Introduction
Inflammation is part of the complex biological
response of vascular tissue to harmful stimuli, including
pathogens, damaged cells or irritants (1). This inflammation is associated with
cytokines and pro-inflammatory mediators secreted from macrophages.
An allergic reaction is the result of an inappropriate immune
response triggering inflammation (2). A common example is hay fever, which
is caused by a hypersensitive response by skin mast cells to
allergens (3). During allergic
inflammation, immunoglobulin E (IgE) is produced against allergen
infiltration resulting in activation of mast cells, which releases
histamine, TNF-α, IL-6, IL-8 and NF-κB (4). Histamine is produced by basophils and
by mast cells located in nearby connective tissues (5). Histamine binds to the H1 receptor in
target cells to contract gut and bronchus smooth muscle and to
increase venular permeability and rheum (6). Histamine increases the permeability
of capillaries to white blood cells and various proteins to allow
them to engage pathogens in the infected tissue (6). TNF-α is secreted during the allergic
mechanism by mast cells, macrophages and T cells. TNF-α causes the
expression of adhesion factors to vascular endothelial cells and
accumulates white blood cells resulting in an inflammatory response
(7,8). IL-6 causes a chronic inflammatory
response, activating T cells and producing IgE (9). IL-8 functions as a chemotactic factor
for neutrophils, eosinophils and T lymphocytes activating an
inflammatory response (10). NF-κB
acts as a transcription factor binding to an NF-κB response element
located at the promoter of target genes, regulating TNF-α, IL-6 and
IL-8 (11,12). The secretion of histamine, TNF-α,
IL-6, IL-8 and NF-κB is important in the inflammatory response and
appropriate regulation of these molecules may be useful for the
treatment of inflammatory disease.
Houttuynia cordata Thunb (HCT), a perennial
herb, known as ‘E-Sung-Cho’ in Korea, is widely distributed
throughout Southeast Asia. Antiviral, anticancer, antileukemic,
antioxidant and antiallergic activities of HCT have previously been
reported (13–17). HCT may be beneficial for the
treatment of mast cell-mediated inflammation (18,19).
In addition, HCT extract induces apoptosis via the
mitochondrial-dependent pathway in HT-29 human colon adenocarcinoma
cells (20).
In the present study, the therapeutic effect of HCT
on allergic inflammatory disease was investigated. The levels of
TNF-α, IL-6 and IL-8 in HMC-1 human mast cells activated by PMA and
A23187 under treatment with HCT were measured. HCT inhibited PMA
plus A23187-induced TNF-α and IL-6 secretion and reduced mRNA
levels of TNF-α, IL-6 and IL-8. HCT was observed to block the
nuclear translocation of NF-κB, inhibiting the phosphorylation of
IκBα in stimulated HMC-1 cells. This indicates that HCT inhibits an
allergic inflammatory response via inhibition of the NF-κB
signaling pathway in HMC-1 human mast cells.
Materials and methods
Preparation of HCT ethanol extract
HCT was purchased from Omniherb (Yeongcheon,
Republic of Korea). A 100 g ground powder was extracted twice with
80% v/v ethanol using an ultra-sonicator (Branson, Danbury, CT,
USA) for 30 min at room temperature. The resulting extract was
filtered through a 0.22 μm filter and concentrated to ~100 ml under
reducing pressure. The ethanol extract was evaporated at 40°C
(Eyela, Tokyo, Japan) and freeze-dried for 72 h (Matsushita,
Kadoma, Japan). The powder from the extract was dissolved in DMSO
and stored in aliquots at −80°C until further analysis.
Reagents
Iscove’s modified Dulbecco’s medium (IMDM), fetal
bovine serum (FBS), antibiotic-antimycotic and phosphate-buffered
saline (PBS) were purchased from Gibco-BRL (Carlsbad, CA, USA).
Phorbol 12-myristate 13-acetate (PMA) and A23187 were obtained from
Sigma-Aldrich (St. Louis, MO, USA). The MTS assay kit was purchased
from Promega Corporation (Madison, WI, USA) and the EZ-western
detection kit was obtained from Daeil Lab (Daeil Lab Service Co.,
Seoul, Korea).
Antibodies
Anti-human TNF-α antibody, biotinylated anti-human
TNF-α antibody and human TNF-α recombinant protein were obtained
from R&D Systems (Minneapolis, MN, USA). Anti-human IL-6/IL-8
antibody, biotinylated anti-human IL-6/IL-8 antibody and human
IL-6/IL-8 recombinant protein were purchased from BD Biosciences
(San Jose, CA, USA). Antibodies against NF-κB, p-IκBα and lamin B
were purchased from Santa Cruz Biotechnology, Inc. (Santa Cruz, CA,
USA). α-tubulin antibody was from Sigma-Aldrich.
Cell culture
Human mast cells (HMC-1) were maintained as
monolayer cultures in IMDM supplemented with 10% FBS, 100 U/ml of
penicillin and 100 μg/ml of streptomycin at 37°C in a humidified
incubator under 5% CO2 gas.
MTS assay
HMC-1 cells were seeded at a density of
1×106 cells/well in 24-well plates, pretreated with
various concentrations of HCT (0.05–0.4 mg/ml) for 1 h and
incubated for 24 h in the absence or presence of PMA (25 nM) plus
A23187 (1 μM). Following 24-h incubation, MTS reagents were added
to the culture medium prior to the detection of absorbance at 490
nm. Since the absorbance correlates to the viability of cells, the
number of cells (% of control) was calculated using the formula:
cell number (% of control) = (absorbance of cells treated with CJ
or silibinin - absorbance of blank well)/(absorbance of control
cells - absorbance of blank well) × 100.
