Introduction
Sir2-related proteins or sirtuins are highly
conserved NAD(+) dependent deacetylases that have been demonstrated
to regulate the lifespan of lower organisms (1,2) and
affect diseases associated with aging in mammals, including
diabetes, inflammation and neurodegenerative disorders (3). The Sir2 ortholog Sirtuin 1 (Sirt1)
functions as an intracellular energy sensor to detect the
concentration of NAD(+), and controls in vivo metabolic
changes under caloric restriction and starvation through its
deacetylase activity to numerous targets, including histones,
nuclear transcriptional factors and enzymes important for aging and
disease (4,5). Sirt1 has a range of molecular
functions and has emerged as an important protein in aging and
metabolic regulation (6). During
the past decade, studies have reported a correlation between Sirt1
activity and aging-associated diseases, including diabetes,
cardiovascular disease and neurodegenerative disorders. Transgenic
mice with ~2-fold higher levels of Sirt1 expression globally are
protected against metabolic decline due to aging (7–9).
Sirt1 mutant mice were small and exhibited notable developmental
defects of the retina and heart, and only infrequently survived
postnatally (10,11). However, systematic investigations
reporting the association of changes in Sirt1 expression with age
are lacking.
In vivo animal models facilitate
investigations aimed at elucidating changes in Sirt1 expression.
The senescence-accelerated mouse (SAM) is an accelerated aging
model that was established through phenotypic selection from a
common genetic pool of the AKR/J strain of mice (12). The SAM model was established in
1981, including nine major senescence-accelerated prone,
short-lived mice (SAM-P) substrains and three major
senescence-accelerated resistant, long-lived mice (SAM-R)
substrains. In the SAM-P strains, normal development and maturity
of reproductive function are observed, and the values for numerous
(but not all) physiological and pathological parameters are similar
to those in SAM-R strains at a young to mature age. The SAM-P
strains grow normally, but then they exhibit early signs of
senescence, including reduction in physical activity and skin
quality, hair loss, periophthalmic lesions and increased
lordokyphosis. These characteristic pathological phenotypes are
similar to those observed in elderly humans. The life span of the
SAM-P strains is ~26% shorter than that of the SAM-R strains. The
common aging characteristic of SAM-P strains is senescence
acceleration following normal development and maturation.
To assess the effects of aging on the changes of
Sirt1 expression, an in vivo model of aging, SAM-P8, and a
control counterpart strain, SAM-R1, were used. The life span of
SAM-P8 mice ranges from 10–17 months, which is shorter than that of
SAM-R1, which ranges from 19–21 months (13). The mRNA and protein levels of Sirt1
were detected in four different tissues, including brain, liver,
skeletal muscle and white adipose tissue in both strains at
different ages (1-, 4-, 8- and 12-month old), thus covering various
different life stages, including young age prior to maturation,
adult, middle age and old age. The results indicated that Sirt1
expression progressively decreased with age.
Materials and methods
Animals
Male SAM-R1 and SAM-P8 at 1-, 4-, 8- and 12-months
old were donated by Professor Jianping Cai (Department of Molecular
Biology, The Key Laboratory of Geriatrics, Beijing Hospital and
Beijing Institute of Geriatrics, Ministry of Health, Beijing,
China). All the animals were housed at 21°C in a 12 h light/dark
cycle. The chow and water were provided ad libitum. The
animals were allowed to acclimatize for 1 week. The rats were then
weighed prior to sacrifice and the food intake over this time was
calculated according to the formula: Weight of the initial chow
(Wi) - weight of the leftover chow (Wl) - weight of the spilled
chow (Ws)/number of mice per cage/days. All the mice were
sacrificed by cervical dislocation. The tissues were dissected on
ice, washed with ice-cold phosphate-buffered saline (PBS), snap
frozen in liquid nitrogen, ground into powder with mortar and
pestle in liquid nitrogen and then stored at −80°C.
This study was approved by the Biomedical Ethics
Committee of Beijing Hospital and Beijing Institute of Geriatrics,
Ministry of Health. The National Institutes of Health (Bethesda,
MD, USA) Guidelines for the Care and Use of Laboratory Animals were
strictly followed and all of the experiments were approved by the
Biomedical Ethics Committee of Peking University (Beijing,
China).
RNA extraction and quantitative
polymerase chain reaction (qPCR)
Total RNA was isolated from 2–50 mg tissues using
TRIzol® reagent (Invitrogen Life Technologies, Carlsbad,
CA, USA) following the manufacturer’s instructions. First-strand
cDNA was synthesized from 2 μg of total RNA with random hexamer
oligonuclueotide primers using a 20 μl reverse transcription system
(New England Biolabs, Ipswich, MA, USA). A total of 1 μl of cDNA
was amplified by qPCR (7500; Applied Biosystems, Foster City, CA,
USA) with SYBR-Green (Takara Bio, Inc., Shiga, Japan). HPRT was
used as internal control to normalize the amplification result and
the expression level of the SAM-R1 1-month-old group was normalized
as ‘1’. The primers for each PCR are as follows: Sirt1-US,
CAGTGTCATGGTTCCTTTGC and Sirt1-DS, CACCGAGGAACTACCTGAT; HPRT-US,
TGAC ACTGGCAAAACAATGCA and HPRT-DS, GGTCCTTTTCA CCAGCAAGCT.
