Introduction
Gastric cancer (GC) is one of the most common
cancers of the alimentary tract (1). GC is a major malignancy that
seriously affects patient health and is associated with a high rate
of mortality; with prevalence in men double that in women (2). Research regarding the development of
drugs for the treatment of GC and its complications has recently
gained attention. Chemotherapy is one of major therapeutic methods
for the treatment of cancer; however, cancer gradually evolves
resistance to chemotherapeutic reagents (3). Therefore, the identification of novel
therapeutic strategies is essential to improve the clinical
management of patients with GC. The search for novel therapies has
recently focused on several plants used in traditional medicine
(4–6).
For centuries, traditional Chinese medicine has been
used to treat cancer (7). The
plant Ophiopogon japonicus (Thunb.) Ker-Gawl is an evergreen
perennial medicinal herb, which has been widely used in Southeast
Asia to treat disease for thousands of years (8,9).
O. japonicus contains the following active ingredients:
Saponins, polysaccharides, and homoisoflavonoids (10). This plant has been reported to
exert pharmacological effects, including cardioprotective, diuretic
and antibacterial activities (11). Ophiopogonin B (OP-B), which is
isolated from the traditional Chinese herb Radix O.
japonicus, has been revealed to exert anticancer effects in
human non-small cell lung cancer and cervical cancer (12,13).
However, the role of OP-B in GC has yet to be explored.
The present study aimed to investigate the
anticancer effects of OP-B on cell proliferation, apoptosis,
mitochondrial membrane potential (MMP) and reactive oxygen species
(ROS) in the SGC-7901 GC cell line. In addition, the expression
levels of apoptosis-associated proteins were detected, in order to
explore the mechanisms underlying the effects of OP-B.
Materials and methods
Cell culture and treatment
The SGC-7901 human GC cell line was purchased from
the Cell Bank of Chinese Academy of Sciences (Shanghai, China). The
cells were cultured in RPMI 1640 medium (Hyclone; GE Healthcare
Life Sciences, Logan, UT, USA) supplemented with 10% fetal bovine
serum, penicillin (100 IU/ml) and streptomycin (100 mg/ml) (Gibco;
Thermo Fisher Scientific, Inc., Waltham, MA, USA) at 37°C in a
humidified atmosphere containing 5% CO2. Cells in the
logarithmic phase were used in the subsequent experiments.
Cell viability assay
To detect cell viability following exposure to
various concentrations (0, 5, 10 and 20 mmol/l) of OP-B (Jrdun
Biotechnology Corp., New York, NY, USA), 100 μl SGC-7901
cells (5×104/ml) were seeded in 96-well plates and were
cultured for 0, 12, 24, 48 and 72 h at 37°C. Subsequently, 100
μl serum-free Dulbecco's modified Eagle's medium (Hyclone;
GE Healthcare Life Sciences) containing 10% Cell Counting kit
(CCK)-8 reagents (v/v) (Dojin Laboratories, Kumamoto, Japan) was
added to each well, and the cells were cultured for 1 h in a 5%
CO2 atmosphere at 37°C. Optical density was then
measured at 450 nm using a Model 550 microplate reader (Bio-Rad
Laboratories, Inc., Hercules, CA, USA).
Cell apoptosis assay
Cells were seeded at a density of 5×105
cells/well in 6-well plates, following treatment with OP-B (0, 5,
10 and 20 μmol/l). The cells were subsequently stained using
an Annexin V-fluorescein isothiocyanate (FITC)/propidium iodide kit
(BD Biosciences, Franklin Lakes, NJ, USA). Staining was performed
according to the manufacturer's protocol. Apoptosis of SGC-7091
cells was determined using a FACSCalibur flow cytometer (BD
Biosciences, Franklin Lakes, NJ, USA).
Hoechst staining
Cells were seeded on a 6-well plate at
5×105 cells/well and were treated with OP-B (0, 5, 10
and 20 μmol/l). Subsequently, attached cells were washed
with phosphate-buffered saline (PBS) and were incubated with 10
μg/ml Hoechst 33342 staining solution (Sigma-Aldrich, St.
Louis, MO, USA) for 10 min. Following the incubation, cells were
washed with PBS and anti-fade mounting medium was added. Apoptosis,
as determined by condensed and fragmented nuclei (14), was observed under an RX-DA
fluorescence microscope (Leica Microsystems GmbH, Wetzlar,
Germany).
MMP detection
Rhodamine-123 (Rho-123) dye (Sigma-Aldrich) was used
to detect alterations in MMP. Cells (1×106/ml) were
cultured in a 24-well plate. After a period of exposure to various
concentrations of OP-B, cells were washed with PBS, incubated with
Rho-123 (100 nM), and were subsequently subjected to flow
cytometry.