Measurement of pro-inflammatory cytokines
by ELISA (enzyme-linked immunosorbent assay)
HMC-1 cells (1×106) were incubated with
various concentrations of HCT (0.05–0.2 mM) for 1 h and treated
with PMA plus A23187 for 4 h. To measure pro-inflammatory
cytokines, 96-well plates were coated with anti-human TNF-α, IL-6
and IL-8 monoclonal antibodies in 0.1 M sodium carbonate buffer (pH
9.5) and then incubated overnight at 4°C. Following washing, the
cells were blocked with 10% FBS in PBS and incubated at room
temperature for 1 h. Following additional washing, samples (culture
supernatants) were incubated for 2 h at 37°C and washed with PBS
containing 0.05% Tween-20 (PBST) and incubated with 0.2 μg/ml
biotinylated anti-human TNF-α, IL-6 and IL-8 antibodies at room
temperature for 1 h. Incubation with streptavidin-horseradish
peroxidase and subsequent treatment with tetramethylbenzidine and
hydrogen peroxide substrate in the dark was performed for 30 min
together with washing and the reaction was terminated using
2NH2SO4. Color development was measured using
a microplate reader at 450 nm. The inhibition percentage of
cytokine production was calculated using the equation: % inhibition
= (A – B) × 100/A, where A and B are the cytokine production
without and with HCT, respectively.
RNA extraction and reverse
transcription-polymerase chain reaction (RT-PCR)
Total cellular RNA was isolated using an easy-BLUE™
RNA extraction kit (Intron Biotechnology, Seoul, Korea) according
to the manufacturer’s instructions. Total RNA (2 μg) was
synthesized to cDNA using M-MLV reverse transcriptase (Invitrogen
Life Technologies, Carlsbad, CA, USA) according to the
manufacturer’s instructions. PCR was conducted in a 20 μl reaction
mixture consisting of cDNA template, 10 pmol each gene-specific
primer, 10X Taq buffer, 2.5 mM dNTP mixture and 1 unit
Taq DNA polymerase (Takara Korea Biomedical Inc, Seoul,
Korea). PCR was performed using the primers: TNF-α,
5′-TGAGCACTGAAAGCATGATCC-3′ and 5′-ATCACTCCAAAGTGCAGCAG-3′; IL-6,
5′-AACC TTTCCAAAGATGGCTGAA-3′ and 5′-CAGGAACTGG ATCAGGACTTT-3′;
IL-8, 5′-TCAGTGCATAAAGAC ATACTCC-3′ and
5′-TGGCATCTTCACTGATTCTTG-3′; and GAPDH 5′-CGTCTTCACCACCATGGAGA-3′
and 5′-CGGCCATCACGCCACAGTTT-3′. The sequencing involved thermal
cycling at 95°C for 1 min (denaturation), 50°C for 1 min
(annealing) and 72°C for 1 min (extension). The products were
checked by agarose electrophoresis and analyzed using the ChemiDoc
imaging system (Bio-Rad, Hercules, CA, USA).
Preparation of cytosolic and nuclear
protein
Cells were incubated in buffer A [10 mM HEPES (pH
7.9), 10 mM KCl, 1.5 mM MgCl2, 0.5 mM dithiothreitol
(DTT) and 0.2 mM phenylmethylsulfonyl fluoride (PMSF)]. The cells
were incubated on ice for 5 min and centrifuged at 4,000 × g for 5
min. The pellet was then lysed with buffer B [10 mM HEPES (pH 7.9),
10 mM KCl, 1.5 mM MgCl2, 0.1% NP-40, 0.5 mM DTT and 0.2
mM PMSF] and centrifuged at 5,000 rpm for 5 min at 4°C. The
cytoplasmic proteins were extracted from the supernatant and the
pellet was resuspended in buffer C [20 mM HEPES (pH 7.9), 420 mM
NaCl, 1.5 mM MgCl2, 25% glycerol, 0.2 mM EDTA, 0.5 mM
DTT and 0.2 mM PMSF], incubated on ice for 30 min and then
centrifuged at 5,000 rpm for 10 min at 4°C. Nuclear proteins were
obtained from the supernatant.
Western blot analysis
An equal amount of protein in total cell extracts
was separated by SDS-PAGE. Following electrophoresis, the proteins
were transferred to a nitrocellulose membrane (Schleicher &
Schuell Bioscience, Dassel, Germany). The membrane was blocked,
incubated overnight at 4°C with primary antibodies (anti-NFκB,
anti-p-IκBα, anti-lamin B and anti-α-tubulin), washed with PBST
(0.1% PBS) and incubated with appropriate HRP-conjugated secondary
antibodies at room temperature for 1 h. Immunoreactive protein was
developed using an EZ-western detection kit (Daeillab Service Co.,
Seoul, South Korea).
Statistical analysis
Data are presented as the mean ± SD. A Student’s
t-test was used for single variable comparisons. P<0.05 was
considered to indicate a statistically significant difference.
