Introduction
Doxorubicin (DOX) is widely used to treat
malignancies (1), however, it has
been challenged due to its cardiotoxicity (2). Chronic toxicity is closely associated
with typical features of heart failure and electrocardiographic
abnormalities (3,4). Previous studies have indicated that
cardiomyocyte apoptosis and intracellular calcium dysregulation may
be key in DOX-induced heart injury (5,6).
RNA helicases are responsible for ribonucleoprotein
remodeling (7). DEAD-box (DDX)
proteins are considered the largest family (8). p72 is transcribed from probable
ATP-dependent RNA helicase DDX17 mRNA and it is suggested to be key
in cancer progression (9). In
breast tumors, p72 was demonstrated to be widely overexpressed and
functions in estrogen receptor α (ERα) activation, thus, enhancing
its oncogenic activities (10). In
colorectal tumors and adenocarcinomas, p72 has also been indicated
to be abnormally upregulated by increasing β-catenin (11,12).
However, less research has been conducted into the role of p72 on
cardiomyocyte injury.
ER activation is reported to reduce cardiac infarct
size and ventricular arrhythmias, predominantly via activation of
the downstream PI3K/Akt signaling pathway (13). The present study investigated the
role of p72 in DOX-induced cardiomyocyte injury in neonatal rat
cardiomyocytes, which may elucidate possible underlying mechanisms
in reducing DOX-induced cardiomyocyte apoptosis.
Materials and methods
Isolation and culture of rat
cardiomyocytes
Neonatal rat cardiomyocytes were isolated from
1-day-old Sprague Dawley rats (Sibeifu Co., Beijing, China), the
heart tissue was digested with trypsin and type II collagenase. The
cells were seeded in high glucose Dulbecco's modified Eagle's
medium (DMEM; Hyclone; GE Healthcare Life Sciences, Logan, UT, USA)
with 10% FBS (Hyclone; GE Healthcare Life Sciences) at the density
of 5×104 cells/cm2. The present study was
approved by the ethics committee of Henan Provincial People's
Hospital (Zhengzhou, China).
Cell culture
HEK293T cells were purchased from American Type
Culture Collection (Manassas, VA, USA) and were cultured in
DMEM/F12 (Hyclone; GE Healthcare Life Sciences, Logan, UT, USA)
supplemented with 10% fetal bovine serum, 100 U/ml penicillin and
streptomycin (Invitrogen; Thermo Fisher Scientific, Inc., Waltham,
MA, USA) in a 25 cm2 culture flask at 37°C in a
humidified atmosphere of 5% CO2.
Determination of reactive oxygen species
(ROS)
Cells were cultured on six-well chamber slides and
washed with phosphate-buffered saline (PBS) three times for 5 min).
The slides were incubated with ROS Fluorescent Probe-DHE (Vigorous
Biotechnology Beijing Co., Ltd, Beijing, China) in serum-free
DMEM/F-12 medium for 30 min at 37°C in a dark environment. The
slides were fixed in 4% paraformaldehyde for 20 min at room
temperature, washed with PBS for three times and mounted.
Immunofluorescence images were captured using a fluorescence
microscope.
Dimethyl thiazolyl diphenyl tetrazolium
(MTT) assay
Cell viability was determined using a colormetric
MTT assay (Sigma-Aldrich, St. Louis, MO, USA). To investigate the
effects of DOX on cardiomyocyte viability, cells were cultured at
~70% confluency and cultured in serum-free DMEM overnight.
Subsequently, 1, 10, 100 nM, 1, and 10 µM DOX was incubated
with the primary cardiomyocytes for 24 h at 37°C. MTT (0.5 mg/ml)
was added in fresh medium for 4 h and dimethyl sulfoxide was added
into the wells. The absorbance was detected spectrophotometrically
at a wavelength of 550 nm. To determine the time-dependent effects
of DOX, cells were treated with 1 µM DOX for 8, 16, 24 and
48 h prior to investigation of cell viability according to the
above methods. Each experiment was independently performed at least
3 times.
