1. Introduction
The Nitraria genus, a member of the family
Zygophyllaceae, is one of the dominant species in the Mediterranean
and central Asian deserts. The English name Nitre-bush is from the
Latin word saltpetre, referring to the fact that it can
thrive in saline soil. It serves an key ecological role due to its
superior tolerance to severe drought and high salinity. Nitraria
are shrubs 25–200-cm tall, with spiny branches at the apex and
simple serrated leaves. The flowers are yellowish-gray or white,
2–4 cm pubescent, with five petals and five sepals, while the fruit
is a fleshy drupe. The Nitraria genus consists of 13 species across
the world, which are mainly distributed in Asia, Europe, Africa and
Australia (Table I).
 | Table ISpecies of the Nitraria genus and the
geographical distribution. |
Table I
Species of the Nitraria genus and the
geographical distribution.
| No. | Species | Geographical
distribution |
|---|
| 1 | N.
schoberi | China, Europe |
| 2 | N.
sibirica | China, Siberia |
| 3 | N.
senegalensis | Africa |
| 4 | N.
billardieri | Austraria |
| 5 | N.
retusa | Mediterranean
coast |
| 6 | N.
sphaerocarpa | Northwest
China |
| 7 | N.
roborowskii | Northwest China,
Russia |
| 8 | N.
komarovii | Europe caspian
coast |
| 9 | N.
tangutorum | Northwest
China |
| 10 | N.
praevisa | Northwest
China |
| 11 | N.
pamirica | China |
| 12 | N.
sinensis | China |
| 13 | N.
tridentate | Africa |
A growing body of evidence now demonstrates that
Nitraria extract has numerous biomedical properties, including
antifatigue, antitumor, anti-oxidative and antimutagenic
activities. In addition, the fruit of Nitraria sibirica is
extensively used to treat hypertension, menstrual disorders and
gastroenteritis in folklore medicines of Northwest China (1,2). The
leaves of Nitraria retusa also serve as supplement tea and
are used as a poultice in Africa (3,4).
To provide further support and evidence for the
clinical use of this genus, a systematic review of the modern
phytochemical and pharmacological properties of Nitraria was
performed. The available information on the pharmacology and
phytochemistry of the Nitraria genus was collected via libraries
and electronic searches using PubMed (http://www.ncbi.nim.nih.gov/pubmed), ScienceDirect
(http://sciencedirect.com), Springer (http://springer.com), EMBASE (http://elsevier.com/online-tools/embase) and CNKI
(http://www.cnki.net).
2. Phytochemical studies
Currently, the following six species of the Nitraria
genus have been observed for their phytochemical properties: N.
komarovii, N. schoberi, N. sibirica, N. tangutorum, N. retusa
and N. billardieri. These investigations suggested that
members of the Nitraria genus contain numerous components
categorized as alkaloids, flavonoids and phenolic acid, of which
alkaloids and flavonoids are the most abundant constituents. An
overview of ingredients contained in the Nitraria genus is
presented in Table II.
 | Table IICompounds isolated from the Nitraria
genus. |
Table II
Compounds isolated from the Nitraria
genus.
| A, Alkaloid
compounds |
|---|
|
|---|
| No. | Compound | Species | Part of plant | Reference |
|---|
| 1 | Nitraricine | N.
komarovii | Epigeal | 5 |
| 2 | Nitrarizine | N.
komarovii | Epigeal | 5 |
| 3 | Isokomarovine | N.
komarovii | Epigeal | 6 |
| 4 | Komarovidinine | N.
komarovii | Epigeal | 6 |
| 5 | Komarovinine | N.
komarovii | Epigeal | 7 |
| 6 | Peganine
N-oxide | N.
komarovii | Epigeal | 8 |
| 7 |
Nallylschoberine | N.
komarovii | Epigeal | 8 |
| 8 |
Dehydronitramidine | N.
komarovii | Epigeal | 8 |
| 9 | Komavicine | N.
komaroviii | Epigeal | 9 |
| 10 | Deoxypeganine | N.
komarovii | Epigeal | 10 |
| 11 | Nitraramine | N.
schoberi/N. komarovii | Epigeal | 11 |
| 12 | Komarovine | N.
komarovii | Epigeal | 12 |
| 13 | Komarovidine | N.
komarovii | Epigeal | 12 |
| 14 | Komaroine | N.
komarovii | Epigeal | 13 |
| 15 | Nitraraine | N.
schoberi | Epigeal | 14 |
| 16 | Nitrabirine | N.
sibirica | Epigeal | 14 |
| 17 | Nitramine | N.
sibirica | Epigeal | 15 |
| 18 | Isonitramine | N.
sibirica | Epigeal | 16 |
| 19 | Sibirine | N.
sibirica | Epigeal | 17 |
| 20 | Vasicinone
N-oxide | N.
komarovii | Epigeal | 18 |
| 21 | Deoxyvasicinone
N-oxide | N.
komarovii | Epigeal | 18 |
| 22 |
Dihydronitraraine | N.
komarovii | Epigeal | 19 |
| 23 |
Deoxyvasicinone | N.
komarovii | Epigeal | 20 |
| 24 | Vasicinone | N.
komarovii | Epigeal | 20 |
| 25 | Peganine | N.
komarovii | Epigeal | 20 |
| 26 | Deoxypeganine | N.
komarovii | Epigeal | 20 |
| 27 |
Dehydroschoberine | N.
komarovii | Epigeal | 20 |
| 28 | Nitraroxine | N.
komarovii | Epigeal | 20 |
| 29 | Tryptamine | N.
komarovii | Epigeal | 20 |
| 30 | Schoberine | N.
komarovii | Epigeal | 20 |
| 31 | Schoberidine | N.
komarovii | Epigeal | 21 |
| 32 | Nitrarine | N.
schoberi | Epigeal | 22 |
| 33 |
Tetramethylenetetrahydro-β-carboline
N-oxide | N.
komarovii | Epigeal | 23 |
| 34 | Sibirinine | N.
sibirica | Epigeal | 24 |
| 35 |
Dihydroschoberine | N.
sibirica | Aerial | 25 |
| 36 | Nitrabirine
N-oxide | N.
sibirica | Aerial | 25 |
| 37 |
O-acetylnitraraine | N.
schoberi | Aerial | 26 |
| 38 |
N-methylnitrarine | N.
schoberi | Aerial | 27 |
| 39 | Komavine | N.
komarovii | Aerial | 28 |
| 40 | Acetylkomavine | N.
komarovii | Aerial | 28 |
| 41 |
N-allylnitrarine | N.
komarovii | Aerial | 29 |
| 42 | Komarovidinine
N-oxide | N.
komarovii | Aerial | 29 |
| 43 | Sibiridine | N.
sibirica/N. schoberi | Aerial | 30 |
| 44 | Nitraramidine | N.
sibirica | Aerial | 31 |
| 45 | Nitraraidine | N.
sibirica | Aerial | 31 |
| 46 | Komarin | N.
komarovii | Aerial | 32 |
| 47 | Peganol
N-oxide | N.
komarovii | Aerial | 32 |
| 48 |
N-allylisonitrarine | N.
schoberi | Aerial | 33 |
| 49 | Nitraridine | N.
komarovii | Aerial | 34 |
| 50 |
Dihydronitraridine | N.
komarovii | Aerial | 34 |
| 51 |
Tetrahydronitraridine | N.