Detection of ROS
ROS generation was assessed by flow cytometry
(15). Briefly, cells
(5×104/well) were cultured, washed with PBS and were
re-suspended in complete medium. Subsequently, the cells were
incubated with 50 μM DCFH-DA (Vigorous Biotechnology,
Beijing, China). ROS fluorescence intensity was determined by flow
cytometry at an excitation wavelength of 490 nm and an emission
wavelength of 520 nm.
Western blot analysis
Cells were harvested, and total proteins were
extracted using ice-cold radioimmunoprecipitation assay buffer
(Beyotime Institute of Biotechnology). Protein concentration was
subsequently determined using a Bicinchoninic Acid Protein Assay
reagent (Thermo Fisher Scientific, Inc.). Protein samples (40
μg) were separated by sodium dodecyl sulfate-polyacrylamide
gel electrophoresis, and were then transferred to nitrocellulose
membranes. The membranes were blocked in 5% skim milk solution for
1 h at room temperature, then were incubated with rabbit antibodies
against phosphorylated (p)-c-Jun N-terminal kinases (JNK)1/2
(dilution, 1:800; cat. no. 4668; monoclonal), JNK1/2 (dilution,
1:1,000; cat. no. 9252; polyclonal), p-extracellular
signal-regulated kinases (ERK)1/2 (dilution, 1:1,000; cat. no.
4370; monoclonal), ERK1/2 (dilution, 1:1,000; cat. no. 4695;
monoclonal) (all Cell Signaling Technology, Inc., Danvers, MA,
USA), caspase-3 (dilution, 1:1,500; cat. no. ab32351; Abcam,
Cambridge, UK; monoclonal), B-cell lymphoma 2 (Bcl-2; dilution,
1:400; cat. no. sc-492; polyclonal) and Bcl-2-associated X protein
(Bax; dilution, 1:400; cat. no. sc-493; polyclonal) (both Santa
Cruz Biotechnology, Inc., Dallas, TX, USA) overnight at 4°C. This
was followed by incubation with a horseradish peroxidase-conjugated
goat anti-rabbit secondary antibody (dilution, 1:1,000; cat. no.
A0208; Beyotime Institute of Biotechnology, Haimen, China) for 1 h
at room temperature. Rabbit anti-glyceraldehyde 3-phosphate
dehydrogenase (dilution, 1:1,500; cat. no. 5471; monoclonal) was
used as the loading control. The blots were visualized using
enhanced chemiluminescence (EMD Millipore, Billerica, MA, USA) and
analyzed with ImageJ v. 1.49 software (imagej.nih.gov/ij). Western blots presented in the
figures are representative of three independent experiments.
Statistical analysis
All data are presented as the mean ± standard
deviation from three determinations. Data were analyzed with
two-tailed Student's t-tests for comparison with the 0 μmol
group. For statistical analysis, GraphPad Prism 5.0 (GraphPad
Software, Inc., San Diego, CA, USA) was used. P<0.05 was
considered to indicate a statistically significant difference.
Results
Effects of OP-B on proliferation of
SGC-7901 cells
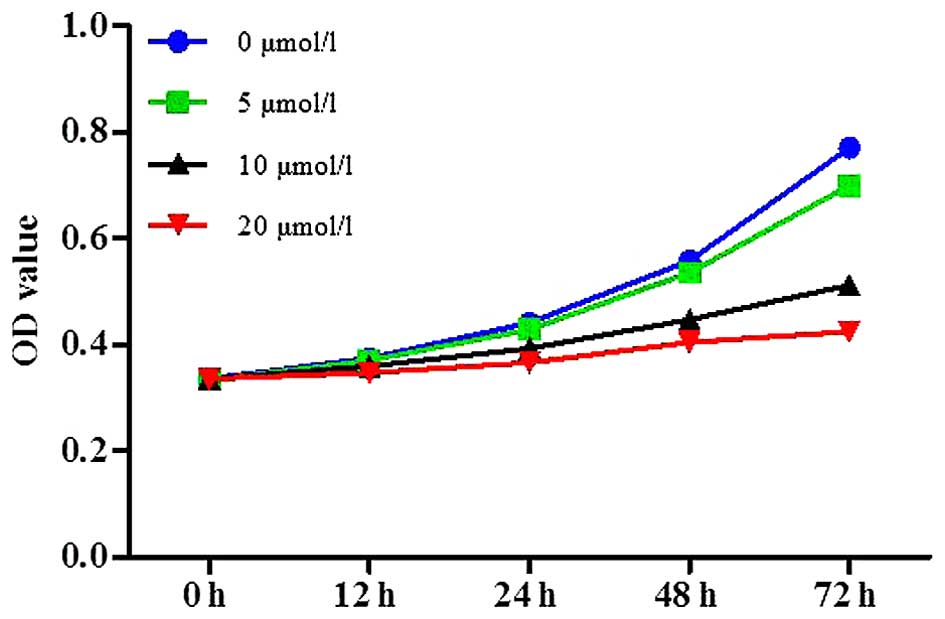
SGC-7901 cells were treated with various
concentrations of OP-B for 0, 12, 24, 48 and 72 h, and the CCK-8
assay was used to measure cell proliferation. As shown in Fig. 1, treatment with OP-B was able to
inhibit GC cell proliferation in a dose-dependent manner.