Western blot analysis
Proteins were isolated from cardiomyocytes in
radioimmunoprecipitation assay buffer [1% Triton X-100, 150 mmol/l
NaCl, 5 mmol/l EDTA and 10 mmol/l Tris-HCl (pH 7.0)] obtained from
Beijing Solarbio Science & Technology Co., Ltd. (Beijing,
China) with supplementation of protease inhibitor cocktail
(Sigma-Aldrich). Protein was quantified using a Pierce BCA Protein
assay kit (Thermo Fisher Scientific, Inc.). Cell lysates (10
µg protein) were separated by 10% SDS-PAGE and transferred
to a polyvinyl difluoride membrane. The membrane was blocked with
8% milk for 2 h at room temperature. The membrane was incubated at
4°C overnight with primary antibodies as follows (all from Cell
Signaling Technology, Inc., Danvers, MA, USA unless otherwise
stated): p72 (1:1,000; Abcam, Cambridge, MA, USA; cat. no.
ab24601), rabbit anti-ERα (1:1,000; cat. no. 8644), rabbit
anti-p-Akt (1:1,000; cat. no. 4060), rabbit anti-Akt (1:1,000; cat.
no. 4691), rabbit anti-p-caspase 3 (1:1000; cat. no. 9664), rabbit
anti-caspase 3 (1:1000; cat. no. 9665) and mouse anti-β-actin
(1:4,000; cat. no. 3700). Following incubation overnight and
washing three times for 5 min with PBS with Tween 20, the
horseradish peroxidase-conjugated goat anti-rabbit IgG secondary
antibodies (1:5,000; Origene Technologies, Beijing, China; cat. no.
ZB-2301) were used at room temperature for 2 h. Immunodetection was
achieved using the Chemilucent Plus ECL Detection kit (EMD
Millipore, Billerica, MA, USA) according to the manufacturer's
protocols. Images of the blots were captured using an imager.
β-actin served as the internal control and Image J 5.0 (imagej.nih.gov) was used to quantify the results.
TUNEL staining
The TUNEL assay was performed using the In
Situ Cell Death Detection kit (Roche Diagnostics, Basel,
Switzerland). Following staining, the cells were washed with cold
PBS and examined under a fluorescence microscope.
Overexpression of p72
Phusion High-Fidelity enzyme (Thermo Fisher
Scientific, Inc.) was used for cloning purposes. The entire p72
cDNA was amplified by RT-PCR using specific primers for
p72-BamHI-forward (GCGGATCCCCGCGGCACTGCCCGGTTTG) and
p72-EcoRI-reverse (GCGAATTCTACAAGTCTTTCAAGTCTTA) and then
cloned into the expression vector, pCDH-CMV-MSC-EF1-copGFP (System
Biosciences, Palo Alto, CA, USA) with a Cold Fusion Cloning kit
(System Biosciences). Recombinant adenovirus was generated from
293T cells with calcium phosphate precipitation. Primary
cardiomyocytes were seeded at 1×106 cells/well in the
6-well plates. Then, the adenovirus vectors were transfected into
cardiomyocytes for 48 h.
Statistical analysis
Data were presented as the mean ± standard error of
the mean from three independent experiments. Statistical analysis
was conducted using Student's t-test on GraphPad Prism 6 (GraphPad,
Inc., La Jolla, CA, USA). P<0.05 was considered to indicate a
statistically significant difference.
