Introduction
MicroRNAs (miRNAs), a class of highly conserved
small non-coding RNAs, play crucial roles in regulating the
expression of target genes in a sequence-specific manner (1,2). It
is acknowledged that the binding of a specific miRNA to the
3′-untranslated region (3′-UTR) of target mRNA may inhibit the
expression of target genes. To date, several classes of miRNAs have
been reported to play significant regulatory roles in various
cellular processes through translational repression, including cell
proliferation, apoptosis and developmental timing (3). Previous studies have revealed that
aberrant expression of miRNA is associated with the development of
human cancer, and its expression profiling has been identified as a
signature of the diagnostic and prognostic characteristics of
specific carcinomas (4,5). Li et al demonstrated that
down-regulation of miR-1297 in Hep-2 cells may inhibit
proliferation of cancer cells, migration and tumor genesis in
vitro (6). Simultaneously,
these authors discovered that miR-1297 may be directly involved in
the downregulation of phosphatase and tensin homolog in Hep-2-cells
and solid tumors. Moreover, Wu et al demonstrated that
overexpression of miR-16 contributed to the cell migration of Hep-2
laryngeal cancer cells and suppression of intercellular adhesion.
Additionally, zyxin, which was negatively regulated by miR-16, was
confirmed to be a direct target gene of miR-16 (7). On this basis, we predicted that
miRNA-mediated regulation of cellular and genetic expression may be
an appropriate treatment option for malignant tumors.
Human telomerase reverse transcriptase (hTERT), a
catalytic subunit of telomerase, is considered to be the most
significant component of the telomerase complex together with the
telomerase RNA component (8,9).
Telomerase activity is crucial in the immortality of cancer cells
(e.g. Hep-2 cancer cells) through continuously adding the telomeric
repeats onto the G-overhang of the 3′ termini of telomeres
(10). Research reveals that only
one study has investigated the correlation between the expression
of miRNA and hTERT (11), in which
miR-138 induced a reduction in hTERT protein in human anaplastic
thyroid carcinoma cell lines. Therefore, it is still unclear which
of the miRNAs could exert a strong suppressive effect on the
expression of hTERT in cancer cells.
In the present study, hTERT expression was found to
be inversely correlated with the endogenous expression of
miR-299-3p in Hep-2 and U2OS cells; moreover, overexpression of
miR-299-3p inhibited the expression of hTERT mRNA and protein by
directly targeting the 3′-UTR of hTERT mRNA. Furthermore,
downregulation of hTERT mediated by miR-299-3p mimic inhibited
Hep-2 cell growth. The results of this study may be of use in the
treatment of human laryngeal cancer.
Materials and methods
Cell culture and transfection
Hep-2 cells and U2OS cells were obtained from the
Cell Bank of Type Culture Collection of the Chinese Academy of
Science (Shanghai, China). The cells were maintained in Dulbecco’s
modified Eagle’s medium (Sigma-Aldrich, St. Louis, MO, USA)
containing 10% fetal bovine serum (Sigma-Aldrich) at 37°C in 5%
CO2 at a humidity of 95%. Transfection of Hep-2 cells
was performed with Lipofectamine 2000 (Invitrogen Life
Technologies, Carlsbad, CA, USA) following the manufacturer’s
instructions. Transfections were performed in triplicate for each
experiment.
Prediction of miRNAs targeting hTERT
To predict the miRNAs that target hTERT and the
conserved sites bound by the seed region of these miRNAs, different
target prediction programs were used, including TargetScan
(http://www.targetscan.org/), miRanda
(http://www.microrna.org/microrna/home.do) and
starBase(http://starbase.sysu.edu.cn/).
