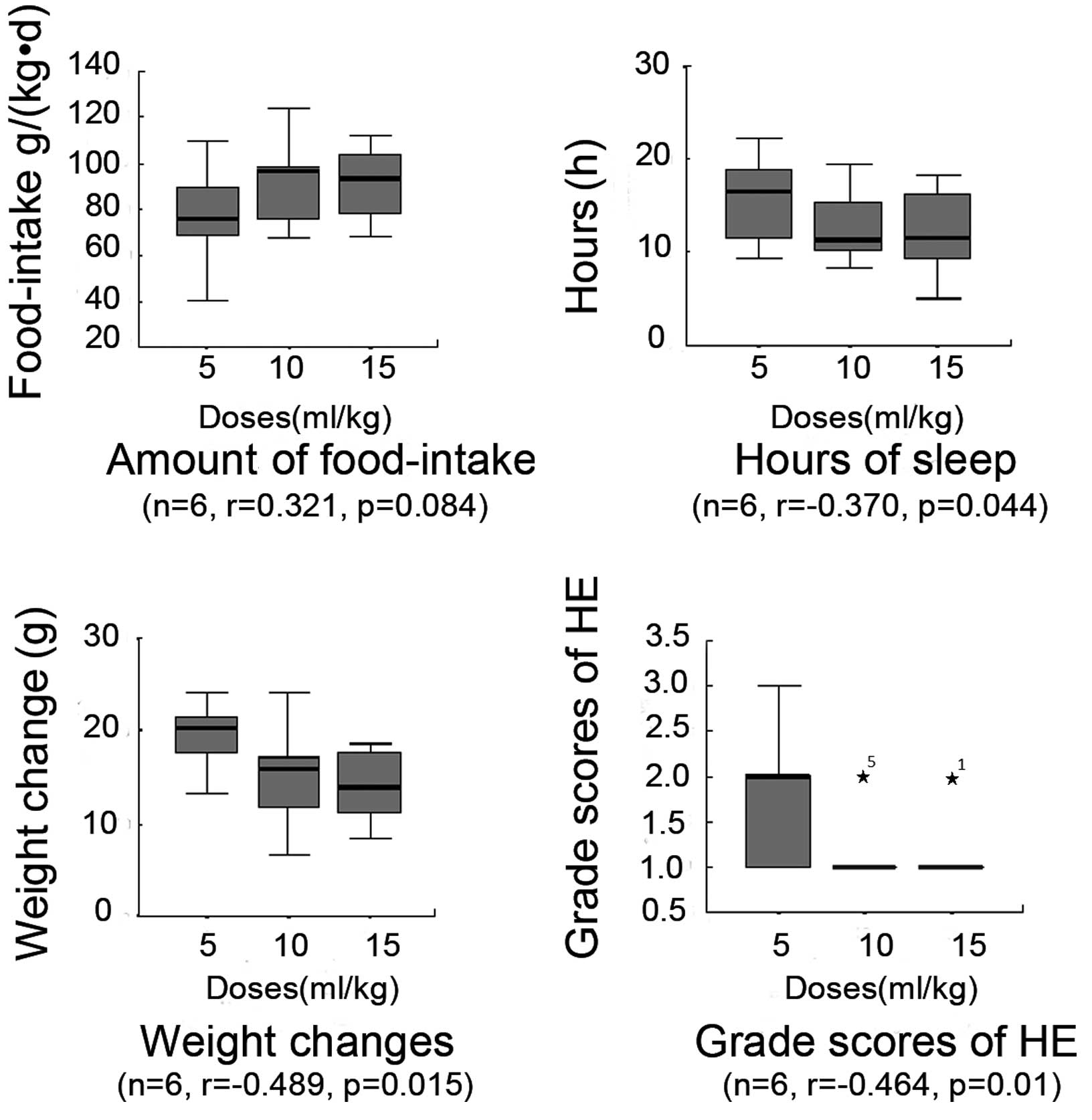
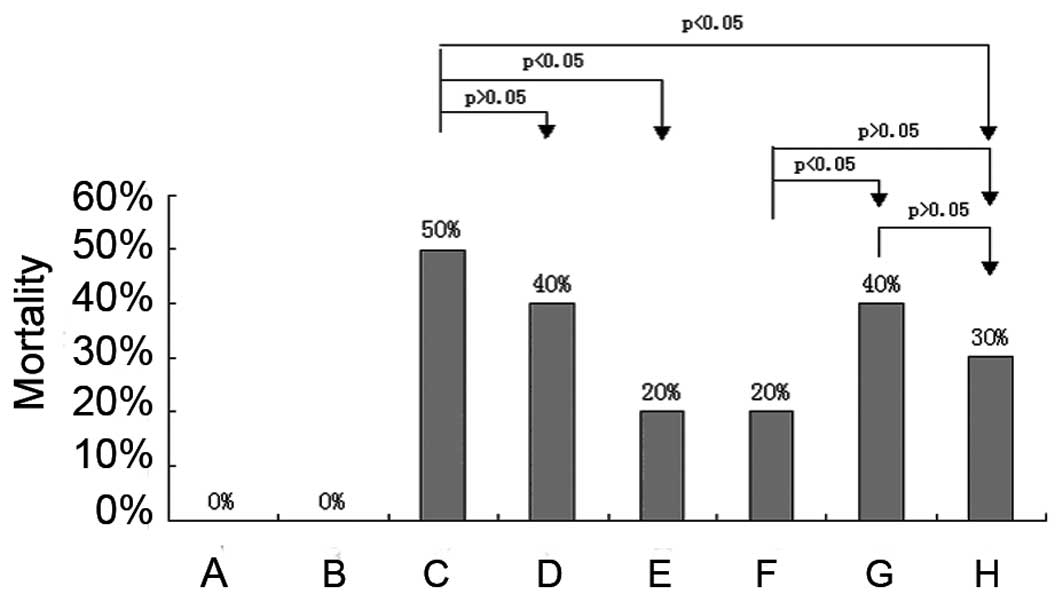
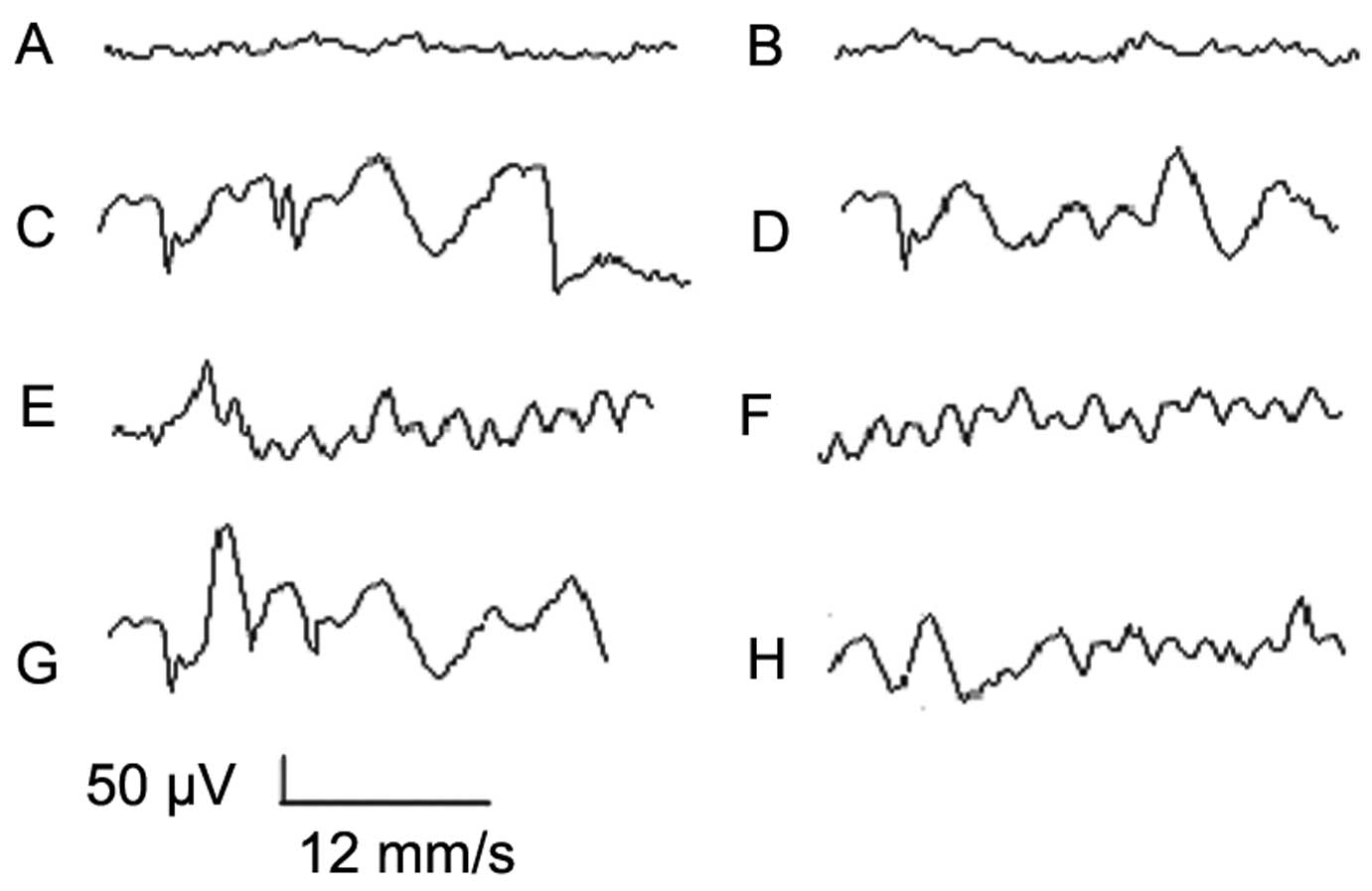
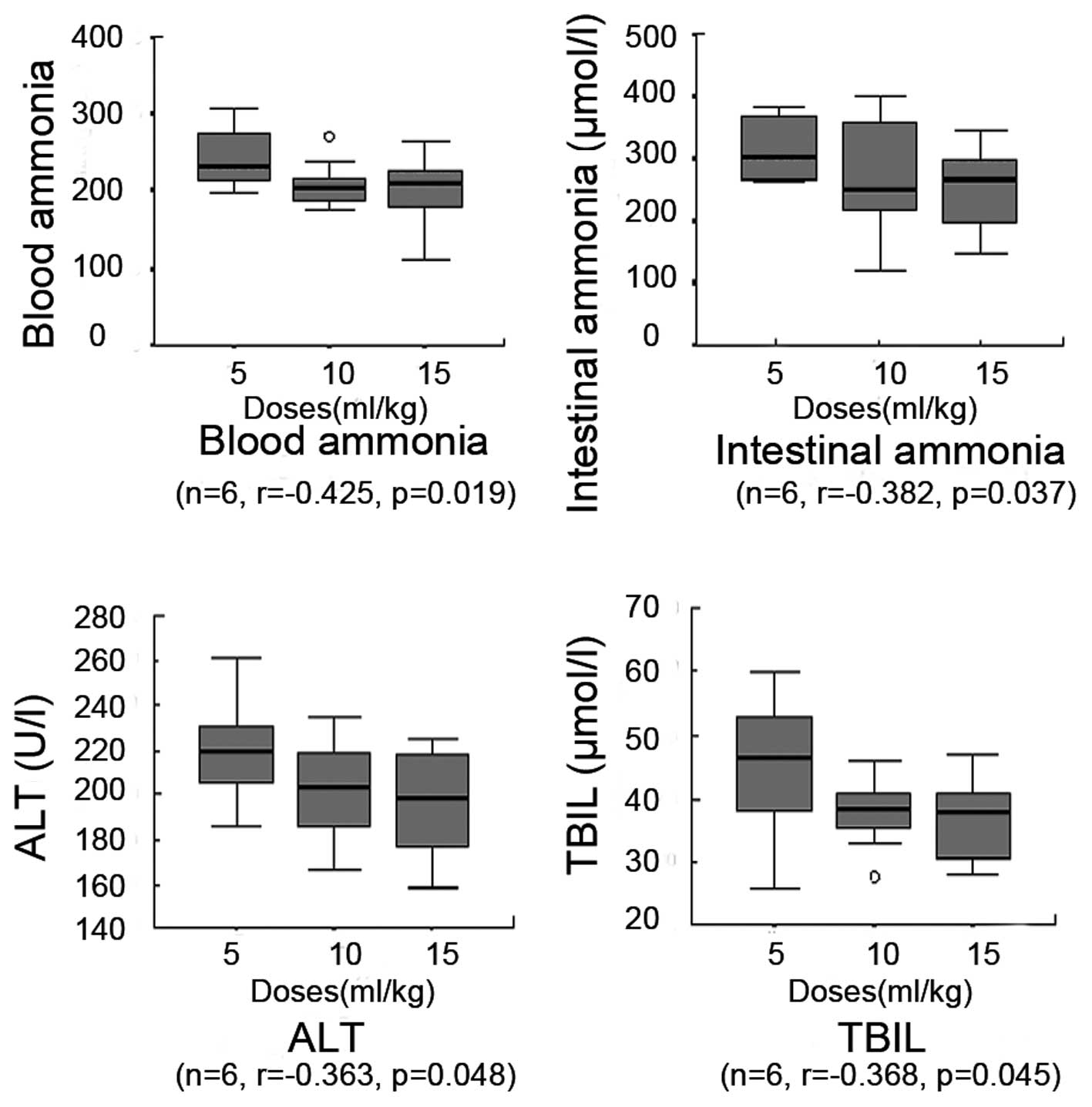
|
1
|
Butterworth RF, Norenberg MD, Felipo V,
Ferenci P, Albrecht J and Blei AT; Members of the ISHEN Commission
on Experimental Models of HE. Experimental models of hepatic
encephalopathy: ISHEN guidelines. Liver Int. 29:783–788. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Huang HC, Chang CC, Wang SS, Chan CY, Lee
