Introduction
Regulated cell death pathways play critical roles in
maintaining physiological homeostasis and contributing to disease
progression. Among these pathways, ferroptosis has attracted
considerable attention due to its distinctive mechanisms and
pathological relevance. In addition to canonical ferroptotic cell
death, iron-dependent lipid peroxidation can also cause sublethal
oxidative membrane injury that affects cellular function without
necessarily inducing ferroptosis. First identified by Dixon et
al (1) in 2012, ferroptosis is a
form of regulated cell death characterized by iron-dependent lipid
peroxidation, primarily driven by reactive oxygen species via the
Fenton reaction. This process is initiated by ferrous ions
(Fe2+), a key factor in ferroptosis. Ferroptosis is a
promising strategy for cancer therapy, and efforts have been made
to explore mechanisms that activate key regulatory pathways to
induce ferroptosis. Elastin, an elastic protein in the dermis, has
been shown to induce ferroptosis by inhibiting cystine
transporters, reducing glutathione (GSH) synthesis, and promoting
the generation of reactive oxygen species (2). As previously demonstrated., under X-ray
irradiation, the administration of elastin decreases GSH levels and
glutathione peroxidase 4 (GPX4) expression, thereby enhancing cell
death in HeLa and adenocarcinoma cell lines and a tumor xenograft
model (3). Previous studies have
indicated that oxygen levels and iron modulation, including
nanoform iron or iron chelators, influence ferroptosis in cancer
cells under X-ray irradiation. Hyperbaric oxygen could sensitize
radioresistant oral squamous cell carcinoma cells to X-rays by
promoting ferroptosis and reducing tumor growth in xenograft mice
(4). On the other hand, deferoxamine
weakly binds free iron and inhibits ferroptosis in bone marrow
nucleated cells of X-ray-irradiated mice, thereby facilitating
hematopoiesis in bone marrow (5). A
nanocarrier composed of a hyperbranched copolymer with
1O2-sensitive linkers and a RAS-selective
lethal agent triggers 1O2-mediated GSH
depletion and GPX4 inactivation, leading to ferroptotic cell death
in breast cancer-bearing mice (6).
Fibroblasts are cells found in connective tissue and
become activated by inflammatory cytokines when tissue damage
occurs due to radiation (7). As
fibroblasts play a central role in radiation-induced fibrosis,
wound healing and long-term normal tissue responses, understanding
oxidative membrane injury in these cells is particularly relevant
in radiobiology. Activated fibroblasts produce components such as
collagen and fibronectin, which serve as scaffolds for cells in the
damaged tissue, thereby promoting tissue regeneration (8). As previously demonstrated, when human
fibroblasts were irradiated with 20 Gy X-rays,
mitochondria-dependent energy metabolism increased, and the cells
underwent cell-cycle arrest (9).
Therapeutic levels of radiation have cytotoxic effects on most
cancer cells, inhibiting proliferation and inducing apoptosis
(10). Irradiation with X-rays
causes DNA damage, triggers mitochondrial signaling and
AMP-activated protein kinase activity, suppresses mitochondrial
metabolism and increases reactive oxygen species production in lung
fibroblasts cultured under 5 and 20% O2 (11). Moreover, fibroblasts treated with a
low dose of 550 µGy X-rays have been shown to exhibit increased
cell proliferation and protein production (12).
Liposomes containing phosphatidylcholine
hydroperoxide induce ferroptosis by targeting divalent metal
transporter 1, which promotes lysosomal Fe2+ efflux in
breast cancer cells and xenografts (13). Prussian blue
(Fe3+-CN--Fe2+) and hyaluronic
acid-based nanoparticles have been shown to induce ferroptosis,
enhancing therapeutic efficacy under 6 Gy X-rays in human lung
carcinoma and melanoma cells (14).
Liposomes have been used to induce ferroptosis under radiation
exposure; however, their interactions with Fe2+ under
X-rays remain poorly understood, and fundamental insights are
lacking. In this context, simplified liposome systems provide a
reductionist approach to isolate Fe2+-driven lipid
peroxidation at membrane surfaces, rather than to recapitulate full
cellular ferroptosis pathways.
The author has recently investigated cell growth
inhibition in glioblastoma and neuroblastoma cells using lithium
carbonate or platinum nano colloids under X-rays (15-17).
While X-rays generate free radicals from water radiolysis (18), the role of Fe2+ in
promoting the oxidation of lipid membranes and ferroptosis-related
oxidative membrane damage is yet to be clarified. Alternatively,
liposomes serve as a lipid membrane model reflecting membrane lipid
peroxidation in ferroptosis research. The present study employed
liposomes composed of the unsaturated lipids,
L-α-dioleoylphosphatidylcholine (DOPC) and
L-α-dioleoylphosphatidylserine (DOPS), at an 8:2 molar ratio as a
lipid membrane model, without cholesterol, proteins, or antioxidant
enzymes. The present study focused on early oxidative membrane
damage and lipid peroxidation as mechanistic events associated with
ferroptosis, rather than on establishing definitive ferroptotic
cell death. The present study investigated the impact of
Fe2+ on cell membrane damage in human fibroblast
OUMS-36T-1 cells treated with X-rays, complemented by model
experiments on membrane lipid peroxidation and Fe2+
oxidation using DOPC/DOPS (8:2 mol/mol) liposomes.
Materials and methods
Cells and cell culture
The OUMS-36T-1 cell line, an hTERT
gene-transfected normal human embryo fibroblast, was obtained from
the JCRB cell bank (cat. no. JCRB1006.1). The OUMS-36T-1 cells were
cultured in Dulbecco's modified Eagle's (DMEM) medium with
L-glutamine (FUJIFILM Wako Pure Chemical Corp.) supplemented with
10% fetal bovine serum (S-FBS-NL-015; Serana Europe GmbH) and
penicillin-streptomycin-amphotericin B suspension (FUJIFILM Wako
Pure Chemical Corp.) at 37˚C with 5% CO2.
Cell proliferation assay using
WST-8
Cell proliferation was evaluated using the WST-8
assay, which utilises a water-soluble tetrazolium salt (19). The OUMS-36T-1 cells were seeded at
2,000 cells/well in a 96-well culture plate (Sumitomo Bakelite Co.,
Ltd.) with n=5 wells and incubated for 24 h at 37˚C with 5%
CO2. Ferrous chloride tetrahydrate (Fe2+;
FUJIFILM Wako Pure Chemical Corp.) or ferric chloride hexahydrate
(Fe3+; FUJIFILM Wako Pure Chemical Corp.) were
administered at 0-40 µM. Fresh Fe2+ solutions were
prepared immediately prior to each experiment to minimize
spontaneous oxidation to Fe3+ under culture conditions.