Results
Effect of HCT on cell viability in
activated mast cells
Mast cells are activated by PMA and A23187 and
secrete inflammatory mediators, including histamine, serotonin,
hydrolase, heparin and prostaglandin. In the current study, the
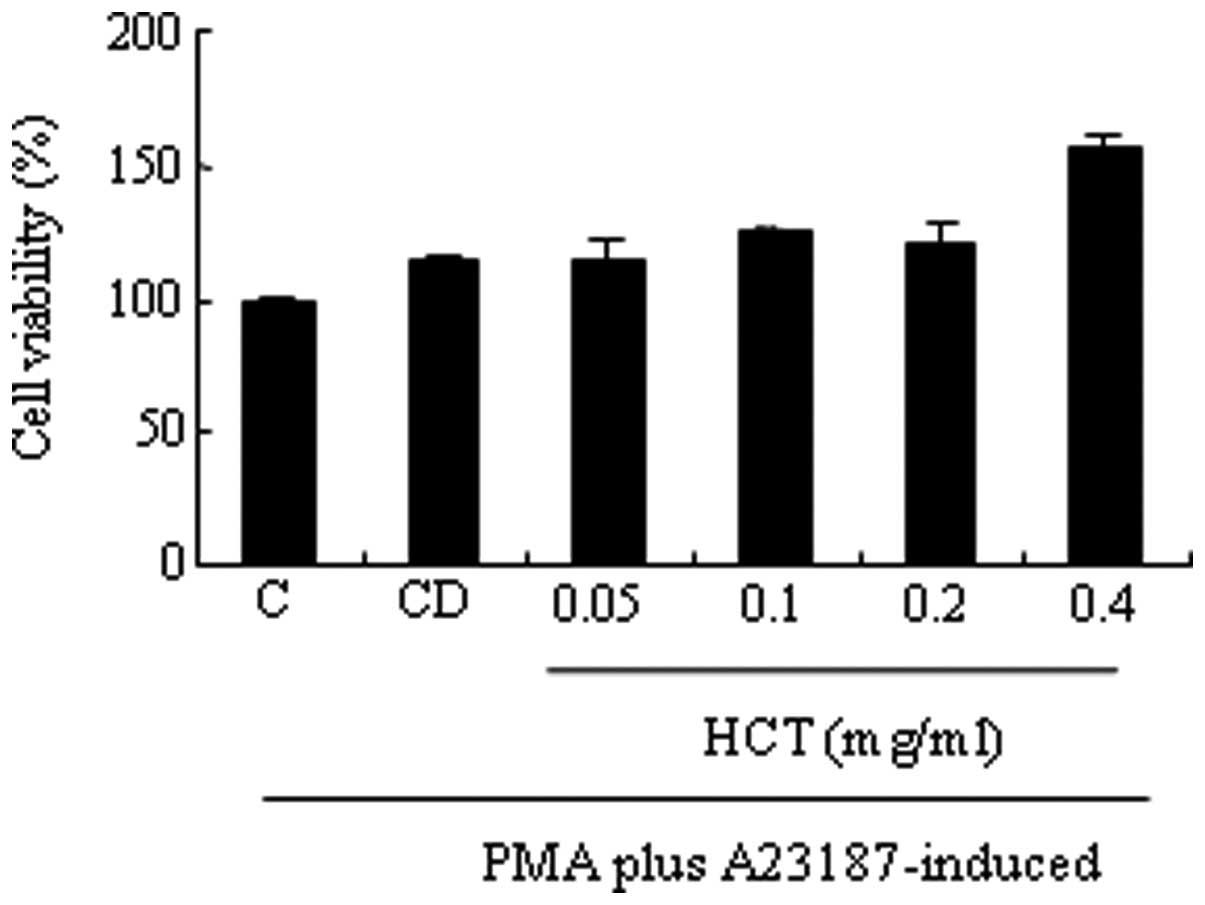
effect of HCT on cell viability in HMC-1 cells was investigated. It
was observed that HCT did not affect cell viability in HMC-1 cells
(Fig. 1). HCT failed to decrease
histamine release induced by PMA plus A23187 (data not shown).
Effect of HCT on PMA plus
A23187-stimulated TNF-α expression
Pro-inflammatory cytokines are important factors of
allergic inflammation. Therefore, the production and expression of
TNF-α was determined by ELISA or RT-PCR to evaluate the effect of
HCT on the pro-inflammatory cytokines. It was observed that HCT
significantly decreased TNF-α production induced by PMA plus A23187
(Fig. 2A). In addition, mRNA
levels of TNF-α induced by PMA plus A23187 was reduced by HCT
treatment (Fig. 2B).
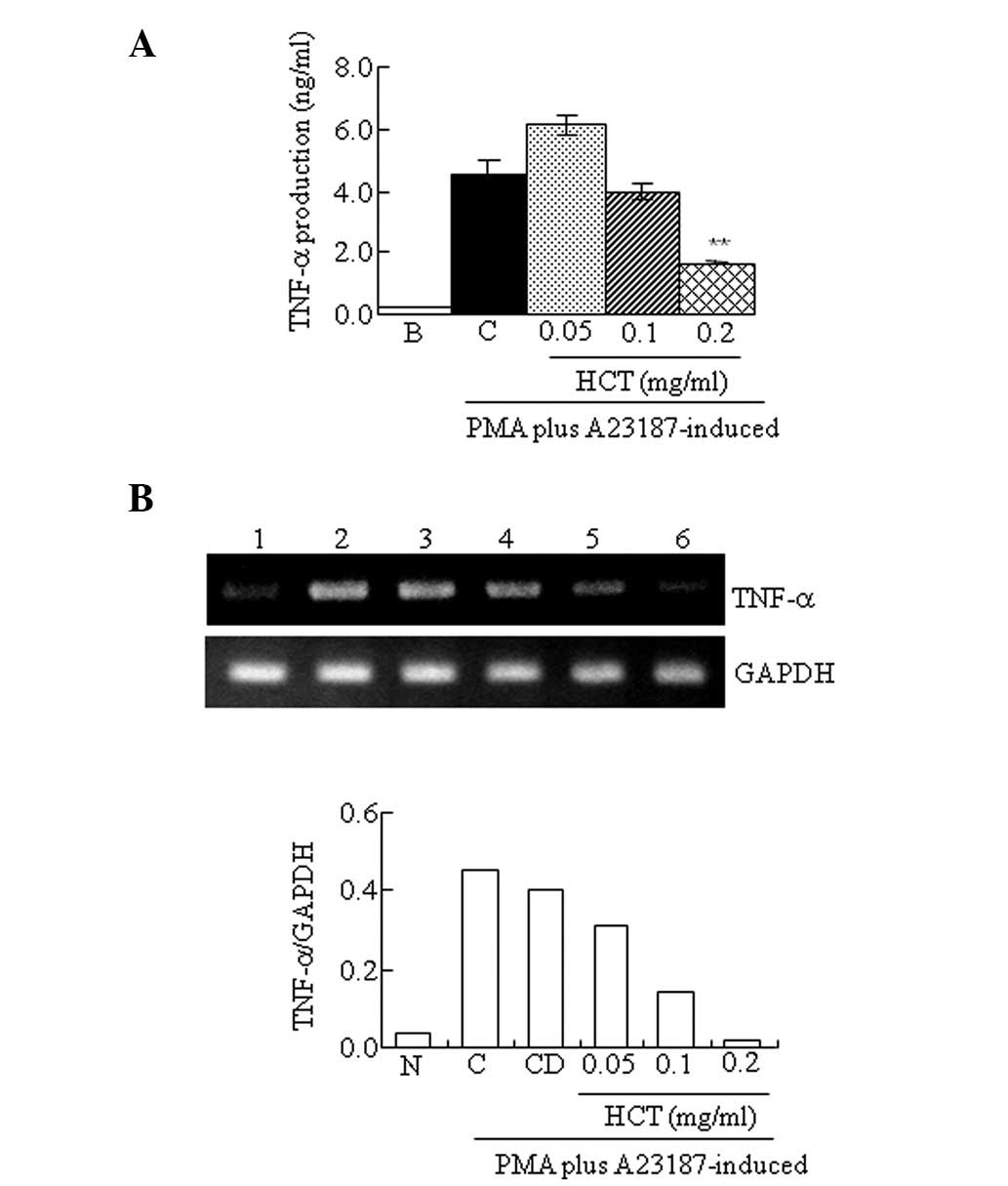 | Figure 2Effect of HCT on PMA plus
A23187-stimulated TNF-α expression. HMC-1 cells (1×106
cells/ml) were pre-incubated with various concentrations of HCT
(0.05–0.2 mg/ml) for 1 h and treated with PMA plus A23187 for 4 h.
