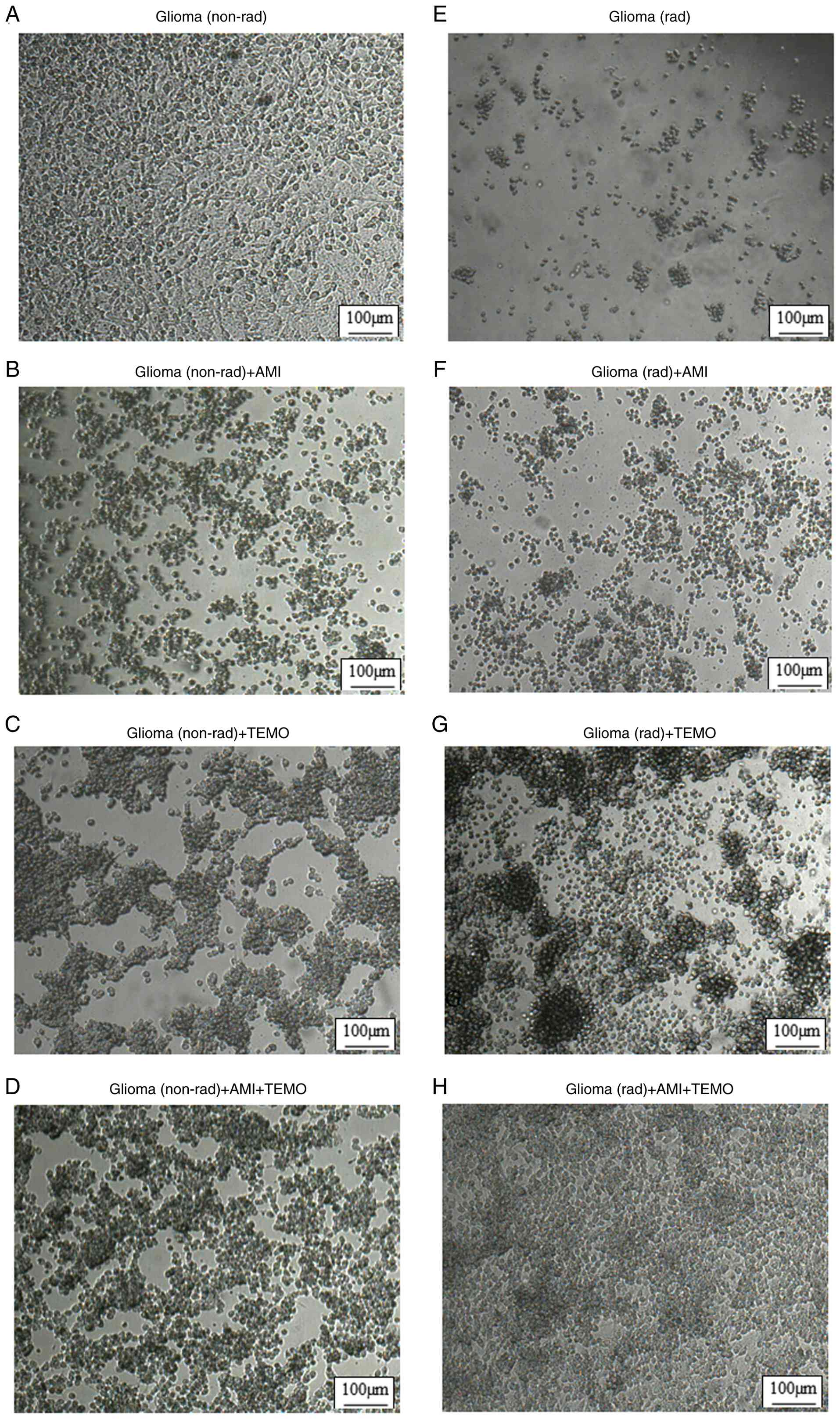
Representative pictures are presented in Fig. 2.
Statistical analysis
The normality of the distribution of the data was
assessed using the Shapiro-Wilk test (α=0.05). The homogeneity of
the variance was calculated using the Levene's test with the η 2
effect type (α=0.05). If the obligatory assumption was established,
the significance of the differences between the groups was
evaluated using a two-way ANOVA (factor 1: radiation; factor 2: the
studied drugs) followed by a post hoc Bonferroni's test. The data
are expressed as the mean ± SEM. P<0.05 was considered to
indicate a statistical significance. Statistical analyses were
performed using GraphPad Prism 9 software (GraphPad Software Inc.;
Dotmatics).
Results
Viability of the cells (%) that were
analyzed in the MTT assay (mitochondrial activity)
The alterations in the viability of the cells (%)
are presented in Fig. 1A. The
results of the two-way ANOVA revealed a significant interaction
between the two analyzed factors (factor 1: radiation; factor 2:
studied drugs). Differences between the groups of cells that had
been treated with the drugs and the untreated cells were strongly
significant (F(3,64)=892.9; P<0.0001). The same
statistical pattern was observed between the groups that had been
exposed to radiation or those that had not been exposed
(F(1,64)=1621.0; P<0.0001). Drug administration (AMI,
TEMO and combined AMI + TEMO) significantly reduced the viability
of the cells in the cultures that had not been exposed to radiation
[decrease of ~36, 32 and 32%, respectively, vs. Glioma (non-rad)
P<0.0001; post hoc Bonferroni]. The radiation was stronger than
the drugs and decreased the viability of cells (~67%, P<0.0001;
post hoc Bonferroni). The AMI effect [a decrease in the viability
of the cells by ~36% vs. Glioma (non-rad)] was potentiated in the
radiated cells [decrease ~11% vs. Glioma (non-rad) + AMI,
P<0.0001, post hoc Bonferroni)]. Radiation did not change the
TEMO effect. By contrast, treatment with AMI or TEMO attenuated the
effect of radiation vs. Glioma (rad), P<0.0001; post hoc
Bonferroni) and the co-administration of AMI + TEMO even reversed
its effect. In the radiated cultures that had been treated with AMI
and TEMO, the viability of the cells was comparable to the
viability of Glioma (non-rad) cells.
