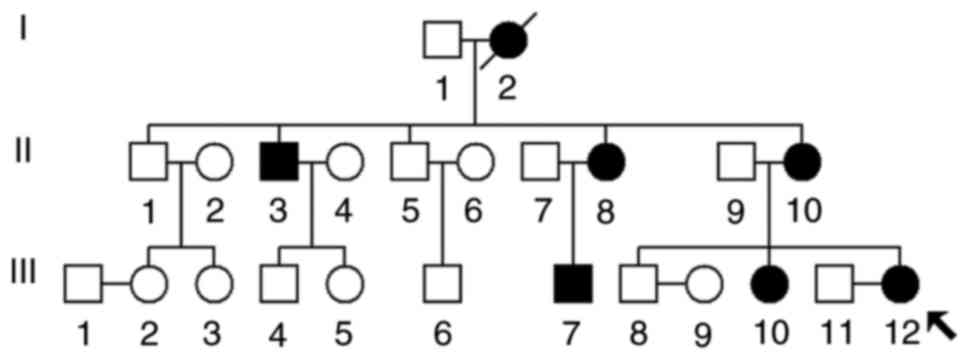
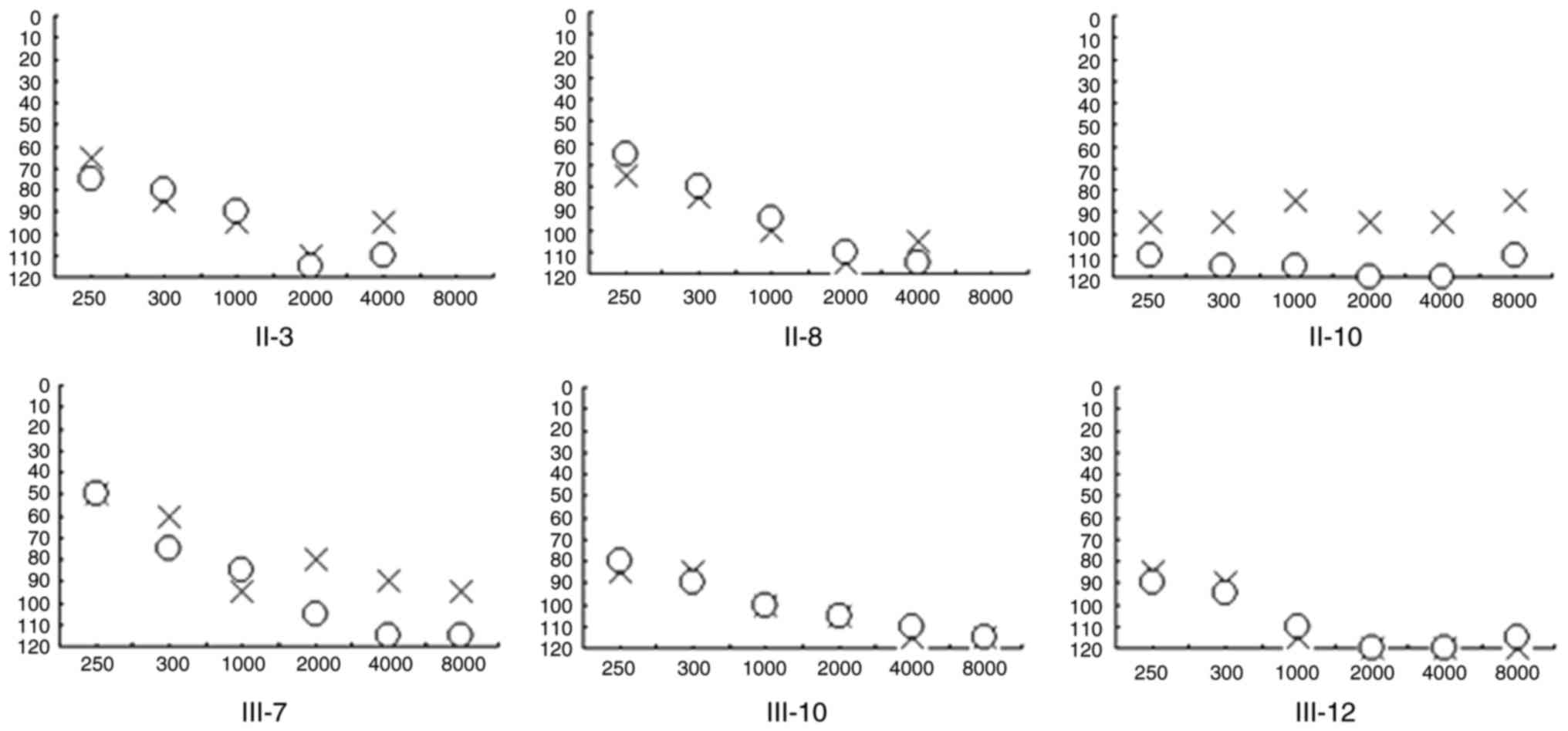
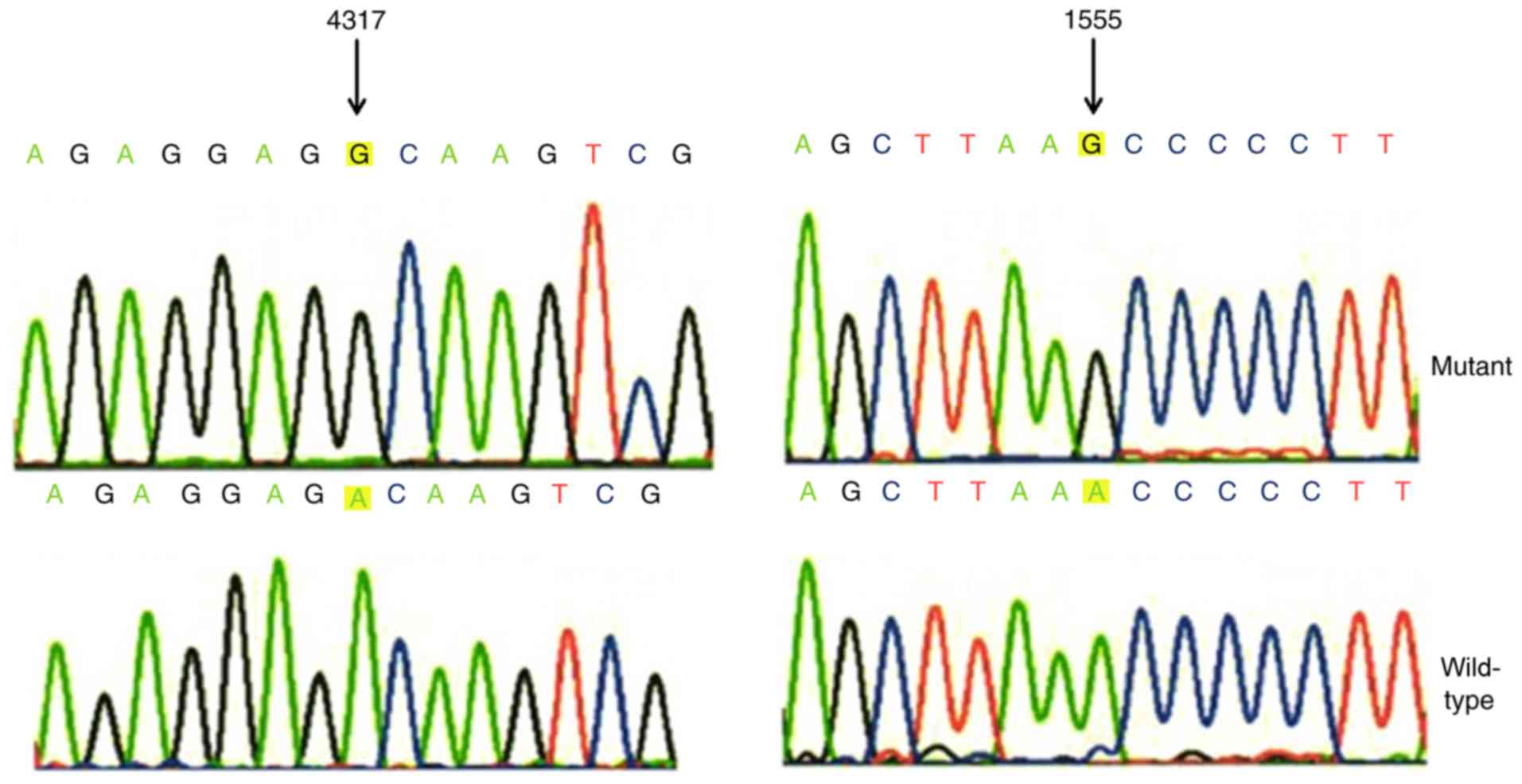
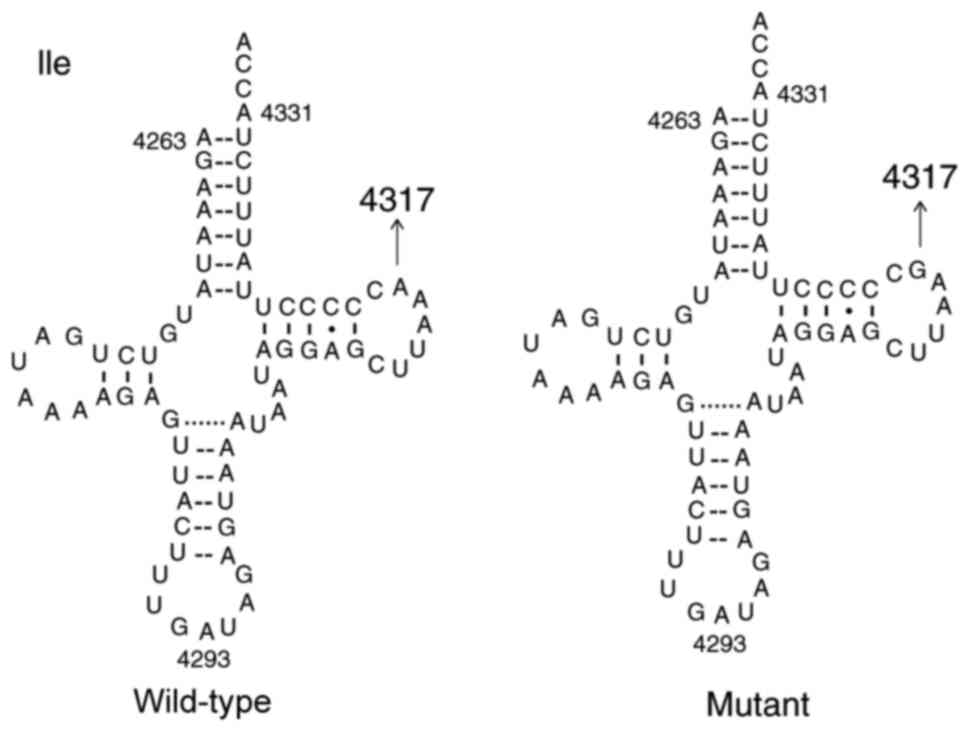
|
1
|
Lin X, Teng Y, Lan J, He B, Sun H and Xu
F: GRHL2 genetic polymorphisms may confer a protective effect
against sudden sensorineural hearing loss. Mol Med Rep.
13:2857–2863. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Nikopoulos K, Farinelli P, Giangreco B,
Tsika C, Royer-Bertrand B, Mbefo MK, Bedoni N, Kjellström U, El
Zaoui I, Di Gioia SA, et al: Mutations in CEP78 cause cone-rod
dystrophy and hearing loss associated with primary-cilia defects.