Protein extraction and western blot
analysis
Approximately 50 mg of the tissues were lysed in
RIPA buffer supplied with 5 mM ethylenediaminetetraacetic acid
(EDTA), 1 mM PMSF and complete protease inhibitor cocktail (Sigma,
St. Louis, MO, USA). Following a brief sonication and incubation
for 30 min on ice, the lysate was centrifuged at 14,000 × g at 4°C
for 15 min and the supernatant was used as a whole cell extract.
The protein concentration was determined with a BCA protein assay
kit (Pierce Biotechnology, Inc., Rockford, IL, USA) using BSA as a
standard. The proteins in the sample buffer were denatured by
maintaining them at 100°C for 3–5 min. A total of 30 μg of the
total proteins were separated on a 12% (W/V) sodium dodecyl
sulfate-polyacrylamide gel (SDS-PAGE) and transferred onto a PVDF
membrane (EMD Millipore, Billerica, MA, USA). After blocking the
membrane with 5% (W/V) non-fat milk in Tris-buffered saline
Tween-20 (TBST), the membrane was incubated overnight with primary
antibody, washed in TBST, incubated with horseradish peroxidase
(HRP) conjugated secondary antibody (anti-rabbit IgG from Sigma;
anti-mouse IgG from Upstate Biotechnology, Lake Placid, NY, USA) at
a dilution of 1:5,000 in TBST, washed and developed with ECL
(Pierce Biotechnology, Inc.). The following primary antibodies were
used: Sirt1 (05–707; Upstate Biotechnology), α-tubulin (ab7291;
Abcam, Cambridge, MA, USA) and β-actin (A5441; Sigma).
Statistical analysis
Statistical analyses were performed using SPSS
statistical software. One-way analysis of variance (ANOVA) was
used. Data are expressed as the group mean ± standard error of the
mean (SEM). P<0.05 was considered to indicate a statistically
significant difference.
Results
Changes in body weight of SAM-P8 and
SAM-R1 with age
To assess the effects of aging on the changes of
Sirt1 expression, four different age groups of both SAM-R1 and
SAM-P8 were established; 1-, 4-, 8- and 12-months old, thus
covering various different life stages, including young age prior
to maturation, adult, middle age and old age.
Body weight (Fig.
1A) and appearance did not differ between the SAM-R1 and SAM-P8
at 1-month old (21.5±0.52 and 20.4±0.45 g, respectively). However,
the body weights of SAM-P8 were 8.7% (P<0.05), 8.4% (P<0.05)
and 11.0% (P<0.05) lower than SAM-R1 in the 4-, 8- and 12-month
old groups, respectively (Fig.
1A). These data indicated that following developmental
maturation, the SAM-P8 mice had a higher reduction in body weight
than that of the SAM-R1 mice (Fig.
1A). Of note, there were no significant differences in the
amount of food intake between the two strains at the different ages
(Fig. 1B). These results suggested
that the amount of food intake is not or at least not the only
reason for the differences in body weight between the different
ages and strains.
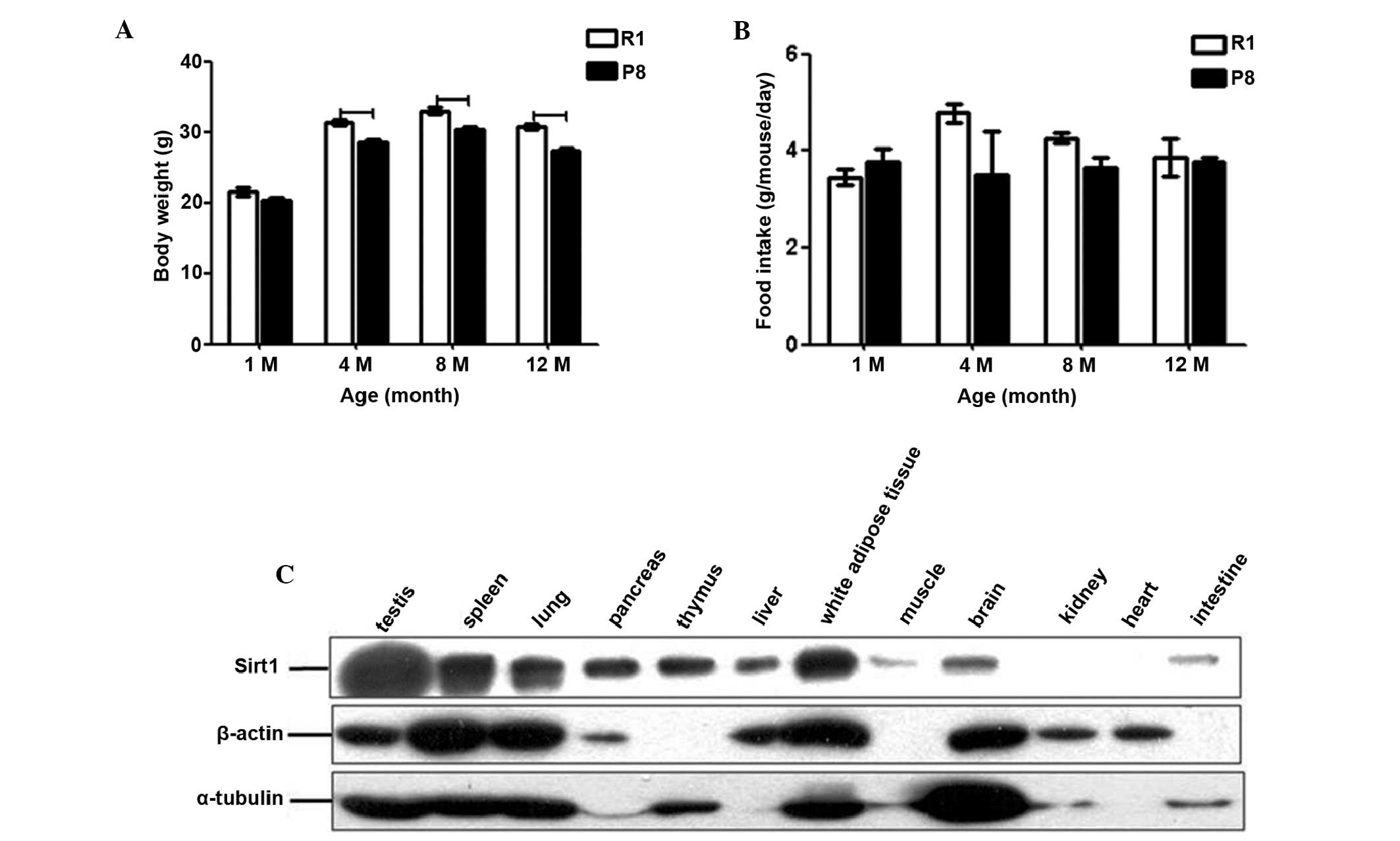 | Figure 1Basic information of SAM. The changes