The cells were exposed to 4 Gy X-rays (CAX-150-20; Chubu Medical
Co., Ltd.; 150 kV-20 mA, 1 mm Al + 0.1 mm Cu filters, 0.60 Gy/min)
and incubated for 3 days at 37˚C with 5% CO2. A single
dose of 4 Gy was selected as a representative moderate-to-high dose
commonly used in in vitro radiobiological studies to
reliably induce oxidative stress (15), while maintaining sufficient cell
viability for early mechanistic analyses. The medium was replaced
with 5% of
2-(2-methoxy-4-nitrophenyl)-3-(4-nitrophenyl)-5-(2,4-disulfophenyl)-2H-tetrazolium
(WST-8; Dojindo Laboratories) diluted with DMEM medium and
incubated for 1.5 h at 37˚C with 5% CO2. The absorbance
at 450 nm, corresponding to the formation of yellowish-orange
formazan resulting from the reduction of WST-8 by intracellular
mitochondrial dehydrogenase, was measured using a
multi-spectrophotometer (Viento; Dainippon Sumitomo Pharma Co.,
Ltd.). The cells were then stained with Hoechst 33342 (Dojindo
Laboratories) at room temperature for 15 min and observed using a
fluorescence microscope (BZ-X710; KEYENCE Corporation) at x200
magnification with Ex/Em: 350/461 nm.
Analysis of intracellular reactive
oxygen species using dichlorodihydrofluorescein diacetate
(DCFH-DA)
Ferrous ion (Fe2+) concentrations (0-40
µM) were selected based on preliminary concentration-response
experiments (data not shown) assessing cell proliferation and
oxidative stress. A concentration of 40 µM was used as a threshold
condition that induces intracellular oxidative stress and lipid
peroxidation without causing overt membrane rupture in the absence
of X-ray irradiation. Intracellular reactive oxygen species levels
were evaluated using DCFH-DA (20).
The OUMS-36T-1 cells were seeded at 3,500 cells/well in a 96-well
culture plate with n=5 wells and incubated as described above. The
DCFH-DA solution (ROS Assay Kit-Highly Sensitive DCFH-DA; Dojindo
Laboratories) was added to each well and incubated for 30 min at
37˚C with 5% CO2. After aspirating the medium,
Fe2+ was administered at 40 µM, either alone or combined
with trisodium citrate dihydrate (FUJIFILM Wako Pure Chemical
Corp.) or reduced glutathione (GSH; FUJIFILM Wako Pure Chemical
Corp.) at 40-120 µM. Following incubation for 0.2 h at 37˚C with 5%
CO2, the cells were exposed to 4 Gy X-rays. The early
time point (0.2 h) was selected to capture immediate oxidative
responses at the membrane level, prior to the onset of secondary
transcriptional or cell-death-related processes. To prevent
light-induced auto-oxidation of the probe, all experiments using
DCFH-DA were performed on black plates with room lights turned off
and under dark conditions. The fluorescence intensity was measured
at 0.2 h following X-ray irradiation using a multimode microplate
reader (TriStar LB941; Berthold Technologies GmbH & Co. KG) at
Ex/Em: 485/535 nm. Fluorescence intensity indicates intracellular
reactive oxygen species levels, since DCFH-DA is enzymatically
converted to DCFH, which is rapidly oxidized by reactive oxygen
species into the fluorescent products.
Membrane lipid peroxidation detection
and lactate dehydrogenase leakage
The disruption of the cell membrane was assessed by
membrane lipid peroxidation and lactate dehydrogenase leakage. The
OUMS-36T-1 cells were seeded at 3,500 cells/well and incubated as
mentioned in 2.2. Fe2+ was administered at 40 µM, either
alone or combined with trisodium citrate dihydrate or reduced
glutathione at 40-120 µM.
N-(4-Diphenylphosphinophenyl)-N'-(3,6,9,12-tetraoxatridecyl)
perylene-3,4,9,10-tetracarboxydiimide (Liperfluo, Dojindo
Laboratories) was added to each well at 7.5 µmol/l for the
detection of lipid hydroperoxides (21). Following 0.2 h of incubation at 37˚C
with 5% CO2, the cells were exposed to 4 Gy of X-rays.
After 0.2 h, fluorescence intensity, which is proportional to lipid
peroxide in membrane lipids, was measured at Ex/Em=485/535 nm with
the multimode microplate reader TriStar LB941. This early
measurement was intended to assess primary membrane lipid
