Introduction
Superior mesenteric artery (SMA) syndrome (SMAS) is
a rare gastrointestinal disease, initially having been reported in
1842. Wilkie (1) reported the
disease in more detail in 1927. Based on this report, SMAS has been
termed as ‘Wilkie's syndrome’. The angle between the aorta and SMA
of a normal individual ranges from 38˚ to 56˚. A mesenteric fat pad
is present within this angle to relieve compression. SMAS occurs
when this angle becomes narrow due to various reasons, including
gastrointestinal dyskinesia, malabsorption, peritoneal adhesions
and corrective surgery for spinal diseases, leading to the
oppression of the distal end of the duodenum (2). Patients usually present with symptoms
similar to intestinal obstruction, including abdominal distension
and frequent vomiting (3). Frequent
vomiting and the loss of digestive juices may lead to further
weight loss and aggravated duodenal compression, resulting in the
formation of a continuous cycle of severe symptoms. This syndrome
is usually related to certain anatomical variations, including a
weak body type, shortening of the Treitz ligament, a higher
physiological position of the duodenum and an abnormally low
position of the SMA (4). The risk
factors which have been reported include gastrointestinal
dyskinesia, malabsorption, peritoneal adhesion, and corrective
surgery for spinal diseases (2).
SMAS can be caused by aortomesenteric angle
reduction. In patients presenting with abdominal pain, nausea,
anorexia, weight loss and vomiting, the diagnosis of SMA syndrome
should be considered (5). The
diagnostic criteria for SMA include an aortomesenteric angle
<20˚ and a aortomesenteric distance <8 mm with gastric and
proximal duodenal dilatation (6).
The typical barium swallow (upper gastrointestinal series) of SMAS
reveals abrupt or nearly total cessation of the barium flow from
the duodenum to the jejunum (7).
Nutritional support treatment leads to a higher rate of symptom
relief. In a previous study, in a modern case series of 22 children
managed for SMAS, the symptom relief rate of non-surgical treatment
was 86% (8). Weight gain may
increase fat in the mesenteric fat pad, covering the third part of
the duodenum to relieve duodenal compression (9). Early surgery should be considered for
patients who have failed nutritional support treatment. Surgical