(A) TNF-α secreted protein levels in the supernatant were measured
by ELISA assay. (B) TNF-α mRNA levels were measured by RT-PCR.
Lanes 1, normal cells; 2, control cells; 3, DMSO control cells; 4,
HCT (0.05 mg/ml) + PMA plus A23187; 5, HCT (0.1 mg/ml) + PMA plus
A23187; 6, HCT (0.2 mg/ml) + PMA plus A23187. Data are presented as
the mean ± SD of three independent experiments
(*P<0.05 and **P<0.01, vs. control). N,
no treatment; C, induced control; CD, induced control with DMSO
treatment; HCT, Houttuynia cordata Thunb; ELISA,
enzyme-linked immunosorbent assay; RT-PCR, reverse transcription
polymerase chain reaction. |
Effect of HCT on PMA plus
A23187-stimulated IL-6 expression
The production and expression of IL-6 by ELISA or
RT-PCR was determined. HCT significantly decreased IL-6 production
induced by PMA plus A23187 (Fig.
3A). In addition, IL-6 mRNA levels induced by PMA plus A23187
were also reduced by HCT treatment (Fig. 3B).
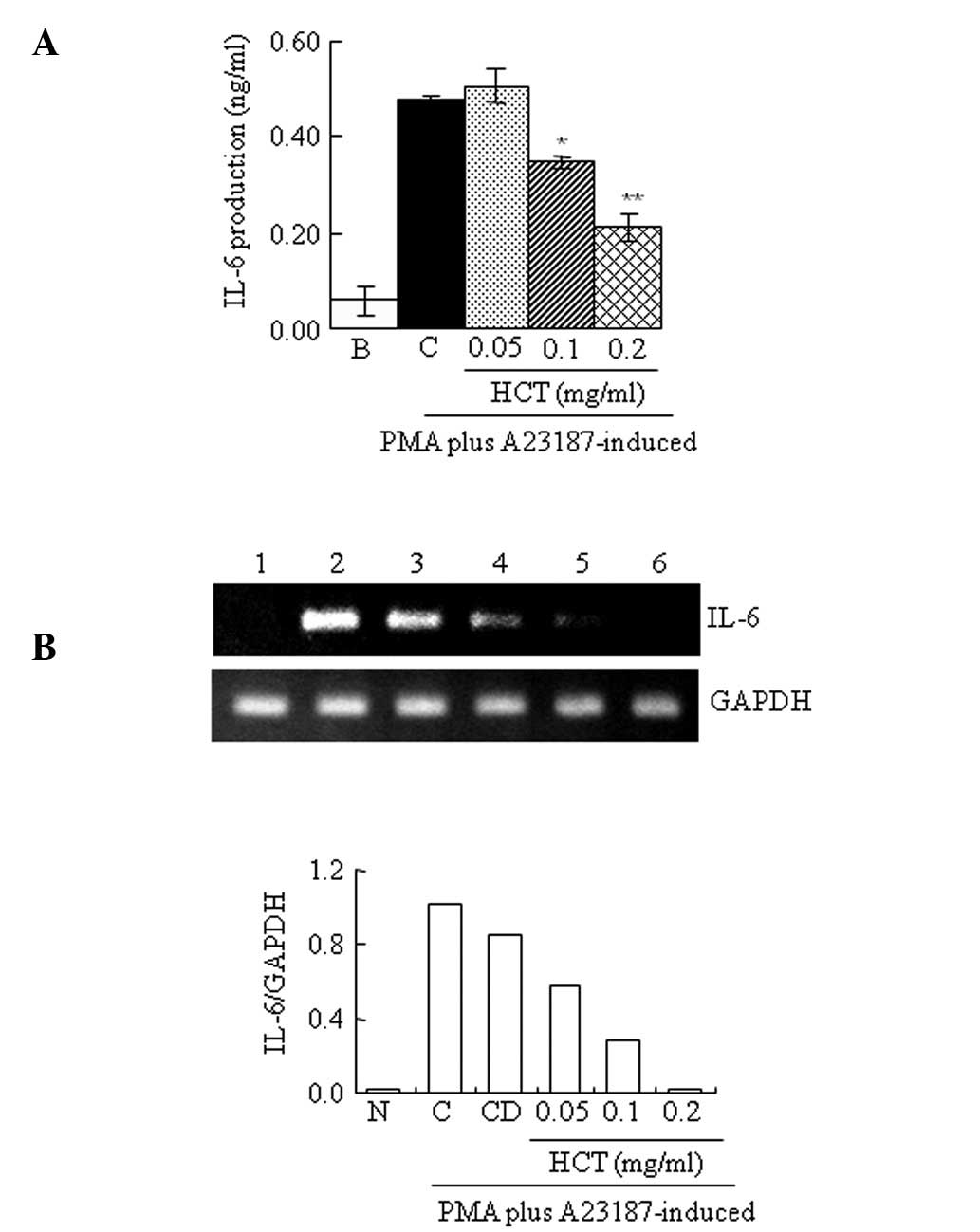 | Figure 3Effect of HCT on PMA plus
A23187-stimulated IL-6 expression. HMC-1 cells (1×106
cells/ml) were preincubated with various concentrations of HCT
(0.05–0.2 mg/ml) for 1 h and treated with PMA plus A23187 for 4 h
(A and B). (A) IL-6 secreted protein levels in the supernatant were
measured by ELISA assay. (B) IL-6 mRNA levels were measured by
RT-PCR. Lanes 1, normal cells; 2, control cells; 3, DMSO control
cells; 4, HCT (0.05 mg/ml) + PMA plus A23187; 5. HCT (0.1 mg/ml) +
PMA plus A23187; 6, HCT (0.2 mg/ml) + PMA plus A23187. Data are
presented as the mean ± SD of three independent experiments
(*P<0.05 and **P<0.01 vs. control). N,
no treatment; C, induced control; CD, induced control with DMSO
treatment; HCT, Houttuynia cordata Thunb; ELISA,
enzyme-linked immunosorbent assay; RT-PCR, reverse transcription
polymerase chain reaction. |
Effect of HCT on PMA plus
A23187-stimulated IL-8 expression
The production and expression of IL-8 was measured
by ELISA and RT-PCR. HCT was not found to significantly decrease
the production levels of IL-8 induced by PMA plus A23187 (Fig. 4A). However, mRNA levels of IL-8
induced by PMA plus A23187 were reduced by HCT (Fig. 4B).