Results
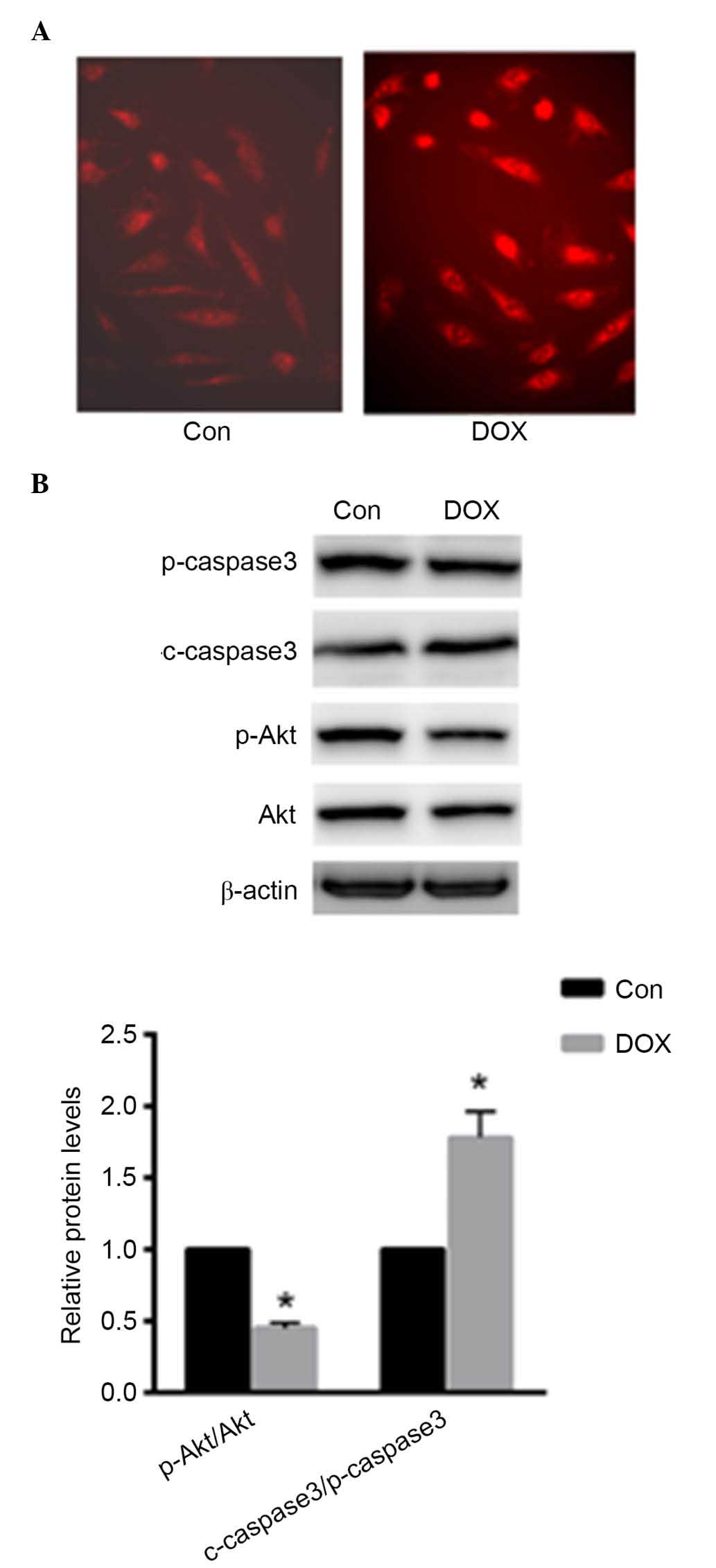
DOX induced ROS production in primary rat
cardiomyocytes
Primary cardiomyocytes were treated with 1 µM
DOX for 24 h. As presented in Fig.
1A, ROS production was enhanced in primary cardiomyocytes as
demonstrated using DHE staining. Furthermore, the present study
investigated the protein expression levels of p-caspase-3,
c-caspase-3, p-Akt and Akt. Caspase-3 activation was enhanced with
DOX treatment, while the phosphorylation level of Akt was reduced
(P<0.05; Fig. 1B). These data
indicated that DOX induced cardiomyocyte injury by increasing ROS
production and apoptosis.
DOX treatment reduced cardiomyocyte
viability and decreased p72 expression
Following 1 µM DOX treatment for 8, 16, 24
and 48 h, cell viability was decreased by 22 and 35% at 24 and 48
h, respectively (each P<0.05; Fig.
2A). Preincubation with 10, 100 nM, 1 and 10 µM DOX for
24 h decreased cell viability by 26 and 54% at 1 and 10 µM
DOX, respectively (P<0.05 and P<0.01, respectively; Fig. 2B). These results suggested that DOX
reduced cardiomyocyte viability in a time- and dose-dependent
manner. Furthermore, protein expression levels of p72 were also
detected under the same conditions. As presented in Fig. 2C and D, p72 expression levels were
decreased following DOX treatment at the concentration of 1
µM for 24 h. Thus, 1 µM DOX was used for 24 h in the
remaining experiments.
