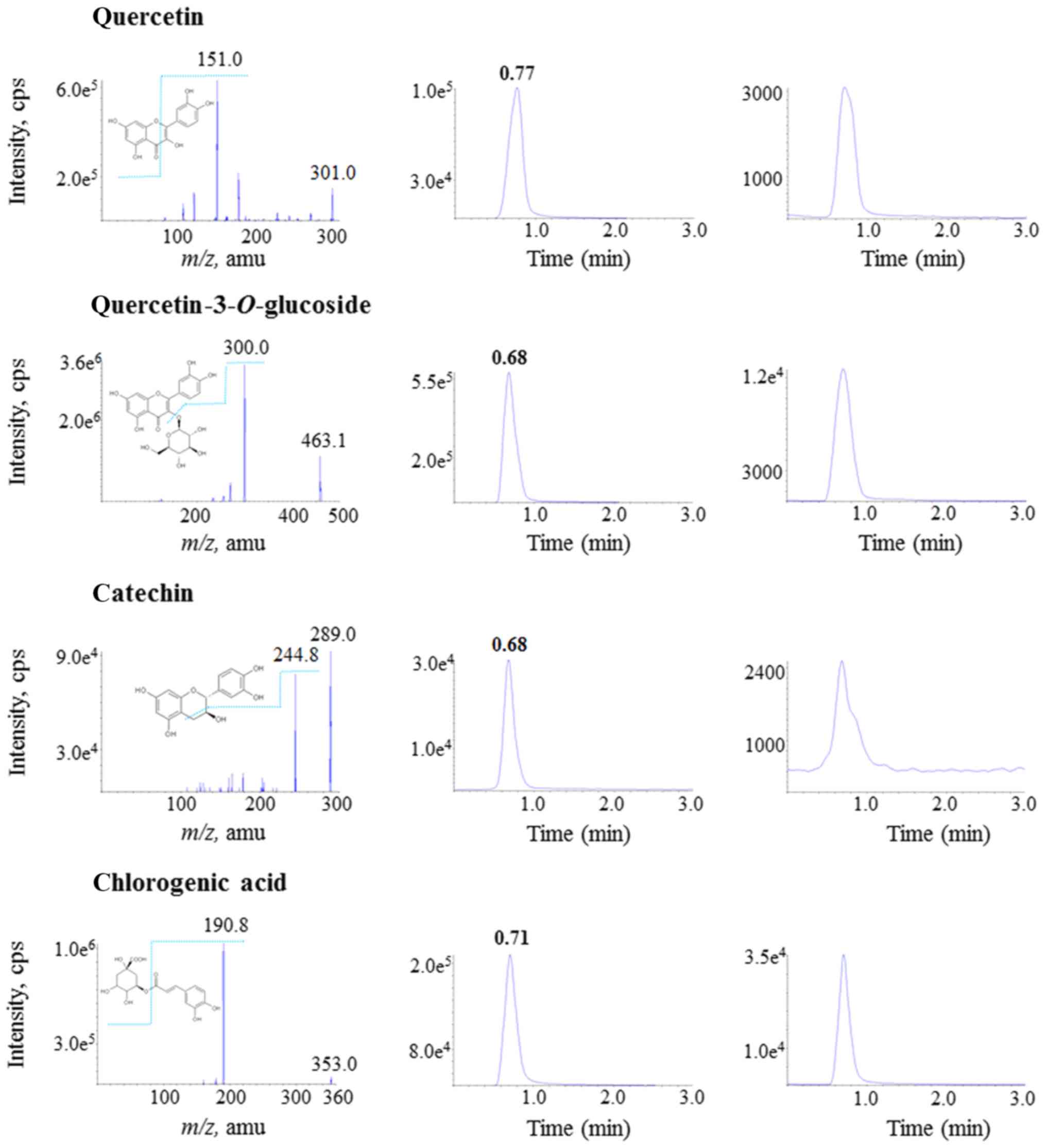
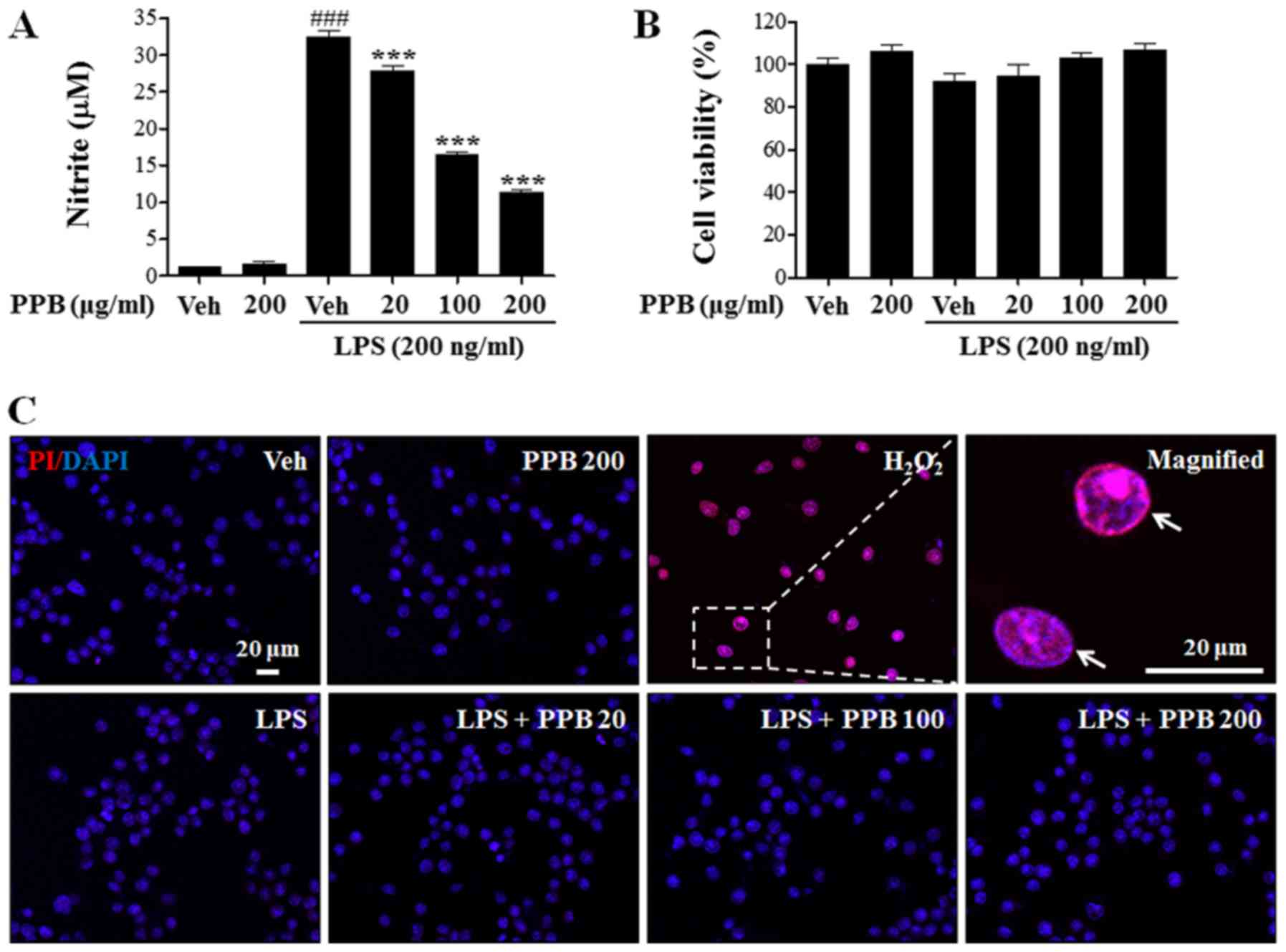
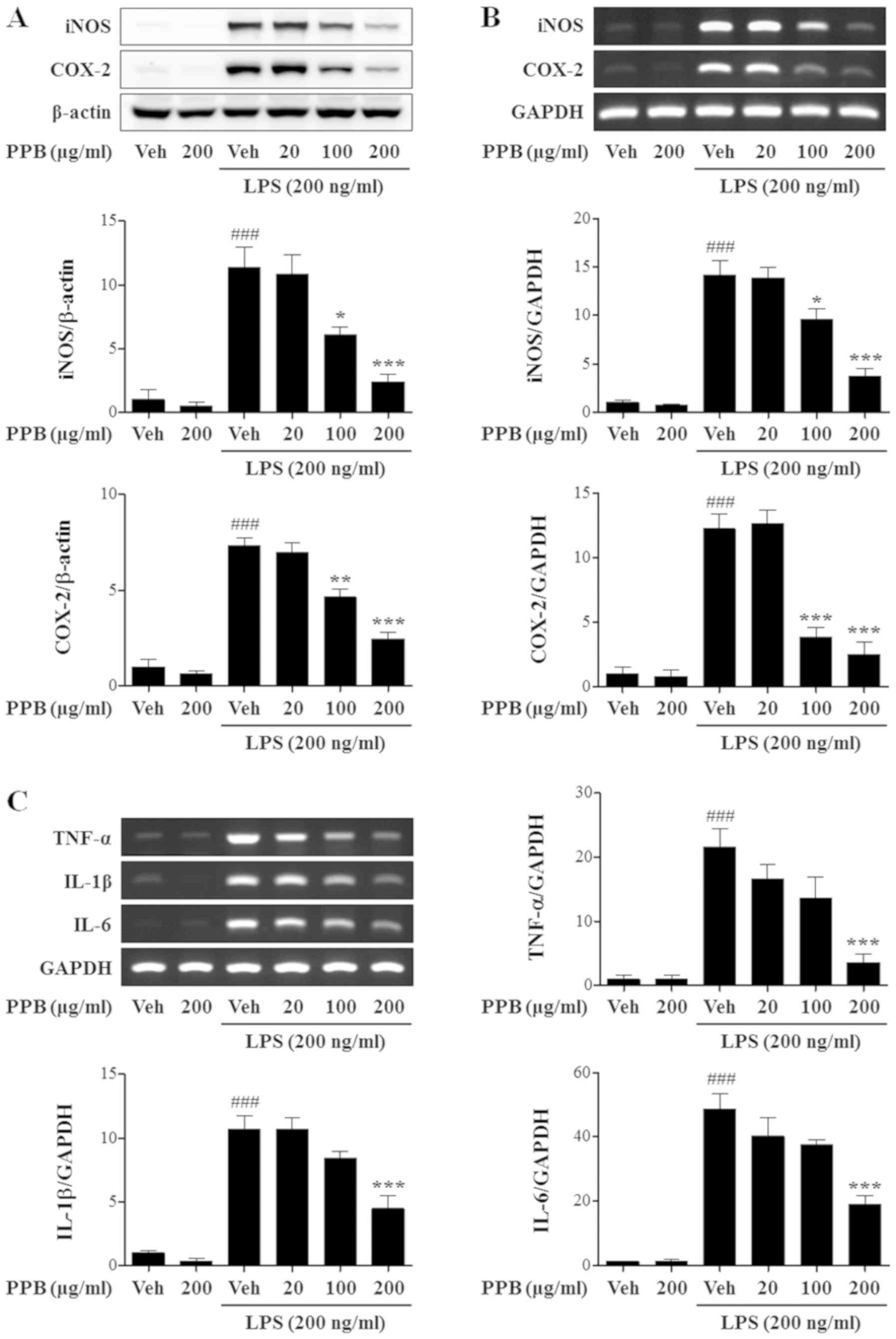
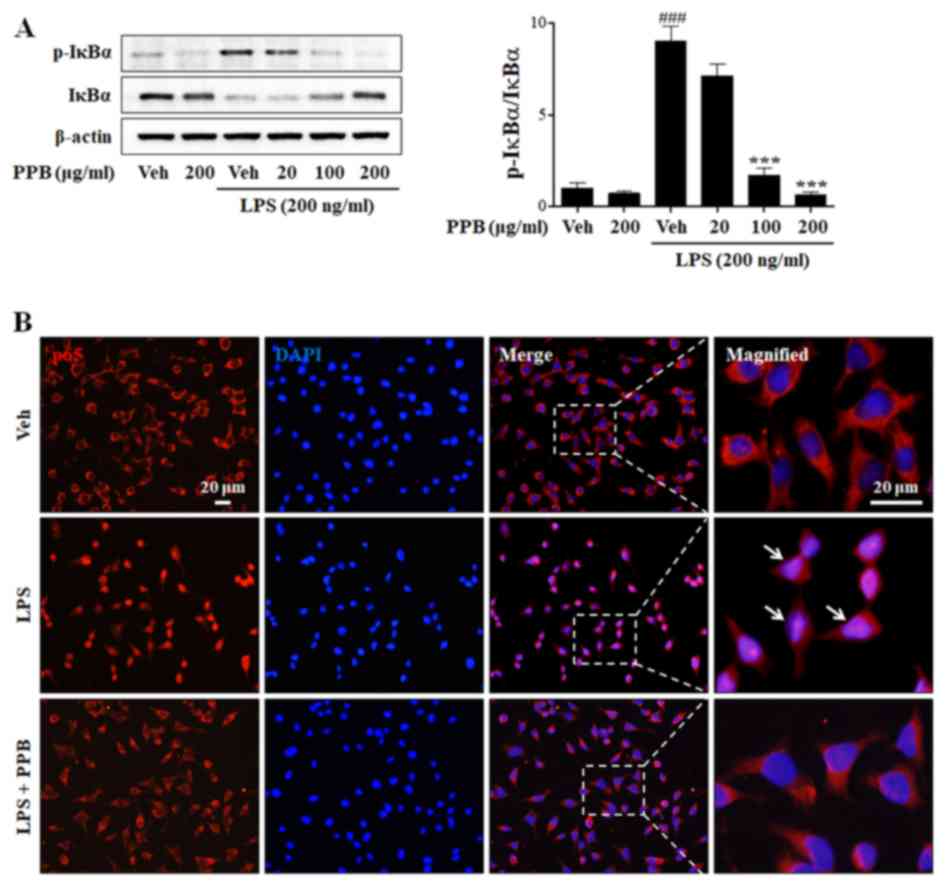
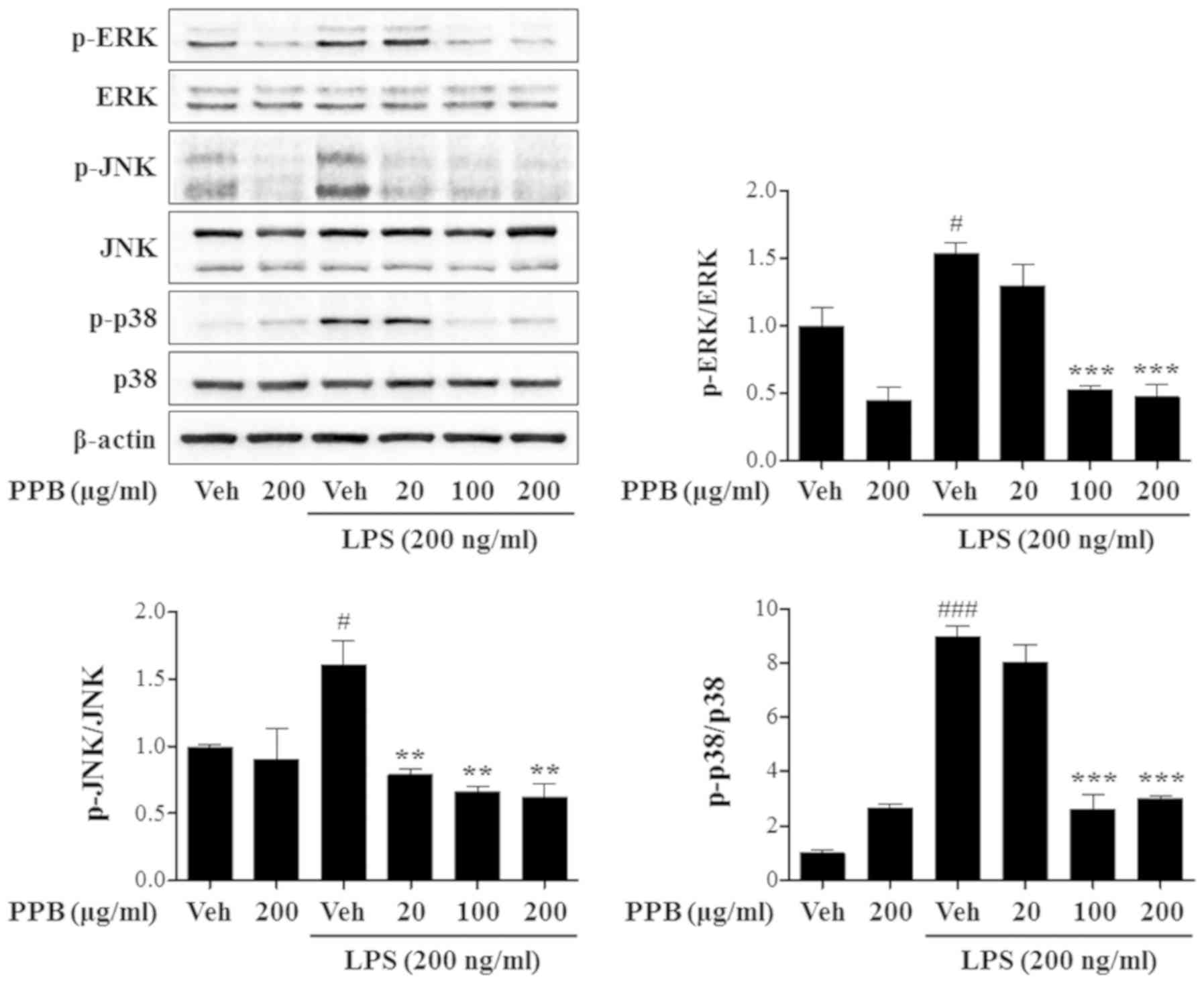
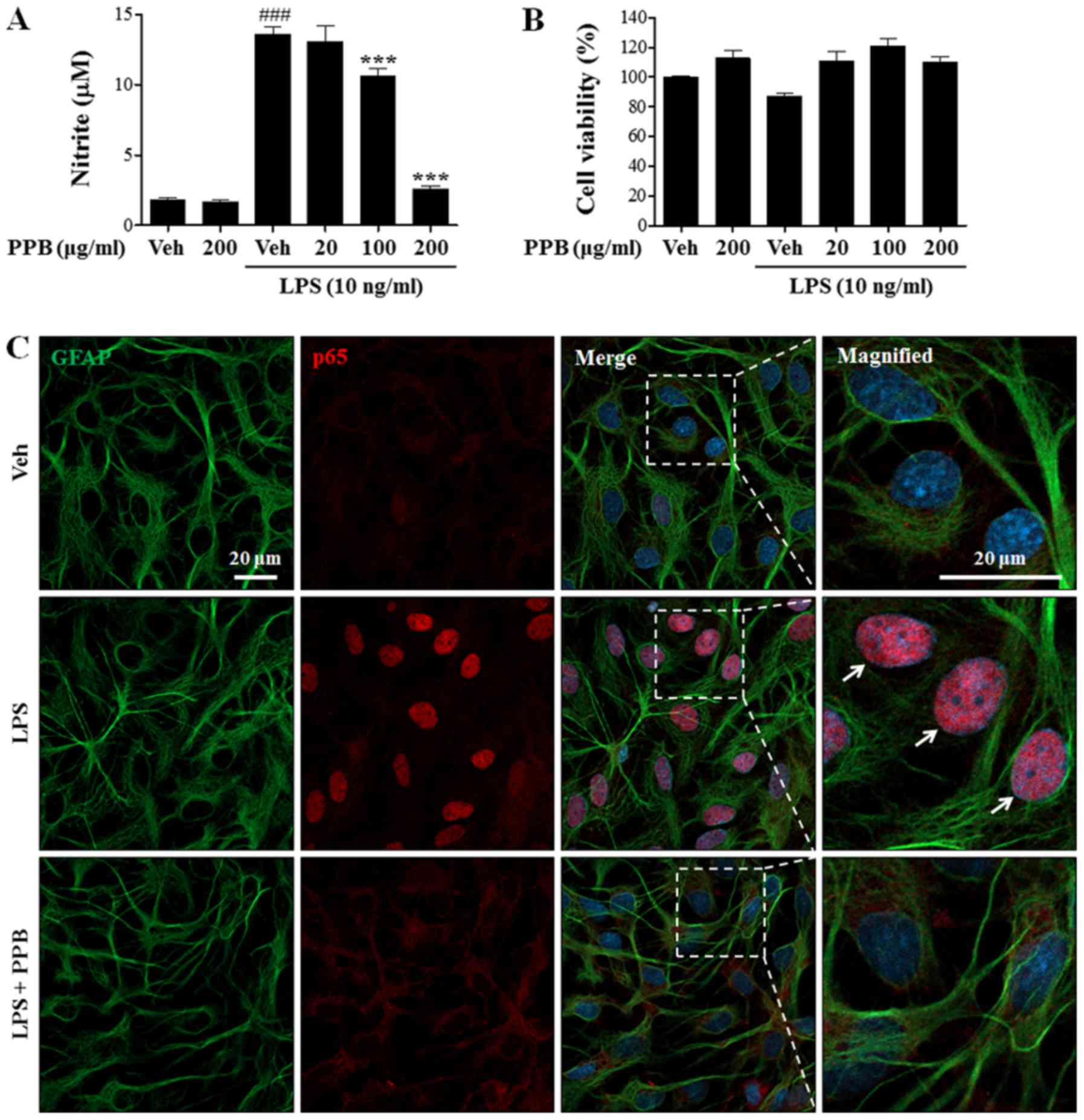
|
1
|
González-Scarano F and Baltuch G:
Microglia as mediators of inflammatory and degenerative diseases.
