|
1
|
Bishop M and Rumrill PD: Multiple
sclerosis: Etiology, symptoms, incidence and prevalence, and
implications for community living and employment. Work. 52:725–734.
2015.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Kamm CP, Uitdehaag BM and Polman CH:
Multiple sclerosis: Current knowledge and future outlook. Eur
Neurol. 72:132–141. 2014.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Zakaria M, Sharawy M and Anan I: Economic
burden of multiple sclerosis in Egypt: A societal perspective. Mult
Scler Relat Disord. 37(101563)2020.
|
|
4
|
Ghasemi N, Razavi S and Nikzad E: Multiple
sclerosis: Pathogenesis, symptoms, diagnoses and cell-based
therapy. Cell J. 19:1–10. 2017.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Hauser SL, Bhan AK, Gilles F, Kemp M, Kerr
C and Weiner HL: Immunohistochemical analysis of the cellular
infiltrate in multiple sclerosis lesions. Ann Neurol. 19:578–587.
1986.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Shi L, Yang X, Froelich CJ and Greenberg
AH: Purification and use of granzyme B. Methods Enzymol.
322:125–143. 2000.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Osińska I, Popko K and Demkow U: Perforin:
An important player in immune response. Cent Eur J Immunol.
39:109–115. 2014.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Alexander JS, Zivadinov R, Maghzi AH,
Ganta VC, Harris MK and Minagar A: Multiple sclerosis and cerebral
endothelial dysfunction: Mechanisms. Pathophysiology. 18:3–12.
2011.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Bascones-Martinez A, Mattila R, Gomez-Font
R and Meurman JH: Immunomodulatory drugs: Oral and systemic adverse
effects. Med Oral Patol Oral Cir Bucal. 19:e24–e31. 2014.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Critchfield JM, Racke MK, Zúñiga-Pflücker
JC, Cannella B, Raine CS, Goverman J and Lenardo MJ: T cell
deletion in high antigen dose therapy of autoimmune
encephalomyelitis. Science. 263:1139–1143. 1994.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Ding SW, Li H, Lu R, Li F and Li WX: RNA
silencing: A conserved antiviral immunity of plants and animals.
Virus Res. 102:109–115. 2004.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Paraboschi EM, Soldà G, Gemmati D, Orioli
E, Zeri G, Benedetti MD, Salviati A, Barizzone N, Leone M, Duga S
and Asselta R: Genetic association and altered gene expression of
mir-155 in multiple sclerosis patients. Int J Mol Sci.
12:8695–8712. 2011.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Ma X, Zhou J, Zhong Y, Jiang L, Mu P, Li
Y, Singh N, Nagarkatti M and Nagarkatti P: Expression, regulation
and function of microRNAs in multiple sclerosis. Int J Med Sci.
11:810–818. 2014.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Junker A, Krumbholz M, Eisele S, Mohan H,
Augstein F, Bittner R, Lassmann H, Wekerle H, Hohlfeld R and Meinl
E: MicroRNA profiling of multiple sclerosis lesions identifies
modulators of the regulatory protein CD47. Brain. 132:3342–3352.
2009.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Seddiki N, Brezar V, Ruffin N, Lévy Y and
Swaminathan S: Role of miR-155 in the regulation of lymphocyte
immune function and disease. Immunology. 142:32–38. 2014.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Niwald M, Migdalska-Sęk M,
Brzeziańska-Lasota E and Miller E: Evaluation of selected MicroRNAs
expression in remission phase of multiple sclerosis and their
potential link to cognition, depression, and disability. J Mol
Neurosci. 63:275–282. 2017.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Baulina N, Kulakova O, Kiselev I, Osmak G,
Popova E, Boyko A and Favorova O: Immune-related miRNA expression
patterns in peripheral blood mononuclear cells differ in multiple
sclerosis relapse and remission. J Neuroimmunol. 317:67–76.
