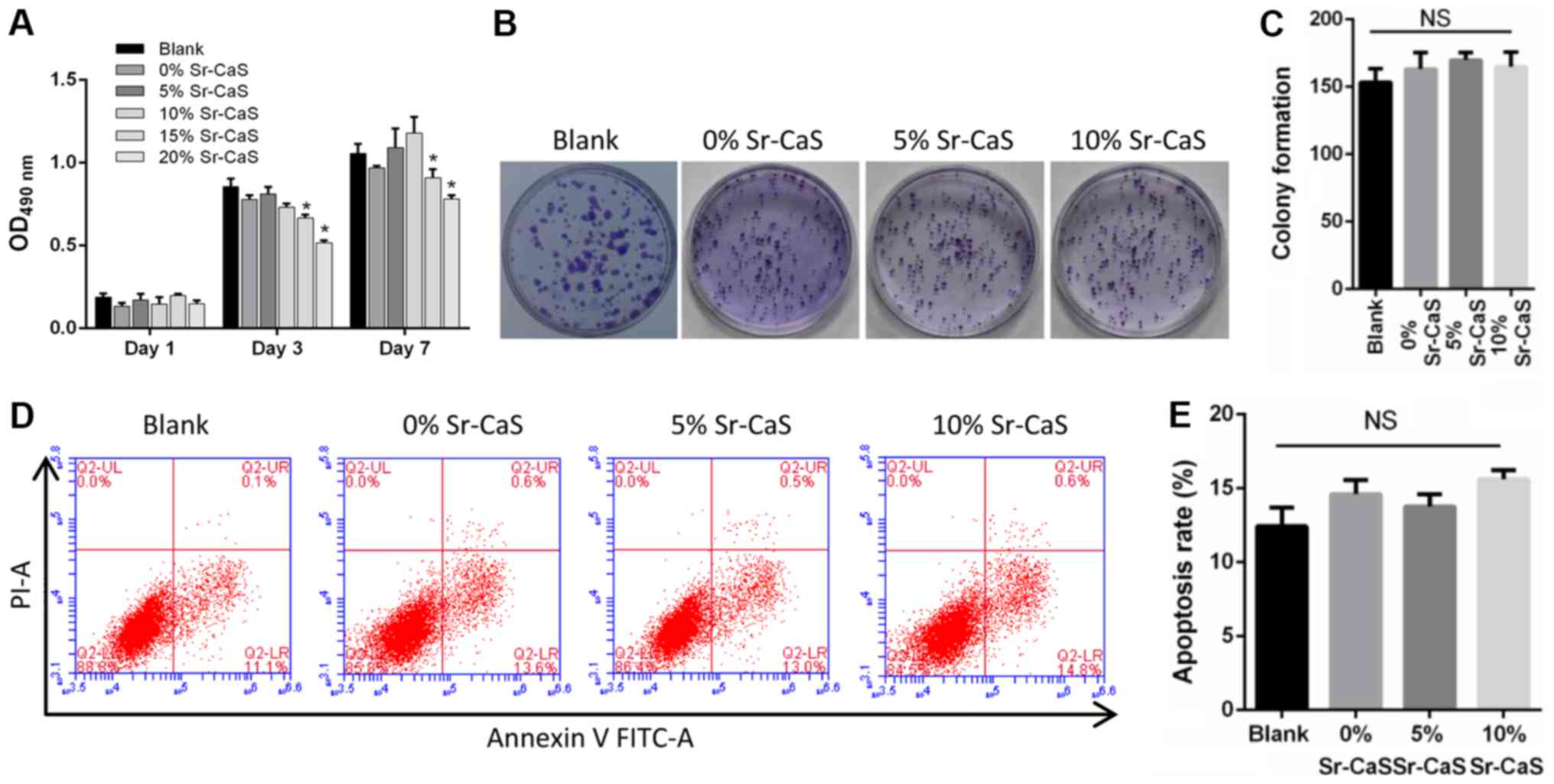
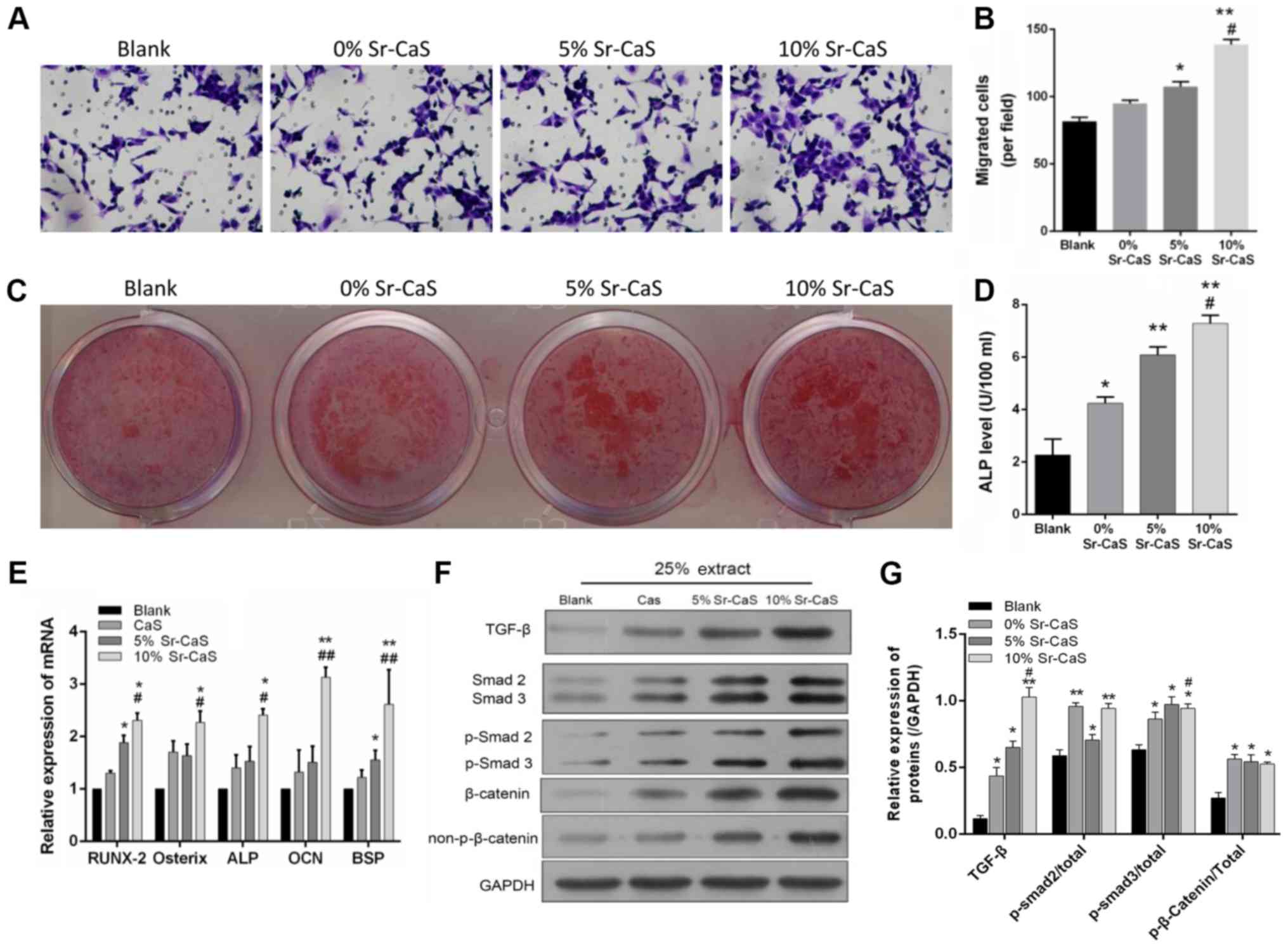
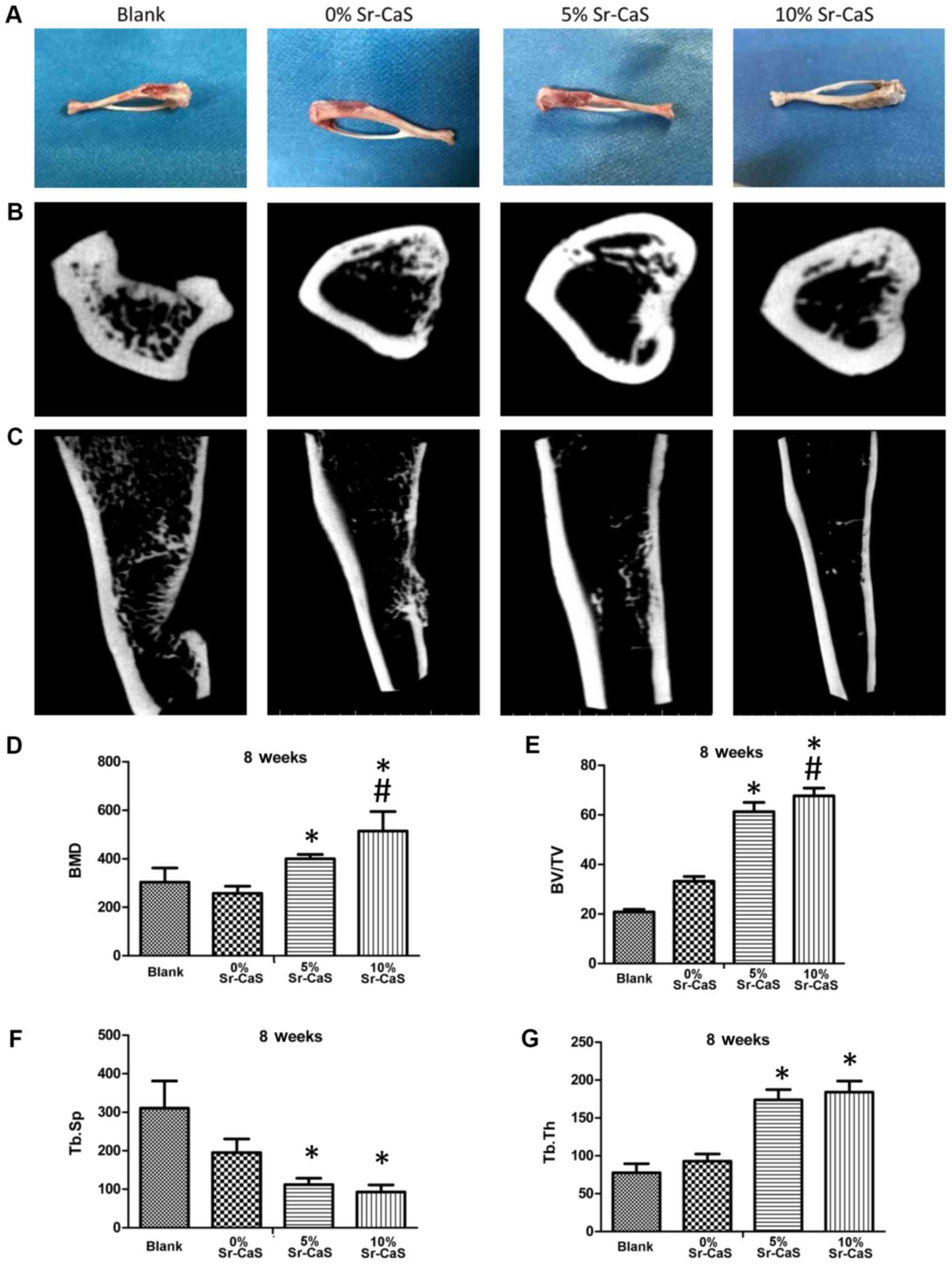
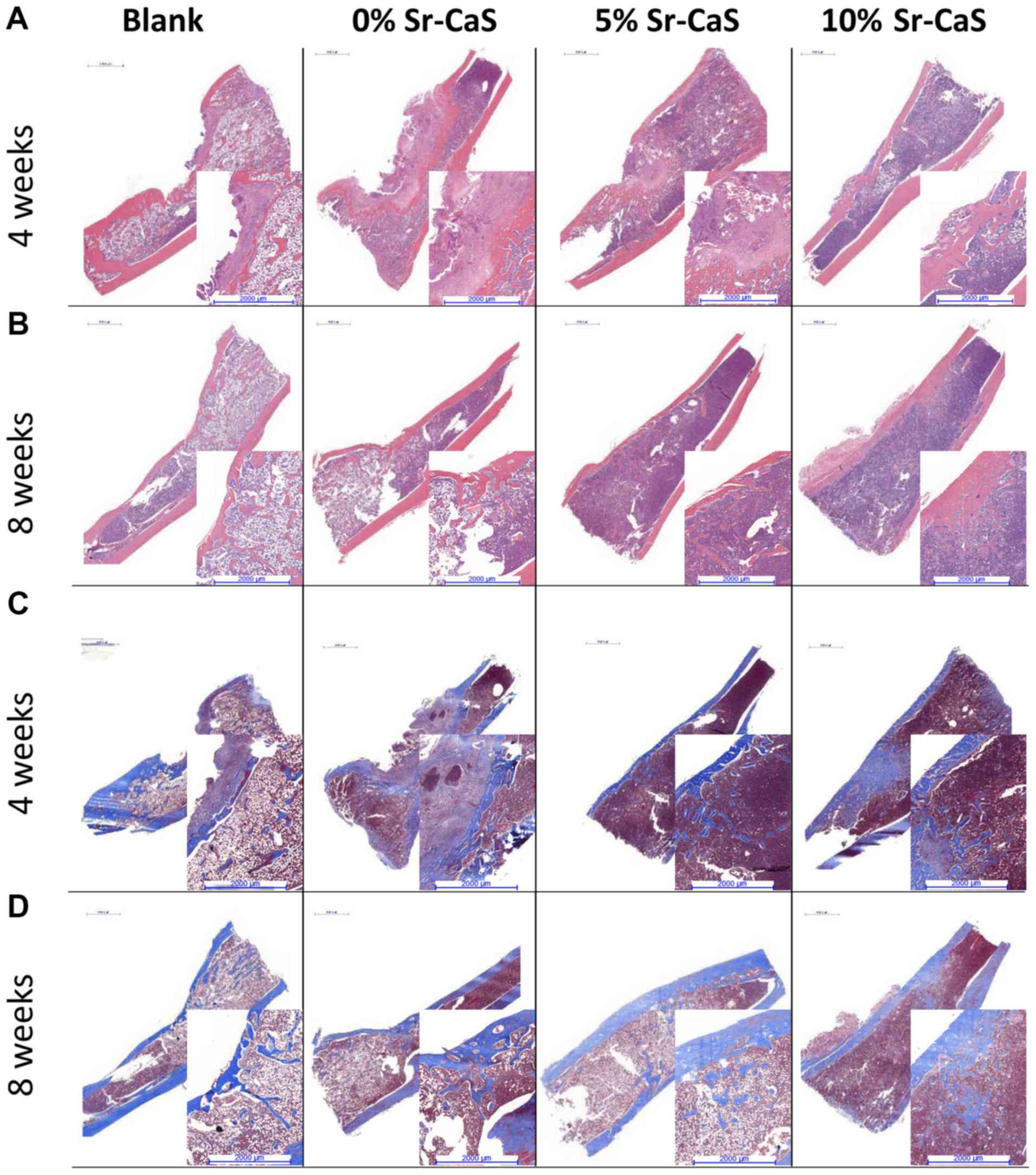
|
1
|
Ishack S, Mediero A, Wilder T, Ricci JL
and Cronstein BN: Bone regeneration in critical bone defects using
three-dimensionally printed beta-tricalcium
phosphate/hydroxyapatite scaffolds is enhanced by coating scaffolds
with either dipyridamole or BMP-2. J Biomed Mater Res B Appl
Biomater. 105:366–375. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Garcia-Gareta E, Coathup MJ and Blunn GW:
Osteoinduction of bone grafting materials for bone repair and
regeneration. Bone. 81:112–121. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
van Houdt CI, Tim CR, Crovace MC, Zanotto
ED, Peitl O, Ulrich DJ, Jansen JA, Parizotto NA, Renno AC and van
den Beucken JJ: Bone regeneration and gene expression in bone
defects under healthy and osteoporotic bone conditions using two
commercially available bone graft substitutes. Biomed Mater.