Komarovii | Aerial | 34 |
| 52 | Schobericine | N.
Schoberi | Aerial | 35 |
| 53 | Komaroidine | N.
Komarovii/N. Schoberi | Aerial | 35 |
| 54 |
Acetylkomaroidine | N.
Komarovii/N. Schoberi | Aerial | 35 |
| 55 |
Tetrahydronitramarine | N.
Komarovii. | Epigeal | 36 |
| 56 |
Tetrahydrokomarovinine | N.
Komarovii | Epigeal | 36 |
| 57 |
Dihydroisokomarovine | N.
Komarovii | Epigeal | 36 |
| 58 |
Tetrahydroisokomarovine | N.
Komarovii | Epigeal | 36 |
| 59 | Nazlinin | N.
Schoberi | Epigeal | 39 |
| 60 | Tangutorine | N.
Tangutorum | Leaves | 40 |
| 61 | Nitrarine | N.
Billardieri | Aerial | 41 |
| 62 |
1-Epinitraramine | N.
Billardieri | Aerial | 41 |
| 63 | 3-Epinitrarine | N.
Billardieri | Aerial | 41 |
| 64 |
5,7-Dihydroxy-3-deoxy-vasicine | N.
Retusa | Aerial | 42 |
| 65 |
7-Hydroxy-3-deoxy-1-vasiciene | N.
Retusa | Aerial | 42 |
| 66 | Allantoin | N.
Tangutorum | Seed | 48 |
|
| B, Flavanoid
compounds |
|
| No. | Compound | Species | Part of plant | Reference |
|
| 67 | Narcissin | N.
Komarovii | Leaves | 28 |
| 68 | Rutin | N.
Retusa | Leaves and
stems | 43 |
| 69 | Kaempferol | N.
Retusa | Leaves and
stems | 43 |
| 70 | Isorhamnetin
3-O-4rham-galactosylrobinobioside | N.
Retusa | Leaves and
stems | 44 |
| 71 | Isorhamnetin
3-robinobioside | N.
Retusa | Leaves and
stems | 44 |
| 72 | Isorhamnetin
3-rutinoside | N.
Retusa | Leaves and
stems | 44 |
| 73 | Isorhamnetin
3-galactoside | N.
Retusa | Leaves and
stems | 44 |
| 74 | Isorhamnetin
3-glucoside | N.
Retusa | Leaves and
stems | 44 |
| 75 | Isorhamnetin | N.
Retusa | Leaves and
stems | 44 |
| 76 | Isorhamnetin
3-xylosylrobinobioside | N.
Retusa | Leaves and
stems | 44 |
| 77 |
Isorhamnetin-7-O-α-L-rhamnoside | N.
Tangutorum | Seeds | 45 |
| 78 |
Isorhamnetin-7-O-β-D-glucoside | N.
Tangutorum | Seeds | 45 |
| 79 |
Kaempferol-7-O-α-L-rhamnoside | N.
Tangutorum | Seeds | 45 |
| 80 |
Quercetin-7-O-α-L-rhamnoside | N.
Tangutorum | Seeds | 45 |
| 81 | Quercetin | N.
Tangutorum | Seeds | 45 |
| 82 |
3,5-Dimethylether-kaempfrol-7-O-β-D-glucoside | N.
Tangutorum | Leaf | 46 |
| 83 |
3-Methylether-kaempferol-7-O-β-D-glucoside | N.
Tangutorum | Leaf | 46 |
| 84 |
Isorhamnetin-3-O-β-D-glucopyranosyl-(1–2)-α-L-rhamnopyranoside | N.
Tangutorum | Fruit | 47 |
| 85 |
5,7,2′-Trihydroxyflavonol | N.
Tangutorum | Fruit | 47 |
| 86 | Cyaniding
3-[6′-(6-trans-p-coumaroyl-β-D-glucopyranosyl)-β-D-galactopyranoside] | N.
Tangutorum | Fruit | 47 |
| 87 | Apigenin
5-O-(2′-O-E-P-coumaroyl)-β-D-glucopyranoside | N.
Tangutorum | Fruit | 47 |
Numerous studies (5–36)
have been conducted to explore the alkaloids contained in the
members of the Nitraria genus. Nearly all known alkaloids (Table II, nos. 1–36) identified in plants
of the Nitraria genus were initially isolated in these studies.
These studies also observed that the content of alkaloids in the
leaves is higher than the content in the roots, stems and seeds of
Nitraria komarovii and Nitraria sibirica (37–38).
There are also various types of alkaloids that have been isolated
and purified in other studies. In the crude MeOH extracts of N.
rhoberi, nazlinin was purified by Üstunes et al
(39). In a proceeding study,
tangutorine was isolated from the leaves of N. tangutorum
(40). Three novel alkaloids,
3-epinitrarine, 1-epinitraramine and nitrarine (41) were identified in N.
billardieri, which is a species that is mainly distributed in
Australia. Phytochemical studies of the aerial sections of N.