Effects of OP-B on the apoptosis of
SGC-7901 cells
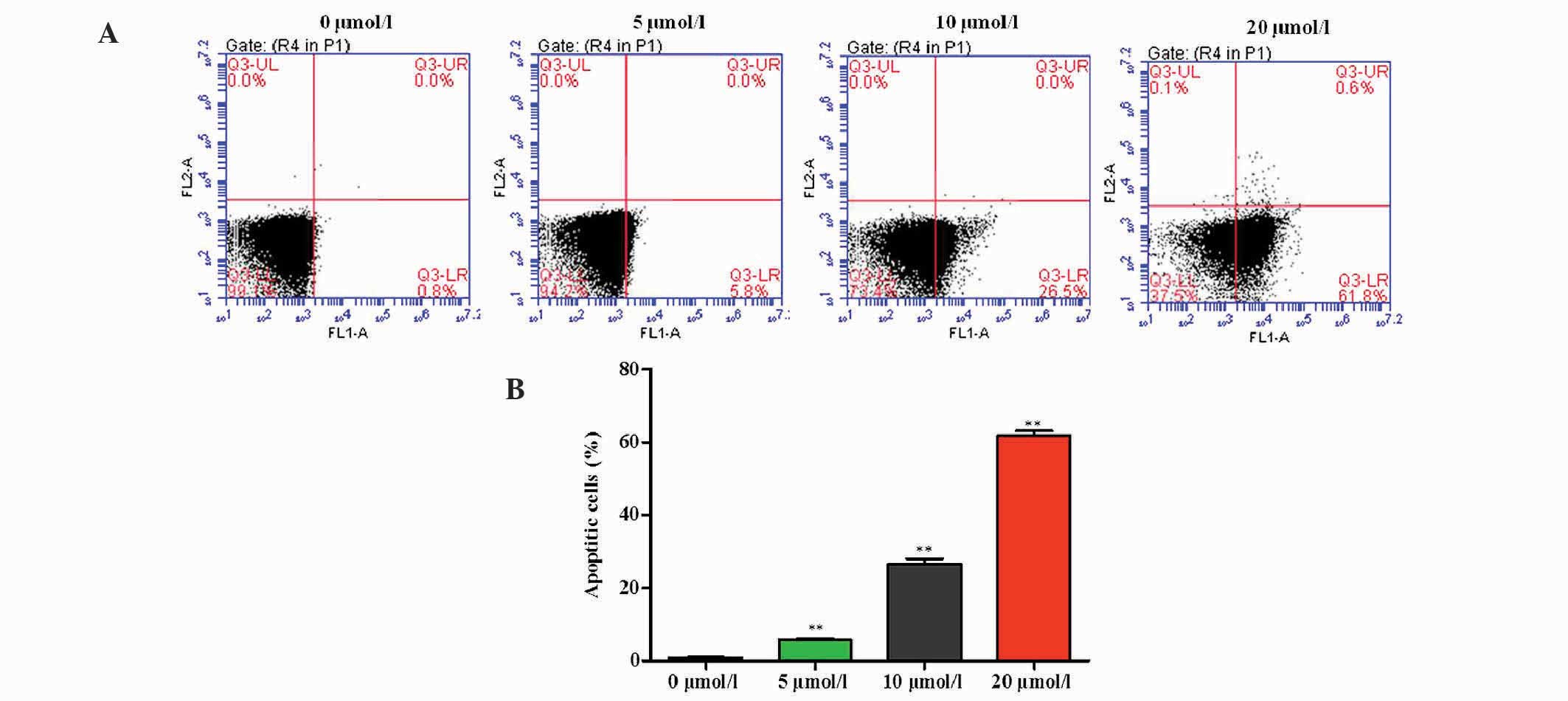
Three different concentrations of OP-B were used to
treat SGC-7901 cells for 24 h. Flow cytometric analysis of Annexin
V-FITC staining demonstrated that OP-B significantly increased the
apoptotic rate of SGC-7901 cells in a dose-dependent manner
(Fig. 2).