10:0350032015. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Lazar MA, Rotaru H, Baldea I, Bosca AB,
Berce CP, Prejmerean C, Prodan D and Campian RS: Evaluation of the
biocompatibility of new fiber-reinforced composite materials for
craniofacial bone reconstruction. J Craniofac Surg. 27:1694–1699.
2016. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Sahoo N, Roy ID, Desai AP and Gupta V:
Comparative evaluation of autogenous calvarial bone graft and
alloplastic materials for secondary reconstruction of cranial
defects. J Craniofac Surg. 21:79–82. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Wang W and Yeung KW: Bone grafts and
biomaterials substitutes for bone defect repair: A review.
Bioactive Mater. 2:224–247. 2017. View Article : Google Scholar
|
|
7
|
Kenley RA, Yim K, Abrams J, Ron E, Turek
T, Marden LJ and Hollinger JO: Biotechnology and bone graft
substitutes. Pharm Res. 10:1393–1401. 1993. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Barrere F, van Blitterswijk CA and de
Groot K: Bone regeneration: Molecular and cellular interactions
with calcium phosphate ceramics. Int J Nanomedicine. 1:317–332.
2006.PubMed/NCBI
|
|
9
|
Scarano A, Orsini G, Pecora G, Iezzi G,
Perrotti V and Piattelli A: Peri-implant bone regeneration with
calcium sulfate: A light and transmission electron microscopy case
report. Implant Dent. 16:195–203. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Shaffer CD and App GR: The use of plaster
of paris in treating infrabony periodontal defects in humans. J
Periodontol. 42:685–690. 1971. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Hing KA, Wilson LF and Buckland T:
Comparative performance of three ceramic bone graft substitutes.
Spine J. 7:475–490. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Frenkel SR, Simon J, Alexander H, Dennis M
and Ricci JL: Osseointegration on metallic implant surfaces:
Effects of microgeometry and growth factor treatment. J Biomed
Mater Res. 63:706–713. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Liu T, Zhang X, Li Z and Peng D:
Management of combined bone defect and limb-length discrepancy
after tibial chronic osteomyelitis. Orthopedics. 34:e363–e367.