2018.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Song J and Lee JE: miR-155 is involved in
Alzheimer's disease by regulating T lymphocyte function. Front
Aging Neurosci. 7(61)2015.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Tsai CY, Allie SR, Zhang W and Usherwood
EJ: MicroRNA miR-155 affects antiviral effector and effector Memory
CD8 T cell differentiation. J Virol. 87:2348–2351. 2013.PubMed/NCBI View Article : Google Scholar
|
|
20
|
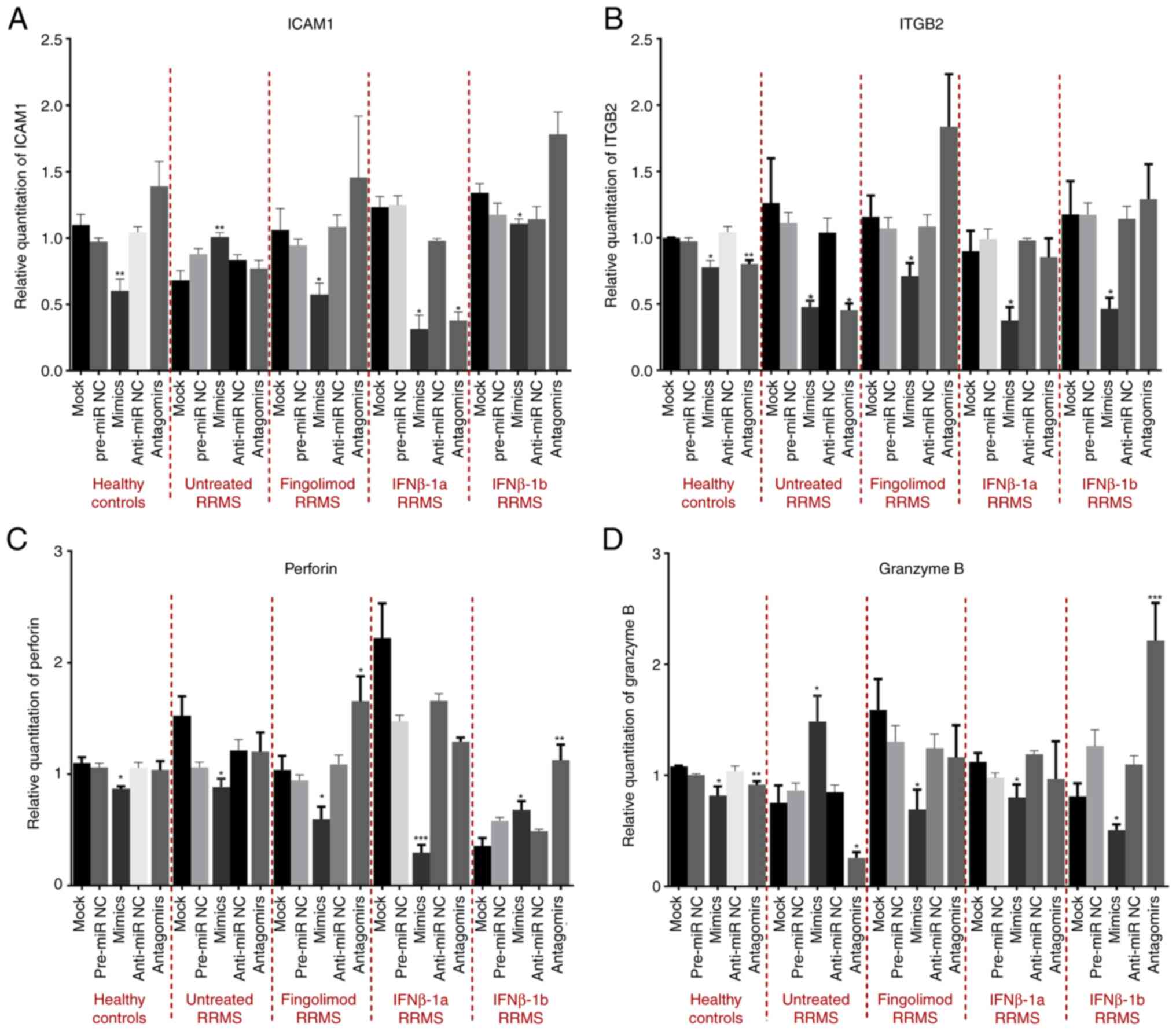
