Introduction
As a traditional Chinese medicine, arsenic has been
widely used for over 2,000 years. It is effectively used in
traditional remedies for the treatment of inflammation, ulcers,
convulsions and schistosomiasis, and studies have demonstrated that
arsenic produces positive effects in cancer therapy (1–3). One
study demonstrated that arsenic trioxide
(As2O3) was clinically effective in patients
with acute promyelocytic leukemia (APL) (4). As2O3 was
approved for the therapy of APL in the year 2000, and subsequently
has been widely used therapeutically in liver, cervical and
esophageal solid tumors (5–7). A
number of studies have demonstrated that the induction of apoptosis
and inhibition of proliferation are involved in the antitumor
mechanism of As2O3 in hematopoietic
malignancies and solid tumors (8–11).
Arsenic sulfide (As4S4),
another arsenic compound, is the main active component of realgar,
an orange-red crystalline mineral that has been extensively used in
traditional Chinese medicine. Compared with
As2O3, As4S4 is less
toxic, but may elicit a similar anti-neoplastic action. The
therapeutic potential of arsenic sulfide in malignancies,
particularly hematopoietic tumors, has been the focus of a number
of studies (12–15). Its antitumor effects are associated
with the inhibition of proliferation, apoptosis and the suppression
of BCR-ABL oncoprotein activity (16,17).
However, the action of As4S4 as a treatment
for solid tumor is unclear. Thus, the aim of the current study was
to investigate the role of As4S4 in the
treatment of solid tumors and its potential as an anticancer agent.
In the present study, the anti-cancer effects of
As4S4 were investigated in vitro and
in vivo.
Materials and methods
Materials
A total of 6 g As4S4 was
dissolved in 200 ml RPMI 1640 medium (Gibco Life Technologies,
Carlsbad, CA, USA) for 12 h, then the concentration of arsenic was
measured by atomic absorption spectrometry (IRIS 1000, Thermo,
Waltham, MA, USA). The
3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT)
was purchased from Sigma-Aldrich (St. Louis, MO, USA).
Cells and animals
The MKN45 gastric cancer, A375 malignant melanoma,
8898 pancreatic carcinoma, HepG2 hepatocellular carcinoma and L02
embryonic liver cell lines were purchased from the cell bank of the
Type Culture Collection of The Chinese Academy of Sciences
(Shanghai, China). All cells were cultured in RPMI 1640 medium
supplemented with 10% heat-inactivated fetal bovine serum (FBS;
Gibco Life Technologies), penicillin (100 U/ml) and streptomycin
(100 U/ml; both Gibco Life Technologies). Cells were incubated at
37°C in a humidified atmosphere of 95% air and 5% CO2.
Male C57BL/6 mice (n=32; age, six weeks), were obtained from the
Animal Center of Fudan University (Shanghai, China; license no.,
2007-0002 SCXK). Mice implanted with Lewis lung carcinoma (LLC)
cells were purchased from the Institute of Materia Medica, Chinese
Academy of Sciences (Shanghai, China; license no., SCXK 2004-0002).
Mice were maintained in an animal facility under pathogen-free
conditions (license no., SYXK 2003-0031). This study was approved
by the ethics committee of Xin Hua Hospital Affiliated to Shanghai
Jiao Tong University School of Medicine (Shanghai, China).
Cytotoxicity assay
The cytotoxicity assay was performed using MTT.
Cells were seeded into and allowed to attach to 96-well culture
plates (1×104 cells/well), for 4 h prior to treatment.
As4S4 at concentrations of 0, 1.25, 2.5, 5
and 10 μg/ml was administered to the cells. After 24-h
treatment, cell viability was evaluated by MTT assay. MTT solution
(20 μl; Sigma-Aldrich) was added to each well and the plates were
incubated for an additional 4 h at 37°C. The supernatant from each
well was then carefully removed, and 100 μl dimethyl sulfoxide
(Sigma-Aldrich) was added to each well and thoroughly mixed. The
optical density (OD) was measured on a Model 550 microplate reader
(Bio-Rad Laboratories, Hercules, CA, USA) at an absorbance
wavelength of 492 nm and a reference wavelength of 630 nm. It was
denoted that the percentage of cell viability = (average OD of
experimental group/average OD of control group) × 100%. The
experiment was repeated three times. The IC50
(concentration causing 50% inhibition of cell growth compared with
the control) value of As4S4 for each of the
tumor cell lines was also calculated after 24 h.
Determination of time-activity curve
The effect of As4S4 on cell
viability was determined by measuring the MTT absorbance of living
cells, which were seeded in and allowed to attach to 96-well
plates. Following 0, 6, 12, 24, 36 and 48 h incubation of the tumor
cells with As4S4 (at IC50) while
L02 cells with 10 μg/ml As4S4, the cell
viability was evaluated by MTT assay. The experiment was repeated
three times.
