Introduction
Acute myocardial infarction (AMI) is one of the most
life threatening diseases in the modern world in terms of its high
mortality rate. The most successful therapeutic strategy, in which
coronary circulation is restored, was established to provide
essential preservation of viable myocardium following AMI (1). However, reperfusion therapy may
induce oxidative stress and inflammatory responses, which causes
cell damage and can lead to death. This pathophysiological process
has been defined as ischemia/reperfusion (I/R) injury (2). To alleviate this injury, numerous
investigations have focused on interventions, which can attenuate
I/R injury (3); however, the
conversion of these strategies into clinical practice has not been
satisfactory. To improve the clinical efficacy of AMI, it is
important to identify novel pharmaceutical preparations for the
prevention of myocardial I/R injury.
As a form of traditional medicine, Schisandra
chinensis has been used extensively in East Asia for thousands
of years and several symptoms can be alleviated with its use
(4,5). Schisandrin B (Sch B) is the natural
medicinal monomer extracted from Schisandra chinensis, and
has been confirmed in several studies to have multiple functions,
including an antioxidant effect on the liver (6,7). In
addition, studies have found that Sch B protects cerebral tissue
from ischemia and I/R injury (8,9). It
has also been reported that Sch B can attenuate myocardial I/R
injury in vitro (10);
however, the precise mechanism underlying the effects of Sch B
in vivo remain to be elucidated.
The phosphoinositide 3-kinases (PI3K)/Akt (also
known as PKB) signaling pathway is considered to be involved in the
regulation of cellular activation and apoptosis (11). Investigations have indicated that
the PI3K/Akt signaling pathway can elevate cellular viability by
limiting the apoptotic events induced by noxious stimuli (12). When the PI3K/Akt signaling pathway
is activated, the myocardial tissue injury induced by I/R is
relieved (13).
In the present study, an I/R rat model was
established to investigate whether Sch B protected myocardial
tissue from I/R injury by suppressing apoptosis, and to determine
the possible mechanisms underlying the cardioprotective effects of
Sch B in vivo.
Materials and methods
Animal models and experimental
design
Male Sprague-Dawley (SD) rats (180–200 g, 7 weeks
old) were purchased from the Experimental Animal Center of Zhejiang
Province (Zhejiang, China). The involvement of animals in the
experiments was reviewed and approved by the Animal Policy and
Welfare Committee of Wenzhou Medical University (Wenzhou, China).
All animals were housed in groups of 4 rats per cage and maintained
in controlled conditions (12 h light-dark cycle; 20–24°C; 45–55%
humidity). Food and water were available ad libitum.
Chemicals and reagents
Sch B was obtained from Xi'an Plant Bio-Engineering
Co., Ltd. (Xi'an, China). LY294002, a PI3K inhibitor, was obtained
from Sigma-Aldrich; Merck Millipore (Darmstadt, Germany).
Anti-phosphorylated (p)-Akt (Ser473; cat. no. 4060), anti-Akt (cat.
no. 4685), anti-B-cell lymphoma 2 (Bcl-2; cat. no. 2876),
anti-cleaved caspase-3 (cat. no. 9661), anti-GAPDH (cat. no. 5174)
antibodies, and anti-rabbit horseradish-peroxidase (HRP)-conjugated
immunoglobulin G (cat. no. 7074) secondary antibodies were
purchased from Cell Signaling Technology, Inc. (Danvers, MA, USA).
Anti-Bcl-2-like protein 4 (Bax; cat. no. sc-6236) antibody was
purchased from Santa Cruz Biotechnology, Inc. (Dallas, TX,
USA).
Experimental groups
According to previous studies (9,10),
preliminary experiments to assess three doses of Sch B (10, 30 and
60 mg/kg) showed that 60 mg/kg Sch B was the optimal dose based on
the myocardial infarction size (81.3±7.1, 74.3±6.7 and 62.8±11.2%,
respectively). In the subsequent experiments, the rats were
randomly divided into five groups: i) Sham group; ii) I/R group;
iii) Sch B+I/R group; iv) Sch B+LY+I/R group; and v) LY+I/R group.
The Sch B-treated rats were administered with Sch B (60 mg/kg
intragastrically, dissolved/suspended in olive oil, once a day) for
15 days, and the remaining rats received olive oil only. The
LY-treated rats were administered with LY294002 [0.3 mg/kg
dissolved in dimethyl sulfoxide (DMSO; 0.1%)] 30 min prior to
reperfusion.