Quantitative polymerase chain reaction
(qPCR)
Total RNA was extracted from Hep-2 cells or U2OS
cells using TRIzol reagent (Invitrogen Life Technologies), strictly
following the instructions. The synthesis of cDNA was carried out
using the RevertAid First strand cDNA synthesis kit (Fermentas;
Thermo Fisher Scientific, Waltham, MA, USA). qPCR was performed
using TaqMan miRNA assay kits (Applied Biosystems, Foster City, CA,
USA) on an Applied Biosystems 7500 thermal cycler. The expression
of miRNA was normalized using glyceraldehyde 3-phosphate
dehydrogenase (GAPDH). The primers for hTERT were
5′-CTTCCACTCCCCACATAGGA-3′ (hTERT-F) and 5′-GTA
CAGGGCACACCTTTGGT-3′ (hTERT-R). The primers for GAPDH were
5′-AGAAGGCTGGGGCTCATTTG-3′ (GAPDH-F) and 5′-AGGGGCCATCCACAGTCTTC-3′
(GAPDH-R). PCR amplification was conducted in a volume of 20
μl containing 3 μl RT product, 10 μl 1X SYBR
Select master mix and 7 μl all-in-one miRNA qPCR primer. The
reaction conditions were as follows: 50°C for 2 min and 95°C for 20
sec, followed by 40 cycles of 95°C for 10 sec, 60°C for 34 sec and
70°C for 10 sec. Melting analysis was carried out at the end of the
amplification cycle to verify non-specific amplification. The
expression level was quantified using the 2−ΔΔCt
method.
Construction of luciferase reporter
plasmids and luciferase assays
The 3′-UTR segments of hTERT containing the
miR-299-3p binding sites were amplified by PCR from Hela cell
genomic DNA. The primers were 5′-CCGCTCGAGTGGCCACCCGCCCACAG-3′
(hTERT-F) and 5′-GTTTAGCGGCCGCTTTTACTCCCACAGCACCTCC-3′ (hTERT-R).
Next, the PCR product was cloned into the XhoI/NotI
sites downstream of the Renilla luciferase (Rluc) reporter
gene pmiR-RB-Report™ vector (RiboBio, Guangzhou, China), which was
named pmiR-hTERT-3′-UTR-wt. Mutations in the predicted miR-299-3p
binding sites were established using pmiR-hTERT-3′-UTR-wt as a
template, and was named pmiR-hTERT-3′-UTR-mut. The pmiR-RB-Report
vector containing a synthetic Renilla luciferase (hRluc)
gene and a synthetic firefly luciferase (hluc) gene encoding
firefly luciferase was used as the internal control. The
recombinant plasmid, designated as pmiR-RB-hTERT-3′-UTR, was
confirmed by restriction enzyme digestion and DNA sequencing. Hela
cells were co-transfected in 96-well plates with miR-299-3p mimic
or negative control (RiboBio), pmiR-hTERT-3′-UTR-wt or
pmiR-hTERT-3′-UTR-mut by Lipofectamine 2000. Forty-eight hours
after transfection, luciferase activity was measured using the dual
luciferase assay system (Promega, Heidelberg, Germany). The firefly
luciferase activity of each sample was normalized to Renilla
luciferase activity.
Western blot analysis
Western blot analysis was performed as routinely
described. Briefly, the protein was homogenized in modified RIPA
buffer containing 0.5% sodium dodecyl sulfate (SDS) in the presence
of proteinase inhibitor cocktail (Complete mini; Roche Diagnostics,
Basel, Switzerland). Proteins (40 μg) were separated by
electrophoresis on a 10% SDS-PAGE gel and transferred to a PVDF
membrane (Millipore, Billerica, MA, USA). Subsequently, the
membrane was blocked in 5% non-fat milk and incubated with rabbit
anti-hTERT antibody (1:1000 dilution; Epitomics, Burlingame, CA,
USA) at 4°C overnight, followed by incubation with a secondary
antibody (1:5000; LI-COR, Lincoln, NE, USA) for 4 h at room
temperature. The same membrane was probed for GAPDH (1:1000
dilution; Epitomics) as the loading control. The relative density
of hTERT to GAPDH was analyzed with an Odyssey® infrared
imaging system (LI-COR).