Mortality of the cells (%) analyzed in
EVE cell counter
The alterations in the mortality of the cells (%)
are presented in Fig. 1B. The
results of the two-way ANOVA revealed a significant interaction
between the two analyzed factors (factor 1: radiation; factor 2:
Studied drugs). The differences between the untreated and treated
cell cultures were significant (F(3,64)=311.6,
P<0.0001). The impact of radiation was also relevant
(F(1,64)=677.4, P<0.0001). Drug administration (AMI,
TEMO and combined AMI + TEMO) caused a significantly higher
mortality of the cells than was observed in the untreated cell
cultures [increase by ~23, 20 and 21%, respectively, vs. Glioma
(non-rad) P<0.0001; post hoc Bonferroni]. The mortality of the
cells (%) was definitely higher in the cultures that had been
exposed only to radiation [increase by ~57% vs. Glioma (non-rad),
P<0.0001; post hoc Bonferroni]. Treatment with AMI or TEMO
attenuated the radiation effect vs. Glioma (rad) (P<0.0001; post
hoc Bonferroni) and the combined treatment with both drugs, AMI +
TEMO, caused the most remarkable effect. In this group, the
mortality of the cells was similar as in Glioma (non-rad).
Proliferation of cells (BrdU positive
%)
The differences in the proliferation level (%) are
presented in Fig. 1C. The results
of the two-way ANOVA indicated a significant interaction between
the two analyzed factors (factor 1: radiation; factor 2: Studied
drugs). The influence of drug administration was strongly
significant (F(3,64)=187.1; P<0.0001). The same
statistical pattern was observed between the groups that had been
exposed to or had not been exposed to radiation
(F(1,64)=619.3; P<0.0001). Treatment with AMI, TEMO
and the co-treatment with AMI + TEMO reduced the proliferation of
cells (decrease of ~26, 34 and 34%, respectively). The effect of
radiation was stronger (decrease of ~60% vs. Glioma (non-rad);
P<0.0001, post hoc Bonferroni). Moreover, radiation enhanced the
effect of AMI or TEMO. Each drug reduced the proliferation of cells
that had been exposed to radiation more than the proliferation of
the cells that had not been exposed to radiation [by ~12% vs.
Glioma (non-rad) + AMI; by ~10% vs. Glioma (non-rad) + TEMO;
P<0.0001; post hoc Bonferroni)]. However, when used together,
both drugs attenuated the effect of radiation vs. Glioma (rad;
P<0.0001; post hoc Bonferroni). In the cultures that had been
exposed to radiation and AMI+TEMO, an increased proliferation of
cells was even observed [by ~35% vs. Glioma (non-rad) + AMI + TEMO;
P<0.0001; post hoc Bonferroni].
PDL-1 expression
The alterations in the PDL-1 expression (ng/ml) are
presented in Fig. 1D. The results
of the two-way ANOVA revealed a significant interaction between the
two analyzed factors (factor 1: radiation; factor 2: Studied
drugs). There were significant effects of the treatment (F
(3,64)=213.1; P<0.0001) and radiation
(F(1,64)=60.94, P<0.0001). The cells that had not
been exposed to radiation and that had been exposed to AMI
demonstrated a decrease in the PDL-1 expression (~12%; P<0.0001;
post hoc Bonferroni), but treatment with TEMO or co-treatment with
AMI + TEMO caused an increase in the PDL-1 expression [~11 and 9%,
respectively, vs. Glioma (non-rad); P<0.0001; post hoc
Bonferroni]. The radiation also increased the PDL-1 expression (by
~11%; P<0.0001; post hoc Bonferroni). In the radiated cultures
that had been exposed to AMI, the PDL-1 expression was still lower
than in the non-radiated glioma cultures (~6%; P<0.01; post hoc
Bonferroni) and were also lower than in the radiated cell cultures
(by 11%; P<0.0001; post hoc Bonferroni). Radiation did not
change the effect of TEMO and the co-treatment with AMI + TEMO.
Microscopy observations
The influence of one of the following factors: AMI,
TEMO, or radiation and their interactions on the C6 glioma cells
were not only assessed in the cell analysis but the morphological
changes in the cell cultures were also evaluated during a
microscopic analysis (the representative pictures of the
non-radiated cultures are presented in Fig. 2A-D and the radiated cultures in
Fig. 2F-H. In the control
(non-radiated and untreated) group, the C6 cells created a dense
network with tight connections (full confluence) (A). Exposure to
radiation at a single dose of 10 Gy induced a strong mortality
after 72 h (E). AMI caused a stronger cytotoxic effect on C6 glioma
cells (B) than TEMO (C) and AMI + TEMO (D). TEMO (C) and AMI + TEMO
(D) had a more intense formation of larger colonies than AMI alone
(B). The radiated cultures that had been exposed to AMI (F) were
characterized by a lower number of live cells than the non-radiated
cultures. By contrast, the application of TEMO (G) or AMI + TEMO
(H) did not induce such an effect. These cultures exhibited a full
confluence. TEMO (G) and AMI + TEMO (H) promoted the formation of
larger colonies. The microscopic analysis for each group was
performed in a bright field in 9 views using JuliStage (NanoEntek;
magnification, x40).
