Introduction
Periodontitis is a disease characterized by
inflammation with multiple aetiological factors, resulting in the
deterioration of tissues encasing the tooth structure, including
the gingiva, alveolar bone, cementum and periodontal ligament.
According to estimates from the Global Burden of Disease 2019
study, periodontal disorders influence the health of >1 billion
individuals worldwide (1). The
inflammatory response of the host immune system to bacteraemia,
although protective, can promote chronic infection by modulating
biofilm formation and bacterial virulence, thereby disrupting
crucial homeostatic mechanisms essential for maintaining
periodontal health (2). As the
disease progresses, inflammatory and resident cells in the
periodontium secrete or activate mediators such as prostaglandin
E2, cytokines, chemokines, matrix metalloproteinases and signalling
proteins, working together to accelerate the destruction of both
soft and hard tissues (3,4). Early identification of inflammatory
diseases and prompt treatment can markedly improve health outcomes
and enhance disease prognosis.
A biomarker is a characteristic that is objectively
measured and evaluated as an indicator of normal biological
processes, pathogenic processes or pharmacological responses to a
therapeutic intervention (4).
Biomarkers can be derived from multiple sources, such as saliva,
serum, gingival crevicular fluid (GCF) and dental plaque (3). They are employed for prompt
identification of disease, monitoring its progression, predicting
potential outcomes, evaluating treatment efficacy and customizing
medical interventions (4). During
infection, the immune system initiates an inflammatory response by
activating various immune cells, including lymphocytes, plasma
cells and macrophages, and releasing cytokines. This immune
activation leads to the release of multiple inflammatory biomarkers
into the bloodstream. Maintaining equilibrium between
pro-inflammatory and anti-inflammatory markers is crucial for
modulating the host immune response to antigenic challenges,
ensuring an effective yet controlled defence mechanism (5).
Lactoferrin is a glycoprotein from the transferrin
family that has a high capacity to bind iron. It is produced by
exocrine glands and discharged by immune cells such as neutrophils,
particularly at sites of infection or inflammation (6). Lactoferrin is found in various body
fluids including saliva, dental plaque, GCF, digestive secretions,
bronchial secretions, bile, urine, breast milk, serum and tears
(7). The primary function initially
attributed to lactoferrin was its antimicrobial activity, driven by
its capacity to bind iron, which is crucial for the multiplication
and survival of bacteria. It also has a key function in numerous
bodily processes, including the uptake of iron in the intestines,
immune system modulation, anti-viral and anti-oxidant activities
(8). Lactoferrin concentrations have
been reported to be increased in stimulated whole saliva of
individuals with chronic periodontitis and are positively
associated with a probing pocket depth (PPD) of ≥6 mm (9). In addition, a previous study
demonstrated that lactoferrin levels are elevated in patients with
periodontitis relative to healthy individuals, whereas they decline
after periodontal therapy (10).
Furthermore, higher lactoferrin levels are associated with worse
clinical parameters both pre- and post-therapy. Ramenzoni et
al (11) reported elevated
lactoferrin levels in the periodontal pockets of patients with
periodontitis, with increased concentrations also observed at
clinically healthy sites within the same subjects. These findings
suggest that the role of lactoferrin in periodontitis remains
unclear and warrants further investigation. Assessing lactoferrin
concentration in GCF holds marked potential as a diagnostic tool,
offering insights into the presence of inflammation, the extent of
oxidative stress and the effectiveness of periodontal treatment
interventions (12). Lactoferrin
exhibits a dual function in modulating immunological responses,
functioning as a mediator with pro- or anti-inflammatory effects,
contingent upon the situation and signalling pathways it engages
(13). Due to this dual effect, the
precise role of lactoferrin in periodontal disease remains
unclear.
So far, to the best of our knowledge, one systematic
review and meta-analysis has explored the role of lactoferrin in
bone regeneration (14); however,
there remains a lack of comprehensive reviews focusing on its
potential as a diagnostic biomarker in periodontal disease. To
address this gap, the present meta-analysis investigated the
function of lactoferrin serving as an indicator of periodontitis by
examining its association with key clinical indicators, including
PPD, clinical attachment loss (CAL) and gingival index (GI). The
present analysis aimed to clarify the diagnostic and prognostic
value of lactoferrin in detecting disease presence and progression,
thereby contributing to an improved understanding of its relevance
in periodontal diagnostics. The clinical question in the present
meta-analysis, formulated using the PICOS framework, aimed to
determine whether lactoferrin levels vary between patients with
periodontitis (Population) and healthy subjects (Comparison). The
study assessed lactoferrin concentrations in saliva, serum or GCF
(Outcome) with respect to the occurrence of periodontitis
(Intervention), including only studies that used ELISA as the
detection method (Study design). Furthermore, the analysis explored
the association between lactoferrin levels, periodontal disease
severity and standard clinical periodontal indicators to assess its
potential role as a diagnostic and prognostic biomarker.
Materials and methods
Protocol development and research
question
The methodology for the present systematic review
and meta-analysis complied with Preferred Reporting Items for
Systematic Reviews and Meta-Analyses (PRISMA) guidelines to ensure
methodological rigor (15). The
meta-analysis was registered with PROSPERO (https://www.crd.york.ac.uk/prospero/; registration no.
CRD420251003457). The study design included case-control,
cross-sectional, randomized controlled trial and non-randomized
controlled trial studies that utilized ELISA as the detection
method for lactoferrin. Only studies utilizing ELISA were included
to ensure methodological consistency, enable robust quantitative
comparisons and reduce heterogeneity stemming from assay-related
variability. ELISA remains the most standardized, validated and
widely adopted platform for lactoferrin detection in both clinical
and translational research settings (16).
Data collection and search
procedures
A thorough and systematic electronic search was
independently conducted by three researchers using major databases,
including PubMed (https://pubmed.ncbi.nlm.nih.gov/), Google Scholar
(https://scholar.google.com/), the
Cochrane Library (https://www.cochranelibrary.com/) and ResearchGate
(https://www.researchgate.net/);
additional records were identified through manual screening of
reference lists and citation tracking of relevant studies. The
literature search was conducted between December 1, 2024 and
February 28, 2025, ensuring thorough coverage of relevant
literature. Additionally, pertinent studies were identified through
hand-searching the bibliographies of the selected articles and
related review articles. The following MeSH terms were used for
searching: ‘Periodontitis’, ‘periodontal disease’, ‘lactoferrin’,
‘probing pocket depth’, ‘clinical attachment level’ and ‘gingival
index’. Combining these terms with logical operators such as AND or
OR search phrases such as [‘Periodontitis’ (MeSH) OR ‘Periodontal
Diseases’(MeSH)] AND ‘Lactoferrin Protein’(MeSH)] [‘Periodontal
Pocket’(MeSH) OR ‘Clinical Attachment Loss’(MeSH) OR ‘Gingival
Index’(MeSH)] were used to identify relevant studies for the
present meta-analysis.
Eligibility criteria
Studies assessing the association between
lactoferrin concentration and periodontal indicators such as PPD,
CAL and GI in relation to periodontal disease were included. The
studies included were original, peer-reviewed, full-text articles
in English. Eligible studies involved human participants, used
ELISA to quantify lactoferrin in biological fluids (serum, saliva
or GCF), considered lactoferrin as a biomarker, and included
periodontitis and healthy control groups for comparison. The
present meta-analysis encompasses studies published between 1993
and 2022, offering a broad and longitudinal perspective on the
evolving body of evidence. The present study excluded case reports,
reviews, letters, commentaries and conference abstracts regardless
of data availability, as well as unpublished, preprint or grey
literature with poor methodology. In addition, animal or in
vitro studies without human data were not considered. Research
not focused on periodontal disease or without proper periodontal
assessment was excluded, along with studies failing to measure
lactoferrin levels, lacking a control group comparison, or not
evaluating the diagnostic accuracy of lactoferrin. Non-English
studies were also excluded unless a translation was available.
Data extraction
Independent, comprehensive searches of the
literature were undertaken by the authors across various databases.