options include the lysis of the Treitz ligament, gastrojejunostomy
or duodenojejunostomy (5,10-13).
The present study describes two cases of SMAS which
occurred following esophageal cancer surgery and describes the
symptoms presented and the treatments used. It is hoped that the
findings presented herein may aid in the treatment of other similar
cases in the future.
Case report
The first case included a 68-year-old female patient
who was diagnosed with squamous cell carcinoma, due to weight loss
(4 kg) and intermittent dysphagia. The computerized tomography (CT)
scan (PHILIPS Brilliance 64) of the abdomen prior to surgery did
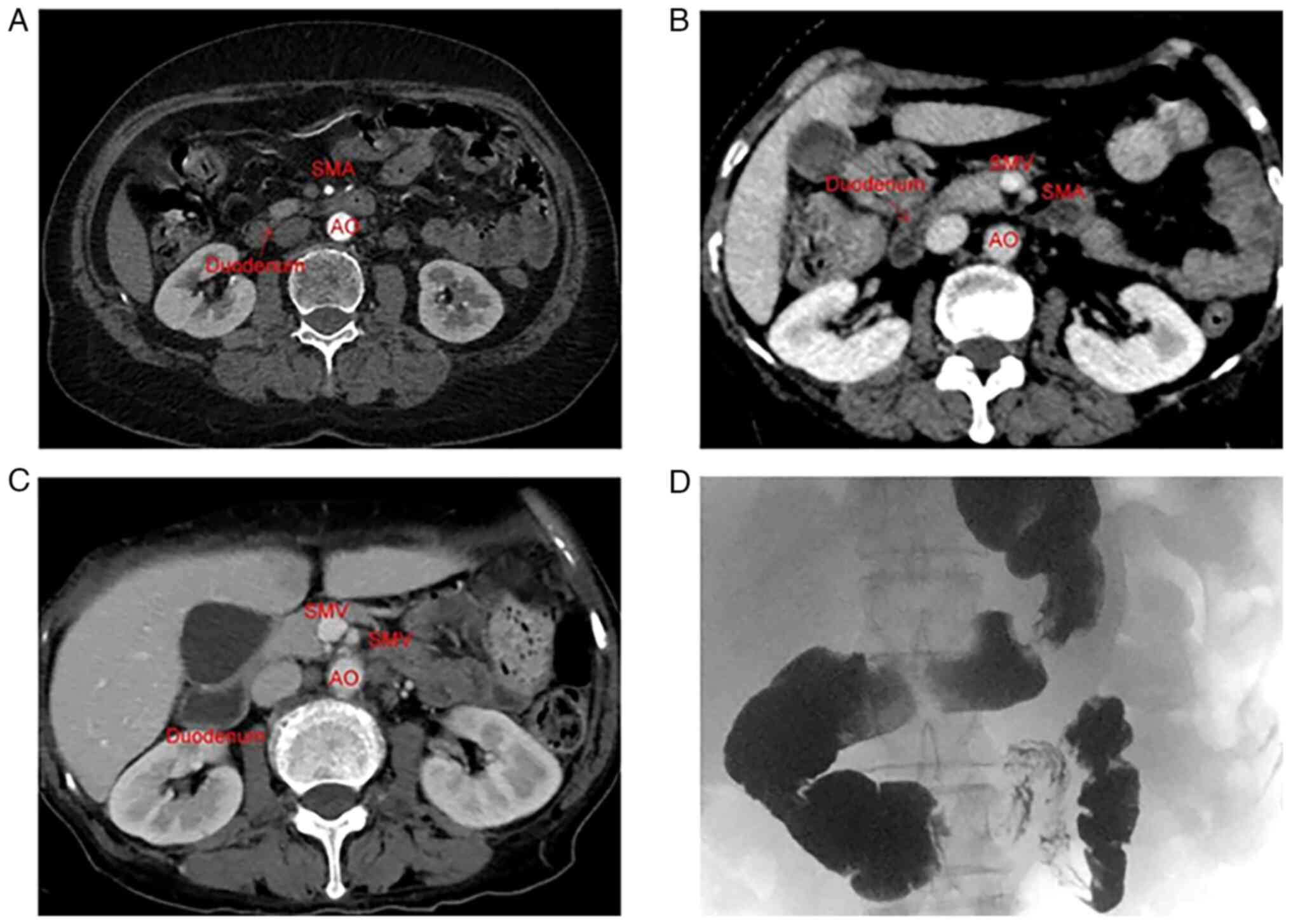
not reveal any abnormalities (Figs.
1A and 2A). Surgical resection
of the esophageal cancer was performed by trans-left thoracic
esophagectomy at the People's Hospital of Kai Zhou District on
January 26, 2021. The post-operative pathological examination
revealed moderately differentiated esophageal squamous cell
carcinoma. The pathological stage was determined as T3N0M0.
An esophageal lipiodol angiography confirmed the
absence of esophagogastric anastomotic fistula on post-operative
day 7. Therefore, the patient began drinking water and receiving
oral liquid food; however, the patient also presented with a poor
appetite and exhibited satiety following the consumption of only a
limited portion of food. On the 10th day after surgery, the patient
began to complain of symptoms of acid reflux and belching, which
became more apparent following food consumption. These symptoms
were not relieved after the administration of mosapride and
omeprazole. The patient developed symptoms of bloating and frequent
vomiting within a few minutes to half an hour after eating on
post-operative day 13. Initially, it was considered that these
symptoms were caused by post-operative gastroparesis and the
patient was asked to suspend oral intake. The patient underwent a
CT scan of the abdomen on post-operative day 16 (Fig. 1B), and no apparent abnormalities were
observed. Following 7 days of parenteral nutritional support, the
patient's vomiting symptoms gradually subsided and she was
discharged on the 20th day post-surgery. The patient's
pre-operative body mass index (BMI) was 25.5 kg/m2,
which was decreased to 20.8 kg/m2 at the time of
discharge.