Establishment of myocardial I/R
model
Following sacrifice of the rats with intraperitoneal
pentobarbital (60 mg/kg), the rats were mechanically ventilated
using a rodent respirator (DW-3000B; Huaibei Zhenghua Technology
Co., Ltd., Anhui, China). A limb lead II electrocardiogram (ECG)
was continuously monitored and recorded during the entire
procedure. Following cardiac exposure, the left anterior descending
(LAD) coronary artery was ligated with a 6–0 polypropylene suture
below the inferior margin of the left auricle. At the top of the
vessel, a vinyl tube was placed for reversible coronary occlusion.
The success of ischemia was judged by the ST-segment elevation on
the ECG. The myocardium underwent ischemia for 45 min, following
which the slipknot was released for 24 h to mimic reperfusion.
Assessment of myocardial infarction
size
Infarction size was evaluated using Evans Blue
(EB)/triphenyl tetrazolium chloride (TTC) staining (14). In brief, at the end of reperfusion,
2 ml EB (2%; Sigma-Aldrich; Merck Millipore) was injected
intravenously following ligation of the LAD. The heart was removed
immediately and stored at −20°C for 20 min. The tissues were
sectioned into five transverse slices perpendicular to the long
axis (2 mm thick). The slices were incubated in a 1% TTC solution
at 37°C for 20 min. The EB-stained area represented the area with
normal blood supply. The infarct area (IA) was paler and the area
stained red represented the viable tissue at risk. The IA and area
at risk were analyzed using Image J software (version 1.44;
National Institutes of Health, Bethesda, MD, USA).
Measurements of cardiac troponin T
(cTn-T) and creatinine kinase-MB (CK-MB)
Blood samples were collected from the rats at the
end of reperfusion and were centrifuged at 3,000 × g for 10 min at
4°C. The activities of cTn-T and CK-MB in the plasma were estimated
using commercially available assay kits (Shanghai XinFan
Biotechnology Co., Ltd., Shanghai, China).
Morphological changes in the
myocardium
The hearts were collected at the end of reperfusion.
Following excision of the superfluous tissue, the left ventricular
tissue was fixed in 4% paraformaldehyde at room temperature for 24
h. Subsequently, the fixed tissues were embedded in paraffin and
sectioned at 4 µm thicknesses. The myocardial sections were stained
with hematoxylin and eosin at room temperature for 2 min and
visualized under a light microscope (magnification, ×200; Nikon
Corporation, Tokyo, Japan).
Detection of apoptosis
Following dewaxing and rehydration, the myocardial
tissue sections (4 µm thick) were prepared and cell apoptosis was
detected using the terminal deoxyribonucleotide transferase
mediated dUTP nick end-labeling (TUNEL) assay (Ai Mingkang
Bio-Reagent Co., Ltd., Chongqing, China). The stained sections were
viewed using fluorescence microscopy (Nikon Corporation) and the
TUNEL-positive cells were evaluated at ×400 magnification.
Immunohistochemistry staining
Following dewaxing and rehydraton, the myocardial
tissue sections (4 µm thick) were blocked with 5% bovine serum
albumin (BSA; Sigma-Aldrich; Merck Millipore) and incubated with
anti-Bax antibody (1:100) or anti-Bcl-2 antibody (1:100) overnight
at 4°C. Subsequently the sections were incubated with the
HRP-conjugated secondary antibody (1:200) at room temperature for
30 min and visualized under a light microscope (magnification,
×200; Nikon Corporation).
Western blot analysis
Proteins were extracted from the myocardial tissues
using lysis buffer containing protease (Beyotime Institute of
Biotechnology, Haimen, China) and phosphatase inhibitors (Cell
Signaling Technology, Inc., Danvers, MA, USA) under homogenization,
and the concentrations of protein were measured using a
Bicinchoninic Acid Protein Assay kit (Beyotime Institute of
Biotechnology). Following separation by 12% SDS-PAGE, a total of 20
µg proteins were transferred onto a polyvinylidene fluoride
membrane, blocked with 5% BSA at room temperature for 1 h, and
incubated with specific primary antibodies (1:1,000) overnight at
4°C. Subsequently, immunoreactive bands were incubated with a
HRP-conjugated secondary antibody (1:1,000) at room temperature for
1 h and detected using enhanced chemiluminescence (EMD Millipore,
Billerica, MA, USA).
Statistical analysis
Data are expressed as the mean ± standard deviation.
GraphPad Prism software (version 5.0; GraphPad Software, Inc., La
Jolla, CA, USA) was used for statistical analysis. One-way analysis
of variance and Student-Newman-Keuls test were used. P<0.05 was
considered to indicate a statistically significant difference.
Results
Myocardial infarction size
Representative images of the TTC and EB staining for
the rats in each group are shown in Fig. 1. Compared with the control rats,
Sch B administration led to a significant reduction in myocardial
infarction size (62.8±11.2 vs. 84.5±4.1%; P<0.05). However, the
protective effect of the medication was eliminated by LY294002
treatment (I/R+Sch B+LY group, 83.8±2.9%), compared with the I/R+
Sch B group (62.8±11.2%; P<0.05; Fig. 1A).
