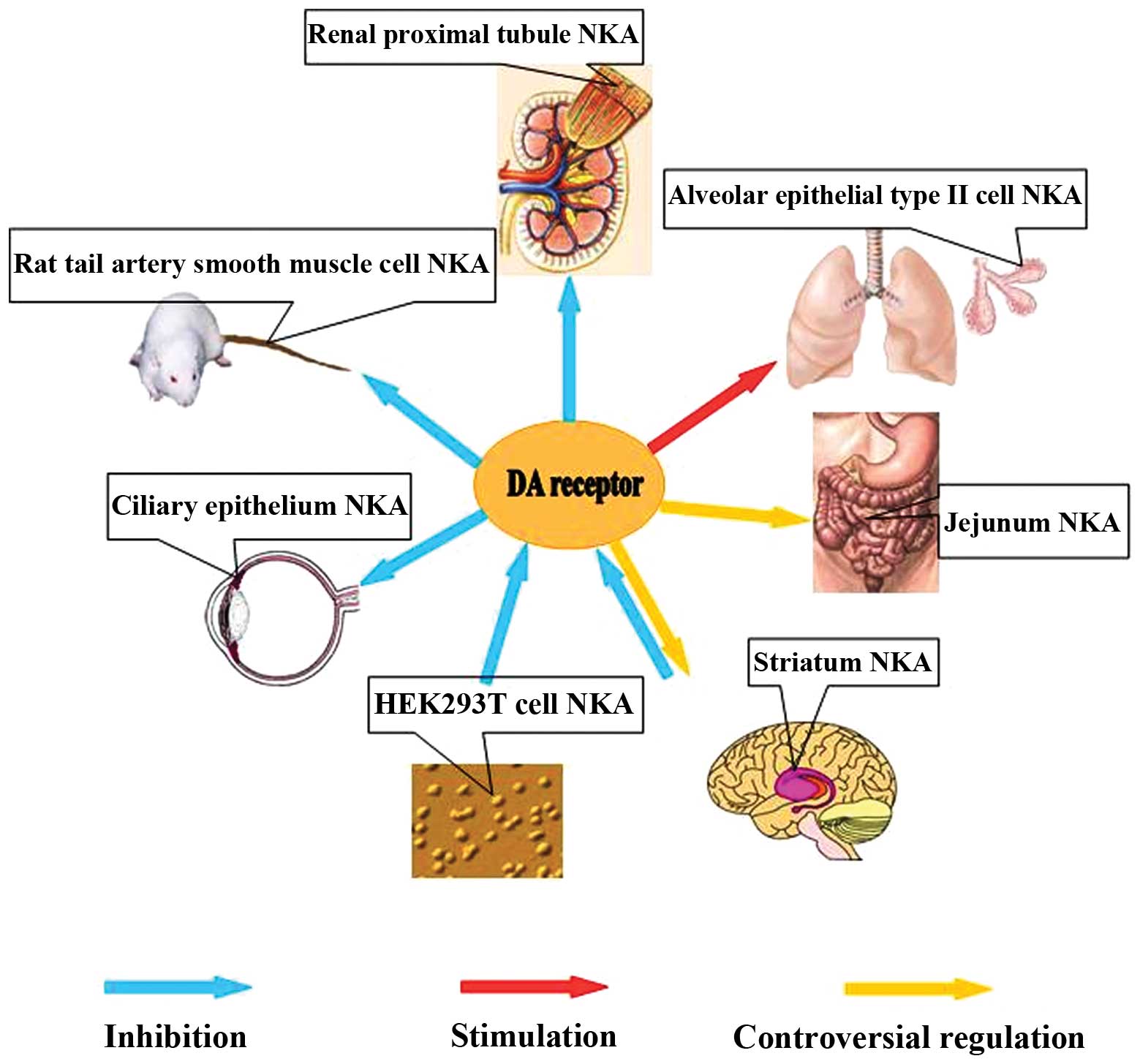
1. Introduction
Over a number of decades, it has become evident that
dopamine (DA) is important in the regulation of blood pressure and
sodium balance by direct actions on renal, lung and intestinal
epithelial, brain, non-pigmented ciliary epithelium and vascular
bed ion transport, in which the regulation of
Na+/K+-ATPase (NKA) exerts a critical role.
There are five subtypes of DA receptors, D1,
D2, D3, D4 and D5. The
D1 and D5 receptors are members of the
D1-like family of DA receptors, whereas the
D2, D3 and D4 receptors are
members of the D2-like family. There is specific
evidence indicating the existence of possible D6 and
D7 receptors, however, such receptors have not yet been
conclusively identified (1).
NKA, or the Na+/K+ pump, is an
energy transducing ion pump first described by Skou in 1957
(2), which uses ATP to transport
Na+ out of cells and K+ into cells. The
resulting gradient drives numerous processes, for example transport
of glucose into intestinal and renal epithelial cells through a
glucose-sodium cotransporter (3),
as well as the transport of other nutrients, including amino acids
(4) and ions, for example
Ca2+(5). This enzyme is
also responsible for generating the resting potential of cells,
which is particularly important in neuronal and muscle function. It
consists of an α subunit (6),
which is responsible for its catalytic and pharmacological
properties, as well as β and γ subunits, which may have regulatory
functions (7,8). The α subunit has four isoforms,
α1, α2, α3 and α4,
while the β subunit has three isoforms, β1,
β2 and β3. The γ subunit is a hydrophobic
protein of ~10 kDa. The α1 isoform is expressed
ubiquitously and plays the main ‘housekeeping’ role; α2
is expressed primarily in skeletal muscle, as well as in the brain
and heart; α3 is expressed in the brain and heart; and
α4 is expressed in testis and skeletal muscle (9). It is reasonable to hypothesize that
the tissue-specific distribution of the α isoforms indicates that
each isoform subtype exhibits a particular function associated with
the tissue in which it is expressed. Animals lacking expression of
the α1 isoform die during embryogenesis; specifically,
embryos fail to develop beyond the blastocyst stage (10). This is an expected result as the
α1 isoform is ubiquitously expressed and is required for
multiple biological functions. Animals lacking the α2
isoform gene are born but die immediately following birth (11). A subsequent study (12) demonstrated that
α2-isoform knockout mice have a defect in the breathing
center of the brain, causing failure to breathe and, thereby, death
from asphyxia. The α1 and α2 isoforms
regulate cardiovascular function. For example, studies indicate
that the α2 isoform of NKA mediates an ouabain-induced
increase in vascular and cardiac contractility, which plays a role
in the development and maintenance of hypertension (13) and cardiac inotropy in mice
(14). Other studies have shown
that the α1 isoform may also regulate cardiac
contractility and functionally and physically couple with the Na/Ca
exchanger in the heart (15).
Ouabain protects the heart against ischemia-reperfusion injury,
which is likely due to activation of the NKA/c-Src receptor complex
and subsequent stimulation of key mediators of preconditioning,
such as phosphatidylinositol (PI)-phospholipase C
(PLC)-γ1 and protein kinase (PK) C-ɛ (16). The effect of NKA on increased
cardiovascular risk, including ischemic heart failure, is likely to
provide insight into the identification of new medicines. An
example is the novel agent, istaroxime, which has inotropic
(inhibition of NKA) and lusitropic (stimulation of sarcoplasmic
reticulum Ca2+-ATPase activity) effects in animal models
of acute heart failure syndromes (17). Previous studies are consistent with
the hypothesis that NKA activity significantly increases
accompanied by an increased surface expression of the α1
and α2 isoforms of NKA, which protects neurons from
subsequent ischemia 24-h following the preconditioning treatment
(18). Therefore, NKA regulation
becomes a crucial step in exploring subsequent responses in various
tissues.