in (A) body weight and (B) food intake of SAM-R1 and SAM-P8 with
age are expressed as the mean ± SEM. R1, SAM-R1; P8, SAM-P8; 1M,
4M, 8M and 12M, 1-, 4-, 8- and 12-month old groups, respectively.
(C) Western blot analysis of Sirt1 (upper panel), β-actin (middle
panel) and α-tubulin (lower panel) expression in 4-month old SAM-R1
tissues: testis, spleen, lung, pancreas, thymus, liver, white
adipose tissue, skeletal muscle, brain, kidney, heart and
intestine. The dash indicates the difference is significant between
the two groups (P<0.05). SAM, senescence-accelerated mouse;
SAM-R1, senescence-accelerated mouse resistant 1; SAM-P8,
senescence-accelerated mouse prone 8; Sirt1, sirtuin 1; SEM,
standard error of the mean. |
Sirt1 expression pattern in adult SAM-R1
tissues
To attempt to elucidate the expression pattern of
Sirt1 in different tissues of normal adult mice, the protein level
of Sirt1 in 12 tissues from 4-month old SAM-R1 rats were
investigated (Fig. 1C). Among
these 12 tissues, the testis, spleen, white adipose tissue and lung
had the highest expression level, while pancreas, thymus, liver and
brain had moderate expression levels, and in the skeletal muscle
and intestine, the signals were extremely weak. The expression of
Sirt1 in the kidney and heart was too low to be detectable.
Age-dependent changes in Sirt1 mRNA
expression level in different tissues of SAM
Then, the changes of Sirt1 expression patterns with
age were investigated in the different tissues, including the
liver, skeletal muscle, white adipose tissue and brain. These
tissues cover high (white adipose tissue), moderate (liver and
brain) and low (skeletal muscle) expression level in all of the 12
tissues scanned. Furthermore, the liver, white adipose tissue and
skeletal muscle belong to the metabolic system and regulate energy
storage and consumption, and the brain is part of the central
nervous system and thus secretes metabolism-regulating
hormones.
Sirt1 mRNA levels decreased with age in all four
tissues in both SAM-R1 and SAM-P8 (Fig. 2). The hypothesized maximal
difference was to be between SAM-R1 1-month and SAM-P8 12-month old
groups. Compared with the SAM-R1 1-month old group, the Sirt1 mRNAs
level in the SAM-P8 12-month old group reduced to 31.7% (P<0.01)
in the brain (Fig. 2A), 38.3%
(P<0.01) in the liver (Fig.
2B), 46.4% in skeletal muscle (Fig. 2C) and 38.3% (P=0.064) in white
adipose tissue (Fig. 2D). These
differences were enhanced compared with those observed between the
12-month and 1-month old groups within the same strain. In the
brain tissue compared with 1-month old mice, the Sirt1 mRNA level
of 12-month old mice decreased to 38.1% (P<0.01) in SAM-R1 and
38.3% (P<0.01) in SAM-P8 (Fig.
2A); in the liver, decreased to 52.0% (P<0.01) in SAM-R1 and
48.8% (P<0.01) in SAM-P8 (Fig.
2B); in skeletal muscle, decreased to 55.7% in SAM-R1 and 46.5%
in SAM-P8 (Fig. 2C) and in white
adipose tissue, decreased to 45.4% in SAM-R1 and 47.5% in SAM-P8
(Fig. 2D).