Annu Rev Neurosci. 22:219–240. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Maragakis NJ and Rothstein JD: Mechanisms
of disease: Astrocytes in neurodegenerative disease. Nat Clin Pract
Neurol. 2:679–689. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Prinz M and Priller J: Microglia and brain
macrophages in the molecular age: From origin to neuropsychiatric
disease. Nat Rev Neurosci. 15:300–312. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Liu B and Hong JS: Role of microglia in
inflammation-mediated neurodegenerative diseases: Mechanisms and
strategies for therapeutic intervention. J Pharmacol Exp Ther.
304:1–7. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Sanchez Guajardo V, Tentillier N and
Romero Ramos M: The relation between α-synuclein and microglia in
Parkinson's disease: Recent developments. Neuroscience. 302:47–58.
2015. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Keren Shaul H, Spinrad A, Weiner A,
Matcovitch Natan O, Dvir Szternfeld R, Ulland TK, David E, Baruch
K, Lara Astaiso D, Toth B, et al: A unique microglia type
associated with restricting development of Alzheimer's disease.
Cell. 169:1276–1290. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Kim DC, Lee DS, Ko W, Kim KW, Kim HJ, Yoon
CS, Oh H and Kim YC: Heme oxygenase-1-inducing activity of
4-methoxydalbergione and 4′-hydroxy-4-methoxydalbergione from
Dalbergia odorifera and their anti-inflammatory and cytoprotective
effects in murine hippocampal and BV2 microglial cell line and
primary rat microglial cells. Neurotox Res. 33:337–352. 2018.