retusa reported the novel alkaloids 5,7-dihydroxy-3-deoxy
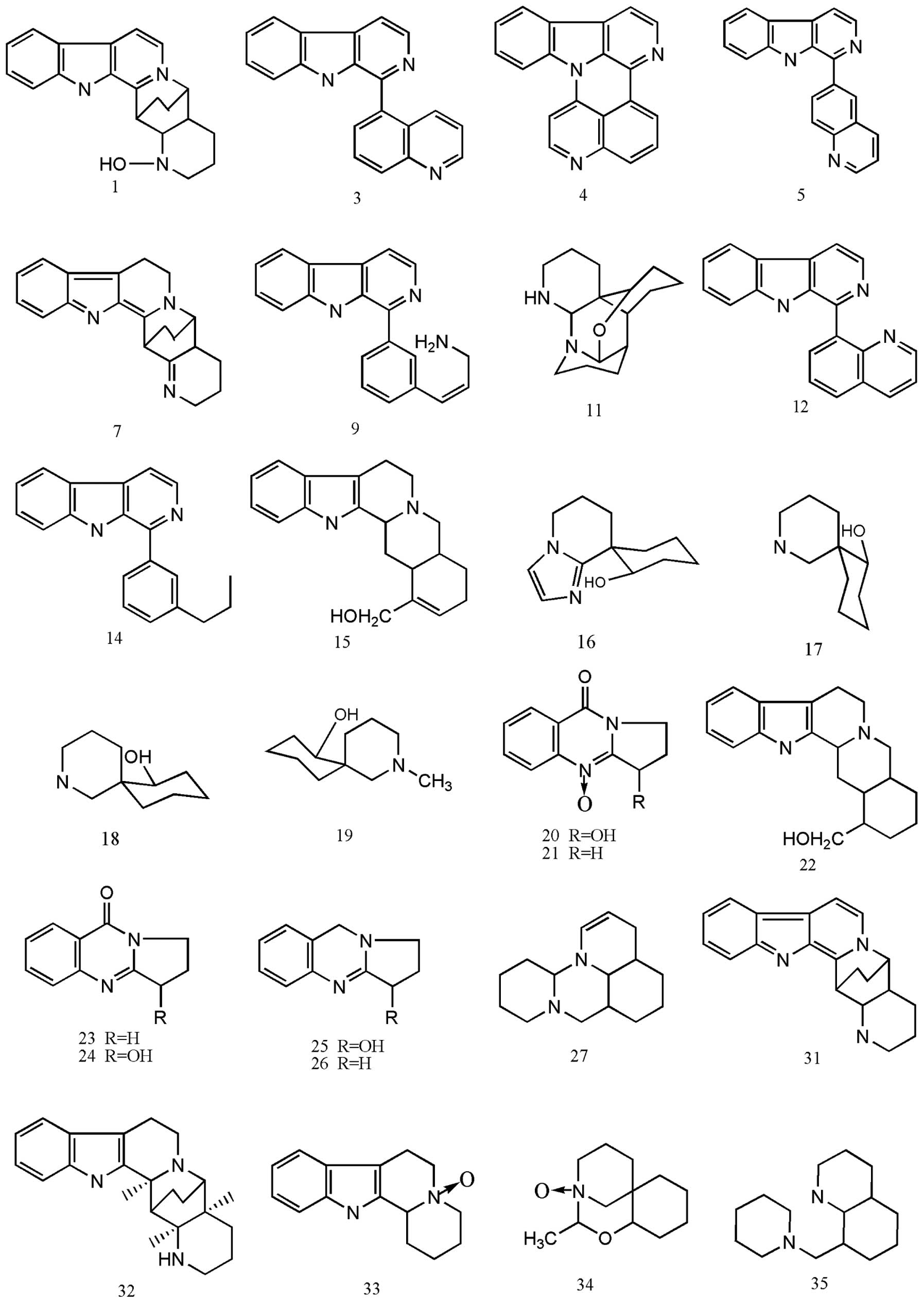
vasicine and 7-hydroxy-3-deoxy-1-vasiciene (42) (Fig.
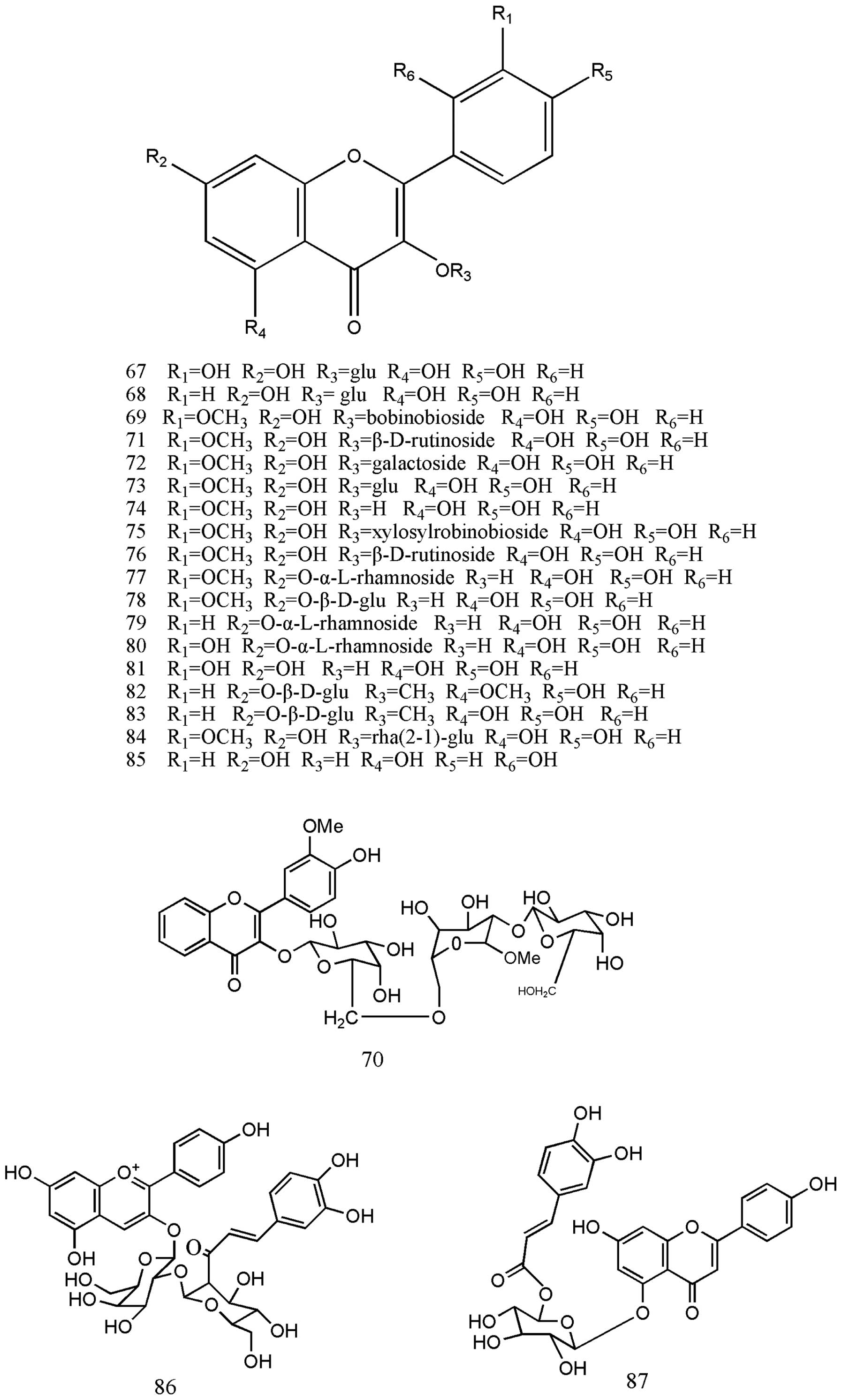
1). Nitraria was also reported by Saleh et al (43) to contain the flavonoids rutin,
kaempferol and isorhamnetin. The flavonoids isolated in other
studies are listed in Table II,
nos. 67–87 (44–48) (Fig.