Multiple factors acting through signaling pathways,
including protein kinases (cAMP/PKA, PKC and cGMP/PKG),
Ca2+/calmodulin and reactive oxygen species, modulate
the activity of NKA (19). DA
receptor-mediated NKA regulation is of fundamental and clinical
value and functions through the stimulation of multiple signaling
pathways, including PLC/PKC, cAMP/PKA, PI3K, adaptor protein 2
(AP-2) and tyrosine phosphatase, resulting in diverse responses,
for example the natriuretic effect, increased lung edema clearance,
vascular tone, neuronal excitability and the neuroprotective
effect. However, the DA receptor-NKA complex interacts and each
part performs reciprocal functional modulation of the other
(20). Therefore, this review
presents a detailed discussion on the regulation of NKA by DA
receptors via multiple pathways, followed by specific cellular
responses and the modulation of DA receptors induced by NKA.
2. DA receptor-mediated regulation of
NKA
It is well documented that DA increases or decreases
the activity of NKA in an organ-specific manner. This regulation
occurs, at least partially, via receptor-mediated second messenger
activation or phosphorylation/dephosphorylation and promotes NKA
insertion or removal from the plasma membrane. The activation of DA
receptors induces opposing modulatory effects upon NKA in various
tissue and cells, for example inhibition in the kidney and
stimulation in alveolar epithelial cells (AECs). For example, the
difference is due to phosphorylation of the NKA α1
subunit in the kidney and dephosphorylation in the lung, leading to
endocytosis/exocytosis, which in turn results in the
decrease/increase of NKA activity and alternate second messenger
activation. It is generally accepted that the signal pathways of
NKA regulation by DA involve the stimulation of serine/threonine
protein kinases, primarily PKA and PKC, as well as other pathways,
including tyrosine kinase, AP-2, PI3K-PKC-G protein-coupled
receptor kinase type 2 (GRK2) cascade and mitogen-activated protein
kinase (MAPK)/extracellular signal-regulated kinase (ERK)
pathways.
DA receptor-mediated inhibition of NKA in
the kidney
DA synthesized within the rat renal proximal tubule
(PCT) is important for the regulation of renal sodium excretion,
thus maintaining sodium homeostasis, a major determinant of blood
pressure (21–23). The natriuretic effect of DA depends
on its ability to increase the glomerular filtration rate and/or to
directly modulate tubular sodium reabsorption by affecting NKA
(24). It has previously been
shown that phosphorylation of the NKA α1 subunit Ser-11
or -18, in response to DA, provides the signal for NKA endocytosis.
The inhibition of NKA activity in PCTs or a cell line derived from
opossum proximal tubules (OK cells) (25,26)
and the activation of PI3K, is critical for NKA endocytosis, as it
favors binding of the AP-2 μ2 subunit to Tyr-537 within the
α1 subunit and, thereby, promotes clathrin recruitment
(27–30).
Moreover, receptor-mediated internalization of NKAα
and β subunits into early and late endosomes from the basolateral
membrane (BLM) via a clathrin-dependent pathway and inhibition of
NKA activity by DA in rat renal PCTs indicates a key role for
activation of PI3K (27,31) in the endocytic sequence and
demonstrates that this process is PKC dependent (32). In wild-type OK cells (33), early inhibition of NKA activity by
DA is dependent on the stimulation of D1 and
D2 receptors through activation of the PLC/PKC- and
PLA2-dependent pathways, as well as late inhibition through PKA-
and PLA2-dependent pathways. These pathways involve pertussis toxin
(PTX)-sensitive G protein, resulting in the early downregulation of
BLM expression of the α subunit. By contrast, the total cell
expression remains constant. Besides, the stimulation of
D2-like receptors leads to the inhibition of NKA
activity and hyperpolarization. These two effects are associated
with the opening of K+ channels and activation of
PTX-sensitive G protein in OK cells (34). Furthermore, the DA-mediated
regulation of NKA requires the PDZ-2 domain of sodium hydrogen
regulatory factor-1 in OK cells (35). Ouabain, as an NKA inhibitor, was
previously shown to decrease D1 receptor-induced NKA
inhibition in a human PCT line, resulting in increased renal sodium
reabsorption and eventual ouabain-induced hypertension (36). In human renal PCT cells, the
scaffolding protein, caveolin-1, was revealed to be necessary for
the association of D1-like receptors with G
protein-coupled receptor kinase type 4 (GRK4) and the
AP-2-associated reduction in plasma membrane NKA (37), among which the presence of gene
variants at 3 exons in GRK4 is consistent with the known effects of
GRK4 variants on uncoupling D1 receptors from adenylate
cyclase, demonstrating that GRK4 is a potential therapeutic target
(38).
In PCT or OK cells, NKA inhibition via DA receptors
is mediated by diverse signaling pathways in addition to
phosphorylation of the NKA α1 subunit. For example, DA
produces a natriuresis, attributed, in part, to inhibition of NKA
activity in PCT, and the impairment of this inhibition has been
linked to several forms of hypertension in animals. These effects
are largely mediated via D1 or D1-like
receptors (36) or by synergistic
action of the D1 and D2 receptors (39) coupled to adenylyl cyclase
activation and cAMP generation (40), as well as PLC/PKC signaling in rat
renal PCT cells or Madin-Darby canine kidney epithelial cells
(41,42). Dahl salt-sensitive rats lack the
capacity to inhibit tubular NKA activity due to defective
D1 receptor adenylate cyclase coupling. This defect may
contribute to the impaired natriuretic capacity in prehypertensive
Dahl salt-sensitive rats (43).
Those studies indicate that the failure of DA receptors to inhibit
NKA activity contributes to animal models of hypertension (44), among which the adenylate cyclase
pathway is important. Specific studies provided direct evidence
that the D1-like receptor-mediated inhibition of NKA
activity in OK cells involves a G protein of the Gsα, but not of
the Gq/11α class, positively coupled to adenylyl cyclase.
Subsequent activation of PKA and PKC pathways follows in a single
sequence of events with PKA activation prior to PKC activation,
which likely includes phosphorylation of PLC by PKA (45). These observations suggest that the
cAMP-PKA-PLC signaling cascade is important for the DA-induced
PKC-mediated inhibition of renal NKA activity (45). Furthermore, among these signaling
pathways, PKC is also important for DA receptor-induced NKA
regulation. In OK cells, PKC-dependent inhibition of NKA activity
by DA requires the integrity of the association between actin
cytoskeleton and NKA (46).
However, various PKC isoforms mediate with differing and even
opposing effects. For example, PKC-β and -ζ mediate opposing
effects on PCT NKA activity. Phorbol 12-myristate
13-acetate-dependent stimulation of NKA is mediated by the
activation of PKC-β, whereas inhibition by DA requires the
activation of PKC-ζ (47). Lipoic
acid, while reducing oxidative stress, normalizes PKC activity and
restores D1 receptor-G(q/11)α-PLC signaling, along with
the ability of SKF-38393 (D1 receptor agonist) to
inhibit NKA activity (41). These
effects may infer value in clinical medication.
A previous study demonstrated that stimulation of
the D5 receptor decreases NKA activity in renal PCT
cells from Wistar-Kyoto rats. Furthermore, pretreatment with a
D3 receptor agonist for 24 h enhanced D5
receptor expression and the D5 receptor-mediated
inhibitory effect on NKA activity in Wistar-Kyoto cells, but
decreased these effects in spontaneously hypertensive rat cells
(48), indicating crosstalk of
D3 and D5 receptors in NKA activity
regulation. Wistar fatty rats tend to develop salt-sensitive
hypertension, which may be caused by excessive sodium retention
occurring as a result of a defective dopaminergic system in the
kidney that fails to inhibit NKA activity (49).