Am J Hum Genet. 99:770–776. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Namburi P, Ratnapriya R, Khateb S, Lazar
CH, Kinarty Y, Obolensky A, Erdinest I, Marks-Ohana D, Pras E,
Ben-Yosef T, et al: Bi-allelic truncating mutations in CEP78,
encoding centrosomal protein 78, cause cone-rod degeneration with
sensorineural hearing loss. Am J Hum Genet. 99:777–784. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Franzè A, Esposito G, Di Domenico C, Iossa
S, Sauchelli G, Fioretti T, Cavaliere M, Auletta G, Corvino V,
Laria C, et al: SLC26A4 genotypes associated with enlarged
vestibular aqueduct malformation in south Italian children with
sensorineural hearing loss. Clin Chem Lab Med. 54:e259–e263.
2016.PubMed/NCBI
|
|
5
|
Bakhchane A, Bousfiha A, Charoute H,
Salime S, Detsouli M, Snoussi K, Nadifi S, Kabine M, Rouba H, Dehbi
H, et al: Update of the spectrum of GJB2 gene mutations in 152
Moroccan families with autosomal recessive nonsyndromic hearing
loss. Eur J Med Genet. 59:325–329. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Kim SJ, Lee S, Park HJ, Kang TH, Sagong B,
Baek JI, Oh SK, Choi JY, Lee KY and Kim UK: Genetic association of
MYH genes with hereditary hearing loss in Korea. Gene. 591:177–182.
2016. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Guan J, Wang D, Cao W, Zhao Y, Du R, Yuan
H, Liu Q, Lan L, Zong L, Yang J, et al: SIX2 haploinsufficiency
causes conductive hearing loss with ptosis in humans. J Hum Genet.
61:917–922. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Zhu GJ, Shi LS, Zhou H, Yang Y, Chen J and
Gao X: A novel compound heterozygous mutation of SLC26A4 in two
Chinese families with nonsyndromic hearing loss and enlarged
vestibular aqueducts. Mol Med Rep. 16:9011–9016. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Wong SH, Wang WH, Chen PH, Li SY and Yang
JJ: Functional analysis of a nonsyndromic hearing loss-associated
mutation in the transmembrane II domain of the GJC3 gene. Int J Med
Sci. 14:246–256. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Xia W, Hu J, Liu F, Ma J, Sun S, Zhang J,
Jin K, Huang J, Jiang N, Wang X, et al: New role of LRP5,
associated with nonsyndromic autosomal-recessive hereditary hearing
loss. Hum Mutat. 38:1421–1431. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Lee KY, Choi SY, Bae JW, Kim S, Chung KW,
Drayna D, Kim UK and Lee SH: Molecular analysis of the GJB2, GJB6
and SLC26A4 genes in Korean deafness patients. Int J Pediatr
Otorhinolaryngol. 72:1301–1309. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Zhou XL, He LX, Yu LJ, Wang Y, Wang XJ,
Wang ED and Yang T: Mutations in KARS cause early-onset hearing
loss and leukoencephalopathy: Potential pathogenic mechanism. Hum
Mutat. 38:1740–1750. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Cai XZ, Li Y, Xia L, Peng Y, He CF, Jiang
L, Feng Y, Xia K, Liu XZ, Mei LY and Hu ZM: Exome sequencing
identifies POU4F3 as the causative gene for a large chinese family
with non-syndromic hearing loss. J Hum Genet. 62:317–320. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Palombo F, Al-Wardy N, Ruscone GA, Oppo M,
Kindi MN, Angius A, Al Lamki K, Girotto G, Giangregorio T, Benelli
M, et al: A novel founder MYO15a frameshift duplication is the
major cause of genetic hearing loss in oman. J Hum Genet.
62:259–264. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Ryu N, Lee S, Park HJ, Lee B, Kwon TJ, Bok
J, Park CI, Lee KY, Baek JI and Kim UK: Identification of a novel
splicing mutation within SLC17A8 in a Korean family with hearing
loss by whole-exome sequencing. Gene. 627:233–238. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Li D, Sun J, Zhao L, Guo W, Sun W and Yang
S: Aminoglycoside increases permeability of osseous spiral laminae
of cochlea by interrupting MMP-2 and MMP-9 balance. Neurotox Res.