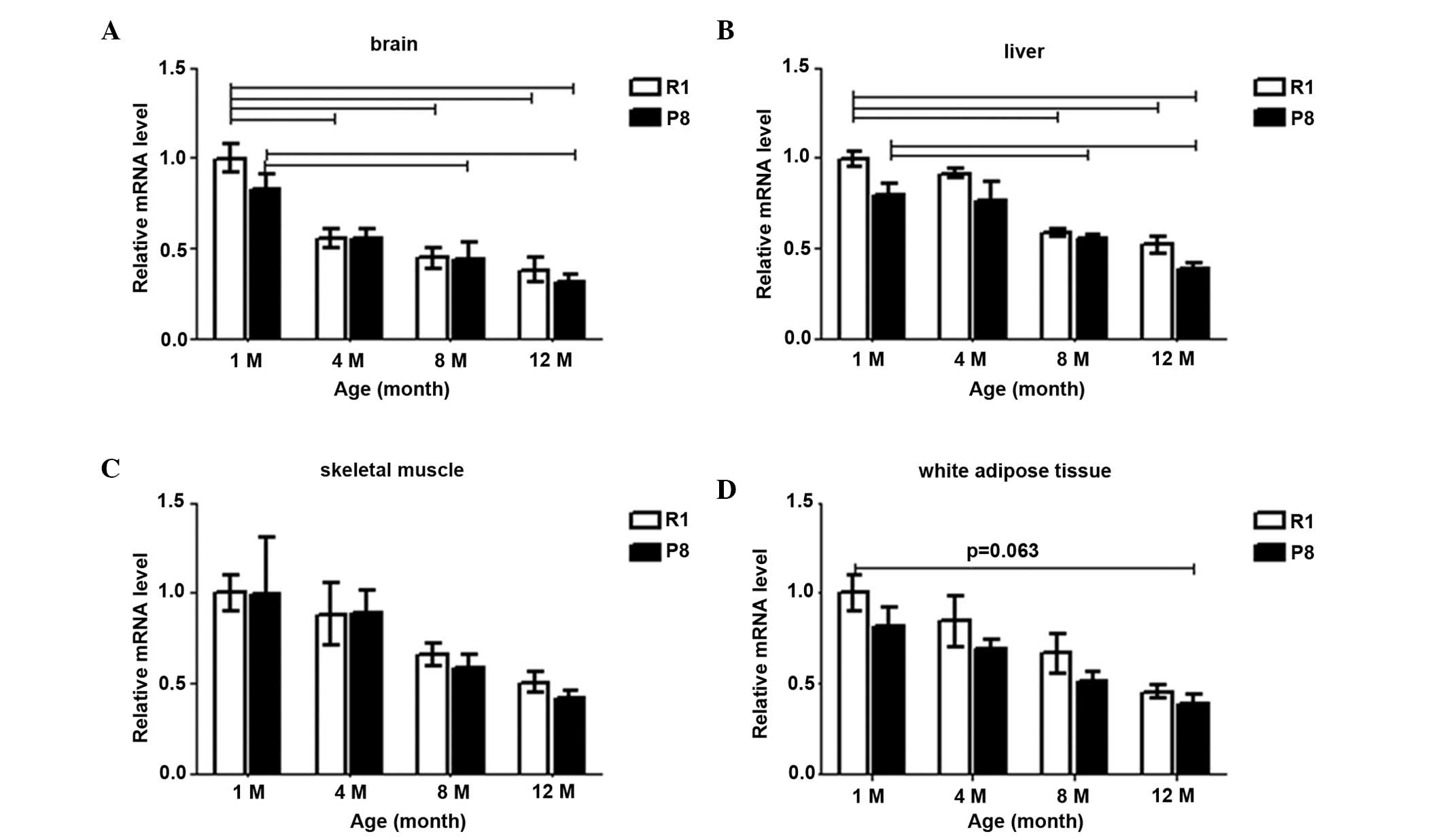 | Figure 2The changes of Sirt1 mRNA expression
patterns in SAM-R1 and SAM-P8 in (A) brain, (B) liver, (C) skeletal
muscle and (D) white adipose tissue with age. The values (mean ±
SEM) are expressed as a percentage relative to the SAM-R1
1-month-old group. n=3–6 animals/group. R1, SAM-R1; P8, SAM-P8; 1M,
4M, 8M and 12M, 1-, 4-, 8- and 12-month old groups, respectively.
The dash indicates the difference is significant between the two
groups (P<0.05). SAM, senescence-accelerated mouse; SAM-R1,
senescence-accelerated mouse resistant 1; SAM-P8,
senescence-accelerated mouse prone 8; Sirt1, sirtuin 1; SEM,
standard error of the mean. |
The majority of the Sirt1 mRNA expression in SAM-P8
was lower than that in the SAM-R1 strain at the same age. The
levels in SAM-P8 were 82.9, 98.9, 89.7 and 83.4% of that in the
SAM-R1 from 1- to 12-months old, respectively, in the brain
(Fig. 2A); 79.5, 80.1, 86.8 and
74.6% in the liver (Fig. 2B);
81.6, 82.1, 86.3 and 85.5% in white adipose tissue (Fig. 2D); and 89.1 and 83.3% of SAM-R1 at
8-months and 12-months old, respectively, in the skeletal muscle
(Fig. 2C).
In the brain tissues (Fig. 2A), in SAM-R1, the Sirt1 mRNA level
reduced to 55.8% (4-month; P<0.01), 45.0% (8-month; P<0.01)
and 38.1% (12-month; P<0.01) sequentially, compared with the
1-month old group; in SAM-P8, it reduced to 66.6% (4-month), 48.7%
(8-month; P<0.05) and 38.3% (12-month; P<0.01). In the liver
(Fig. 2B), compared with the
1-month old group, the Sirt1 mRNA level gradually declined to 95.6,
58.8% (P<0.01) and 52.0% (P<0.01) in SAM-R1, and to 96.3,
64.2% (P<0.01) and 48.8% (P<0.01) in SAM-P8 at 4-, 8- and
12-month old, respectively. The differences between 4-month old
group with older groups were also significant (P<0.01) in both
strains. In the skeletal muscle (Fig.