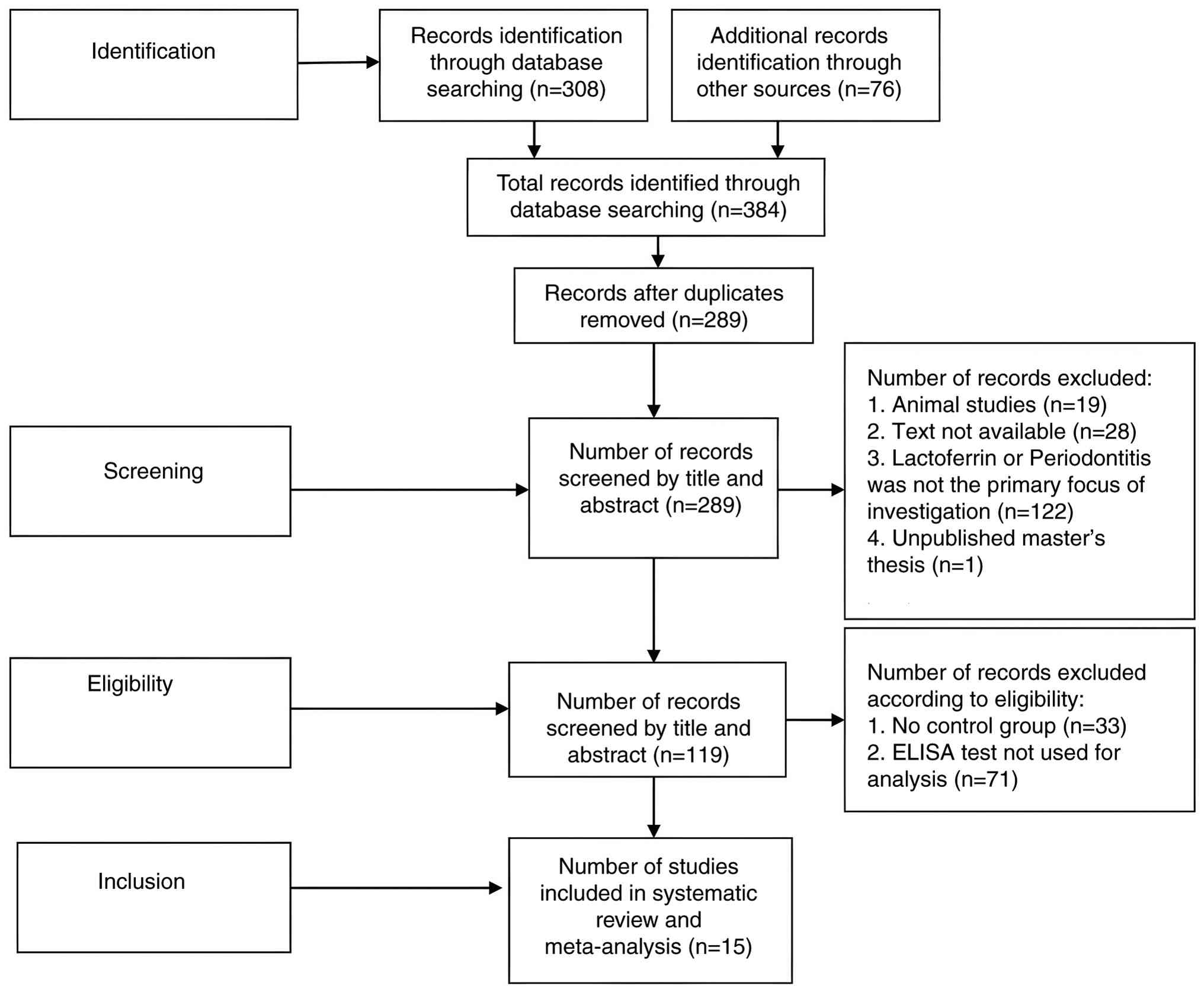
The search process, outlined according to the PRISMA flowchart, is
shown in Fig. 1.
Evaluation of methodological
quality
The robustness of the study methods and established
tools were used to assess possible sources of bias: Case-control
and cross-sectional studies were evaluated using the
Newcastle-Ottawa Scale; randomized controlled trials were assessed
with the Jadad Scale; and non-randomized controlled trials were
appraised utilizing the Modified Methodological Index for
Non-Randomized Studies (Modified MINORS) (17-19).
Data items
Data from the included studies were exported into
Microsoft Excel (Office 2013; Microsoft Corporation) for analysis.
For the subsequent analysis, the authors independently extracted
key information from each eligible study, which included: The first
author, publication year, country, study design, sample type
(serum, saliva or GCF), molecular analysis method (ELISA), measured
lactoferrin levels, PPD, CAL and GI, and the total number of
participants, including both controls and patients with
periodontitis (Table I). Any
discrepancies in study selection, quality assessment or data
extraction were resolved through consensus discussions among the
authors.
 | Table IStudy characteristics. |
Table I
Study characteristics.
| | | Number of
subjects | Concentration of
lactoferrin, ng/ml | PPD, mm | CAL, mm | GI, % | |
|---|
| First author, year,
country | Study type | Sample type | Case group | Control group | Case group | Control group | Case group | Control group | Case group | Control group | Case group | Control group | New-castle-Ottawa
Scale | (Refs.) |
|---|
| Orhue, 2022,
Nigeria | CS study | Saliva | 51 | 51 | 6,740±590 | 5,270±610 | 5.1±0.7 | 2.1±0.60 | 4.2±0.06 | 1.4±0.9 | 1.8±0.58 | 0.6±0.08 | 8 | (12) |
| Ramenzoni, 2021,
Switzerland | CC study | Saliva | 10 | 10 | 1,253±139.74 | 507.5±64 | 7.15±1.06 | 2.95±0.38 | 6.12±0.60 | 1.95±0.07 | 1.9±0.5 | 0.8±0.9 | 8 | (11) |
| Vozna, 2021,
Ukraine | CC study | GCF | 126 | 20 | 303.19±75.31 | 16.87±30.34 | 5.2±0.8 | 1.9±0.4 | 4.3±0.04 | 1.2±0.07 | 1.6±0.6 | 0.5±0.08 | 7 | (24) |
| Mandra, 2018,
Russia | CC study | Saliva | 45 | 30 | 6,188.2±928.3 | 756.53±100.6 | 6.2±0.9 | 2.2±0.52 | 4.1±0.05 | 1.99±0.09 | 1.5±0.05 | 0.8±0.07 | 7 | (30) |
| Talib, 2018,
Iraq | CC study | Saliva | 33 | 15 | 47.35±2.4 | 3.9±0.64 | 0.93±0.5 | 0.1±0.50 | 0.91±0.07 | 0.2±0.08 | 0.95±0.8 | 0.89±0.6 | 8 | (25) |
| Yadav, 2014,
India | NRI study | GCF | 25 | 25 | 1,857.21±91.5 | 75.34±7.13 | 0.55±0.7 | 0.825±0.8 | 0.50±0.7 | 0.73±0.07 | 0.67±0.02 | 0.72±0.9 | 13a | (26) |
| Glimvall, 2012,
Sweden | CC study | Saliva | 17 | 17 | 2,710±2,360 | 1,120±1,980 | 0.45±0.6 | 0.25±0.08 | 0.41±0.08 | 0.21±0.06 | 0.9±0.04 | 0.8±0.08 | 8 | (9) |
| Shimizu, 2011,
Japan | RCT | Saliva | 37 | 35 | 9,800±6500 | 9,300±3,500 | 4.16±0.09 | 4.04±0.09 | 4.18±0.09 | 4.04±0.09 | 1.34±0.06 | 0.34±0.06 | 4b | (33) |
| Wu, 2009,
China | CC study | Saliva | 5 | 5 | 6,800±800 | 8,900±500 | 5.6±1.02 | 2.2±0.9 | 5.2±0.4 | 1.6±0.05 | 1.4±0.6 | 0.9±0.8 | 7 | (31) |
| Wei, 2004,
Taiwan | CC study | GCF | 19 | 8 |
192,750±156,540 |
193,250±168,120 | 5.12±1.04 | 1.86±0.55 | 4.9±0.6 | 1.08±0.07 | 2.48±0.55 | 0.1±0.6 | 7 | (34) |
| Figueredo, 2000,
Sweden | CC study | GCF | 13 | 12 | 237.1±110.9 | 352.8±191.1 | 5.5±0.6 | 2.4±0.4 | 4.2±0.7 | 1.9±0.08 | 1.9±0.3 | 1.3±0.4 | 7 | (35) |
| Figueredo, 1999,
Sweden | CC study | Serum | 15 | 15 | 558±96 | 193±32 | 5.3±1.03 | 2.6±0.2 | 4.1±0.6 | 1.2±0.08 | 1.8±0.5 | 0.9±0.2 | 8 | (27) |
| Tsai, 1998,
Taiwan | CS study | GCF | 66 | 23 | 552.16±419.4 | 69.36±22.2 | 5.69±1.8 | 2.39±0.50 | 5.2±0.6 | 1.9±0.06 | 2.40±0.61 | 0.22±0.42 | 8 | (28) |
| Murray, 1995,
UK | CS study | GCF | 10 | 10 | 106.2±193.9 | 619.92±531.7 | 5.8±1.81 | 0.9±0.69 | 5.3±0.4 | 1.8±0.8 | 2.3±0.82 | 0.1±0.31 | 7 | (32) |
| Adonogianaki, 1993,
UK | CS study | GCF | 24 | 26 | 217±111 | 95±37 | 5.04±0.29 | 1.54±0.10 | 4.9±0.5 | 1.1±0.5 | 2.46±0.17 | 0.38±0.10 | 7 | (29) |
Statistical analysis
The analysis included 15 studies, utilizing the odds
ratio (OR) as the primary effect size index. The analysis employed
a random-effects model, assuming that the included studies
represent a random sample drawn from a broader population of
relevant studies, which enables the results to be generalized
beyond the specific data analysed (20). To evaluate the association between
lactoferrin levels in saliva, serum or GCF and periodontitis, the
standardized mean difference (SMD) with a 95% confidence interval
(CI) was calculated, incorporating a random-effects model to
account for potential heterogeneity among studies. For all
outcomes, SMD and the corresponding CI were reported as the effect
measures. Additionally, a prediction interval was computed to
estimate the range within which the true effect in future studies
might lie (21). The statistical
significance of the pooled SMD between patients with periodontitis
and healthy controls was assessed using a Z-test. Heterogeneity
across the studies was assessed by applying the Q-statistic,
I2-statistic, τ and τ2, along with visual
examination of the forest plots (22). Publication bias was assessed both
qualitatively and quantitatively through funnel plot analysis,
Begg's rank correlation test, and Egger's linear regression test
(23). Statistical analyses were
executed with the aid of Comprehensive Meta-Analysis Software
(version 4; https://meta-analysis.com).