Discussion
Neuropathic pain and other complications, which are
a consequence surgical resection, radiotherapy, tumor presence, or
its recurrence, are a common problem in patients with cancer
including glioma (17). Several
drugs are used to relieve neuropathic pain, namely, some opioid
analgesics, anticonvulsant drugs as well as some antidepressants,
especially tricyclic antidepressants (AMI, nortriptyline; a
metabolite of AMI and clomipramine) are recommended (18). It is known that AMI induces an
analgesic effect quickly by modulating neuroinflammatory signaling
and later produces an antidepressant effect. There are also data
concerning its anticancer effects (19). Since not only neurons but also the
astroglia cells that built a tumor mass are able to take up
amitriptyline, the role of AMI as a potential adjuvant in glioma
chemoradiotherapy was the research problem analyzed for this
article.
In the present study, in order to mimic the
polytherapy that is used in patients with glioma, AMI (a potential
adjuvant drug) was administered in combination with TEMO and
radiation, which to some extent imitates modern stereotactic
radiotherapy. The effects of the drugs on glioma cells were
assessed after a single dose was administered into a culture medium
for 72 h, which was when a full culture confluence that mimicked
maximal tumor growth was achieved. The short duration of the
experiments was due to the rapid cytotoxic effect that was
observed. In the pilot study, it was observed that a single
exposure of C6 glioma cells to radiation at 10 Gy for 72 h induced
the death of 80% of the cells in the culture.
AMI +/- radio effects
AMI was used at a concentration of 10 µM because
this level can be achieved in the brain during treatment. In the
brain, the level of AMI is 13-16-fold higher than in serum
(20), where it reaches a
concentration from 0.15 to 0.7 µM (21,22).
It was found that AMI induced significant anticancer effects on C6
glioma cells that had been exposed to or had not been exposed to
radiation. AMI alone inhibited the viability, enhanced the
mortality of glioma cells and also induced an antiproliferative
effect. Moreover, its influence on the viability and proliferation
of glioma cells was augmented by radiation however, it was weaker
than the results of radiation alone.
The molecular mechanism of the interaction between
AMI and radiation in our experimental model is undoubtedly complex.
The observed differences in the response to AMI in the cultures
that had or had not been exposed to radiation might be partially
explained through the different time points of the DNA damage
(23) or probably the influence of
radiation and AMI on the cytokine network, especially IL-6. As was
shown in another in vitro study (24), IL-6 is released spontaneously by
glioma cells and stimulates an invasive tumor phenotype (high
proliferation, viability of cells, migration). Moreover, radiation
initiates the biological damage that is mediated by free radicals
and acts on cytokine production at the transcriptional or
translational level (25) in the
acute phase of the reaction. In some in vivo and clinical
studies, dose-and time-dependent effects of AMI on the
pro-inflammatory cytokines were observed. AMI decreased the release
of IL-6 in an acute and chronic model of inflammation (mouse sepsis
model, inflammation reaction to the implant in mice,
carrageenan-induced paw edema model, in animals with neuropathic
pain, or in patients with major depression) (26-29).
It can be assumed that decrease of IL-6 production might have
significance for the reduced viability and proliferation of glioma
cells that have been exposed to AMI and radiation compared with the
AMI effect alone that was observed in the present experiments. This
suggestion may be supported by observations by other authors that
when AMI is used at concentrations between 33-100 µM, it induces an
inhibitory effect on the III cancer respiratory chain while at
lower doses, it induces a radioprotective effect in normal
hippocampal cells (30). Although
numerous promising agents are currently being developed to
ameliorate the negative effects of radiotherapy, the range of doses
at which AMI might interfere with the radioprotective mechanisms
has not been assessed yet.
The unexpected lack of a synergistic effect with
radiation and the stronger effect alone of imipramine (tricyclic
antidepressant drug) was also observed by Royds et al
(31) on prostate cancer cells.
This effect was associated with blocking the voltage-gated
potassium channels (EAG1), and as a result, increased lipid
peroxidation, oxidative stress, and markedly altered cell integrity
and lipid membrane permeability.
The present study revealed that AMI significantly
reduced the expression of protein PD-L1 in glioma C6 cells for the
first time, which expands knowledge about the complex mechanisms of
action of this antidepressant including its anti-inflammatory
action. Similar data were observed in an ovarian cancer model when
a decrease in PDL-1 by AMI was associated with inhibiting
serotonin, TGM2 (transglutaminase 2), and KYN/Indole signaling,
which in effect can help overcome resistance and make immunotherapy
more effective (32,33).
A decrease in the PD-L1 expression was observed in
all of the cultures that had been exposed to or had not been
exposed to radiation, but it was weaker in the radiated cultures.
Immune checkpoints (PD-1/PD-L1) and inflammasomes, which are
characterized by an enhanced production of pro-inflammatory
cytokines such as interleukin (IL)-1β and tumor necrosis factor
(TNF-α), are important for the development of pyroptosis in cancer
cells (34). That is why these
cytokines also appear to be an attractive additional target for
future anticancer therapy. Unfortunately, in clinical studies, the
inhibition of only one factor, for example, only PDL-1 or epidermal
growth factor receptor has not yet produced the expected results in
glioma treatment (35). AMI, in
addition to its influence on the PDL-1 expression, induces
pleiotropic effects. The anti-inflammatory properties of AMI have
been proven in several studies. In the authors' previous in
vitro experiments, it was shown that AMI inhibited the
expression of subunit p65 of the NF-kappa B transcriptional factor,
the secretion of lactic acid into the medium, integrated into the
reactive oxygen species system, reduced the volume of swollen
mitochondria, and silenced the expression of the markers that are
characteristic for cancer cells in glioma (36). Moreover, in a previous study on
LPS-stimulated primary mixed glial and microglial cultures, it was
demonstrated that AMI and its metabolite nortriptyline inhibited
the release of the proinflammatory cytokines, TNF-α and IL-1β
(37).