FY, et al: Pravastatin for thioacetamide-induced hepatic failure
and encephalopathy. Eur J Clin Invest. 42:139–145. 2012. View Article : Google Scholar
|
|
3
|
Rama Rao KV, Reddy PV, Tong X and
Norenberg MD: Brain edema in acute liver failure: inhibition by
L-histidine. Am J Pathol. 176:1400–1408. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Butterworth RF: Altered glial-neuronal
crosstalk: cornerstone in the pathogenesis of hepatic
encephalopathy. Neurochem Int. 57:383–388. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Strekalova OS, Uchaĭkin VF, Ipatova OM,
Torkhovskaia TI, Medvedeva NV, et al: Comatose states:
etiopathogenesis, experimental studies, treatment of hepatic coma.
Biomed Khim. 55:380–396. 2009.(In Russian). PubMed/NCBI
|
|
6
|
Hassanein T, Blei AT, Perry W, Hilsabeck
R, Stange J, et al: Performance of the hepatic encephalopathy
scoring algorithm in a clinical trial of patients with cirrhosis
and severe hepatic encephalopathy. Am J Gastroenterol.
104:1392–1400. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Kochar DK, Agarwal P, Kochar SK, Jain R,
Rawat N, et al: Hepatocyte dysfunction and hepatic encephalopathy
in Plasmodium falciparum malaria. QJM. 96:505–512. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Bémeur C, Vaquero J, Desjardins P and
Butterworth RF: N-acetylcysteine attenuates cerebral complications
of non-acetaminophen-induced acute liver failure in mice:
antioxidant and anti-inflammatory mechanisms. Metab Brain Dis.
