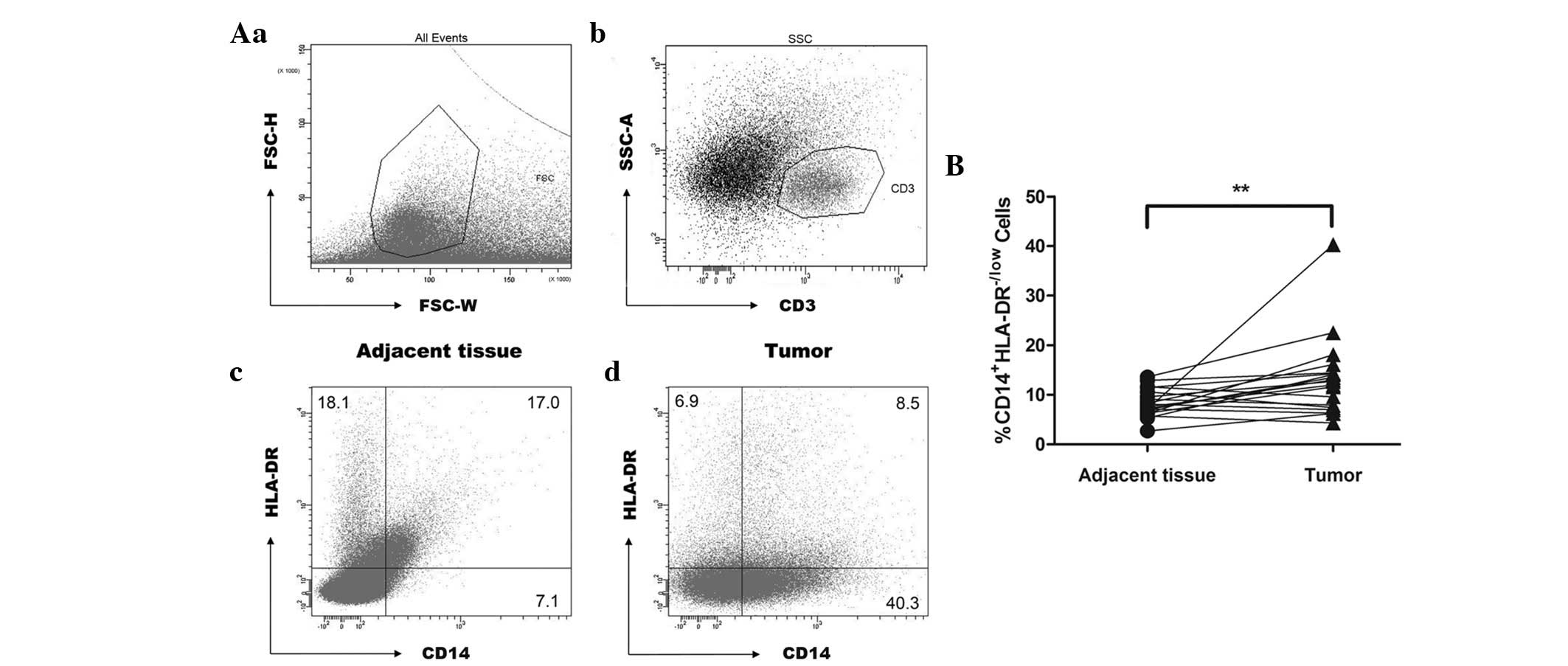
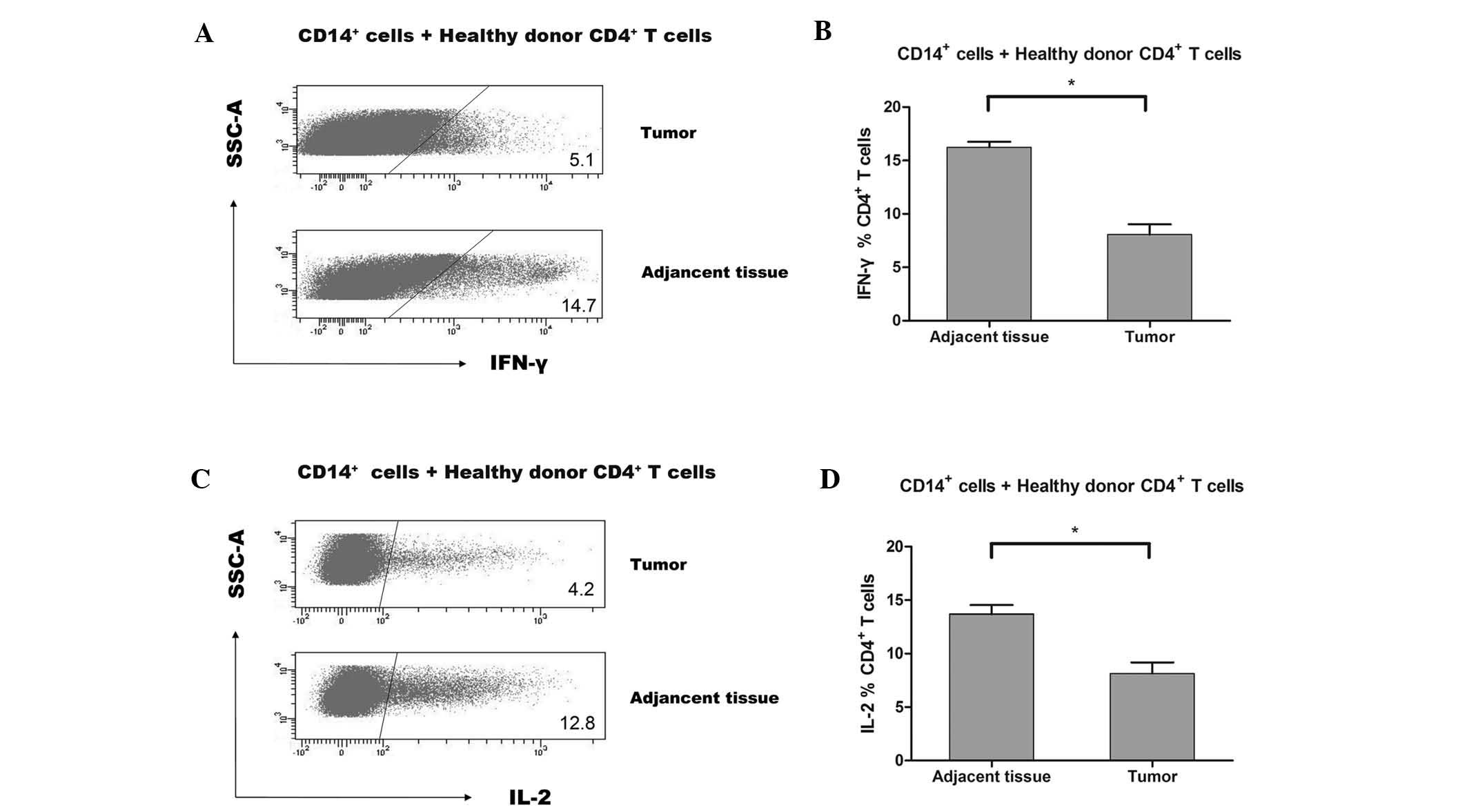
|
1
|
Almand B, Clark JI, Nikitina E, et al:
Increased production of immature myeloid cells in cancer patients:
a mechanism of immunosuppression in cancer. J Immunol. 166:678–689.