NKA is a transducer of signals between extracellular
and intracellular compartments. Ouabain-stimulated NKA signaling
(at oubain concentrations far lower than those inhibiting pump
activity) has recently demonstrated clinical promise by protecting
malnourished embryonic kidneys from adverse developmental
programming. A deeper understanding of the tissue-protective role
of NKA signaling, as well as the regulation of NKA pumping
activity, is of fundamental importance for the understanding and
treatment of kidney diseases and kidney-associated hypertension
(50).
DA receptor-mediated stimulation of NKA
in the lung
By contrast, in AECs of normal rats, the stimulation
of DA receptors increases lung edema clearance by increasing NKA
function. Specific studies have shown that DA promotes protein
phosphatase 2A translocation to the membrane fraction, leading to
dephosphorylation of the NKAα1 subunit at the Ser-18
residue and its recruitment to the cell plasma membrane of alveolar
epithelial type II (ATII) cells via DA receptors, which results in
increased NKA activity (51,52)
and lung fluid clearance (53).
Moreover, new evidence has revealed that administration of a
tyrosine-enriched diet to rats increases endogenous DA production,
which causes increased NKA protein abundance in AECs, leading to
increased NKA activity via the activation of D1
receptors (54). Conceivably, the
ability of AECs to produce DA may constitute a physiological
mechanism favoring alveolar fluid reabsorption. In addition, the
regulation of NKA by DA under pathological conditions exerts an
important role. Studies have reported that rats exposed to 100%
oxygen for 64 h demonstrate a decreased ability to clear edema in
the isolated-perfused lung model, in association with decreased NKA
activity in ATII cells (55).
During acute hyperoxic lung injury, DA restores the ability of the
lung to clear edema, which is mediated by recruitment and
translocation of NKA from intracellular pools to the plasma
membrane of the alveolar epithelium (56). This may be beneficial in the
management of patients with acute hypoxemic respiratory failure. A
previous study provided new evidence that DA exerts its effect by
upregulating active Na+ transport via the activation of
amiloride-sensitive sodium channels and the basolateral NKA within
minutes, which has been shown to be beneficial in a rat model of
ventilator-induced lung injury (57). This information is relevant to
current clinical trials exploring the effects of alveolar fluid
clearance stimulation in patients with acute lung injury.
Furthermore, overexpression of the NKAβ1 subunit led to
increased alveolar fluid clearance in the rat model of acute
hydrostatic pulmonary edema (58).
Therefore, DA receptor-mediated stimulation of NKA in the lung
provides a potential therapeutic option for the treatment of
pulmonary edema.
DA-induced exocytosis of NKA is dependent on
activation of PKC isozymes, PKC-ɛ and -δ (59). Additionally, DA enhances NKA
activity in ATII cells by MAPK/ERK pathway proteins via the
D2 receptor by a mechanism involving de novo
synthesis of β subunits and, possibly, recruitment of pre-existing
α subunits (60). Furthermore, DA
activates MAPK/ERK via Ras proteins, the serine/threonine kinase
Raf-1 and diacylglycerol-dependent PKC isoenzymes through the
D2 receptor. However, importantly and contrary to the
classical model, this pathway does not involve Grb2/SOS (adapter
protein/guanine nucleotide exchange factor) complex formation in
rat ATII cells (61). More studies
are therefore required to elucidate the post-transcriptional
regulatory mechanisms involved in DA-mediated NKA regulation.
DA receptor-mediated regulation of NKA in
the intestine
The activity of intestinal NKA may also be affected
or modulated by DA. The inhibition of jejunal NKA activity through
D1 receptors is mediated by locally formed DA and is
dependent on salt intake in 20-day-old rats (62) and obese rats (63). By contrast, in lean Zucker rats,
NKA fails to respond to the activation of D1 receptors
irrespective of their salt intake (63). Furthermore, the association between
salt intake, increased DA formation and inhibition of NKA at the
intestinal level is not as straightforward as that described in
renal tissues. For example, in old-aged rats, a high salt diet
fails to alter the intestinal dopaminergic tonus or NKA activity,
whereas in adult rats this diet decreases NKA activity (64). DA inhibits jejunal NKA in young
breast-fed Wistar rats through the activation of D1
receptors, but not in adult rats or in young rats fed solid food
for two days. The lack of DA sensitivity is accompanied by markedly
elevated basal jejunal NKA activity (65). Therefore, food intake in young rats
is important in the development of the DA insensitivity by NKA
activity. These studies indicate that DA receptor-mediated
regulation of NKA in the intestine remains controversial.
Additionally, jejunal NKA activity in young rats is
regulated by cholera toxin (CTX)- and PTX-sensitive G proteins,
however, regulation of NKA by CTX-sensitive G proteins is absent in
adult animals. This difference may explain the failure of DA to
inhibit intestinal NKA activity in adult rats (66). Furthermore, the uninephrectomised
and three-quarter nephrectomised rats (3/4nx rats, a model of
chronic renal insufficiency) present DA-sensitive enhanced
natriuresis. In uninephrectomised rats, this is accompanied by a
reduced jejunal NKA activity (67), whereas in 3/4nx rats, by an
increased jejunal NKA activity (68). These studies indicate that the
changes in jejunal NKA activity may contribute to the maintenance
of sodium homeostasis in conditions of compromised renal function
and that there are complementary functions between the intestine
and kidney during development and renal-intestinal crosstalk.
DA receptor-mediated inhibition of NKA in
the brain, non-pigmented ciliary epithelium and vascular bed
The modulation of NKA by DA also occurs in other
tissues, for example, the synergism between D1 and
D2 receptors underlies numerous electrophysical and
behavioural effects of DA in the mammalian brain. Previous studies
have provided evidence for joint and separated confinement of the
D1 receptor and NKA in the postsynaptic areas of
dendritic spines (69). DA
inhibits the NKA activity of isolated striatal neurons through a
synergistic effect on D1 and D2 receptors
(70,71), while fencamfamine, acting as an
indirect DA agonist, reduces NKA activity through cAMP-dependent
PKA mechanisms via D1 and D2 receptors in rat
striatum (71). These studies
support the hypothesis that DA and other neurotransmitters regulate
neuronal excitability through the novel mechanism of pump
inhibition. Besides, in rat brain, inhibition of NKA activity and
mitochondrial respiratory chain function (various toxic actions) by
DA may trigger intracellular damage pathways leading to the
mortality of nigral dopaminergic neurons (72). D1- and
D2-like receptor activation contribute to neuronal death
in the striatum of newborn piglets after hypoxic-ischemic
encephalopathy in association with increased nitrotyrosine and
decreased NKA activity. Additionally, the mechanisms of
D1-like receptor toxicity may involve cAMP-regulated
phosphoprotein of 32 kDa-dependent phosphorylation of
N-methyl-D-aspartate (NMDA) receptor NR1 and NKA
(73). However, the maintenance of
adequate cerebral perfusion pressure, by pharmacologically
preventing systemic hypotension with DA infusion, has been found to
prevent cerebral ischemia and attenuates energy depletion and
neuronal injury in a model of newborn piglets with meningitis. In
this model, the decreased cerebral cortical cell membrane NKA
activity and increased lipid peroxidation products, indicative of
meningitis-induced brain damage, are significantly attenuated by DA
infusion (74). Similarly, the
decreased DA levels in the brain may be partly responsible for the
decrease in NKA activity in the striatum of newborn piglets during
posthypoxic reoxygenation (75). A
further study revealed that D2 receptors are involved in
stimulating striatal NKA activity following short-term morphine
treatment, whereas D1 receptors are involved in
inhibiting striatal NKA activity subsequent to long-term morphine
treatment (76). Therefore,
bidirectional regulation of NKA by DA in the brain indicates its
complexity and implications in neuroprotective therapy (Fig. 1).