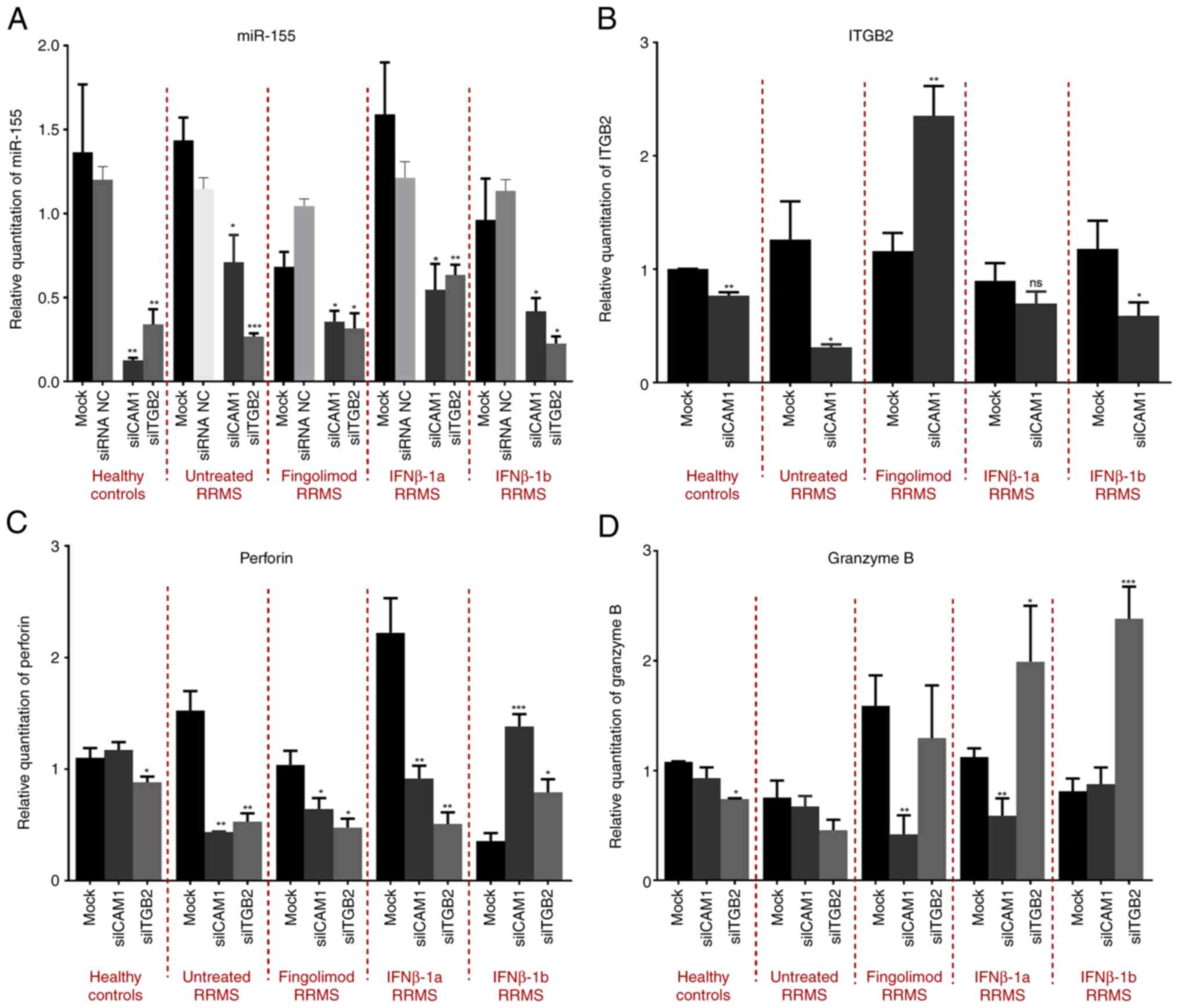
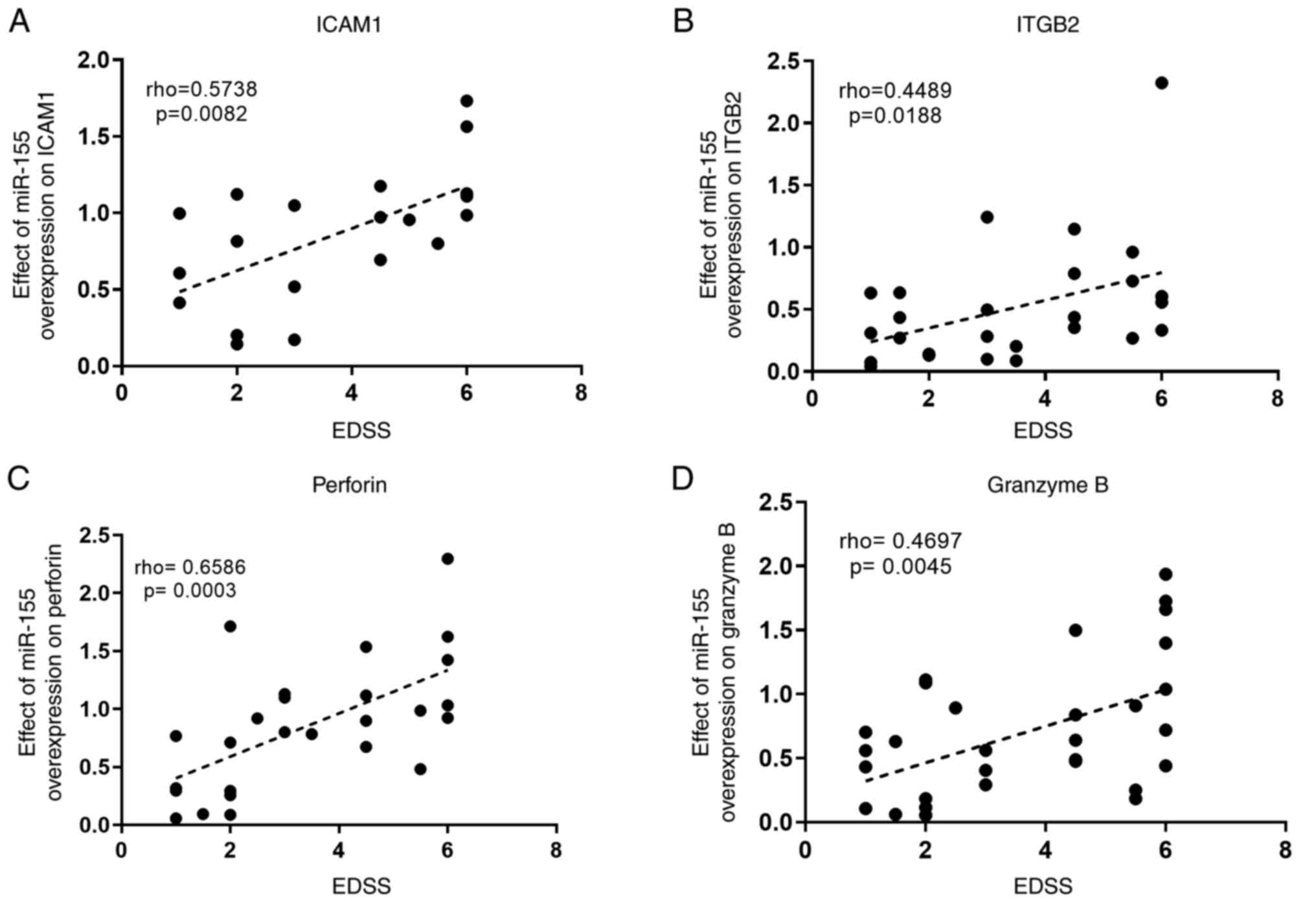
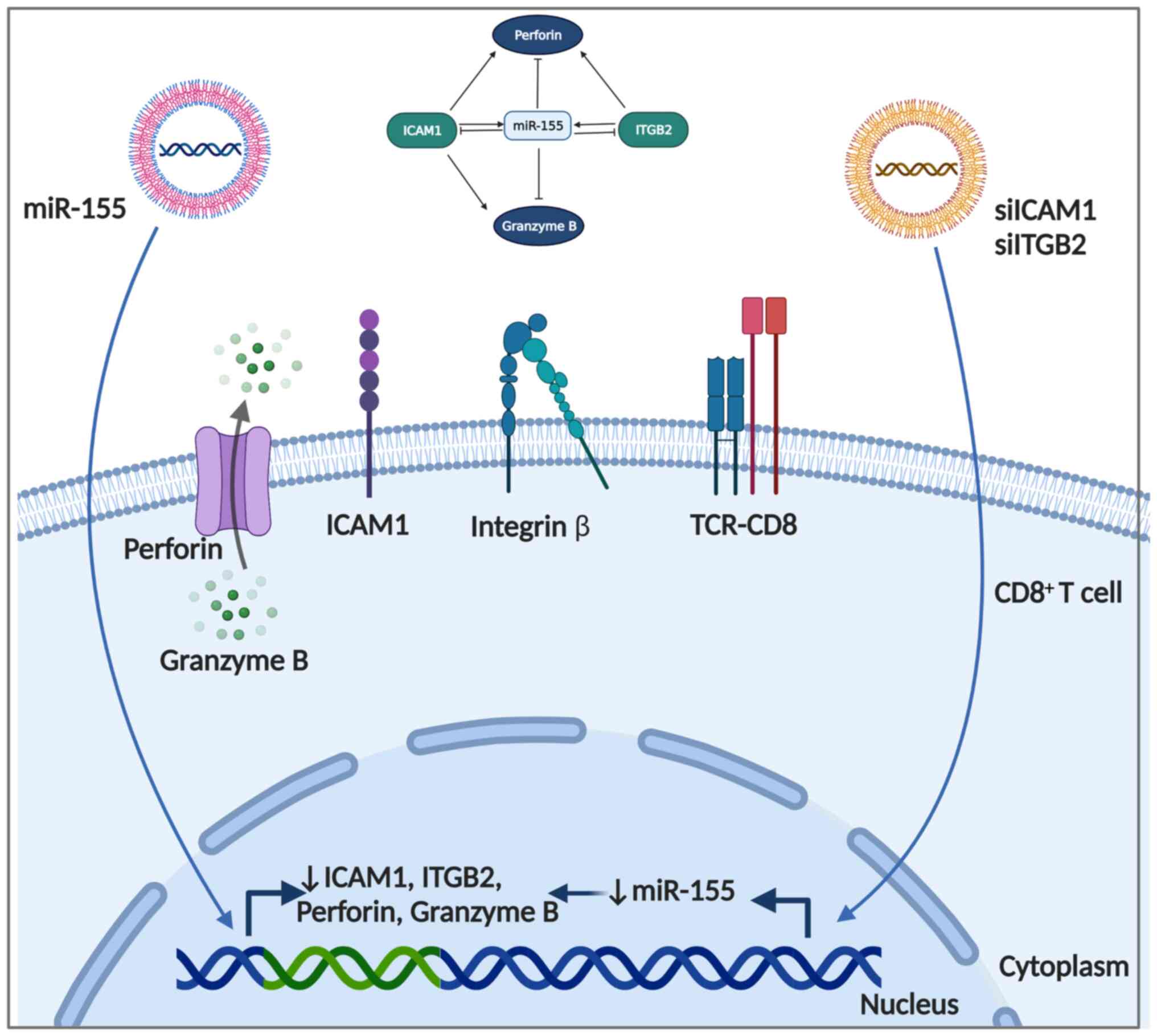
Elkhodiry AA, Zamzam DA and El Tayebi HM:
miR-155 and functional proteins of CD8+ T cells as potential
prognostic biomarkers for relapsing-remitting multiple sclerosis.
Mult Scler Relat Disord. 53(103078)2021.PubMed/NCBI View Article : Google Scholar
|
|
21
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) method. Methods. 25:402–408.