CCK-8 assay
Hep-2 Cells were seeded in a 96-well plate at a
concentration of 103 cells per well. Following
transfection, the cells were maintained at 37°C for 24, 48 and 72
h, respectively. The cells were treated with CCK-8 (Dojindo Inc.,
Kunamoto, Japan) for 4 h at 37°C. Subsequently, the medium was
removed and the pellet was dissolved in 100 μl DMSO.
Finally, the number of cells was determined using a microplate
reader at a wavelength of 450 nm.
Statistical analysis
SPSS 11.5 software (SPSS Inc., Chicago, IL, USA) was
used for the data analysis. Student’s t-test was performed to
analyze the differences between the data obtained from three
independent experiments. P<0.05 was considered to indicate a
statistically significant difference.
Results
hTERT expression is inversely correlated
with endogenous expression of miR-299-3p in Hep-2 and U2OS
cells
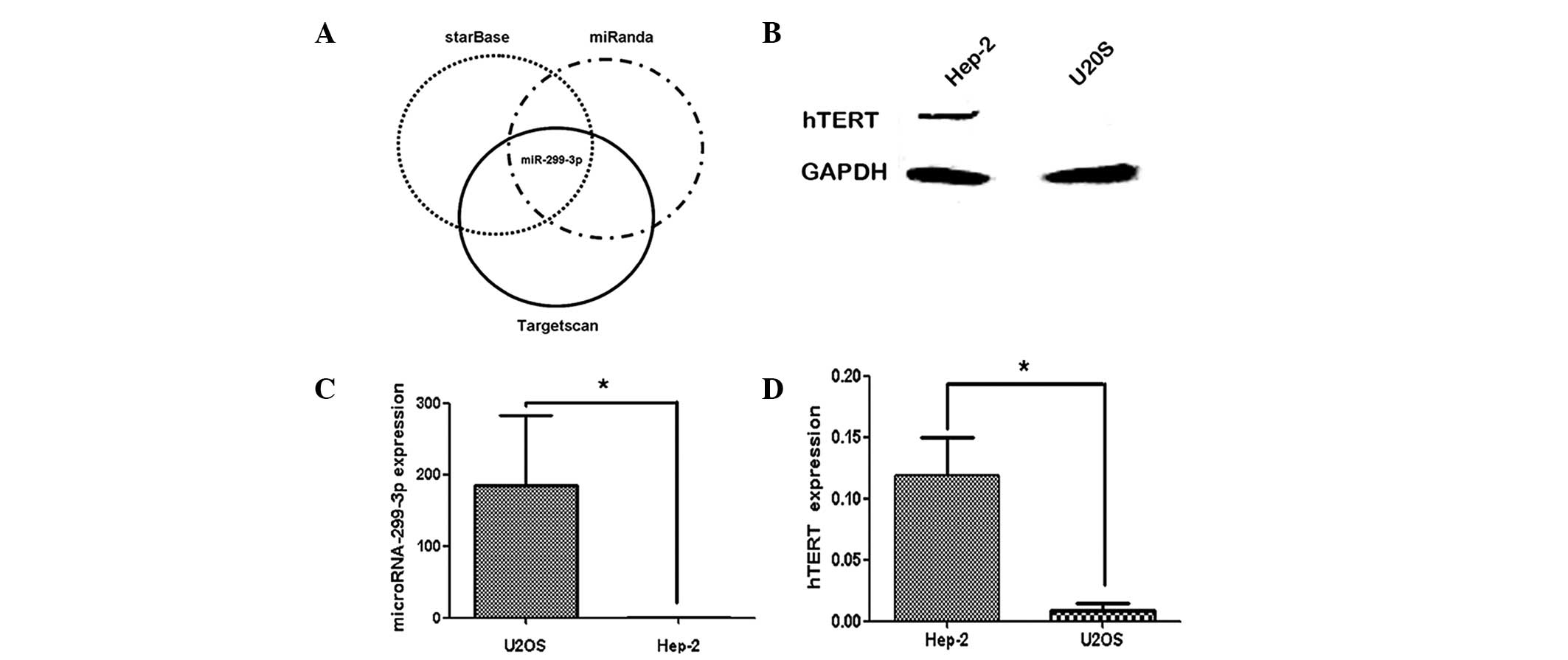
Fig. 1 shows the
correlation between miR-299-3p expression and hTERT protein in
Hep-2 and U2OS cells. Western blot analysis revealed that hTERT was
positively expressed in the Hep-2 cells, while it was negatively
expressed in U2OS cells (Fig. 1B).
