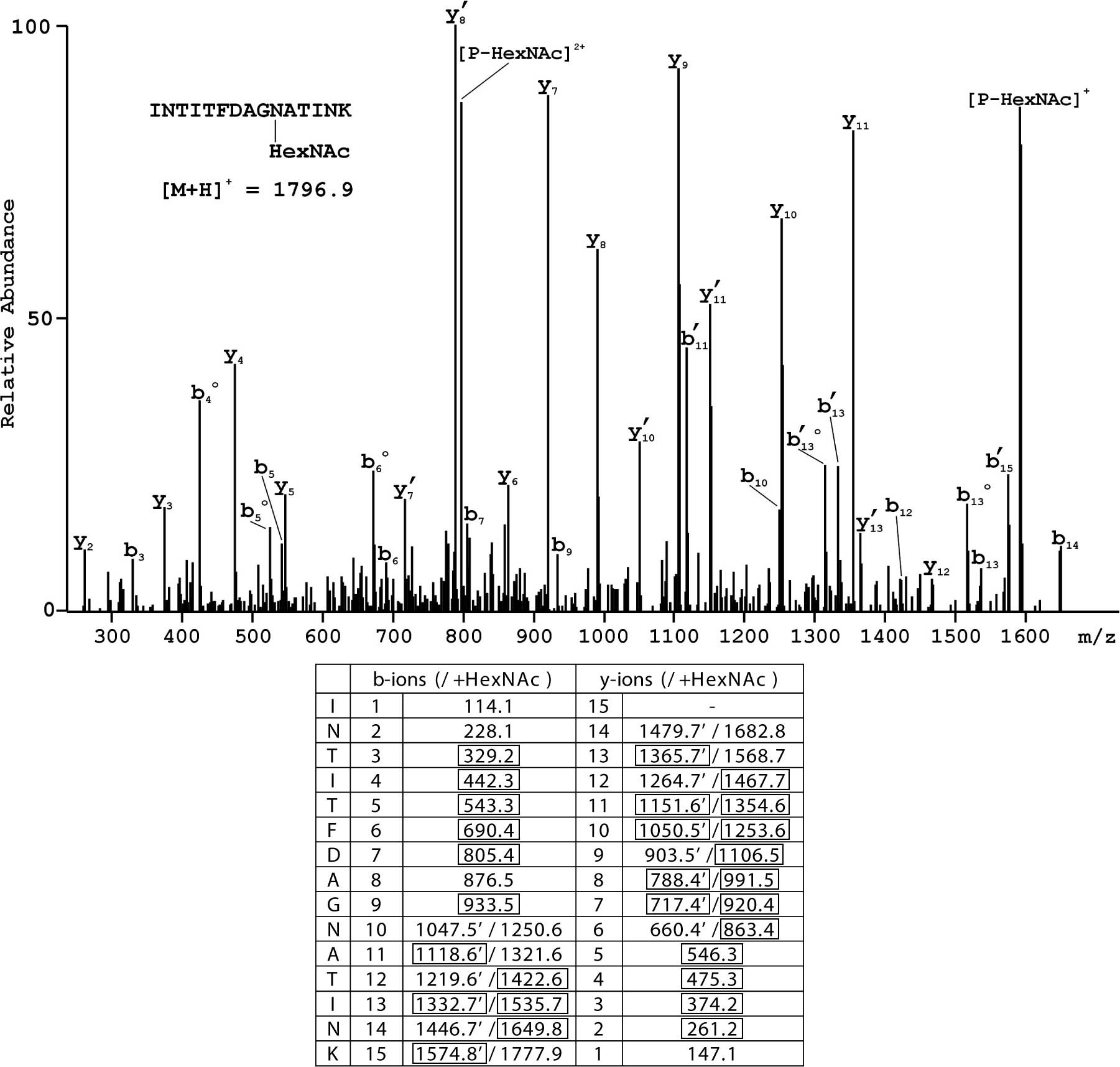
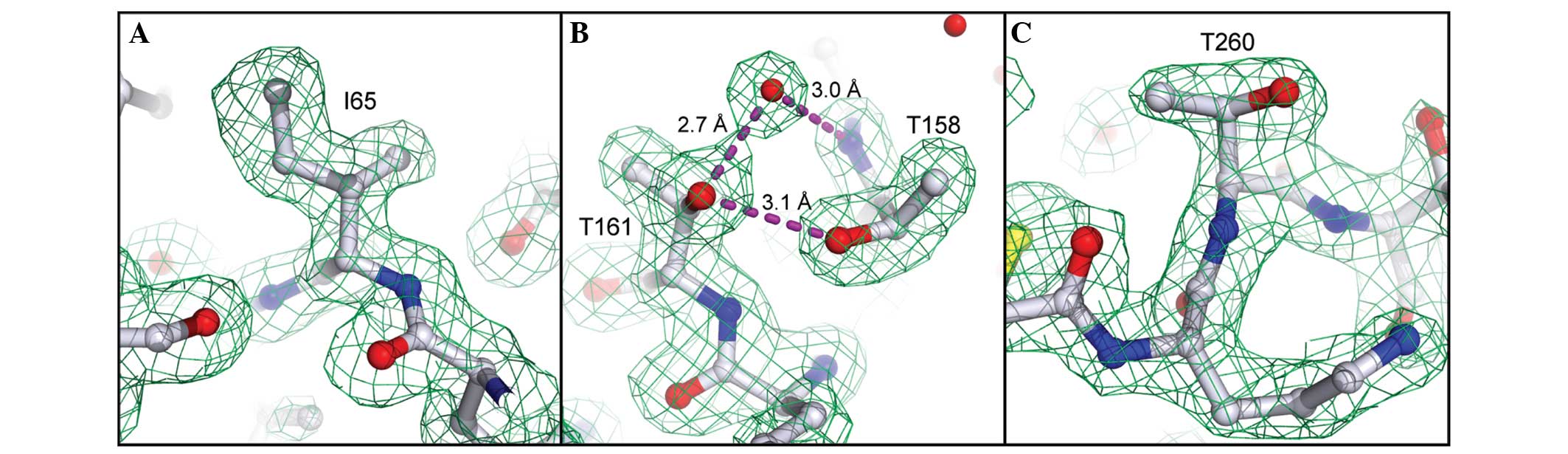
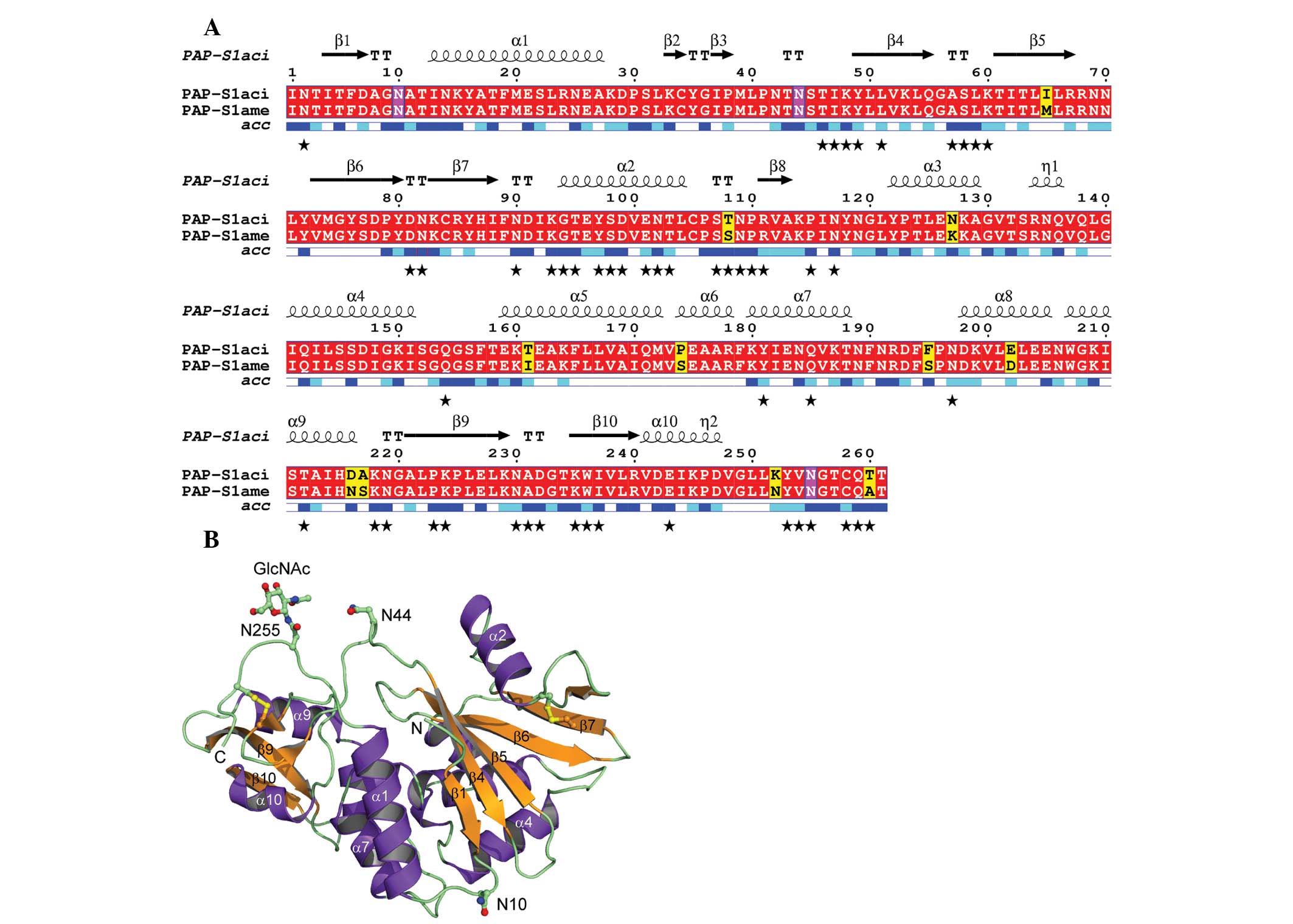
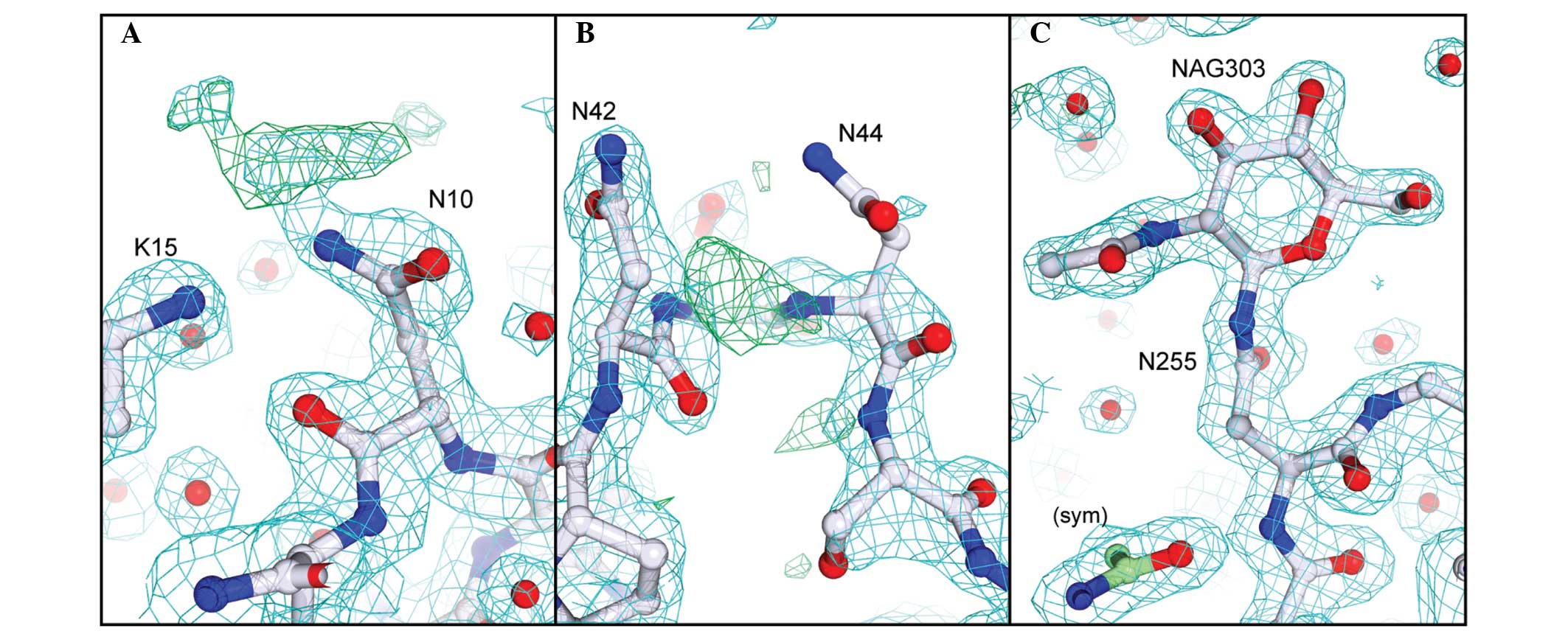
|
1
|
Puri M, Kaur I, Perugini MA and Gupta RC:
Ribosome-inactivating proteins: current status and biomedical
applications. Drug Discov Today. 17:774–783. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Stirpe F: Ribosome-inactivating proteins:
from toxins to useful proteins. Toxicon. 67:12–16. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Endo Y, Tsurugi K and Lambert JM: The site
of action of six different ribosome-inactivating proteins from
plants on eukaryotic ribosomes: the RNA N-glycosidase activity of
the proteins. Biochem Biophys Res Commun. 150:1032–1036. 1988.