The reported decrease in the PD-L1 expression in the
cells treated with AMI may be important for its anti-glioma effects
because other results have shown that glioma cells secrete PD-L1
into the peritumoral areas, particularly microglia, which contain
highly expressed PD-1(15). PD-L1
stimulates the PD-L1 receptors on microglial cells, which then
promotes tumor growth and invasion. Recently, researchers have
focused their attention on microglia as a potential and promising
target for the future immunotherapy of glioma. Microglia, which are
resident brain macrophages, play a key role in mediating the local
immune response. Specific microglial phenotypes (M1, M2), which are
characterized by their pro-or anti-inflammatory molecule
expression, have different functions (38) and may promote or inhibit tumor
growth. Although microglia cells determine the recruitment of
immunological cells into the tumor microenvironment (TME), there
are currently no methods for glioma therapy that target macrophage
polarization. Therefore, AMI may be considered as a potential
adjuvant drug in glioma therapy because of its anti-inflammatory
impact on glia as well as some peripheral immunomodulatory
properties. AMI suppresses interferon-γ production by the
Th1 cells, reduces T cell proliferation, and also has an
influence on the T cell phenotype by reducing the frequency of the
occurrence of CD8+CD45RA,
CD27+CD4+, and
CD27+CD8+ in peripheral blood mononuclear
cells (39).
Although the use of PD-1/PD-L1 inhibitors
(Nivolumab, Pembolizumab, Cemiplimab, Atezolizumab, Avelumab and
Durvalumab) have improved therapy in numerous types of cancer
(lung, melanoma and prostate cancer) (38), no therapeutic benefits have been
found in glioma. Despite the high expectations, in a large,
randomized study III phase Checkmate 143 (NCT02617589), Nivolumab
did not exhibit any significant benefits in glioma therapy
(40). The available literature
presents different, and occasionally, conflicting effects of the
interactions between the PD-L1 inhibitors and the drugs that are
used in the conventional therapy. Wang et al (41) demonstrated that the concomitant use
of TEMO and a PD-L1 inhibitor strongly reduced tumor growth and
improved the survival rate in a mouse model of glioma; however, in
patients with a recurrent tumor, this therapy did not have such
effects.
TEMO +/- radio effects
In the present study, TEMO (1 mM) affected the
viability and proliferation of glioma cells that were similar to
AMI but increased the expression of PD-L1 at a rate that was
similar to radiation alone. Other in vitro studies also
reported that TEMO induced the PD-L1 overexpression and researchers
have suggested that the mechanism of this effect can be mediated by
signal transducer and activator of transcription 3 signalization.
The increase in the PD-L1 expression induced by radiation is in
line with the observations of Xing et al (42), who proved that radiotherapy
increased the PD-L1 expression that was detected in glioma
specimens from 64 patients. Radiotherapy appears to increase the
MHC1-mediated antigen presentation and tumor lymphocyte
infiltration. These radiation effects appear to be partially
modulated by an increased expression of interferon-Y19(43). It has been hypothesized that in
response to the aforementioned radiation effects, the expression of
PD-L1 is higher in tumor cells in order to evade immune detection.
Furthermore, an acquired glioma resistance to the PD-1 inhibitors
can occur via mutations of the apelin receptor 20 and
interferon-receptor-associated Janus kinase 1 (JAK1) 21 both of
which lead to a decreased expression of interferon-Y as well as
tumor infiltration by immune cells (44).
AMI + TEMO +/- radio effects
Surprisingly, the concomitant use of rad + AMI +
TEMO has not shown synergistic anticancer effect, which was also
confirmed by the restoration of the full confluence or colony
formation that was observed during microscopic analysis. The
radiated cell cultures that had been treated with both drugs were
characterized by a significantly increased percentage of living
cells, cell division, and a reduced mortality of cells compared
with the radiated non-treated cultures, which means that
concomitant use of AMI + TEMO reversed the effect of radiation
alone. Although the molecular mechanism of this synergistic adverse
effect requires further investigation, it may be hypothesized that
GSCs could play an important role; on the one hand generated in
response to TEMO/radiotherapy and on the other had initiate
resistance to therapy (45).
Moreover, it has been proven that in in vitro conditions,
TEMO increased the HMGB1 expression that also promotes GSC
phenotype (46). The measurement
of the mitochondrial activity (MTT test) in the present experiments
found that the sensitivity of this parameter to the studied
combinations may also indicate the engagement of mitochondria in
the mechanism of AMI action and the effects that were observed as a
result of concomitant use of the AMI + TEMO interactions. The
modulation of mitochondrial activity by AMI has already been
described (47). These effects may
at least partially explain the formation of larger cellular
colonies in the radiated cultures treated with TEMO and, after they
had been treated with the concomitant use of AMI + TEMO.