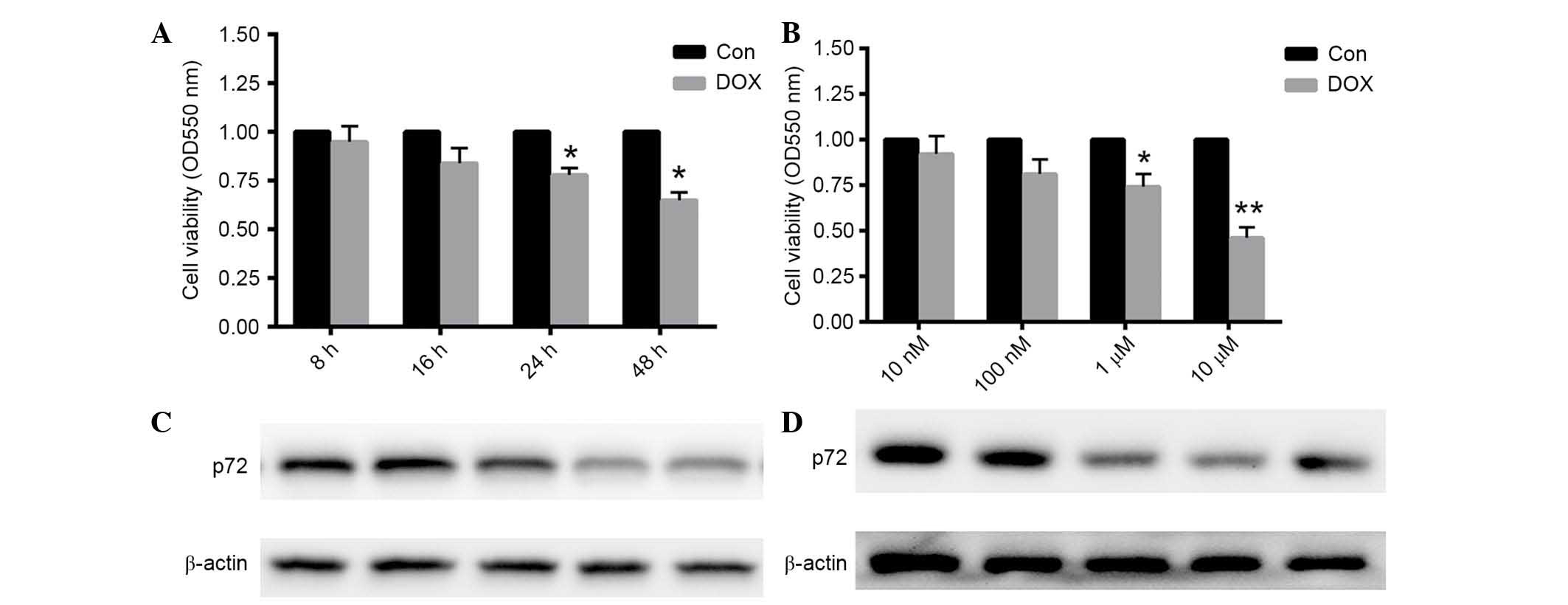 | Figure 2Cardiomyocytes viability was decreased
in a time- and dose-dependent manner, accompanied by a reduction in
p72 expression. (A) Rat cardiomyocytes were treated with 1
µM DOX for 8, 16, 24 or 48 h. (B) Cardiomyocytes were
preincubated with 1, 10, 100 nM, and 1 µM DOX for 24 h. The
MTT assay was conducted to determine cell viability. (C and D)
Protein expression levels of p72 were dertermined under the same
conditions. Data are presented as the mean ± standard error of the
mean, n=6 independent experiments. *P<0.05,