View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Gessner SL and Irvin JD: Inhibition of
elongation factor 2-dependent translocation by the pokeweed
antiviral protein and ricin. J Biol Chem. 255:3251–3253.
1980.PubMed/NCBI
|
|
5
|
Stirpe F and Battelli MG:
Ribosome-inactivating proteins: progress and problems. Cell Mol
Life Sci. 63:1850–1866. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Rajamohan F, Kurinov IV, Venkatachalam TK
and Uckun FM: Deguanylation of human immunodeficiency virus (HIV-1)
RNA by recombinant pokeweed antiviral protein. Biochem Biophys Res
Commun. 263:419–424. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Hudak KA, Bauman JD and Tumer NE: Pokeweed
antiviral protein binds to the cap structure of eukaryotic mRNA and
depurinates the mRNA downstream of the cap. RNA. 8:1148–1159. 2002.
View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Parikh BA, Coetzer C and Tumer NE:
Pokeweed antiviral protein regulates the stability of its own mRNA
by a mechanism that requires depurination but can be separated from
depurination of the alpha-sarcin/ricin loop of rRNA. J Biol Chem.
277:41428–41437. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Parikh BA and Tumer NE: Antiviral activity
of ribosome inactivating proteins in medicine. Mini Rev Med Chem.
4:523–543. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Mansouri S, Choudhary G, Sarzala PM,
Ratner L and Hudak KA: Suppression of human T-cell leukemia virus I
gene expression by pokeweed antiviral protein. J Biol Chem.
284:31453–31462. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
D'Cruz OJ, Waurzyniakt B and Uckun FM: A
13-week subchronic intravaginal toxicity study of pokeweed
antiviral protein in mice. Phytomedicine. 11:342–351. 2004.
View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Uckun FM, Rustamova L, Vassilev AO,
Tibbles HE and Petkevich AS: CNS activity of pokeweed anti-viral
protein (PAP) in mice infected with lymphocytic choriomeningitis
virus (LCMV). BMC Infect Dis. 5:92005. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Shapira A and Benhar I: Toxin-based
therapeutic approaches. Toxins (Basel). 2:2519–2583. 2010.