2).
Wang et al (49) determined fatty acids in N.
tangutorum seed by supercritical carbon dioxide extraction and
high-performance liquid chromatography/atmospheric pressure
chemical ionization/mass spectrometry. Using gas chromatograph-mass
spectrometry, numerous volatile substances were detected in the
extract of the stem, leaves and fruits of N. tangutorum and
N. sibirica (50–52). Additionally, Wu et al
(53) isolated phenolic acid from
the water extraction of N. tangutorum.
3. Pharmacological effects
Antifatigue activity
The antifatigue activity of polysaccharides from the
fruits of N. tangutorum was assessed in mice using the
forced swim test (FST). The results demonstrated that the
FST-induced reductions in glucose, superoxide dismutase (SOD) and
glutathione peroxidase (GPx), and the increases in creatine
phosphokinase, lactic dehydrogenase, blood urea nitrogen,
triglyceride (TG) and malondialdehyde (MDA) levels, were inhibited
by the polysaccharides from N. tangutorum. Additionally, at
the same dosage, the extract of N. tangutorum is more potent
than Hippophae rhamnoides and Lycium ruthenicum,
which are traditionally used as medicinal foods with antifatigue
and antioxidant potential in Tibet (54). Fruit extracts of N.
tangutorum markedly prolonged the swimming time, climbing time
and survival time in low temperature of mice compared with the
control group in a study by Suo et al (55). The seed oil of N.
tangutorum, when extracted by supercritical CO2,
displayed similar effects as the fruit, and upregulated the
contents of serum urea-nitrogen and hepatic glycogen, but
downregulated the serum lactic acid contents, consequently
improving the swimming and climbing time. This result has been
repeated in other studies (56,57).
Antitumor activity
Boubaker et al investigated the apoptotic
potential of N. retusa ethyl acetate (EA) extract and
isorhamnetin 3-O-rutinoside (I3-O-R) isolated from
the ethyl acetate extract, in K562 human chronic myelogenous
erythroleukemia cells. After 48 h incubation with N. retusa
extract and I3-O-R, K562 cell viability was significantly
suppressed by inducing apoptosis, and the caspase 3 and caspase 8
activity was increased (58). The
extract and the component inhibited the genotoxicity induced by
hydroxyl radicals in K562 cells (59). Another similar study indicated that
EA extract of N. retusa and I3-O-R have a strong
antiproliferative effect on TK6 human lymphoblastoid cells,
possibly due to their involvement in the apoptotic pathway
(60).
The hexane (Hex), chloroform (Chl) and methanol
(MeOH) extracts of N. retusa were utilized to test their
antiproliferative effects on K562 cells. The Hex and Chl extracts
were demonstrated to induce stronger antiproliferative effects than
the MeOH extracts, by ameliorating the DNA fragmentation, poly
ADPribose polymerase cleavage, and caspase 3 and caspase 8 activity
(61). In another study,
3H-thymidine incorporation-induced proliferation of the HT29 human
colon cancer cells was reduced in a dose-dependent manner following
treatment with tangutorine, a β-carboline alkaloid from the leaves
of N. tangutorum. Tangutorine may induce p21 suppression of
all cyclins and their associated kinases, such as the topoisomerase
II, and thus inhibit normal DNA replication and mitosis (62). The activities of fractions/extracts
of N. retusa were compared with their flavanoid contents,
which consisted of the following four major flavonoids:
Isorhamnetin; isorhamnetin-3-O-glucoside (I3-O-G);
I3-O-R; and isorhamnetin-3-O-robinobioside
(I3-O-Rb). They inhibited the proliferation of Caco-2 cells
in vitro (63).
The total flavones from the N. tangutorum
fruits repress proliferation of the SGC-7901 human gastric
adenocarcinoma cell line and A-704 human kidney adenocarcinoma cell
line by regulating the levels of Ca2+, K+ and
P3+ in the cell (64–65).
The in vivo experiment demonstrated that flavones, in
addition to the aqueous extract of N. tangutorum, combined
with 5-fluorouracil, induced a significantly increased inhibitory
rate in the Hep human throat cancer cell line and U14 human
cervical cancer cell line by regulating the weight of immune
organs, the formation value of serolysin and phagocytic index
(66–67). Additionally, the lipids of N.
tangutorum were cytotoxic against the MGC-803 human gastric
carcinoma cell line (68).
Antioxidant activity
EA extract of N. retusa and I3-O-R
indicated a protective effect against lipid peroxidation induced by
H2O2. It demonstrated significant antioxidant
effects on oxidation induced by 2,2′-azobis (2-amidinopropane)
dihydrochloride in K562 cells with 50% inhibitory concentration
values (IC50) of 0.225 mg/ml and 0.31 mg/ml (59). Sterols, the main constituents of
Hex extract, and sterol and polyphenolic compounds, the main
constituents of Chl extract, may participate in the protective
effect against lipid peroxidation induced by
H2O2 in K562 cells (61). Chl extract exhibited the highest
ability to protect plasmid DNA against hydroxyl radical-induced DNA
damage, and the highest antioxidant capacity, with 0.95 mM trolox
equivalent capacity when tested using the ferric
reducing/antioxidant method (69).
Furthermore, the extract of fruits from N. tangutorum
displayed a significantly different antioxidant activity when
assessed with the 1,1-diphenyl-2-picrylhydrazy (DPPH),
2,2′-Azinobis (3-ethylbenzothiaz-oline-6-sulfonate) (ABTS) and
ferric-reducing antioxidant power assays (70).