Hematoxylin and eosin (HE) staining
assay
Cells from exponentially growing cultures were
seeded in 24-well culture plates and treated with
As4S4 (at IC50) for 36 h, and L02
cells were treated with 10 μg/ml
As4S4. Cells were washed with
phosphate-buffered saline (PBS; Gibco Life Technologies), fixed in
4% paraformaldehyde (Sigma-Aldrich) for 15 min, stained with
hematoxylin (Beyotime Institute of Biotechnology, Jiangsu, China)
for 8 min, washed again with PBS, stained with eosin (Beyotime
Institute of Biotechnology) for 5 min, and then examined and imaged
with with the Nikon Eclipse 55i microscope (Nikon Corporation,
Tokyo, Japan).
Flow cytometric analysis of cellular DNA
content
Three cell lines (A375, MKN45 and L02) were seeded
in 6-well culture plates (2×105 cells/well). Following
12-h incubation, A375 and MKN45 cells were treated with the
respective IC50 of As4S4 for 36 h,
and L02 cells were treated with 10 μg/ml
As4S4. Floating and attached cells were
collected in centrifuge tubes. Cells were washed with PBS, then
resuspended and fixed in 70% ice-cold ethanol (Yangyuan, Changshu,
China) for 4 h at 4°C. Subsequently, they were treated with RNase A
(50 μg/ml; Sigma-Aldrich) for 30 min. Cells were stained
with propidium iodide (50 μg/ml), then analyzed in a flow
cytometer (BD Accuri C6, BD Biosciences, Franklin Lakes, NJ, USA).
The percentages of cells in the G0/G1, S,
G2/M and sub-G1 phases were analyzed using
standard ModiFit LT 3.1and CellQuest Pro software (BD, Mac OS X.1;
San Jose, CA, USA).
Lactate dehydrogenase (LDH) release
assay
The A375 and MKN45 cells were treated with the
respective IC50 doses of As4S4,
and L02 cells were treated with 10 μg/ml
As4S4. After 36 h, supernatants were
harvested to measure the levels of LDH using the LDH kit (BHKT
Clinical Reagent Co., Beijing, China)
Caspase activity assay
MKN45 cells were seeded in 10-cm dishes. Following a
resting period of 12 h, cells were treated with various
concentrations (0 μg/ml, 0.25 × IC50, 0.5 ×
IC50, 1 × IC50) of
As4S4 for 36 h. Following treatment, the
cells (floating and attached) were collected and washed three times
with PBS and resuspended in 50 mM Tris-HCl (pH 7.4; Sigma-Aldrich),
1 mM EDTA (Sigma-Aldrich) and 10 mM ethyleneglycoltetraacetic acid
(Sigma-Aldrich). Cell lysates were clarified by centrifugation at
18,000 × g for 3 min and clear lysates containing 50 μg
protein were incubated with 100 μM enzyme-specific
colorigenic substrates (Ac-DEVD-pNA; Beyotime Institute of
Biotechnology) at 37°C for 1 h. The activity of caspase-3 and −9
was denoted as the cleavage of colorimetric substrate measured at
an absorbance of 405 nm.
In vivo experiments with C57BL/6
mice
The mice implanted with LLC cells were sacrificed by
cervical dislocation. Under sterile conditions, tumor tissues were
dissected and the tumor cells were suspended in RPMI 1640 medium
containing 10% FBS. The cell suspension was injected into the
flanks of the experimental mice (106 cells in 200 μl PBS
for each mouse). The tumor-bearing mice were divided into four
groups: Negative control (NC) group, positive control group and
high- and low- dose groups, each containing eight mice. The
tumor-bearing mice were administered with an intraperitoneal
injection of either 30 (low) or 60 (high) mg/kg dose of
As4S4, daily for eight days. The NC group was
treated with 0.9% normal saline (Rongbai, Shanghai, China) and the
positive group was treated with 20 mg/kg cyclophosphamide (CTX;
Yili, Shanghai, China), respectively. Subsequent to euthanization,
the solid tumors were harvested and weighed, and blood was drawn to
measure the level of interleukin-2 (IL-2). The solid tumor weights
were statistically analyzed. The rate of inhibition (RI) was
calculated according to the following formula: RI = [(mean tumor
weight of the NC group - mean tumor weight of the drug group)/mean
tumor weight of the NC group] × 100%.
Statistical analysis
Each experimental value was expressed as the mean ±
standard deviation. Statistical analysis was performed using Origin
software, version 7.0 (OriginLab, Northampton, MA, USA) to evaluate
the differences between groups. P<0.05 was considered to
indicate a statistically significant difference.