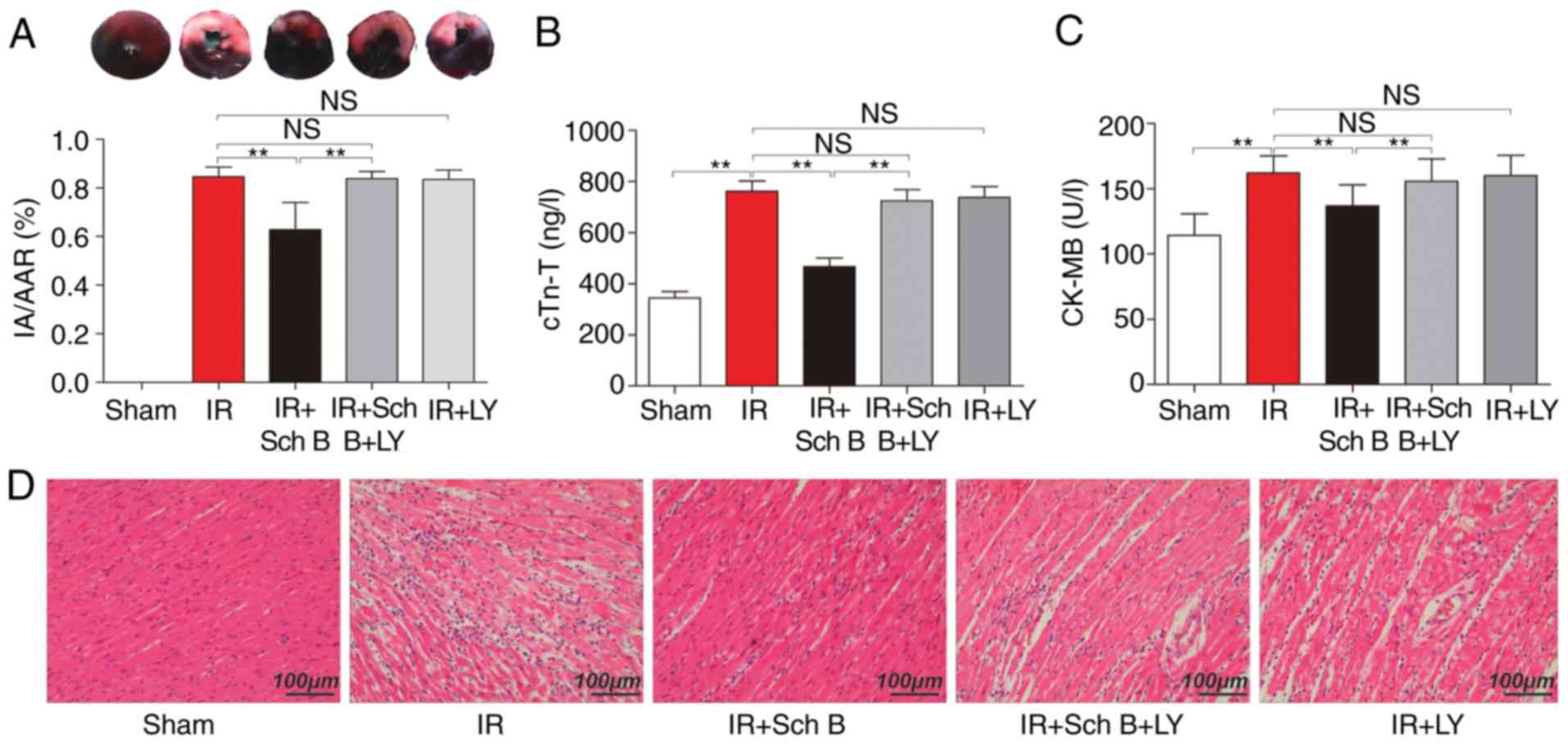 | Figure 1.Effects of Sch B and LY on the area of
myocardial infarction (n=6 for each group), serum-specific cardiac
injury biomarkers (n=8 for each group) and myocardial tissue. (A)
Representative images of tetrazolium chloride and Evans Blue
staining of sections of the heart. Blue, not at risk; paler, IA;
paler and red, AAR. The bar graph represents the percentage of IA
vs. AAR. AAR, as a percentage of the area of the whole left
ventricle did not differ among the groups. (B) Serum levels of
cTn-T. (C) Serum levels of CK-MB. (D) Hematoxylin and eosin
staining of myocardial tissue. Data are presented as the mean ±
standard deviation. **P<0.05. Sch B, schisandrin B; LY,
LY294002; IR, ischemia/reperfusion; IA, infarct area; AAR, area at
risk; cTn-T cardiac troponin T; CK-MB, creatinine kinase-MB; NS,
not significant. |
Plasma cTn-T and CK-MB
Compared with the sham-operation rats, the levels of
myocardial infarction-associated markers in plasma samples were
distinctly increased in the control rats. Consistently, Sch B
treatment reduced the levels of cTn-T, compared with that in the
control rats (468.3±92.0 vs. 761.8±114.0 ng/l, respectively;
P<0.05; Fig. 1A) and reduced
the level of CK-MB, compared with that in the control rats
(136.8±16.3 vs. 162.1±12.9 U/l, respectively; P<0.05 Fig. 1B). However, the inhibitory effect
of Sch B on the level of cTn-T was eliminated by LY294002
administration, compared with that in the Sch B group (468.3±92.0
vs. 725.6±122.2 ng/l, respectively; P<0.05), with the same
observed for CK-MB (136.8±16.3 vs. 155.7±17.2 U/l, respectively;
P<0.05; Fig. 1).