The DPPH scavenging activity, xanthine oxidase
inhibition and superoxide scavenging activity of various N.
retusa extracts and compounds, including isorhamnetin,
I3-O-G, I3-O-R and I3-O-Rb, were evaluated to
confirm the association between the activities of the fractions and
their flavonoid contents. The EA extracts were identified to be
most effective at scavenging the DPPH stable free radical, and the
CHCl3 extracts exhibited the highest xanthine oxidase
inhibition activity; however, only BuOH extract exhibited a
scavenging activity toward superoxide radicals. Overall, all the
compounds exhibited some level of DPPH and superoxide scavenging
and xanthine oxidase inhibition activity, and the aglycone
compounds were more active than their glycosylated derivatives
(63). Antiradical activities
against DPPH, and β-carotene and Fe-reducing power were more
efficient in leaf non-polar fractions compared with polar fractions
of N. retusa (61). Similar
studies investigating anti-oxidant properties were also performed
by Bouaziz et al (71) and
demonstrated that the EA fraction and MeOH fraction of N.
retusa indicated DPPH scavenging activity and reduction of the
ABTS radical cation.
Anthocyanins have also been demonstrated to elicit
scavenging effects against O2, OH and DPPH in a
dose-dependent manner. Notably, these scavenging capacities were
greater than those of vitamin C according to results of in
vitro anti-oxidative tests (72). Nazlinin isolated from Nitraria and
its derivative 1-(4-butylamino)-3,4-dihydro-β-carboline have been
indicated to be inhibitors of pig kidney diamine oxidase, while
1-(4-butylamino)-β-carbolin was demonstrated as a substrate
(73).
Antimicrobial activity
The EA, ethanol and Chl extracts from the fruits of
N. tangutorum had antibacterial effects against
Escherichia coli, Bacillus subtilis and Staphyloccocus
aureus. The EA fraction presented the highest level of
antibacterial activity (74,75).
In addition, Chl extract from N. retusa leaves was more
efficient against all human pathogen strains, particularly
Escherichia coli and Staphylococcus aureus (76). The EA and MeOH extracts of N.
retusa revealed antimicrobial effects against Pseudomonas
aeruginosa and Aspergillus niger in vitro (71). The ethanol extracts of N.
retusa also exhibited cytotoxicity in brine shrimp with LC50
values of 6.2 μg/ml (77).
Antimutagenic activity
The protection of N. retusa against
mutagenicity induced by methyl methanesulfonate and
2-aminoanthracene in Salmonella typhimurium TA102 and TA104
strains was observed. The highest protection was elicited by Chl
and MeOH extracts of N. retusa, with inhibition percentages
of 44.93% at 50 μg/plate in the presence of TA102 strain and 38% at
10 μg/plate in the presence of TA104 strain. Hex and Chl extracts
have been demonstrated to reduce the mutagenicity induced by
2-aminoanthracene with 83.4% in TA104 and 65.3% in the TA102 strain
(69).
Hypotensive effects
Senejouxa et al conducted a study on the
vasorelaxant activity and underlying mechanisms of hydroalcoholic
extract from the fruits of N. sibirica on thoracic aortic
rings isolated from Wistar rats. The study revealed that the
hydroalcoholic extract was more effective in the induction of
vasodilation of phenylephrine- than high KCl-pre-contracted aortic
rings with respective Emax values of 82.9±2.2 and
34.8±3.6%. The acute intravenous injection of hydroalcoholic
extract induced an immediate and transient hypotensive effect in
anesthetized spontaneously hypertensive and control rats through an
endothelium-dependent pathway involving nitric oxide synthase (NOS)
activation, endothelium-derived hyperpolarizing factor production
and muscarinic receptor stimulation (78). Flavonoids of different
concentrations increased the repair of impaired human umbilical
vein endothelial cells induced by high glucose or
H2O2 by increasing the ratio of NOS, SOD and
GPx activity, and NO level (79,80).
Notably, the inhibition activity of ethanol extracts from 10
halophytes on angiotensin converting enzyme (ACE) has been
investigated extensively. The Nitraria sibirica fraction has
been demonstrated to significantly inhibit ACE with an IC50 value
of 69.36 g/l (81).
Hypoglycemic effects
The fruit of N. tangutorum at a dosage of 1.8
g/kg and 3.6 g/kg not only had a therapeutic action in a mouse
diabetes model induced by alloxan, but also led to an increase in
the glucose-toleration in similar models in rats. It has also been
demonstrated to reduce glucose levels in hyperglycemic animal
models induced by epinephrine and glucose (82). Shabana et al (83) investigated the hypoglycemic
activity of 31 desert plants from different Egyptian localities in
normal fasting and alloxanised rats, and Nitraria retusa had
hypoglycemic effects in normal fasting rats.
Lipid lowering effects
An in vivo study on the effects of the fruit
extracts of N. tangutorum on rat and mouse models of
hyperlipermia induced by high lipid levels was conducted by Suo
et al. N. tangutorum significantly reduced the serum level
of total cholesterol (TC) and TG in a rat hyperlipermia model, and
the level of low-density lipoprotein (LDL) in Kunming strain rats.
It also increased the ratio of high-density lipoprotein cholesterol
(HDL)/TC, HDL/LDL and SOD activity, and reduced the MDA content
in vivo and in vitro (84,85).
Hepatoprotective effects
A study by Zhang et al (86) indicated that seed oil of N.
tangutorum alleviated the increased levels of aspartate
aminotransferase, alanine aminotransferase and MDA induced by CCl4,
and it also enhanced the level of GPx in liver.
4. Conclusion
The studies summarized above strongly support the
theory that the Nitraria genus has favorable therapeutic
properties, indicating its potential for clinical use. The present
review presents and assesses the previous pharmacological and
phytochemical studies published on the Nitraria genus, and may aid
the easy identification and further research into properties of
members of the Nitraria genus.
Nitraria as a halophyte, is ecologically central in
stabilizing wind-blown sand and loess soils and thus reduces
erosion. Hence, combined with the pharmacological effects, the
rational development and utilization of Nitraria may be beneficial
for the local environment and public health.
Numerous alkaloids have been isolated from the
Nitraria genus. However, there is currently no research on the
pharmacological properties of the alkaloid components, which are
the most abundant constituents in nature. Further studies on the
antitumor and anti-oxidative activities of these components are
required. Furthermore, few molecular mechanisms are known, which
may hamper the further clinical application of Nitraria. The
possible synergistic action among the bioactive compounds of the
plants must be evaluated prior to their use in clinical
practice.
Acknowledgements
The current study was supported by grants from the
National Natural Science Foundation of China (grant no.
U1203104).