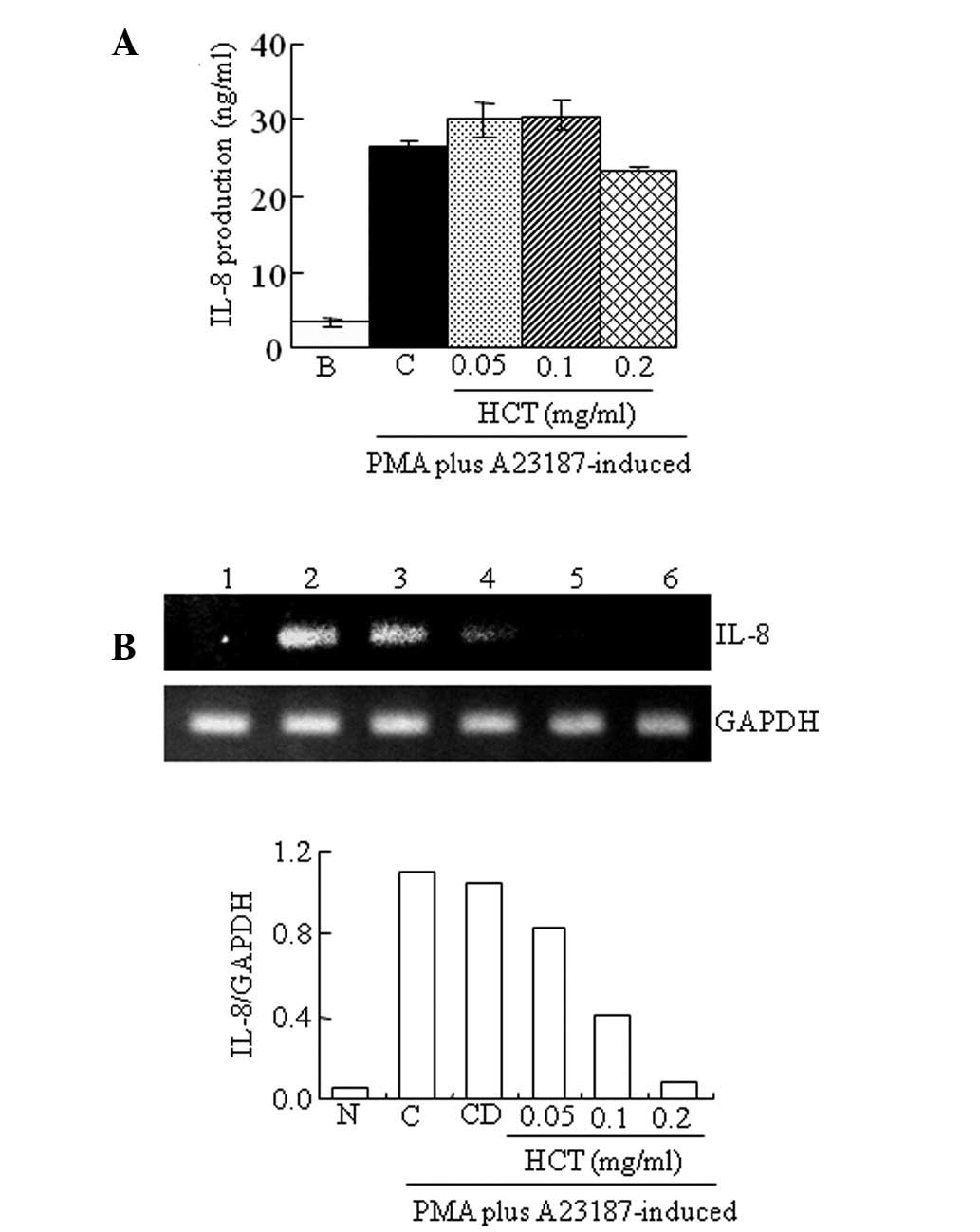 | Figure 4Effect of HCT on PMA plus
A23187-stimulated IL-8 expression. HMC-1 cells (1×106
cells/ml) were preincubated with various concentrations of HCT
(0.05–0.2 mg/ml) for 1 h and treated with PMA plus A23187 for 4 h
(A and B). (A) IL-8 secreted protein levels in the supernatant were
measured by ELISA assay. (B) IL-8 mRNA levels were measured by
RT-PCR. Lanes 1, normal cells; 2, control cells; 3, DMSO control
cells; 4, HCT (0.05 mg/ml) + PMA plus A23187; 5, HCT (0.1 mg/ml) +
PMA plus A23187; 6, HCT (0.2 mg/ml) + PMA plus A23187. Data are
presented as the mean ± SD of three independent experiments. N, no
treatment; C, induced control; CD, induced control with DMSO
treatment; HCT, Houttuynia cordata Thunb ELISA,
enzyme-linked immunosorbent assay; RT-PCR, reverse transcription
polymerase chain reaction. |
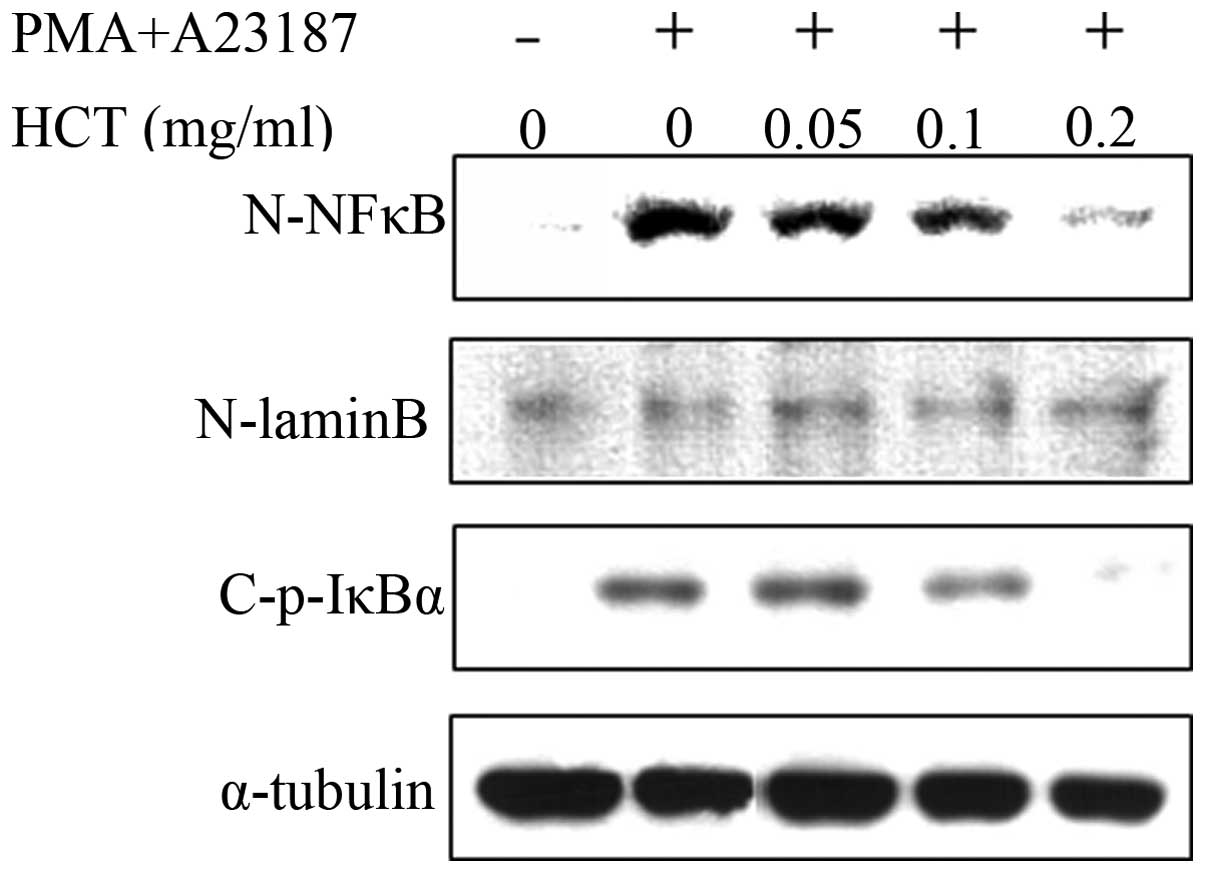
Effect of HCT on PMA plus
A23187-stimulated NF-κB activation and IκBα phosphorylation
Expression of pro-inflammatory cytokines, including
TNF-α, IL-6 and IL-8, is regulated by NF-κB signaling. The effect
of HCT on the expression of NF-κB signaling molecules was analyzed
and HCT was found to suppress the expression of nuclear NF-κB
induced by PMA plus A23187 (Fig.
5). In addition, HCT inhibited the phosphorylation of IκBα in