31:348–357. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Sagwa EL, Souverein PC, Ribeiro I,
Leufkens HG and Mantel-Teeuwisse AK: Differences in
VigiBase® reporting of aminoglycoside and
capreomycin-suspected ototoxicity during tuberculosis treatment.
Pharmacoepidemiol Drug Saf. 26:1–8. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Ding Y, Leng J, Fan F, Xia B and Xu P: The
role of mitochondrial DNA mutations in hearing loss. Biochem Genet.
51:588–602. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Li Z, Li R, Chen J, Liao Z, Zhu Y, Qian Y,
Xiong S, Heman-Ackah S, Wu J, Choo DI and Guan MX: Mutational
analysis of the mitochondrial 12S rRNA gene in Chinese pediatric
subjects with aminoglycoside-induced and non-syndromic hearing
loss. Hum Genet. 117:9–15. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Zheng J, Ji Y and Guan MX: Mitochondrial
tRNA mutations associated with deafness. Mitochondrion. 12:406–413.
2012. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Guan MX, Fischel-Ghodsian N and Attardi G:
A biochemical basis for the inherited susceptibility to
aminoglycoside ototoxicity. Hum Mol Genet. 9:1787–1793. 2000.
View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Guan MX, Fischel-Ghodsian N and Attardi G:
Biochemical evidence for nuclear gene involvement in phenotype of
non-syndromic deafness associated with mitochondrial 12S rRNA
mutation. Hum Mol Genet. 5:963–971. 1996. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Lu J, Qian Y, Li Z, Yang A, Zhu Y, Li R,
Yang L, Tang X, Chen B, Ding Y, et al: Mitochondrial haplotypes may
modulate the phenotypic manifestation of the deafness-associated
12S rRNA 1555A>G mutation. Mitochondrion. 10:69–81. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Rieder MJ, Taylor SL, Tobe VO and
Nickerson DA: Automating the identification of DNA variations using
quality-based fluorescence re-sequencing: Analysis of the human
mitochondrial genome. Nucleic Acids Res. 26:967–973. 1998.
View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Andrews RM, Kubacka I, Chinnery PF,
Lightowlers RN, Turnbull DM and Howell N: Reanalysis and revision
of the cambridge reference sequence for human mitochondrial DNA.
Nat Genet. 23:1471999. View
Article : Google Scholar : PubMed/NCBI
|
|
26
|
Ding Y, Xia BH, Liu Q, Li MY, Huang SX and
Zhuo GC: Allele-specific PCR for detecting the deafness-associated
mitochondrial 12S rRNA mutations. Gene. 591:148–152. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Guan MX, Yan Q, Li X, Bykhovskaya Y,
Gallo-Teran J, Hajek P, Umeda N, Zhao H, Garrido G, Mengesha, et
al: Mutation in TRMU related to transfer RNA modification modulates
the phenotypic expression of the deafness-associated mitochondrial
12S ribosomal RNA mutations. Am J Hum Genet. 79:291–302. 2006.
View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Schmittgen TD, Zakrajsek BA, Mills AG,
Gorn V, Singer MJ and Reed MW: Quantitative reverse
transcription-polymerase chain reaction to study mRNA decay:
Comparison of endpoint and real-time methods. Anal Biochem.
285:194–204. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Bibb MJ, Van Etten RA, Wright CT, Walberg
MW and Clayton DA: Sequence and gene organization of mouse
mitochondrial DNA. Cell. 26:167–180. 1981. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Gadaleta G, Pepe G, De Candia G,
Quagliariello C, Sbisà E and Saccone C: The complete nucleotide
sequence of the Rattus norvegicus mitochondrial genome: Cryptic
signals revealed by comparative analysis between vertebrates. J Mol
Evol. 28:497–516. 1989. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Roe BA, Ma DP, Wilson R and Wong JF: The
complete nucleotide sequence of the xenopus laevis mitochondrial
genome. J Biol Chem. 260:9759–9774. 1985.PubMed/NCBI
|
|
32
|
Dai ZY, Sun BC, Huang SS, Yuan YY, Zhu YH,
Su Y and Dai P: Correlation analysis of phenotype and genotype of
GJB2 in patients with non-syndromic hearing loss in china. Gene.