In rabbit non-pigmented ciliary epithelium,
stimulation of the D1 receptor causes a reduction of
NKA-mediated ion transport and NKA activity by a mechanism that may
involve a tyrosine kinase step and the cAMP/PKA pathway (77).
Moreover, DA inhibits rat tail artery NKA activity,
which the D1 and D2 receptor subtypes
mediate. It appears that D1, but not D2
receptor agonist-induced inhibition of NKA is mediated by a
PTX-sensitive mechanism and may be coupled to the activation of the
PLC system. Modulation of NKA by DA may contribute to vascular tone
(78). Moreover, DA and DA
receptor agonists inhibit NKA activity through D1
receptors, which are linked to PTX sensitive-mechanisms and the PLC
signaling pathway in cultured rat aortic smooth muscle cells
(79) (Fig. 1).
3. NKA-mediated regulation of DA receptors
or DA metabolism
It has been well documented that DA increases or
decreases the activity of NKA in an organ-specific manner via
receptor-mediated second messenger activation and promotes NKA
insertion or removal from the plasma membrane. However, the first
evidence of a direct interaction between NKA and DA receptors has
also been reported. Furthermore, NKA and DA receptors are able to
functionally regulate one another via protein-protein interactions
in the absence of ligands or downstream signaling events,
indicating that, in addition to traditional second
messenger-mediated communication, NKA and DA receptors associate in
a complex to provide a more rapid and immediate response to
external stimuli or changes to the cellular environment.
Additionally, NKA association with the D1 and
D2 receptors leads to decreased signaling through a
novel mechanism that is independent of receptor density or
trafficking. This novel finding of DA receptor and NKA interaction
was confirmed by co-immunoprecipitation and western blot analyses
in HEK293T cells and this was further substantiated by verifying
the interaction between native DA receptors and NKA in brain tissue
(20). Moreover, an increased
intracellular sodium concentration induces the increased
colocalization of DA receptors with NKA molecules in the region of
the plasma membrane of renal epithelial cells (80). Ouabain, as a NKA inhibitor,
decreased D1 receptor-induced NKA inhibition in the
human PCT cell line, which was responsible for the increase in
renal sodium reabsorption and eventually led to ouabain-induced
hypertension (36). This indicates
that NKA inhibition is important in the regulation of DA
receptor-induced sodium homeostasis and the subsequent regulation
of blood pressure. Cannabinoids and arachidonic acid have been
found to inhibit DA and 5-HT uptake into rat neocortical
synaptosomes; this effect was neither cannabinoid receptor-mediated
nor due to competitive inhibition of membrane transporters, but was
partly affected by a decreased NKA activity (81). Furthermore, in HEK293T cells with
NKA and DA receptor co-expression, the expression of NKA was found
to markedly decrease D1 and D2 receptor
densities with a concomitant functional decrease in DA
receptor-mediated regulation of cAMP levels. However,
pharmacological inhibition of endogenous or overexpressed NKA
enhance DA receptor function without altering receptor number or
localization. Similarly, DA receptor function was also enhanced by
small-interfering RNA reduction of endogenous NKA. These
observations indicate that, under basal conditions, NKA negatively
regulates DA receptor function via protein-protein interactions.
Expression of DA receptors decreases endogenous NKA function in a
reciprocal manner in the absence of DA, suggesting DA receptor
proteins as regulators of NKA activity (20). This crosstalk of reciprocal
regulation between DA receptors and NKA provides a novel control
mechanism for DA receptor signaling and cellular ion balance.
4. Dicussion and future prospects
DA receptor-mediated regulation of NKA by
synergism with other substances
The synergism of DA and 20-hydroxyeicosatetraenoic
acid, a major arachidonic acid metabolite of cytochrome P450,
inhibits NKA activity in PCT via the D1 signaling
pathway (82). Urodilatin or DA
decreases NKA activity, while urodilatin and DA combined, further
decrease NKA activity, demonstrating an additive effect on the
sodium pump, suggesting that urodilatin and DA act via a common
intracellular pathway to decrease sodium and water tubular
reabsorption, contributing to its natriuretic and diuretic effects
(83). Additionally,
synergistically interacting D1 and NMDA receptors
mediate non-vesicular transporter-dependent γ-aminobutyric acid
(GABA) release in rat striatal medium spiny neurons through
cAMP-dependent inhibition of NKA. This induces the accumulation of
intracellular sodium, reversal of the GABA carrier and potentiation
of NMDA-induced release (84).
Stimulation of the D3 receptor inhibited NKA activity,
which was increased by pretreatment with the endothelin B-receptor
agonist, BQ3020, in renal PCT cells of Wistar-Kyoto rats, but not
in renal PCT cells of spontaneously hypertensive rats (85). By contrast, pretreatment with
PD128907, a D3 receptor agonist, elevated the inhibitory
effect of BQ3020 on NKA activity in Wistar-Kyoto, but not in
spontaneously hypertensive cells (86). Furthermore, insulin increases
D5 receptor-mediated NKA inhibition and the aberrant
interaction between insulin and D5 receptors regulates
renal sodium transport and participates in the pathogenesis of
hypertension (87). These
synergistic effects of DA receptors and other substances play a
crucial role in the regulation of NKA and associated clinical
diseases.
DA receptor-mediated regulation of NKA by
antagonism with other substances
The selective activation of 5-HTIA
receptors increases NKA activity in renal cortical tubules,
responsible for the antinatriuretic effect of 5-HT and antagonizes
NKA inhibition by DA (88).
Similarly, in the intestine, the inhibitory action of maximal doses
of DA on NKA activity is significantly prevented by coincubation
with 5-HT, indicating the presence of a functional antagonism
between the two amines in the control of NKA activity (62).
Additionally, angiotensin receptor subtypes
(AT1) and D1 receptors function as a unit of
opposites, which may provide a highly versatile and sensitive
system for short-term regulation of sodium excretion (89). The stimulation of renal
AT1 receptors by angiotensin II (Ang II) delivers
signals through the PLC pathway to inhibit extraneuronal DA uptake.
Ang II was able to stimulate renal NKA activity alone, while DA and
Ang II functioned together through a common pathway involving
reversible renal tubular NKA deactivation and activation,
respectively (90). Ang II induces
a rapid partial internalization of D1 receptors and
elimination of D1 receptor signaling in rat renal PCT
cells, resulting in a change in NKA activity. Similarly, exposure
to a D1 agonist results in rapid partial internalization
of AT1 receptors and complete elimination of
AT1 receptor signaling. D1 and AT1
receptors are partners in a multiprotein complex. NKA, the target
for the two receptors, is included in this complex and a region in
the COOH-terminal tail of D1 receptors (residues
397–416) is found to interact with AT1 receptors and NKA
(89). These studies indicate that
AT1 and D1 receptors function as a unit of
opposites, which provides a highly versatile and sensitive system
for short-term regulation of sodium excretion.