Two-tailed P<0.05 was considered to indicate a statistically
significant difference.
Results
Overview of studies included. Number
of studies
Overall, 384 studies were retrieved from various
databases between December 1, 2024 and February 28, 2025. Among the
384 studies, 15 eligible studies, as determined by the inclusion
and exclusion criteria, were included in the meta-analysis
representing data from 798 participants (496 cases and 302
controls).
Study characteristics. The 15 included
studies consisted of nine case-control studies, four
cross-sectional studies, one non-randomized interventional study
and one randomized controlled trial. Lactoferrin levels were
evaluated across various studies in different biological fluids,
including saliva, serum and GCF, to compare lactoferrin
concentration and periodontal parameters (PPD, CAL and GI) between
patients with periodontitis and healthy individuals. The studies
were conducted across different regions of Asia, Europe and Africa,
with publication dates ranging from 1993 to 2022. All studies
assessed with the Newcastle-Ottawa Scale had quality scores of 7 or
8. For the randomized controlled trial and non-randomized
interventional study, the Jadad scale and Modified MINORS scale
were used as the Newcastle-Ottawa Scale cannot be used for these
studies (17). The Jadad scale score
was 4 for the randomized controlled trial and the Modified MINORS
scale score was 13 for the non-randomized interventional study.
Analysis revealed notable differences in lactoferrin levels and
periodontal parameters comparing patients with periodontitis to
healthy controls: Salivary lactoferrin was increased in 156
patients out of 230 in the test group; in GCF, lactoferrin levels
were increased in 241 patients out of 297 patients in the test
group; and serum lactoferrin was elevated in 15 patients compared
with 15 controls. In addition, the clinical parameters (PPD, CAL,
GI) were higher in 471 patients out of 496 patients in the test
group.
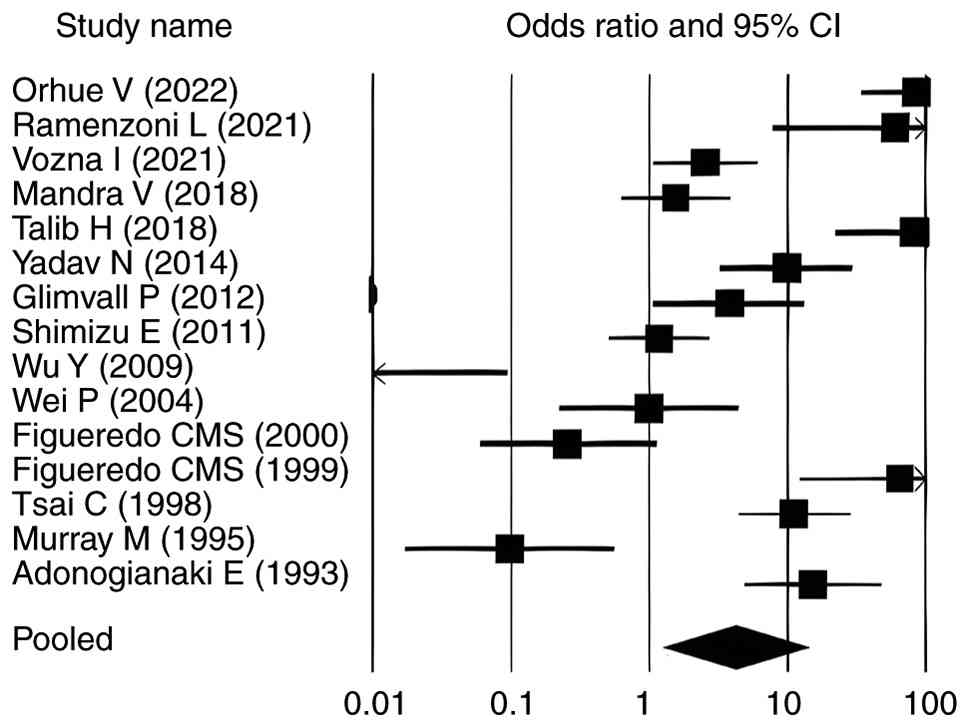
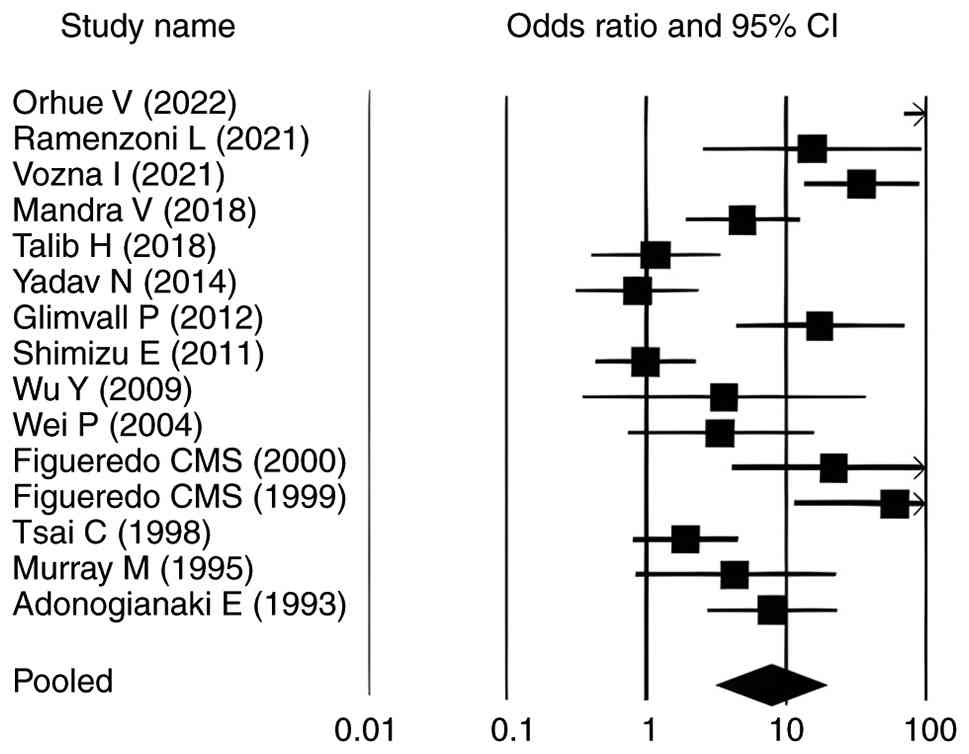
Overall effect size analysis
The pooled analysis of the 15 included studies
revealed a significant association between periodontitis and
elevated lactoferrin levels (SMD=2.630, P<0.010; 95% CI:
1.140-11.180) (Table II). Among
these, 10 studies (9,11,12,24-30)
reported statistically significant positive associations
(P<0.05) with OR>1. The remaining five studies (31-35)
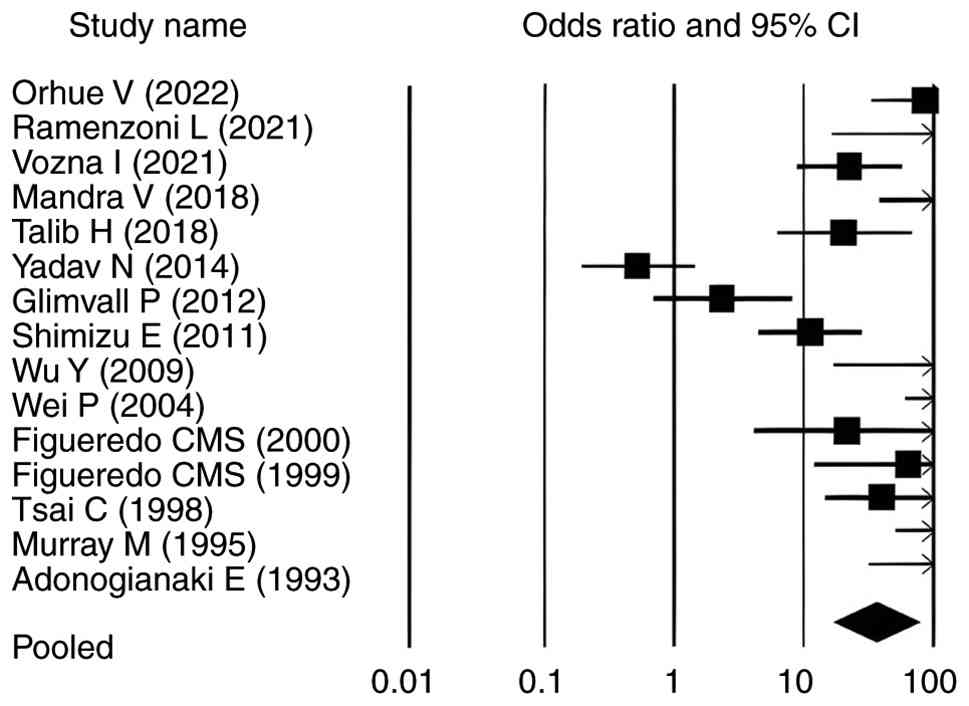
did not show statistically significant associations. For PPD, the
pooled SMD was 8.630 (P<0.010; 95% CI: 17.070-90.340) and 13
studies (11,12,24,25,27-35)
demonstrated a statistically significant association between
elevated lactoferrin levels and increased PPD in individuals with
periodontitis (OR>1; P<0.05). In contrast, studies by Yadav
et al (26) and Glimvall