References
|
1
|
Liu YX: Desert Flora in China. 2. Science
Press; Beijing China: pp. 3061988
|
|
2
|
Zhengyi Wu, Raven H and Deyuan H: Flora of
China. 43(1)Science Press; Beijing, China: pp. 120–121. 1988
|
|
3
|
Tackholm V: Students Flora of Egypt. Cairo
University; 31. pp. 313pp. 8681974
|
|
4
|
Jafri S and El-Gadi A: Flora of Libya.
Department of Botany, Al Fateh University; pp. 551977
|
|
5
|
Tulyaganov TS and Abdullaev ND: Alkaloids
of Nitraria komarovii Nitraricine and Nitrarizine. Chem Nat
Compd. 3:76–78. 1995.
|
|
6
|
Tulyaganov TS, Ibragimov AA and Yunusov
SY: Alkaloids of Nitraria komarovii. VI. structure and
synthesis of isokomarovine and of komarovidinine. Chem Nat Compd.
18:601–603. 1982.
|
|
7
|
Tulyaganov TS, Ibragimov AA and Yunusov
SY: Alkaloids of Nitraria komarovii. Vll. Structure and
synthesis of komarovinine. Chem Nat Compd. 18:604–606. 1982.
|
|
8
|
Tulyaganov TS: Alkaloids of Nitraria
komarovii XVII. Peganine N-oxide, N-allylschoberine and
dehydronitramidine. Chem Nat Compd. 30:727–729. 1994.
|
|
9
|
Tulyaganov TS: Alkaloids of Nitraria
komarovii. XI. Structure of komavicine and the products of the
dehydrogenation of the nitrarine method of dehydrogenation in the
quinolinyl-β-carboline series. Chem Nat Compd. 29:26–30. 1993.
|
|
10
|
Tulyaganov TS: Alkaloids of Nitraria
komarovii. XIV. Deoxypeganine N-oxide. Chem Nat Compd.
29:731993.
|
|
11
|
Ibragimov AA and Yunusov SY: Alkaloids of
Nitraria schoberi. Ring-chain tautomerism of the hydrolysis
product of nitraramine. Chem Nat Compd. 22:680–684. 1986.
|
|
12
|
Tulyaganov TS, Ibragimov AA and Yunusov
SY: Alkaloids of Nitraria komarovii. IV. Total synthesis of
komarovine and komarovidine. Chem Nat Compd. 17:149–152. 1981.
|
|
13
|
Tulyaganov TS, Ibragimov AA and Yunusov
SY: Alkaloids of Nitraria komarovii. IX. Structure of
komaroine. Chem Nat Compd. 20:378–379. 1984.
|
|
14
|
Ibragimov AA and Yunusov SY: Alkaloids of
Nitraria schoberi. Structure of nitraraine. Chem Nat Compd.
21:502–509. 1985.
|
|
15
|
Novgorodova NY, Maekh SK and Yunusov SY:
Nitramine-A new alkaloid from Nitraria schoberi. Chem Nat
Compd. 11:455–456. 1975. View Article : Google Scholar
|
|
16
|
Osmanov Z, Ibragimov AA and Yunusov SY:
Isonitramine-A new alkaloid from Nitraria sibirica. Chem Nat
Compd. 13:607–608. 1977. View Article : Google Scholar
|
|
17
|
Osmanov Z, Ibragimov AA and Yunusov SY:
Structure of Sibirine. Chem Nat Compd. 18:206–208. 1982. View Article : Google Scholar
|
|
18
|
Tulyaganov TS, Atadzhanov RS, Abdullaev
ND, et al: Alkloids of Nitraria komarovii. XV. Vasicinone
and deoxyvasicinone N-oxides. Chem Nat Compd. 29:509–511. 1993.
|
|
19
|
Ibragimov AA and Yunusov SY: Alkaloids of
Nitraria schoberi. Structure of dihydronitraraine. Chem Nat
Compd. 21:510–512. 1985.
|
|
20
|
Tulyaganov TS: Alkaloids of Nitraria
komarovii. XIII. Nitraramine n-oxide and the structure of
dehydroschoberine. Chem Nat Compd. 29:31–34. 1993.
|
|
21
|
Ibragimov AA, Maekh SK and Yunusov SY: The
structure of schoberine. Chem Nat Compd. 11:2971975. View Article : Google Scholar
|
|
22
|
Normatov M and Yunusov SY: A study of the
alakloids of Nitraria Schoberi. The structure of nitrarine.
Chem Nat Compd. 4:1201968.
|
|
23
|
Tulyaganov TS and Shorakhimov NN:
Alkaloids of Nitraria komarovii XII.
Tetramethylenetetrahydro-β-carboline N-oxide. Chem Nat Compd.
26:478–479. 1990.
|
|
24
|
Ibragimov AA, Abdullaev ND, Osmanov Z, et
al: Alkaloids of Nitraria sibirica. Structure and absolute
configuration of sibirinine. Chem Nat Compd. 23:569–573. 1987.
|
|
25
|
Tulyaganov TS and Alaberdiev FK: Alkaloids
of Nitraria sibirica. Dihydroschoberine and nitrabirine
N-oxide. Chem Nat Compd. 37:556–557. 2007. View Article : Google Scholar
|
|
26
|
Tulyaganov TS and Kozimova NM: Alkaloids
from Nitraria schoberi. O-acetylnitraraine. Chem Nat Compd.
41:578–579. 2005.
|
|
27
|
Tulyaganov TS and Nazarov OM: Alkaloids of
Nitraria schoberi. N-methylnitrarine. Chem Nat Compd.
36:393–394. 2000. View Article : Google Scholar
|
|
28
|
Tulyaganov TS, Nazarov OM, Levkovich MG,
et al: Alkaloids of the Nitraria genus. Komavine and
acetylkomavine. Chem Nat Compd. 37:61–64. 2001. View Article : Google Scholar
|
|
29
|
Tulyaganov TS and Makhmudov OE: Alkaloids
of Nitraria komarovii. N-allylnintrarine and komarovidinine
N-oxide. Chem Nat Compd. 36:396–398. 2000. View Article : Google Scholar
|
|
30
|
Tulyaganov TS and Allaberdiev FK:
Alkaloids from plants of the Nitraria genus. Structure of
sibiridine. Chem Nat Compd. 39:292–293. 2003. View Article : Google Scholar
|
|
31
|
Tulyaganov TS and Allaberdiev FK:
Alkaloids of Nitraria sibirica. Structures of nitraramidine
and nitraraidine. Chem Nat Compd. 38:602–604. 2002. View Article : Google Scholar
|
|
32
|
Tulyaganov TS and Makhmudov OE: Alkaloids
of Nitraria komarovii. Structures of komarin and
peganol-N-oxide. Chem Nat Compd. 36:76–78. 2000.
|
|
33
|
Tulyaganov TS, Nazarov OM, Makhmudov OE,
et al: N-allylisonitratine and narcissin from plants of the
Nitraria genus. Chem Nat Compd. 37:470–473. 2001. View Article : Google Scholar
|
|
34
|
Tulyaganov TS: Alkaloids from Nitraria
komarovii. Structures of nitraridine, dihydronitraridine and
tetrahydronitraridine. Chem Nat Compd. 42:459–461. 2006. View Article : Google Scholar
|
|
35
|
Tulyaganov TS, Kozimova NM and Allaberdiev
FK: Alkaloids from plants of the genus Nitraria. Chem Nat Compd.