peroxidation induced by Fe2+ and X-rays.
On the other hand, following irradiation with 4 Gy
X-rays, the cells were incubated for 21 h at 37˚C with 5%
CO2. The cell culture medium was then replaced with DMEM
containing the cytotoxicity LDH assay kit (Dojindo Laboratories),
and the mixture was incubated for 0.5 h at room temperature in the
dark. The absorbance at 490 nm, which is proportional to lactate
dehydrogenase leakage (22), was
measured with the multi-spectrophotometer Viento (Dainippon
Sumitomo Pharma, Co. Ltd.).
Preparation of liposomes
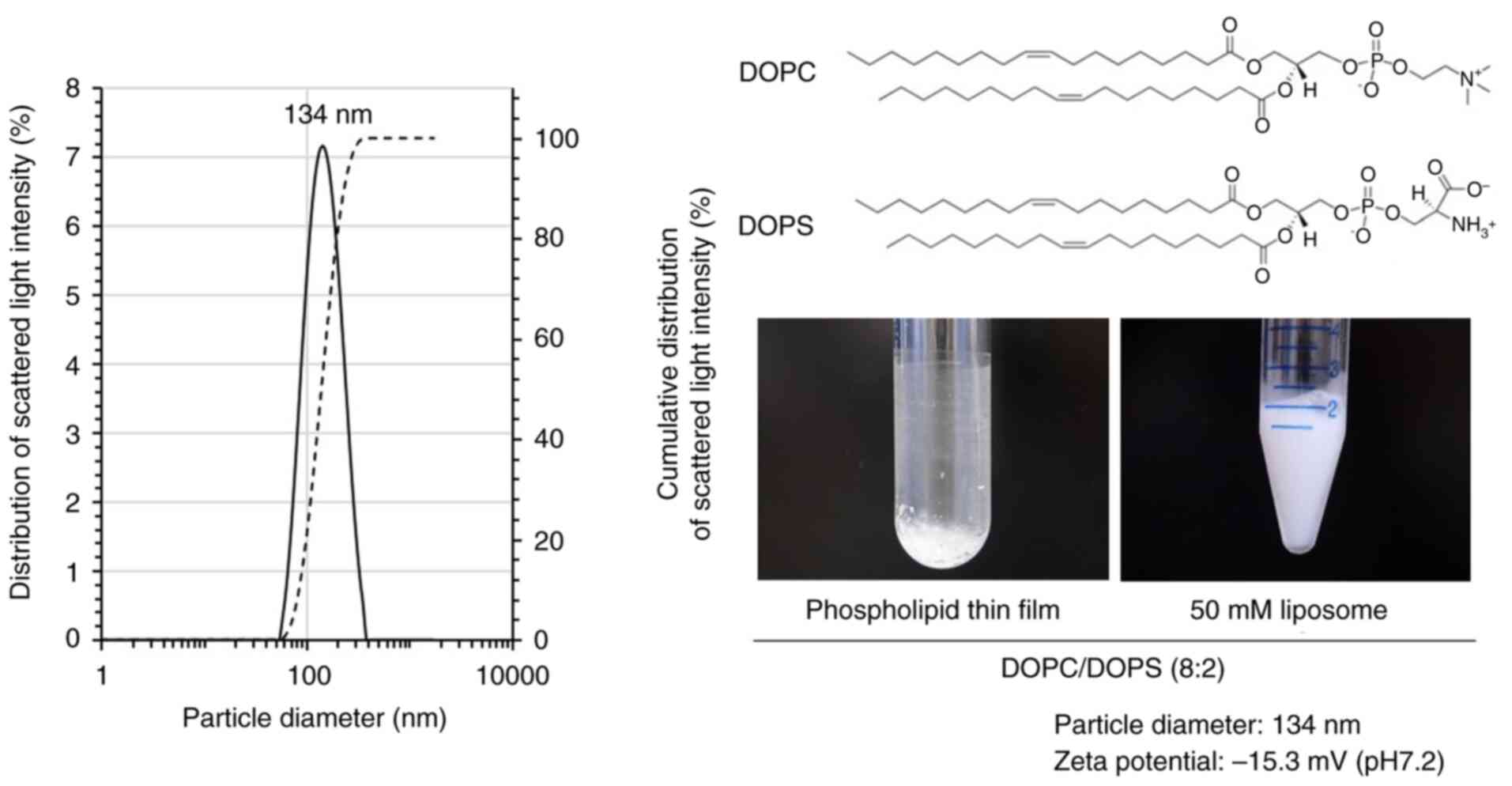
Liposomes composed of DOPC and DOPS at an 8:2 molar
ratio were prepared by mixing
1,2-dioleoyl-sn-glycero-3-phosphocholine (DOPC; NOF Corp.) and
1,2-dioleoyl-sn-glycero-3-phospho-L-serine (DOPS; NOF Corp.). A
phospholipid thin film was formed by evaporating the chloroform
solution of DOPC/DOPS (8:2) under reduced pressure using a custom
apparatus developed by Dr Yoshimura [image of the device is shown
in a previous study by the author (23)]. The resulting film was rehydrated in
phosphate-buffered saline at pH 7.2 (PBS (-), FUJIFILM Wako Pure
Chemical Corp.). The average particle diameter and zeta potential
were 134 nm and -15.3 mV, respectively, as measured using a zeta
potential and particle size analyzer (ELSZ-2; Otsuka Electronics
Co. Ltd.).
Analysis of membrane lipid
peroxidation using Liperfluo
The DOPC/DOPS (8:2) liposomes were added at 100 µM
in a 96-well culture plate with n=5 wells. Fe2+ or
Fe3+ was added at 0-50 µM, and Liperfluo was added to
each well at 7.5 µmol/l. The plate was left to stand for 0.5 h, and
then exposed to 4 Gy of X-rays. After 0.2 h, the fluorescence
intensity was measured as described above.
Oxidation of ferrous ion using the
Nitroso-PSAP method
The DOPC/DOPS (8:2) liposomes were added at 100 µM
in a 1/2 area 96-well plate (UV-STAR; Greiner Bio-One International
GmbH) with n=5 wells. Assays were performed under conditions
containing or lacking the DOPC/DOPS (8:2) liposomes.
Fe2+ or Fe3+ was added at 50 µM, and
nitroso-N, N-dimethyl-p-phenylenediamine (Nitroso-PSAP; Dojindo
Laboratories) was added to each well at 0.004 w/v% for the
detection of ferrous ions (Fe2+) (24). The plate was left to stand for 0.2 h,
and then exposed to 4 Gy X-rays. After 0.2 h, the absorbance at 750
nm, corresponding to the residual ferrous ions (Fe2+),
was measured using the multi-spectrophotometer Viento (Dainippon
Sumitomo Pharma, Co. Ltd.).
Statistical analysis
Data are presented as the mean ± standard deviation
(SD), n=5 wells. Statistical analyses were performed using one-way
analysis of variance (ANOVA) followed by Tukey's honestly
significant difference (HSD) test for comparisons with the
corresponding control group. For two-group comparisons, the
Student's t-test was used. A value of P<0.05 was considered to
indicate a statistically significant difference.
Results
The present study investigated the effects of X-rays
on iron-dependent oxidative membrane damage relevant to ferroptosis
by assessing intracellular levels of reactive oxygen species,
membrane lipid peroxidation and lactate dehydrogenase leakage from
human fibroblasts treated with ferrous ion (Fe2+) and
X-rays.
Effects of Fe2+ and
Fe3+on cell proliferation
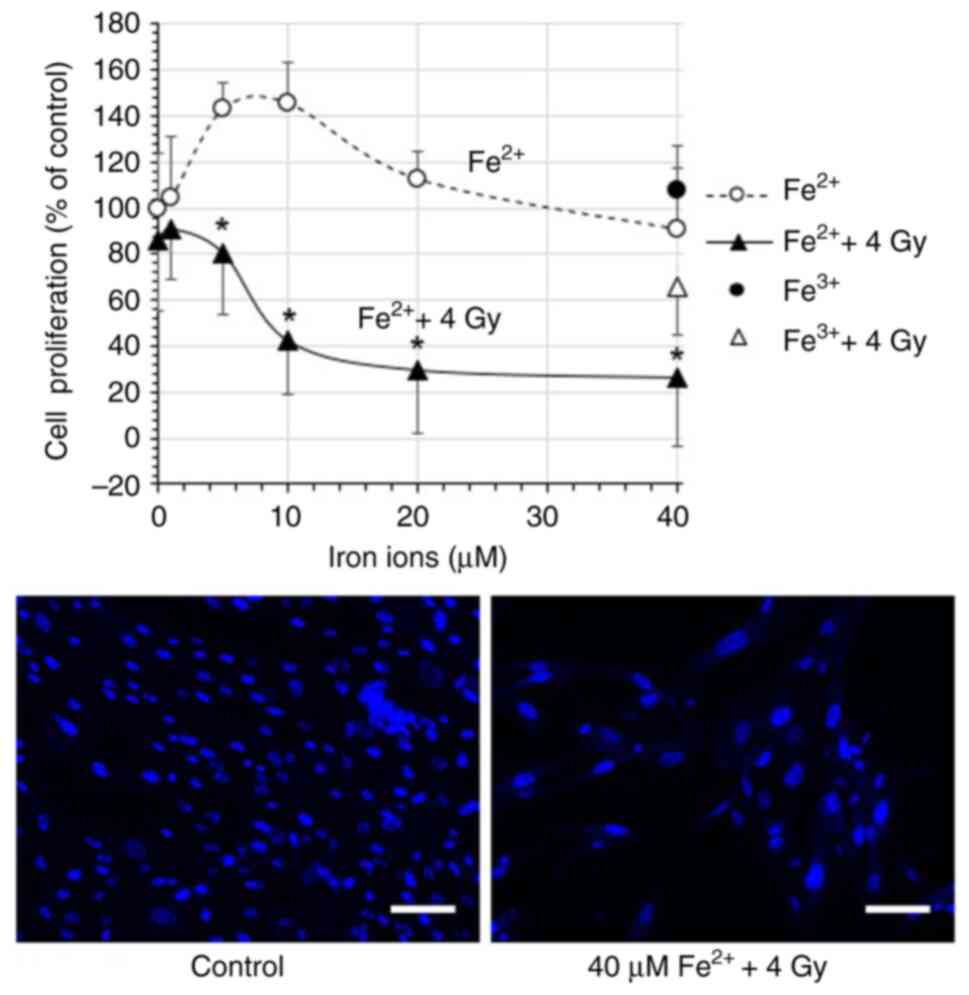
Cell proliferation increased to 145.8% as the
concentration of Fe2+ rose from 0 to 10 µM (Fig. 1), but decreased at higher
concentrations, reaching 90.9% at 40 µM. Exposure to 4 Gy X-rays
alone reduced cell proliferation to 85.6%. Combined treatment with
Fe2+ and X-rays resulted in a concentration-dependent
decline in cell proliferation, reaching a minimum of 26.5% at 40
µM. These trends were confirmed through fluorescence microscopy
(Fig. 1). By contrast, ferric ion
(Fe3+) had a minimal effect on cell viability. Cell
proliferation remained at 107.5% at 40 µM Fe3+ and
decreased to 65.6% following X-ray irradiation, which was
comparable to the untreated control (Fig. 1). This indicates that Fe3+
alone did not impair cell proliferation.