stimulated HMC-1 cells indicating that HCT abrogates the
dissociation of IκBα from the NF-κB heterodimer (p65 and p50) to
suppress NF-κB signaling (Fig.
5).
Discussion
Mast cells are major immune cells involved in
allergies with versatile physiological functions and are important
for allergic activities in response to the stimuli-induced release
of histamine (21–23). In addition, the inhibition of
TNF-α, IL-6, IL-8 and NF-κB activation has been shown as an
indicator of anti-inflammatory events in mast cells (21,24).
Although several traditional herbal medicines have been shown to
have inhibitory effects on allergy or inflammation using mast cells
(25,26), there is little information with
regard to the effects of HCT on mast cell-derived allergic
inflammation or the molecular mechanisms involved. Therefore, in
the present study, we investigated whether HCT has a therapeutic
effect on allergic inflammatory disease. Since PMA alone fails to
induce the degranulation of mast cells, HMC-1 cells were stimulated
by PMA and A23187. Notably, HCT inhibited PMA plus A23187-induced
TNF-α and IL-6 secretion and reduced mRNA levels of TNF-α, IL-6 and
IL-8. In agreement with the current observations, it was previously
reported that HCT inhibits the secretion of TNF-α in the activated
macrophage-like cell line, RAW 264.7 (27).