During the first few months after the surgery, the
patient repeatedly experienced a series of symptoms, including
abdominal distension, vomiting and loss of appetite. Therefore, she
received parenteral nutrition treatment at our department several
times. Due to repeated hospitalizations, she was examined using
barium swallow (upper gastrointestinal series) and an enhanced CT
scan of the abdomen was performed on May 15, 2021, which indicated
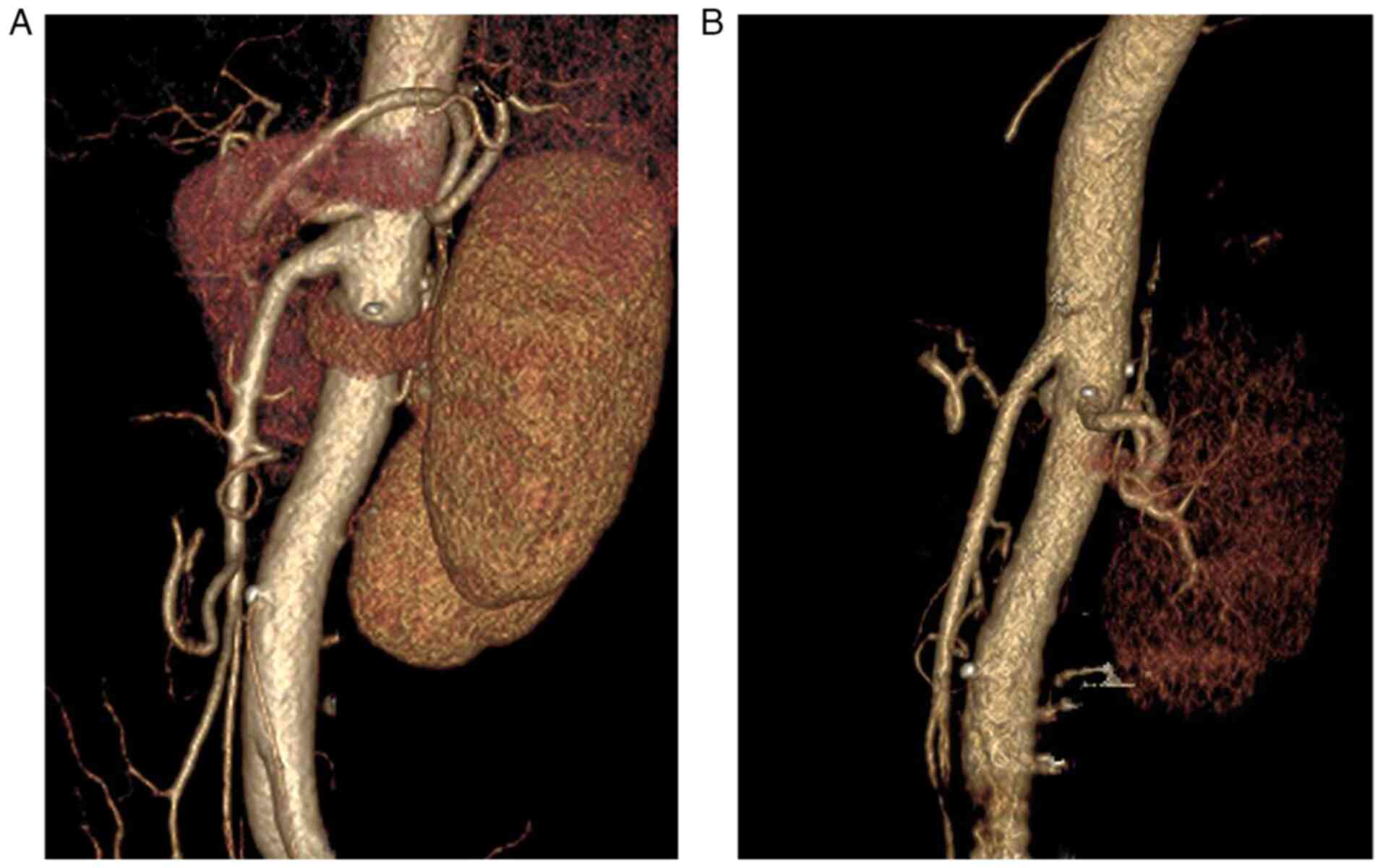
an apparent duodenal compression (Figs.