**P<0.01 vs. the control. p72, probable ATP-dependent
RNA helicase DDX17; DOX, doxorubicin; Con, control. |
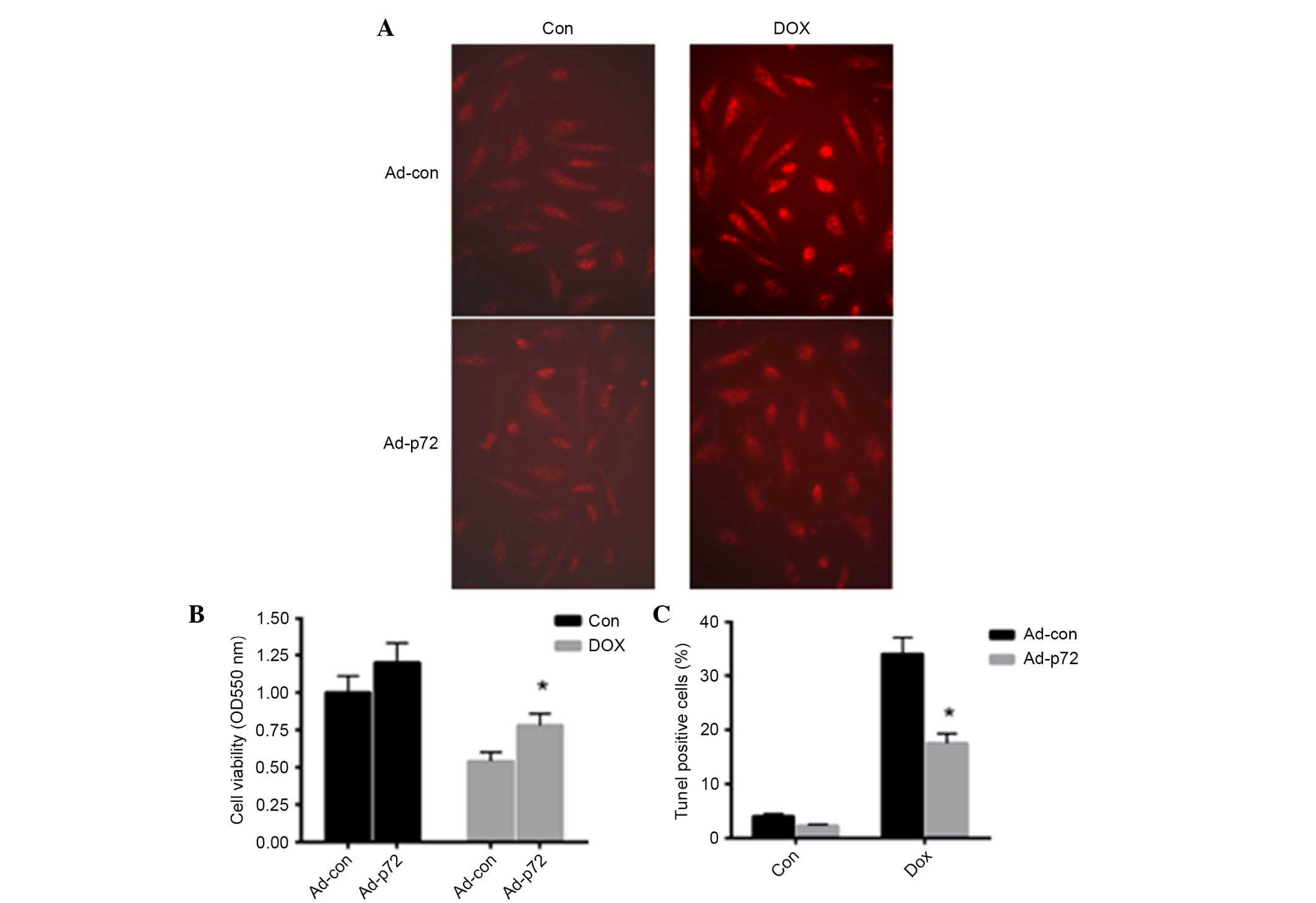
p72 exerts a protective effect on
DOX-induced cardiomyocyte injury
To investigate the effect of p72 on cardiomyocyte
apoptosis, adenovirus vectors expressing p72 were introduced into
primary cardiomyocytes. As presented in Fig. 3A, overexpression of p72 reduced
DOX-induced ROS production in cardiomyocytes. Following induction
of p72 overexpression, cell viability was significantly enhanced in
the DOX-induced group (P<0.05; Fig.