View Article : Google Scholar
|
|
14
|
Benigni F, Canevari S, Gadina M, Adobati
E, Ferreri AJ, Di Celle EF, Comolli R and Colnaghi MI: Preclinical
evaluation of the ribosome-inactivating proteins PAP-1, PAP-S and
RTA in mice. Int J Immunopharmacol. 17:829–839. 1995. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Monzingo AF, Collins EJ, Ernst SR, Irvin
JD and Robertus JD: The 2.5 A structure of pokeweed antiviral
protein. J Mol Biol. 233:705–715. 1993. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Kurinov IV, Myers DE, Irvin JD and Uckun
FM: X-ray crystallographic analysis of the structural basis for the
interactions of pokeweed antiviral protein with its active site
inhibitor and ribosomal RNA substrate analogs. Protein Sci.
8:1765–1772. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Zeng ZH, He XL, Li HM, Hu Z and Wang DC:
Crystal structure of pokeweed antiviral protein with well-defined
sugars from seeds at 1.8 A resolution. J Struct Biol. 141:171–178.
2003. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Hogg T, Kuta Smatanova I, Bezouska K,
Ulbrich N and Hilgenfeld R: Sugar-mediated lattice contacts in
crystals of a plant glycoprotein. Acta Crystallogr D Biol
Crystallogr. 58:1734–1739. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Brune DC: Alkylation of cysteine with
acrylamide for protein sequence analysis. Anal Biochem.
207:285–290. 1992. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Perkins DN, Pappin DJ, Creasy DM and
Cottrell JS: Probability-based protein identification by searching
sequence databases using mass spectrometry data. Electrophoresis.
20:3551–3567. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Read RJ: Improved Fourier coefficients for
maps using phases from partial structures with errors. Acta Cryst
A. 42:140–149. 1986. View Article : Google Scholar
|
|
22
|
Emsley P and Cowtan K: Coot:
model-building tools for molecular graphics. Acta Crystallogr D
Biol Crystallogr. 60:2126–2132. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Brünger AT, Adams PD, Clore GM, DeLano WL,
Gros P, Grosse-Kunstleve RW, Jiang JS, Kuszewski J, Nilges M, Pannu
NS, et al: Crystallography & NMR system: A new software suite
for macromolecular structure determination. Acta Crystallogr D Biol
Crystallogr. 54:905–921. 1998. View Article : Google Scholar
|
|
24
|
Lovell SC, Davis IW, Arendall WB 3rd, de
Bakker PI, Word JM, Prisant MG, Richardson JS and Richardson DC:
Structure validation by Calpha geometry: phi, psi and Cbeta
deviation. Proteins. 50:437–450. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Kabsch W and Sander C: Dictionary of
protein secondary structure: pattern recognition of hydrogen-bonded
and geometrical features. Biopolymers. 22:2577–2637. 1983.
View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Kabsch W: A solution for the best rotation
to relate two sets of vectors. Acta Cryst A. 32:922–923. 1976.
View Article : Google Scholar
|
|
27
|
Collaborative Computational Project,
Number 4: The CCP4 suite: programs for protein crystallography.
Acta Crystallogr D Biol Crystallogr. 50:760–763. 1994. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
DeLano WL: The PyMOL Molecular Graphics
System. DeLano Scientific; Palo Alto, USA: 2002, http://www.pymol.org.
|
|
29
|
McGinnis S and Madden TL: BLAST: at the
core of a powerful and diverse set of sequence analysis tools.
Nucleic Acids Res. 32:W20–W25. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Honjo E, Dong D, Motoshima H and Watanabe
K: Genomic clones encoding two isoforms of pokeweed antiviral
protein in seeds (PAP-S1 and S2) and the N-glycosidase activities
of their recombinant proteins on ribosomes and DNA in comparison
with other isoforms. J Biochem. 131:225–231. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Corpet F: Multiple sequence alignment with
hierarchical clustering. Nucl Acids Res. 16:10881–10890. 1988.
View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Gouet P, Courcelle E, Stuart DI and Métoz
F: ESPript: analysis of multiple sequence alignments in PostScript.
Bioinformatics. 15:305–308. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Rajamohan F, Pugmire MJ, Kurinov IV and
Uckun FM: Modeling and alanine scanning mutagenesis studies of
recombinant pokeweed antiviral protein. J Biol Chem. 275:3382–3390.
2000. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Altschul SF, Gish W, Miller W, Myers EW
and Lipman DJ: Basic local alignment search tool. J Mol Biol.