Results
Cytotoxic effect of
As4S4 on tumor cells
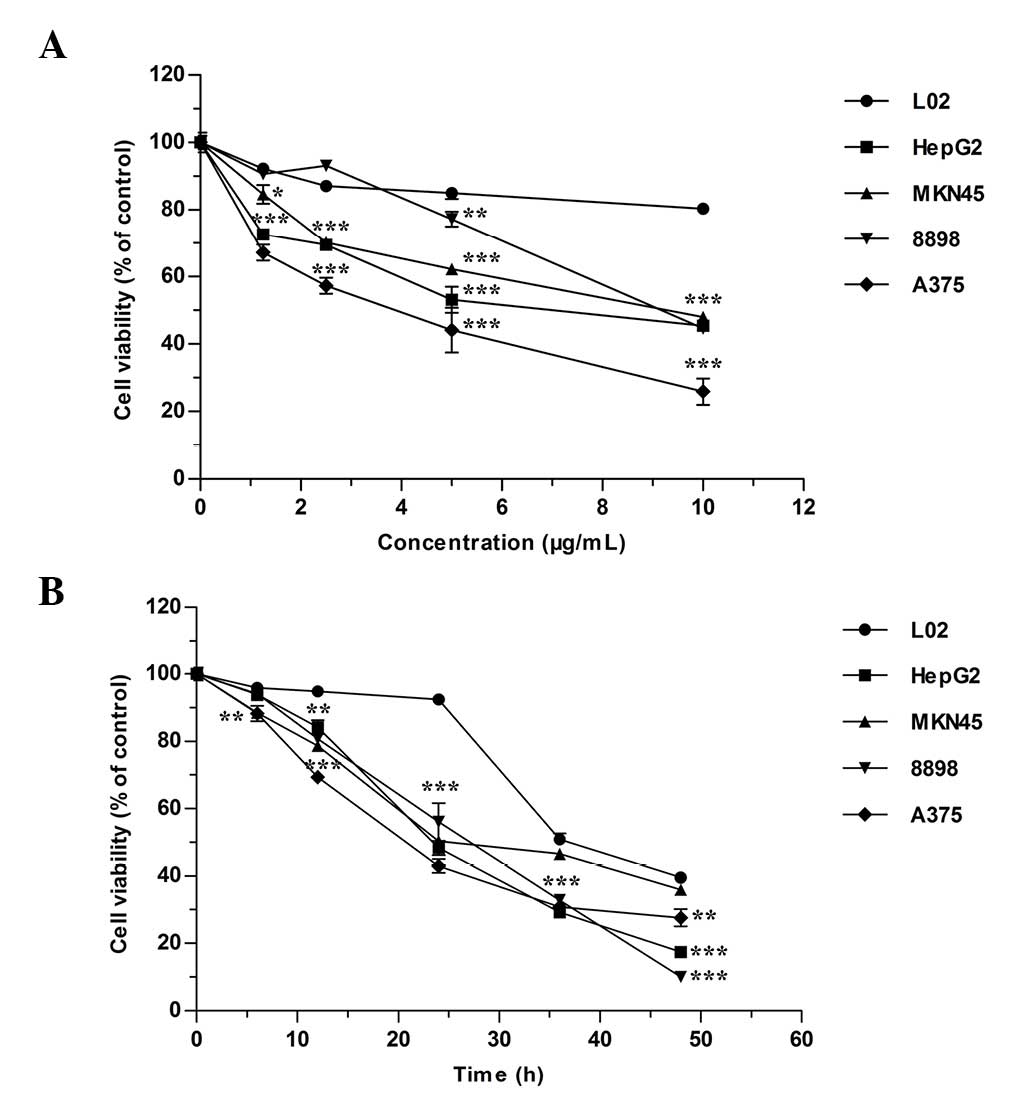
The cytotoxicity assay results for
As4S4 on the five cell lines are presented in
Fig. 1. The data indicated that
the cell proliferation was inhibited by As4S4
in a dose-dependent (Fig. 1A) and
time-dependent (Fig. 1B) manner
(P<0.001), and each cell line presented a different sensitivity
to the inhibitory effect of As4S4. The
IC50 values of the tumor cell lines following 24-h
treatment are presented in Table
I. As4S4 generated a weaker effect on L02
cells compared with the four tumor cell lines.
 | Table IIC50 value of arsenic
sulfide for each tumor cell line following 24-h treatment. |
Table I
IC50 value of arsenic
sulfide for each tumor cell line following 24-h treatment.
