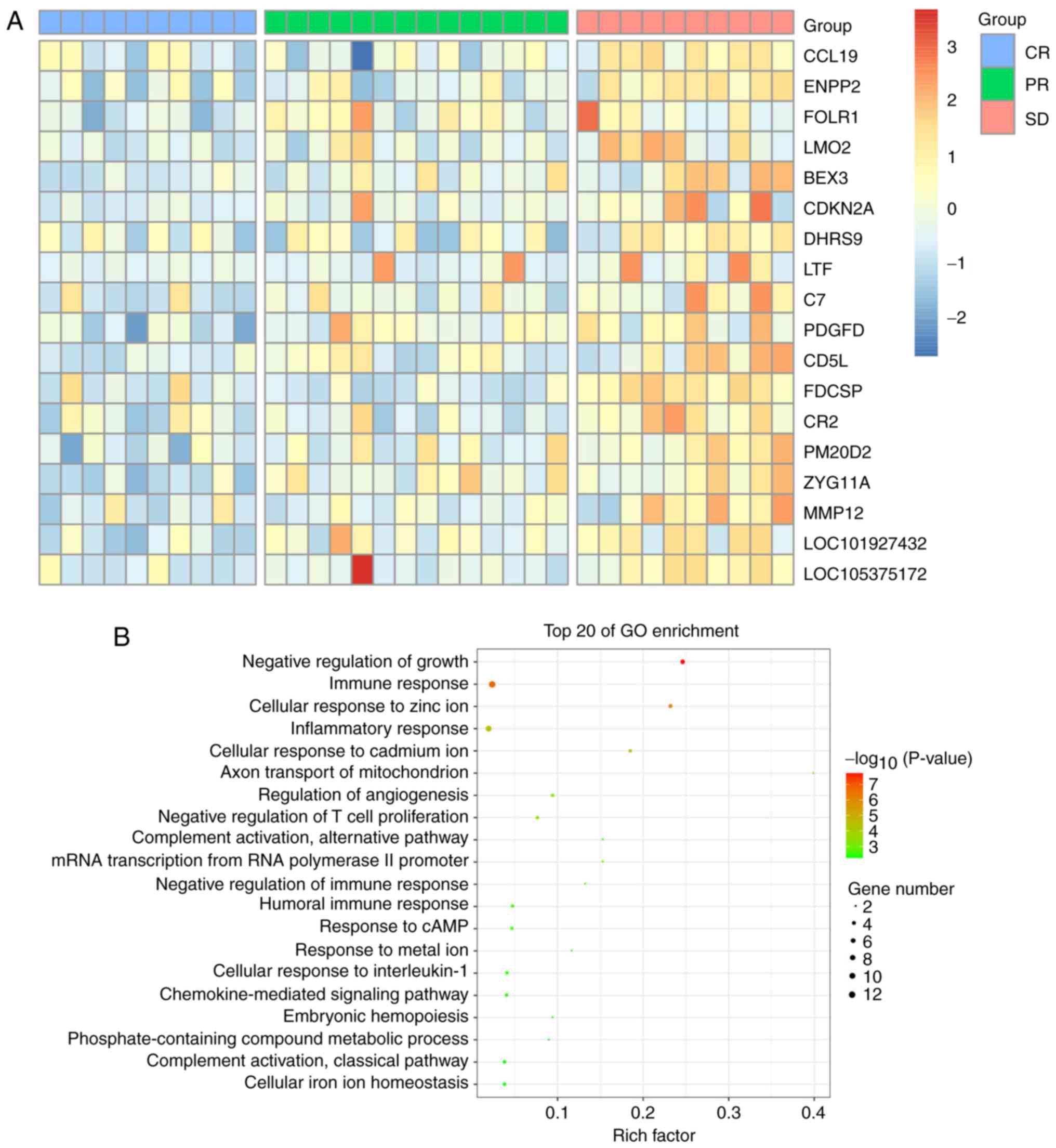
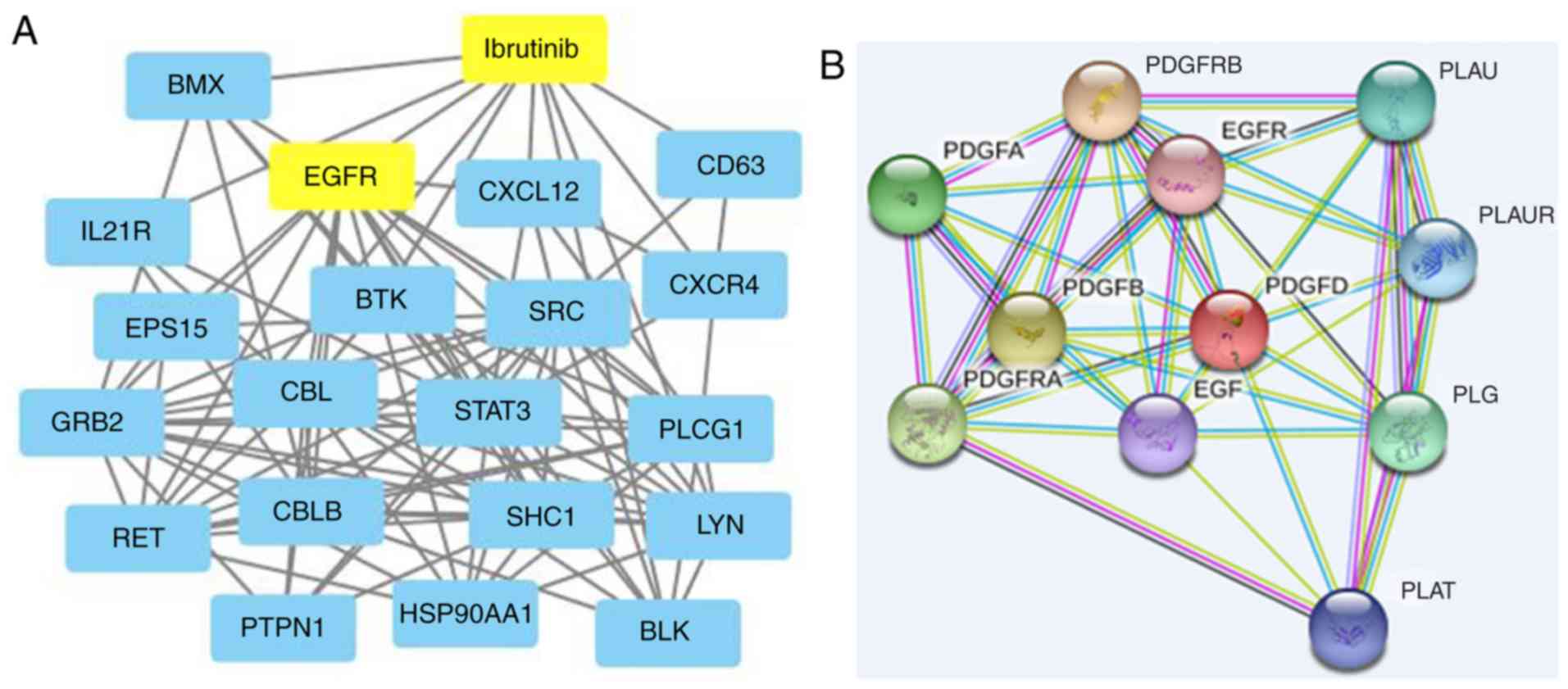
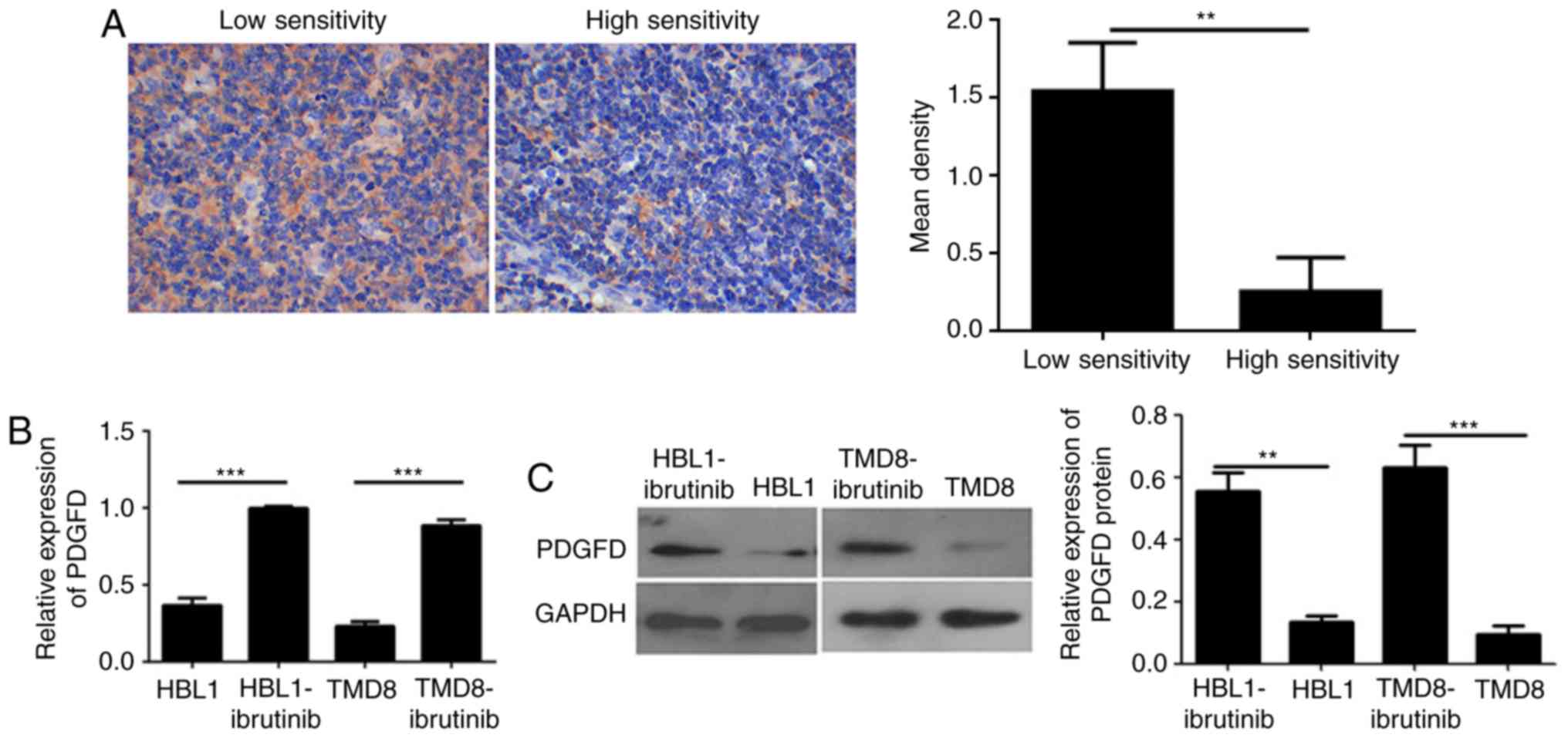