1C and D, and 2B). Therefore, the diagnosis of SMAS was
confirmed. Since the symptoms of bloating and vomiting were not
relieved following nutritional support, the patient underwent a
duodenojejunostomy on May 20, 2021. During the surgery, the
proximal duodenal bowel of the SMA was dilated, whereas the distal
intestinal tube was free and smooth, indicating that the duodenum
was compressed by the SMA. Following post-operative nutritional
support, the symptoms of vomiting and abdominal distension were
relieved. She was discharged 2 weeks after the surgery, having a
BMI of 23.5 kg/m2.
The second case included a 66-year-old female
patient who underwent radical esophageal cancer surgery four years
ago. She did not receive systemic chemotherapy following the
surgery, and other detailed surgical information was not available.
The post-operative pathological examination revealed moderately
differentiated esophageal squamous cell carcinoma. The pathological
stage was determined as T2N0M0. At 1 month prior to admission to
our hospital, the patient exhibited abdominal distension and
intermittent vomiting without apparent cause. The symptoms did not
resolve spontaneously during that time, and she was admitted to our
hospital for further treatment. Following admission to the
hospital, the patient underwent barium swallow (upper
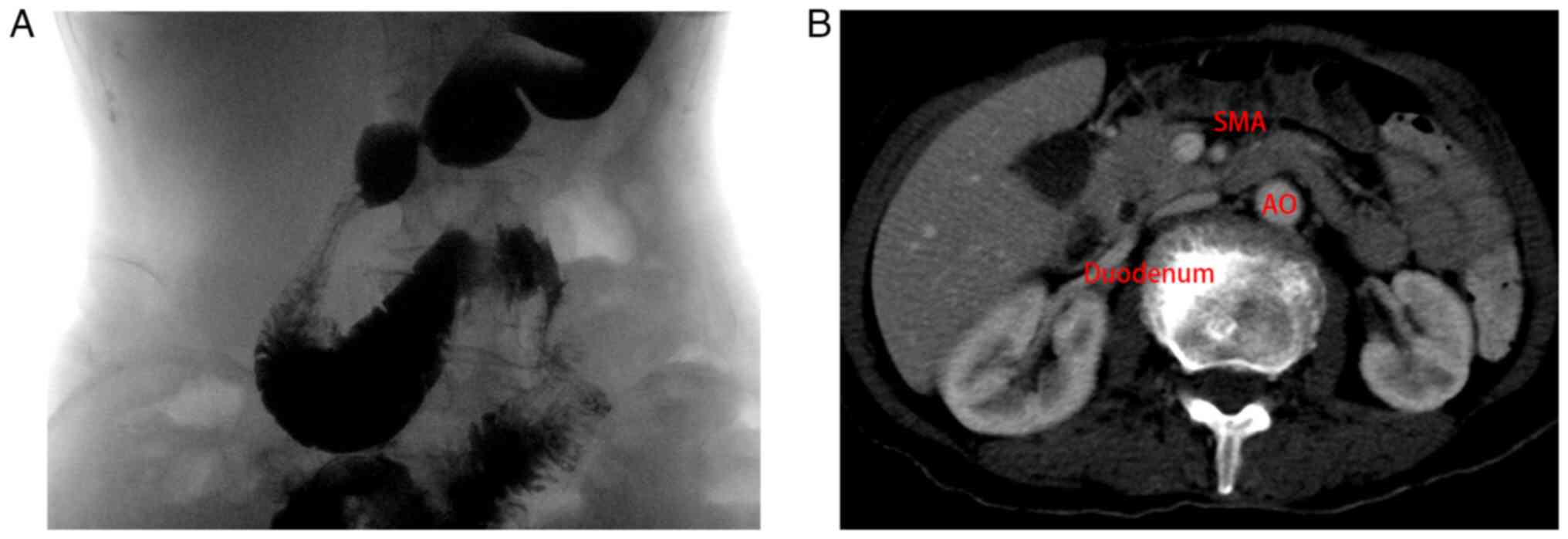
gastrointestinal series) (Fig. 3),
which indicated that the third part of the duodenum was compressed,
and the enhanced CT scan of the abdomen revealed that the proximal
duodenal bowel of SMA was dilated. She was diagnosed with SMAS due
to her typical clinical symptoms and specific imaging studies.