Radiation triggers inter alia the depolarization of
the mitochondrial membrane, induces the release of cytochrome c
into the cytosol in response to apoptosis stimulus, and causes
oxidative stress (37). However, a
too low dose of radiation (single dose 10 Gy) could also escalate
the repair processes and accelerate the repopulation despite a
temporary therapeutic effect manifested by high mortality of C6
glioma cells, which in consequence could reverse the effect
concomitant use of AMI + TEMO (48). Moreover, exposure to a single dose
of radiation induces different effects than exposure to multiple
doses. Tzadok et al (49)
reported a dose-dependent inhibition of the viability of
astrocytoma U87 cells as a result of using antidepressants
(fluoxetine or sertraline) in increasing doses combined with
radiation. However, the inclusion of TEMO into the studied
combinations, namely, radiation + TEMO + antidepressant, did not
intensify this inhibition. This unexpected drug interaction may be
associated with a specificity of C6 glioma cell line in which
cytochrome P450 activation has been discovered, which potentially
alters drugs level, their effectiveness, and could lead to drug
antagonism (50). Similarly, in
another study, the concomitant use of fluoxetine + imatinib induced
a stronger anticancer effect than their combination with radiation
(51).
The last issue that could have affected the
anticancer effect of the studied drug combinations is the sequence
in which they were used. In clinical practice, in patients with
glioma, after surgical resection, the cytostatic drug TEMO is used
first followed by radiation (TEMO plays the role of a sensitizer on
radiation) (52). In experimental
in vitro models, following this order is not always possible
within one passage.
The aforementioned data indicate that further
research using other glioma cell lines and in vivo models
are undoubtedly required to properly explain the mechanisms of the
important interactions between radiation and potential anticancer
drugs.
Finally, the lack of synergism between AMI + TEMO +
radio can be explained by the fact that AMI is a modulator of
autophagy acting as ‘Trojan horse’. Depending on context, AMI in C6
cells can be an inducer or inhibitor of autophagy - the process
that protects cells against oxidative and genotoxic stressors (in
the initial stages, but in later phases it may contribute to cell
death) (53). Other studies
described that AMI may inhibit the efficacy of TEMO and
radiotherapy, particularly in gliomas, by interfering with cell
death pathways, potentially inducing autophagy, blocking
proapoptotic signals (such as TRAIL-dependent apoptosis), and
affecting DNA repair or cell cycle regulation. Although it may also
exhibit complex, occasionally synergistic, effects depending on the
context and cell type, often related to its effects on calcium,
ceramide, and various receptors, ultimately complicating the
efficacy of standard treatment (54,55).
Moreover, in C6 glioma cells, AMI may also have a
direct ‘postsynaptic’ effect, increasing the availability of
signaling molecules such as Gsα, potentially modulating cell
survival pathways. Indeed, AMI's effects on C6 cells span a broad
network of non-dopaminergic receptors and intracellular pathways,
creating unique drug interaction profiles that are currently being
investigated for cancer therapy (56).
In conclusion, AMI is a drug that has a high
biological activity and a multi-directional mechanism of action
that induces dose- and time-dependent effects. Although this
antidepressant has been used for numerous years, its
pharmacological properties against cancers, and its position in
cancer therapy have not yet been fully recognized.
The presented results of an in vitro study
support other studies concerning the role of antidepressants as
active players in the TME. The present study highlights some
promising anticancer effects in C6 glioma cells cultures that were
induced by AMI alone. The results suggest that the position of AMI
in glioma therapy may be important especially for its novel
mechanism, which is associated with decreasing expression of
PDL-immune checkpoints. In the clinic, this immunomodulatory action
might help overcome resistance, enhance immunotherapy, improve OS
in patients with glioma, or open the door to new combination
strategies (57,58). However, to achieve this effect, AMI
should be probably used with TEMO and radiation in time intervals,
which will require further analyses. At the same time, the current
study presents a second ‘face’ and type of action of AMI on C6
glioma that depends on its being used alone or concomitantly with
TEMO or radiation.
Although anticancer effects of AMI are interesting,
there are certain limitations to the present study. An in
vitro model does not reflect the immunological, hormonal and
cross-talk interactions between a tumor and its niche, which is why
it is difficult to uncritically extrapolate the obtained results to
clinical practice. In in vitro experiments, only the direct
effects of the tested drugs on cell cultures were examined but not
the effects of their active metabolites. However, this does not
change the fact that AMI appears to be a favorable candidate for
drug repositioning strategy in further research of glioma. Finally,
an estimation the cytotoxic/antiproliferative properties of AMI,
its modulatory effect on the PD-L1 expression, and its influence on
colonies should be verified in in vivo studies in order to
precisely estimate its potential as an adjuvant therapy.
Acknowledgements
Not applicable.
Funding
Funding: The present study was supported by the School of
Medicine, Medical University of Silesia, (grant no.
PCN-1-141/N/0/O).
Availability of data and materials
The datasets generated during and/or analyzed during
the current study are available from the corresponding author upon
reasonable request.
Authors' contributions
AMBW and EO conceptualized and supervised the study,
prepared and revised the manuscript. AMBW performed the cell
cultures. WM provided substantive support in the analysis of
results. KS operated the radiotherapy machine and assisted during
the radiation process. TC assisted in preparing the C6 cells for
radiation and enabling cell irradiation. MG performed statistical
analysis and the graphical presentation of the results. All authors
read and approved the final version of the manuscript. All authors
confirm the authenticity of all the raw data.
Ethics approval and consent to
participate
Not applicable.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
McKinnon C, Nandhabalan M, Murray SA and
Plaha P: Glioblastoma: Clinical presentation, diagnosis, and
management. BMJ. 374(n1560)2021.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Tan SK, Jermakowicz A, Mookhtiar AK,
Nemeroff CB, Schürer SC and Ayad NG: Drug repositioning in
glioblastoma: A pathway perspective. Front Pharmacol.