View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Liddelow SA, Guttenplan KA, Clarke LE,
Bennett FC, Bohlen CJ, Schirmer L, Bennett ML, Münch AE, Chung WS,
Peterson TC, et al: Neurotoxic reactive astrocytes are induced by
activated microglia. Nature. 541:481–487. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Dong Y and Benveniste EN: Immune function
of astrocytes. Glia. 36:180–190. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Wang X, Yang L, Yang L, Xing F, Yang H,
Qin L, Lan Y, Wu H, Zhang B, Shi H, et al: Gypenoside IX suppresses
p38 MAPK/Akt/NFκB signaling pathway activation and inflammatory
responses in astrocytes stimulated by proinflammatory mediators.
Inflammation. 40:2137–2150. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Zhang J, Benveniste H, Klitzman B and
Piantadosi CA: Nitric oxide synthase inhibition and extracellular
glutamate concentration after cerebral ischemia/reperfusion.
Stroke. 26:298–304. 1995. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Law A, Gauthier S and Quirion R: Say NO to
Alzheimer's disease: The putative links between nitric oxide and
dementia of the Alzheimer's type. Brain Res Brain Res Rev.
35:73–96. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Block ML, Zecca L and Hong JS:
Microglia-mediated neurotoxicity: Uncovering the molecular
mechanisms. Nat Rev Neurosci. 8:57–69. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Jung WK, Lee DY, Park C, Choi YH, Choi I,
Park SG, Seo SK, Lee SW, Yea SS, Ahn SC, et al: Cilostazol is
anti-inflammatory in BV2 microglial cells by inactivating nuclear
factor-kappaB and inhibiting mitogen-activated protein kinases. Br
J Pharmacol. 159:1274–1285. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Wilms H, Sievers J, Rickert U,
Rostami-Yazdi M, Mrowietz U and Lucius R: Dimethylfumarate inhibits
microglial and astrocytic inflammation by suppressing the synthesis
of nitric oxide, IL-1beta, TNF-alpha and IL-6 in an in-vitro model
of brain inflammation. J Neuroinflammation. 7:302010. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Liu D, Wang Z, Liu S, Wang F, Zhao S and
Hao A: Anti-inflammatory effects of fluoxetine in
lipopolysaccharide (LPS)-stimulated microglial cells.
Neuropharmacology. 61:592–599. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Boche D, Perry VH and Nicoll JA: Review:
Activation patterns of microglia and their identification in the
human brain. Neuropathol Appl Neurobiol. 39:3–18. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Recio MC, Andujar I and Rios JL:
Anti-inflammatory agents from plants: Progress and potential. Curr
Med Chem. 19:2088–2103. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Piotrowska H, Kucinska M and Murias M:
Biological activity of piceatannol: Leaving the shadow of
resveratrol. Mutat Res. 750:60–82. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Lee H, Kim YO, Kim H, Kim SY, Noh HS, Kang
SS, Cho GJ, Choi WS and Suk K: Flavonoid wogonin from medicinal
herb is neuroprotective by inhibiting inflammatory activation of
microglia. FASEB J. 17:1943–1944. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Li FQ, Wang T, Pei Z, Liu B and Hong JS:
Inhibition of microglial activation by the herbal flavonoid
baicalein attenuates inflammation-mediated degeneration of
dopaminergic neurons. J Neural Transm (Vienna). 112:331–347. 2005.
View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Suk K, Lee H, Kang SS, Cho GJ and Choi WS:
Flavonoid baicalein attenuates activation-induced cell death of
brain microglia. J Pharmacol Exp Ther. 305:638–645. 2003.
View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Park JS, Park EM, Kim DH, Jung K, Jung JS,
Lee EJ, Hyun JW, Kang JL and Kim HS: Anti-inflammatory mechanism of
ginseng saponins in activated microglia. J Neuroimmunol. 209:40–49.
2009. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
He LF, Chen HJ, Qian LH, Chen GY and Buzby
JS: Curcumin protects pre-oligodendrocytes from activated microglia
in vitro and in vivo. Brain Res. 1339:60–69. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Li R, Huang YG, Fang D and Le WD:
(−)-Epigallocatechin gallate inhibits lipopolysaccharide-induced
microglial activation and protects against inflammation-mediated
dopaminergic neuronal injury. J Neurosci Res. 78:723–731. 2004.
View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Choi DK, Koppula S and Suk K: Inhibitors
of microglial neurotoxicity: Focus on natural products. Molecules.
16:1021–1043. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Shen H, Wang H, Wang L, Wang L, Zhu M,
Ming Y, Zhao S, Fan J and Lai EY: Ethanol extract of root of Prunus
persica inhibited the growth of liver cancer cell HepG2 by inducing
cell cycle arrest and migration suppression. Evid Based Complement
Alternat Med. 2017:82319362017. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Rho JR, Jun CS, Ha Y, Yoo MJ, Cui MX, Baek
HS, Lim JA, Lee YH and Chai KY: Isolation and characterization of a
new alkaloid from the seed of Prunus persica L. and its
anti-inflammatory activity. Bull Korean Chem Soc. 28:1289–1293.