Effects of Fe2+ on the
generation of intracellular reactive oxygen species, membrane lipid
peroxidation and lactate dehydrogenase leakage
The present study then examined the effects of 40 µM
Fe2+ on the generation of intracellular reactive oxygen
species, membrane lipid peroxidation and lactate dehydrogenase
leakage. Fe2+ was added extracellularly; however, based
on the observed increases in intracellular reactive oxygen species
and lipid peroxidation, it was hypothesized that Fe2+
may have contributed, directly or indirectly, to intracellular
oxidative processes.
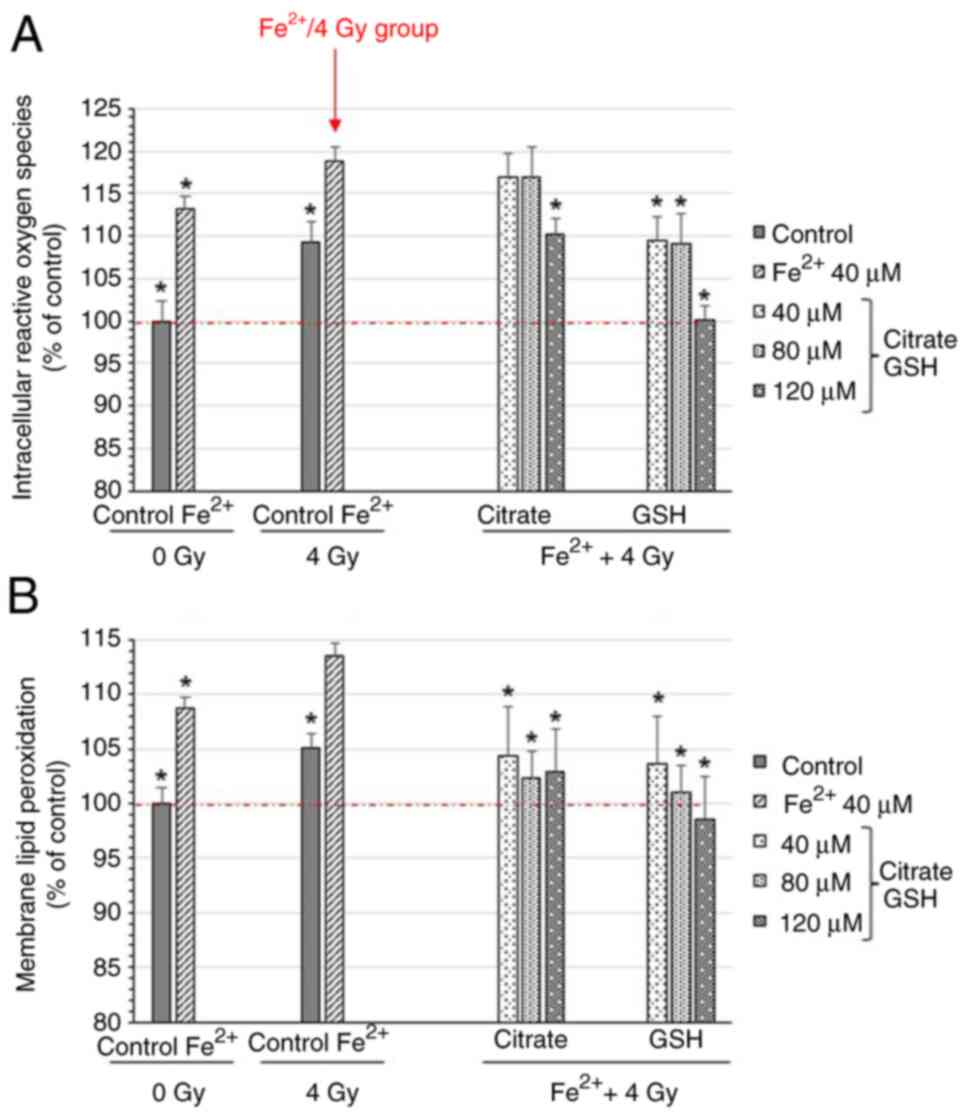
The levels of intracellular reactive oxygen species
increased to 113.1% with Fe2+ alone and to 109.0%
following X-ray irradiation (Fig.
2). The combination of Fe2+ and X-rays further
elevated reactive oxygen species to 118.9%, suggesting a
synergistic effect (Fig. 2). When
trisodium citrate, a chelator of Fe2+, was added at
molar ratios of 1:1 to 1:3 (Fe2+: citrate),
intracellular reactive oxygen species decreased from 116.9 to
110.3%. Similarly, treatment with reduced GSH lowered intracellular
reactive oxygen species from 109.4% to near baseline (Fig. 2).
A similar trend was observed in membrane lipid
peroxidation. Treatment with 40 µM Fe2+ increased lipid
peroxidation to 108.7%, and X-ray irradiation alone elevated it to
105.1% (Fig. 2). Combined treatment
further increased it to 113.5%. Trisodium citrate and reduced GSH
were more effective in suppressing lipid peroxidation than
intracellular reactive oxygen species, reducing membrane lipid
peroxidation to 102.9 and 98.5%, respectively (Fig. 2). These results indicate that
Fe2+-induced oxidative stress is especially significant
near membrane surfaces or within lipid-rich microenvironments.