3B). TUNEL staining was also conducted to detect apoptotic
cells. Notably, p72 overexpression significantly reduced
DOX-induced cell apoptosis (Fig.
3C). These data indicated that p72 exerts a protective effect
in DOX-induced cardiomyocyte injury.
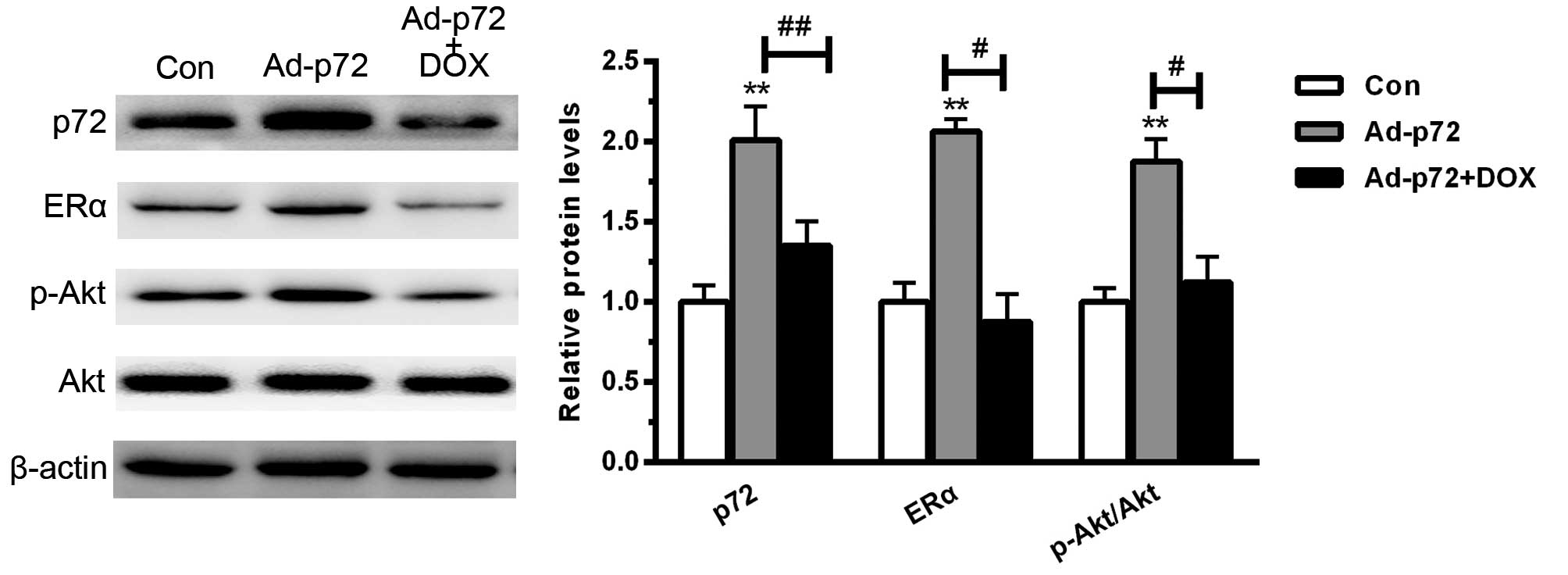
p72 enhances ERα activation and Akt
phosphorylation
To investigate the underlying mechanism by which p72
regulates cardiomyocyte viability, the downstream signaling pathway
was assessed. As presented in Fig.
4, overexpression of p72 significantly enhanced ERα activation
and increased the phosphorylation level of Akt (each P<0.01). By
contrast, DOX significantly reduced ERα activation (P<0.05) and
increased the phosphorylation level of Akt (P<0.05). These data
indicated that p72 overexpression protected against cardiomyocyte
injury predominantly by enhancing ERα activation and Akt
phosphorylation.
Discussion
In numerous types of malignancy, DOX is commonly
applied as an effective antitumor agent. However, it is also found
to result in irreversible chronic cardiomyopathy and heart failure
(14). A previous study has
indicated that oxidative stress may be key in DOX-induced
cardiomyocytes (14). In the
present study, ROS production and cell apoptosis were demonstrated
to be significantly enhanced in cardiomyocytes. Furthermore, DOX
reduced cardiomyocyte viability in a time- and dose-dependent
manner. These data indicated that DOX induced cardiomyocyte
apoptosis predominantly via increased ROS production and activation
of caspase-3.