2001.PubMed/NCBI View Article : Google Scholar
|
|
22
|
Yusuf-Makagiansar H, Anderson ME,
Yakovleva TV, Murray JS and Siahaan TJ: Inhibition of LFA-1/ICAM-1
and VLA-4/VCAM-1 as a therapeutic approach to inflammation and
autoimmune diseases. Med Res Rev. 22:146–167. 2002.PubMed/NCBI View Article : Google Scholar
|
|
23
|
Yang X, Wu Y, Zhang B and Ni B: Noncoding
RNAs in multiple sclerosis. Clin Epigenetics.
10(149)2018.PubMed/NCBI View Article : Google Scholar
|
|
24
|
Lukiw WJ, Surjyadipta B, Dua P and
Alexandrov PN: Common micro RNAs (miRNAs) target complement factor
H (CFH) regulation in Alzheimer's disease (AD) and in age-related
macular degeneration (AMD). Int J Biochem Mol Biol. 3:105–116.
2012.PubMed/NCBI
|
|
25
|
El Tayebi HM, Waly AA, Assal RA, Hosny KA,
Esmat G and Abdelaziz AI: Transcriptional activation of the
IGF-II/IGF-1R axis and inhibition of IGFBP-3 by miR-155 in
hepatocellular carcinoma. Oncol Lett. 10:3206–3212. 2015.PubMed/NCBI View Article : Google Scholar
|
|
26
|
Atwa S, Hosny K, Handoussa H and El Tayebi
H: Paradoxically functioning onco-miR-155 and the tumor suppressor
miR-194 consensus on PD-L1 immune checkpoint upregulation via
MALAT-1 and XIST in hepatocellular carcinoma. J Hepatol. 68 (Suppl
1):S610–S611. 2018.
|
|
27
|
Junker A: Pathophysiology of translational
regulation by microRNAs in multiple sclerosis. FEBS Lett.
585:3738–3746. 2011.PubMed/NCBI View Article : Google Scholar
|
|
28
|
Reyes-Long S, Cortes-Altamirano JL,
Clavijio-Cornejo D, Gutiérrez M, Bertolazzi C, Bandala C, Pineda C
and Alfaro-Rodríguez A: Nociceptive related microRNAs and their
role in rheumatoid arthritis. Mol Biol Rep. 47:7265–7272.
2020.PubMed/NCBI View Article : Google Scholar
|
|
29
|
Leng RX, Pan HF, Qin WZ, Chen GM and Ye
DQ: Role of microRNA-155 in autoimmunity. Cytokine Growth Factor
Rev. 22:141–147. 2011.PubMed/NCBI View Article : Google Scholar
|
|
30
|
Azzaoui K, Blommers M, Götte M, Zimmermann
K, Liu H and Fretz H: Discovery of small molecule drugs targeting
the biogenesis of microRNA-155 for the treatment of systemic lupus
erythematosus. Chimia (Aarau). 74:798–802. 2020.PubMed/NCBI View Article : Google Scholar
|
|
31
|
Kamphuis WW, Troletti C, Reijerkerk A,
Romero IA and de Vries HE: The blood-brain barrier in multiple
sclerosis: microRNAs as key regulators. CNS Neurol Disord Drug
Targets. 14:157–167. 2015.PubMed/NCBI View Article : Google Scholar
|
|
32
|
Landgraf P, Rusu M, Sheridan R, Sewer A,
Iovino N, Aravin A, Pfeffer S, Rice A, Kamphorst AO, Landthaler M,
et al: A mammalian microRNA expression atlas based on small RNA
library sequencing. Cell. 129:1401–1414. 2007.PubMed/NCBI View Article : Google Scholar
|
|
33
|
Wu H, Neilson JR, Kumar P, Manocha M,
Shankar P, Sharp PA and Manjunath N: miRNA profiling of naïve,
effector and memory CD8 T cells. PLoS One. 2(e1020)2007.PubMed/NCBI View Article : Google Scholar
|
|
34
|
Neilson JR, Zheng GXY, Burge CB and Sharp
PA: Dynamic regulation of miRNA expression in ordered stages of
cellular development. Genes Dev. 21:578–589. 2007.PubMed/NCBI View Article : Google Scholar
|
|
35
|
Elovaara I, Ukkonen M, Leppäkynnäs M,
Lehtimäki T, Luomala M, Peltola J and Dastidar P: Adhesion
molecules in multiple sclerosis: Relation to subtypes of disease
and methylprednisolone therapy. Arch Neurol. 57:546–551.