| Cell line | IC50
(μg/ml) × ± standard deviation |
|---|
| HepG2 | 6.89±1.078 |
| MKN45 | 9.37±0.948 |
| 8898 | 9.06±0.984 |
| A375 | 3.78±0.827 |
Effect of As4S4 on
cell morphology
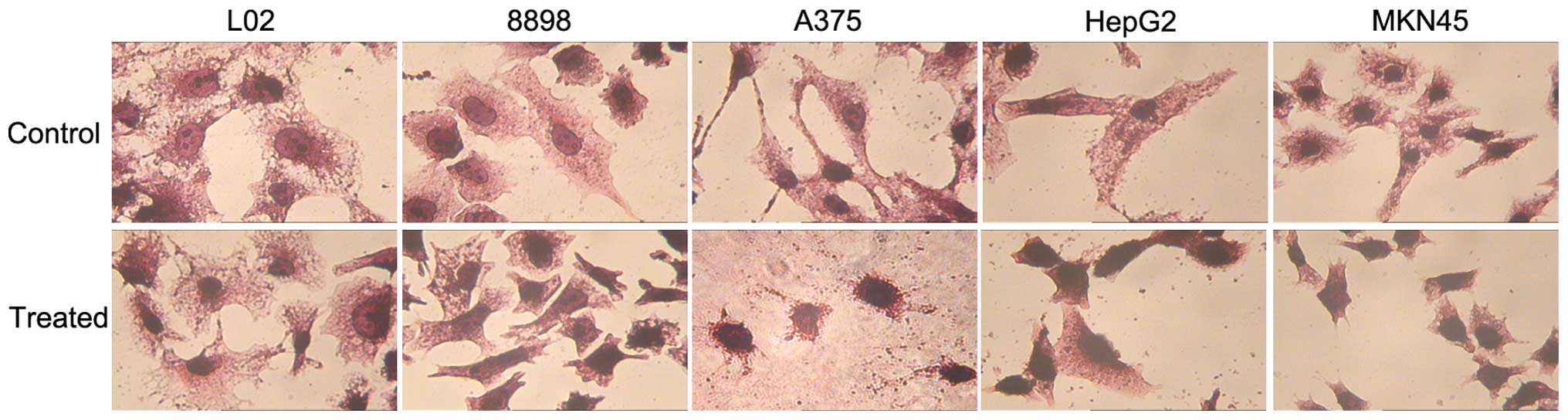
The HE staining assay identified that the tumor
cells (8898, A375, HepG2 and MKN45) treated with
As4S4 exhibited cell shrinkage, nuclear
condensation and fragmentation, which are typical characteristics
of apoptosis. However, the treated L02 cells did not exhibit
significant morphological changes (Fig. 2).
Effect of As4S4 on
G2/M phase arrest and the apoptotic sub-G1
population
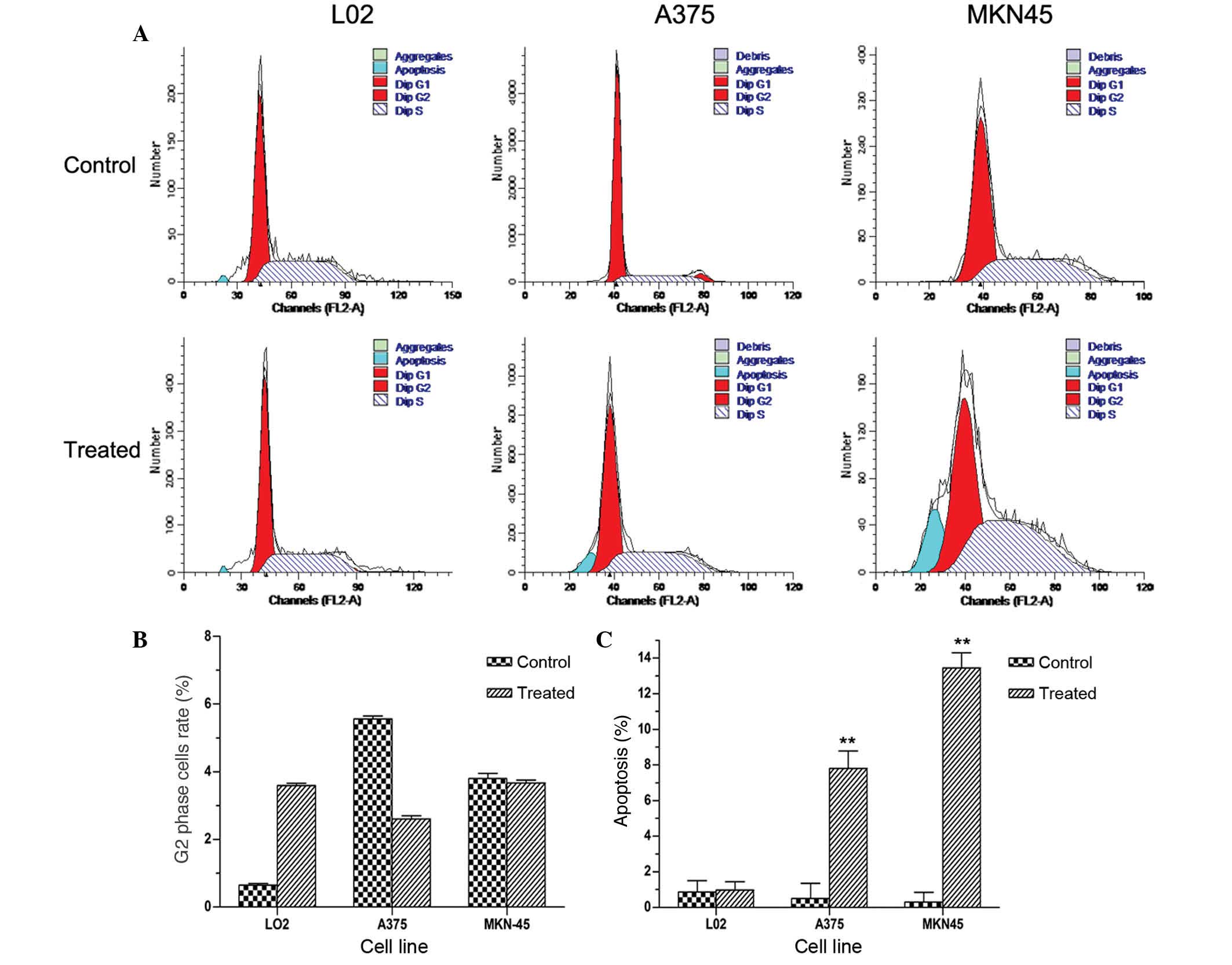
To determine whether the reduction in cell viability
observed involved alterations to the cell cycle, the effect of
As4S4 on the cell cycle distribution in the
A375, MKN45 and L02 cell lines was investigated using
fluorescence-activated cell sorting analysis (Fig. 3A). The apoptotic index was
calculated by measuring the number of cells in the
sub-G1 population following treatment with
As4S4. Subsequent to exposure of A375, MKN45
cells to the respective IC50s of
As4S4 and of L02 cells to 10 μg/ml
As4S4 for 36 h, no marked G2/M
phase arrest was observed, as demonstrated in Fig. 3B. These results suggest that
As4S4 produced no significant effect on
G2/M phase arrest. A significant increase in the
sub-G1 fraction was identified in tumor cells treated
with As4S4, whilst no significant difference
was observed in the L02 cells compared with the untreated control
cells (Fig. 3C).