2011.PubMed/NCBI
|
|
14
|
Villar CC and Cochran DL: Regeneration of
periodontal tissues: Guided tissue regeneration. Dent Clin North
Am. 54:73–92. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Huang Z, Li B, Li Q, Huang Z, Yin B, Ma P,
Xu D, Wu Z and Qiu G: Effect of injectable composites of calcium
sulfate and hyaluronate in enhancing osteogenesis. Zhongguo Xiu Fu
Chong Jian Wai Ke Za Zhi. 31:730–737. 2017.(In Chinese). PubMed/NCBI
|
|
16
|
Cao L, Weng W, Chen X, Zhang J, Zhou Q,
Cui J, Zhao Y, Shin JW and Su J: Promotion of in vivo
degradability, vascularization and osteogenesis of calcium
sulfate-based bone cements containing nanoporous lithium doping
magnesium silicate. Int J Nanomedicine. 12:1341–1352. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Liu X, Liu HY, Lian X, Shi XL, Wang W, Cui
FZ and Zhang Y: Osteogenesis of mineralized collagen bone graft
modified by PLA and calcium sulfate hemihydrate: In vivo study. J
Biomater Appl. 28:12–19. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Yin W, Sun Q and Ma L: Study on the
effects of curculigoside on proliferation, differentiation, and
calcification of mouse osteoblastic MC3T3-E1 cells. World Sci
Technol. 13:852–855. 2011. View Article : Google Scholar
|
|
19
|
Beuerlein MJ and Mckee MD: Calcium
sulfates: What is the evidence? J Orthop Trauma. 24 (Suppl
1):S46–S51. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Bonnelye E, Chabadel A, Saltel F and
Jurdic P: Dual effect of strontium ranelate: Stimulation of
osteoblast differentiation and inhibition of osteoclast formation
and resorption in vitro. Bone. 42:129–138. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Marie PJ, Hott M, Modrowski D, De Pollak
C, Guillemain J, Deloffre P and Tsouderos Y: An uncoupling agent
containing strontium prevents bone loss by depressing bone
resorption and maintaining bone formation in estrogen-deficient
rats. J Bone Miner Res. 8:607–615. 1993. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Canalis E, Hott M, Deloffre P, Tsouderos Y
and Marie PJ: The divalent strontium salt S12911 enhances bone cell
replication and bone formation in vitro. Bone. 18:517–523. 1996.
View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Zhang J, Zhao S, Zhu Y, Huang Y, Zhu M,
Tao C and Zhang C: Three-dimensional printing of
strontium-containing mesoporous bioactive glass scaffolds for bone
regeneration. Acta Biomater. 10:2269–2281. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Poh PS, Hutmacher DW, Stevens MM and
Woodruff MA: Fabrication and in vitro characterization of bioactive
glass composite scaffolds for bone regeneration. Biofabrication.
5:0450052013. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Ren J, Blackwood KA, Doustgani A, Poh PP,
Steck R, Stevens MM and Woodruff MA: Melt-electrospun
polycaprolactone strontium-substituted bioactive glass scaffolds
for bone regeneration. J Biomed Mater Res A. 104:21092016.
View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Li X, Xu CP, Hou YL, Song JQ, Cui Z, Wang
SN, Huang L, Zhou CR and Yu B: A novel resorbable
strontium-containing α-calcium sulfate hemihydrate bone substitute:
A preparation and preliminary study. Biomed Mater. 9:0450102014.
View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Li Y, Chen SK, Li L, Qin L, Wang XL and
Lai YX: Bone defect animal models for testing efficacy of bone
substitute biomaterials. J Orthop Translat. 3:95–104. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Del Rosario C, Rodriguez-Evora M, Reyes R,
Delgado A and Evora C: BMP-2, PDGF-BB, and bone marrow mesenchymal
cells in a macroporous beta-TCP scaffold for critical-size bone
defect repair in rats. Biomed Mater. 10:0450082015. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) method. Methods. 25:402–408. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Yu J, Xu L, Li K, Xie N, Xi Y, Wang Y,
Zheng X, Chen X, Wang M and Ye X: Zinc-modified calcium silicate
coatings promote osteogenic differentiation through TGF-β/smad
pathway and osseointegration in osteopenic rabbits. Sci Rep.
7:34402017. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Yamada Y, Ueda M, Hibi H and Nagasaka T:
Translational research for injectable tissue-engineered bone
regeneration using mesenchymal stem cells and platelet-rich plasma:
From basic research to clinical case study. Cell Transplant.
13:343–355. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Bajada S, Harrison PE, Ashton BA,
Cassar-Pullicino VN, Ashammakhi N and Richardson JB: Successful
treatment of refractory tibial nonunion using calcium sulphate and
bone marrow stromal cell implantation. J Bone Joint Surg Br.