In addition, qPCR was used to measure the expression of miR-299-3p
in the two cell lines. As shown in Fig. 1C, higher levels of miR-299-3p were
observed in U2OS cell lines compared with those in Hep-2 cells. An
inverse correlation was observed between the expression of
miR-299-3p and hTERT protein in these two cell lines. These results
indicated that the high expression of hTERT in Hep-2 cell lines was
inversely correlated with the endogenous expression of
miR-299-3p.
miR-299-3p regulates the expression of
hTERT in Hep-2 cells
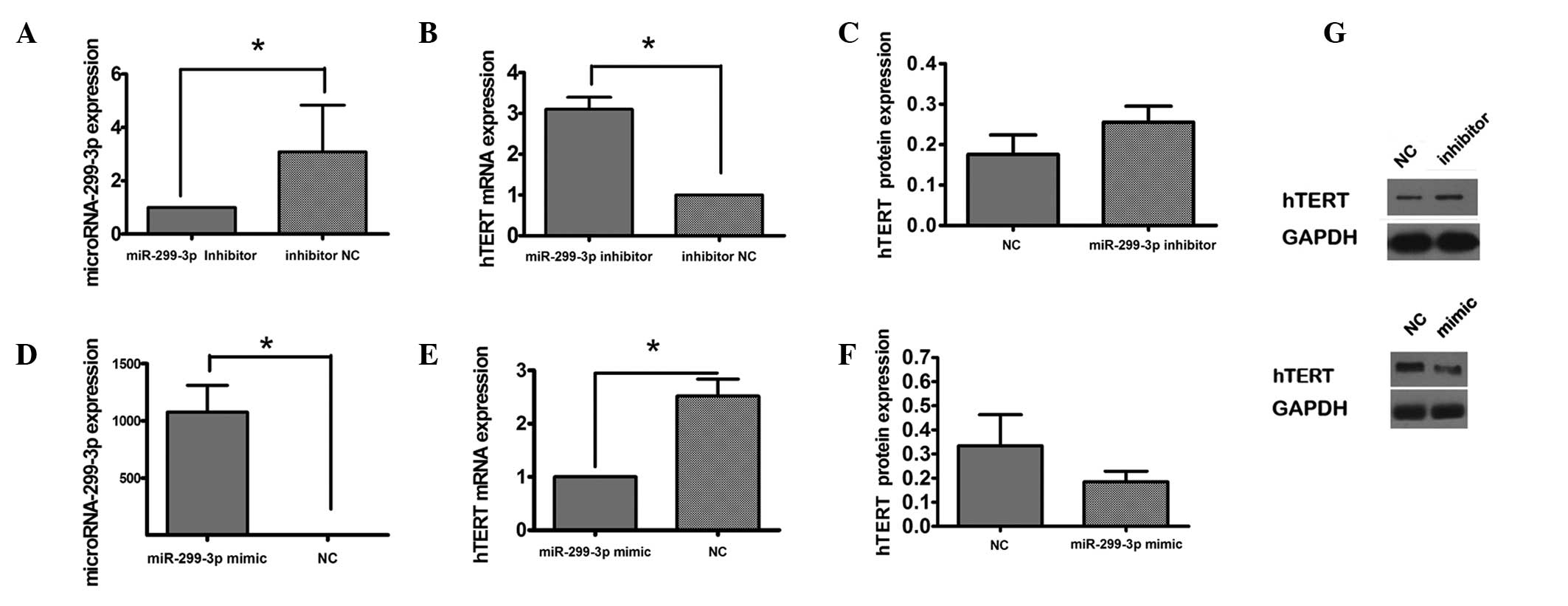
To investigate the association between miR-299-3p
and hTERT, the expression of hTERT was measured following knockdown
of miR-299-3p in vitro (Fig.