2001. View Article : Google Scholar
|
|
2
|
Bronte V, Serafini P, Apolloni E and
Zanovello P: Tumor-induced immune dysfunctions caused by myeloid
suppressor cells. J Immunother. 24:431–446. 2001. View Article : Google Scholar
|
|
3
|
Young MR, Petruzzelli GJ, Kolesiak K,
Achille N, Lathers DM and Gabrilovich DI: Human squamous cell
carcinomas of the head and neck chemoattract immune suppressive
CD34(+) progenitor cells. Hum Immunol. 62:332–341. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Huang A, Zhang B, Wang B, Zhang F, Fan KX
and Guo YJ: Increased CD14(+)HLA-DR (−/low) myeloid-derived
suppressor cells correlate with extrathoracic metastasis and poor
response to chemotherapy in non-small cell lung cancer patients.
Cancer Immunol Immunother. 62:1439–1451. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Zea AH, Rodriguez PC, Atkins MB, et al:
Arginase-producing myeloid suppressor cells in renal cell carcinoma
patients: a mechanism of tumor evasion. Cancer Res. 65:3044–3048.
2005.PubMed/NCBI
|
|
6
|
Hanson EM, Clements VK, Sinha P, Ilkovitch
D and Ostrand-Rosenberg S: Myeloid-derived suppressor cells
downregulate L-selectin expression on CD4+ and
CD8+ T cells. J Immunol. 183:937–944. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Gabrilovich DI and Nagaraj S:
Myeloid-derived suppressor cells as regulators of the immune
system. Nat Rev Immunol. 9:162–174. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Duffy A, Zhao F, Haile L, et al:
Comparative analysis of monocytic and granulocytic myeloid-derived
suppressor cell subsets in patients with gastrointestinal
malignancies. Cancer Immunol Immunother. 62:299–307. 2013.
View Article : Google Scholar
|
|
9
|
Hoechst B, Ormandy LA, Ballmaier M, et al:
A new population of myeloid-derived suppressor cells in
hepatocellular carcinoma patients induces CD4(+)CD25(+)Foxp3(+) T
cells. Gastroenterology. 135:234–243. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Khaled YS, Ammori BJ and Elkord E:
Myeloid-derived suppressor cells in cancer: recent progress and
prospects. Immunol Cell Biol. 91:493–502. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Lechner MG, Liebertz DJ and Epstein AL:
Characterization of cytokine-induced myeloid-derived suppressor
cells from normal human peripheral blood mononuclear cells. J
Immunol. 185:2273–2284. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Kusmartsev S and Gabrilovich DI: Effect of
tumor-derived cytokines and growth factors on differentiation and
immune suppressive features of myeloid cells in cancer. Cancer
Metastasis Rev. 25:323–331. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Fernandez-Martinez AB, Collado B, Bajo AM,
Sanchez-Chapado M, Prieto JC and Carmena MJ: Vasoactive intestinal
peptide induces cyclooxygenase-2 expression through nuclear
factor-kappaB in human prostate cell lines Differential
time-dependent responses in cancer progression. Mol Cell
Endocrinol. 270:8–16. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Valdehita A, Carmena MJ, Bajo AM and
Prieto JC: RNA interference-directed silencing of VPAC1 receptor
inhibits VIP effects on both EGFR and HER2 transactivation and VEGF
secretion in human breast cancer cells. Mol Cell Endocrinol.
348:241–246. 2012. View Article : Google Scholar
|
|
15
|
Sotomayor S, Carmena MJ, Schally AV, et
al: Transactivation of HER2 by vasoactive intestinal peptide in
experimental prostate cancer: Antagonistic action of an analog of
growth-hormone-releasing hormone. Int J Oncol. 31:1223–1230.