Allergic inflammation is classified into early-phase
(or type I immediate hypersensitivity) and late-phase reactions,
which result in subsequent chronic allergic inflammation. Release
of histamine and other mediators following the crosslinking of the
Fc receptor subsequent to the binding of IgE (FcɛRI) to allergen in
mast cells is known as early-phase reaction. The IgE-mediated PCA
reaction is a sensitive reaction for the detection of small
quantities of antibodies and has been used to evaluate the
mechanisms of immediate allergy reaction (28). These early-phase responses are
followed by a late-phase reaction that typically develops 2–9 h
following allergen exposure. In late-phase reactions, the
recruitment of leucocytes, including T-cells and neutrophils, is
featured (21,29,30).
The transition to the late-phase reaction is characterized as the
recruitment of leukocytes by upregulating mediators, including IL-8
and TNF-α (21,30). Mast cell-derived IL-8 is
hypothesized to activate neurophils in allergic inflammation
(31). In a previous study, it was
reported that monomeric IgE induces long-lasting IL-8 synthesis in
mast cells (32). It was also
reported that TNF-α is crucial in the development of late-phase
anaphylactic reactions via the PAF-mediated NF-κB-dependent pathway
and initiates late-phase allergic inflammation (33). Therefore, the observation of no
significant change in histamine release, but significant
suppression of inflammatory cytokines, indicates that HCT may have
inhibitory effects on allergic inflammation through transition or
late-phase reaction.
NF-κB plays a key role in the cellular stress
response and in inflammation by controlling the expression of a
network of genes, including TNF-α, IL-6 and IL-8. Following
infection, microbial pathogens are sensed by the host and activate
NF-κB transcription factors via triggering of various sensors,
including the toll-like receptors, which are expressed on cells of
the innate immune system, macrophages, dendritic cells and mucosal
epithelial cells (34,35). NF-κB activation is closely
controlled by a pathway that regulates the proteolysis of the
inhibitory IκB and IκB-associated proteins. In unstimulated cells,
NF-κB dimers are sequestered in the cytoplasm via physical
association with NF-κB inhibitory proteins, IκBs (34). Upon stimulation, signal
transduction events rapidly lead to the activation of the IκB
kinase (IKK) complex, composed of two catalytic subunits (IKKα and
IKKβ) and a regulatory subunit, NF-κB essential modulator (34). Activated IKK phosphorylates IκBα,
predominantly via the action of IKKβ, triggering its
polyubiquitination and proteasomal degradation and inducing the
nuclear translocation of associated NF-κB subunits (34). NF-κB subunits bind to specific DNA
to induce the transcription of target genes. The current
observations indicate that HCT suppresses the nuclear translocation
of NF-κB as well as the phosphorylation of IκBα, which may inhibit
the expression of proinflammatory cytokines, including TNF-α, IL-6
and IL-8.
Results of the present study suggest that HCT
inhibits the production of pro-inflammatory cytokines via
inhibition of the NF-κB signaling pathway in HMC-1 human mast
cells. We hypothesized that HCT may be a potential therapeutic
target for the treatment of allergies and inflammatory
diseases.
Acknowledgements
This study was supported by a grant of the
Traditional Korean Medicine R&D Project, Ministry of Health
& Welfare, Republic of Korea (no. B110043). This research was
also supported by Basic Science Research Program through the
National Research Foundation of Korea (NRF) funded by the Ministry
of Education, Science and Technology (no. 2012R1A1A3004797).
References
|
1
|
Ferrero-Miliani L, Nielsen OH, Andersen PS
and Girardin SE: Chronic inflammation: importance of NOD2 and NALP3
in interleukin-1beta generation. Clin Exp Immunol. 147:227–235.
2007.PubMed/NCBI
|
|
2
|
Rosenwasser LJ: Current understanding of
the pathophysiology of allergic rhinitis. Immunol Allergy Clin
North Am. 31:433–439. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Rose S, Weld-Moore R, Ghazali N and Newman
L: ‘I’ve got hay-fever and my mouth is stinging!’. Br Dent J.
211:369–370. 2011.
|
|
4
|
Olivera A and Rivera J: An emerging role
for the lipid mediator sphingosine-1-phosphate in mast cell
effector function and allergic disease. Adv Exp Med Biol.
716:123–142. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Kuna P, Reddigari SR, Rucinski D,
Oppenheim JJ and Kaplan AP: Monocyte chemotactic and activating
factor is a potent histamine-releasing factor for human basophils.
J Exp Med. 175:489–493. 1992. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Wang SL, Malany S, Wang Q, Santos MA,
Crowe PD and Maki RA: Histamine induces interleukin-6 expression in
the human synovial sarcoma cell line (SW982) through the H1
receptor. Inflamm Res. 55:393–398. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Thomas PS: Tumor necrosis factor-alpha:
the role of this multifunctional cytokine in asthma. Immunol Cell
Biol. 79:132–140. 2001. View Article : Google Scholar
|
|
8
|
Nakae S, Suto H, Kakurai M, Sedgwick JD,
Tsai M and Galli SJ: Mast cells enhance T cell activation:
Importance of mast cell-derived TNF. Proc Natl Acad Sci USA.
102:6467–6472. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Bodreau RT, Hoskin DW and Lin TJ:
Phosphatase inhibition potentiates IL-6 production by mast cells in
response to FcepsilonRI-mediated activation: involvement of p38
MAPK. J Leukoc Biol. 76:1075–1081. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Murayama T, Mukaida N, Sadanari H,
Yamaguchi N, Khabar KS, Tanaka J, Matsushima K, Mori S and Eizuru
Y: The immediate early gene 1 product of human cytomegalovirus is
sufficient for up-regulation of interleukin-8 gene expression.