Effects of OP-B on morphological
alterations to SGC-7901 cells
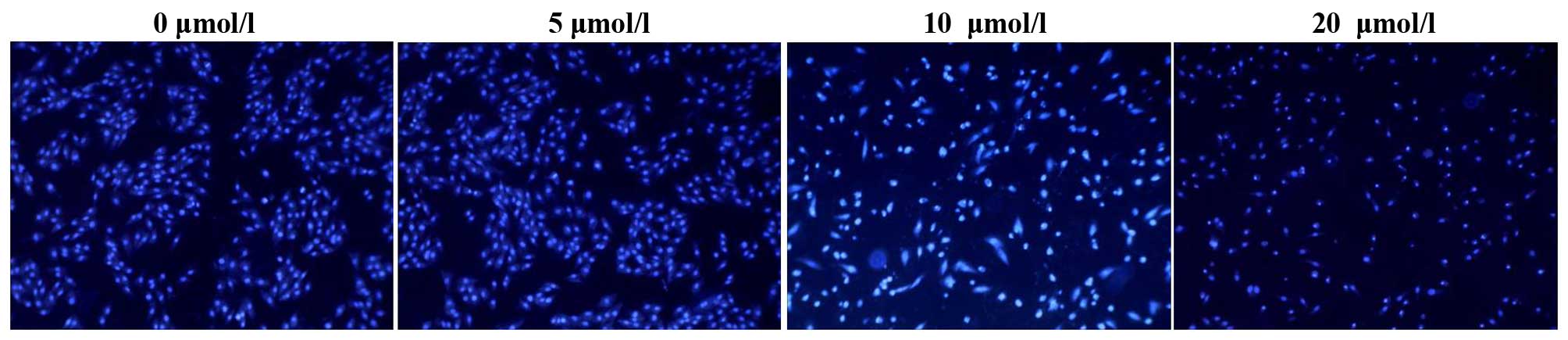
To confirm that OP-B induced apoptosis,
morphological alterations to SGC-7901 cells were observed following
Hoechst 33342 staining. As shown in Fig. 3, following OP-B treatment, blue
emission was much brighter than in the control cells, indicating
the presence of apoptotic cells. In addition, condensed chromatin
was detected in several of the OP-B-treated cells and apoptotic
bodies had formed in some cells.
Effects of OP-B on MMP in SGC-7901
cells