Enhanced ROS production results in aberrant
downstream signaling pathways in different cell types (15,16).
Thus, the present study investigated the downstream signaling and
observed decreased phosphorylation levels of Akt, which has been
suggested to enhance cell survival (15). This is consistent with other
previous studies, which demonstrate DOX effects PI3K/Akt signaling
(15,16).
Estrogen receptors are suggested to be important in
various pathophysiologies, including cardiac dysfunctions (17,18).
In clinic practice, estrogen treatment markedly improves myocardiac
infarct size and heart failure (19). Previous studies have determined
that ERs activate the downstream PI3K/Akt signaling pathway,
thereby limiting the inflammatory responses in vivo and
in vitro (19,20).
As an RNA helicase, p72 binds to double and single
stranded RNA. By stimulating ATPase activity, it provides enough
energy to unwind RNA duplexes (21). Previous studies have indicated that
p72 functions as a transcription activator in an estrogen-dependent
manner (21,22). Similarly to p68, p72 directly binds
to ERα, thereby stimulating its transcription (23).
The present study demonstrated p72 was reduced
following treatment with DOX, suggesting it exerts a possible
protective effect. Furthermore, the current study indicated that
overexpression of p72 resulted in reduced DOX-induced cardiomyocyte
injury. Notably, p72 enhanced ERα activation and downstream Akt
phosphorylation. This is consistent with the present study.
In conclusion, the present study demonstrated a
protective role of p72 in DOX-induced cardiomyocyte apoptosis,
predominantly via ERα activation and PI3K/Akt phosphorylation.
References
|
1
|
Smith LA, Cornelius VR, Plummer CJ, Levitt
G, Verrill M, Canney P and Jones A: Cardiotoxicity of anthracycline
agents for the treatment of cancer: Systematic review and
meta-analysis of randomised controlled trials. BMC Cancer.
10:3372010. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Lefrak EA, Pitha J, Rosenheim S and
Gottlieb JA: A clinicopathologic analysis of adriamycin
cardiotoxicity. Cancer. 32:302–314. 1973. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Wojtacki J, Lewicka-Nowak E and
Leśniewski-Kmak K: Anthracycline-induced cardiotoxicity: Clinical
course, risk factors, pathogenesis, detection and prevention-review
of the literature. Med Sci Monit. 6:411–420. 2000.
|
|
4
|
Von Hoff DD, Layard MW, Basa P, Davis HL
Jr, Von Hoff AL, Rozencweig M and Muggia FM: Risk factors for
doxorubicin-induced congestive heart failure. Ann Intern Med.
91:710–717. 1979. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Zhang S, Liu X, Bawa-Khalfe T, Lu LS, Lyu
YL, Liu LF and Yeh ET: Identification of the molecular basis of
doxorubicin-induced cardiotoxicity. Nat Med. 18:1639–1642. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Zhou S, Starkov A, Froberg MK, Leino RL
and Wallace KB: Cumulative and irreversible cardiac mitochondrial
dysfunction induced by doxorubicin. Cancer Res. 61:771–777.
2001.PubMed/NCBI
|
|
7
|
Bleichert F and Baserga SJ: The long
unwinding road of RNA helicases. Mol Cell. 27:339–352. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Jankowsky E, Gross CH, Shuman S and Pyle
AM: Active disruption of an RNA-protein interaction by a DExH/D RNA
helicase. Science. 291:121–125. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Uhlmann-Schiffler H, Rössler OG and Stahl
H: The mRNA of DEAD box protein p72 is alternatively translated
into an 82-kDa RNA helicase. J Biol Chem. 277:1066–1075. 2002.
View Article : Google Scholar
|
|
10
|
Yager JD and Davidson NE: Estrogen
carcinogenesis in breast cancer. N Engl J Med. 354:270–282. 2006.