2A). RT-qPCR analysis demonstrated that transfection of Hep-2
cells with miR-299-3p inhibitor contributed to the expression of
hTERT mRNA compared with the control group transfected with a
scrambled inhibitor negative control (Fig. 2B). In addition, the expression of
hTERT protein was increased in the presence of miR-299-3p
downregulation (Fig. 2C). The
effect of overexpression of miR-299-3p induced by miR-299-3p mimic
on the expression of hTERT protein was investigated by RT-qPCR and
western blot analysis (Fig. 2D–F).
It was observed that upregulation of miR-299-3p reduced the
expression of hTERT mRNA and protein. These results indicated that
miR-299-3p negatively regulated hTERT expression.
hTERT is a direct target of
miR-299-3p
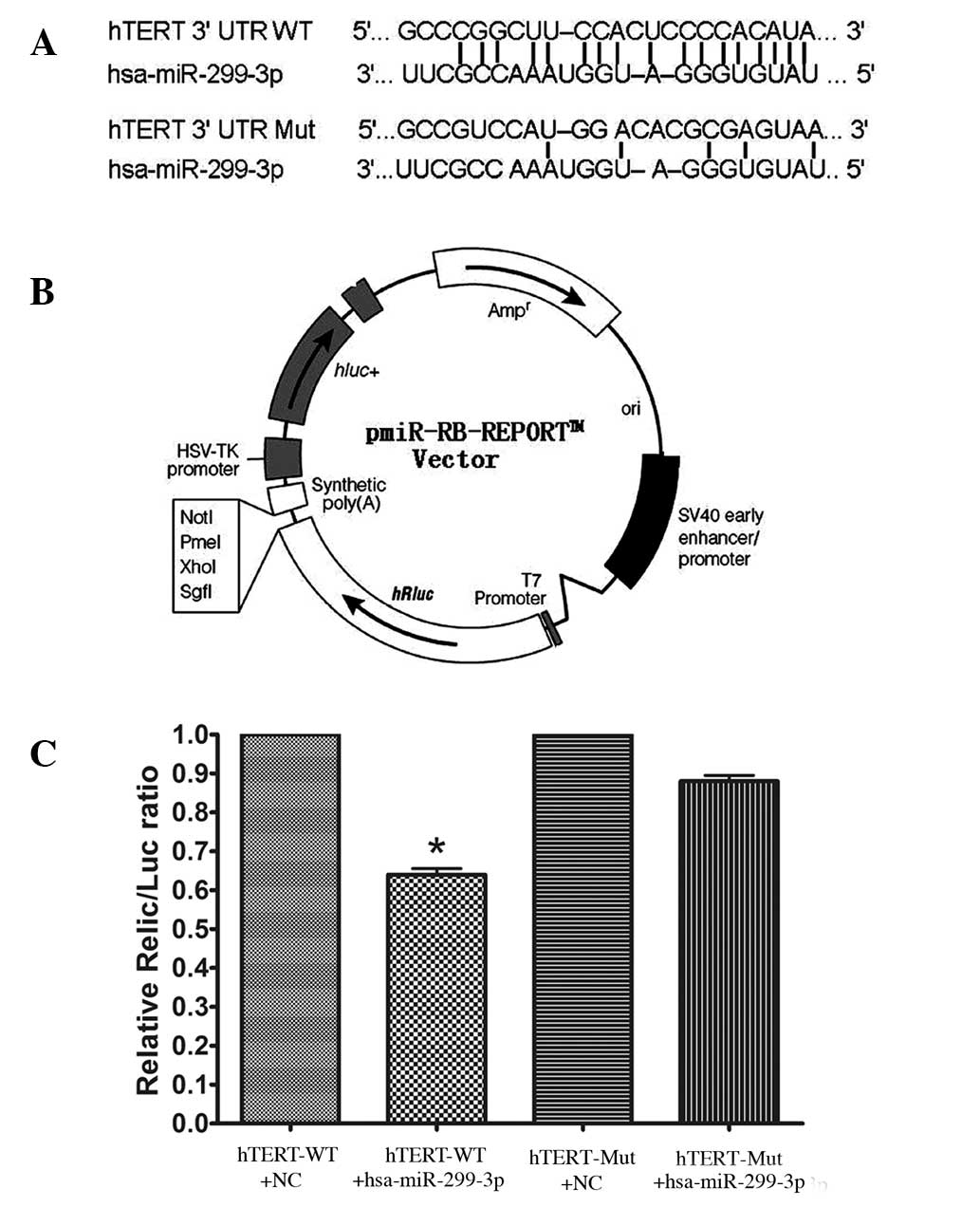
In Hela cells co-transfected with the reporter
plasmids and miR-299-3p mimic or negative control duplex, the
activity of the luciferase reporter containing wild-type 3′-UTR was
significantly suppressed by miR-299-3p mimic. To be precise, the
luciferase activity declined by 36% in comparison with the negative
control (P<0.05). However, the luciferase activity of the mutant
reporter recovered by 24% compared with the wild type, indicating
that miR-299-3p may suppress the expression of hTERT by binding
with the 3′-UTR of hTERT mRNA. Together, the results revealed that
miR-299-3p regulated the expression of endogenous human hTERT by
directly targeting the 3′-UTR of hTERT mRNA (as shown in Fig. 3).
miR-299-3p affects the growth of Hep-2
cancer cells
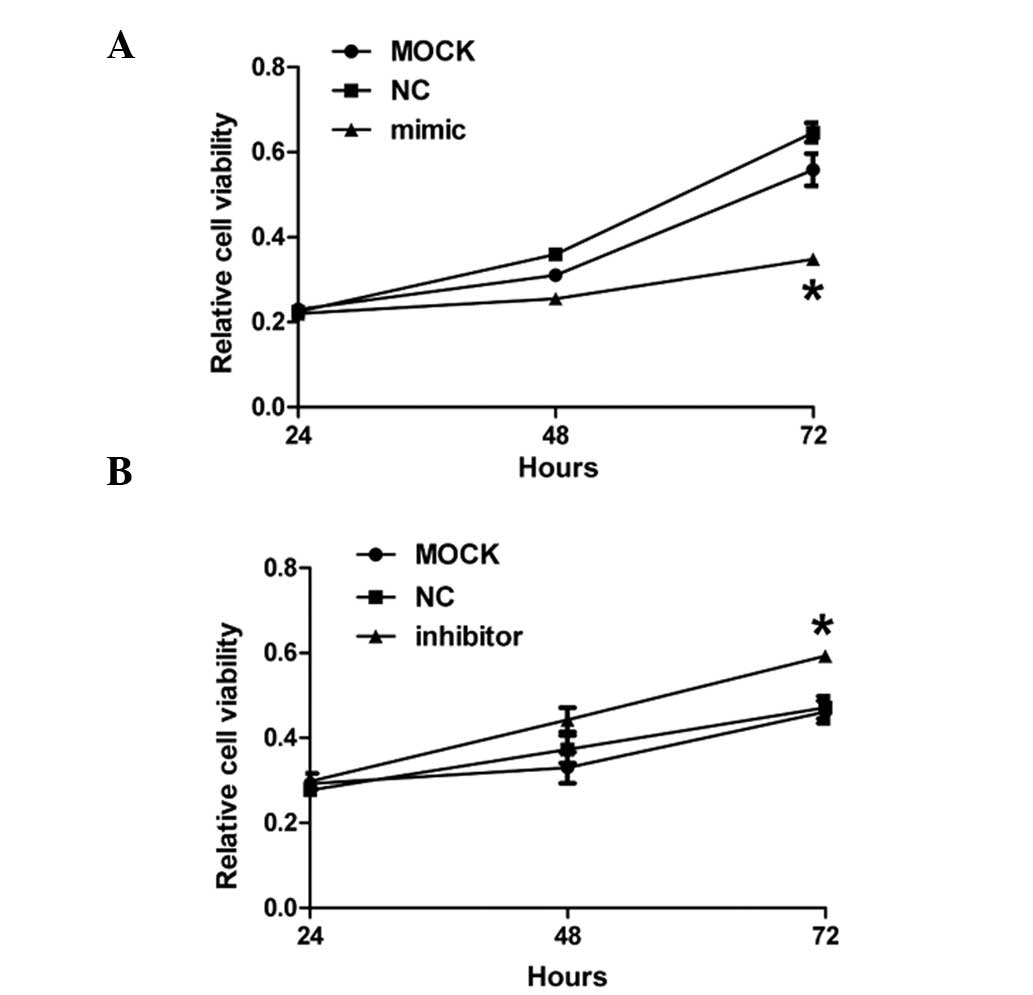
To investigate the effects of miR-299-3p
