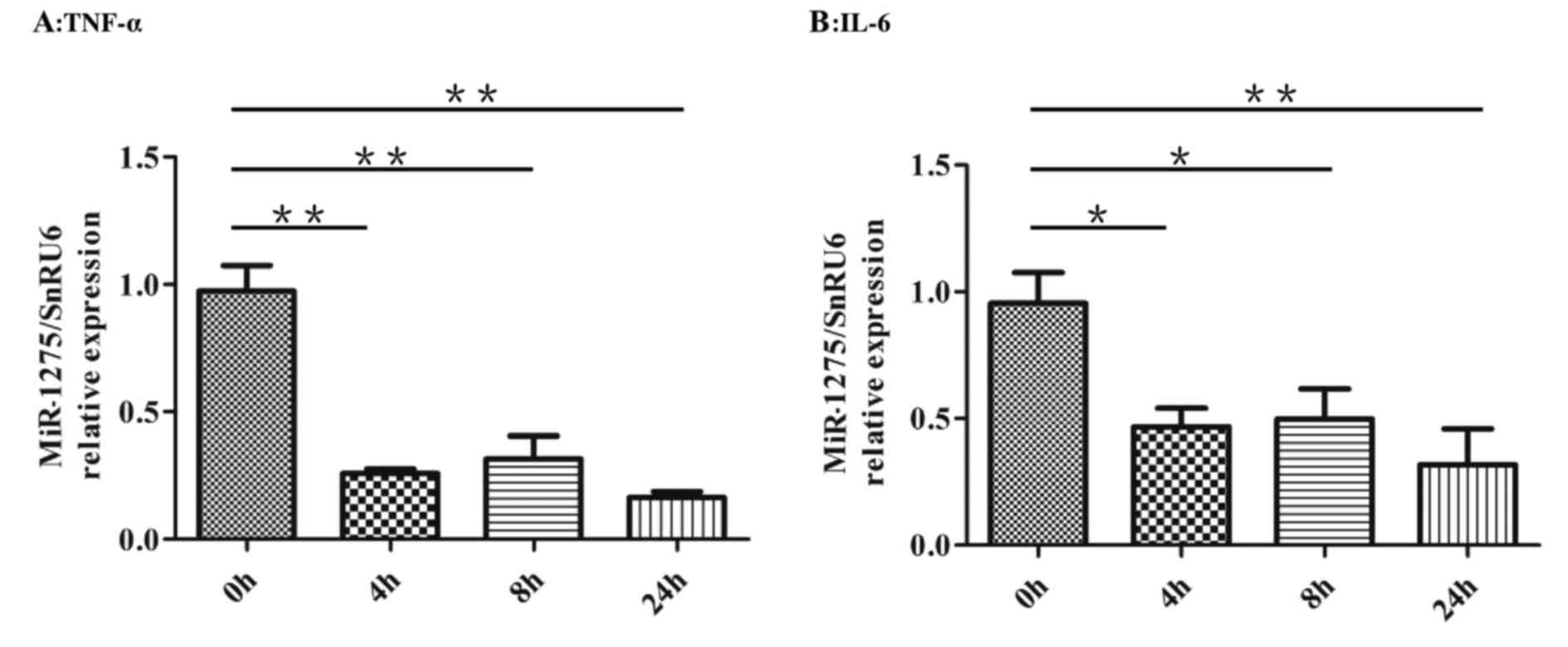
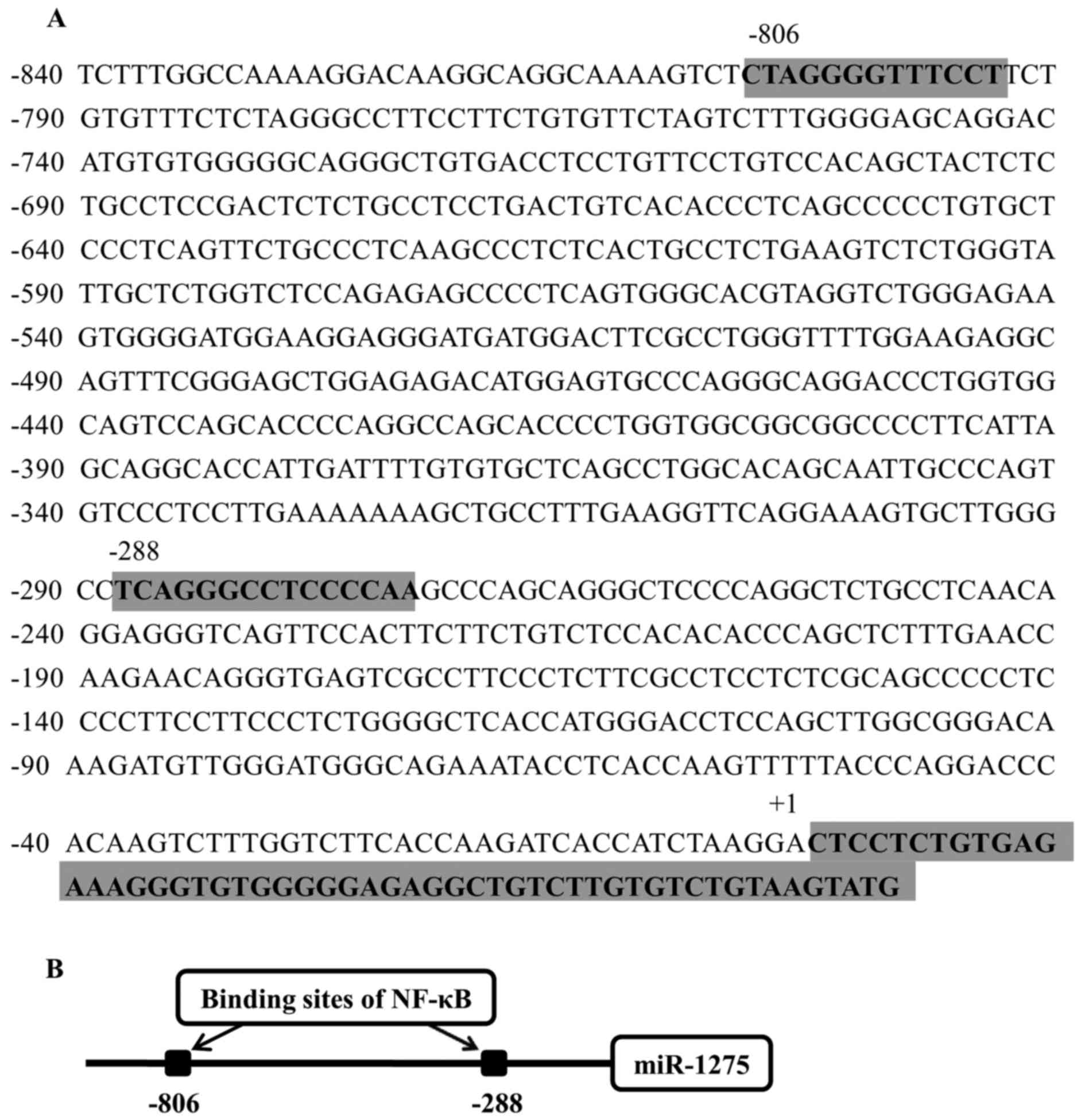
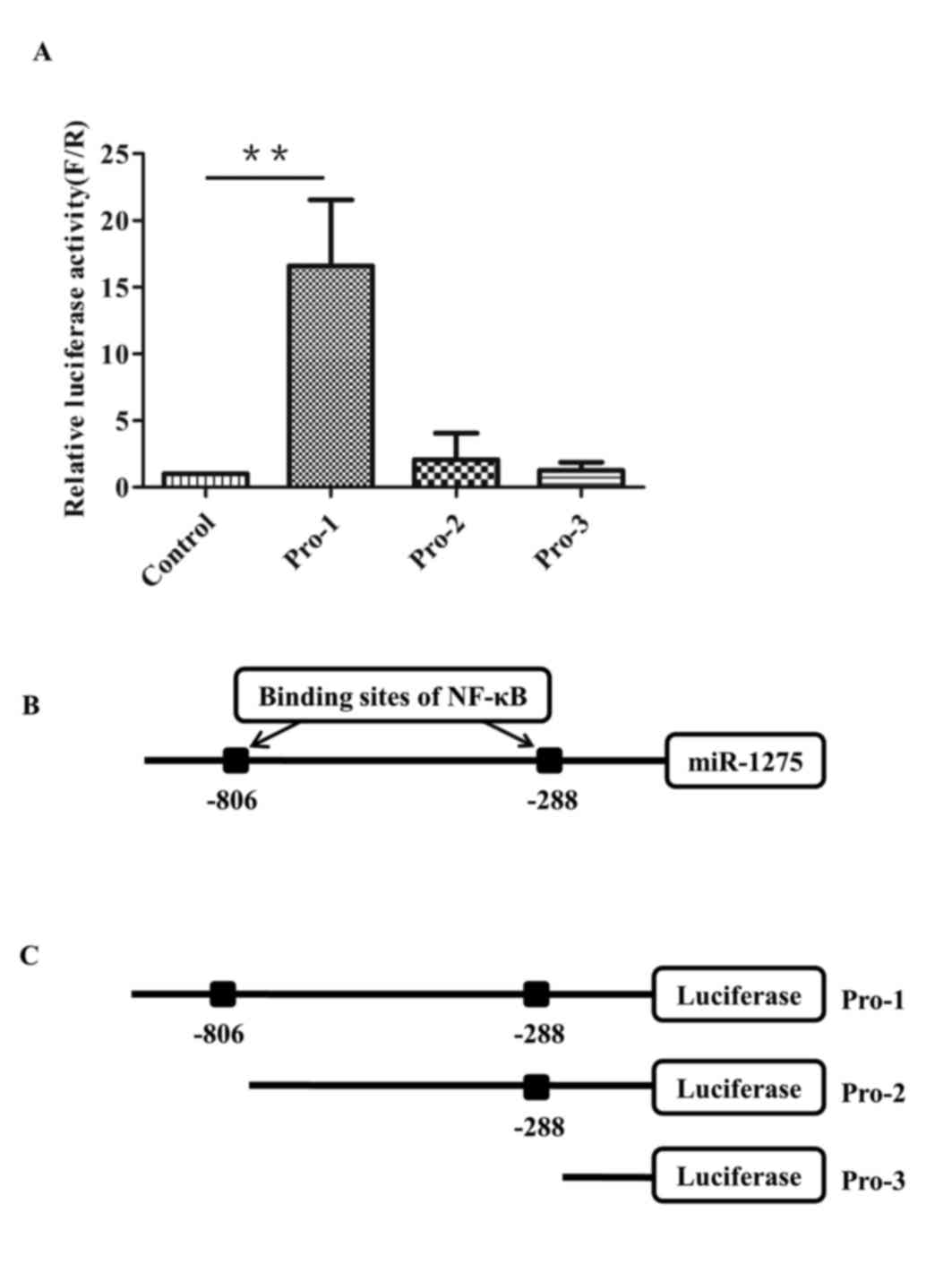
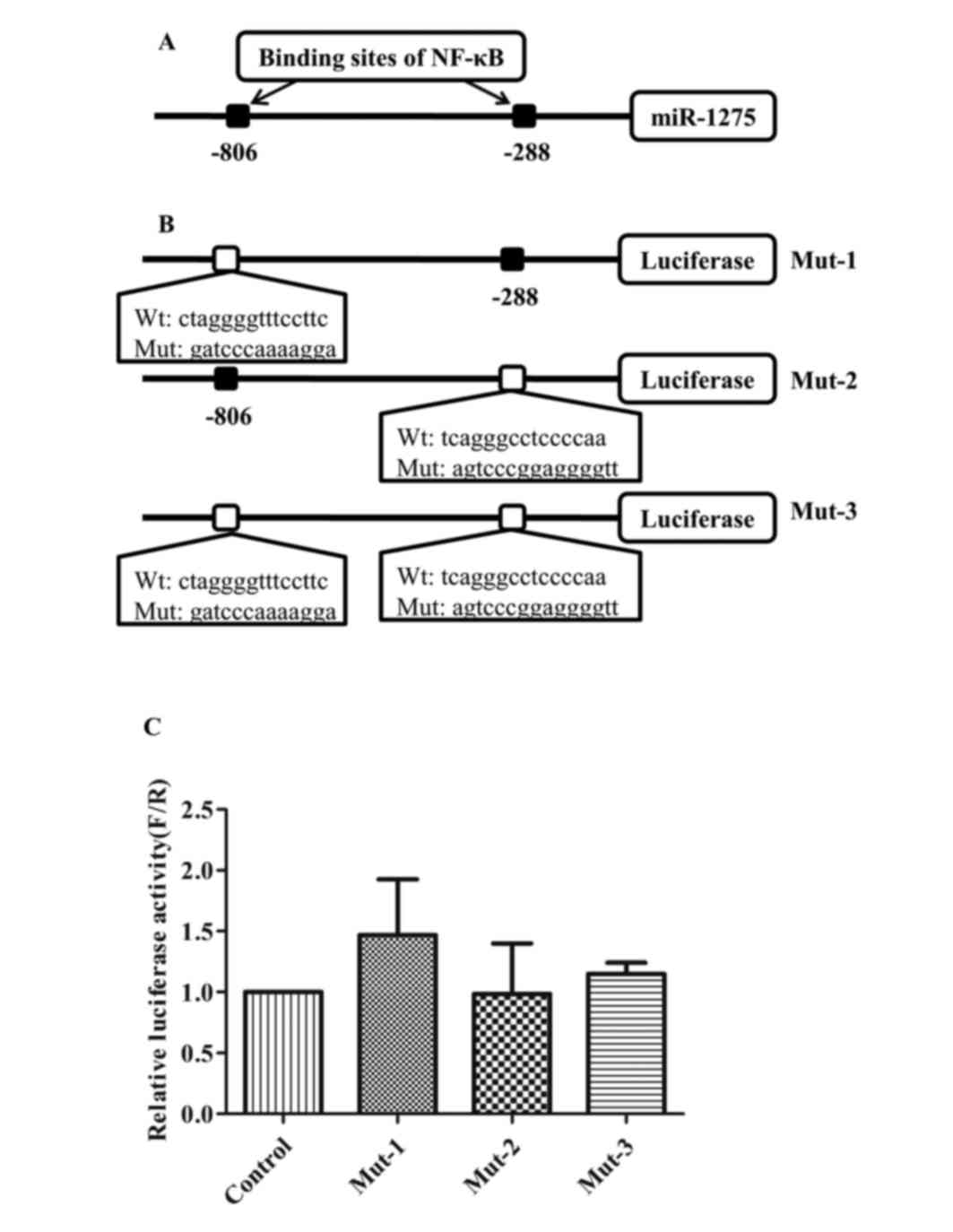
|
1
|
World Health Organization, . Obesity and
Overweight Fact Sheet. June. 2016http://www.who.int/mediacentre/factsheets/fs311/en/3–August.
2016
|
|
2
|
Sandfort V, Lai S, Ahlman MA, Mallek M,
Liu S, Sibley CT, Turkbey EB, Lima JA and Bluemke DA: Obesity is
associated with progression of atherosclerosis during statin
treatment. J Am Heart Assoc. 5(pii): e0036212016. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Peng Y, Yu S, Li H, Xiang H, Peng J and
Jiang S: MicroRNAs: Emerging roles in adipogenesis and obesity.