Effect of As4S4 on
the level of LDH and the activation of caspase
The LDH release assay measured the leakage of LDH
into the extracellular medium following cellular lysis. The release
of intracellular LDH was detected following exposure to
As4S4 (Fig.
4A). Meanwhile, caspase-3 and −9 were activated in MKN45 cells
treated with As4S4 for 36 h (Fig. 4B). These results indicate the
involvement of caspase-3 and −9 in
As4S4-mediated cell apoptosis.
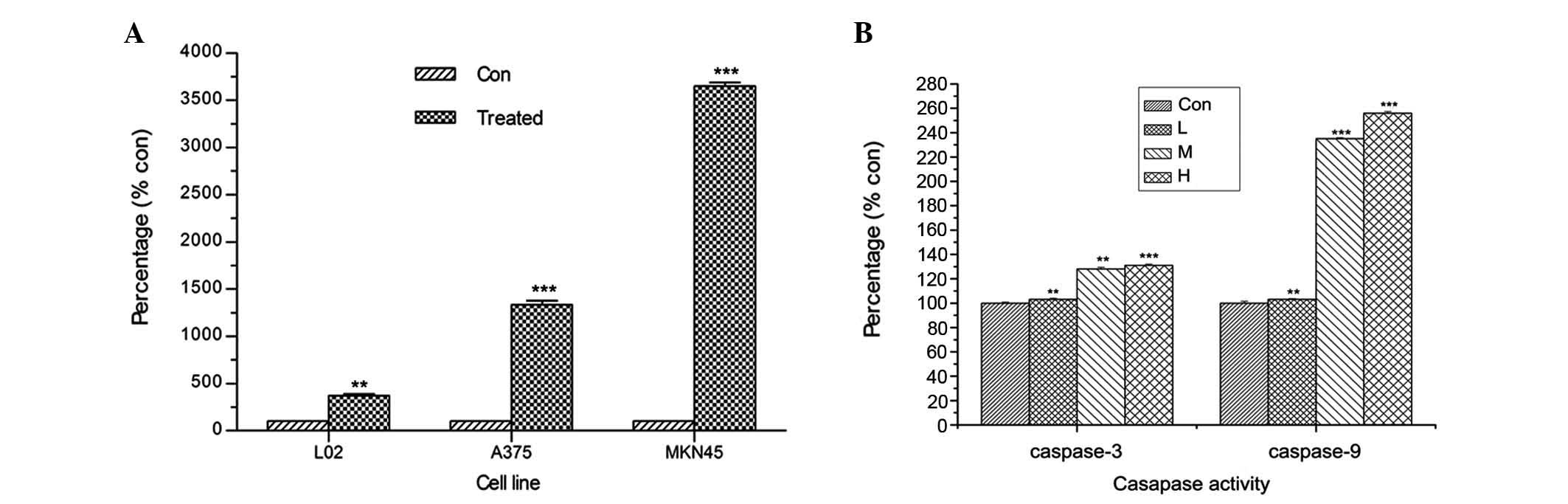 | Figure 4Effect of As4S4
on the release of LDH and the activation of caspase-3 and −9. (A)
Subsequent to exposure of A375 and MKN45 cells to the respective
IC50s of As4S4 for 36 h, and
exposure of L02 cells to 10 μg/ml
As4S4, the leakage of LDH from cells was
analyzed. (B) Caspase-3 and −9 activation was analyzed in MKN45
cells treated with different concentrations of
As4S4 for 36 h: Control, 0 μg/ml; L, 0.25 ×
IC50; M, 0.5 × IC50 and H, 1 ×
IC50. Data are expressed as the mean ± standard
deviation; *P<0.05, **P<0.01 and
***P<0.001 vs. control. As4S4,
arsenic sulfide; LDH, lactate dehydrogenase; L, low; M, medium; H,
high. |
As4S4 inhibits the
growth of solid tumors and elevates the levels of IL-2 in
blood
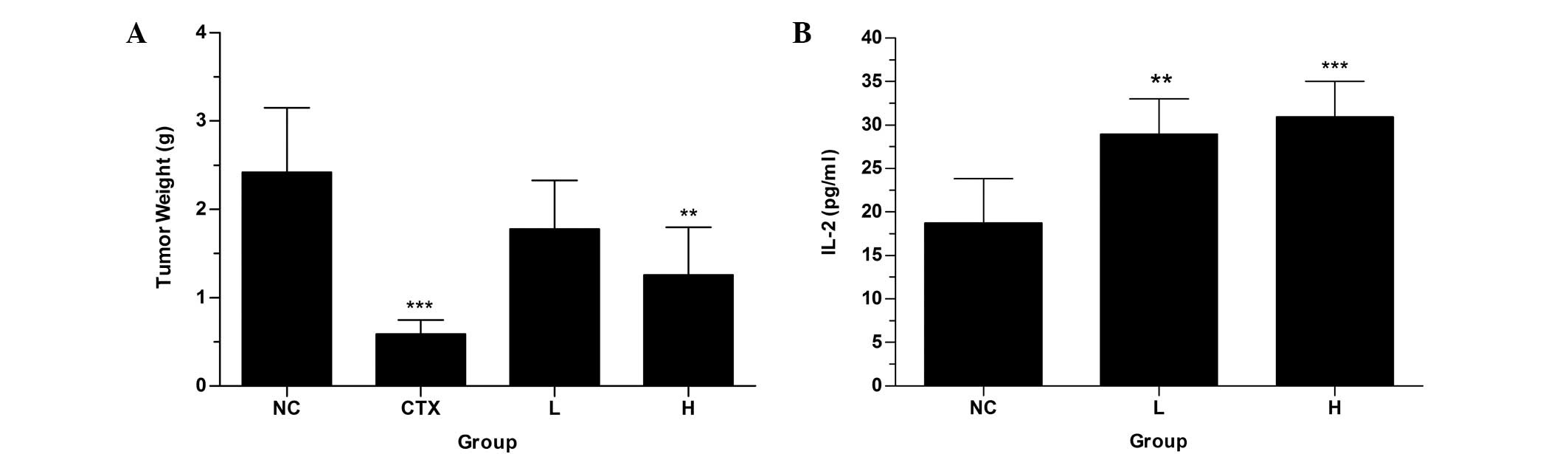
Mouse lung cancer LLC cells were implanted in
C57BL/6 mice. Following treatment with As4S4
for eight days, the suppression of tumor growth was observed
(Fig. 5A). The inhibition ratios
of the low-dose group (As4S4, 30 mg/kg) and
high-dose group (As4S4, 60 mg/kg) were 26.45
and 47.93%, respectively (Table
II). The high dosage exhibited a greater anticancer effect than
the low dosage, and produced a significantly reduced tumor weight
compared with that of the NC group. In addition,
As4S4 treatment did not significantly alter