42:198–200. 2006. View Article : Google Scholar
|
|
36
|
Tulyaganov TS and Yunusov SY: Alkaloids of
Nitraria komarovii. X Structures of tetrahydronitramarine,
tetrahydrokomarovinine, dihydroisokomarovine, and
tetrahydroisokomarovine. Chem Nat Compd. 26:49–53. 1990.
|
|
37
|
Tulyaganov TS, Ibragimov AA and Yunusov
SY: Alkaloids of Nitraria komarovii. I. Chem Nat Compd.
15:6541979.
|
|
38
|
Osmanov Z, Ibragimov AA, Yunusov SY, et
al: Dynamics of the accumulation of the alkaloids of Nitraria
sibirica. Chem Nat Compd. 18:3721982. View Article : Google Scholar
|
|
39
|
Üstunes L, Özer A, Laekeman GM, et al:
Chemical characterization and pharmacological activity of nazlinin,
a novel indole alkaloid from Nitraria schoberi. J Nat Prod.
54:959–966. 1991.PubMed/NCBI
|
|
40
|
Duan JA, Williams ID, Che CT, et al:
Tangutorine: A novel β-carboline alkaloid from Nitraria
tangutorum. Tetrahedron Lett. 40:2593–2596. 1999.
|
|
41
|
Shen MY, Zuanazzi JA, Kan C, et al:
Alkaloids from Nitraria billardieri. Nat Prod Lett.
6:119–125. 1995. View Article : Google Scholar
|
|
42
|
El-Alali A, Zoubi AA, Gharaibeh M, et al:
Phytochemical and biological investigation of Nitraria
retusa. Asch. Jor J Pharm Sci. 5:155–163. 2012.
|
|
43
|
Saleh NAM and El-Hadidi MH: An approach to
the chemosystematics of the Zygophyllaceae. Biochem Syst Ecol.
5:121–128. 1977. View Article : Google Scholar
|
|
44
|
Halim AF, Saad HE and Hashish NE: Flavonol
glycosides from Nitraria retusa. Phytochemistry. 40:349–351.
1995. View Article : Google Scholar
|
|
45
|
Jia ZJ, Zhu GJ and Wang JH: Flavonoid
constituents from the seeds of Nitraria tangutorum Bolor.
Acta Botanica Sinica. 31:241–243. 1989.
|
|
46
|
Duan JA, Zhou RH and Zhao SX: The chemical
constituents of flavonoids and phenolic acid compounds of leaves
from Nitraria tangutorum Bor. J Plant Resour Environ. 8:6–9.
1999.
|
|
47
|
Lu HL, Ouyang F, Su YL, et al: Study on
chemical constituents of the fruits of Nitraria tangutorum
Bobr. Northwest Pharm J. 28:221–223. 2013.
|
|
48
|
Wang HL, Li YL, Wang XY, et al: Chemical
constituents of Nitraira tangutorum seed from Qaidam basin.
Nat Prod Res Dev. 19:614–616. 2007.
|
|
49
|
Wang HL, Suo YR, Wang XY, et al:
Extraction of Nitraria tangutorum seed oil by supercritical
carbon dioxide and determination of free fatty acids by
HPLC/APCI/MS with fluorescence detection. Sep Purif Technol.
56:371–377. 2007.
|
|
50
|
Wang JM and Kang WY: Volatiles from the
leaves of Nitraria tangutorum and Nitraria sibirica.
Nat Prod Res Dev. 23:680–683. 2011.
|
|
51
|
Wang JM and Kang WY: The volatiles in the
stem of Nitraria tangutorum and Nitraria sibirica.
Fine Chem. 26:773–775. 2009.
|
|
52
|
Zhu Y, Liu JR, Wang HY, et al: Comparison
and analysis on fruit oils of Nitraria prepared by
supercritical CO2, fluid extraction and impregnation
method. Food Fermentation Ind. 32:84–86. 2006.
|
|
53
|
Wu ZB, Li ML, Hang M, et al: Study on
water-soluble chemical constituents of Nitraria tangutorum
Bobr. J Shihezi Univ (Nat sci). 30:728–730. 2012.
|
|
54
|
Ni W, Gao T, Wang H, et al: Anti-fatigue
activity of polysaccharides from the fruits of four Tibetan plateau
indigenous medicinal plants. J Ethnopharmacol. 150:529–535. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
55
|
Suo YR, Li YL, Wang HL, et al: Effect of
fruit of Nitraria tangutorum Bobr. from Qaidam basin on
immunomodulatory, anti-fatigue and cold-tolerance. Nat Prod Res
Dev. 17:717–721. 2005.
|
|
56
|
Suo YR, Wang HL, Li YL, et al: The
anti-fatigue effect of Nitraria tangutorum Bobr. Seed oil
from Qaidam basin on the mice. Nat Prod Res Dev. 18:88–91.
2006.
|
|
57
|
Ma TJ and Xu YQ: The anti-fatigue effect
of Nitraria seed oil on mice. Acta Lab Anim Sci Sin.
20:77–79. 2012.
|
|
58
|
Boubaker J, Bhouri W, Sghaier MB, et al:
Ethyl acetate extract and its major constituent, isorhamnetin
3-O-rutinoside, from Nitraria retusa leaves, promote
apoptosis of human myelogenous erythroleukaemia cells. Cell Prolif.
44:453–461. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
59
|
Boubaker J, Ben Sghaier M, Skandrani I, et
al: Isorhamnetin 3-O-robinobioside from Nitraria retusa
leaves enhance antioxidant and antigenotoxic activity in human
chronic myelogenous leukemia cell line K562. BMC Complem Altern
Med. 12:1352012.
|
|
60
|
Boubaker J, Bhouri W, Ben Sghaier M, et
al: Flavonoids products from Nitraria retusa leaves promote
lymphoblastoid cells apoptosis. Nutr Cancer. 64:1095–1102.
