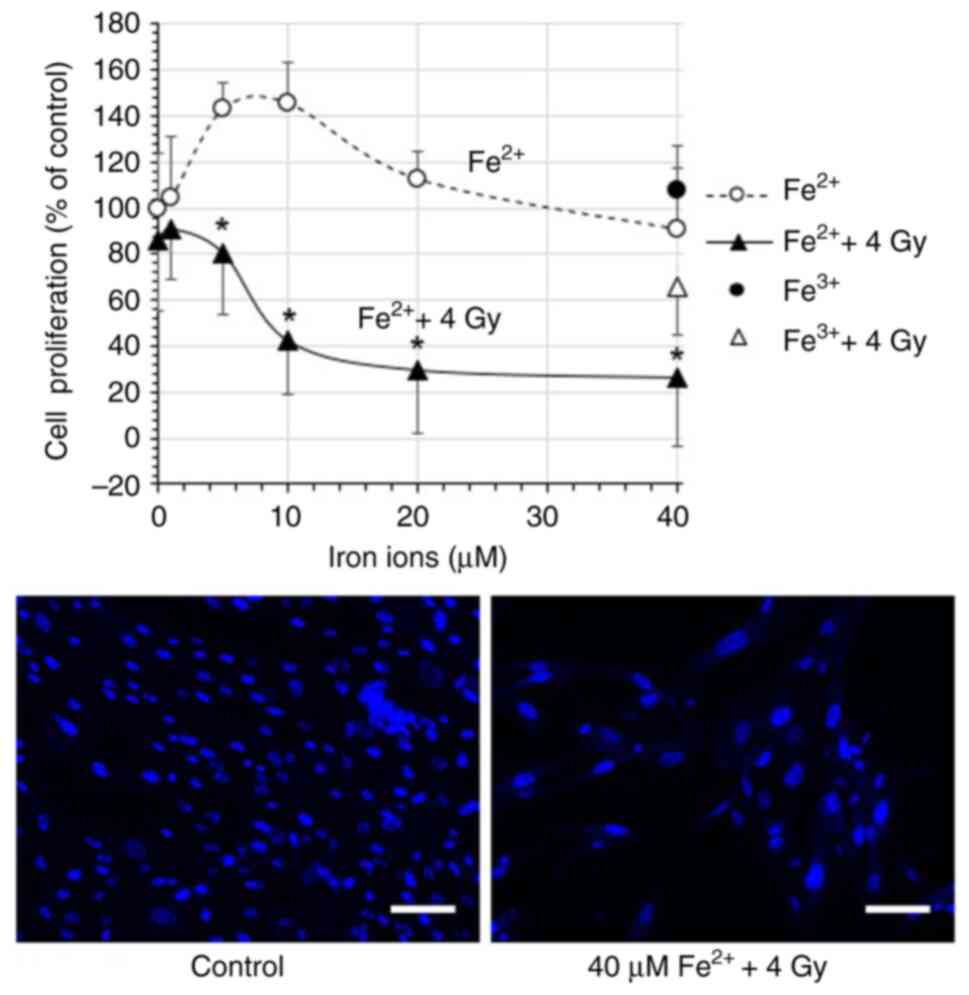
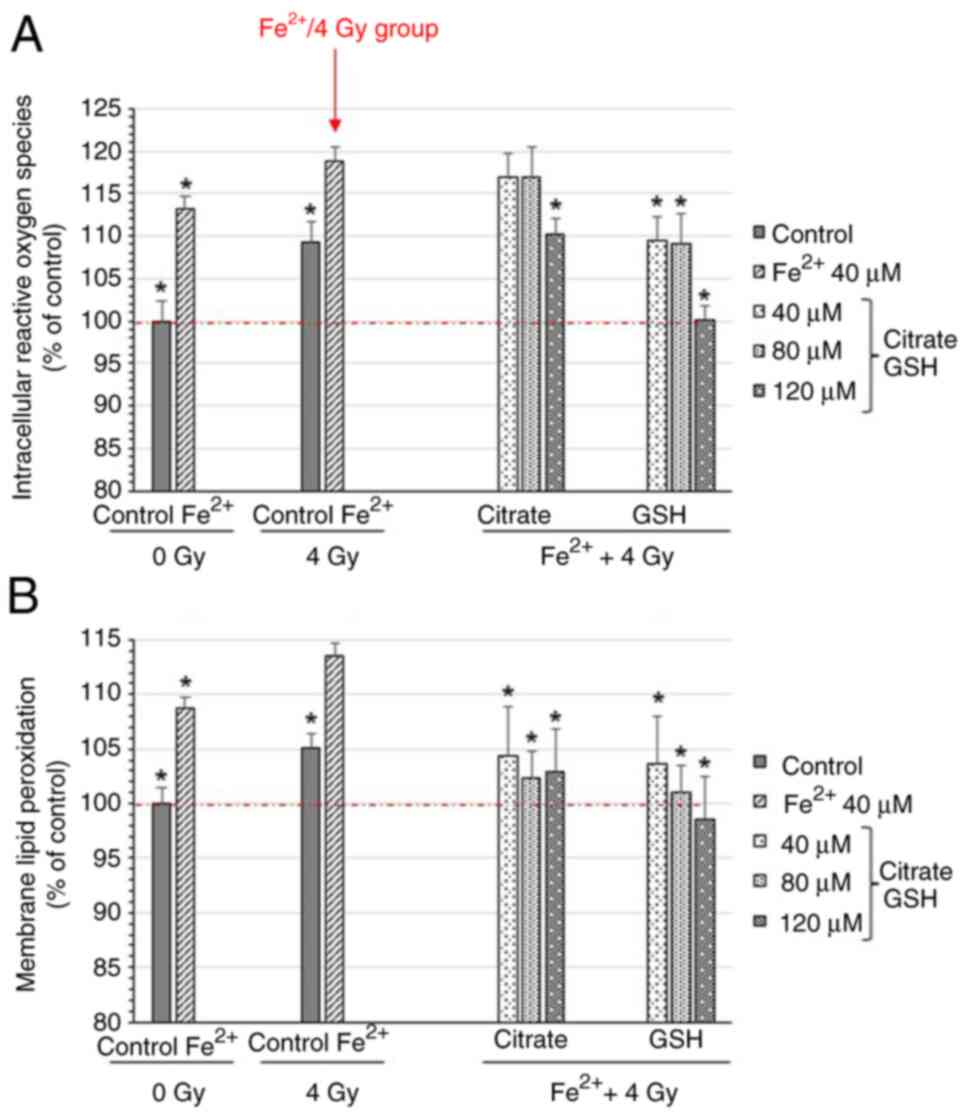
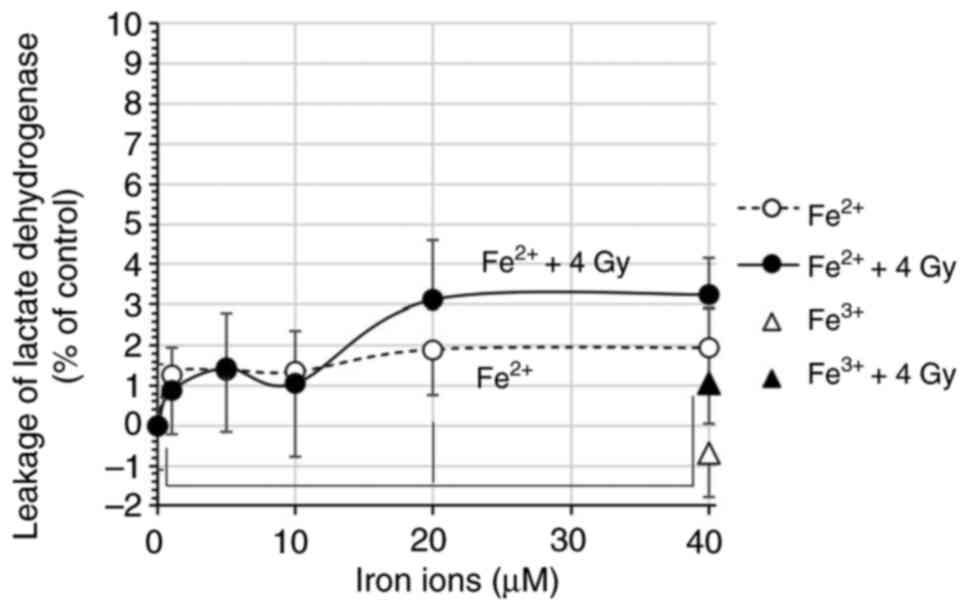
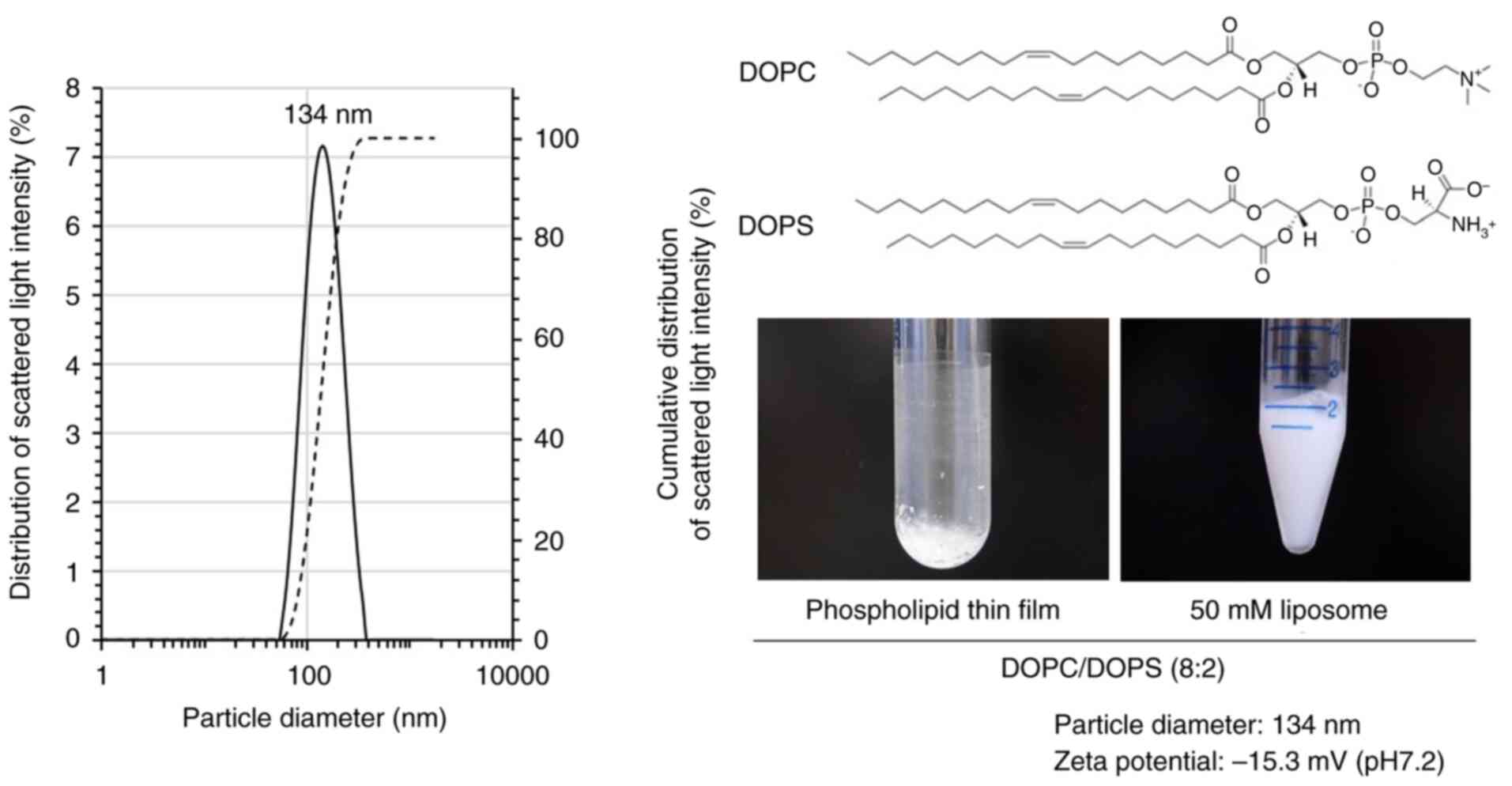
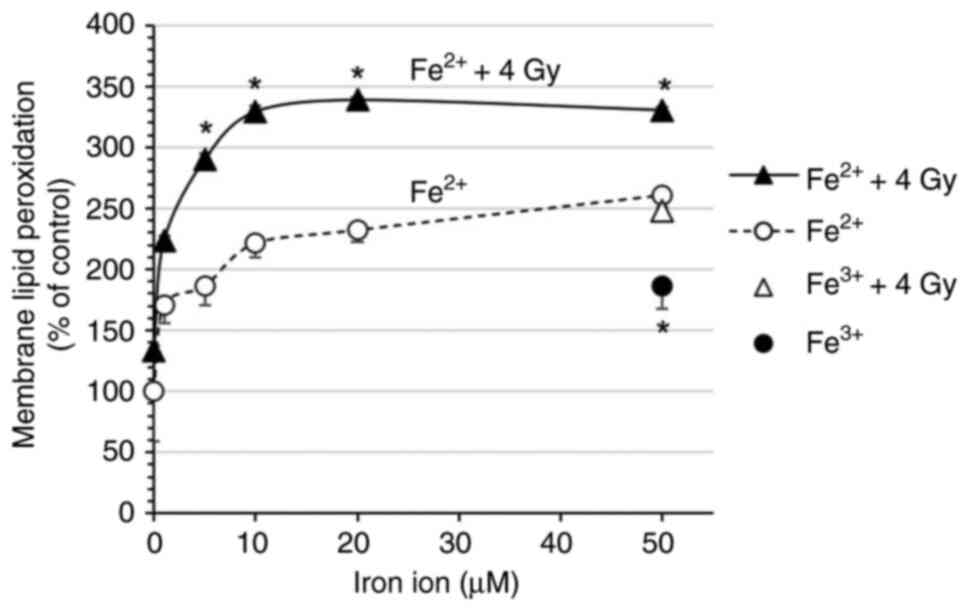
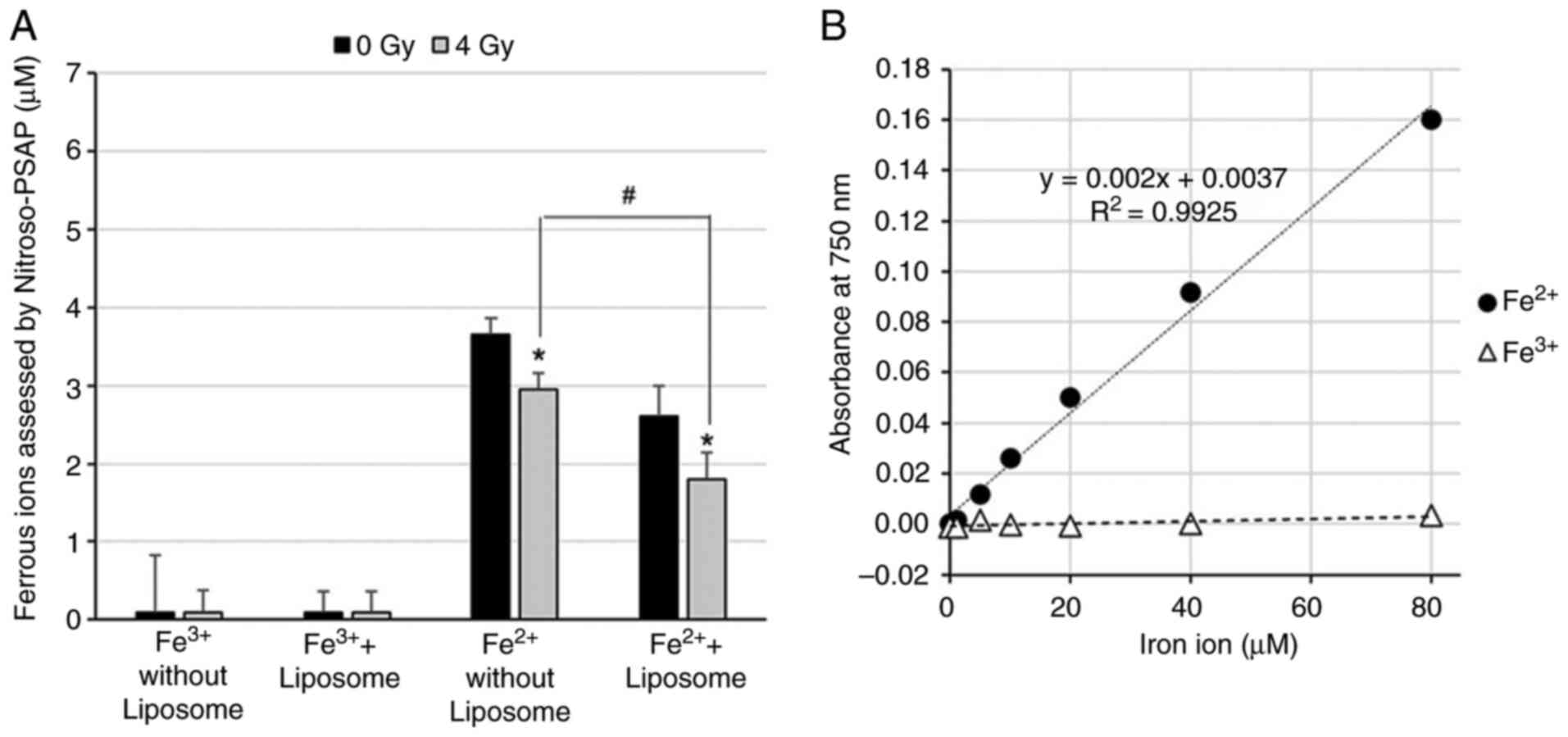
|
1
|
Dixon SJ, Lemberg KM, Lamprecht MR, Skouta
R, Zaitsev EM, Gleason CE, Patel DN, Bauer AJ, Cantley AM, Yang WS,
et al: Ferroptosis: an iron-dependent form of nonapoptotic cell
death. Cell. 149:1060–1072. 2012.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Dixon SJ, Patel DN, Welsch M, Skouta R,