et al (9), despite reporting
OR>1, did not show statistically significant associations, as
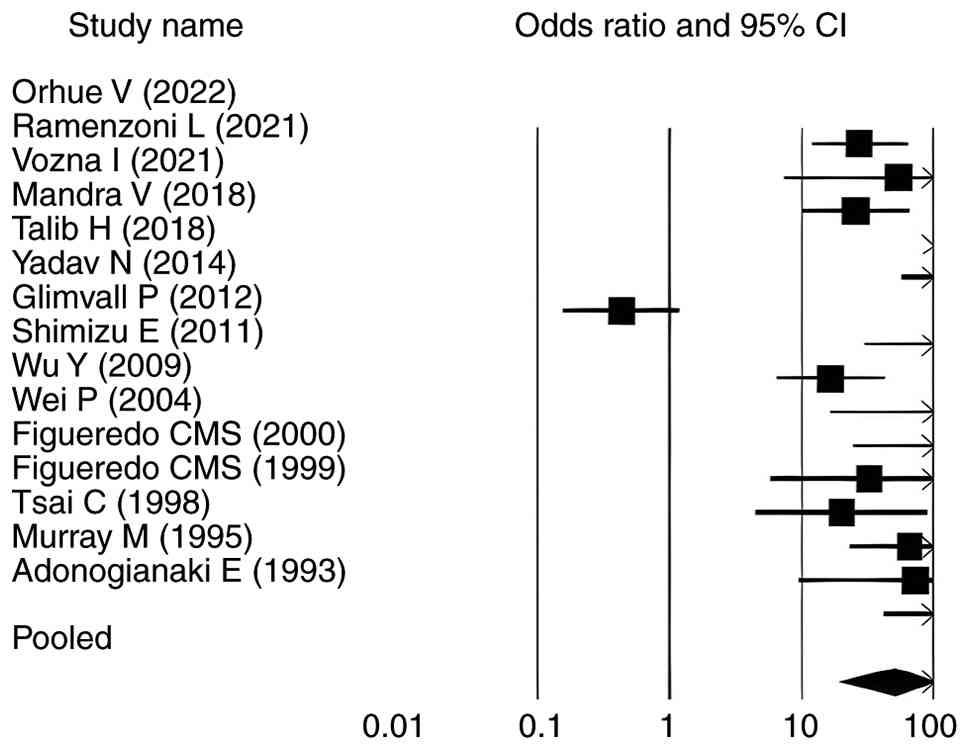
their corresponding P-values were >0.05 (Table III). Similarly, for CAL, the pooled
SMD was 8.382 (P<0.010; 95% CI: 19.855-123.053), with 14 studies
(9,11,12,24,25,27-35)
showing a significant association between elevated lactoferrin
levels and increased CAL in individuals with periodontitis and one
[Yadav et al (26)] reported
no association (Table IV).
Regarding GI, the pooled SMD was 4.288 (P<0.010; 95% CI:
2.914-17.654). Eight studies (9,11,12,24,27,29,30,35)
demonstrated a statistically significant association between
elevated lactoferrin levels and increased GI in individuals with
periodontitis (P<0.05). Among the remaining studies, five
reported (25,28,31,32,34)
OR>1 but did not reach statistical significance (P>0.05).
Shimizu et al (33) reported
a neutral OR of 1 with P>0.05, whereas Yadav et al
(26) showed an OR<1 with
P>0.05, indicating no significant association (Table V). Table
III, Table IV and Table V present the association between
elevated lactoferrin levels and the periodontal clinical parameters
PPD, CAL and GI in individuals with periodontitis.
 | Table IIPooled ORs with 95% CIs, Z-values and
P-values for lactoferrin levels across included studies under a
random-effects model. |
Table II
Pooled ORs with 95% CIs, Z-values and
P-values for lactoferrin levels across included studies under a
random-effects model.
| First author,
year | OR | 95% CI | Z-value | P-value | (Refs.) |
|---|
| Orhue, 2022 | 85.046 | 33.511-215.835 | 9.351 | <0.010 | (12) |
| Ramenzoni,
2021 | 60.171 | 7.866460.274 | 3.351 | <0.010 | (11) |
| Vozna, 2021 | 2.528 | 1.067-5.988 | 2.108 | <0.010 | (24) |
| Mandra, 2018 | 1.544 | 0.628-3.799 | 0.946 | <0.010 | (30) |
| Talib, 2018 | 82.474 | 21.996-309.231 | 6.544 | <0.010 | (25) |
| Yadav, 2014 | 9.857 | 3.278-29.642 | 4.073 | <0.010 | (26) |
| Glimvall, 2012 | 3.758 | 1.067-13.240 | 2.061 | 0.039 | (9) |
| Shimizu, 2011 | 1.188 | 0.514-2.749 | 0.403 | 0.687 | (33) |
| Wu, 2009 | 0.003 | 0.000-0.096 | -3.327 | 0.001 | (31) |
| Wei, 2004 | 0.994 | 0.222-4.449 | -0.007 | 0.994 | (34) |
| Figueredo,
2000 | 0.257 | 0.059-1.121 | -1.808 | 0.710 | (35) |
| Figueredo,
1999 | 65.531 | 12.277-349.797 | 4.895 | <0.010 | (27) |
| Tsai, 1998 | 11.184 | 4.408-28.375 | 5.083 | <0.010 | (28) |
| Murray, 1995 | 0.097 | 0.017-0.559 | -2.614 | 0.009 | (32) |
| Adonogianaki,
1993 | 15.186 | 4.862-47.426 | 4.682 | <0.010 | (29) |
| Random-effects
model | 3.960 | 1.140-11.180 | 2.630 | <0.010 | |
 | Table IIIPooled ORs with 95% CIs, Z-values and
P-values for probing pocket depth across included studies under a
random-effects model. |
Table III
Pooled ORs with 95% CIs, Z-values and
P-values for probing pocket depth across included studies under a
random-effects model.