570:272–276. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Kong QP, Bandelt HJ, Sun C, Yao YG, Salas
A, Achilli A, Wang CY, Zhong L, Zhu CL, Wu SF, et al: Updating the
East Asian mtDNA phylogeny: A prerequisite for the identification
of pathogenic mutations. Hum Mol Genet. 15:2076–2086. 2006.
View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Prezant TR, Agapian JV, Bohlman MC, Bu X,
Oztas S, Qiu WQ, Arnos KS, Cortopassi GA, Jaber L and Rotter JI:
Mitochondrial ribosomal RNA mutation associated with both
antibiotic-induced and non-syndromic deafness. Nat Genet.
4:289–294. 1993. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
el-Schahawi M, de Munain López A, Sarrazin
AM, Shanske AL, Basirico M, Shanske S and DiMauro S: Two large
Spanish pedigrees with nonsyndromic sensorineural deafness and the
mtDNA mutation at nt 1555 in the 12s rRNA gene: Evidence of
heteroplasmy. Neurology. 48:453–456. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Yuan EF, Xia W, Huang JT, Hu L, Liao X,
Dai X and Liu SM: A sensitive and convenient method for clinical
detection of non-syndromic hearing loss-associated common
mutations. Gene. 628:322–328. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Lu J, Li Z, Zhu Y, Yang A, Li R, Zheng J,
Cai Q, Peng G, Zheng W, Tang X, et al: Mitochondrial 12S rRNA
variants in 1642 han chinese pediatric subjects with
aminoglycoside-induced and nonsyndromic hearing loss.
Mitochondrion. 10:380–390. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Suzuki T and Nagao A: Human mitochondrial
tRNAs: Biogenesis, function, structural aspects, and diseases. Annu
Rev Genet. 45:299–329. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Pütz J, Giegé R and Florentz C: Diversity
and similarity in the tRNA world: Overall view and case study on
malaria-related tRNAs. FEBS Lett. 584:350–358. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Tanaka M, Ino H, Ohno K, Hattori K, Sato
W, Ozawa T, Tanaka T and Itoyama S: Mitochondrial mutation in fatal
infantile cardiomyopathy. Lancet. 336:14521990. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Lechowicz U, Pollak A, Frączak A,
Rydzanicz M, Stawiński P, Lorens A, Skarżyński PH, Skarżyński H,
Płoski R and Ołdak M: Application of next-generation sequencing to
identify mitochondrial mutations: Study on m.7511T>C in patients
with hearing loss. Mol Med Rep. 17:1782–1790. 2018.PubMed/NCBI
|
|
42
|
Zhu HY, Wang SW, Liu L, Chen R, Wang L,
Gong XL and Zhang ML: Genetic variants in mitochondrial tRNA genes
are associated with essential hypertension in a chinese Han
population. Clin Chim Acta. 410:64–69. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Degoul F, Brule H, Cepanec C, Helm M,
Marsac C, Leroux J, Giegé R and Florentz C: Isoleucylation
properties of native human mitochondrial tRNAIle and tRNAIle
transcripts. Implications for cardiomyopathy-related point
mutations (4269, 4317) in the tRNAIle gene. Hum Mol Genet.
7:347–354. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Ding Y, Zhuo G and Zhang C: The
mitochondrial tRNALeu(UUR) A3302G mutation may be associated with
insulin resistance in woman with polycystic ovary syndrome. Reprod
Sci. 23:228–233. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Jeng JY, Yeh TS, Lee JW, Lin SH, Fong TH
and Hsieh RH: Maintenance of mitochondrial DNA copy number and
expression are essential for preservation of mitochondrial function
and cell growth. J Cell Biochem. 103:347–357. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Shibata SB, Ranum PT, Moteki H, Pan B,
Goodwin AT, Goodman SS, Abbas PJ, Holt JR and Smith RJH: RNA
interference prevents autosomal-dominant hearing loss. Am J Hum
Genet. 98:1101–1113. 2016. View Article : Google Scholar : PubMed/NCBI
|