89:1382–1386. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Quarto R, Mastrogiacomo M, Cancedda R,
Kutepov SM, Mukhachev V, Lavroukov A, Kon E and Marcacci M: Repair
of large bone defects with the use of autologous bone marrow
stromal cells. N Engl J Med. 344:385–386. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Muschler GF, Nakamoto C and Griffith LG:
Engineering principles of clinical cell-based tissue engineering. J
Bone Joint Surg Am. 86:1541–1558. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Guanghua Chen, Guizhi Huang, Hao Lin,
Haojun Wu and Chen H: Bone marrow mesenchymal stem cell
transplatation increases bone mineral density of an ovariectomized
rat model of osteoporosis. Chin J Tissue Engineering Res. 21:49–53.
2017.
|
|
36
|
Cesareo R, Napolitano C and Iozzino M:
Strontium ranelate in postmenopausal osteoporosis treatment: A
critical appraisal. Int J Womens Health. 2:1–6. 2010.PubMed/NCBI
|
|
37
|
Kostenuik PJ and Shalhoub V:
Osteoprotegerin: A physiological and pharmacological inhibitor of
bone resorption. Curr Pharm Des. 7:613–635. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Chen QY, Liang GQ, Lin Y, Liu BL and
Zhao-Hui LI: Effects of strontium ranelate on titanium particles
stimulating mononuclear macrophage to secrete osteolysis factor and
its RANK expression. Rheum Arthritis. 2015.
|
|
39
|
Li Y, Li J, Zhu S, Luo E, Feng G, Chen Q
and Hu J: Effects of strontium on proliferation and differentiation
of rat bone marrow mesenchymal stem cells. Biochem Biophys Res
Commun. 418:725–730. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Gentleman E, Fredholm YC, Jell G,
Lotfibakhshaiesh N, O'Donnell MD, Hill RG and Stevens MM: The
effects of strontium-substituted bioactive glasses on osteoblasts
and osteoclasts in vitro. Biomaterials. 31:3949–3956. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Nahass HE, Din NNE and Nasry SA: The
effect of strontium ranelate gel on bone formation in calvarial
critical size defects. Open Access Maced J Med Sci. 5:994–999.
2017. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Zhao S, Wang X, Li N, Chen Y, Su Y and
Zhang J: Effects of strontium ranelate on bone formation in the
mid-palatal suture after rapid maxillary expansion. Drug Des Devel
Ther. 9:2725–2734. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Karatas OH, Toy E, Demir A, Toy H and
Kozacioglu S: Effects of strontium ranelate on sutural bone
formation: A histological and immunohistochemical study. Aust
Orthod J. 32:139–147. 2016.PubMed/NCBI
|
|
44
|
Meunier PJ, Roux C, Seeman E, Ortolani S,
Badurski JE, Spector TD, Cannata J, Balogh A, Lemmel EM,
Pors-Nielsen S, et al: The effects of strontium ranelate on the
risk of vertebral fracture in women with postmenopausal
osteoporosis. N Engl J Med. 350:459–468. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Chen Y, Zhou Y, Yang S, Li JJ, Li X, Ma Y,
Hou Y, Jiang N, Xu C, Zhang S, et al: Novel bone substitute
composed of chitosan and strontium-doped α-calcium sulfate
hemihydrate: Fabrication, characterisation and evaluation of
biocompatibility. Mater Sci Eng C Mater Biol Appl. 66:84–91. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Neves N, Linhares D, Costa G, Ribeiro CC
and Barbosa MA: In vivo and clinical application of
strontium-enriched biomaterials for bone regeneration: A systematic
review. Bone Joint Res. 6:366–375. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Yang D, Okamura H and Qiu L: Upregulated
osterix expression elicited by Runx2 and Dlx5 is required for the
accelerated osteoblast differentiation in PP2A Cα-knockdown cells.
Cell Biol Int. 42:403–410. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Komori T: Runx2, an inducer of osteoblast
and chondrocyte differentiation. Histochem Cell Biol. 149:313–323.
2018. View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Mundlos S, Otto F, Mundlos C, Mulliken JB,
Aylsworth AS, Albright S, Lindhout D, Cole WG, Henn W, Knoll JH, et
al: Mutations involving the transcription factor CBFA1 cause
cleidocranial dysplasia. Cell. 89:773–779. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Nakashima K, Zhou X, Kunkel G, Zhang Z,
Deng JM, Behringer RR and de Crombrugghe B: The novel zinc
finger-containing transcription factor osterix is required for
osteoblast differentiation and bone formation. Cell. 108:17–29.