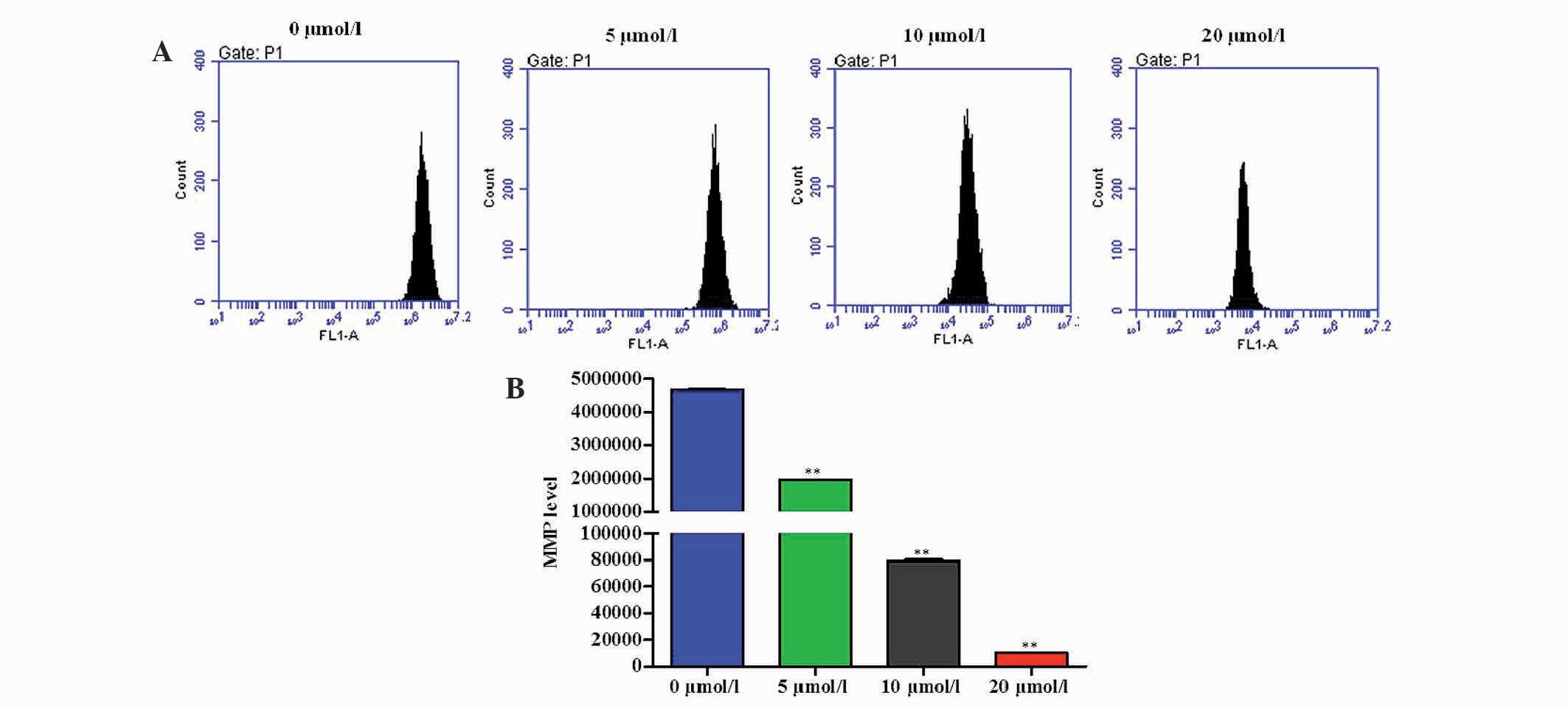
To assess the effects of OP-B on alterations to the
MMP in SGC-7901 cells, flow cytometry was conducted to detect the
fluorescence intensity of Rho-123. As shown in Fig. 4, treatment of SGC-7901 cells with
OP-B (5, 10, and 20 μmol/l) for 24 h resulted in a moderate
depolarization of MMP in a dose-dependent manner.
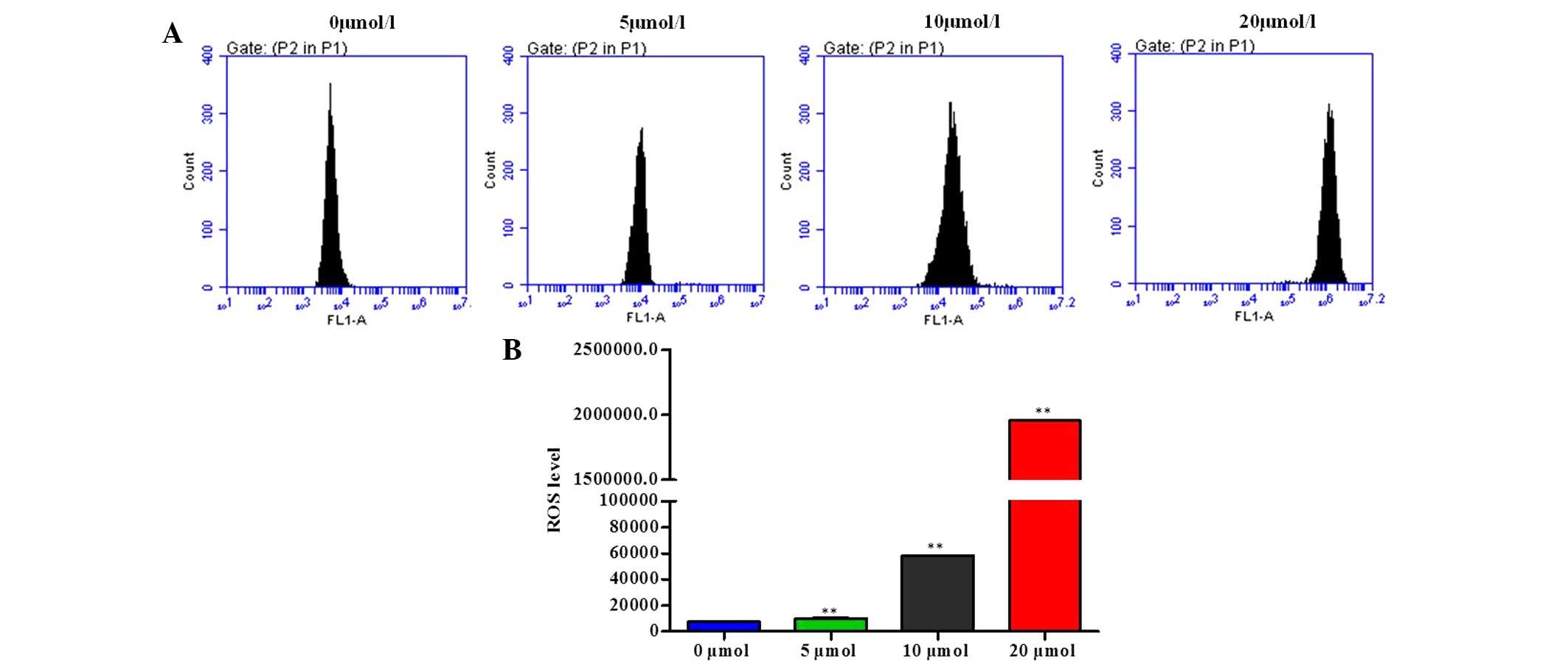
Effects of OP-B on ROS generation in
SGC-7901 cells
The fluorescent probe dichlorodihydrofluorescein
diacetate was used to determine the levels of ROS generation in
SGC-7901 cells. As shown in Fig.