2012.
|
|
61
|
Boubaker J, Bhouri W, Sghaier MB, et al:
Leaf extracts from Nitraria retusa promote cell population
growth of human cancer cells by inducing apoptosis. Cancer Cell
Int. 11:372011.
|
|
62
|
Liu BPL, Chong EYY, Cheung FWK, et al:
Tangutorine induces p21 expression and abnormal mitosis in human
colon cancer HT-29 cells. Biochem Pharmacol. 70:287–299. 2005.
View Article : Google Scholar : PubMed/NCBI
|
|
63
|
Salem JH, Chevalot I, Harscoat-Schiavo C,
et al: Biological activities of flavonoids from Nitraria
retusa (Forssk.) Asch and their acylated derivatives. Food
Chem. 124:486–494. 2011.
|
|
64
|
Liu CS, Wu XG, Xie MR, et al: Inhibition
effects of Nitraria tangutorum flavone on SGC-7901 cell
proliferation. J Gansu Sci. 21:66–68. 2009.
|
|
65
|
Yu FR, Lian SJ and Lian XZ: Cytotoxic and
proliferous effects of Nitraria tangutorum flavone on human
A-704 strain cells. Chin Pharmacol Bull. 24:236–239. 2008.
|
|
66
|
Yu FR, Wei KQ and Lian XZ: Effect and
mechanism of nitrary flavone on Hep, U14 tumor of mice. Chin
Pharmacol Bull. 19:886–888. 2003.
|
|
67
|
Wei KQ, Yu FR and Lian XZ: Effect of
nitrary decotion on rehabilitation of Hep tumor and reinforcement
of immunological function. J Clin Rehab Tissue Eng. 7:2418–2419.
2003.
|
|
68
|
Li YP, Sun LP, Wang SZ, et al: Inhibition
effects of shayingtao lipids on the human gastric carcinoma MGC-803
cells. Chin J Clin Res. 26:108–109. 2013.
|
|
69
|
Boubaker J, Skandrani I, Bouhlel I, et al:
Mutagenic, antimutagenic and antioxidant potency of leaf extracts
from Nitraria retusa. Food Chem Toxicol. 48:2283–2290. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
70
|
Zheng J, Li H, Ding CX, et al:
Anthocyanins composition and antioxidant activity of two major wild
Nitraria tangutorun Bobr. variations from Qinghai-Tibet
Plateau. Food Res Int. 44:2041–2046. 2011. View Article : Google Scholar
|
|
71
|
Bouaziz M, Dhouib A, Loukil S, et al:
Polyphenols content, antioxidant and antimicrobial activities of
extracts of some wild plants collected from the south of Tunisia.
Afric J Biotechn. 8:7017–7027. 2009.
|
|
72
|
Bai XM, Ding CX, Guo YQ, et al: Free
radical scavenging effects of anthocyanins from the fruits of
Nitraria tangutorun. Nat Prod Res Dev. 20:884–887. 2008.
|
|
73
|
Cheng E, Dekker HL, van Gelder BF, et al:
Inhibition of pig kidney diamine oxidase by nazlinin and nazlinin
derivatives. Biochim Biophys Acta. 1253:189–192. 1995. View Article : Google Scholar : PubMed/NCBI
|
|
74
|
Zhang GX, Qi JH, Ren X, et al: In vitro
antimicrobial activity of extracts from fruits of Nitraria
sibirica pall. Sci Tech Food Ind. 13:104–106. 2012.
|
|
75
|
Li J, Wu XL, Chen Y, et al: Antidiarrheal
properties of different extracts of Chinese herbal medicine formula
Bao-Xie-Ning. J Integr Med. 11:125–134. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
76
|
Zaouali Y, Ksouri WM, Saada M, et al:
Solvent effects on antioxidants and biological activities of the
halophyte Nitraria retusa (Forssk.). Asch Planta Med.
77:PM2032011.
|
|
77
|
Tawaha KA: Cytotoxicity evaluation of
Jordanian wild plants using brine shrimp lethality test. Jor J App
Sci-Nat Sci. 8:12–17. 2006.
|
|
78
|
Senejouxa F, Girarda C, Aisab HA, et al:
Vasorelaxant and hypotensive effects of a hydroalcoholic extract
from the fruits of Nitraria sibirica Pall. (Nitrariaceae). J
Ethnopharmacol. 141:629–634. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
79
|
Fan LL, Tang H, Zhang YJ, et al: The
protection of total flavonoids of Nitraria tangutorun Bobr.
on vascular endothelial cells which was injured by high glucose.
Pharm Clin Chin Mater Med. 26:21–23. 2010.(In Chinese).
|
|
80
|
Suo YR, Gao H and Wang HL: The protective
effect of Nitraria tangutorum Bobr. seed oil from Qaidam
basin on the liver injury of mice. Nat Prod Res Dev. 17:573–576.
2005.
|
|
81
|
Liu Y, Tan CY, Wu D, et al: Study on
inhibition activity of angiotensin I-converting enzyme (ACE) on 10
halophytes. Chin Pharm J. 45:736–738. 2010.(In Chinese).
|
|
82
|
Suo YR and Wang HQ: Studies on
hypoglycemic effect of fruit of Nitraria tangutorum Bobr.
from Qinghai Tsaidam basin. Food Sci. 25:164–167. 2004.
|
|
83
|
Shabana MM, Mirhom YW, Genenah AA, et al:
Study into wild Egyptian plants of potential medicinal activity.
Ninth communication: hypoglycaemic activity of some selected plants
in normal fasting and alloxanised rats. Arch Exp Veterinarmed.
44:389–394. 1990.
|
|
84
|
Suo YR, Wang HL and Wang HQ: Research on
decreasing blood lipid and anti-oxidative effect of fruit of
Nitraria Tangutorum Bobr. From Qaidam basin. Nat Prod Res
Deve. 16:54–58. 2004.
|
|
85
|
Song HY, Zhang L, Pan JL, et al:
Bioactivity of five components of Chinese herbal formula Jiangzhi
granules against hepatocellular steatosis. J Integr Med.
11:262–268. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
86
|
Zhang YJ, Fan LL, Zhang YJ, et al:
Protective effect of total flavonoids of Nitraria tangutorun
Bobr. against vascular endothelial cells injury induced by hydrogen
peroxide. Lishizhen Med Materia Med Res. 20:562–564. 2009.(In
Chinese).
|