Morphological changes in the
myocardium
Compared with the hearts of the sham group,
disorganization of cell structures was observed in the myocardium
of untreated rats. Cardiomyocyte necrosis and inflammatory cell
infiltration were decreased in the Sch B treatment group, compared
with the untreated rats. However, treatment with LY294002
eliminated the protective effects of Sch B in myocardial injury
(Fig. 1D).
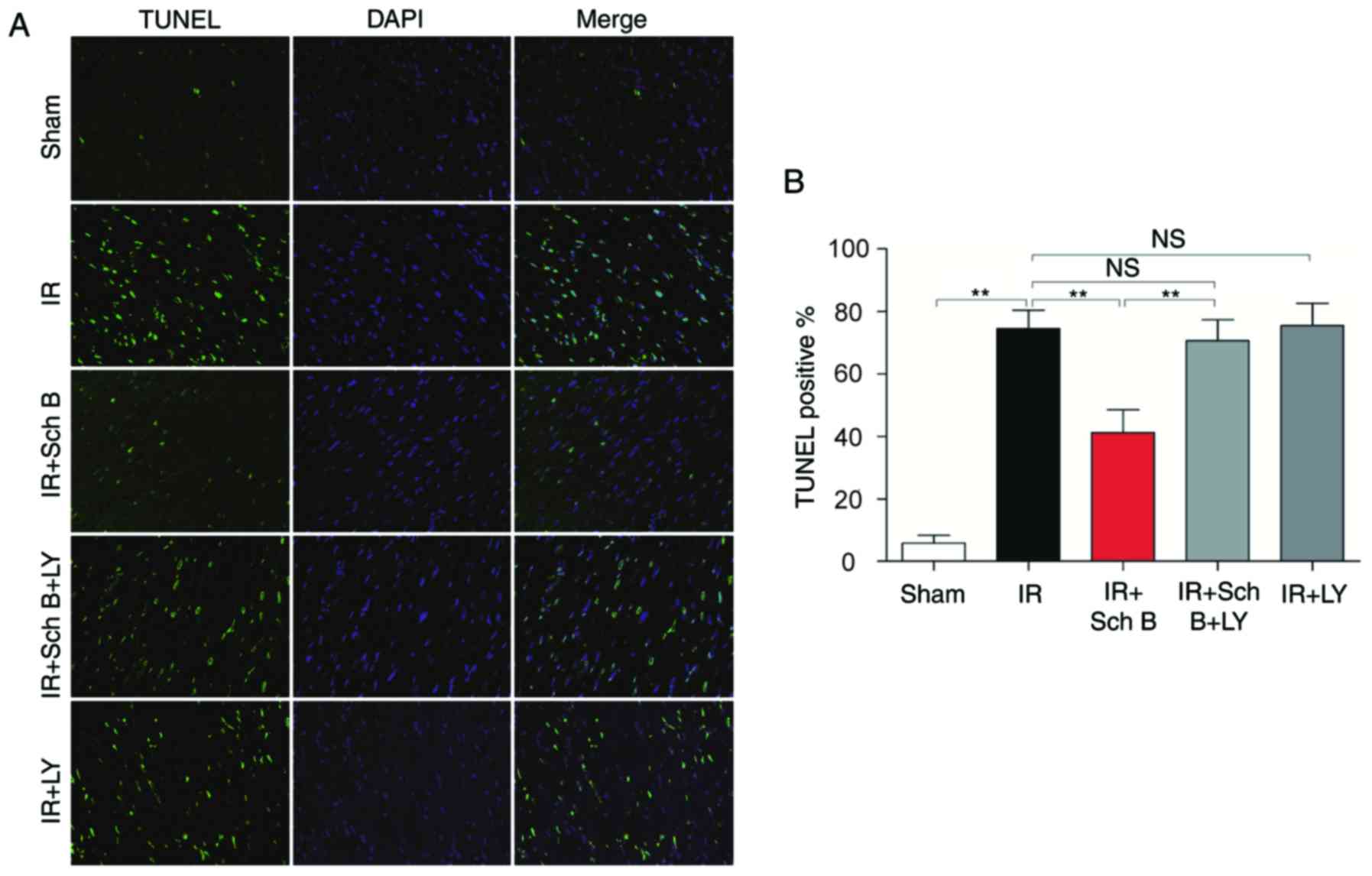
Myocardial apoptosis
The results of the TUNEL assay revealed that the
number of TUNEL-positive cells was higher in the untreated rats
(74.50±5.97%), compared with that in the sham group (5.75±2.50%).