2007. View Article : Google Scholar
|
|
29
|
Lee JY and An BJ: Anti-oxidant and
anti-inflammation activities of prunus persica flos. J Appl Biol
Chem. 53:162–169. 2010. View Article : Google Scholar
|
|
30
|
Kwak CS, Yang J, Shin CY and Chung JH:
Topical or oral treatment of peach flower extract attenuates
UV-induced epidermal thickening, matrix metalloproteinase-13
expression and pro-inflammatory cytokine production in hairless
mice skin. Nutr Res Pract. 12:29–40. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Shin TY, Park SB, Yoo JS, Kim IK, Lee HS,
Kwon TK, Kim MK, Kim JC and Kim SH: Anti-allergic inflammatory
activity of the fruit of prunus persica: Role of calcium and
NF-kappaB. Food Chem Toxicol. 48:2797–2802. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Benmehdi H, Fellah K, Amrouche A, Memmou
F, Malainine H, Dalile H and Siata W: Phytochemical study,
antioxidant activity and kinetic behaviour of flavonoids fractions
isolated from prunus persica L. Leaves. Asian J Chem. 29:132017.
View Article : Google Scholar
|
|
33
|
Deb L, Gupta R, Dutta A, Yadav A, Bhowmik
D and Kumar KS: Evaluation of antioxidant activity of aqueous
fraction of Prunus persica L. aqueous extract. Der Pharmacia
Sinica. 1:157–164. 2010.
|
|
34
|
Prakash V, Rana S and Sagar A: Studies on
analysis of antibacterial and antioxidant activity of Prunus
persica (L.) Batsch. Int J Sci Nat. 8:54–58. 2017.
|
|
35
|
Zhao X, Zhang W, Yin X, Su M, Sun C, Li X
and Chen K: Phenolic composition and antioxidant properties of
different peach [Prunus persica (L.) Batsch] cultivars in China.
Int J Mol Sci. 16:5762–5778. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
McCarthy KD and de Vellis J: Preparation
of separate astroglial and oligodendroglial cell cultures from rat
cerebral tissue. J Cell Biol. 85:890–902. 1980. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
NIH, . Laboratory animal welfare. Special
Edition of the NIH Guide for Grants and Contracts. NIH; Bethesda,
MD, USA: pp. 85–23. 1985
|
|
38
|
Wrobel K, Claudio E, Segade F, Ramos S and
Lazo PS: Measurement of cytotoxicity by propidium iodide staining
of target cell DNA: Application to the quantification of murine
TNF-alpha. J Immunol Methods. 189:243–249. 1996. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Baldwin AS Jr: The NF-kappaB and I kappaB
proteins: New discoveries and insights. Annu Rev Immunol.
14:649–683. 1996. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Mankan AK, Lawless MW, Gray SG, Kelleher D
and McManus R: NF- kappaB regulation: The nuclear response. J Cell
Mol Med. 13:631–643. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Oh YT, Lee JY, Lee J, Kim H, Yoon KS, Choe
W and Kang I: Oleic acid reduces lipopolysaccharide-induced
expression of iNOS and COX-2 in BV2 murine microglial cells:
Possible involvement of reactive oxygen species, p38 MAPK, and
IKK/NF-kappaB signaling pathways. Neurosci Lett. 464:93–97. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Mander P and Brown GC: Nitric oxide,
hypoxia and brain inflammation. Biochem Soc Trans. 32:1068–1069.
2004. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Pautz A, Art J, Hahn S, Nowag S, Voss C
and Kleinert H: Regulation of the expression of inducible nitric
oxide synthase. Nitric Oxide. 23:75–93. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Bishop Bailey D, Calatayud S, Warner TD,
Hla T and Mitchell JA: Prostaglandins and the regulation of tumor
growth. J Environ Pathol Toxicol Oncol. 21:93–101. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Singh AK and Jiang Y: How does peripheral
lipopolysaccharide induce gene expression in the brain of rats?
Toxicology. 201:197–207. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Banks WA and Robinson SM: Minimal
penetration of lipopolysaccharide across the murine blood-brain
barrier. Brain Behav Immun. 24:102–109. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Banks WA, Gray AM, Erickson MA, Salameh
TS, Damodarasamy M, Sheibani N, Meabon JS, Wing EE, Morofuji Y,
Cook DG and Reed MJ: Lipopolysaccharide-induced blood-brain barrier
disruption: Roles of cyclooxygenase, oxidative stress,
neuroinflammation, and elements of the neurovascular unit. J
Neuroinflammation. 12:2232015. View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Tarassishin L, Suh HS and Lee SC: LPS and
IL-1 differentially activate mouse and human astrocytes: Role of
CD14. Glia. 62:999–1013. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Panicker N, Saminathan H, Jin H, Neal M,
Harischandra DS, Gordon R, Kanthasamy K, Lawana V, Sarkar S, Luo J,
et al: Fyn kinase regulates microglial neuroinflammatory responses
in cell culture and animal models of Parkinson's disease. J
Neurosci. 35:10058–10077. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Kim YJ, Hwang SY and Han IO: Insoluble
matrix components of glioma cells suppress LPS-mediated iNOS/NO
induction in microglia. Biochem Biophys Res Commun. 347:731–738.