2C), the mRNA level also gradually declined, as compared with
the 1-month old group in both strains; in SAM-R1, to 88.4, 65.8 and
55.7% at 4-, 8- and 12-month old, respectively; and in SAM-P8, to
88.8, 58.8 and 46.5% at 4-, 8- and 12-month old, respectively. In
white adipose tissue (Fig. 2D),
compared with the 1-month old group, in SAM-R1, the expression
level decreased to 84.7% (4-month), 59.8% (8-month) and 45.4%
(12-month), sequentially; in SAM-P8, decreased to 85.2% (4-month),
63.2% (8-month) and 47.5% (12-month) sequentially.
These data indicated that the Sirt1 mRNA expression
level decreased with age in all four tissues in both strains, and
the most marked reduction was detected in the brain.
Age-associated changes in Sirt1 protein
expression level in different tissues of SAM
The Sirt1 protein level was also detected in the
tissues of the two strains at the various different ages. It also
decreased with age in all of these four tissues in both strains
(Fig. 3). With regard to the
supposed maximal difference, compared with the SAM-R1 1-month old
group, the Sirt1 protein level in the SAM-P8 12-month old group
reduced to 31.8% in the brain (Fig. 3A
and B), 65.6% in the liver (Fig.
3A and C), 65.9% in skeletal muscle (Fig. 3A and D) and 74.5% in white adipose
tissue (Fig. 3A and E). These
differences were not always increased compared with those between
1- and 12-month old within the same strain of all the tissues. In
the brain tissue, compared with the 1-month old mice, the Sirt1
protein level of the 12-month old group decreased to 54.4% in
SAM-R1 and 35.3% in SAM-P8 (Fig. 3A
and B). In the liver tissue, the expression decreased to 67.4%
in SAM-R1 and 80.4% in SAM-P8 (Fig. 3A
and C). In the skeletal muscle, it decreased to 74.4% in SAM-R1
and 65.4% in SAM-P8 (Fig. 3A and
D), and finally, in the white adipose tissue, the expression
decreased to 91.9% in SAM-R1 and 59.2% (P<0.05) in SAM-P8
(Fig. 3A and E).
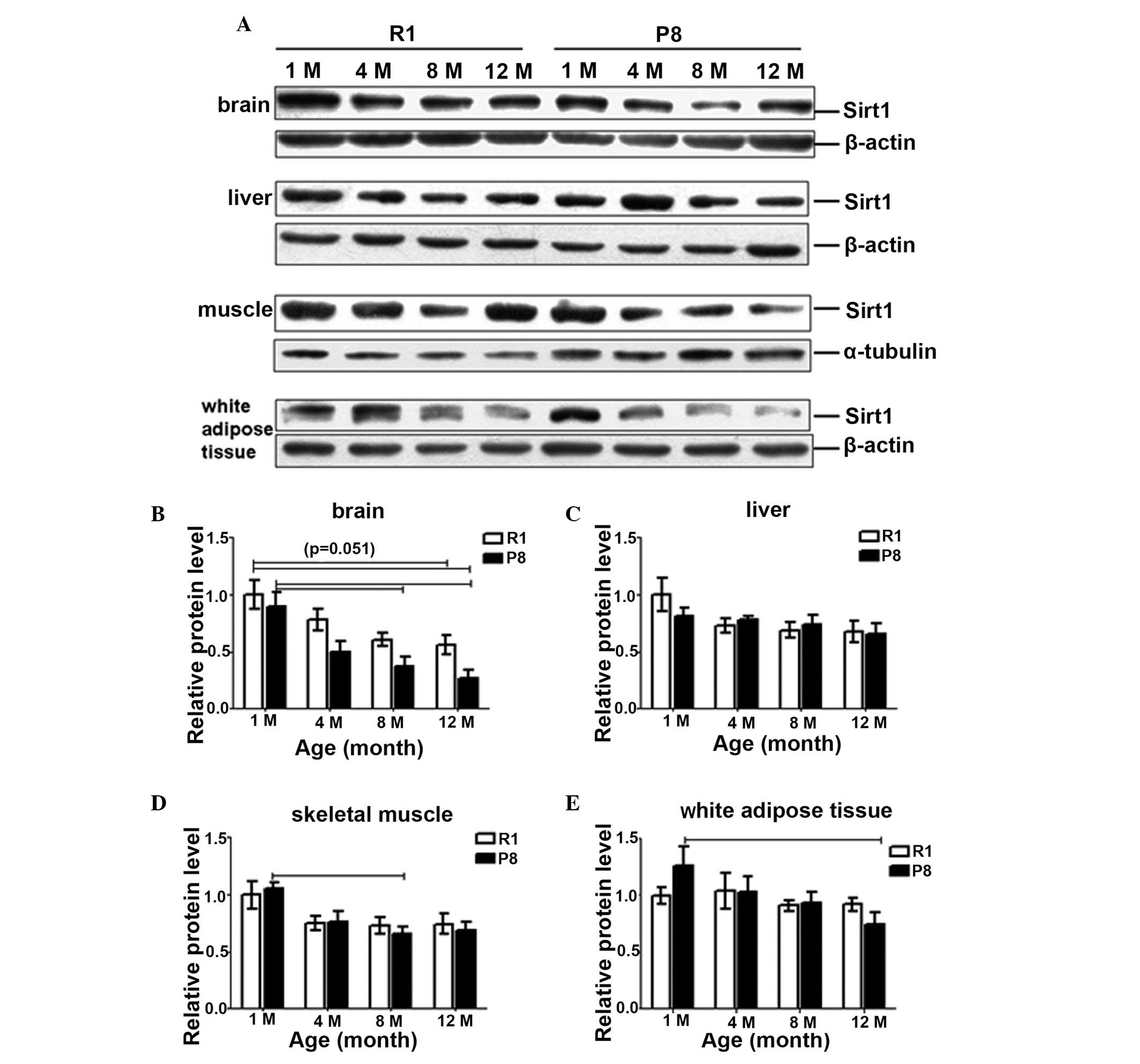 | Figure 3(A) Representative western blotting
images and quantitative densitometric analysis of the changes of
Sirt1 protein expression patterns in SAM-R1 and SAM-P in (B) brain,
(C) liver, (D) skeletal muscle and (E) white adipose tissue with
age. The values (mean ± SEM) are expressed as a percentage relative
to the SAM-R1 1-month-old group. n=3–6 animals/group. R1, SAM-R1;
P8, SAM-P8; 1M, 4M, 8M and 12M, 1-, 4-, 8- and 12-month old groups,
respectively. The dash indicates the difference is significant
between the two groups (P<0.05). SAM, senescence-accelerated
mouse; SAM-R1, senescence-accelerated mouse resistant 1; SAM-P8,
senescence-accelerated mouse prone 8; Sirt1, sirtuin 1; SEM,
standard error of the mean. |
As for the differences between SAM-P8 and SAM-R1 in
the brain at the same age (Fig. 3A and
B), the levels of SAM-P8 were lower than those of SAM-R1 at the
same age and the differences increased with age: 90.0, 64.7, 62.2
and 58.4% (vs. SAM-R1) from 1- to 12-month old, respectively. While
in the liver (Fig. 3A and C),
there were almost no marked differences between the two strains,
except that at 1-month old, the protein level of SAM-P8 was 18%