5, SGC-7901 cells exposed to 5, 10, and 20 μmol/l OP-B
for 12 h exhibited a significant increase in the intracellular
accumulation of ROS in a dose-dependent manner.
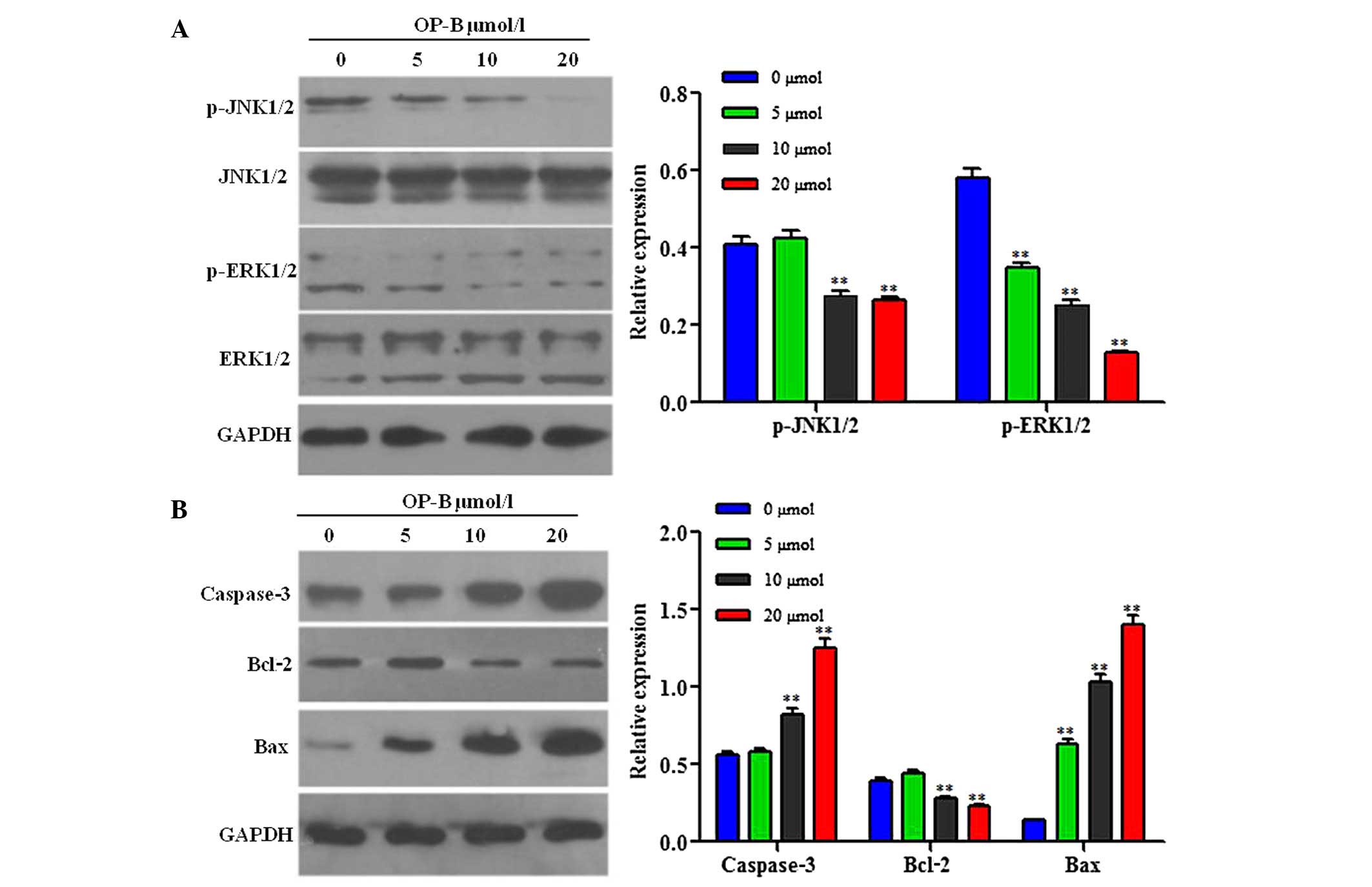
Western blotting results
To clarify the mechanism by which OP-B induced
SGC-7901 cells apoptosis, the expression levels of
apoptosis-associated proteins and the phosphorylation of kinases
were determined by western blotting. As shown in Fig. 6A, treatment with OP-B for 6 h
resulted in down-regulation of the protein expression levels of
p-JNK1/2 and p-ERK1/2. Furthermore, following treatment with OP-B
for 48 h, protein expression levels of caspase-3 and Bax were
increased, whereas the expression levels of Bcl-2 were
down-regulated (Fig. 6B).