25:241–249. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Sharma P, Sharma BC and Sarin SK: Critical
flicker frequency for diagnosis and assessment of recovery from
minimal hepatic encephalopathy in patients with cirrhosis.
Hepatobiliary Pancreat Dis Int. 9:27–32. 2010.PubMed/NCBI
|
|
10
|
Zafirova Z and O’Connor M: Hepatic
encephalopathy: current management strategies and treatment,
including management and monitoring of cerebral edema and
intracranial hypertension in fulminant hepatic failure. Curr Opin
Anaesthesiol. 23:121–127. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Al Sibae MR and McGuire BM: Current trends
in the treatment of hepatic encephalopathy. Ther Clin Risk Manag.
5:617–626. 2009.PubMed/NCBI
|
|
12
|
Zheng Z, Li X, Li Z and Ma X: Artificial
and bioartificial liver support systems for acute and
acute-on-chronic hepatic failure: A meta-analysis and
meta-regression. Exp Ther Med. 6:929–936. 2013.PubMed/NCBI
|
|
13
|
Choi JW, Yoon KT, Park JY, Kim JK, Ahn SH,
et al: Usefulness and safety of extracorporeal liver support
therapy using MARSR for patients with liver failure: a preliminary
report. Korean J Gastroenterol. 54:28–35. 2009.(In Korean).
View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Phongsamran PV, Kim JW, Cupo Abbott J and
Rosenblatt A: Pharmacotherapy for hepatic encephalopathy. Drugs.
70:1131–1148. 2009. View Article : Google Scholar
|
|
15
|
Sharma P and Sharma BC: Disaccharides in
the treatment of hepatic encephalopathy. Metab Brain Dis.
28:313–320. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Sharma BC, Sharma P, Agrawal A and Sarin
SK: Secondary prophylaxis of hepatic encephalopathy: an open-label
randomized controlled trial of lactulose versus placebo.
Gastroenterology. 137:885–891. 891.e12009. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Malaguarnera M, Gargante MP, Malaguarnera
G, Salmeri M, Mastrojeni S, et al: Bifidobacterium combined with
fructo-oligosaccharide versus lactulose in the treatment of
patients with hepatic encephalopathy. Eur J Gastroenterol Hepatol.
22:199–206. 2010. View Article : Google Scholar
|
|
18
|
Weber FL Jr: Effects of lactulose on
nitrogen metabolism. Scand J Gastroenterol Suppl. 222:83–87.
1997.PubMed/NCBI
|
|
19
|
Sotelo N, de los Angeles Durazo M,
Gonzalez A and Dhanakotti N: Early treatment with N-acetylcysteine
in children with acute liver failure secondary to hepatitis A. Ann
Hepatol. 8:353–358. 2009.PubMed/NCBI
|
|
20
|
Sundaram V and Shaikh OS: Hepatic
encephalopathy: pathophysiology and emerging therapies. Med Clin
North Am. 93:819–836. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Wang AH, Duan ZJ, Tian G, Lu D, Zhang WJ,
et al: The addition of a pH-sensitive gel improves microemulsion
stability for the targeted removal of colonic ammonia. BMC
Gastroenterol. 11:502011. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Tønnesen HH and Karlsen J: Alginate in
drug delivery systems. Drug Dev Ind Pharm. 28:621–630. 2002.
View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Pan MD, Li JC, Lin Q, Wang XH and Wang LH:
Progress on sodium alginate application to drug controlled release.
China Pharmaceuticals. 17:3–5. 2008.(In Chinese).
|
|
24
|
Chen SC, Wu YC, Mi FL, et al: A novel
pH-sensitive hydrogel composed of N,O-carboxymethyl chitosan and
alginate crosslinked by genipin for protein drug delivery. J
Control Release. 96:285–300. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Moroni A, Drefko W and Thone G:
Formulations of zero-order, pH-dependent, sustained release matrix
systems by ionotropic gelation of alginate-containing mixtures.