lower than that of SAM-R1, although this difference was not
significant. In the skeletal muscle (Fig. 3A and D), Sirt1 protein levels in
SAM-P8 were also lower than in SAM-R1 at 8-month (10%) and 12-month
old (7.6%). In white adipose tissue (Fig. 3A and E), Sirt1 protein level in
SAM-P8 was marginally higher (1.26-fold vs. SAM-R1) at 1-month old
and lower (0.81-fold vs. SAM-R1) at 12-month old.
In the brain (Fig. 3A
and B), the Sirt1 protein level declined evidently with age
from 1-month old in both strains and more so in SAM-P8 (35.3%) than
in SAM-R1 (54.4%): in SAM-R1, the protein expression reduced to
78.0, 60.7 and 54.4% (P=0.051) and in SAM-P8, it reduced to 56.1,
42.0% (P<0.05) and 35.3% (P<0.05) at 4-, 8- and 12-month old,
respectively. In the liver (Fig. 3A
and C), Sirt1 protein level in SAM-R1 reduced by ~30% at
1-month old, while were relatively stable and only reduced
moderately following 4-months: 72.9, 69.1 and 67.4% at 4-, 8- and
12-month old compared with 1-month old, respectively. In SAM-P8,
the protein levels also only decreased slightly to 95.3, 90.2 and
80.4% at 4-, 8- and 12-month old compared with 1-month old,
respectively. In the skeletal muscle (Fig. 3A and D), the change in Sirt1
protein level expression in SAM-R1 was similar to that in the
liver: reduced from 1- to 4-month old, then maintained relatively
stable: the levels were 75.1, 73.5 and 74.5% in 4-, 8- and 12-month
old group as at 1-month old group, respectively. While in SAM-P8,
the Sirt1 protein level declined gradually from 1- to 8-month old,
then remained stable from 8- to 12-month old: 71.9% (4-month),
62.8% (8-month, P<0.05) and 65.5% (12-month) compared with the
1-month old group. In the white adipose tissue (Fig. 3A and E), the changing trends were
weakly different from the above two tissues. In SAM-P8, the Sirt1
protein level gradually reduced to 81.7, 74.3 and 59.2% (P<0.05)
at 4-, 8- and 12-month old compared with 1-month old, respectively;
while in SAM-R1, it remained stable from 1- to 4-month (1.04-fold
vs. 1-month) and from 8-month (0.91-fold vs. 1-month) to 12-month
(0.92-fold vs. 1-month) and reduced slightly from 4- to 8-month old
(13.6% as 4-month).
These data demonstrate that the Sirt1 protein level
also decreased with age in all of the four tissues in both strains,
however the degree at which the expression changed was not as
marked as that observed at an mRNA level, except in the brain. The
protein expression was reduced most in the brain than in the other
three tissues. The differences between SAM-P8 and SAM-R1 at the
same age were not evident in the liver, muscle and white adipose
tissue, but the levels in SAM-P8 were lower than SAM-R1 in the
brain.
Discussion
Aging in most species is associated with impaired
adaptive and homeostatic mechanisms, leading to susceptibility to
environmental or internal stresses with increasing rates of disease
(14,15). Aging is a multifactorial process
characterized by a progressive decline in physiological function of
bodily organs (16). Sirt1 is
expressed throughout the body and has broad biological effects.