9(218)2018.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Abadi B, Shahsavani Y, Faramarzpour M,
Rezaei N and Rahimi HR: Antidepressants with antitumor potential in
treating glioblastoma: A narrative review. Fundam Clin Pharmacol.
36:35–48. 2022.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Martino M, Rocchi G, Escelsior A and
Fornaro M: Immunomodulation mechanism of antidepressants:
Interactions between serotonin/norepinephrine balance and Th1/Th2
balance. Curr Neuropharmacol. 10:97–123. 2012.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Jia X, Gao Z and Hu H: Microglia in
depression: Current perspectives. Sci China Life Sci. 64:911–925.
2021.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Frizzo ME and Ohno Y: Perisynaptic
astrocytes as a potential target for novel antidepressant drugs. J
Pharmacol Sci. 145:60–68. 2021.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Castren E: Neurotrophic effects of
antidepressant drugs. Curr Opin Pharmacol. 4:58–64. 2004.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Bortolato B, Hyphantis TN, Valpione S,
Perini G, Maes M, Morris G, Kubera M, Köhler CA, Fernandes BS,
Stubbs B, et al: Depression in cancer: The many biobehavioral
pathways driving tumor progression. Cancer Treat Rev. 52:58–70.
2017.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Steingart AB and Cotterchio M: Do
antidepressants cause, promote, or inhibit cancers? J Clin
Epidemiol. 48:1407–1412. 1995.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Dehghani A, Tabaku A and Eshghjoo S: The
tricyclic antidepressant amitriptyline is cytotoxic to human breast
cancer (MCF7) cells. J Biol Stud. 5(1):85–92. 2022.
|
|
11
|
Quail DF and Joyce JA: The
microenvironmental landscape of brain tumors. Cancer Cell.
31:326–341. 2017.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Hab Y, Liu D and Li L: PD-1/PD-L1 pathway:
Current researches in cancer. Am J Cancer Res. 10:727–742.
2020.PubMed/NCBI
|
|
13
|
Dunai C and Murphy WJ: NK cells for
PD-1/PD-L1 blockade immunotherapy: Pinning down the NK cell. J Clin
Invest. 128:4251–4253. 2018.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Wu J and Wang N: Current progress of
antiPD1/PDL1 immunotherapy for glioblastoma (Review). Mol Med Rep.
30(221)2024.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Zeng YF, Wei XY, Guo QH, Chen SY, Deng S,
Liu ZZ, Gong ZC and Zeng WJ: The efficacy and safety of
anti-PD-1/PD-L1 in treatment of glioma: A single-arm meta-analysis.
Front Immunol. 14(1168244)2023.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Bielecka AM and Obuchowicz E: Chronic
physiological hypoxia and high glucose concentration promote
resistance of T98G glioblastoma cell line to temozolomide. Drug
Des. 3(117)2014.
|
|
17
|
Horan NA and Pugh TM: Intractable central
pain in a patient with diffuse glioma. Am J Phys Med Rehabil.
98:e107–e110. 2019.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Sindrup SH, Otto M, Finnerup NB and Jensen
TS: Antidepressants in the treatment of neuropathic pain. Basic
Clin Pharmacol Toxicol. 96:399–409. 2005.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Gałecki P, Wójcik JM and Talarowska M: The
anti-inflammatory mechanism of antidepressants. Prog
Neuropsychopharmacol Biol Psychiatry. 80:291–294. 2018.PubMed/NCBI View Article : Google Scholar
|
|
20
|
Miyake K, Fukuchi K, Kitaura T, Kimura M
and Kimura Y: Pharmacokinetic analysis of amitriptyline and its
demethylated metabolite in serum and brain of rats after acute and
chronic oral administration of amitriptyline. J Pharmacobiodyn.
15:157–166. 1992.PubMed/NCBI View Article : Google Scholar
|
|
21
|
Shulz P, Dick P, Blaschke TF and Hollister
L: Discrepancies between pharmacokinetic studies of amitriptyline.
Clin Pharmacokinet. 10:257–268. 1985.PubMed/NCBI View Article : Google Scholar
|
|
22
|
Daniel WA, Syrek M, Haduch A and
Wójcikowski J: Different effects of amitriptyline and imipramine on
the pharmacokinetics and metabolism of perazine in rats. J Pharm
Pharmacol. 52:1473–1481. 2000.PubMed/NCBI View Article : Google Scholar
|
|
23
|
Morgan MA and Lawrence TS: Molecular
pathways: Overcoming radiation resistance by targeting DNA damage
response pathways. Clin Cancer Res. 21:2898–2904. 2021.PubMed/NCBI View Article : Google Scholar
|
|
24
|
West AJ, Tsui V, Stylli SS, Nguyen HPT,
Morokoff AP, Kaye AH and Luwor RB: The role of interleukin-6-STAT3
signalling in glioblastoma. Oncol Lett. 16:4095–4104.