2007.PubMed/NCBI
|
|
16
|
Couvineau A and Laburthe M: VPAC
receptors: structure, molecular pharmacology and interaction with
accessory proteins. Br J Pharmacol. 166:42–50. 2012. View Article : Google Scholar :
|
|
17
|
Laburthe M, Couvineau A and Tan V: Class
II G protein-coupled receptors for VIP and PACAP: structure, models
of activation and pharmacology. Peptides. 28:1631–1639. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Pozo D, Anderson P and Gonzalez-Rey E:
Induction of alloantigen-specific human T regulatory cells by
vasoactive intestinal peptide. J Immunol. 183:4346–4359. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Wu K, Kryczek I, Chen L, Zou W and Welling
TH: Kupffer cell suppression of CD8+ T cells in human
hepatocellular carcinoma is mediated by B7-H1/programmed death-1
interactions. Cancer Res. 69:8067–8075. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Kryczek I, Wu K, Zhao E, et al: IL-17+
regulatory T cells in the microenvironments of chronic inflammation
and cancer. J Immunol. 186:4388–4395. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Vuk-Pavlovic S, Bulur PA, Lin Y, et al:
Immunosuppressive CD14+HLA-DRlow/- monocytes in prostate cancer.
Prostate. 70:443–455. 2010.
|
|
22
|
Lechner MG, Megiel C, Russell SM, et al:
Functional characterization of human Cd33+ and Cd11b+
myeloid-derived suppressor cell subsets induced from peripheral
blood mononuclear cells co-cultured with a diverse set of human
tumor cell lines. J Translat Med. 9:902011. View Article : Google Scholar
|
|
23
|
Youn JI and Gabrilovich DI: The biology of
myeloid-derived suppressor cells: the blessing and the curse of
morphological and functional heterogeneity. Eur J Immunol.
40:2969–2975. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Li JM, Southerland L, Hossain MS, et al:
Absence of vasoactive intestinal peptide expression in
hematopoietic cells enhances Th1 polarization and antiviral
immunity in mice. J Immunol. 187:1057–1065. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Sinha P, Clements VK, Bunt SK, Albelda SM
and Ostrand-Rosenberg S: Cross-talk between myeloid-derived
suppressor cells and macrophages subverts tumor immunity toward a
type 2 response. J Immunol. 179:977–983. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Huang B, Pan PY, Li Q, et al: Gr-1+CD115+
immature myeloid suppressor cells mediate the development of
tumor-induced T regulatory cells and T-cell anergy in tumor-bearing
host. Cancer Res. 66:1123–1131. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Porembka MR, Mitchem JB, Belt BA, et al:
Pancreatic adenocarcinoma induces bone marrow mobilization of
myeloid-derived suppressor cells which promote primary tumor
growth. Cancer Immunol Immunother. 61:1373–1385. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Movahedi K, Guilliams M, Van den Bossche
J, et al: Identification of discrete tumor-induced myeloid-derived
suppressor cell subpopulations with distinct T cell-suppressive
activity. Blood. 111:4233–4244. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Youn JI, Nagaraj S, Collazo M and
Gabrilovich DI: Subsets of myeloid-derived suppressor cells in
tumor-bearing mice. J Immunol. 181:5791–5802. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Poschke I, Mougiakakos D, Hansson J,
Masucci GV and Kiessling R: Immature immunosuppressive
CD14+HLA-DR−/low cells in melanoma patients
are Stat3hi and overexpress CD80, CD83 and DC-sign. Cancer Res.
70:4335–4345. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Mantovani A, Sozzani S, Locati M, Allavena
P and Sica A: Macrophage polarization: tumor-associated macrophages
as a paradigm for polarized M2 mononuclear phagocytes. Trends
Immunol. 23:549–555. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Colombo MP and Piconese S:
Regulatory-T-cell inhibition versus depletion: the right choice in
cancer immunotherapy. Nat Rev Cancer. 7:880–887. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Brimnes MK, Vangsted AJ, Knudsen LM, et
al: Increased level of both CD4+FOXP3+ regulatory T
cells and CD14+HLA-DR (−)/low myeloid-derived suppressor cells and
decreased level of dendritic cells in patients with multiple
myeloma. Scand J Immunol. 72:540–547. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Rodrigues JC, Gonzalez GC, Zhang L, et al:
Normal human monocytes exposed to glioma cells acquire
myeloid-derived suppressor cell-like properties. Neuro Oncol.