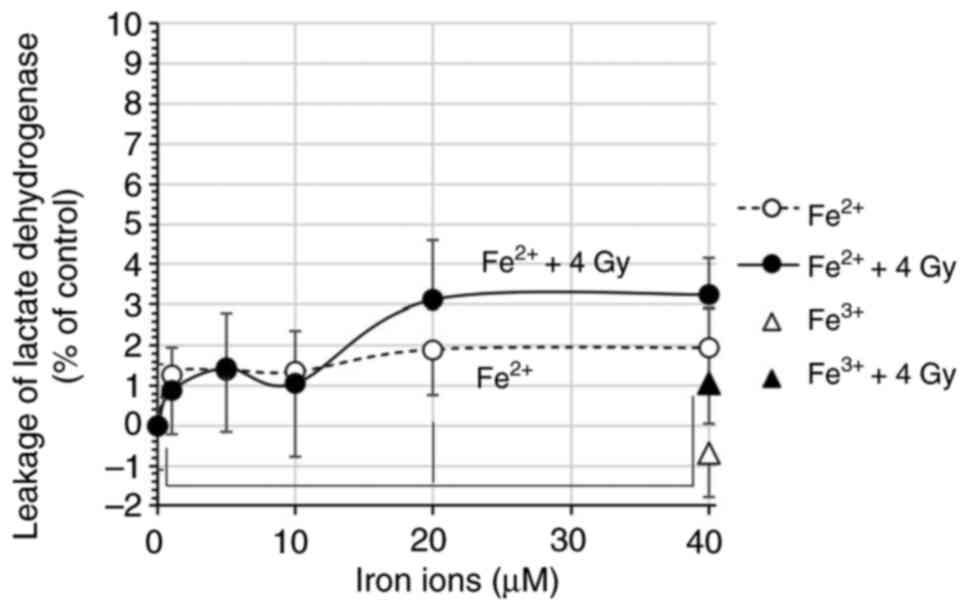
The leakage of lactate dehydrogenase, a marker for
loss of membrane integrity, increased with rising concentrations of
Fe2+, reaching 1.9-fold at 40 µM (Fig. 3). When combined with X-rays, the
lactate dehydrogenase leakage increased to 3.2-fold compared to the
control, indicating exacerbated membrane disruption.
Fe3+ at the same concentration did not increase lactate
dehydrogenase leakage, further highlighting the distinct effects of
Fe2+ (Fig. 3).
DOPC and DOPS model membranes
Subsequently, model membranes composed of DOPC and
DOPS in an 8:2 molar ratio were prepared to investigate
membrane-specific oxidative stress further. The resulting liposomes
had an average diameter of 134 nm and a zeta potential of -15.3 mV
(Fig. 4). The presence of anionic
lipids such as DOPS likely facilitates electrostatic interactions
with Fe2+, while the unsaturated acyl chains of DOPC
contribute to high membrane fluidity, rendering the system more
susceptible to lipid peroxidation. While lipid droplets have been
proposed to buffer ferroptotic stress by sequestering iron or
storing lipophilic antioxidants, our study focused on phospholipid
bilayer liposomes as a model of membrane lipids. Thus, the observed
effects mainly reflect direct Fe2+/X-ray interactions
with phospholipid membranes rather than lipid droplet-mediated
mechanisms.
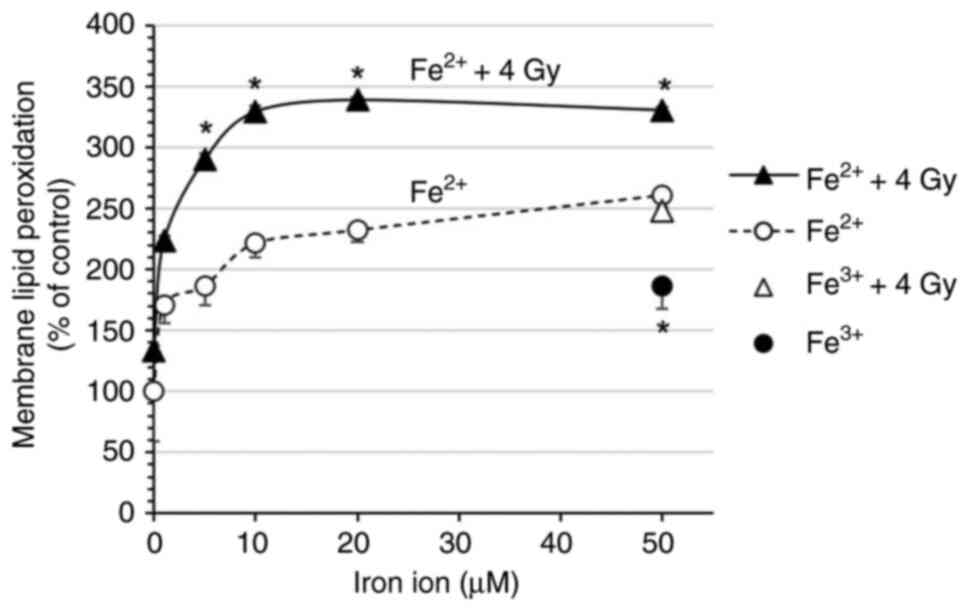
Effects of Fe2+ on lipid
peroxidation
Lipid peroxidation in 100 µM liposomes, measured
using Liperfluo, increased with Fe2+ treatment, reaching
261.1% at a concentration of 50 µM (Fig.
5). X-ray exposure alone increased peroxidation to 133.6%, and
the combination of Fe2+ and X-rays further elevated it
to 330.4%. Fe2+ induced a more modest increase and
exhibited less synergy with X-rays, indicating that Fe2+
has greater redox activity under X-ray exposure.
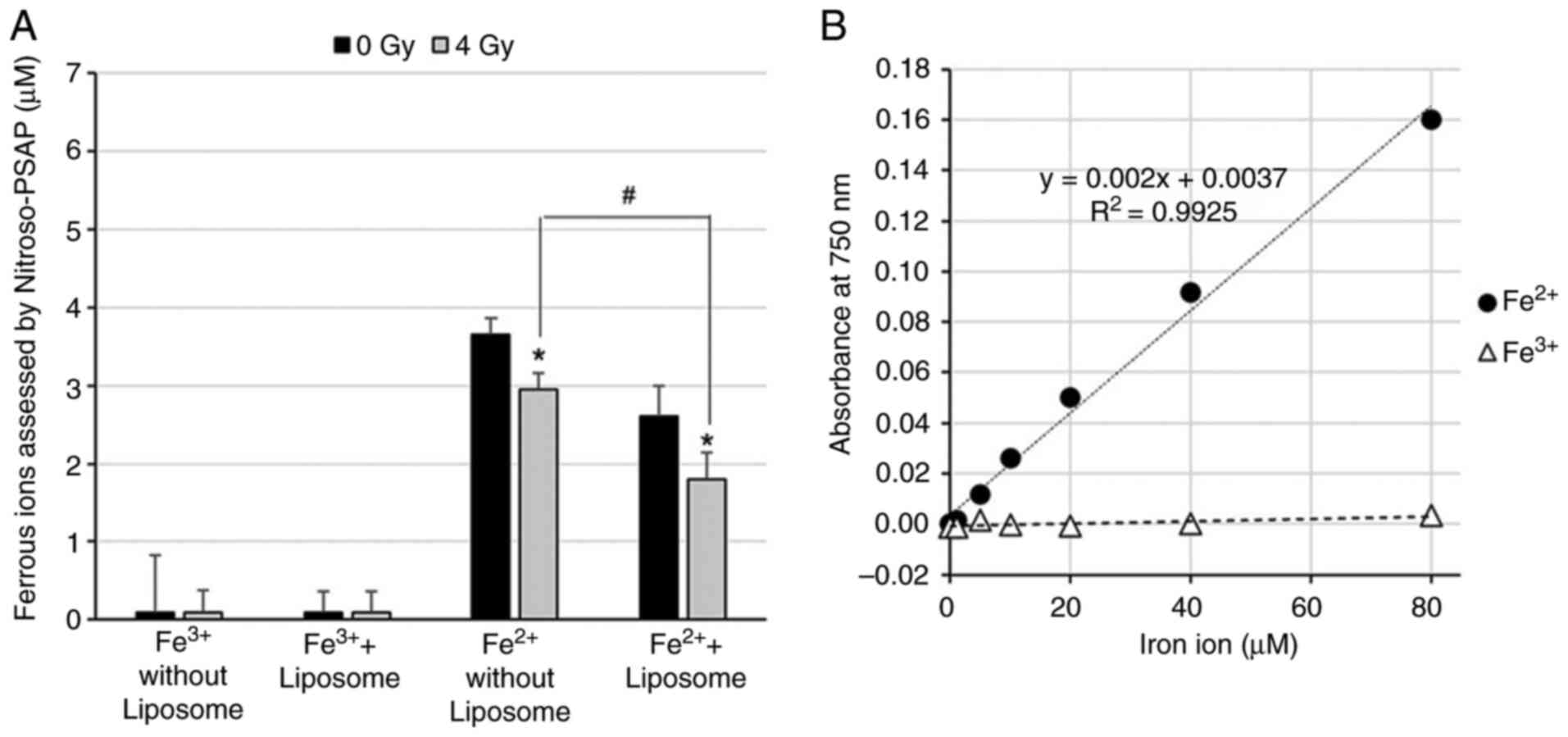
To investigate the oxidation of Fe2+, its
residual concentration was measured using the Nitroso-PSAP method.