Sirt1 has been demonstrated to have a crucial role in mammalian
health and disease, and the regulation of metabolism, stress
responses, genome stability and aging. To clarify systematically
the natural changing pattern of Sirt1 expression may facilitate our
understanding of the natural aging process. It is possible that the
level of Sirt1 expression in each tissue may be a potential marker
that reflects the degree of physiological function decline in a
tissue with age.
In the present study, various different groups of
animals which covered multi-facet parameters were established: i)
age comparisons were selected between the young, maturing and old
mice (1-, 4-, 8- and 12-month old); ii) both senescence-accelerated
prone and resistant strains, SAM-P8 and SAM-R1, which have
different aging speeds were utilized; iii) four different tissues
were examined, including brain, liver, skeletal muscle and white
adipose tissue, which not only cover from low, moderate to high
expression levels in all the 12 tissues that were originally
scanned, but also include organs important in regulating metabolism
and central nervous functioning; iv) finally, both transcriptional
and translational Sirt1 expression levels were examined. These
parameters contributed to the reliability of this systematic
investigation of Sirt1 expression level with age.
In general, the results of the present study
demonstrated that Sirt1 expression decreased with age in all four
tissues investigated in both strains at a transcriptional and
translational level (Figs. 2 and
3). At a transcriptional level,
the age-dependent reduction in Sirt1 expression was more marked in
SAM-P8 than SAM-R1 in the liver and skeletal muscle, or similar to
SAM-R1 in white adipose tissue and brain. In all the four tissues,
the largest differences were evident between 1-month old SAM-R1 and
12-month old SAM-P8. At a translational level, the expression in
SAM-P8 decreased more than SAM-R1 in skeletal muscle, white adipose
tissue and brain. Furthermore, Sirt1 transcriptional level in
SAM-P8 was ~10–25% lower than in SAM-R1 at almost all the time
points in these tissues following maturation. The protein level in
SAM-P8 was also lower than in SAM-R1 at least at old age (12-month
old) in all the four tissues. Previously, Sasaki et al
(17) identified that the loss of
Sirt1 with age was accelerated in mice with accelerated aging but
was not observed in long-lived growth hormone receptor knockout
mice. In another study, diabetic animals demonstrated increased
cellular senescence in renal glomerulus and retinal blood vessels,
along with reduced Sirt1 mRNA expression in these tissues (18). Hyperglycemia accelerates
aging-associated processes in the vascular endothelial cells and it
appears such processes are mediated via the downregulation of Sirt1
(18). In the kidney, aging
markedly decreased Sirt1 protein level in 24-month old animals as
compared with 2- and 12-month old animals (16). These studies are in accordance with
our findings and these data suggested there is a parallel
correlation between Sirt1 expression level and the degree of aging,
and the loss of Sirt1 may be an important trigger for the aging
process.
Loss of Sirt1 with age has tissue specificity to a
certain degree, particularly at a protein level. From 1- to
12-month old, Sirt1 mRNA decreased gradually in the liver, skeletal
muscle and white adipose tissue to ~50% (45.4–55.7%), while in the
brain it reduced to <40%: 38.1% in SAM-P8 and 38.3% in SAM-R1.
At a protein level, Sirt1 also decreased gradually in the brain and
reduced more in SAM-P8 (to 35.3%) than in SAM-R1 (54.4%). However,
in the other three tissues, this effect was weaker than in the
brain, particularly following maturation. In the brain, Sirt1
protein levels of SAM-P8 were lower than those of SAM-R1 at all the
ages and the differences increased with age. These results
suggested that the brain was the most sensitive to changes in Sirt1
induced by age, particularly in the SAM-P8 model. Since the SAM
model was established in 1981 (12), the substrain SAM-P8 mice have
attracted notable attention in the gerontological research of
dementia. This model is characterized by a marked increase in
oxidative stress in the brain and the development of early learning
and memory deficits (between 8 and 10 months) together with other
characteristics similar to those observed in Alzheimer’s disease
(AD). Other characteristics of these mice include an altered
circadian rhythm, reduced anxiety behavior (19), immune dysfunction late in their
lifespan (20) and reduced life
expectancy (21,22). Sirt1 has been demonstrated to be
involved in all of the above functions, including circadian rhythms
(23,24), immune responses (25,26),
oxidative response (27) and AD
(28,29). As demonstrated in the results that
the Sirt1 expression level decreased more in SAM-P8 than in SAM-R1,
it is possible that the loss of Sirt1 in SAM-P8 has an important
role in the pathological characteristics of SAM-P8.
In conclusion, the present study systematically
investigated age-associated changes in Sirt1 expression pattern and
found that Sirt1 expression declined with age at the
transcriptional and translational levels in liver, skeletal muscle,
white adipose and brain tissues in two in vivo models of
aging, SAM-P8 and SAM-R1. It was demonstrated that the Sirt1 level
was lower in SAM-P8 than in SAM-R1, particularly at old age, and
the most sensitive organ to this reduction was the brain.
Acknowledgements
The present study was supported by the Research
Special Fund for Public Welfare Industry of Health to T.M.Z. (grant
no. 201302008) and the National Natural Science Foundation of China
to H.G. (grant no. 81300693)
References
|
1
|
Tissenbaum HA and Guarente L: Increased
dosage of a sir-2 gene extends lifespan in Caenorhabditis
elegans. Nature. 410:227–230. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Viswanathan M and Guarente L: Regulation
of Caenorhabditis elegans lifespan by sir-2.1 transgenes.