Treatment with Sch B reduced the number of TUNEL-positive nuclei
stained (41.25±7.27%; P<0.05). However, this anti-apoptotic
effect of Sch B was attenuated by LY294002 administration
(41.25±7.27 vs. 70.50±6.76%, respectively; P<0.05), as shown in
Fig. 2A and B.
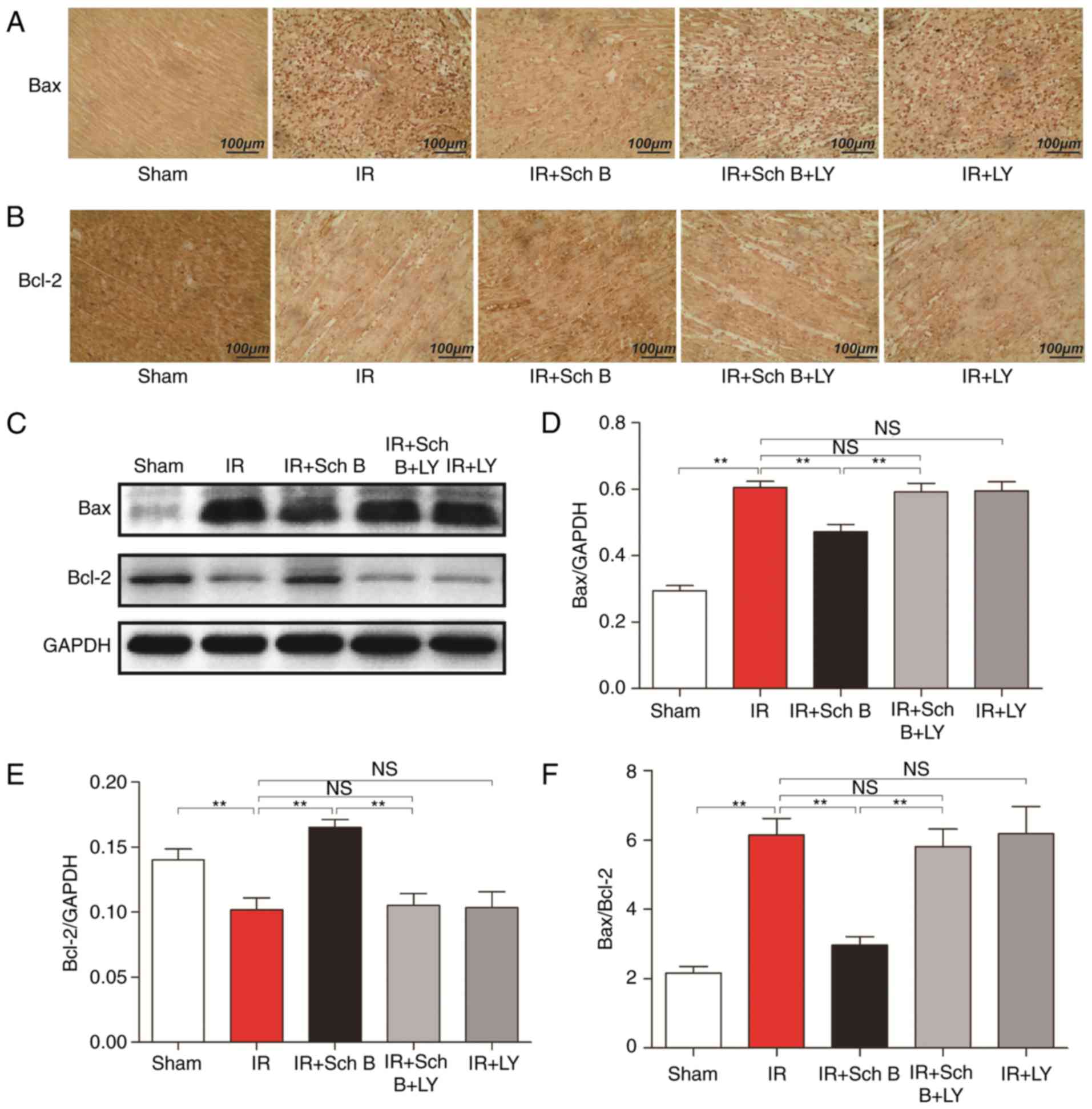
Immunohistochemical staining
Compared with the untreated rats, the concentrations
of Bax were significantly decreased and the concentrations of Bcl-2
were increased in the Sch B treatment groups, however, these
effects of Sch B were attenuated by LY294002 treatment (Fig. 3A-F).