2000.PubMed/NCBI View Article : Google Scholar
|
|
36
|
Fujii C, Kondo T, Ochi H, Okada Y, Hashi
Y, Adachi T, Shin-Ya M, Matsumoto S, Takahashi R, Nakagawa M and
Mizuno T: Altered T cell phenotypes associated with clinical
relapse of multiple sclerosis patients receiving fingolimod
therapy. Sci Rep. 6(35314)2016.PubMed/NCBI View Article : Google Scholar
|
|
37
|
Cerutti C, Soblechero-Martin P, Wu D,
Lopez-Ramirez MA, de Vries H, Sharrack B, Male DK and Romero IA:
MicroRNA-155 contributes to shear-resistant leukocyte adhesion to
human brain endothelium in vitro. Fluids Barriers CNS.
13(8)2016.PubMed/NCBI View Article : Google Scholar
|
|
38
|
Lind EF, Elford AR and Ohashi PS:
Micro-RNA 155 is required for optimal CD8+ T cell responses to
acute viral and intracellular bacterial challenges. J Immunol.
190:1210–1216. 2013.PubMed/NCBI View Article : Google Scholar
|
|
39
|
Blüml S, Bonelli M, Niederreiter B,
Puchner A, Mayr G, Hayer S, Koenders MI, van den Berg WB, Smolen J
and Redlich K: Essential role of microRNA-155 in the pathogenesis
of autoimmune arthritis in mice. Arthritis Rheum. 63:1281–1288.
2011.PubMed/NCBI View Article : Google Scholar
|
|
40
|
Lashine YA, Salah S, Aboelenein HR and
Abdelaziz AI: Correcting the expression of miRNA-155 represses
PP2Ac and enhances the release of IL-2 in PBMCs of juvenile SLE
patients. Lupus. 24:240–247. 2015.PubMed/NCBI View Article : Google Scholar
|
|
41
|
Yasuda M, Tanaka Y, Tamura M, Fujii K,
Sugaya M, So T, Takenoyama M and Yasumoto K: Stimulation of beta1
integrin down-regulates ICAM-1 expression and ICAM-1-dependent
adhesion of lung cancer cells through focal adhesion kinase. Cancer
Res. 61:2022–2030. 2001.PubMed/NCBI
|
|
42
|
Scholer A, Hugues S, Boissonnas A, Fetler
L and Amigorena S: Intercellular adhesion molecule-1-dependent
stable interactions between T cells and dendritic cells determine
CD8+ T cell memory. Immunity. 28:258–270. 2008.PubMed/NCBI View Article : Google Scholar
|
|
43
|
Bullard DC, Hu X, Schoeb TR, Collins RG,
Beaudet AL and Barnum SR: Intercellular adhesion molecule-1
expression is required on multiple cell types for the development
of experimental autoimmune encephalomyelitis. J Immunol.
178:851–857. 2007.PubMed/NCBI View Article : Google Scholar
|
|
44
|
Bullard DC, Hu X, Schoeb TR, Axtell RC,
Raman C and Barnum SR: Critical requirement of CD11b (Mac-1) on T
cells and accessory cells for development of experimental
autoimmune encephalomyelitis. J Immunol. 175:6327–6333.
2005.PubMed/NCBI View Article : Google Scholar
|
|
45
|
Faraoni I, Antonetti FR, Cardone J and
Bonmassar E: miR-155 gene: A typical multifunctional microRNA.
Biochim Biophys Acta. 1792:497–505. 2009.PubMed/NCBI View Article : Google Scholar
|