Cell Signal. 26:1888–1896. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Ambros V: The functions of animal
microRNAs. Nature. 431:350–355. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Abente EJ, Subramanian M, Ramachandran V
and Najafi-Shoushtari SH: MicroRNAs in obesity-associated
disorders. Arch Biochem Biophys. 589:108–119. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Yang L, Shi CM, Chen L, Pang LX, Xu GF, Gu
N, Zhu LJ, Guo XR, Ni YH and Ji CB: The biological effects of
hsa-miR-1908 in human adipocytes. Mol Biol Rep. 42:927–935. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Fawzy IO, Hamza MT, Hosny KA, Esmat G, El
Tayebi HM and Abdelaziz AI: miR-1275: A single microRNA that
targets the three IGF2-mRNA-binding proteins hindering tumor growth
in hepatocellular carcinoma. FEBS Lett. 589:2257–2265. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Pang L, You L, Ji C, Shi C, Chen L, Yang
L, Huang F, Zhou Y, Zhang J, Chen X and Guo X: MiR-1275 inhibits
adipogenesis via ELK1 and its expression decreases in obese
subjects. J Mol Endocrinol. 57:33–43. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Das K, Saikolappan S and Dhandayuthapani
S: Differential expression of miRNAs by macrophages infected with
virulent and avirulent Mycobacterium tuberculosis. Tuberculosis
(Edinb). 93 Suppl:S47–S50. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Katsushima K, Shinjo K, Natsume A, Ohka F,
Fujii M, Osada H, Sekido Y and Kondo Y: Contribution of
microRNA-1275 to Claudin11 protein suppression via a
polycomb-mediated silencing mechanism in human glioma stem-like
cells. J Biol Chem. 287:27396–27406. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Akamatsu S, Hayes CN, Tsuge M, Miki D,
Akiyama R, Abe H, Ochi H, Hiraga N, Imamura M, Takahashi S, et al:
Differences in serum microRNA profiles in hepatitis B and C virus
infection. J Infect. 70:273–287. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Dandona P, Aljada A and Bandyopadhyay A:
Inflammation: The link between insulin resistance, obesity and
diabetes. Trends Immunol. 25:4–7. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Hotamisligil GS, Arner P, Caro JF,
Atkinson RL and Spiegelman BM: Increased adipose tissue expression
of tumor necrosis factor-alpha in human obesity and insulin
resistance. J Clin Invest. 95:2409–2415. 1995. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Karkeni E, Astier J, Tourniaire F, El Abed
M, Romier B, Gouranton E, Wan L, Borel P, Salles J, Walrand S, et
al: Obesity-associated inflammation induces microRNA-155 expression
in adipocytes and adipose tissue: Outcome on adipocyte function. J
Clin Endocrinol Metab. 101:1615–1626. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Chen L, Dai YM, Ji CB, Yang L, Shi CM, Xu
GF, Pang LX, Huang FY, Zhang CM and Guo XR: MiR-146b is a regulator
of human visceral preadipocyte proliferation and differentiation
and its expression is altered in human obesity. Mol Cell
Endocrinol. 393:65–74. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Xu LL, Shi CM, Xu GF, Chen L, Zhu LL, Zhu
L, Guo XR, Xu MY and Ji CB: TNF-alpha, IL-6 and leptin increase the
expression of miR-378, an adipogenesis-related microRNA in human
adipocytes. Cell Biochem Biophys. 70:771–776. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Shi C, Zhu L, Chen X, Gu N, Chen L, Zhu L,
Yang L, Pang L, Guo X, Ji C and Zhang C: IL-6 and TNF-α induced
obesity-related inflammatory response through transcriptional
regulation of miR-146b. J Interferon Cytokine Res. 34:342–348.