Following 11 days of nutritional support, the symptoms of vomiting
and abdominal distension were relieved. At 1 week after being
discharged from the hospital, the patient's vomiting or abdominal
distension after eating was completely relieved, and the current
BMI has increased from 17.9 kg/m2 at admission to 19.2
kg/m2.
Discussion
In the first case of the present case report, the
most frequently used duodenal-jejunal anastomosis was used. On the
7th day following operation, the patient had his stomach tube
removed and started eating. The symptoms of bloating and vomiting
were significantly relieved. A duodenojejunostomy involves an
end-to-side anastomosis between the duodenum and jejunum, bypassing
the SMA and reducing compression. It has been reported that the
symptom relief rate of duodenojejunostomy reaches 90% (14). The symptoms of the second patient
were relieved by providing nutritional support treatment for 11
days.
No standard guidelines have been reported regarding
the indications for surgery in this rare syndrome. In the second
case, the patient's symptoms were gradually relieved after 11 days
of parenteral nutrition treatment. It is considered that
conservative treatment for patients with mild SMAS can be extended
to 4 weeks. During treatment, oral enteral nutrition preparations
can be attempted (15). When
vomiting and abdominal distension are relieved, enteral and
parenteral nutrition can be performed simultaneously. If the
patient presents with severe abdominal distension, frequent
vomiting, electrolyte balance disorder, or the symptoms of
conservative treatment for a long period of time without relief,
early surgical intervention should be considered. By contrast, for
those patients with esophageal cancer and severe preoperative
nutritional impairment, long-term eating difficulties, and/or in
elderly patients, post-operative prefabricated jejunostomy may be
used to reduce the incidence of SMAS. However, the risk of
prefabricated jejunostomy must be carefully evaluated prior to the
surgery.
SMAS is a rare disease encountered in clinical
practice and it is even rarer in patients undergoing radical
resection of esophageal cancer combined with SMAS. The authors
believe that the main reasons for this include the following: i)
The typical symptoms of milder SMAS are similar to the
gastrointestinal symptoms of gastrointestinal reconstruction,
following esophageal cancer resection; ii) routine examinations,
including esophageal iodine contrast, chest and abdomen CT scans,
and electronic gastroscopy exhibit decreased sensitivity in disease
diagnosis; and iii) the severe obstructive symptoms of SMAS are
similar to the symptoms of viscous intestinal obstructions, which
may mislead the attending physician. Both patients reported in the
present study underwent several CT scans, esophageal lipiodol and
electronic gastroscopy. The SMAS of these two patients was not
diagnosed at an early stage by multiple examinations, prolonging
the treatment period and hospital stay. The early diagnosis of
esophageal cancer is difficult. The slow progress of eating
obstruction in patients with esophageal cancer may lead to a poor
nutritional status, and the radical resection of esophageal cancer
requires the removal of the majority of the stomach and the
repositioning of the stomach into the thoracic cavity, which may
result in an upward displacement of the horizontal part of the
duodenum. Those processes may also cause SMAS.