215:403–410. 1990. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Hogg T and Hilgenfeld R: Protein
crystallography in drug discovery. Comprehensive Medicinal
Chemistry II. Taylor JB, Triggle DJ and Kubinyi H: 3. 2nd edition.
Elsevier; Amsterdam: pp. 875–900. 2007, View Article : Google Scholar
|
|
36
|
Ho BK and Brasseur R: The Ramachandran
plots of glycine and pre-proline. BMC Struct Biol. 5:142005.
View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Hase S, Fujimura K, Kanoh M and Ikenaka T:
Studies on heterogeneity of Taka-amylase A: isolation of an amylase
having one N-acetylglucosamine residue as the sugar chain. J
Biochem. 92:265–270. 1982.PubMed/NCBI
|
|
38
|
Chalkley RJ, Thalhammer A, Schoepfer R and
Burlingame AL: Identification of protein O-GlcNAcylation sites
using electron transfer dissociation mass spectrometry on native
peptides. Proc Natl Acad Sci USA. 106:8894–8899. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Wang Z, Udeshi ND, Slawson C, Compton PD,
Sakabe K, Cheung WD, Shabanowitz J, Hunt DF and Hart GW: Extensive
crosstalk between O-GlcNAcylation and phosphorylation regulates
cytokinesis. Sci Signal. 3:ra22010.PubMed/NCBI
|
|
40
|
Kim YC, Jahren N, Stone MD, Udeshi ND,
Markowski TW, Witthuhn BA, Shabanowitz J, Hunt DF and Olszewski NE:
Identification and origin of N-linked β-D-N-acetylglucosamine
monosaccharide modifications on Arabidopsis proteins. Plant
Physiol. 161:455–464. 2013. View Article : Google Scholar :
|
|
41
|
Islam MR, Kung SS, Kimura Y and Funatsu G:
N-acetyl-D-glu cosamine-asparagine structure in
ribosome-inactivating proteins from the seeds of Luffa cylindrica
and Phytolacca americana. Agric Biol Chem. 55:1375–1381. 1991.
View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Wells L, Whelan SA and Hart GW: O-GlcNAc:
a regulatory post-translational modification. Biochem Biophys Res
Commun. 302:435–441. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Khalkhall Z and Marshall RD: Glycosylation
of ribonuclease A catalysed by rabbit liver extracts. Biochem J.
146:299–307. 1975. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Khalkhali Z and Marshall RD:
UDP-N-acetyl-D-glucosamine-as paragine sequon
N-acetyl-b-D-glucosaminyl-transferase-activity in human serum.
Carbohydr Res. 49:455–473. 1976. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Khalkhali Z, Marshall RD, Reuvers F,
Habets-Willems C and Boer P: Glycosylation in vitro of an
asparagine sequon catalysed by preparations of yeast cell
membranes. Biochem J. 160:37–41. 1976. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Rajamohan F, Mao C and Uckun FM: Binding
interactions between the active center cleft of recombinant
pokeweed antiviral protein and the alpha-sarcin/ricin stem loop of
ribosomal RNA. J Biol Chem. 276:24075–24081. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Bonness MS, Ready MP, Irvin JD and Mabry
TJ: Pokeweed antiviral protein inactivates pokeweed ribosomes;
implications for the antiviral mechanism. Plant J. 5:173–183. 1994.
View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Poyet JL and Hoeveler A: cDNA cloning and
expression of pokeweed antiviral protein from seeds in Escherichia
coli and its inhibition of protein synthesis in vitro. FEBS Lett.
406:97–100. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Barbieri L, Battelli MG and Stirpe F:
Ribosome-inactivating proteins from plants. Biochem Biophys Acta.
1154:237–282. 1993.PubMed/NCBI
|
|
50
|
Gross V, Heinrich PC, vom Berq D, Steube
K, Andus T, Tran-Thi TA, Decker K and Gerok W: Involvement of
various organs in the initial plasma clearance of differently
glycosylated rat liver secretory proteins. Eur J Biochem.
173:653–659. 1988. View Article : Google Scholar : PubMed/NCBI
|