View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Causevic M, Hislop RG, Kernohan NM, Carey
FA, Kay RA, Steele RJ and Fuller-Pace FV: Overexpression and
poly-ubiquitylation of the DEAD-box RNA helicase p68 in colorectal
tumours. Oncogene. 20:7734–7743. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Yang L, Lin C, Zhao S, Wang H and Liu ZR:
Phosphorylation of p68 RNA helicase plays a role in
platelet-derived growth factor-induced cell proliferation by
up-regulating cyclin D1 and c-Myc expression. J Biol Chem.
282:16811–16819. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Wu CH, Liu JY, Wu JP, Hsieh YH, Liu CJ,
Hwang JM, Lee SD, Chen LM, Chang MH, Kuo WW, et al:
17beta-estradiol reduces cardiac hypertrophy mediated through the
up-regulation of PI3K/Akt and the suppression of calcineurin/NF-AT3
signaling pathways in rats. Life Sci. 78:347–356. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Ivanov D, Shabalov N, Petrenko Y,
Shabalova N and Treskina NA: The specific characteristics of DIC
syndrome vary with different clinical settings in the newborn. J
Matern Fetal Neonatal Med. 27:1088–1092. 2014. View Article : Google Scholar
|
|
15
|
Xu J, Qian J, Xie X, Lin L, Zou Y, Fu M,
Huang Z, Zhang G, Su Y and Ge J: High density lipoprotein protects
mesenchymal stem cells from oxidative stress-induced apoptosis via
activation of the PI3K/Akt pathway and suppression of reactive
oxygen species. Int J Mol Sci. 13:17104–17120. 2012. View Article : Google Scholar
|
|
16
|
Muzi-Filho H, Bezerra CG, Souza AM,
Boldrini LC, Takiya CM, Oliveira FL, Nesi RT, Valença SS,
Einicker-Lamas M, Vieyra A, et al: Undernutrition affects cell
survival, oxidative stress, Ca2+ handling and signaling pathways in
vas deferens, crippling reproductive capacity. PLoS One.
8:e696822013. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Zhong L, Zhou XL, Liu YS, Wang YM, Ma F,
Guo BL, Yan ZQ and Zhang QY: Estrogen receptor α mediates the
effects of notoginsenoside R1 on endotoxin-induced inflammatory and
apoptotic responses in H9c2 cardiomyocytes. Mol Med Rep.
12:119–126. 2015.PubMed/NCBI
|
|
18
|
Wang T, McDonald C, Petrenko NB, Leblanc
M, Wang T, Giguere V, Evans RM, Patel VV and Pei L:
Estrogen-related receptor α (ERRα) and ERRγ are essential
coordinators of cardiac metabolism and function. Mol Cell Biol.
35:1281–1298. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Mahmoodzadeh S, Leber J, Zhang X, Jaisser
F, Messaoudi S, Morano I, Furth PA, Dworatzek E and Regitz-Zagrosek
V: Cardiomyocyte-specific estrogen receptor alpha increases
angiogenesis, lymphangiogenesis and reduces fibrosis in the female
mouse heart post-myocardial infarction. J Cell Sci Ther. 5:1532014.
View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Wu KL, Chen CH and Shih CD:
Nontranscriptional activation of PI3K/Akt signaling mediates
hypotensive effect following activation ofestrogen receptor β in
the rostral ventrolateral medulla of rats. J Biomed Sci. 19:762012.
View Article : Google Scholar
|
|
21
|
Huang Y and Liu ZR: The ATPase, RNA
unwinding, and RNA binding activities of recombinant p68 RNA
helicase. J Biol Chem. 277:12810–12815. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Watanabe M, Yanagisawa J, Kitagawa H,
Takeyama K, Ogawa S, Arao Y, Suzawa M, Kobayashi Y, Yano T,
Yoshikawa H, et al: A subfamily of RNA-binding DEAD-box proteins
acts as an estrogen receptor alpha coactivator through the
N-terminal activation domain (AF-1) with an RNA coactivator, SRA.
EMBO J. 20:1341–1352. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Metivier R, Penot G, Hübner MR, Reid G,
Brand H, Kos M and Gannon F: Estrogen receptor-alpha directs
ordered, cyclical and combinatorial recruitment of cofactors on a
natural target promoter. Cell. 115:751–763. 2003. View Article : Google Scholar
|