| First author,
year | OR | 95% CI | Z-value | P-value | (Refs.) |
|---|
| Orhue, 2022 | 85.763 | 33.768-217.818 | 9.361 | <0.010 | (12) |
| Ramenzoni,
2021 | 150.129 |
16.265-1385.759 | 4.419 | <0.010 | (11) |
| Vozna, 2021 | 22.311 | 8.831-56.368 | 6.566 | <0.010 | (24) |
| Mandra, 2018 | 331.138 | 80.674 451.545 | 7.729 | <0.010 | (30) |
| Talib, 2018 | 20.306 | 6.184-66.676 | 4.963 | <0.010 | (25) |
| Yadav, 2014 | 3.174 | 0.823-8.437 | -1.426 | 0.154 | (26) |
| Glimvall, 2012 | 2.334 | 0.678-8.032 | -1.340 | 0.179 | (9) |
| Shimizu, 2011 | 11.224 | 4.026 31.307 | 4.330 | <0.010 | (33) |
| Wu, 2009 | 608.723 |
16.655-222,482.563 | 3.492 | <0.010 | (31) |
| Wei, 2004 | 61.250 | 6.106-558.127 | 3.369 | <0.010 | (34) |
| Figueredo,
2000 | 21.557 | 4.106-113.174 | 3.629 | <0.010 | (35) |
| Figueredo,
1999 | 49.358 | 11.337-215.196 | 4.876 | <0.010 | (27) |
| Tsai, 1998 | 40.351 | 14.583-111.617 | 7.120 | <0.010 | (28) |
| Murray, 1995 | 649.82 |
50.210-8410.164 | 6.853 | <0.010 | (32) |
| Adonogianaki,
1993 | 125.443 | 31.500-499.535 | 4.988 | <0.010 | (29) |
| Random-effects
model | 39.270 | 17.070-90.340 | 8.630 | <0.010 | |
 | Table IVPooled ORs with 95% CIs, Z-values and
P-values for clinical attachment loss across included studies under
a random-effects model. |
Table IV
Pooled ORs with 95% CIs, Z-values and
P-values for clinical attachment loss across included studies under
a random-effects model.
| First author,
year | OR | 95% CI | Z-value | P-value | (Refs.) |
|---|
| Orhue, 2022 | 27.661 | 11.959-63.979 | 7.760 | <0.010 | (12) |
| Ramenzoni,
2021 | 54.683 | 7.281-410.686 | 3.890 | <0.010 | (11) |
| Vozna, 2021 | 25.851 | 10.164-65.746 | 6.829 | <0.010 | (24) |
| Mandra, 2018 | 1,064.485 |
23.739-4,827.187 | 9.037 | <0.010 | (30) |
| Talib, 2018 | 244.912 |
57.657-1,040.328 | 7.454 | <0.010 | (25) |
| Yadav, 2014 | 0.432 | 0.156-1.197 | -1.613 | 0.107 | (26) |
| Glimvall, 2012 | 169.051 | 30.138-948.250 | 5.831 | <0.010 | (9) |
| Shimizu, 2011 | 16.801 | 6.455-43.730 | 5.781 | <0.010 | (9) |
| Wu, 2009 | 607.663 |
16.636-2,2196.580 | 3.491 | <0.010 | (31) |
| Wei, 2004 | 189.269 |
24.377-1,469.517 | 5.014 | <0.010 | (34) |
| Figueredo,
2000 | 32.631 | 5.843-182.234 | 3.971 | <0.010 | (35) |
| Figueredo,
1999 | 20.176 | 4.482-90.816 | 3.915 | <0.010 | (27) |
| Tsai, 1998 | 66.449 | 23.050-191.561 | 7.768 | <0.010 | (28) |
| Murray, 1995 | 73.833 | 9.272-587.925 | 4.064 | <0.010 | (32) |
| Adonogianaki,
1993 | 174.835 | 41.956-728.564 | 7.091 | <0.010 | (29) |
| Random-effects
model | 49.430 | 19.855-123.053 | 8.382 | <0.010 | |
 | Table VPooled ORs with 95% CIs, Z-values and
P-values for gingival index across included studies under a
random-effects model. |
Table V
Pooled ORs with 95% CIs, Z-values and
P-values for gingival index across included studies under a
random-effects model.
| First author,
year | OR | 95% CI | Z-value | P-value | (Refs.) |
|---|
| Orhue, 2022 | 191.968 | 70.058-526.017 | 10.222 | <0.010 | (12) |
| Ramenzoni,
2021 | 15.496 | 2.555-93.978 | 2.980 | 0.003 | (11) |
| Vozna, 2021 | 35.314 | 13.680-91.159 | 7.366 | <0.010 | (24) |
| Mandra, 2018 | 4.91 | 1.920-12.555 | 3.322 | 0.001 | (30) |
| Talib, 2018 | 1.183 | 0.410-3.415 | 0.310 | 0.756 | (25) |
| Yadav, 2014 | 0.861 | 0.315-2.355 | -0.291 | 0.771 | (26) |
| Glimvall, 2012 | 17.599 | 4.353-71.151 | 4.024 | <0.010 | (9) |
| Shimizu, 2011 | 1.000 | 0.432-2.312 | 0.000 | >0.999 | (33) |
| Wu, 2009 | 3.606 | 0.355-36.602 | 1.085 | 0.278 | (31) |
| Wei, 2004 | 3.391 | 0.7832-15.711 | 1.561 | 0.119 | (34) |
| Figueredo,
2000 | 22.132 | 4.200-116.632 | 3.652 | <0.010 | (35) |
| Figueredo,
1999 | 60.125 | 11.419-316.585 | 4.833 | <0.010 | (27) |
| Tsai, 1998 | 1.894 | 0.797-4.502 | 1.446 | 0.148 | (28) |
| Murray, 1995 | 4.293 | 0.822-22.416 | 1.728 | 0.084 | (32) |
| Adonogianaki,
1993 | 7.978 | 2.695-23.623 | 3.750 | <0.010 | (29) |
| Random-effects
model | 7.173 | 2.914-17.654 | 4.288 | <0.010 | |
Heterogeneity
The heterogeneity analysis across the 15 included
studies revealed substantial variability in outcomes under the
random-effects model. For lactoferrin concentration, the Q-value
was 153.664 (P<0.010) with 14 degrees of freedom and an
I2 of 90.89%, indicating significant heterogeneity
beyond chance. Similarly, PPD, CAL and GI showed Q-values of
103.060, 105.864 and 120.646, respectively (all P<0.010), with
corresponding I2 values of 83.15, 85.83 and 88.40%,
further supporting substantial heterogeneity across studies.
Despite this, the between-study variance (τ2) remained
low for all parameters (lactoferrin τ2=3.617; PPD
τ2=2.060; CAL τ2=2.806; GI
τ2=2.700), suggesting that the pooled effect sizes were
relatively stable across different populations and study
settings.
Forest plots
The forest plots for all analysed parameters, such
as lactoferrin concentration, PPD, CAL and GI, illustrate
individual study result, with the square indicating the OR and the
line representing the 95% CI. In each plot, the pooled effect size
is depicted by a diamond at the bottom, indicating a statistically
significant positive association. In each plot, the pooled effect
size is depicted by a diamond at the bottom, indicating a
statistically significant positive association. For lactoferrin
concentration, the CIs ranged from 1.140-11.180, whereas for PPD,
CAL and GI in relation to elevated lactoferrin in individuals with
periodontitis, the intervals were 17.070-90.340, 19.855-123.053 and
2.914-17.654, respectively (Fig. 2,
Fig. 3, Fig. 4 and Fig.
5). Most individual study outcomes aligned closely with the
pooled estimates, reflecting a generally consistent trend across
studies. Although some studies displayed wider CIs, suggesting
slight variance, the overall pattern supports a low level of
heterogeneity and reinforces the robustness of the findings across
different clinical indicators.