Lee ED, Hayano M, Thomas AG, Gleason CE, Tatonetti NP, Slusher BS
and Stockwell BR: Pharmacological inhibition of cystine-glutamate
exchange induces endoplasmic reticulum stress and ferroptosis.
Elife. 3(e02523)2014.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Shibata Y, Yasui H, Higashikawa K,
Miyamoto N and Kuge Y: Erastin, a ferroptosis-inducing agent,
sensitised cancer cells to X-ray irradiation via glutathione
starvation in vitro and in vivo. PLoS One.
14(e0225931)2019.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Liu J, An W, Zhao Q, Liu Z, Jiang Y, Li H
and Wang D: Hyperbaric oxygen enhances X-ray induced ferroptosis in
oral squamous cell carcinoma cells. Oral Dis. 30:116–127.
2024.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Zhang X, Wu Z, Lan H, Chen S, Wu J, Zhu L
and Xiao Y: Deferoxamine promotes recovery of bone marrow
hematopoietic function in mice exposed to a sublethal dose of X-ray
irradiation. Nan Fang Yi Ke Da Xue Xue Bao. 43:1577–1584.
2023.PubMed/NCBI View Article : Google Scholar : (In Chinese).
|
|
6
|
Zhang B, Liu H, Wang Y, Zhang Y and Cheng
J: Application of singlet oxygen-activatable nanocarriers to boost
X-ray-induced photodynamic therapy and cascaded ferroptosis for
breast cancer treatment. J Mater Chem B. 11:9685–9696.
2023.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Citrin DE, Prasanna PGS, Walker AJ,
Freeman ML, Eke I, Barcellos-Hoff MH, Arankalayil MJ, Cohen EP,
Wilkins RC, Ahmed MM, et al: Radiation-induced fibrosis: mechanisms
and opportunities to mitigate. report of an NCI workshop, September
19, 2016. Radiat Res. 188:1–20. 2017.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Müller K and Meineke V: Radiation-induced
alterations in cytokine production by skin cells. Exp Hematol. 35
(Suppl 1):S96–S104. 2007.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Hirose E, Noguchi M, Ihara T and Yokoya A:
Mitochondrial metabolism in X-Irradiated cells undergoing
irreversible cell-cycle arrest. Int J Mol Sci.