Drug Dev Ind Pharm. 37:216–224. 2011. View Article : Google Scholar
|
|
26
|
El-Sherbiny IM, Salama A and Sarhan AA:
Ionotropically cross-linked pH-sensitive IPN hydrogel matrices as
potential carriers for intestine-specific oral delivery of protein
drugs. Drug Dev Ind Pharm. 37:121–130. 2011. View Article : Google Scholar
|
|
27
|
Xiong W, Gao X, Zhao Y, Xu H and Yang X:
The dual temperature/pH-sensitive multiphase behavior of poly
(N-isopropylacrylamide-co-acrylic acid) microgels for potential
application in in situ gelling system. Colloids Surf B
Biointerfaces. 84:103–110. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Yin YH, Yang YJ and Xu BH: Swelling
kinetics of hydrogels for colonic-site drug delivery. Acta
Polymerica Sinica. 2001.650–655. 2001.(In Chinese).
|
|
29
|
Yin YH, Yang YJ and Xu BH: Hydrogels
containing 4,4′-di(methacryloylmino)azobenzenes: Synthesis and
animal experiment for colonic-site drug release. Acta Polymerica
Sinica. 2002.408–413. 2002.(In Chinese).
|
|
30
|
Kim S, Kim JH and Kim D: pH sensitive
swelling and releasing behavior of nano-gels based on
polyaspartamide graft copolymers. J Colloid Interface Sci.
356:100–106. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Kono K, Tabata F and Takagishi T:
pH-responsive permeability of poly (acrylic acid)-poly
(ethylenimine) complex capsule membrane. J Memb Sci. 76:233–243.
1993. View Article : Google Scholar
|
|
32
|
Li XQ, Dong L, Liu ZH and Luo JY:
Expression of gamma-aminobutyric acid A receptor subunits alpha1,
beta1, gamma2 mRNA in rats with hepatic encephalopathy. World J
Gastroenterol. 11:3319–3322. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Zimmermann C, Ferenci P, Pifl C, Yurdaydin
C, Ebner J, et al: Hepatic encephalopathy in thioacetamide-induced
acute liver failure in rats: characterization of an improved model
and study of amino acid-ergic neurotransmission. Hepatology.
9:594–601. 1989. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Chu CJ, Lee FY, Wang SS, Chang FY, Lin HC,
et al: Establishment of an animal model of hepatic encephalopathy.
Zhonghua Yi Xue Za Zhi (Taipei). 63:263–269. 2000.
|
|
35
|
Yurdaydin C, Walsh TJ, Engler HD, Ha JH,
Li Y, et al: Gut bacteria provide precursors of benzodiazepine
receptor ligands in a rat model of hepatic encephalopathy. Brain
Res. 679:42–48. 1995. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Bongaerts G, Severijnen R and Timmerman H:
Effect of antibiotics, prebiotics and probiotics in treatment for
hepatic encephalopathy. Med Hypotheses. 64:64–68. 2005. View Article : Google Scholar
|
|
37
|
Häberle J: Clinical practice: the
management of hyperammonemia. Eur J Pediatr. 170:21–34. 2011.
View Article : Google Scholar
|
|
38
|
Kowdley KV and Burman BE: ACP Journal
Club. Adding rifaximin to lactulose increased reversal and
decreased mortality in hepatic encephalopathy. Ann Intern Med.
159:JC82013. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Sharma BC, Sharma P, Lunia MK, et al: A
randomized, double-blind, controlled trial comparing rifaximin plus
lactulose with lactulose alone in treatment of overt hepatic
encephalopathy. Am J Gastroenterol. 108:1458–1463. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Debray D, Yousef N and Durand P: New
management options for end-stage chronic liver disease and acute
liver failure: potential for pediatric patients. Paediatr Drugs.
8:1–13. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Sharma P, Sharma BC and Sarin SK:
Predictors of nonresponse to lactulose for minimal hepatic
encephalopathy in patients with cirrhosis. Liver Int. 29:1365–1371.
2009. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Maclayton DO and Eaton-Maxwell A:
Rifaximin for treatment of hepatic encephalopathy. Ann
Pharmacother. 43:77–84. 2009. View Article : Google Scholar
|