Additionally, in vivo short-term morphine
treatment has been found to stimulate NKA activity by inhibiting
PKA activity and subsequently decreasing NKA phosphorylation in a
dose-dependent manner, which may be significantly inhibited by the
D2-like receptor antagonist, eticlopride. Contrary to
short-term morphine treatment, long-term morphine treatment
significantly suppressed NKA activity by stimulating PKA activity
and subsequently increasing the NKA phosphorylation, which may be
significantly inhibited by the D1-like receptor
antagonist, SCH 23390 (76). These
observations demonstrate that DA receptors are involved in the
regulation of NKA activity following activation of opioid receptors
by morphine (76).
Similarly, in rat renal PCT cells, NKA activity is
stimulated by Ang II and oxymetazoline (an α-adrenergic agonist) at
physiological, nonsaturating Na+ concentrations, while
these stimulatory effects are blocked by DA and atrial natriuretic
peptide, as well as by their respective second messengers, cAMP and
cGMP (91). DA receptor-induced
NKA regulation is antagonized by other hormones, including insulin.
Insulin activates the PI3K-PKC-GRK2 cascade, causing D1
receptor serine phosphorylation, which leads to D1
receptor downregulation and uncoupling from G proteins. This
results in the failure of D1 receptor agonists to
stimulate G proteins and inhibit NKA activity (92,93).
Furthermore, DA oxidation products, for example
H2O2 and reactive quinones, have been
responsible for various toxic actions of DA in rat brain, including
the inhibition of NKA activity and mitochondrial respiratory chain
function, which may trigger intracellular damage pathways leading
to the death of the nigral dopaminergic neuron. The
H2O2- and quinone-scavenging properties of
N-acetylcysteine are likely to account for its protective
effect against NKA inhibition induced by DA. The results have
important implications in the neuroprotective therapy of sporadic
Parkinson’s disease (72).
Therefore, the regulation of NKA by DA may be important for the
development of neuroprotective therapy.
The bidirectionally regulated activity of NKA
indicates the complexity of NKA regulation in sodium reabsorption
in vivo. These may infer certain value in clinical
medication.
DA receptor-mediated regulation of NKA by
affecting mutual release or turnover, as well as catabolism between
DA and other substances
DA modulates NKA by affecting the mutual release or
turnover and catabolism between DA and other hormones. For example,
D2 receptors have been implicated in stimulating
striatal NKA activity following short-term morphine treatment,
whereas D1 receptors are involved in inhibiting striatal
NKA activity subsequent to long-term morphine treatment. In this
case, in vivo short-term injection of morphine is able to
promote DA release and activate D2 receptors, resulting
in NKA stimulation, whereas in vitro direct administration
of morphine to isolated striatal synaptosomes is not the result of
non-functional striatal circuits or inhibition of the afferent
input signal (76).
Atrial natriuretic factor (ANF) may decrease renal
DA turnover and catabolism, favor DA accumulation into renal cells
and increase its endogenous content and availability. This permits
D1 receptor recruitment and stimulation and, in turn,
inhibition of NKA activity resulting in decreased sodium
reabsorption. ANF and DA may act in this way via a common pathway
to enhance natriuresis and diuresis (94). Therefore, increased effort into the
study of NKA regulation in vivo is essential and is also
critical for clinically relevant medication in hormone synergism
and counteraction of NKA regulation, which results in differing
responses.
5. Conclusion
DA-mediated regulation of NKA through multiple
signal transduction pathways (cAMP/PKA, PI-PLC/PKC, PI3K, AP-2,
tyrosine phosphatase and MAPK/ERK) results in diverse responses,
including the natriuretic effect, lung edema clearance, vascular
tone, neuronal excitability and neuroprotective effects. These
results suggest that NKA regulation by DA is affected by complex
functional networks that are either specific or non-specific and
involve distinct and often mutually interacting intracellular
signal transduction pathways. Conversely, NKA also regulates DA
receptors, indicating the complicated crosstalk between DA
receptors and NKA. Accordingly, more experiments should be
conducted on the complexity of various substances interacting with
NKA activity in vivo, as these may be of value in clinically
relevant medication. In conclusion, understanding the crosstalk of
DA receptors and NKA, as well as further investigation into this
function, are important for the interest of drug development.
Acknowledgements
The authors acknowledge support from the Natural
Science Foundation of China (nos. 30801444 and 81200886), the
Natural Science Foundation of Hebei Province (nos. H2012208020,
H2012208080 and C2011208081), the Hebei University of Science and
Technology Discipline Construction Office and the State Key
Laboratory Breeding Base-Hebei Key Laboratory of Molecular
Chemistry For Drugs.
References
|
1
|
Contreras F, Fouillioux C, Bolivar A, et
al: Dopamine, hypertension and obesity. J Hum Hypertens. 16(Suppl
1): S13–S17. 2002. View Article : Google Scholar
|
|
2
|
Skou JC: The influence of some cations on
an adenosine triphosphatase from peripheral nerves. Biochim Biophys
Acta. 23:394–401. 1957. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Wright EM, Hirayama BA and Loo DF: Active
sugar transport in health and disease. J Intern Med. 261:32–43.
2007. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Namekata K, Harada C, Kohyama K, Matsumoto
Y and Harada T: Interleukin-1 stimulates glutamate uptake in glial
cells by accelerating membrane trafficking of
Na+/K+-ATPase via actin depolymerization. Mol
Cell Biol. 28:3273–3280. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Swift F, Birkeland JA, Tovsrud N, et al:
Altered Na+/Ca2+-exchanger activity due to
downregulation of Na+/K+-ATPase
alpha2-isoform in heart failure. Cardiovasc Res. 78:71–78.
2008.PubMed/NCBI
|
|
6
|
Blanco G, DeTomaso AW, Koster J, Xie ZJ
and Mercer RW: The alpha-subunit of the Na, K-ATPase has catalytic
activity independent of the beta-subunit. J Biol Chem.
269:23420–23425. 1994.PubMed/NCBI
|
|
7
|
Jaisser F, Jaunin P, Geering K, Rossier BC
and Horisberger JD: Modulation of the Na, K-pump function by beta
subunit isoforms. J Gen Physiol. 103:605–623. 1994. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Beguin P, Wang X, Firsov D, et al: The
gamma subunit is a specific component of the Na, K-ATPase and
modulates its transport function. EMBO J. 16:4250–4260. 1997.
View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Shamraj OI and Lingrel JB: A putative
fourth Na+, K(+)-ATPase alpha-subunit gene is expressed
in testis. Proc Natl Acad Sci USA. 91:12952–12956. 1994.PubMed/NCBI
|
|
10
|
Barcroft LC, Moseley AE, Lingrel JB and
Watson AJ: Deletion of the Na/K-ATPase alpha1-subunit gene (Atp1a1)
does not prevent cavitation of the preimplantation mouse embryo.
Mech Dev. 121:417–426. 2004.PubMed/NCBI
|
|
11
|
James PF, Grupp IL, Grupp G, et al:
Identification of a specific role for the Na, K-ATPase alpha 2
isoform as a regulator of calcium in the heart. Mol Cell.
3:555–563. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Ikeda K, Onimaru H, Yamada J, et al:
Malfunction of respiratory-related neuronal activity in
Na+, K+-ATPase alpha2 subunit-deficient mice
is attributable to abnormal Cl− homeostasis in brainstem
neurons. J Neurosci. 24:10693–10701. 2004.PubMed/NCBI
|
|
13
|
Dostanic I, Paul RJ, Lorenz JN, Theriault
S, Van Huysse JW and Lingrel JB: The alpha2-isoform of Na-K-ATPase
mediates ouabain-induced hypertension in mice and increased
vascular contractility in vitro. Am J Physiol Heart Circ Physiol.