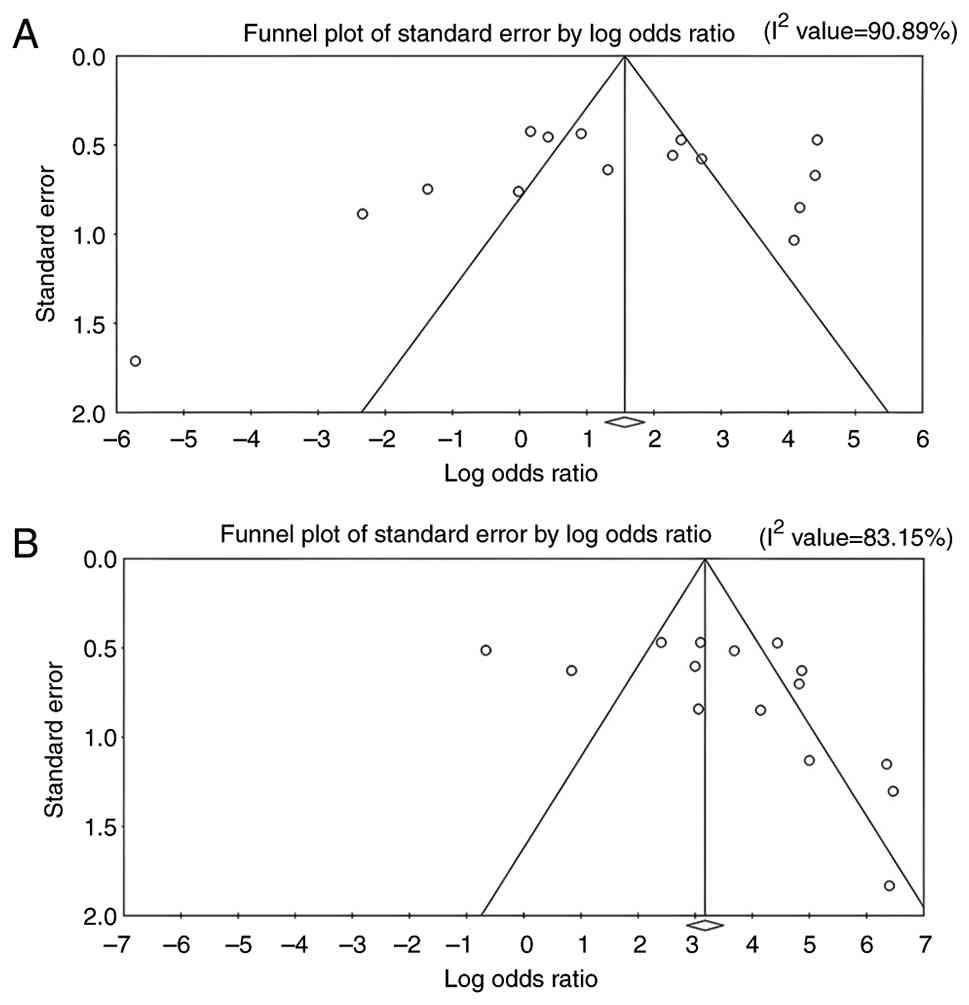
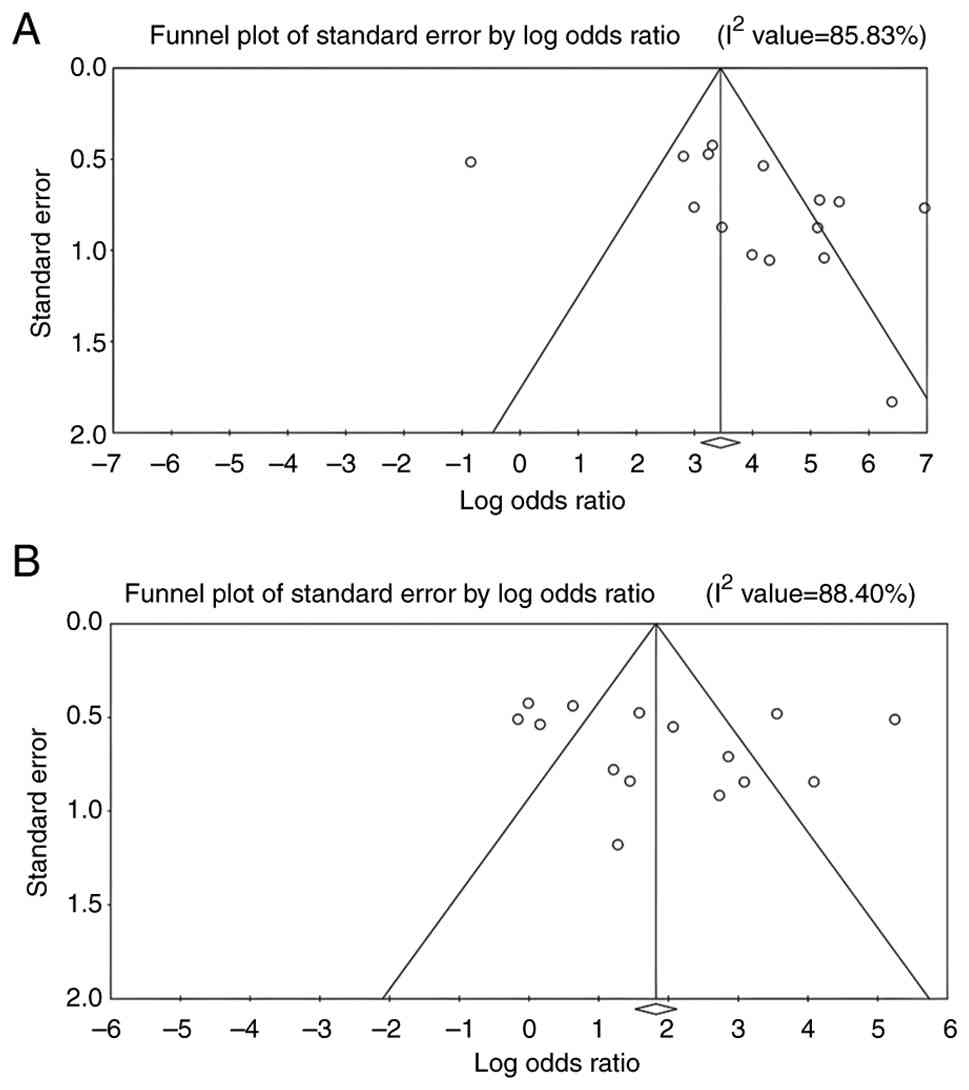
Publication bias. Funnel plots
The funnel plots for all outcome measures, including
lactoferrin concentration, PPD, CAL and GI, appeared visually
symmetrical, indicating a balanced distribution of study effect
sizes around the pooled estimates (Figs.
6 and 7). Although minor
asymmetries were observed in a few studies, these did not suggest
the presence of marked publication bias.
Eggers's regression intercepts. Potential
publication bias across all outcome variables was assessed using
Egger's test. No statistically significant evidence of publication
bias was observed for lactoferrin concentration (T=-0.646;
P=0.529). Likewise, Egger's test did not indicate evidence of
small-study effects for PPD (T=0.335; P=0.743) or GI (T=0.930;
P=0.370). By contrast, Egger's test for CAL yielded a statistically
significant result (T=2.327; P=0.035), suggesting the possible
presence of small-study effects, which may indicate publication
bias or methodological heterogeneity among the included
studies.
Begg's rank correlation test: The Begg's test
results are interpreted solely in the context of publication bias.
Specifically, for lactoferrin concentration, the Kendall's τ-value
of -0.105 with P=0.620 indicated no evidence of publication bias.
Similarly, no statistically significant evidence of publication
bias was observed for PPD (τ=0.230; P=0.230), CAL (τ=0.510;
P=0.610) or GI (τ=0.238; P=0.239). Overall, these findings suggest
that publication bias is unlikely to have materially influenced the
pooled estimates.
Discussion
Periodontitis is a chronic inflammatory condition
with multiple contributing factors, which is mainly triggered by
the build-up of dental plaque, which progressively damages the
periodontium (1). Lactoferrin serves
a pivotal role in safeguarding oral and periodontal health by
serving as a frontline defence protein, exerting antimicrobial
effects, modulating immune responses and reducing inflammation
(8). Lactoferrin influences cytokine
activity by upregulating anti-inflammatory cytokines, including
IL-4 and IL-10, while also modulating the production of
pro-inflammatory cytokines such as TNF-α, IL-1, IL-6 and
granulocyte-macrophage colony-stimulating factor (8,9).
Lactoferrin levels have been shown to be associated with various
periodontal indicators, such as the plaque index, GI, bleeding on
probing, PPD and CAL. Elevated lactoferrin levels in saliva, serum
and GCF have been recognized as potential biomarkers of periodontal
disease activity, emphasizing its utility as both a diagnostic tool
and a target for therapeutic intervention (12,24,27).
The present meta-analysis included 15 studies
comprising saliva, serum and GCF samples from patients with
periodontitis and healthy controls. The pooled analysis revealed
significantly elevated lactoferrin levels in patients with
periodontitis compared with controls, with a SMD of 2.630,
demonstrating a strong positive association with disease presence.