Discussion
OP-B is a natural active compound extracted from the
Chinese herbal medicine O. japonicus, which is widely used
in clinical Chinese medicine (16). The present study confirmed that
high concentrations of OP-B were able to significantly inhibit the
proliferation of SGC-7901 cells in a time- and dose-dependent
manner. In addition, apoptotic rates were increased and cell
morphology was altered following OP-B treatment, thus suggesting
that induction of apoptosis may be the main mechanism underlying
the antiproliferative effects of OP-B in SGC-7901 cells.
Apoptosis is a physiological process that leads to
cell self-destruction. Dysregulation of this process is implicated
in various diseases (17).
Mitochondria are important cell organelles, the functions of which
include respiration, oxygen metabolism and enzyme activity, and are
associated with membrane permeability and MMP (18). Breakdown of the MMP is an early
stage of the apoptotic process and precedes nuclear disintegration
(19). In the present study, MMP
fluorescence intensity was gradually reduced in response to high
dose OP-B, thus suggesting that OP-B may induce cell apoptosis by
decreasing MMP. Furthermore, the expression levels of
apoptosis-associated proteins caspase-3 and Bax were significantly
increased, whereas the protein expression levels of Bcl2 were
decreased.
ROS generation and subsequent oxidative stress have
long been implicated in induction of cell apoptosis (20). Mitochondria have been identified as
the major organelles in which ROS are generated (21). As expected, intracellular ROS
levels were significantly increased when SGC-7901 cells were
exposed to OP-B. The MAPK family comprises three main parallel
pathways: ERK, JNK and p38. Previous studies have indicated that
MAPK is an important pathway involved in cell proliferation and
apoptosis (22–24). In the present study, the
phosphorylation levels of JNK1/2 and ERK1/2 were increased in
SGC-7901 cells following OP-B treatment. These results suggested
that the anticancer mechanisms of OP-B may be associated with the
JNK1/2 and ERK1/2 signaling pathways.
The pharmacological assays conducted in the present
study suggested that OP-B may significantly decrease proliferation,
and induce apoptosis of GC cells. In conclusion, OP-B may be
considered a potential candidate agent for the treatment of GC.
Further studies regarding the underlying mechanism of OP-B are
currently ongoing.
References
|
1
|
Jemal A, Bray F, Center MM, Ferlay J, Ward
E and Forman D: Global cancer statistics. CA Cancer J Clin.
61:69–90. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
de Martel C, Forman D and Plummer M:
Gastric cancer: Epidemiology and risk factors. Gastroenterol Clin
North Am. 42:219–240. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Wong H and Yau T: Targeted therapy in the
management of advanced gastric cancer: Are we making progress in
the era of personalized medicine? Oncologist. 17:346–358. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Qi F, Li A, Inagaki Y, Gao J, Li J, Kokudo
N, Li XK and Tang W: Chinese herbal medicines as adjuvant treatment
during chemo-or radio-therapy for cancer. Biosci Trends. 4:297–307.
2010.
|
|
5
|
Li R, Chen WC, Wang WP, Tian WY and Zhang
XG: Extraction, characterization of Astragalus polysaccharides and
its immune modulating activities in rats with gastric cancer.