Nature. 477:E1–E2. 2011.
|
|
3
|
Donmez G and Guarente L: Aging and
disease: connections to sirtuins. Aging Cell. 9:285–290. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Guarente L: Franklin H. Epstein Lecture:
Sirtuins, aging, and medicine. N Engl J Med. 364:2235–2244. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Imai S, Armstrong CM, Kaeberlein M and
Guarente L: Transcriptional silencing and longevity protein Sir2 is
an NAD-dependent histone deacetylase. Nature. 403:795–800. 2000.
View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Yamamoto H, Schoonjans K and Auwerx J:
Sirtuin functions in health and disease. Mol Endocrinol.
21:1745–1755. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Banks AS, Kon N, Knight C, et al: SirT1
gain of function increases energy efficiency and prevents diabetes
in mice. Cell Metab. 8:333–341. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Bordone L, Cohen D, Robinson A, et al:
SIRT1 transgenic mice show phenotypes resembling calorie
restriction. Aging Cell. 6:759–767. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Herranz D, Muñoz-Martin M, Cañamero M, et
al: Sirt1 improves healthy ageing and protects from metabolic
syndrome-associated cancer. Nat Commun. 1:32010. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Cheng HL, Mostoslavsky R, Saito S, et al:
Developmental defects and p53 hyperacetylation in Sir2 homolog
(SIRT1)-deficient mice. Proc Natl Acad Sci USA. 100:10794–10799.
2003. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
McBurney MW, Yang X, Jardine K, Bieman M,
Th’ng J and Lemieux M: The absence of SIR2alpha protein has no
effect on global gene silencing in mouse embryonic stem cells. Mol
Cancer Res. 1:402–409. 2003.PubMed/NCBI
|
|
12
|
Takeda T, Hosokawa M, Takeshita S, et al:
A new murine model of accelerated senescence. Mech Ageing Dev.
17:183–194. 1981. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Flood JF and Morley JE: Learning and
memory in the SAMP8 mouse. Neurosci Biobehav Rev. 22:1–20. 1998.
View Article : Google Scholar
|
|
14
|
Campbell KH and O’Hare AM: Kidney disease
in the elderly: update on recent literature. Curr Opin Nephrol
Hypertens. 17:298–303. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
O’Hare AM, Bertenthal D, Covinsky KE, et
al: Mortality risk stratification in chronic kidney disease: one
size for all ages? J Am Soc Nephrol. 17:846–853. 2006.PubMed/NCBI
|
|
16
|
Lim JH, Kim EN, Kim MY, et al:
Age-associated molecular changes in the kidney in aged mice. Oxid
Med Cell Longev. 2012:1713832012.PubMed/NCBI
|
|
17
|
Sasaki T, Maier B, Bartke A and Scrable H:
Progressive loss of SIRT1 with cell cycle withdrawal. Aging Cell.
5:413–422. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Mortuza R, Chen S, Feng B, Sen S and
Chakrabarti S: High glucose induced alteration of SIRTs in
endothelial cells causes rapid aging in a p300 and FOXO regulated
pathway. PLoS One. 8:e545142013. View Article : Google Scholar
|
|
19
|
Miyamoto M: Characteristics of age-related
behavioral changes in senescence-accelerated mouse SAMP8 and
SAMP10. Exp Gerontol. 32:139–148. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Powers DC, Morley JE and Flood JF:
Age-related changes in LFA-1 expression, cell adhesion, and
PHA-induced proliferation by lymphocytes from
senescence-accelerated mouse (SAM)-P/8 and SAM-R/1 substrains. Cell
Immunol. 141:444–456. 1992. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Morley JE: The SAMP8 mouse: a model of
Alzheimer disease? Biogerontology. 3:57–60. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Morley JE, Armbrecht HJ, Farr SA and Kumar
VB: The senescence accelerated mouse (SAMP8) as a model for
oxidative stress and Alzheimer’s disease. Biochim Biophys Acta.
1822:650–656. 2012.
|
|
23
|
Asher G, Gatfield D, Stratmann M, et al:
SIRT1 regulates circadian clock gene expression through PER2
deacetylation. Cell. 134:317–328. 2008. View Article : Google Scholar
|
|
24
|
Belden WJ and Dunlap JC: SIRT1 is a
circadian deacetylase for core clock components. Cell. 134:212–214.
2008. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Salminen A, Kauppinen A, Suuronen T and
Kaarniranta K: SIRT1 longevity factor suppresses NF-kappaB -driven
immune responses: regulation of aging via NF-kappaB acetylation?
Bioessays. 30:939–942. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Sequeira J, Boily G, Bazinet S, et al:
sirt1-null mice develop an autoimmune-like condition. Exp Cell Res.
314:3069–3074. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Chong ZZ, Shang YC, Wang S and Maiese K:
SIRT1: new avenues of discovery for disorders of oxidative stress.
Expert Opin Ther Targets. 16:167–178. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Qin W, Yang T, Ho L, et al: Neuronal SIRT1
activation as a novel mechanism underlying the prevention of
Alzheimer disease amyloid neuropathology by calorie restriction. J
Biol Chem. 281:21745–21754. 2006. View Article : Google Scholar
|
|
29
|
Kim D, Nguyen MD, Dobbin MM, et al: SIRT1
deacetylase protects against neurodegeneration in models for
Alzheimer’s disease and amyotrophic lateral sclerosis. EMBO J.
26:3169–3179. 2007.
|

