2014. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) method. Methods. 25:402–408. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Mathelier A, Fornes O, Arenillas DJ, Chen
CY, Denay G, Lee J, Shi W, Shyr C, Tan G, Worsley-Hunt R, et al:
JASPAR 2016: A major expansion and update of the open-access
database of transcription factor binding profiles. Nucleic Acids
Res. 44:D110–D115. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Kaur S, Anjali G, Bhardwaj P, Taneja J and
Singh R: Data in support of FSH induction of IRS-2 in human
granulosa cells: Mapping the transcription factor binding sites in
human IRS-2 promoter. Data Brief. 6:162–167. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Farré D, Roset R, Huerta M, Adsuara JE,
Roselló L, Albà MM and Messeguer X: Identification of patterns in
biological sequences at the ALGGEN server: PROMO and MALGEN.
Nucleic Acids Res. 31:3651–3653. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Kuang Q, Li J, You L, Shi C, Ji C, Guo X,
Xu M and Ni Y: Identification and characterization of NF-kappaB
binding sites in human miR-1908 promoter. Biomed Pharmacother.
74:158–163. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Parafioriti A, Bason C, Armiraglio E,
Calciano L, Daolio PA, Berardocco M, Di Bernardo A, Colosimo A,
Luksch R and Berardi AC: Ewing's sarcoma: An analysis of miRNA
expression profiles and target genes in paraffin-embedded primary
tumor tissue. Int J Mol Sci. 17(pii): E6562016. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Hotamisligil GS: Inflammation and
metabolic disorders. Nature. 444:860–867. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Zhu L, Chen L, Shi CM, Xu GF, Xu LL, Zhu
LL, Guo XR, Ni Y, Cui Y and Ji C: MiR-335, an adipogenesis-related
microRNA, is involved in adipose tissue inflammation. Cell Biochem
Biophys. 68:283–290. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Ozes ON, Mayo LD, Gustin JA, Pfeffer SR,
Pfeffer LM and Donner DB: NF-kappa B activation by tumour necrosis
factor requires the Akt serine-threonine kinase. Nature. 401:82–85.
1999. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Wang D and Baldwin AS Jr: Activation of
nuclear factor-kappaB-dependent transcription by tumor necrosis
factor-alpha is mediated through phosphorylation of RelA/p65 on
serine 529. J Biol Chem. 273:29411–29416. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Chen LF and Greene WC: Shaping the nuclear
action of NF-kappa B. Nat Rev Mol Cell Bio. 5:392–401. 2004.
View Article : Google Scholar
|
|
29
|
Taganov KD, Boldin MP, Chang KJ and
Baltimore D: NF-kappaB-dependent induction of microRNA miR-146, an
inhibitor targeted to signaling proteins of innate immune
responses. Proc Natl Acad Sci USA. 103:12481–12486. 2006;
View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Iliopoulos D, Hirsch HA and Struhl K: An
epigenetic switch involving NF-kappaB, Lin28, Let-7 MicroRNA and
IL6 links inflammation to cell transformation. Cell. 139:693–706.
2009. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Xu Z, Xiao SB, Xu P, Xie Q, Cao L, Wang D,
Luo R, Zhong Y, Chen HC and Fang LR: miR-365, a novel negative
regulator of interleukin-6 gene expression, is cooperatively
regulated by Sp1 and NF-kappaB. J Biol Chem. 286:21401–21412. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Borghi A, Verstrepen L and Beyaert R:
TRAF2 multitasking in TNF receptor-induced signaling to NF-κB, MAP
kinases and cell death. Biochem Pharmacol. 116:1–10. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Vranková S, Barta A, Klimentová J,
Dovinová I, Líšková S, Dobešová Z, Pecháňová O, Kuneš J and Zicha
J: The regulatory role of nuclear factor Kappa B in the heart of
hereditary hypertriglyceridemic rat. Oxid Med Cell Longev.
2016:98140382016. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Sasaki CT and Vageli DP: miR-21, miR-155,
miR-192, and miR-375 deregulations related to NF-kappaB activation
in gastroduodenal fluid-induced early preneoplastic lesions of
laryngeal mucosa in vivo. Neoplasia. 18:329–338. 2016. View Article : Google Scholar : PubMed/NCBI
|