Biochem Biophys Res Commun. 279:298–304. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Kuprsh DV, Udalova IA, Turetskaya RL, Rice
NR and Nedospasov SA: Conserved kappa B element located downstream
of the tumor necrosis factor alpha gene: distinct NF-kappa B
binding pattern and enhancer activity in LPS activated murine
macrophages. Oncogene. 11:97–106. 1995.
|
|
12
|
Galien R, Evans HF and Garcia T:
Involvement of CCAAT/enhancer-binding protein and nuclear
factor-kappa B binding site in interleukin-6 promoter inhibition by
estrogens. Mol Endocrinol. 10:713–722. 1996.PubMed/NCBI
|
|
13
|
Chang JS, Chiang LC, Chen CC, Liu LT, Wang
KC and Lin CC: Antileukemic activity of Bidens pilosa L.
var. minor(Blume) Sherff and Houttuynia cordata
Thunb. Am J Chin Med. 29:303–312. 2001.
|
|
14
|
Chiang LC, Chang JS, Chen CC, Ng LT and
Lin CC: Anti-herpes simplex virus activity of Bidens pilosa
and Houttuynia cordata. Am J Chin Med. 31:355–362. 2003.
View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Li GZ, Chai OH, Lee MS, Han EH, Kim HT and
Song CH: Inhibitory effects of Houttuynia cordata water
extracts on anaphylactic reaction and mast cell activation. Biol
Pharm Bull. 28:1864–1868. 2005.
|
|
16
|
Lu HM, Liang YZ, Yi LZ and Wu XJ:
Anti-inflammatory effect of Houttuynia cordata injection. J
Ethnopharmacol. 104:245–249. 2006. View Article : Google Scholar
|
|
17
|
Chen YY, Liu JF, Chen CM, Chao PY and
Chang TJ: A study of the antioxidative and antimutagenic effects of
Houttuynia cordata Thunb. using an oxidized frying oil-fed
model. J Nutr Sci Vitaminol (Tokyo). 49:327–333. 2003.PubMed/NCBI
|
|
18
|
Kim IS, Kim JH, Kim JS, Yun CY, Kim DH and
Lee JS: The inhibitory effect of Houttuynia cordata extract
on stem cell factor-induced HMC-1 cell migration. J Ethnopharmacol.
112:90–95. 2007.
|
|
19
|
Lee JS, Kim IS, Kim JH, Kim JS, Kim DH and
Yun CY: Suppressive effects of Houttuynia cordata Thunb
(Saururaceae) extract on Th2 immune response. J Ethnopharmacol.
117:34–40. 2008.
|
|
20
|
Tang YJ, Yang JS, Lin CF, Shyu WC, Tsuzuki
M, Lu CC, Chen YF and Lai KC: Houttuynia cordata Thunb
extract induces apoptosis through mitochondrial-dependent pathway
in HT-29 human colon adenocarcinoma cells. Oncol Rep. 22:1051–1056.
2009.
|
|
21
|
Bischoff SC: Role of mast cells in
allergic and non-allergic immune responses: comparison of human and
murine data. Nat Rev Immunol. 7:93–104. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Mekori YA and Metcalfe DD: Mast cells in
innate immunity. Immunol Rev. 173:131–140. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Prussin C and Metcalfe DD: 4. IgE, mast
cells, basophils and eosinophils. J Allergy Clin Immunol. 111(2
Suppl): S486–S494. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Kim SJ, Lee EJ, Song YS, Jeong HJ, Lee KM,
Kim HR, Chae HJ, Shin TY, Kim YK, Hong SH and Kim HM:
Cheongyeolsaseuptang inhibits production of TNF-alpha, IL-6 and
IL-8 as well as NF-kappa B activation in human mast cells. J
Ethnopharmacol. 97:83–88. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Cha JY, Jung JY, Jung JY, Lee JR, Cho IJ,
Ku SK, Byun SH, Ahn YT, Lee CW, Kim SC and An WG: Inhibitory
effects of traditional herbal formula pyungwi-san on inflammatory
response in vitro and in vivo. Evid Based Complement Alternat Med.
2013:6301982013.PubMed/NCBI
|
|
26
|
Kim SR, Choi HS, Seo HS, Choi YK, Shin YC
and Ko SG: Topical application of herbal mixture extract inhibits
ovalbumin- or 2,4-dinitrochlorobenzene-induced atopic dermatitis.
Evid Based Complement Alternat Med. 2012:5454972012.PubMed/NCBI
|
|
27
|
Park E, Kum S, Wang C, Park SY, Kim BS and
Schuller-Levis G: Anti-inflammatory activity of herbal medicines:
inhibition of nitric oxide production and tumor necrosis
factor-alpha secretion in an activated macrophage-like cell line.
Am J Chin Med. 33:415–424. 2005. View Article : Google Scholar
|
|
28
|
Harada M, Nagata M and Takeuchi M:
Production of passive cutaneous anaphylaxis (PCA) and reversed PCA
by rat IgE antibody in the mouse. Experientia. 44:459–462. 1988.
View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Valenta R: The future of antigen-specific
immunotherapy of allergy. Nat Rev Immunol. 2:446–453.
2002.PubMed/NCBI
|
|
30
|
Galli SJ, Tsai M and Piliponsky AM: The
development of allergic inflammation. Nature. 454:445–454. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Lippert U, Möller A, Welker P, Artuc M and
Henz BM: Inhibition of cytokine secretion from human leukemic mast
cells and basophils by H1- and H2-receptor antagonists. Exp
Dermatol. 9:118–124. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Cruse G, Kaur D, Yang W, Duffy SM,
Brightling CE and Bradding P: Activation of human lung mast cells
by monomeric immunoglobulin E. Eur Respir J. 25:858–863. 2005.
View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Choi IW, Kim YS, Kim DK, Choi JH, Seo KH,
Im SY, Kwon KS, Lee MS, Ha TY and Lee HK: Platelet-activating
factor-mediated NF-kappaB dependency of a late anaphylactic
reaction. J Exp Med. 198:145–151. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Mohamed MR and McFadden G: NFkB
inhibitors: strategies from poxviruses. Cell Cycle. 8:3125–3132.
2009. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Medzhitov R: Toll-like receptors and
innate immunity. Nat Rev Immunol. 1:135–145. 2001. View Article : Google Scholar : PubMed/NCBI
|