the body weight of the mice (data not shown). Subsequent to
treatment with As4S4, the concentrations of
IL-2 in the treatment groups were higher compared with the NC group
(Fig. 5B). These results
demonstrate that As4S4 is able to suppress
tumor growth.
 | Table IIAntitumor effect of
As4S4 in vivo. |
Table II
Antitumor effect of
As4S4 in vivo.
| Group | Injection dosage
(mg/kg) × ± standard deviation | Tumor weight
(g) | Tumor inhibition
(%) |
|---|
| Negative
control | 0 | 2.42±0.73 | 0 |
| Positive control
(cyclophosphamide) | 20 | 0.59±0.16b | 75.62 |
| High
As4S4 | 60 | 1.26±0.54a | 47.93 |
| Low
As4S4 | 30 | 1.78±0.55 | 26.45 |
Discussion
As4S4 has attracted worldwide
interest in recent years due to the successful clinical application
of arsenic compounds in the treatment of APL (12,18).
However, its efficacy in the treatment of solid tumors remains to
be thoroughly elucidated. Hence, in the present study, the
antitumor effect of As4S4 on solid tumors and
its possible mechanism of action were investigated.
Apoptosis, the most common form of tumor cell death,
is a biological process of programmed cell death (PCD). The typical
morphological and molecular changes that occur during the course of
apoptosis include cell shrinkage, nuclear fragmentation, chromatin
condensation, DNA fragmentation and changes in apoptotic protein
expression (19–21). In the current study, using MTT
assay, As4S4 was observed to be able to
inhibit the proliferation of tumor cells in a dose- and
time-dependent manner, but produced a less marked effect on healthy
L02 cells, which were used as a control to assess the
hepatotoxicity of As4S4. Flow cytometry
indicated that the inhibition of A375 and MKN45 tumor cell growth
by As4S4 may be due to cell apoptosis.