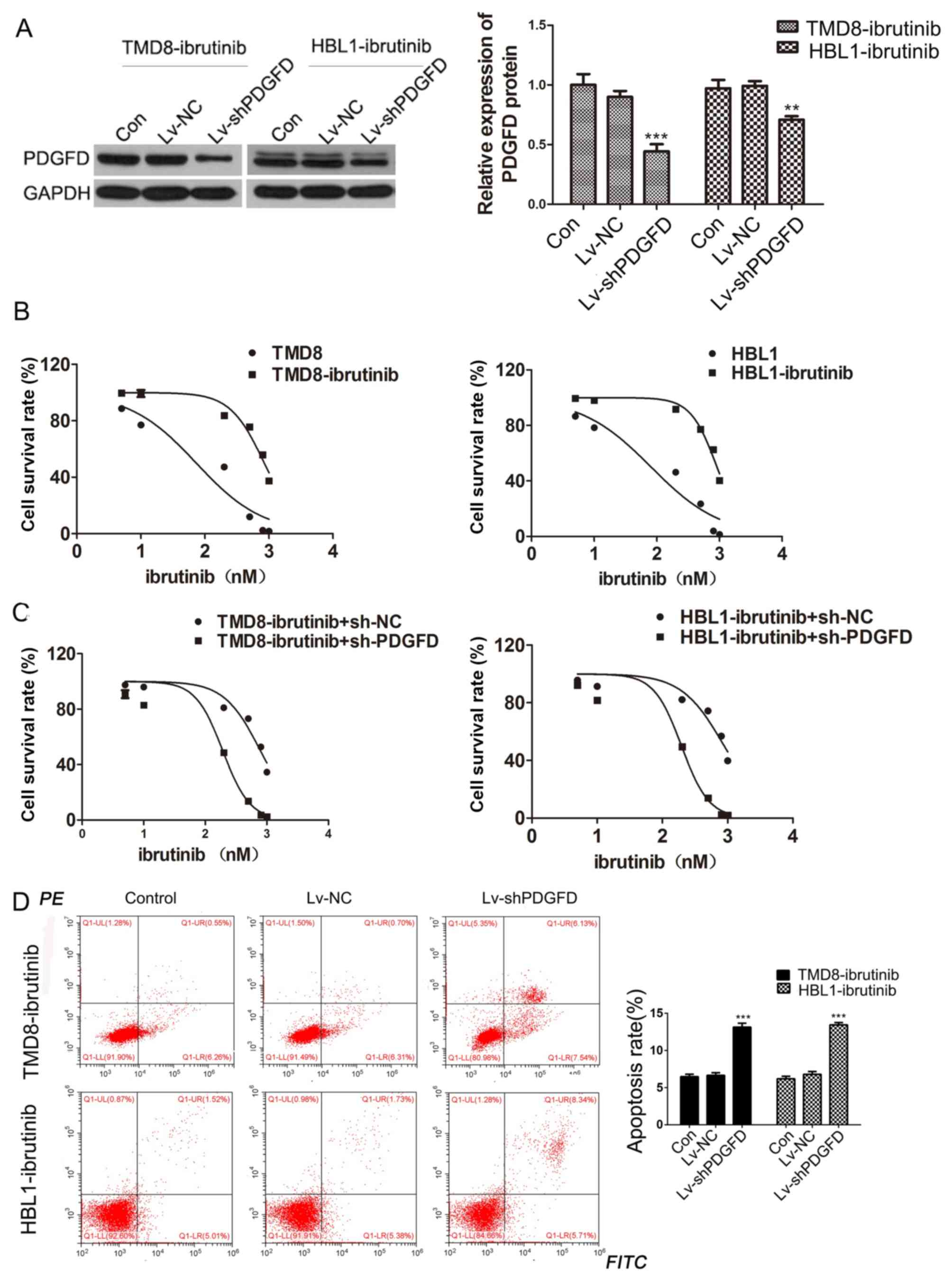
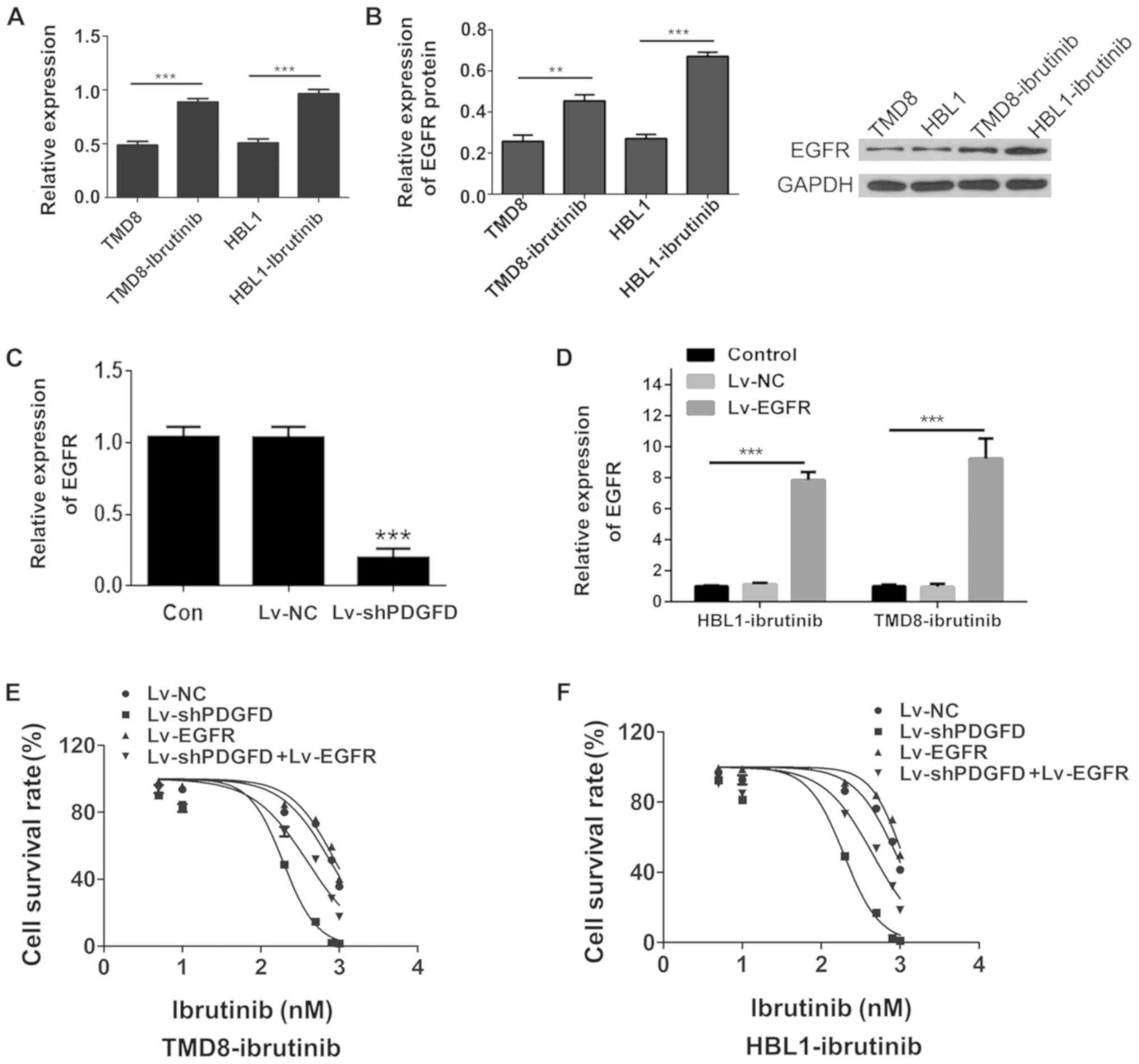
|
1
|
Sun J, Yang Q, Lu Z, He M, Gao L, Zhu M,
Sun L, Wei L, Li M, Liu C, et al: Distribution of lymphoid
neoplasms in China: Analysis of 4,638 cases according to the world
health organization classification. Am J Clin Pathol. 138:429–434.
2012. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Lossos IS and Morgensztern D: Prognostic
biomarkers in diffuse large B-cell lymphoma. J Clin Oncol.