Carbohydr Polym. 78:738–742. 2009. View Article : Google Scholar
|
|
6
|
Li X, Yang G, Li X, Zhang Y, Yang J, Chang
J, Sun X, Zhou X, Guo Y, Xu Y, et al: Traditional Chinese medicine
in cancer care: A review of controlled clinical studies published
in Chinese. PLoS One. 8:e603382013. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Ling Y: Traditional Chinese medicine in
the treatment of symptoms in patients with advanced cancer. Ann
Palliat Med. 2:141–152. 2013.PubMed/NCBI
|
|
8
|
Boyang Y and Guojun X: Studies on resource
utilization of Chinese drug dwarf lilyturf (Ophiopogon japonicus).
Zhongyao Maidong De Ziyuan Liyong Yanjiu. 26:205–210. 1995.In
Chinese.
|
|
9
|
Xiao P, Li DP and Yang SL: Modern Chinese
materia medica. Chemical Industry Press. 4:253–272. 2002.
|
|
10
|
Kou J, Sun Y, Lin Y, Cheng Z, Zheng W, Yu
B and Xu Q: Anti-inflammatory activities of aqueous extract from
Radix Ophiopogon japonicus and its two constituents. Biol Pharm
Bull. 28:1234–1238. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Lin X, Zhou QF and Xu D: Research progress
of the pharmacological actions of ophiopogon root. Shang Hai Zhong
Yi Yao Za Zhi She. 38:59–61. 2004.In Chinese.
|
|
12
|
Chen M, Du Y, Qui M, Wang M, Chen K, Huang
Z, Jiang M, Xiong F, Chen J, Zhou J, et al: Ophiopogonin B-induced
autophagy in non-small cell lung cancer cells via inhibition of the
PI3 K/Akt signaling pathway. Oncol Rep. 29:430–436. 2013.
|
|
13
|
Xu QJ, Hou LL, Hu GQ and Xie SQ: Molecular
mechanism of ophiopogonin B induced cellular autophagy of human
cervical cancer HeLa cells. Yao Xue Xue Bao. 48:855–859. 2013.In
Chinese. PubMed/NCBI
|
|
14
|
Kabeer FA, Sreedevi GB, Nair MS,
Rajalekshmi DS, Gopalakrishnan LP, Kunjuraman S and Prathapan R:
Antineoplastic effects of deoxyelephantopin, a sesquiterpene
lactone from Elephantopus scaber, on lung adenocarcinoma (A549)
cells. J Integr Med. 11:269–277. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Mondal J, Bishayee K, Panigrahi AK and
Khuda-Bukhsh AR: Low doses of ethanolic extract of Boldo (Peumus
boldus) can ameliorate toxicity generated by cisplatin in normal
liver cells of mice in vivo and in WRL-68 cells in vitro, but not
in cancer cells in vivo or in vitro. J Integr Med. 12:425–438.
2014. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Wang YH, Qiu C, Wang DW, Hu ZF, Yu BY and
Zhu DN: Identification of multiple constituents in the traditional
Chinese medicine formula Sheng-Mai San and rat plasma after oral
administration by HPLC-DAD-MS/MS. J Pharm Biomed Anal.
54:1110–1127. 2011. View Article : Google Scholar
|
|
17
|
Thompson CB: Apoptosis in the pathogenesis
and treatment of disease. Science. 267:1456–1462. 1995. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Mohamad N, Gutiérrez A, Núñez M, Cocca C,
Martín G, Cricco G, Medina V, Rivera E and Bergoc R: Mitochondrial
apoptotic pathways. Biocell. 29:149–161. 2005.PubMed/NCBI
|
|
19
|
Lucken-Ardjomande S and Martinou JC:
Newcomers in the process of mitochondrial permeabilization. J Cell
Sci. 118:473–483. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Ott M, Gogvadze V, Orrenius S and
Zhivotovsky B: Mitochondria, oxidative stress and cell death.
Apoptosis. 12:913–922. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Götz ME, Künig G, Riederer P and Youdim
MB: Oxidative stress: Free radical production in neural
degeneration. Pharmacol Ther. 63:37–122. 1994. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Cagnol S and Chambard JC: ERK and cell
death: Mechanisms of ERK-induced cell death-apoptosis, autophagy
and senescence. FEBS J. 277:2–21. 2010. View Article : Google Scholar
|
|
23
|
Kralova J, Dvorak M, Koc M and Kral V: P38
MAPK plays an essential role in apoptosis induced by
photoactivation of a novel ethylene glycol porphyrin derivative.
Oncogene. 27:3010–3020. 2008. View Article : Google Scholar
|
|
24
|
Dhanasekaran DN and Reddy EP: JNK
signaling in apoptosis. Oncogene. 27:6245–6251. 2008. View Article : Google Scholar : PubMed/NCBI
|