overexpression on the growth of Hep-2 cancer cells, CCK-8 assay was
performed on Hep-2 cells 24, 48 and 72 h after treatment with
miR-299-3p mimic, inhibitor and negative control, respectively. As
demonstrated in Fig. 4A, the
growth of Hep-2 cancer cells notably decreased 72 h after
transfection of miR-299-3p mimic compared with the mock and
negative control. However, downregulation of miR-299-3p contributed
to a significant increase in cell growth compared with the other
two groups 72 h after transfection with miR-299-3p inhibitor. All
of these results demonstrated that miR-299-3p affected the growth
of Hep-2 cancer cells.
Discussion
Our study firstly demonstrated that miR-299-3p may
significantly downregulate the expression of hTERT in human Hep-2
cells. Compared with the hTERT-negative U2OS cells, overexpression
of miR-299-3p was noted in hTERT-positive Hep-2 cells. Thus,
miR-299-3p may act as a tumor suppressor in Hep-2 cancer cells.
As the core catalytic subunit of the enzyme
telomerase, hTERT protein has a significant effect on
tumorigenesis. Previous studies have shown that telomerase is
activated at various levels, including the transcriptional and
post-transcriptional levels of hTERT (8,12).
In addition, a positive correlation has been reported between hTERT
expression and telomerase activity in a variety of cancer cell
lines (13,14). To our knowledge, several protein
kinases were involved in the post-transcriptional modification of
hTERT protein; for instance, AKT kinase and protein kinase C have
been reported to mediate the phosphorylation of hTERT protein,
leading to activation of telomerase activity (15,16).