288:H477–H485. 2005. View Article : Google Scholar
|
|
14
|
Dostanic I, Lorenz JN, Schultz Jel J, et
al: The alpha2 isoform of Na, K-ATPase mediates ouabain-induced
cardiac inotropy in mice. J Biol Chem. 278:53026–53034. 2003.
View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Dostanic I, Schultz Jel J, Lorenz JN and
Lingrel JB: The alpha 1 isoform of Na, K-ATPase regulates cardiac
contractility and functionally interacts and co-localizes with the
Na/Ca exchanger in heart. J Biol Chem. 279:54053–54061. 2004.
View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Pierre SV, Yang C, Yuan Z, et al: Ouabain
triggers preconditioning through activation of the Na+,
K+-ATPase signaling cascade in rat hearts. Cardiovasc
Res. 73:488–496. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Blair JE, Macarie C, Ruzyllo W, et al;
HORIZON-HF investigators. Rationale and design of the hemodynamic,
echocardiographic and neurohormonal effects of istaroxime, a novel
intravenous inotropic and lusitropic agent: a randomized controlled
trial in patients hospitalized with heart failure (HORIZON-HF)
trial. Am J Ther. 15:231–240. 2008. View Article : Google Scholar
|
|
18
|
Tian D, Dmitrieva RI, Doris PA, et al:
Protein kinase M zeta regulation of Na/K ATPase: a persistent
neuroprotective mechanism of ischemic preconditioning in
hippocampal slice cultures. Brain Res. 1213:127–139. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Zhang L, Zhang Z, Guo H and Wang Y:
Na+/K+-ATPase-mediated signal transduction
and Na+/K+-ATPase regulation. Fundam Clin
Pharmacol. 22:615–621. 2008.
|
|
20
|
Hazelwood LA, Free RB, Cabrera DM,
Skinbjerg M and Sibley DR: Reciprocal modulation of function
between the D1 and D2 dopamine receptors and
the Na+, K+-ATPase. J Biol Chem.
283:36441–36453. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Carey RM: Theodore Cooper Lecture: Renal
dopamine system: paracrine regulator of sodium homeostasis and
blood pressure. Hypertension. 38:297–302. 2001. View Article : Google Scholar
|
|
22
|
Jose PA, Eisner GM and Felder RA:
Regulation of blood pressure by dopamine receptors. Nephron
Physiol. 95:p19–p27. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Jose PA, Eisner GM and Felder RA: Renal
dopamine and sodium homeostasis. Curr Hypertens Rep. 2:174–183.
2000. View Article : Google Scholar
|
|
24
|
Felder RA, Felder CC, Eisner GM and Jose
PA: The dopamine receptor in adult and maturing kidney. Am J
Physiol. 257:F315–F327. 1989.PubMed/NCBI
|
|
25
|
Chibalin AV, Ogimoto G, Pedemonte CH, et
al: Dopamine- induced endocytosis of Na+,
K+-ATPase is initiated by phosphorylation of Ser-18 in
the rat alpha subunit and is responsible for the decreased activity
in epithelial cells. J Biol Chem. 274:1920–1927. 1999.PubMed/NCBI
|
|
26
|
Chibalin AV, Pedemonte CH, Katz AI,
Feraille E, Berggren PO and Bertorello AM: Phosphorylation of the
catalyic alpha-subunit constitutes a triggering signal for
Na+, K+-ATPase endocytosis. J Biol Chem.
273:8814–8819. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Chibalin AV, Zierath JR, Katz AI, Berggren
PO and Bertorello AM: Phosphatidylinositol 3-kinase-mediated
endocytosis of renal Na+, K+-ATPase alpha
subunit in response to dopamine. Mol Biol Cell. 9:1209–1220. 1998.
View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Chen Z, Krmar RT, Dada L, et al:
Phosphorylation of adaptor protein-2 mu2 is essential for
Na+, K+-ATPase endocytosis in response to
either G protein-coupled receptor or reactive oxygen species. Am J
Respir Cell Mol Biol. 35:127–132. 2006.PubMed/NCBI
|
|
29
|
Ogimoto G, Yudowski GA, Barker CJ, et al:
G protein-coupled receptors regulate Na+,
K+-ATPase activity and endocytosis by modulating the
recruitment of adaptor protein 2 and clathrin. Proc Natl Acad Sci
USA. 97:3242–3247. 2000.PubMed/NCBI
|
|
30
|
Done SC, Leibiger IB, Efendiev R, et al:
Tyrosine 537 within the Na+, K+-ATPase
alpha-subunit is essential for AP-2 binding and clathrin-dependent
endocytosis. J Biol Chem. 277:17108–17111. 2002.PubMed/NCBI
|
|
31
|
Yudowski GA, Efendiev R, Pedemonte CH,
Katz AI, Berggren PO and Bertorello AM: Phosphoinositide-3 kinase
binds to a proline- rich motif in the Na+,
K+-ATPase alpha subunit and regulates its trafficking.
Proc Natl Acad Sci USA. 97:6556–6561. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Chibalin AV, Katz AI, Berggren PO and
Bertorello AM: Receptor-mediated inhibition of renal
Na(+)-K(+)-ATPase is associated with endocytosis of its alpha- and
beta-subunits. Am J Physiol. 273:C1458–C1465. 1997.
|
|
33
|
Khundmiri SJ and Lederer E: PTH and DA
regulate Na-K ATPase through divergent pathways. Am J Physiol Renal
Physiol. 282:F512–F522. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Gomes P and Soares-Da-Silva P:
D2-like receptor-mediated inhibition of
Na+-K+-ATPase activity is dependent on the
opening of K+ channels. Am J Physiol Renal Physiol.
283:F114–F123. 2002.PubMed/NCBI
|
|
35
|
Salyer S, Lesousky N, Weinman EJ, Clark
BJ, Lederer ED and Khundmiri SJ: Dopamine regulation of
Na+-K+-ATPase requires the PDZ-2 domain of
sodium hydrogen regulatory factor-1 (NHERF-1) in opossum kidney
cells. Am J Physiol Cell Physiol. 300:C425–C434. 2011.PubMed/NCBI
|
|
36
|
Zhang YR and Yuan ZY: Dopamine-mediated
inhibition of renal Na+/K+-ATPase in HK-2
cells is reduced by ouabain. Clin Exp Pharmacol Physiol.
37:613–618. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Gildea JJ, Israel JA, Johnson AK, Zhang J,
Jose PA and Felder RA: Caveolin-1 and dopamine-mediated
internalization of NaKATPase in human renal proximal tubule cells.
Hypertension. 54:1070–1076. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Gildea JJ, Shah I, Weiss R, et al: HK-2
human renal proximal tubule cells as a model for G protein-coupled
receptor kinase type 4-mediated dopamine 1 receptor uncoupling.
Hypertension. 56:505–511. 2010. View Article : Google Scholar
|
|
39
|
Bertorello A and Aperia A: Inhibition of
proximal tubule Na(+)-K(+)-ATPase activity requires simultaneous
activation of DA1 and DA2 receptors. Am J Physiol. 259:F924–F928.
1990.
|
|
40
|
Felder CC, Campbell T, Albrecht F and Jose
PA: Dopamine inhibits Na(+)-H+ exchanger activity in renal BBMV by
stimulation of adenylate cyclase. Am J Physiol. 259:F297–F303.
1990.
|
|
41
|
Banday AA and Lokhandwala MF: Oxidative
stress reduces renal dopamine D1 receptor-Gq/11alpha G
protein-phospholipase C signaling involving G protein-coupled
receptor kinase 2. Am J Physiol Renal Physiol. 293:F306–F315.