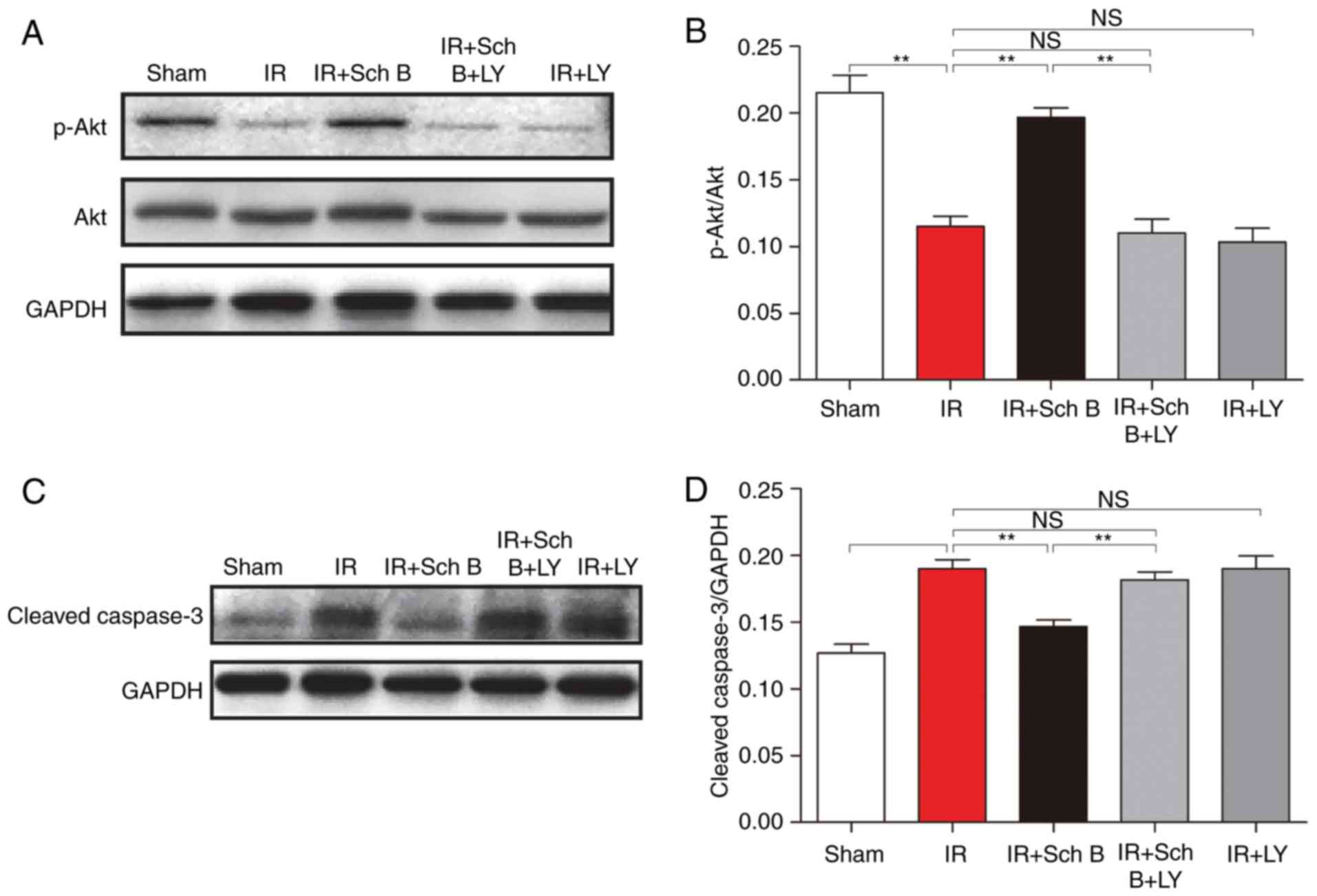
Western blot analysis
Treatment with Sch B significantly increased the
concentrations of p-Akt, compared with that in the untreated rats
(0.20±0.02 vs. 0.12±0.02; P<0.05; Fig. 4A and B). However, the
administration of LY294002, an inhibitor of PI3K, attenuated the
Sch B-induced upregulation of myocardial p-Akt in the I/R + Sch B +
LY group (0.11±0.03). The concentrations of cleaved caspase-3 in
the myocardium of the Sch B+I/R group were also lower, compared
with those in the I/R and Sch B+LY+I/R groups (Fig. 4C and D).