2018.PubMed/NCBI View Article : Google Scholar
|
|
25
|
Singh M, Raghaw A and Gautam KA: Role of
the circulatory interleukin-6 in the pathogenesis of gliomas: A
systematic review. World J Methodol. 12:428–437. 2022.PubMed/NCBI View Article : Google Scholar
|
|
26
|
Jiang S, Shi D, Bai L, Niu T, Kang R and
Liu Y: Inhibition of interleukin-6 trans-signaling improves
survival and prevents cognitive impairment in a mouse model of
sepsis. Int Immunopharmacol. 119(110169)2023.PubMed/NCBI View Article : Google Scholar
|
|
27
|
Cruz-Garcia L, Nasser F, O'Brien G, Grepl
J, Vinnikov V, Starenkiy V, Artiukh S, Gramatiuk S and Badie C:
Transcriptional dynamics of DNA damage responsive genes in
circulating leukocytes during radiotherapy. Cancers (Basel).
14(2649)2022.PubMed/NCBI View Article : Google Scholar
|
|
28
|
Serizawa K, Tomizawa-Shinohara H, Miyake
S, Yogo K and Matsumoto Y: Interleukin-6: Evolving role in the
management of neuropathic pain in neuroimmunological disorders.
Inflamm Regen. 41(34)2021.PubMed/NCBI View Article : Google Scholar
|
|
29
|
Amdekar S, Roy P, Singh V, Kumar A, Singh
R and Sharma P: Anti-inflammatory activity of lactobacillus on
carrageenan-induced paw edema in male Wistar rats. Int J Inflam.
2012(752015)2012.PubMed/NCBI View Article : Google Scholar
|
|
30
|
Guo YR, Liu ZW, Peng S, Duan MY, Feng JW,
Wang WF, Xu YH, Tang X, Zhang XZ, Ren BX and Tang FR: The
neuroprotective effect of amitriptyline on radiation-induced
impairment of hippocampal neurogenesis. Dose Response.
17(1559325819895912)2019.PubMed/NCBI View Article : Google Scholar
|
|
31
|
Royds J, Conroy MJ, Dunne MR, McCrory C
and Lysaght J: An investigation into the modulation of T cell
phenotypes by amitriptyline and nortriptyline Eur.
Neuropsychopharmacol. 31:131–144. 2020.PubMed/NCBI View Article : Google Scholar
|
|
32
|
Zaltron E, Vianello F, Ruzza A, Palazzo A,
Brillo V, Celotti I, Scavezzon M, Rossin F, Leanza L and Severin F:
The role of transglutaminase 2 in cancer: An update. Int J Mol Sci.
25(2797)2024.PubMed/NCBI View Article : Google Scholar
|
|
33
|
Li J, Mei B, Feng L, Wang X, Wang D, Huang
J and Zhang G: Amitriptyline revitalizes ICB response via dually
inhibiting Kyn/Indole and 5-HT pathways of tryptophan metabolism in
ovarian cancer. iScience. 27(111488)2024.PubMed/NCBI View Article : Google Scholar
|
|
34
|
Jiang M, Liu C, Ding D, Tian H and Yu C:
Comparative efficacy and safety of Anti-PD-1/PD-L1 for the
Treatment of non-small cell lung cancer: A network meta-analysis of
13 Randomized controlled studies. Front Oncol.
12(827050)2022.PubMed/NCBI View Article : Google Scholar
|
|
35
|
Perrichet A, Ghiringhelli F and Rébé C:
Understanding inflammasomes and PD-1/PD-L1 crosstalk to improve
cancer treatment efficiency. Cancers (Basel).
12(3550)2020.PubMed/NCBI View Article : Google Scholar
|
|
36
|
Bielecka-Wajdman AM, Lesiak M, Ludyga T,
Sieroń A and Obuchowicz E: Reversing glioma malignancy: A new look
at the role of antidepressant drugs as adjuvant therapy for
glioblastoma multiforme. Cancer Chemother Pharmacol. 79:1249–1256.
2017.PubMed/NCBI View Article : Google Scholar
|
|
37
|
Bielecka-Wajdman AM, Ludyga T, Machnik G,
Gołyszny M and Obuchowicz E: Tricyclic antidepressants modulate
stressed mitochondria in glioblastoma multiforme cells. Cancer
Control. 25(1073274818798594)2018.PubMed/NCBI View Article : Google Scholar
|
|
38
|
Obuchowicz E, Kowalski J, Labuzek K,
Krysiak R, Pendzich J and Herman ZS: Amitriptyline and
nortriptyline inhibit interleukin-1 release by rat mixed glial and
microglial cell cultures. Int J Neuropsychopharmacol. 9:27–35.
2006.PubMed/NCBI View Article : Google Scholar
|
|
39
|
Geribaldi-Doldán N, Fernandez-Ponce C,
Quiroz RN, Sanchez-Gomar I, Escorcia LG, Velasquez EP and Qiiroz
EN: The role of microglia in glioblastoma. Front.Oncol.
10(603495)2021.PubMed/NCBI View Article : Google Scholar
|
|
40
|
Li W, Wu F, Zhao S, Shi P, Wang S and Cui
D: Correlation between PD-1/PD-L1 expression and polarization in
tumor-associated macrophages: A key player in tumor immunotherapy.
Cytokine Growth Factor Rev. 67:49–57. 2022.PubMed/NCBI View Article : Google Scholar
|
|
41
|
Wang Y, Zhou S, Yang F, Qi X, Wang X, Guan
X, Shen C, Duma N, Vera Aguilera J, Chintakuntlawar A, et al:
Treatment-related adverse events of PD-1 and PD-L1 inhibitors in
clinical trials: A systematic review and meta-analysis. JAMA Oncol.
5:1008–1019. 2019.PubMed/NCBI View Article : Google Scholar
|
|
42
|
Xing Y, Chand G, Liu C, Cook GJR,
O'Doherty J, Zhao L, Wong NCL, Meszaros LK, Ting HH and Zhao J:
Early phase I Study of a Tc-Labeled anti-programmed death ligand-1
(PD-L1) single-domain antibody in SPECT/CT assessment of PD-L1
expression in non-small cell lung cancer. J Nucl Med. 60:1213–1220.