2007.PubMed/NCBI
|
|
42
|
Shahedi M, Laborde K, Bussieres L, Dechaux
M and Sachs C: Protein kinase C activation causes inhibition of
Na/K-ATPase activity in Madin-Darby canine kidney epithelial (MDCK)
cells. Pflugers Arch. 420:269–274. 1992. View Article : Google Scholar
|
|
43
|
Nishi A, Eklof AC, Bertorello AM and
Aperia A: Dopamine regulation of renal Na+, K(+)-ATPase
activity is lacking in Dahl salt-sensitive rats. Hypertension.
21:767–771. 1993.
|
|
44
|
Banday AA, Lau YS and Lokhandwala MF:
Oxidative stress causes renal dopamine D1 receptor
dysfunction and salt-sensitive hypertension in Sprague-Dawley rats.
Hypertension. 51:367–375. 2008. View Article : Google Scholar
|
|
45
|
Gomes P and Soares-da-Silva P: Role of
cAMP-PKA-PLC signaling cascade on dopamine-induced PKC-mediated
inhibition of renal Na(+)-K(+)-ATPase activity. Am J Physiol Renal
Physiol. 282:F1084–F1096. 2002.PubMed/NCBI
|
|
46
|
Gomes P and Soares-da-Silva P:
Dopamine-induced inhibition of Na+-K+-ATPase
activity requires integrity of actin cytoskeleton in opossum kidney
cells. Acta Physiol Scand. 175:93–101. 2002.
|
|
47
|
Efendiev R, Bertorello AM and Pedemonte
CH: PKC-beta and PKC-zeta mediate opposing effects on proximal
tubule Na+, K+-ATPase activity. FEBS Lett.
456:45–48. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Huang H, Ren H, Chen C, et al:
D3 dopamine receptor regulation of D5
receptor expression and function in renal proximal tubule cells.
Hypertens Res. 35:639–647. 2012.
|
|
49
|
Tsuchida H, Imai G, Shima Y, Satoh T and
Owada S: Mechanism of sodium load-induced hypertension in
non-insulin dependent diabetes mellitus model rats: defective
dopaminergic system to inhibit Na-K-ATPase activity in renal
epithelial cells. Hypertens Res. 24:127–135. 2001. View Article : Google Scholar
|
|
50
|
Aperia A: 2011 Homer Smith Award: To serve
and protect: classic and novel roles for Na+,
K+-adenosine triphosphatase. J Am Soc Nephrol.
23:1283–1290. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
51
|
Lecuona E, Garcia A and Sznajder JI: A
novel role for protein phosphatase 2A in the dopaminergic
regulation of Na, K-ATPase. FEBS Lett. 481:217–220. 2000.
View Article : Google Scholar : PubMed/NCBI
|
|
52
|
Lecuona E, Dada LA, Sun H, et al: Na,
K-ATPase alpha1-subunit dephosphorylation by protein phosphatase 2A
is necessary for its recruitment to the plasma membrane. FASEB J.
20:2618–2620. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
53
|
Helms MN, Chen XJ, Ramosevac S, Eaton DC
and Jain L: Dopamine regulation of amiloride-sensitive sodium
channels in lung cells. Am J Physiol Lung Cell Mol Physiol.
290:L710–L722. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
54
|
Adir Y, Azzam ZS, Lecuona E, et al:
Augmentation of endogenous dopamine production increases lung
liquid clearance. Am J Respir Crit Care Med. 169:757–763. 2004.
View Article : Google Scholar : PubMed/NCBI
|
|
55
|
Olivera WG, Ridge KM and Sznajder JI: Lung
liquid clearance and Na, K-ATPase during acute hyperoxia and
recovery in rats. Am J Respir Crit Care Med. 152:1229–1234. 1995.
View Article : Google Scholar : PubMed/NCBI
|
|
56
|
Saldias FJ, Lecuona E, Comellas AP, Ridge
KM and Sznajder JI: Dopamine restores lung ability to clear edema
in rats exposed to hyperoxia. Am J Respir Crit Care Med.
159:626–633. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
57
|
Jaitovich A and Sznajder JI: Improving
survival by increasing lung edema clearance: is airspace delivery
of dopamine a solution? Crit Care. 12:1352008. View Article : Google Scholar : PubMed/NCBI
|
|
58
|
Azzam ZS, Dumasius V, Saldias FJ, Adir Y,
Sznajder JI and Factor P: Na, K-ATPase overexpression improves
alveolar fluid clearance in a rat model of elevated left atrial
pressure. Circulation. 105:497–501. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
59
|
Ridge KM, Dada L, Lecuona E, et al:
Dopamine-induced exocytosis of Na, K-ATPase is dependent on
activation of protein kinase C-epsilon and -delta. Mol Biol Cell.
13:1381–1389. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
60
|
Guerrero C, Lecuona E, Pesce L, Ridge KM
and Sznajder JI: Dopamine regulates Na-K-ATPase in alveolar
epithelial cells via MAPK-ERK-dependent mechanisms. Am J Physiol
Lung Cell Mol Physiol. 281:L79–L85. 2001.PubMed/NCBI
|
|
61
|
Guerrero C, Pesce L, Lecuona E, Ridge KM
and Sznajder JI: Dopamine activates ERKs in alveolar epithelial
cells via Ras-PKC-dependent and Grb2/Sos-independent mechanisms. Am
J Physiol Lung Cell Mol Physiol. 282:L1099–L1107. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
62
|
Vieira-Coelho MA, Teixeira VA, Finkel Y,
Soares-Da-Silva P and Bertorello AM: Dopamine-dependent inhibition
of jejunal Na+-K+-ATPase during high-salt
diet in young but not in adult rats. Am J Physiol. 275:G1317–G1323.
1998.PubMed/NCBI
|
|
63
|
Lucas-Teixeira VA, Hussain T, Serrao P,
Soares-da-Silva P and Lokhandwala MF: Intestinal dopaminergic
activity in obese and lean Zucker rats: response to high salt
intake. Clin Exp Hypertens. 24:383–396. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
64
|
Vieira-Coelho MA, Serrao P, Hussain T,
Lokhandwala MF and Soares-da-Silva P: Salt intake and intestinal
dopaminergic activity in adult and old Fischer 344 rats. Life Sci.
69:1957–1968. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
65
|
Lucas-Teixeira V, Vieira-Coelho MA and
Soares-Da-Silva P: Food intake abolishes the response of rat
jejunal Na(+), K(+)-ATPase to dopamine. J Nutr. 130:877–881.
2000.PubMed/NCBI
|
|
66
|
Vieira-Coelho MA and Soares-da-Silva P:
Ontogenic aspects of D1 receptor coupling to G proteins
and regulation of rat jejunal Na+, K+ ATPase
activity and electrolyte transport. Br J Pharmacol. 129:573–581.
2000.
|
|
67
|
Vieira-Coelho MA, Serrao P, Guimaraes JT,
Pestana M and Soares-da-Silva P: Concerted action of dopamine on
renal and intestinal Na(+)-K(+)-ATPase in the rat remnant kidney.
Am J Physiol Renal Physiol. 279:F1033–F1044. 2000.PubMed/NCBI
|
|
68
|
Sampaio-Maia B, Serrao P, Moura M and
Pestana M: Jejunal dopamine and Na, K-ATPase activity in early
chronic renal insufficiency. Nephrology (Carlton). 11:63–67. 2006.