2002. View Article : Google Scholar : PubMed/NCBI
|
|
51
|
Zhou H, Choong P, McCarthy R, Chou ST,
Martin TJ and Ng KW: In situ hybridization to show sequential
expression of osteoblast gene markers during bone formation in
vivo. J Bone Miner Res. 9:1489–1499. 1994. View Article : Google Scholar : PubMed/NCBI
|
|
52
|
van Leeuwen JP, van Driel M, van den Bemd
GJ and Pols HA: Vitamin D control of osteoblast function and bone
extracellular matrix mineralization. Crit Rev Eukaryot Gene Expr.
11:199–226. 2001.PubMed/NCBI
|
|
53
|
Takahashi T, Kato S, Suzuki N, Kawabata N
and Takagi M: Autoregulatory mechanism of Runx2 through the
expression of transcription factors and bone matrix proteins in
multipotential mesenchymal cell line, ROB-C26. J Oral Sci.
47:199–207. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
54
|
Riminucci M, Corsi A, Peris K, Fisher LW,
Chimenti S and Bianco P: Coexpression of bone sialoprotein (BSP)
and the pivotal transcriptional regulator of osteogenesis,
Cbfa1/Runx2, in malignant melanoma. Calcif Tissue Int. 73:281–289.
2003. View Article : Google Scholar : PubMed/NCBI
|
|
55
|
Chen G, Deng C and Li YP: TGF-β and BMP
signaling in osteoblast differentiation and bone formation. Int J
Biol Sci. 8:272–288. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
56
|
Rahman MS, Akhtar N, Jamil HM, Banik RS
and Asaduzzaman SM: TGF-β/BMP signaling and other molecular events:
Regulation of osteoblastogenesis and bone formation. Bone Res.
3:150052015. View Article : Google Scholar : PubMed/NCBI
|
|
57
|
Wu M, Chen G and Li YP: TGF-β and BMP
signaling in osteoblast, skeletal development, and bone formation,
homeostasis and disease. Bone Res. 4:160092016. View Article : Google Scholar : PubMed/NCBI
|
|
58
|
Leah E: Osteoarthritis: TGF-β overload at
bones of cartilage degeneration. Nat Rev Rheumatol. 9:3822013.
View Article : Google Scholar : PubMed/NCBI
|
|
59
|
Saito M, Ichikawa J, Ando T, Schoenecker
JG, Ohba T, Koyama K, Suzuki-Inoue K and Haro H: Platelet-derived
TGF-β induces tissue factor expression via the smad3 pathway in
osteosarcoma cells. J Bone Miner Res. 33:2048–2058. 2018.
View Article : Google Scholar : PubMed/NCBI
|
|
60
|
Li H, Fan J, Fan L, Li T, Yang Y, Xu H,
Deng L, Li J, Li T, Weng X, et al: MiRNA-10b reciprocally
stimulates osteogenesis and inhibits adipogenesis partly through
the TGF-β/SMAD2 signaling pathway. Aging Dis. 9:1058–1073. 2018.
View Article : Google Scholar : PubMed/NCBI
|
|
61
|
Chen X, Hu C, Wang G, Li L, Kong X, Ding Y
and Jin Y: Nuclear factor-κB modulates osteogenesis of periodontal
ligament stem cells through competition with β-catenin signaling in
inflammatory microenvironments. Cell Death Dis. 4:e5102013.
View Article : Google Scholar : PubMed/NCBI
|
|
62
|
Amini Nik S, Ebrahim RP, Van Dam K,
Cassiman JJ and Tejpar S: TGF-beta modulates beta-Catenin stability
and signaling in mesenchymal proliferations. Exp Cell Res.
313:2887–2895. 2007. View Article : Google Scholar : PubMed/NCBI
|