Elevated lactoferrin levels were associated with higher PPD, CAL
and GI across the included studies. Across the 15 included studies,
statistically significant findings (OR>1, P<0.05) were
reported in 10 studies for lactoferrin concentration in individuals
with periodontitis compared with healthy controls. Collectively,
these findings suggest that elevated lactoferrin levels are a
common feature of periodontitis, supporting its role as a marker of
periodontal inflammation. For periodontal clinical parameters,
statistically significant results were observed in 13 studies for
PPD, 14 studies for CAL and 8 studies for GI. In the majority of
included studies, OR>1 with statistically significant P-values
were reported, indicating an overall positive association between
elevated lactoferrin levels and worsening periodontal parameters in
patients with periodontitis. A few studies, including Murray et
al (32), Wu et al
(31), Wei et al (34) and Figueredo and Gustafsson (35), reported neutral or slightly decreased
lactoferrin levels, likely due to small sample sizes, dilution
effects in whole saliva or proteolytic degradation by bacterial
enzymes. Nonetheless, the majority of evidence consistently
indicated elevated lactoferrin in periodontitis, underscoring its
clinical relevance.
Studies by Legrand et al (36) and Glimvall et al (9) have demonstrated that elevated
lactoferrin levels primarily derived from polymorphonuclear
neutrophils are a consistent finding across various inflammatory
conditions, including arthritis, inflammatory bowel disease and
chronic periodontitis, reinforcing its relevance as a key
biomarker. According to Actor et al (37), lactoferrin has a role in modulating
adaptive immunity by attracting sentinel immune cells, driving the
development of T-cell precursors into active T-helper cells and
enhancing the maturation of immature B cells into competent
antigen-presenting cells. Kivadasannavar et al (10) observed a notable decline in
lactoferrin concentrations in GCF following periodontal surgery,
suggesting that measuring lactoferrin levels in GCF could be an
effective approach to monitor treatment progress in
periodontitis.
The present meta-analysis specifically focused on
the role of lactoferrin in periodontitis and its association with
key periodontal indicators, including PPD, CAL and GI. By
consolidating findings from the included studies, the meta-analysis
indicates that elevated lactoferrin levels in saliva, serum and GCF
are generally associated with increased periodontal disease
severity, reflecting its antimicrobial activity through neutrophil
recruitment, enhanced phagocytic bacterial clearance and modulation
of immune responses during inflammation (30). These results underscore the potential
of lactoferrin as a clinically relevant, non-invasive biomarker for
early detection, risk stratification and monitoring of treatment
response in periodontitis. Furthermore, its association with
inflammatory activity highlights its utility in guiding therapeutic
interventions and evaluating treatment efficacy, while future
studies with standardized sampling, sensitive assays and
longitudinal monitoring may support the development of
lactoferrin-based diagnostic and prognostic platforms in routine
clinical practice.
The present meta-analysis additionally revealed that
elevated lactoferrin levels were consistently associated with
increased periodontal indicators, such as PPD, CAL and GI in
patients with periodontitis compared with healthy individuals. For
periodontal clinical parameters, statistically significant results
were observed in 13 studies for PPD, 14 studies for CAL and eight
studies for GI. These studies documented OR>1 and statistically
significant P-values (<0.05), reinforcing a robust association
between increased PPD, CAL and GI, and the severity of periodontal
disease. Additionally, potential variability in diagnostic
criteria, sampling methods or unaccounted confounding factors may
have weakened the observed association in pooled analyses. In
support of the link between lactoferrin and disease severity, Orhue
et al (12) demonstrated
elevated salivary lactoferrin concentration in individuals with
chronic periodontitis compared with healthy subjects, along with a
positive association between lactoferrin and key periodontal
parameters such as bleeding on probing and PPD.
Recent advances in bio-detection technologies can
enhance the diagnostic potential of lactoferrin in periodontal
disease. Nano-enabled platforms, including iron molybdenum
disulfide-assisted lateral flow systems and nanozyme-based
biosensors, offer improved sensitivity, speed and stability for
detecting inflammatory biomarkers (38,39).
Innovations such as ferric yolk-shell nanostructures and label-free
metabolic profiling further increase diagnostic precision across
biological samples (40). Together,
these approaches enable rapid, non-invasive and high-resolution
detection, supporting the development of lactoferrin-based assays
for monitoring periodontal disease activity.
The present meta-analysis has several limitations:
The number of studies examining the association between lactoferrin
and periodontitis was limited, underscoring the need for further
research. Analysis was restricted to saliva, serum and GCF,
excluding dental plaque, which may provide additional insight.
Substantial heterogeneity likely arose from differences in assay
methods, sample collection, diagnostic criteria, study populations,
immune responses and oral hygiene practices. In addition, although
ELISA was widely used, variations in sensitivity and specificity
may have affected results. In addition, Egger's test indicated the
presence of small-study effects for CAL, suggesting that the pooled
estimate may be influenced by publication bias or methodological
heterogeneity among the included studies. Standardizing collection
and assay protocols is essential, and future studies should clarify
the biological role of lactoferrin and its potential as a biomarker
for systemic inflammatory conditions.
In conclusion, the present findings highlight the
notable involvement of lactoferrin in the development and
progression of periodontitis, establishing it as a crucial
modulator of inflammation with considerable therapeutic promise.
The present study demonstrated a positive association between
lactoferrin concentrations and periodontal parameters, including
PPD, CAL and GI. Furthermore, the consistently elevated lactoferrin
levels detected in saliva, blood serum and GCF highlight its
potential as a non-invasive biomarker for diagnosing
periodontitis.
Acknowledgements
Not applicable.
Funding
Funding: No funding was received.
Availability of data and materials
The data generated in the present study may be
requested from the corresponding author.
Authors' contributions
GVP, JM and DP confirm the authenticity of all the
raw data. GVP performed the formal analysis, curated the data,
interpreted the results, drafted the manuscript and revised it
critically. JM contributed to the study conceptualization,
interpretation of findings and supervision. DP contributed to the
conceptualization and methodology, conducted the investigation,
curated and validated the data, interpreted the results, and
drafted and revised the manuscript. RKK validated the data and
analyses, contributed to interpretation and revised the manuscript
critically. SK designed and interpreted the visualizations,
contributed to interpretation and revised the manuscript
critically. ADJ contributed to the investigation and data
acquisition, and to data interpretation. KST contributed to
visualization design and interpretation of the results. BM
validated the data and analyses, and contributed to interpretation.
AN contributed to visualization design and interpretation of
results. All authors read and approved the final manuscript.
Ethics approval and consent to
participate
Not applicable.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
GBD 2019 Diseases and Injuries
Collaborators. Global burden of 369 diseases and injuries in 204
countries and territories, 1990-2019: A systematic analysis for the
global burden of disease study 2019. Lancet. 396:1204–1222.
2020.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Souza JA, Rossa C Jr, Garlet GP, Nogueira
AV and Cirelli JA: Modulation of host cell signaling pathways as a
therapeutic approach in periodontal disease. J Appl Oral Sci.
20:128–138. 2012.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Giannobile WV: Host-response therapeutics
for periodontal diseases. J Periodontol. 79 (8 Suppl):S1592–S1600.
2008.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Califf RM: Biomarker definitions and their
applications. Exp Biol Med (Maywood). 243:213–221. 2018.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Georgiou AC, Crielaard W, Armenis I, de
Vries R and van der Waal SV: Apical periodontitis is associated
with elevated concentrations of inflammatory mediators in
peripheral blood: A systematic review and meta-analysis. J Endod.
45:1279–1295.e3. 2019.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Moreno-Expósito L, Illescas-Montes R,
Melguizo-Rodríguez L, Ruiz C, Ramos-Torrecillas J and de
Luna-Bertos E: Multifunctional capacity and therapeutic potential
of lactoferrin. Life Sci. 195:61–64. 2018.PubMed/NCBI View Article : Google Scholar
|
|
7
|
El-Fakharany EM, Ashry M, Abd-Elaleem AH,
Romeih MH, Morsy FA, Shaban RA and Abdel-Wahhab KG: Therapeutic
efficacy of Nano-formulation of lactoperoxidase and lactoferrin via
promoting immunomodulatory and apoptotic effects. Int J Biol
Macromo. 220:43–55. 2022.PubMed/NCBI View Article : Google Scholar
|
|
8
|
El Amrousy D, El-Afify D, Elsawy A,
Elsheikh M, Donia A and Nassar M: Lactoferrin for iron-deficiency
anemia in children with inflammatory bowel disease: A clinical
trial. Pediatr Res. 92:762–766. 2022.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Glimvall P, Wickström C and Jansson H:
Elevated levels of salivary lactoferrin, a marker for chronic
periodontitis? J Periodontal Res. 47:655–660. 2012.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Kivadasannavar AV, Pudakalakatti PS,
Hattarki SA and Kambali SS: Estimation of lactoferrin levels in
gingival crevicular fluid before and after periodontal therapy in
patients with chronic periodontitis. Contemp Clin Dent. 5:25–30.