A calibration curve was confirmed to be linear in the range of 0-80
µM we measured Fe2+ before measurements (Fig. 5). Upon adding 100 µM liposomes to 50
µM Fe2+, the detectable amount of Fe2+ was
3.65 µM. X-ray exposure alone reduced Fe2+ to 2.95 µM,
and the combination of liposomes and X-rays further reduced it to
1.80 µM (Fig. 6). Scatter plot data
were fitted with linear trendlines, and the calibration curve shown
in Fig. 6 was obtained using the
linear regression (least-squares) function in Microsoft Excel.
Discussion
The results of the present study demonstrated that
Fe2+ at low concentrations promoted fibroblast
proliferation; however, at higher concentrations, it induced
oxidative stress and membrane damage, with X-ray exposure
amplifying these effects. By contrast, Fe3+ had a
minimal effect, underscoring the distinct redox activity of
Fe2+. While the present study did not directly assess
canonical ferroptosis markers, such as GPX4 or acyl-CoA synthetase
long-chain family member 4 (ACSL4), the observed iron-dependent
lipid peroxidation and membrane damage are consistent with early
ferroptosis-related oxidative processes.
It is known that Fe2+ rapidly binds to
intracellular proteins, such as ferritin and transferrin, thereby
modulating its redox activity (25).
These mechanisms may help explain the oxidative stress observed
herein, although further experiments are required for direct
validation. In addition, lysosomal iron release under oxidative
stress has been reported as a potential contributor to cytosolic
Fe2+ accumulation and lipid peroxidation (26,27). The
results of the present study suggest that Fe2+ plays a
central role in oxidative stress generation, likely via Fenton-type
reactions (1,28). Although the present study did not
verify lysosomal escape, future studies using lysosomal markers or
inhibitors may clarify this mechanism.
Previous studies have reported that lipophilic
antioxidants, such as ferrostatin-1 and liproxstatin-1 inhibit the
Fe2+-driven peroxidation of polyunsaturated
phospholipids during ferroptosis (29). Although reduced GSH is a hydrophilic
and non-lipid-permeable antioxidant, it significantly suppressed
lipid peroxidation in the present study. It was noted that
physiological intracellular reduced GSH concentrations are
generally in the millimolar range (30), whereas the supplementation in the
present study was at the micromolar level. The observed protective
effect may therefore reflect localized antioxidant enhancement or
the stabilization of extracellular Fe2+, rather than a
direct mimic of intracellular reduced GSH levels. This effect is
likely attributable to the antioxidant properties of extracellular
reduced GSH, which may scavenge reactive oxygen species generated
under X-ray irradiation and Fe2+ exposure, thereby
suppressing lipid peroxidation at the plasma membrane. Therefore,
the GSH effects observed herein should be interpreted within the
context of an in vitro model, rather than as a direct
representation of intracellular antioxidant regulation.
Cell membranes, rich in polyunsaturated fatty acids,
are highly susceptible to lipid peroxidation, a key trigger of
ferroptosis following irradiation (31). It has been shown that 8 Gy X-rays
induce significant membrane lipid peroxidation in human skin
fibroblasts, as evidenced by the accumulation of malondialdehyde
(32). Furthermore, X-ray exposure
affects the utilization of reduced GSH and increases mitochondrial
reactive oxygen species, ultimately disrupting cellular redox
balance (33). Previous research
using ultraviolet A has also demonstrated close associations among
reactive oxygen species production, lipid peroxidation and lactate
dehydrogenase leakage in fibroblasts (34). The findings of the present study are
consistent with these reports, confirming that oxidative stress
plays a central role in radiation-induced membrane damage. Notably,
while Fe2+ alone induced oxidative stress and inhibited
cell proliferation, significant membrane lipid peroxidation was
only observed when Fe2+ was combined with X-ray
irradiation, suggesting a synergistic cytotoxic effect.
The present study used DOPC/DOPS (8:2) liposomes as
a model system to evaluate membrane-specific oxidative stress.
These anionic, highly fluid membranes are expected to interact
electrostatically with Fe2+. They are prone to lipid
peroxidation (23), allowing the
focus on direct Fe2+/X-ray effects on phospholipid
bilayers, rather than lipid droplet-mediated mechanisms.
Consistent with this expectation, both
Fe2+ and X-ray exposure enhanced lipid peroxidation, and
their combination had a synergistic effect, whereas Fe3+
induced only modest changes. This indicates that Fe2+
possesses higher redox activity under X-rays and is a more potent
driver of membrane damage. Notably, Fe2+ concentration
measurements revealed accelerated oxidation to Fe3+ in
the presence of membranes and X-rays, supporting the notion that
phospholipid bilayers facilitate iron redox cycling. This behavior
qualitatively resembles the mechanism of the Fricke dosimeter, in
which irradiation drives the Fe2+ → Fe3+
conversion. These findings highlight that membrane environments are
not passive targets, but active modulators of iron redox chemistry.
Notably, previous research has shown that the Fenton reaction at
air-water interfaces proceeds over 1,000 times faster than in bulk
solution, producing high-valent iron-oxo species (Fe(IV)=O) without
generating hydroxyl radicals (35).
This is likely due to partial solvation and to increased
accessibility of oxidants, such as hydrogen peroxide, to the iron
center at the interface. In a related study, lactoferrin enhanced
the Fenton reaction and increased hydroxyl radical production under
X-ray exposure (15), suggesting
that iron-binding proteins may influence oxidative reactions in
biological systems. In the present study, it was observed that
X-ray exposure promoted Fe2+ oxidation in the presence
of DOPC/DOPS (8:2) liposomes, supporting the hypothesis that
cellular or membrane-like environments modulate redox reactions
involving Fe2+.