In addition, certain accessory proteins (e.g. 14-3-3 protein and
nuclear factor-κB) may modulate telomerase activity by interacting
with hTERT protein (17,18). miRNA has always been considered to
be one of the key factors in the post-transcriptional mechanism.
However, few studies have noted the suppressive effects of miRNA on
hTERT expression (11). To
identify a miRNA that can directly target hTERT in human laryngeal
cancer, we examined the expression of miR-299-3p and its effects on
hTERT in human Hep-2 laryngeal cancer cells.
Computational algorithms reveal that miR-299-3p may
directly target the binding site within the 3′-UTR of hTERT through
base pairing, as it revealed high complementarity to the 3′-UTR of
hTERT mRNA. To confirm that hTERT was a target of miR-299-3p, we
cloned the hTERT 3′-UTR containing the target sequence, or a
fragment with several mutations in the target site, into a
luciferase reporter vector, respectively. Our results demonstrated
that miR-299-3p suppressed the luciferase activity of
pmiR-hTERT-3′-UTR-wt by 36% compared with the negative control. The
suppressive effect was blocked when treating with the vector
integrated with mutation of the miR-299-3p binding sites. Moreover,
the expression of endogenous hTERT mRNA and protein decreased in
miR-299-3p mimic-transfected Hep-2 cells, suggesting that
miR-299-3p may regulate hTERT expression by inducing mRNA
degradation and translational suppression. Finally, downregulation
of miR-299-3p promotes the proliferation of Hep-2 cells. All of
these results demonstrate that overexpression of miR-299-3p
inhibits Hep-2 cell growth by targeting the 3′-UTR of the hTERT
mRNA.