2006. View Article : Google Scholar : PubMed/NCBI
|
|
51
|
Bhatt D and Ghosh S: Regulation of the
NF-κB-mediated transcription of inflammatory genes. Front Immunol.
5:712014. View Article : Google Scholar : PubMed/NCBI
|
|
52
|
Lawrence T: The nuclear factor NF-kappaB
pathway in inflammation. Cold Spring Harb Perspect Biol.
1:a0016512009. View Article : Google Scholar : PubMed/NCBI
|
|
53
|
Lu YC, Yeh WC and Ohashi PS: LPS/TLR4
signal transduction pathway. Cytokine. 42:145–151. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
54
|
Korcheva V, Wong J, Corless C, Iordanov M
and Magun B: Administration of ricin induces a severe inflammatory
response via nonredundant stimulation of ERK, JNK, and P38 MAPK and
provides a mouse model of hemolytic uremic syndrome. Am J Pathol.
166:323–339. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
55
|
Da Silva J, Pierrat B, Mary JL and
Lesslauer W: Blockade of p38 mitogen-activated protein kinase
pathway inhibits inducible nitric-oxide synthase expression in
mouse astrocytes. J Biol Chem. 272:28373–28380. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
56
|
Xia Q, Hu Q, Wang H, Yang H, Gao F, Ren H,
Chen D, Fu C, Zheng L, Zhen X, et al: Induction of COX-2-PGE2
synthesis by activation of the MAPK/ERK pathway contributes to
neuronal death triggered by TDP-43-depleted microglia. Cell Death
Dis. 6:e17022015. View Article : Google Scholar : PubMed/NCBI
|
|
57
|
Bhat NR, Zhang P, Lee JC and Hogan EL:
Extracellular signal-regulated kinase and p38 subgroups of
mitogen-activated protein kinases regulate inducible nitric oxide
synthase and tumor necrosis factor-α gene expression in
endotoxin-stimulated primary glial cultures. J Neurosci.
18:1633–1641. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
58
|
Abidi W, Jiménez S, Moreno MÁ and
Gogorcena Y: Evaluation of antioxidant compounds and total sugar
content in a nectarine [Prunus persica (L.) Batsch] progeny. Int J
Mol Sci. 12:6919–6935. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
59
|
Gasparotto J, Somensi N, Bortolin RC,
Moresco KS, Girardi CS, Klafke K, Rabelo TK, Morrone Mda S,
Vizzotto M and Raseira Mdo C: Effects of different products of
peach (Prunus persica L. Batsch) from a variety developed in
southern Brazil on oxidative stress and inflammatory parameters in
vitro and ex vivo. J Clin Biochem Nutr. 55:110–119. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
60
|
Lee JY and An BJ: Antioxidant and
anti-inflammatory effects of fractions from Pruni persicae Flos.
Korea J Herbol. 27:55–63. 2012. View Article : Google Scholar
|
|
61
|
Mei X, Zhou L, Zhang T, Lu B, Sheng Y and
Ji L: Chlorogenic acid attenuates diabetic retinopathy by reducing
VEGF expression and inhibiting VEGF-mediated retinal
neoangiogenesis. Vascul Pharmacol. 101:29–37. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
62
|
Kim M, Choi SY, Lee P and Hur J:
Neochlorogenic acid inhibits lipopolysaccharide-induced activation
and pro-inflammatory responses in BV2 microglial cells. Neurochem
Res. 40:1792–1798. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
63
|
Li N, Wang Y, Li X, Zhang H, Zhou D, Wang
W, Li W, Zhang X, Li X, Hou Y and Meng D: Bioactive phenols as
potential neuroinflammation inhibitors from the leaves of
Xanthoceras sorbifolia Bunge. Bioorg Med Chem Lett. 26:5018–5023.
2016. View Article : Google Scholar : PubMed/NCBI
|
|
64
|
Kim CY, Lee C, Park GH and Jang JH:
Neuroprotective effect of epigallocatechin-3-gallate against
β-amyloid-induced oxidative and nitrosative cell death via
augmentation of antioxidant defense capacity. Arch Pharm Res.
32:869–881. 2009. View Article : Google Scholar : PubMed/NCBI
|