View Article : Google Scholar : PubMed/NCBI
|
|
69
|
Blom H, Ronnlund D, Scott L, et al:
Nearest neighbor analysis of dopamine D1 receptors and
Na(+)-K(+)-ATPases in dendritic spines dissected by STED
microscopy. Microsc Res Tech. 75:220–228. 2012.
|
|
70
|
Bertorello AM, Hopfield JF, Aperia A and
Greengard P: Inhibition by dopamine of (Na(+)+K+)ATPase activity in
neostriatal neurons through D1 and D2
dopamine receptor synergism. Nature. 347:386–388. 1990.
|
|
71
|
Pinto Ferreira M, DeLucia R, Luiz
Aizenstein M, Glezer I and Scavone C: Fencamfamine modulates
sodium, potassium-ATPase through cyclic AMP and cyclic
AMP-dependent protein kinase in rat striatum. J Neural Transm.
105:549–560. 1998.PubMed/NCBI
|
|
72
|
Bagh MB, Maiti AK, Jana S, Banerjee K, Roy
A and Chakrabarti S: Quinone and oxyradical scavenging properties
of N-acetylcysteine prevent dopamine mediated inhibition of
Na+, K+-ATPase and mitochondrial electron
transport chain activity in rat brain: implications in the
neuroprotective therapy of Parkinson’s disease. Free Radic Res.
42:574–581. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
73
|
Yang ZJ, Torbey M, Li X, et al: Dopamine
receptor modulation of hypoxic-ischemic neuronal injury in striatum
of newborn piglets. J Cereb Blood Flow Metab. 27:1339–1351. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
74
|
Park WS, Chang YS, Shim JW, et al: Effects
of dopamine infusion on cerebral blood flow, brain cell membrane
function and energy metabolism in experimental Escherichia
coli meningitis in the newborn piglet. J Korean Med Sci.
18:869–875. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
75
|
Zaitseva T, Shen J, Schears G, Creed J,
Wilson DF and Pastuszko A: Effect of catecholamines on activity of
Na(+), K(+)-ATPase in neonatal piglet brain during posthypoxic
reoxygenation. Comp Biochem Physiol A Mol Integr Physiol.
132:139–145. 2002.
|
|
76
|
Wu ZQ, Chen J, Chi ZQ and Liu JG:
Involvement of dopamine system in regulation of Na+,
K+-ATPase in the striatum upon activation of opioid
receptors by morphine. Mol Pharmacol. 71:519–530. 2007.PubMed/NCBI
|
|
77
|
Nakai Y, Dean WL, Hou Y and Delamere NA:
Genistein inhibits the regulation of active sodium-potassium
transport by dopaminergic agonists in nonpigmented ciliary
epithelium. Invest Ophthalmol Vis Sci. 40:1460–1466. 1999.
|
|
78
|
Rashed SM and Songu-Mize E: Regulation of
Na(+)-pump activity by dopamine in rat tail arteries. Eur J
Pharmacol. 284:289–297. 1995.
|
|
79
|
Rashed SM and Songu-Mize E: Regulation of
Na+, K(+)-ATPase activity by dopamine in cultured rat
aortic smooth muscle cells. Eur J Pharmacol. 305:223–230. 1996.
|
|
80
|
Cinelli AR, Efendiev R and Pedemonte CH:
Trafficking of Na-K-ATPase and dopamine receptor molecules induced
by changes in intracellular sodium concentration of renal
epithelial cells. Am J Physiol Renal Physiol. 295:F1117–F1125.
2008. View Article : Google Scholar
|
|
81
|
Steffens M and Feuerstein TJ:
Receptor-independent depression of DA and 5-HT uptake by
cannabinoids in rat neocortex - involvement of Na(+)/K(+)-ATPase.
Neurochem Int. 44:529–538. 2004.PubMed/NCBI
|
|
82
|
Kirchheimer C, Mendez CF, Acquier A and
Nowicki S: Role of 20-HETE in D1/D2 dopamine
receptor synergism resulting in the inhibition of
Na+-K+-ATPase activity in the proximal
tubule. Am J Physiol Renal Physiol. 292:F1435–F1442.
2007.PubMed/NCBI
|
|
83
|
Citarella MR, Choi MR, Gironacci MM,
Medici C, Correa AH and Fernandez BE: Urodilatin and dopamine: a
new interaction in the kidney. Regul Pept. 153:19–24. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
84
|
Schoffelmeer AN, Vanderschuren LJ, De
Vries TJ, Hogenboom F, Wardeh G and Mulder AH: Synergistically
interacting dopamine D1 and NMDA receptors mediate
nonvesicular transporter-dependent GABA release from rat striatal
medium spiny neurons. J Neurosci. 20:3496–3503. 2000.PubMed/NCBI
|
|
85
|
Zhang Y, Fu C, Ren H, et al: Impaired
stimulatory effect of ETB receptor on D(3) receptor in immortalized
renal proximal tubule cells of spontaneously hypertensive rats.
Kidney Blood Press Res. 34:75–82. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
86
|
Yu C, Yang Z, Ren H, et al: D3
dopamine receptor regulation of ETB receptors in renal proximal
tubule cells from WKY and SHRs. Am J Hypertens. 22:877–883.
2009.
|
|
87
|
Yang J, Cui Z, He D, et al: Insulin
increases D5 dopamine receptor expression and function
in renal proximal tubule cells from Wistar-Kyoto rats. Am J
Hypertens. 22:770–776. 2009.
|
|
88
|
Soares-da-Silva P, Pinto-do OP and
Bertorello AM: Antagonistic actions of renal dopamine and
5-hydroxytryptamine: increase in Na+, K(+)-ATPase
activity in renal proximal tubules via activation of 5-HT1A
receptors. Br J Pharmacol. 117:1199–1203. 1996. View Article : Google Scholar : PubMed/NCBI
|
|
89
|
Khan F, Spicarova Z, Zelenin S, Holtback
U, Scott L and Aperia A: Negative reciprocity between angiotensin
II type 1 and dopamine D1 receptors in rat renal
proximal tubule cells. Am J Physiol Renal Physiol. 295:F1110–F1116.
2008. View Article : Google Scholar : PubMed/NCBI
|
|
90
|
Choi MR, Medici C, Gironacci MM, Correa AH
and Fernandez BE: Angiotensin II regulation of renal dopamine
uptake and Na(+), K(+)-ATPase activity. Nephron Physiol.
111:p53–p58. 2009.PubMed/NCBI
|
|
91
|
Aperia A, Holtback U, Syren ML, Svensson
LB, Fryckstedt J and Greengard P: Activation/deactivation of renal
Na+, K(+)-ATPase: a final common pathway for regulation
of natriuresis. FASEB J. 8:436–439. 1994.PubMed/NCBI
|
|
92
|
Banday AA, Fazili FR and Lokhandwala MF:
Insulin causes renal dopamine D1 receptor
desensitization via GRK2-mediated receptor phosphorylation
involving phosphatidylinositol 3-kinase and protein kinase C. Am J
Physiol Renal Physiol. 293:F877–F884. 2007.PubMed/NCBI
|
|
93
|
Banday AA, Asghar M, Hussain T and
Lokhandwala MF: Dopamine-mediated inhibition of renal Na, K-ATPase
is reduced by insulin. Hypertension. 41:1353–1358. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
94
|
Correa AH, Choi MR, Gironacci M, Aprile F
and Fernandez BE: Atrial natriuretic factor decreases renal
dopamine turnover and catabolism without modifying its release.
Regul Pept. 146:238–242. 2008. View Article : Google Scholar : PubMed/NCBI
|