In cancer settings, such as clear cell renal cell
carcinoma (ccRCC), the composition of lipid droplets (e.g., the
enrichment of polyunsaturated fatty acids vs. monounsaturated fatty
acids) and the accumulation of droplets have been reported to
influence ferroptosis sensitivity (36,37).
While the fibroblast and liposome system used herein does not
directly address this mechanism, these findings highlight how
membrane lipid composition can broadly shape susceptibility to
ferroptosis. Furthermore, long non-coding RNAs (lncRNAs), such as
LINC00336 and MALAT1 have been shown to regulate ferroptosis by
sponging miRNAs in cancer models (38). In ccRCC, ferroptosis-related genes,
such as ACSL4 have been identified as potential regulatory targets
(39). Key effectors of ferroptosis,
including GPX4 and ACSL4, may be regulated by lncRNA-miRNA networks
in specific cancer contexts. Although these regulatory pathways
were not addressed in the present fibroblast-based model, they
illustrate how membrane lipid oxidation may intersect with broader
ferroptosis regulatory networks in cancer contexts.
In summary, in the present study, Fe2+
promoted cell proliferation at low concentrations, whereas it
induced oxidative stress and membrane damage at higher
concentrations, effects that were amplified by X-ray exposure.
Antioxidants suppressed these effects, and a liposome model
confirmed that the membrane is involved in Fe2+
oxidation. These findings provide insight into ferroptosis
mechanisms and highlight the therapeutic potential and
environmental risks associated with iron and ionizing
radiation.
In conclusion, the present study reveals a
synergistic effect of Fe2+ and X-rays in promoting
oxidative membrane damage in a fibroblast complemented by a
liposome model. Fe2+ induces lipid peroxidation through
membrane interactions, impairing cell proliferation, while X-rays
amplify this oxidative stress. Given the essential role of
fibroblasts in tissue regeneration, these findings underscore the
biological importance of Fe2+-mediated damage
exacerbated by radiation. This insight contributes to a more
in-depth understanding of iron-dependent oxidative mechanisms
relevant to ferroptosis and may inform future strategies exploring
ferroptosis-targeted cancer therapies under radiation exposure.
Further in vivo studies and antioxidant evaluations are
required to advance therapeutic and environmental applications.
Acknowledgements
The author would like to express his sincere
gratitude to Mie University (Tsu, Japan), particularly the
Radioisotope Experimental Facility and its dedicated staff, for
providing a supportive environment that enabled this research. The
author is also grateful to the Kanagawa Institute of Industrial
Science and Technology (KISTEC) for their invaluable assistance
with the particle size and zeta potential measurements of
liposomes, and to Emeritus Professor Tetsuro Yoshimura (Mie
University) for developing the liposome production apparatus and
for his kind guidance on the liposome preparation method.
Funding
Funding: No funding was received.
Availability of data and materials
The data generated in the present study may be
requested from the corresponding author.
Author's contributions
SK was involved in the conceptualization of the
study, as well as in the study methodology, and investigation, and
in the writing of the manuscript. SK confirms the authenticity of
all the raw data. The author has read and approved the final
manuscript.
Ethics approval and consent to
participate
Not applicable.
Patient consent for publication
Not applicable.
Competing interests
The author declares that he has no competing
interests.
References
|
1
|
Dixon SJ, Lemberg KM, Lamprecht MR, Skouta
R, Zaitsev EM, Gleason CE, Patel DN, Bauer AJ, Cantley AM, Yang WS,
et al: Ferroptosis: an iron-dependent form of nonapoptotic cell
death. Cell. 149:1060–1072. 2012.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Dixon SJ, Patel DN, Welsch M, Skouta R,
Lee ED, Hayano M, Thomas AG, Gleason CE, Tatonetti NP, Slusher BS
and Stockwell BR: Pharmacological inhibition of cystine-glutamate
exchange induces endoplasmic reticulum stress and ferroptosis.
Elife. 3(e02523)2014.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Shibata Y, Yasui H, Higashikawa K,
Miyamoto N and Kuge Y: Erastin, a ferroptosis-inducing agent,
sensitised cancer cells to X-ray irradiation via glutathione
starvation in vitro and in vivo. PLoS One.
14(e0225931)2019.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Liu J, An W, Zhao Q, Liu Z, Jiang Y, Li H
and Wang D: Hyperbaric oxygen enhances X-ray induced ferroptosis in
oral squamous cell carcinoma cells. Oral Dis. 30:116–127.
2024.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Zhang X, Wu Z, Lan H, Chen S, Wu J, Zhu L
and Xiao Y: Deferoxamine promotes recovery of bone marrow
hematopoietic function in mice exposed to a sublethal dose of X-ray
irradiation. Nan Fang Yi Ke Da Xue Xue Bao. 43:1577–1584.
2023.PubMed/NCBI View Article : Google Scholar : (In Chinese).
|
|
6
|
Zhang B, Liu H, Wang Y, Zhang Y and Cheng
J: Application of singlet oxygen-activatable nanocarriers to boost
X-ray-induced photodynamic therapy and cascaded ferroptosis for
breast cancer treatment. J Mater Chem B. 11:9685–9696.
2023.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Citrin DE, Prasanna PGS, Walker AJ,
Freeman ML, Eke I, Barcellos-Hoff MH, Arankalayil MJ, Cohen EP,
Wilkins RC, Ahmed MM, et al: Radiation-induced fibrosis: mechanisms
and opportunities to mitigate. report of an NCI workshop, September
19, 2016. Radiat Res. 188:1–20. 2017.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Müller K and Meineke V: Radiation-induced
alterations in cytokine production by skin cells. Exp Hematol. 35
(Suppl 1):S96–S104. 2007.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Hirose E, Noguchi M, Ihara T and Yokoya A:
Mitochondrial metabolism in X-Irradiated cells undergoing
irreversible cell-cycle arrest. Int J Mol Sci.
24(1833)2023.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Busato F, Khouzai BE and Mognato M:
Biological mechanisms to reduce radioresistance and increase the
efficacy of radiotherapy: State of the art. Int J Mol Sci.
23(10211)2022.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Shimura T, Totani R, Ogasawara H, Inomata
K, Sasatani M, Kamiya K and Ushiyama A: Effects of oxygen on the
response of mitochondria to X-irradiation and reactive oxygen
species-mediated fibroblast activation. Int J Radiat Biol.
99:769–778. 2023.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Truong K, Bradley S, Baginski B, Wilson
JR, Medlin D, Zheng L, Wilson RK, Rusin M, Takacs E and Dean D: The
effect of well-characterized, very low-dose x-ray radiation on
fibroblasts. PLoS One. 13(e0190330)2018.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Jiang J, Yang L, Xie Q, Liu X, Jiang J,
Zhang J, Zhang S, Zheng H, Li W, Cai X, et al: Synthetic vectors
for activating the driving axis of ferroptosis. Nat Commun.