In conclusion, the diagnosis of SMAS should be
considered in patients following esophagectomy who have a poor oral
intake and present with bloating, vomiting and other
gastrointestinal obstructions.
Acknowledgements
Not applicable.
Funding
Funding: No funding was received.
Availability of data and materials
Not applicable.
Authors' contributions
YZ and ZLY designed the study and drafted the
initial manuscript. ZLY and ZW collected the original data and
information. YZ and ZLY confirm the authenticity of all the raw
data. All authors have read and approved the final manuscript.
Ethics approval and consent to
participate
The patients described herein provided their consent
for their inclusion in the present case report, and written consent
was obtained and stored electronically as a PDF.
Patient consent for publication
The patients provided their consent for the
publication of personal data, and written consent was obtained and
stored electronically as a PDF.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Wilkie DP: Chronic duodenal ileus. Br J
Surg. 9:204–214. 1921.
|
|
2
|
Diab S and Hayek F: Combined superior
mesenteric artery syndrome and nutcracker syndrome in a young
patient: A case report and review of the literature. Am J Case Rep.
21(e922619)2020.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Baltazar U, Dunn J, Floresguerra C,
Schmidt L and Browder W: Superior mesenteric artery syndrome: An
uncommon cause of intestinal obstruction. South Med J. 93:606–608.
2000.PubMed/NCBI
|
|
4
|
Rai RR, Shah S, Palliyil NS, Dalvie S and
Shah R: Superior mesenteric artery syndrome complicating spinal
deformity correction surgery a case report and review of the
literature. JBJS Case Connect. 9(e0497)2019.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Merrett ND, Wilson RB, Cosman P and
Biankin AV: Superior mesenteric artery syndrome: Diagnosis and
treatment strategies. J Gastrointest Surg. 13:287–292.
2009.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Hines JR, Gore RM and Ballantyne GH:
Superior mesenteric artery syndrome. Diagnostic criteria and
therapeutic approaches. Am J Surg. 148:630–632. 1984.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Reckler JM, Bruck HM, Munster AM, Curreri
PW and Pruitt BA Jr: Superior mesenteric artery syndrome as a
consequence of burn injury. J Trauma. 12:979–985. 1972.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Biank V and Werlin S: Superior mesenteric
artery syndrome in children: A 20-year experience. J Pediatr
Gastroenterol Nutr. 42:522–525. 2006.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Sun Z, Rodriguez J, McMichael J, Walsh RM,
Chalikonda S, Rosenthal RJ, Kroh MD and El-Hayek K: Minimally
invasive duodenojejunostomy for superior mesenteric artery
syndrome: A case series and review of the literature. Surg Endosc.
29:1137–1144. 2015.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Wilson-Storey D and MacKinlay GA: The
superior mesenteric artery syndrome. J R Coll Surg Edinb.
31:175–178. 1986.PubMed/NCBI
|
|
11
|
Yao SY, Mikami R and Mikami S: Minimally
invasive surgery for superior mesenteric artery syndrome: A case
report. World J Gastroenterol. 21:12970–12975. 2015.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Strong EK: Mechanics of arteriomesentric
duodenal obstruction and direct surgical attack upon etiology. Ann
Surg. 148:725–730. 1958.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Ha CD, Alvear DT and Leber DC: Duodenal
derotation as an effective treatment of superior mesenteric artery
syndrome: A thirty-three-year experience. Am Surg. 74:644–653.
2008.PubMed/NCBI
|
|
14
|
Morris TC, Devitt PG and Thompson SK:
Laparoscopic duodenojejunostomy for superior mesenteric artery
syndrome-how I do it. J Gastrointest Surg. 13:1870–1873.
2009.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Sinagra E, Raimondo D, Albano D, Guarnotta
V, Blasco M, Testai S, Marasà M, Mastrella V, Alaimo V, Bova V, et
al: Superior mesenteric artery syndrome: Clinical, endoscopic, and
radiological findings. Gastroenterol Res Pract.
2018(1937416)2018.PubMed/NCBI View Article : Google Scholar
|