However, no significant effect of As4S4 on
G2/M phase arrest was observed. The HE staining assay
demonstrated that four tumor cell lines treated with the
IC50 of As4S4 for 36 h exhibited
cell shrinkage, nuclear condensation and fragmentation (typical
characteristics of apoptosis), while L02 cells treated with
As4S4 demonstrated no differences prior to
and post administration.
LDH is clinically significant as a marker of injury
and disease, since it is released during cell or tissue damage. The
LDH release assay measures the leakage of the soluble cytoplasmic
LDH enzyme into the extracellular medium via cellular lysis. The
current study demonstrated that As4S4
increased the release of LDH, indicating the induction of apoptosis
or necrosis.
Caspases are cysteine proteases activated by a
cascade, involving the cleavage of their precursors. Caspase-3 is
an executioner caspase that disassembles cells through the cleavage
of proteins such as PARP, in order to inactivate them. Caspase-3 is
commonly activated by either the caspase-9-mediated mitochondrial
pathway, or by the caspase-8-mediated death receptor pathway
(22–24). In the current study,
As4S4 treatment was demonstrated to lead to
caspase-3 and −9 activation, which suggests the involvement of the
caspase pathways in As4S4-induced
apoptosis.
The antitumor activity of
As4S4 was further examined in a mouse model.
Similar to the in vitro results, As4S4
was capable of inhibiting tumor growth in vivo. In the high
dosage group, the tumors grew slower compared to the NC group.
However, As4S4 did not produce a significant
inhibitory effect on tumor growth in the low
As4S4 dosage group compared with the NC
group.
Studies have demonstrated that IL-2 serves an
important function in the regulation of antigen-specific T-cell
responses (25,26). The cytokines expressed by a T-cell
in response to an antigen indicate the specific pathway, and IL-2
is associated with the T helper 1 (Th1) pathway. IL-2 has been
demonstrated to serve an important role in specific immunological
responses to tumor cell growth (27,28).
Thus, in the present study, IL-2 production was investigated to
evaluate the hypothesis that As4S4 increases
T-cell activation by modulating IL-2. The results demonstrated that
the serum concentrations IL-2 were enhanced in
As4S4-treated C57BL/6 mice bearing LLC,
compared with the NC mice, suggesting that it may be a potent
inducer of Th1-type cytokines.
In the present study, As4S4
was observed to induce apoptosis in cancer cells. The detailed
mechanism of apoptosis induction in solid tumors by
As4S4 requires further investigation.
Previous studies have reported that As4S4
treatment induced differentiation of hematological tumor cells
(29,30), suggesting that the antitumor action
of As4S4 in solid tumors may involve cellular
differentiation.
In the current study, the results suggested that
As4S4 may be able to inhibit cell growth and
increase the release of LDH. Furthermore, the apoptosis was
suggested to involve caspase-3 and −9 activation. Additionally,
As4S4 was demonstrated to have the effect of
suppressing tumor growth in vivo. These results suggest that
As4S4 may be a potential therapeutic
candidate in the treatment of solid tumors.
Acknowledgements
The current study was supported by the National
Natural Science Foundation of China (grant nos. 81274142 and
30300139); the Science and Technology Commission of Shanghai
Municipality (grant no. 11ZR1423400); the Key Project of Shanghai
Municipal Education Commission (grant no. 07zz43); and Shanghai
Jiao Tong University School of Medicine Foundation of Science and
Technology (05XJ21030).
References
|
1
|
Nakagawa Y, Akao Y, Morikawa H, et al:
Arsenic trioxide-induced apoptosis through oxidative stress in
cells of colon cancer cell lines. Life Sci. 70:2253–2269. 2002.
View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Ramanathan K, Anusuyadevi M, Shila S and
Panneerselvam C: Ascorbic acid and alpha-tocopherol as potent
modulators of apoptosis on arsenic induced toxicity in rats.
Toxicol Lett. 156:297–306. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Waxman S and Anderson KC: History of the
development of arsenic derivatives in cancer therapy. Oncologist.
6(Suppl 2): 3–10. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Niu C, Yan H, Yu T, et al: Studies on
treatment of acute promyelocytic leukemia with arsenic trioxide:
remission induction, follow-up, and molecular monitoring in 11
newly diagnosed and 47 relapsed acute promyelocytic leukemia
patients. Blood. 94:3315–3324. 1999.PubMed/NCBI
|
|
5
|
Murgo AJ: Clinical trials of arsenic
trioxide in hematologic and solid tumors: overview of the National
Cancer Institute Cooperative Research and Development Studies.
Oncologist. 6(Suppl 2): 22–28. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Zheng J, Deng YP, Lin C, Fu M, Xiao PG and
Wu M: Arsenic trioxide induces apoptosis of HPV16 DNA-immortalized
human cervical epithelial cells and selectively inhibits viral gene
expression. Int J Cancer. 82:286–292. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Shen ZY, Zhang Y, Chen JY, et al:
Intratumoral injection of arsenic to enhance antitumor efficacy in
human esophageal carcinoma cell xenografts. Oncol Rep. 11:155–159.