24(1833)2023.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Busato F, Khouzai BE and Mognato M:
Biological mechanisms to reduce radioresistance and increase the
efficacy of radiotherapy: State of the art. Int J Mol Sci.
23(10211)2022.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Shimura T, Totani R, Ogasawara H, Inomata
K, Sasatani M, Kamiya K and Ushiyama A: Effects of oxygen on the
response of mitochondria to X-irradiation and reactive oxygen
species-mediated fibroblast activation. Int J Radiat Biol.
99:769–778. 2023.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Truong K, Bradley S, Baginski B, Wilson
JR, Medlin D, Zheng L, Wilson RK, Rusin M, Takacs E and Dean D: The
effect of well-characterized, very low-dose x-ray radiation on
fibroblasts. PLoS One. 13(e0190330)2018.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Jiang J, Yang L, Xie Q, Liu X, Jiang J,
Zhang J, Zhang S, Zheng H, Li W, Cai X, et al: Synthetic vectors
for activating the driving axis of ferroptosis. Nat Commun.
15(7923)2024.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Bae C, Hernández Millares R, Ryu S, Moon
H, Kim D, Lee G, Jiang Z, Park MH, Kim KH, Koom WS, et al:
Synergistic effect of ferroptosis-inducing nanoparticles and X-Ray
irradiation combination therapy. Small. 20(e2310873)2024.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Kato S: Lactoferrin inhibits the
proliferation of IMR-32 neuroblastoma cells even under X-rays. Med
Int (Lond). 3(33)2023.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Kato S: Effects of platinum-coexisting
dopamine with X-ray irradiation upon human glioblastoma cell
proliferation. Hum Cell. 34:1653–1661. 2021.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Kato S: Under lithium carbonate
administration, nicotine triggers cell dysfunction in human
glioblastoma U-251MG cells, which is distinct from cotinine. Med
Int (Lond). 2(19)2022.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Jay-Gerin JP: Fundamentals of Water
Radiolysis. Encyclopedia. 5(38)2025.
|
|
19
|
Ishiyama M, Tominaga H, Shiga M, Sasamoto
K, Ohkura Y and Ueno K: A combined assay of cell viability and in
vitro cytotoxicity with a highly water-soluble tetrazolium salt,
neutral red and crystal violet. Biol Pharm Bull. 19:1518–1520.
1996.PubMed/NCBI View Article : Google Scholar
|
|
20
|
Eruslanov E and Kusmartsev S:
Identification of ROS using oxidized DCFDA and flow-cytometry.
Methods Mol Biol. 594:57–72. 2010.PubMed/NCBI View Article : Google Scholar
|
|
21
|
Soh N, Ariyoshi T, Fukaminato T, Nakajima
H, Nakano K and Imato T: Swallow-tailed perylene derivative: A new
tool for fluorescent imaging of lipid hydroperoxides. Org Biomol
Chem. 5:3762–3768. 2007.PubMed/NCBI View Article : Google Scholar
|
|
22
|
Kumar P, Nagarajan A and Uchil PD:
Analysis of cell viability by the lactate dehydrogenase assay. Cold
Spring Harb Protoc: Jun 1, 2018 (Epub ahead of print).
|
|
23
|
Kato S and Kuwata K: Pro-/anti-oxidative
properties of dopamine on membrane lipid peroxidation upon X-ray
irradiation. Radiat Phys Chem. 185(109518)2021.
|
|
24
|
El Behery M, Fujimura M, Kimura T and
Tsubaki M: Direct measurements of ferric reductase activity of
human 101F6 and its enhancement upon reconstitution into
phospholipid bilayer nanodisc. Biochem Biophys Rep.
21(100730)2020.PubMed/NCBI View Article : Google Scholar
|
|
25
|
Muckenthaler MU, Rivella S, Hentze MW and
Galy B: A red carpet for iron metabolism. Cell. 168:344–361.