Discussion
The results of the present study revealed that
treatment with the natural medicinal monomer, Sch B, reduced the
infract size induced by I/R. The increased levels of cTn-T and
CK-MB were also attenuated by treatment with Sch B, and treatment
with Sch B reduced the number of TUNEL-positive nuclei stained.
These results indicated that Sch B exerted protective effects in
the myocardium via the decrease in TUNEL-positive cells, which
indicated that the cardioprotective effects of Sch B were
associated with cardiomyocyte apoptosis.
Cardiomyocyte apoptosis is important in the progress
of myocardial infarction and cardiac dysfunction following ischemia
(15). As an important regulator,
the Bcl-2 protein family is considered to be involved in the
myocardial cell apoptotic pathway (16). In addition, Bcl-2 has been shown to
have several effects, including the prevention of mitochondria from
releasing cytochrome c and inhibiting caspase activity,
whereas Bax is known to increase cell apoptosis (16,17).
Consequently, the Bax/Bcl-2 ratio affects apoptotic signaling. In
the present study, it was confirmed that Sch B decreased the
Bax/Bcl-2 ratio, which suggested that the anti-apoptotic effect of
Sch B was associated with the Bcl-2 protein family.
Additionally, the caspase family is considered to be
involved in the cell apoptotic process, with caspase-3 being an
essential protease in the apoptotic cascade reaction (18). When caspase-3 is activated, it
becomes cleaved caspase-3. In the present study, the results showed
that the concentrations of cleaved caspase-3 in the Sch
B-pretreated rats were lower, compared with those in the untreated
rats, which indicated that the anti-apoptotic effect of Sch B was
associated with the caspase family.
The activation of the PI3K/Akt-dependent pathway is
also considered to be involved in myocardial tissue protection and
the inhibition of I/R-induced cardiomyocyte apoptosis (19–21)
via involvement of the caspase family and the incremental
expression of Bcl-2 (22). In the
present study, the results showed that the concentrations of p-Akt
in the Sch B-pretreated rats were higher, compared with those in
the untreated rats. Of note, the use of LY294002 in the present
study revealed that the inhibitory action of PI3K eliminated the
protective effects of Sch B. Taken together, these results
suggested that the potential benefits of Sch B in I/R injury were
likely mediated by the activation of PI3K/Akt pathways via
involvement of the caspase family and the Bcl-2 protein family.
It has been reported that Sch B administration in an
isolated perfused heart preparation protected the myocardium from
I/R injury by decreasing mitochondrial sensitivity to the
permeability transition induced by calcium ions (10). Additionally, it has been shown that
the activation of Akt is vital in maintaining mitochondrial
morphology and function, and modulating the sensitivity of
mitochondrial permeability transition pore opening (23). Therefore, it is likely that Sch B
has a protective effect on myocardial tissues via the PI3K/Akt
pathway.
The limitations of the present study require
consideration when interpreting the results. First, the
investigation is likely to be improved by the addition of a control
group treated with DMSO only. Secondly, as the use of TUNEL
staining for the detection of apoptosis is controversial, further
investigations are required to validate the data. Thirdly, the
effects of Sch B on cardiac function require evaluation in further
investigations, and other signaling pathways involved in the
myocardial protective effects of Sch B require identification.
In conclusion, the present study revealed that Sch B
exerted cardioprotective effects by reducing cardiomyocyte
apoptosis in an in vivo model. These cardioprotective
effects of Sch B required activation of the PI3K/Akt signaling
pathway.
Acknowledgements
The present study was funded by the Science and
Technology Bureau of Lishui (grant no. 2012cxtd10).
References
|
1
|
Lapointe-Shaw L and Bell CM: Acute
myocardial infarction. Br Med J. 348:f76962014. View Article : Google Scholar
|
|
2
|
Fröhlich GM, Meier P, White SK, Yellon DM
and Hausenloy DJ: Myocardial reperfusion injury: Looking beyond
primary PCI. Eur Heart J. 34:1714–1722. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Iliodromitis K, Farmakis D, Andreadou I,
Zoga A, Bibli SI, Manolaki T, Dagres N, Iliodromitis EK,
Anastasiou-Nana M and Kremastinos DT: Various models of cardiac
conditioning in single or sequential periods of ischemia:
Comparative effects on infarct size and intracellular signaling.