2014.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Ramenzoni LL, Hofer D, Solderer A,
Wiedemeier D, Attin T and Schmidlin PR: Origin of MMP-8 and
lactoferrin levels from gingival crevicular fluid, salivary glands
and whole saliva. BMC Oral Health. 21(385)2021.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Orhue VE, Ehizele AO, Akhionbare O and
Ojehanon P: Salivary lactoferrin levels, disease severity and
correlates in patients with chronic periodontitis presenting to a
tertiary health facility in Nigeria. Nig J Dent Res. 7:60–66.
2022.
|
|
13
|
Hu P, Zhao F, Wang J and Zhu W:
Lactoferrin attenuates lipopolysaccharide-stimulated inflammatory
responses and barrier impairment through the modulation of
NF-κB/MAPK/Nrf2 pathways in IPEC-J2 cells. Food Funct.
11:8516–8526. 2020.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Trybek G, Jedliński M, Jaroń A, Preuss O,
Mazur M and Grzywacz A: Impact of lactoferrin on bone regenerative
processes and its possible implementation in oral surgery-a
systematic review of novel studies with metanalysis and
metaregression. BMC Oral Health. 20(232)2020.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Vrabel M: Preferred reporting items for
systematic reviews and meta-analyses. Oncol Nurs Forum. 42:552–554.
2015.
|
|
16
|
Aydin S, Emre E, Ugur K, Aydin MA, Sahin
İ, Cinar V and Akbulut T: An overview of ELISA: A review and update
on best laboratory practices for quantifying peptides and proteins
in biological fluids. J Int Med Res.
53(3000605251315913)2025.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Wells G, Shea B, O'Connell D, Robertson J,
Peterson J, Welch V, Losos M and Tugwell P: The Newcastle-Ottawa
Scale (NOS) for assessing the quality of nonrandomised studies in
meta-analyses. In 3rd Symposium on Systematic Reviews: Beyond the
Basics, Oxford, UK, pp2000, 2014.
|
|
18
|
Jadad AR, Moore RA, Carroll D, Jenkinson
C, Reynolds DJ, Gavaghan DJ and McQuay HJ: Assessing the quality of
reports of randomized clinical trials: Is blinding necessary?
Control Clin Trials. 17:1–12. 1996.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Slim K, Nini E, Forestier D, Kwiatkowski
F, Panis Y and Chipponi J: Methodological index for non-randomized
studies (minors): Development and validation of a new instrument.
ANZ J Surg. 73:712–716. 2003.PubMed/NCBI View Article : Google Scholar
|
|
20
|
Wetterslev J, Thorlund K, Brok J and Gluud
C: Estimating required information size by quantifying diversity in
random-effects model meta-analyses. BMC Med Res Methodol.
9(86)2009.PubMed/NCBI View Article : Google Scholar
|
|
21
|
Andrade C: Mean difference, standardized
mean difference (SMD), and their use in meta-analysis: As simple as
it gets. J Clin Psychiatry. 81(20f13681)2020.PubMed/NCBI View Article : Google Scholar
|
|
22
|
Huedo-Medina TB, Sánchez-Meca J,
Marín-Martínez F and Botella J: Assessing heterogeneity in
meta-analysis: Q statistic or I2 index? Psychol Methods.
11:193–206. 2006.PubMed/NCBI View Article : Google Scholar
|
|
23
|
Lin L and Chu H: Quantifying publication
bias in meta-analysis. Biometrics. 74:785–794. 2018.PubMed/NCBI View Article : Google Scholar
|
|
24
|
Vozna IV, Pavlov SV and Voznyi OV: The
influence of oral antimicrobial peptide content on the quantitative
microflora composition in periodontal pockets among residents of a
large industrial region. Zaporozhye Med J. 23:388–394. 2021.
|
|
25
|
Talib HJ and Ahmed MA: Assessment of
salivary lactoferrin and pH levels and their correlation with
gingivitis and severity of chronic periodontitis (part: 2). J Bagh
Coll Dent. 30:46–52. 2018.
|
|
26
|
Yadav N, Lamba AK, Thakur A, Faraz F,
Tandon S and Pahwa P: Effect of periodontal therapy on lactoferrin
levels in gingival crevicular fluid. Aust Dent J. 59:314–320.
2014.PubMed/NCBI View Article : Google Scholar
|
|
27
|
Figueredo CM, Ribeiro MS, Fischer RG and
Gustafsson A: Increased interleukin-1beta concentration in gingival
crevicular fluid as a characteristic of periodontitis. J
Periodontol. 70:1457–1463. 1999.PubMed/NCBI View Article : Google Scholar
|
|
28
|
Tsai CC, Kao CC and Chen CC: Gingival
crevicular fluid lactoferrin levels in adult periodontitis
patients. Aust Dent J. 43:40–44. 1998.PubMed/NCBI View Article : Google Scholar
|
|
29
|
Adonogianaki E, Moughal NA and Kinane DF:
Lactoferrin in the gingival crevice as a marker of
polymorphonuclear leucocytes in periodontal diseases. J Clin
Periodontol. 20:26–31. 1993.PubMed/NCBI View Article : Google Scholar
|
|
30
|
Mandra EV, Sementsova EA, Polushina LG,
Svetlakova EN, Mandra JV and Bazarnyj VV: Comparance specifics of
lactoferrin in saliva secretion of patients with viral and
bacterial diseases in oral cavity. Materials of the International
Congress ''Dentistry of the Great Urals''. December. 4-6:2019.
|
|
31
|
Wu Y, Shu R, Luo LJ, Ge LH and Xie YF:
Initial comparison of proteomic profiles of whole unstimulated
saliva obtained from generalized aggressive periodontitis patients
and healthy control subjects. J Periodontal Res. 44:636–644.
2009.PubMed/NCBI View Article : Google Scholar
|
|
32
|
Murray MC, Mooney J and Kinane DF: The
relationship between elastase and lactoferrin in healthy,
gingivitis and periodontitis sites. Oral Dis. 1:106–109.
1995.PubMed/NCBI View Article : Google Scholar
|
|
33
|
Shimizu E, Kobayashi T, Wakabayashi H,
Yamauchi K, Iwatsuki K and Yoshie H: Effects of orally administered
lactoferrin and lactoperoxidase-containing tablets on clinical and
bacteriological profiles in chronic periodontitis patients. Int J
Dent. 2011(405139)2011.PubMed/NCBI View Article : Google Scholar
|
|
34
|
Wei PF, Ho KY, Ho YP, Wu YM, Yang YH and
Tsai CC: The investigation of glutathione peroxidase, lactoferrin,
myeloperoxidase and interleukin-1beta in gingival crevicular fluid:
Implications for oxidative stress in human periodontal diseases. J
Periodontal Res. 39:287–293. 2004.PubMed/NCBI View Article : Google Scholar
|
|
35
|
Figueredo CM and Gustafsson A: Increased
amounts of laminin in GCF from untreated patients with
periodontitis. J Clin Periodontol. 27:313–318. 2000.PubMed/NCBI View Article : Google Scholar
|
|
36
|
Legrand D, Elass E, Carpentier M and
Mazurier J: Lactoferrin: A modulator of immune and inflammatory
responses. Cell Mol Life Sci. 62:2549–2559. 2005.PubMed/NCBI View Article : Google Scholar
|
|
37
|
Actor JK, Hwang SA and Kruzel ML:
Lactoferrin as a natural immune modulator. Curr Pharm Des.
15:1956–1973. 2009.PubMed/NCBI View Article : Google Scholar : Qin X, Li D, Qin
X, Chen F, Guo H, Gui Y, Zhao J, Jiang L and Luo D: Electrochemical
detection of the cardiac biomarker cardiac troponin I. View 5:
20240025, 2024.
|
|
38
|
Xu M, Lin C, Zhao S, Zhang W, Li D, Fang
F, Teng Z, Peng Y, Liu M, Huang Z, et al: A versatile catalytic and
photothermal lateral flow immunoassay based on ultrathin
Fe-MoS2 nanosheets for sensitive and accurate detection
of influenza A. View. 5(20240067)2024.
|
|
39
|
Wu J, Wei Y, Shi C, Huang Y, Hong Z, Liu
W, Gu L, Wang R, Li Y, Cao J, et al: Thermally engineered ferric
yolk-shells for vaginal secretion-based cervical cancer screening.
Adv Funct Mater. 34(2312380)2024.
|
|
40
|
Su H, Zhang H, Wu J, Huang L, Zhang M, Xu
W, Cao J, Liu W, Liu N, Jiang H, et al: Fast label-free metabolic
profile recognition identifies phenylketonuria and subtypes. Adv
Sci (Weinh). 11(e2305701)2024.PubMed/NCBI View Article : Google Scholar
|