2017.PubMed/NCBI View Article : Google Scholar
|
|
26
|
Cañeque T, Baron L, Müller S, Carmona A,
Colombeau L, Versini A, Solier S, Gaillet C, Sindikubwabo F,
Sampaio JL, et al: Activation of lysosomal iron triggers
ferroptosis in cancer. Nature. 642:492–500. 2025.PubMed/NCBI View Article : Google Scholar
|
|
27
|
Stockwell BR, Friedmann Angeli JP, Bayir
H, Bush AI, Conrad M, Dixon SJ, Fulda S, Gascón S, Hatzios SK,
Kagan VE, et al: Ferroptosis: A regulated cell death nexus linking
metabolism, redox biology, and disease. Cell. 171:273–285.
2017.PubMed/NCBI View Article : Google Scholar
|
|
28
|
Yang WS and Stockwell BR: Ferroptosis:
Death by lipid peroxidation. Trends Cell Biol. 26:165–176.
2016.PubMed/NCBI View Article : Google Scholar
|
|
29
|
Zilka O, Shah R, Li B, Friedmann Angeli
JP, Griesser M, Conrad M and Pratt DA: On the mechanism of
cytoprotection by ferrostatin-1 and Liproxstatin-1 and the role of
lipid peroxidation in ferroptotic cell death. ACS Cent Sci.
3:232–243. 2017.PubMed/NCBI View Article : Google Scholar
|
|
30
|
Montero D, Tachibana C, Rahr Winther J and
Appenzeller-Herzog C: Intracellular glutathione pools are
heterogeneously concentrated. Redox Biol. 1:508–513.
2013.PubMed/NCBI View Article : Google Scholar
|
|
31
|
Mao C, Lei G, Horbath A and Gan B:
Assessment of lipid peroxidation in irradiated cells. Methods Cell
Biol. 172:37–50. 2022.PubMed/NCBI View Article : Google Scholar
|
|
32
|
Kim BC, Shon BS, Ryoo YW, Kim SP and Lee
KS: Melatonin reduces X-ray irradiation-induced oxidative damages
in cultured human skin fibroblasts. J Dermatol Sci. 26:194–200.
2001.PubMed/NCBI View Article : Google Scholar
|
|
33
|
Shimura T, Nakashiro C, Fujiwara K, Shiga
R, Sasatani M, Kamiya K and Ushiyama A: Radiation affects
glutathione redox reaction by reduced glutathione peroxidase
activity in human fibroblasts. J Radiat Res. 63:183–191.
2022.PubMed/NCBI View Article : Google Scholar
|
|
34
|
Morlière P, Moysan A, Santus R, Hüppe G,
Mazière JC and Dubertret L: UVA-induced lipid peroxidation in
cultured human fibroblasts. Biochim Biophys Acta. 1084:261–268.
1991.PubMed/NCBI View Article : Google Scholar
|
|
35
|
Enami S, Sakamoto Y and Colussi AJ: Fenton
chemistry at aqueous interfaces. Proc Natl Acad Sci USA.
111:623–628. 2014.PubMed/NCBI View Article : Google Scholar
|
|
36
|
Kim JW, Lee JY, Oh M and Lee EW: An
integrated view of lipid metabolism in ferroptosis revisited via
lipidomic analysis. Exp Mol Med. 55:1620–1631. 2023.PubMed/NCBI View Article : Google Scholar
|
|
37
|
Klasson TD, LaGory EL, Zhao H, Huynh SK,
Papandreou I, Moon EJ and Giaccia AJ: ACSL3 regulates lipid droplet
biogenesis and ferroptosis sensitivity in clear cell renal cell
carcinoma. Cancer Metab. 10(14)2022.PubMed/NCBI View Article : Google Scholar
|
|
38
|
Wang M, Mao C, Ouyang L, Liu Y, Lai W, Liu
N, Shi Y, Chen L, Xiao D, Yu F, et al: Long noncoding RNA LINC00336
inhibits ferroptosis in lung cancer by functioning as a competing
endogenous RNA. Cell Death Differ. 26:2329–2343. 2019.PubMed/NCBI View Article : Google Scholar
|
|
39
|
Guo N: Identification of ACSL4 as a
biomarker and contributor of ferroptosis in clear cell renal cell
carcinoma. Transl Cancer Res. 11:2688–2699. 2022.PubMed/NCBI View Article : Google Scholar
|