2019.PubMed/NCBI View Article : Google Scholar
|
|
43
|
Park B, Yee C and Lee KM: The effect of
radiation on the immune response to cancers. Int J Mol Sci.
15:927–943. 2014.PubMed/NCBI View Article : Google Scholar
|
|
44
|
Yang Y, Chen M, Qiu Y, Li X, Huang Y and
Zhang W: The Apelin/APLNR system modulates tumor immune response by
reshaping the tumor microenvironment. Gene.
834(146564)2022.PubMed/NCBI View Article : Google Scholar
|
|
45
|
Snerdi D, Moutafi M, Kotsantis I,
Stavrinou LC and Psyrri A: Overcoming resistance to temozolomide in
glioblastoma: A scoping review of preclinical and clinical data.
Life (Basel). 14(673)2024.PubMed/NCBI View Article : Google Scholar
|
|
46
|
Pineda B, Sánchez García FJ, Olascoaga NK,
Pérez de la Cruz V, Salazar A, Hernández Pedro N, Márquez-Navarro
A, Ortiz Plata A and Sotelo J: Malignant glioma therapy by
vaccination with irradiated C6 cell-derived microvesicles promotes
antitumoral immune response. Mol Ther. 27:1612–1620.
2019.PubMed/NCBI View Article : Google Scholar
|
|
47
|
Gao XY, Zang J, Zheng MH, Zhang YF, Yue
KY, Cao XL, Cao Y, Li XX, Han H, Jiang XF and Liang L: Temozolomide
treatment induces HMGB1 to promote the formation of glioma stem
cells via the TLR2/NEAT1/Wnt pathway in glioblastoma. Front Cell
Dev Biol. 9(620883)2021.PubMed/NCBI View Article : Google Scholar
|
|
48
|
Gogvadze V, Orrenius S and Zhivotovsky B:
Multiple pathways of cytochrome c release from mitochondria in
apoptosis. Biochim Biophys Acta. 1757:639–647. 2006.PubMed/NCBI View Article : Google Scholar
|
|
49
|
Tzadok S, Berry E, Israeli M, Uziel O,
Lahav M, Fenig E, Gil-Ad I, Weizman A and Nordenberg J: In vitro
novel combinations of psychotropics and anticancer modalities in
U87 human glioblastoma cells. Int J Oncol. 37:1043–1051.
2010.PubMed/NCBI View Article : Google Scholar
|
|
50
|
Hu G, Fang Y, Xu H, Wang G, Yang R, Gao F,
Wei Q, Gu Y, Zhang C, Qiu J, et al: Identification of cytochrome
P450 2E1 as a novel target in glioma and development of its
inhibitor as an anti-tumor agent. Adv Sci (Weinh).
10(e2301096)2023.PubMed/NCBI View Article : Google Scholar
|
|
51
|
Hosseinimehr SJ, Najafi SH, Shafiee F,
Hassanzadeh S, Farzipour S, Ghasemi A and Asgarian-Omran H:
Fluoxetine as an antidepressant medicine improves the effects of
ionizing radiation for the treatment of glioma. J Bioenerg
Biomembr. 52:165–174. 2020.PubMed/NCBI View Article : Google Scholar
|
|
52
|
Stupp R, Mason WP, van den Bent MJ, Weller
M, Fisher B, Taphoorn MJ, Belanger K, Brandes AA, Marosi C, Bogdahn
U, et al: Radiotherapy plus concomitant and adjuvant temozolomide
for glioblastoma. N Engl J Med. 352:987–996. 2005.PubMed/NCBI View Article : Google Scholar
|
|
53
|
Hu G, Fang Y, Xu H, Wang G, Yang R, Gao F,
Wei Q, Gu Y, Zhang C, et al: Identification of cytochrome P450 2E1
as a novel target in glioma and development of its inhibitor as an
anti-tumor agent. Adv Sci. 23(e2301096)2023.doi:
10.1002/advs.202301096.
|
|
54
|
Kwon Y, Bang Y, Moon SH, Kim A and Choi
HJ: Amitriptyline interferes with autophagy-mediated clearance of
protein aggregates via inhibiting autophagosome maturation in
neuronal cells. Cell Death Dis. 11(874)2020.PubMed/NCBI View Article : Google Scholar
|
|
55
|
He L, Fu Y, Tian Y, Wang X, Zhou X, Ding
RB, Qi X and Bao J: Antidepressants as autophagy modulators for
cancer therapy. Molecules. 28(7594)2023.PubMed/NCBI View Article : Google Scholar
|
|
56
|
Adornetto A, Lagana ML, Satriano A,
Licastro E, Corasaniti MT, Bagetta G and Russo R: The
antidepressant drug amitriptyline affects human SH-SY5Y
neuroblastoma cell proliferation and modulates autophagy. Int J Mol
Sci. 25(10415)2024.PubMed/NCBI View Article : Google Scholar
|
|
57
|
Pu T, Sun J, Ren G and Li H: Neuro-immune
crosstalk in cancer: Mechanisms and therapeutic implications.
Signal Transduct Target Ther. 10(176)2025.PubMed/NCBI View Article : Google Scholar
|
|
58
|
Pant A and Lim M: Advances in co-opting
anti-depressant drugs in glioma therapy. Cell Rep Med.
3(100837)2022.PubMed/NCBI View Article : Google Scholar
|