Int J Cardiol. 168:1336–1341. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Lam PY and Ko KM: Schisandrin B as a
hormetic agent for preventing age-related neurodegenerative
diseases. Oxid Med Cell Longev. 2012:2508252012. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Panossian A and Wikman G: Pharmacology of
Schisandra chinensis Bail: An overview of Russian research
and uses in medicine. J Ethnopharmacol. 118:183–212. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Chiu PY, Leung HY, Poon MK, Lee SS and Ko
KM: Schisandrin B induced antioxidant response is partly mediated
by cytochrome P-4502E1 catalyzed reaction in mouse liver. Mol Cell
Biochem. 293:87–92. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Chang CY, Chen YL, Yang SC, Huang GC, Tsi
D, Huang CC, Chen JR and Li JS: Effect of schisandrin B and sesamin
mixture on CCl(4)-induced hepatic oxidative stress in rats.
Phytother Res. 23:251–256. 2009. View
Article : Google Scholar : PubMed/NCBI
|
|
8
|
Lee TH, Jung CH and Lee DH:
Neuroprotective effects of Schisandrin B against transient focal
cerebral ischemia in Sprague-Dawley rats. Food Chem Toxicol.
50:4239–4245. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Chen N, Chiu PY and Ko KM: Schisandrin B
enhances cerebral mitochondrial antioxidant status and structural
integrity, and protects against cerebral ischemia/reperfusion
injury in rats. Biol Pharm Bull. 31:1387–1391. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Chiu PY, Leung HY, Siu AH, Poon MK and Ko
KM: Schisandrin B decreases the sensitivity of mitochondria to
calcium ion induced permeability transition and protects against
ischemia/reperfusion injury in rat hearts. Acta Pharmacol Sin.
28:1559–1565. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Qiu Y, Li P and Ji C: Cell death
conversion under hypoxic condition in tumor development and
therapy. Int J Mol Sci. 16:25536–25551. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Zhao GX, Pan H, Ouyang DY and He XH: The
critical molecular interconnections in regulating apoptosis and
autophagy. Ann Med. 47:305–315. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Hu W, Zhang P, Gu J, Yu Q and Zhang D:
NEDD4-1 protects against ischaemia/reperfusion-induced
cardiomyocyte apoptosis via the PI3K/Akt pathway. Apoptosis.
22:437–448. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Black SC and Rodger IW: Methods for
studying experimental myocardial ischemic and reperfusion injury. J
Pharmacol Toxicol Methods. 35:179–190. 1996. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Gottlieb RA and Engler RL: Apoptosis in
myocardial ischemia-reperfusion. Ann New York Acad Sci.
874:412–426. 1999. View Article : Google Scholar
|
|
16
|
Kumar D and Jugdutt BI: Apoptosis and
oxidants in the heart. J Lab Clin Med. 142:288–297. 2003.
View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Othman AI, Elkomy MM, El-Missiry MA and
Dardor M: Epigallocatechin-3-gallate prevents cardiac apoptosis by
modulating the intrinsic apoptotic pathway in isoproterenol-induced
myocardial infarction. Eur J Pharmacol. 794:27–36. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Zhou PY, Zhang Z, Guo YL, Xiao ZZ, Zhu P,
Mai MJ and Zheng SY: Protective effect of antiapoptosis potency of
prolonged preservation by desiccation using high-pressure carbon
monoxide on isolated rabbit hearts. Transplant Proc. 47:2746–2751.
2015. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Lu Y, Zhou J, Xu C, Lin H, Xiao J, Wang Z
and Yang B: JAK/STAT and PI3K/AKT pathways form a mutual
transactivation loop and afford resistance to oxidative
stress-induced apoptosis in cardiomyocytes. Cell Physiol Biochem.
21:305–314. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Bharti S, Golechha M, Kumari S, Siddiqui
KM and Arya DS: Akt/GSK-3β/eNOS phosphorylation arbitrates
safranal-induced myocardial protection against ischemia-reperfusion
injury in rats. Eur J Nutr. 51:719–727. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Bharti S, Singh R, Chauhan SS, Hussain T,
Al-Attas OS and Arya DS: Phosphorylation of Akt/GSK-3β/eNOS
amplifies 5-HT2B receptor blockade mediated anti-hypertrophic
effect in rats. FEBS Lett. 586:180–185. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Ge N, Liu C, Li G, Xie L, Zhang Q, Li L,
Hao N and Zhang J: Hydrosulfide attenuates acute myocardial
ischemic injury through the glycogen synthase kinase-3β/β-catenin
signaling pathway. Int J Mol Med. 37:1281–1289. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Ong SB, Hall AR, Dongworth RK, Kalkhoran
S, Pyakurel A, Scorrano L and Hausenloy DJ: Akt protects the heart
against ischaemia-reperfusion injury by modulating mitochondrial
morphology. Thromb Haemost. 113:513–521. 2015. View Article : Google Scholar : PubMed/NCBI
|