12:351–365. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Gonzalez-Rey E and Delgado M: Vasoactive
intestinal peptide and regulatory T-cell induction: a new mechanism
and therapeutic potential for immune homeostasis. Trends Mol Med.
13:241–251. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Szilasi M, Buglyo A, Treszl A, Kiss L,
Schally AV and Halmos G: Gene expression of vasoactive intestinal
peptide receptors in human lung cancer. Int J Oncol. 39:1019–1024.
2011.PubMed/NCBI
|
|
37
|
Veljkovic M, Dopsaj V, Dopsaj M, et al:
Physical activity and natural anti-VIP antibodies: potential role
in breast and prostate cancer therapy. PloS one. 6:e283042011.
View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Lippitz BE: Cytokine patterns in patients
with cancer: a systematic review. Lancet Oncol. 14:e218–e228. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Boytim M1, Lilly P, Drouvalakis K, et al:
A human class II MHC-derived peptide antagonizes
phosphatidylinositol 3-kinase to block IL-2 signaling. J Clin
Invest. 105:1447–1453. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Szema AM, Hamidi SA, Golightly MG, Rueb TP
and Chen JJ: VIP Regulates the Development & Proliferation of
Treg in vivo in spleen. Allergy Asthma Clin Immunol. 7:192011.
View Article : Google Scholar
|
|
41
|
Dumitriu IE, Dunbar DR, Howie SE, Sethi T
and Gregory CD: Human dendritic cells produce TGF-beta 1 under the
influence of lung carcinoma cells and prime the differentiation of
CD4+CD25+Foxp3+ regulatory T cells. J Immunol.
182:2795–2807. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Vicari AP, Chiodoni C, Vaure C, et al:
Reversal of tumor-induced dendritic cell paralysis by CpG
immunostimulatory oligonucleotide and anti-interleukin 10 receptor
antibody. J Exp Med. 196:541–549. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Toscano MG, Delgado M, Kong W, Martin F,
Skarica M and Ganea D: Dendritic cells transduced with lentiviral
vectors expressing VIP differentiate into VIP-secreting
tolerogenic-like DCs. Mol Ther. 18:1035–1045. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Laborde RR, Lin Y, Gustafson MP, Bulur PA
and Dietz AB: Cancer vaccines in the world of immune suppressive
monocytes (CD14HLA-DR Cells): The gateway to improved responses.
Front Immunol. 5:1472014. View Article : Google Scholar
|
|
45
|
Trinchieri G: Interleukin-12 and the
regulation of innate resistance and adaptive immunity. Nat Rev
Immunol. 3:133–146. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Williams LM, Ricchetti G, Sarma U, Smallie
T and Foxwell BM: Interleukin-10 suppression of myeloid cell
activation-a continuing puzzle. Immunology. 113:281–292. 2004.
View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Hu X, Chen J, Wang L and Ivashkiv LB:
Crosstalk among Jak-STAT, Toll-like receptor and ITAM-dependent
pathways in macrophage activation. J Leukoc Biol. 82:237–243. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Finbloom DS and Winestock KD: IL-10
induces the tyrosine phosphorylation of tyk2 and Jak1 and the
differential assembly of STAT1 alpha and STAT3 complexes in human T
cells and monocytes. J Immunol. 155:1079–1090. 1995.PubMed/NCBI
|
|
49
|
Kusmartsev S, Nagaraj S and Gabrilovich
DI: Tumor-associated CD8+T cell tolerance induced by bone
marrow-derived immature myeloid cells. J Immunol. 175:4583–4592.
2005. View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Gabitass RF, Annels NE, Stocken DD, Pandha
HA and Middleton GW: Elevated myeloid-derived suppressor cells in
pancreatic, esophageal and gastric cancer are an independent
prognostic factor and are associated with significant elevation of
the Th2 cytokine interleukin-13. Cancer Immunol Immunother.
60:1419–1430. 2011. View Article : Google Scholar : PubMed/NCBI
|