Taken together, our study suggests that miR-299-3p
has crucial roles in the progression of Hep-2 cancer cells by
interacting with hTERT mRNA. Therefore, miR-299-3p may act as a
useful diagnostic tool, and its application in gene therapy in
laryngeal cancer is promising.
Acknowledgments
This study was funded by the National Natural
Science Foundation of China (nos. 30901662 and 81172569) and the
Science and Technology Program of Hubei Province of China (no.
302161991).
References
|
1
|
Cummins JM, He Y, Leary RJ, et al: The
colorectal microRNAome. Proc Natl Acad Sci USA. 103:3687–3692.
2006. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
He L and Hannon GJ: MicroRNAs: small RNAs
with a big role in gene regulation. Nat Rev Genet. 5:522–531. 2004.
View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Felekkis K, Touvana E, Stefanou C and
Deltas C: microRNAs: a newly described class of encoded molecules
that play a role in health and disease. Hippokratia. 14:236–240.
2010.
|
|
4
|
Esquela-Kerscher A and Slack FJ: Oncomirs
- microRNAs with a role in cancer. Nat Rev Cancer. 6:259–269. 2006.
View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Calin GA and Croce CM: MicroRNA signatures
in human cancers. Nat Rev Cancer. 6:857–866. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Li X, Wang HL, Peng X, Zhou HF and Wang X:
miR-1297 mediates PTEN expression and contributes to cell
progression in LSCC. Biochem Biophys Res Commun. 427:254–260. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Wu H, Liu T, Wang R, et al: MicroRNA-16
targets zyxin and promotes cell motility in human laryngeal
carcinoma cell line HEp-2. IUBMB Life. 63:101–108. 2011.PubMed/NCBI
|
|
8
|
Daniel M, Peek GW and Tollefsbol TO:
Regulation of the human catalytic subunit of telomerase (hTERT).
Gene. 498:135–146. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Nakano K, Watney E and McDougall JK:
Telomerase activity and expression of telomerase RNA component and
telomerase catalytic subunit gene in cervical cancer. Am J Pathol.
153:857–864. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Smith LL, Coller HA and Roberts JM:
Telomerase modulates expression of growth-controlling genes and
enhances cell proliferation. Nat Cell Biol. 5:474–479. 2003.
View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Mitomo S, Maesawa C, Ogasawara S, et al:
Downregulation of miR-138 is associated with overexpression of
human telomerase reverse transcriptase protein in human anaplastic
thyroid carcinoma cell lines. Cancer Sci. 99:280–286. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Guilleret I, Yan P, Grange F, et al:
Hypermethylation of the human telomerase catalytic subunit (hTERT)
gene correlates with telomerase activity. Int J Cancer.
101:335–341. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Boldrini L, Faviana P, Gisfredi S, et al:
Evaluation of telomerase mRNA (hTERT) in colon cancer. Int J Oncol.
21:493–497. 2002.PubMed/NCBI
|
|
14
|
Ducrest AL, Szutorisz H, Lingner J, et al:
Regulation of the human telomerase reverse transcriptase gene.
Oncogene. 21:541–552. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Kang SS, Kwon T, Kwon DY and Do SI: Akt
protein kinase enhances human telomerase activity through
phosphorylation of telomerase reverse transcriptase subunit. J Biol
Chem. 274:13085–13090. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Kim YW, Hur SY, Kim TE, et al: Protein
kinase C modulates telomerase activity in human cervical cancer
cells. Exp Mol Med. 33:156–163. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Akiyama M, Hideshima T, Hayashi T, et al:
Nuclear factor-kappaB p65 mediates tumor necrosis factor
alpha-induced nuclear translocation of telomerase reverse
transcriptase protein. Cancer Res. 63:18–21. 2003.PubMed/NCBI
|
|
18
|
Seimiya H, Sawada H, Muramatsu Y, et al:
Involvement of proteins in nuclear localization of telomerase. EMBO
J. 19:2652–2661. 2000. View Article : Google Scholar : PubMed/NCBI
|