15(7923)2024.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Bae C, Hernández Millares R, Ryu S, Moon
H, Kim D, Lee G, Jiang Z, Park MH, Kim KH, Koom WS, et al:
Synergistic effect of ferroptosis-inducing nanoparticles and X-Ray
irradiation combination therapy. Small. 20(e2310873)2024.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Kato S: Lactoferrin inhibits the
proliferation of IMR-32 neuroblastoma cells even under X-rays. Med
Int (Lond). 3(33)2023.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Kato S: Effects of platinum-coexisting
dopamine with X-ray irradiation upon human glioblastoma cell
proliferation. Hum Cell. 34:1653–1661. 2021.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Kato S: Under lithium carbonate
administration, nicotine triggers cell dysfunction in human
glioblastoma U-251MG cells, which is distinct from cotinine. Med
Int (Lond). 2(19)2022.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Jay-Gerin JP: Fundamentals of Water
Radiolysis. Encyclopedia. 5(38)2025.
|
|
19
|
Ishiyama M, Tominaga H, Shiga M, Sasamoto
K, Ohkura Y and Ueno K: A combined assay of cell viability and in
vitro cytotoxicity with a highly water-soluble tetrazolium salt,
neutral red and crystal violet. Biol Pharm Bull. 19:1518–1520.
1996.PubMed/NCBI View Article : Google Scholar
|
|
20
|
Eruslanov E and Kusmartsev S:
Identification of ROS using oxidized DCFDA and flow-cytometry.
Methods Mol Biol. 594:57–72. 2010.PubMed/NCBI View Article : Google Scholar
|
|
21
|
Soh N, Ariyoshi T, Fukaminato T, Nakajima
H, Nakano K and Imato T: Swallow-tailed perylene derivative: A new
tool for fluorescent imaging of lipid hydroperoxides. Org Biomol
Chem. 5:3762–3768. 2007.PubMed/NCBI View Article : Google Scholar
|
|
22
|
Kumar P, Nagarajan A and Uchil PD:
Analysis of cell viability by the lactate dehydrogenase assay. Cold
Spring Harb Protoc: Jun 1, 2018 (Epub ahead of print).
|
|
23
|
Kato S and Kuwata K: Pro-/anti-oxidative
properties of dopamine on membrane lipid peroxidation upon X-ray
irradiation. Radiat Phys Chem. 185(109518)2021.
|
|
24
|
El Behery M, Fujimura M, Kimura T and
Tsubaki M: Direct measurements of ferric reductase activity of
human 101F6 and its enhancement upon reconstitution into
phospholipid bilayer nanodisc. Biochem Biophys Rep.
21(100730)2020.PubMed/NCBI View Article : Google Scholar
|
|
25
|
Muckenthaler MU, Rivella S, Hentze MW and
Galy B: A red carpet for iron metabolism. Cell. 168:344–361.
2017.PubMed/NCBI View Article : Google Scholar
|
|
26
|
Cañeque T, Baron L, Müller S, Carmona A,
Colombeau L, Versini A, Solier S, Gaillet C, Sindikubwabo F,
Sampaio JL, et al: Activation of lysosomal iron triggers
ferroptosis in cancer. Nature. 642:492–500. 2025.PubMed/NCBI View Article : Google Scholar
|
|
27
|
Stockwell BR, Friedmann Angeli JP, Bayir
H, Bush AI, Conrad M, Dixon SJ, Fulda S, Gascón S, Hatzios SK,
Kagan VE, et al: Ferroptosis: A regulated cell death nexus linking
metabolism, redox biology, and disease. Cell. 171:273–285.
2017.PubMed/NCBI View Article : Google Scholar
|
|
28
|
Yang WS and Stockwell BR: Ferroptosis:
Death by lipid peroxidation. Trends Cell Biol. 26:165–176.
2016.PubMed/NCBI View Article : Google Scholar
|
|
29
|
Zilka O, Shah R, Li B, Friedmann Angeli
JP, Griesser M, Conrad M and Pratt DA: On the mechanism of
cytoprotection by ferrostatin-1 and Liproxstatin-1 and the role of
lipid peroxidation in ferroptotic cell death. ACS Cent Sci.
3:232–243. 2017.PubMed/NCBI View Article : Google Scholar
|
|
30
|
Montero D, Tachibana C, Rahr Winther J and
Appenzeller-Herzog C: Intracellular glutathione pools are
heterogeneously concentrated. Redox Biol. 1:508–513.
2013.PubMed/NCBI View Article : Google Scholar
|
|
31
|
Mao C, Lei G, Horbath A and Gan B:
Assessment of lipid peroxidation in irradiated cells. Methods Cell
Biol. 172:37–50. 2022.PubMed/NCBI View Article : Google Scholar
|
|
32
|
Kim BC, Shon BS, Ryoo YW, Kim SP and Lee
KS: Melatonin reduces X-ray irradiation-induced oxidative damages
in cultured human skin fibroblasts. J Dermatol Sci. 26:194–200.
2001.PubMed/NCBI View Article : Google Scholar
|
|
33
|
Shimura T, Nakashiro C, Fujiwara K, Shiga
R, Sasatani M, Kamiya K and Ushiyama A: Radiation affects
glutathione redox reaction by reduced glutathione peroxidase
activity in human fibroblasts. J Radiat Res. 63:183–191.
2022.PubMed/NCBI View Article : Google Scholar
|
|
34
|
Morlière P, Moysan A, Santus R, Hüppe G,
Mazière JC and Dubertret L: UVA-induced lipid peroxidation in
cultured human fibroblasts. Biochim Biophys Acta. 1084:261–268.
1991.PubMed/NCBI View Article : Google Scholar
|
|
35
|
Enami S, Sakamoto Y and Colussi AJ: Fenton
chemistry at aqueous interfaces. Proc Natl Acad Sci USA.
111:623–628. 2014.PubMed/NCBI View Article : Google Scholar
|
|
36
|
Kim JW, Lee JY, Oh M and Lee EW: An
integrated view of lipid metabolism in ferroptosis revisited via
lipidomic analysis. Exp Mol Med. 55:1620–1631. 2023.PubMed/NCBI View Article : Google Scholar
|
|
37
|
Klasson TD, LaGory EL, Zhao H, Huynh SK,
Papandreou I, Moon EJ and Giaccia AJ: ACSL3 regulates lipid droplet
biogenesis and ferroptosis sensitivity in clear cell renal cell
carcinoma. Cancer Metab. 10(14)2022.PubMed/NCBI View Article : Google Scholar
|
|
38
|
Wang M, Mao C, Ouyang L, Liu Y, Lai W, Liu
N, Shi Y, Chen L, Xiao D, Yu F, et al: Long noncoding RNA LINC00336
inhibits ferroptosis in lung cancer by functioning as a competing
endogenous RNA. Cell Death Differ. 26:2329–2343. 2019.PubMed/NCBI View Article : Google Scholar
|
|
39
|
Guo N: Identification of ACSL4 as a
biomarker and contributor of ferroptosis in clear cell renal cell
carcinoma. Transl Cancer Res. 11:2688–2699. 2022.PubMed/NCBI View Article : Google Scholar
|