2004.
|
|
8
|
Emadi A and Gore SD: Arsenic trioxide - An
old drug rediscovered. Blood Rev. 24:191–199. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Rojewski MT, Baldus C, Knauf W, Thiel E
and Schrezenmeier H: Dual effects of arsenic trioxide
(As2O3) on non-acute promyelocytic leukaemia
myeloid cell lines: induction of apoptosis and inhibition of
proliferation. Br J Haematol. 116:555–563. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Park WH, Seol JG, Kim ES, et al: Arsenic
trioxide-mediated growth inhibition in MC/CAR myeloma cells via
cell cycle arrest in association with induction of cyclin-dependent
kinase inhibitor, p21, and apoptosis. Cancer Res. 60:3065–3071.
2000.PubMed/NCBI
|
|
11
|
Zhong F, Zhang S, Shao C, Yang J and Wu X:
Arsenic trioxide inhibits cholangiocarcinoma cell growth and
induces apoptosis. Pathol Oncol Res. 16:413–420. 2010. View Article : Google Scholar
|
|
12
|
Lu DP, Qiu JY, Jiang B, et al:
Tetra-arsenic tetra-sulfide for the treatment of acute
promyelocytic leukemia: a pilot report. Blood. 99:3136–3143. 2002.
View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Wang L, Zhou GB, Liu P, et al: Dissection
of mechanisms of Chinese medicinal formula Realgar-Indigo naturalis
as an effective treatment for promyelocytic leukemia. Proc Natl
Acad Sci USA. 105:4826–4831. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Zhang QY, Mao JH, Liu P, et al: A systems
biology understanding of the synergistic effects of arsenic sulfide
and Imatinib in BCR/ABL-associated leukemia. Proc Natl Acad Sci
USA. 106:3378–3383. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Chen S, Fang Y, Ma L, Liu S and Li X:
Realgar-induced apoptosis and differentiation in all-trans retinoic
acid (ATRA)-sensitive NB4 and ATRA-resistant MR2 cells. Int J
Oncol. 40:1089–1096. 2012.
|
|
16
|
Yin T, Wu YL, Sun HP, et al: Combined
effects of As4S4 and imatinib on chronic
myeloid leukemia cells and BCR-ABL oncoprotein. Blood.
104:4219–4225. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Wu J, Shao Y, Liu J, Chen G and Ho PC: The
medicinal use of realgar (As4S4) and its
recent development as an anticancer agent. J Ethnopharmacol.
135:595–602. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Lu DP and Wang Q: Current study of APL
treatment in China. Int J Hematol. 76(Suppl 1): 316–318. 2002.
View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Majno G and Joris I: Apoptosis, oncosis,
and necrosis. An overview of cell death. Am J Pathol. 146:3–15.
1995.PubMed/NCBI
|
|
20
|
McConkey DJ and Orrenius S: Signal
transduction pathways in apoptosis. Stem Cells. 14:619–631. 1996.
View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Hunot S and Flavell RA: Apoptosis. Death
of a monopoly? Science. 292:865–866. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Hengartner M: Apoptosis. Death by crowd
control. Science. 281:1298–1299. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Ashkenazi A and Dixit VM: Death receptors:
signaling and modulation. Science. 281:1305–1308. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
McIlwain DR, Berger T and Mak TW: Caspase
functions in cell death and disease. Cold Spring Harb Perspect
Biol. 5:a0086562013. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Smith KA: Interleukin-2: inception,
impact, and implications. Science. 240:1169–1176. 1988. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Sakaguchi S, Sakaguchi N, Asano M, Itoh M
and Toda M: Immunologic self-tolerance maintained by activated T
cells expressing IL-2 receptor alpha-chains (CD25). Breakdown of a
single mechanism of self-tolerance causes various autoimmune
diseases. J Immunol. 155:1151–1164. 1995.PubMed/NCBI
|
|
27
|
McAdam AJ, Pulaski BA, Harkins SS, Hutter
EK, Lord EM and Frelinger JG: Synergistic effects of co-expression
of the TH1 cytokines IL-2 and IFN-gamma on generation of murine
tumor-reactive cytotoxic cells. Int J Cancer. 61:628–634. 1995.
View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Sakthivel KM and Guruvayoorappan C: Acacia
ferruginea inhibits tumor progression by regulating inflammatory
mediators-(TNF-α, iNOS, COX-2, IL-1β, IL-6, IFN-γ, IL-2, GM-CSF)
and pro-angiogenic growth factor-VEGF. Asian Pac J Cancer Prev.
14:3909–3919. 2013. View Article : Google Scholar
|
|
29
|
Wang LW, Shi YL, Wang N, Gou BD, Zhang TL
and Wang K: Association of oxidative stress with realgar-induced
differentiation in human leukemia HL-60 cells. Chemotherapy.
55:460–467. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Wang N, Wang LW, Gou BD, Zhang TL and Wang
K: Realgar-induced differentiation is associated with MAPK pathways
in HL-60 cells. Cell Biol Int. 32:1497–1505. 2008. View Article : Google Scholar : PubMed/NCBI
|