24:995–1007. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Kubuschok B, Held G and Pfreundschuh M:
Management of diffuse large B-cell lymphoma (DLBCL). Cancer Treat
Res. 165:271–288. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Herman SE, Gordon AL, Hertlein E,
Ramanunni A, Zhang X, Jaglowski S, Flynn J, Jones J, Blum KA, Buggy
JJ, et al: Bruton tyrosine kinase represents a promising
therapeutic target for treatment of chronic lymphocytic leukemia
and is effectively targeted by PCI-32765. Blood. 117:6287–6296.
2011. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Tsukada S, Saffran DC, Rawlings DJ,
Parolini O, Allen RC, Klisak I, Sparkes RS, Kubagawa H, Mohandas T,
Quan S, et al: Deficient expression of a B cell cytoplasmic
tyrosine kinase in human X-linked agammaglobulinemia. 1993. J
Immunol. 188:2936–2947. 2012.PubMed/NCBI
|
|
6
|
Herman SE, Mustafa RZ, Gyamfi JA,
Pittaluga S, Chang S, Chang B, Farooqui M and Wiestner A: Ibrutinib
inhibits BCR and NF-kB signaling and reduces tumor proliferation in
tissue-resident cells of patients with CLL. Blood. 123:3286–3295.
2014. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Hendriks RW, Yuvaraj S and Kil LP:
Targeting Bruton's tyrosine kinase in B cell malignancies. Nat Rev
Cancer. 14:219–232. 2014. View
Article : Google Scholar : PubMed/NCBI
|
|
8
|
Wang ML, Rule S, Martin P, Goy A, Auer R,
Kahl BS, Jurczak W, Advani RH, Romaguera JE, Williams ME, et al:
Targeting BTK with ibrutinib in relapsed or refractory mantle-cell
lymphoma. N Engl J Med. 369:507–516. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Charalambous A, Schwarzbich MA and
Witzens-Harig M: Ibrutinib. Recent Results Cancer Res. 212:133–168.
2018. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Zhang SQ, Smith SM, Zhang SY and Lynn Wang
Y: Mechanisms of ibrutinib resistance in chronic lymphocytic
leukaemia and non-Hodgkin lymphoma. Br J Haematol. 170:445–456.
2015. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Kong D, Banerjee S, Huang W, Li Y, Wang Z,
Kim HR and Sarkar FH: Mammalian target of rapamycin repression by
3,3′-diindolylmethane inhibits invasion and angiogenesis in
platelet-derived growth factor-D-overexpressing PC3 cells. Cancer
Res. 68:1927–1934. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Wang Z, Kong D, Banerjee S, Li Y, Adsay
NV, Abbruzzese J and Sarkar FH: Down-Regulation of platelet-derived
growth factor-D inhibits cell growth and angiogenesis through
inactivation of notch-1 and nuclear factor-kappaB signaling. Cancer
Res. 67:11377–11385. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Lokker NA, Sullivan CM, Hollenbach SJ,
Israel MA and Giese NA: Platelet-derived growth factor (PDGF)
autocrine signaling regulates survival and mitogenic pathways in
glioblastoma cells: Evidence that the novel PDGF-C and PDGF-D
ligands may play a role in the development of brain tumors. Cancer
Res. 62:3729–3735. 2002.PubMed/NCBI
|
|
14
|
Ustach CV, Taube ME, Hurst NJ Jr, Bhagat
S, Bonfil RD, Cher ML, Schuger L and Kim HR: A potential oncogenic
activity of platelet-derived growth factor D in prostate cancer
progression. Cancer Res. 64:1722–1729. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Ustach CV and Kim HR: Platelet-derived
growth factor D is activated by urokinase plasminogen activator in
prostate carcinoma cells. Mol Cell Biol. 25:6279–6288. 2005.
View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Xu L, Tong R, Cochran DM and Jain RK:
Blocking platelet-derived growth factor-D/platelet-derived growth
factor receptor beta signaling inhibits human renal cell carcinoma
progression in an orthotopic mouse model. Cancer Res. 65:5711–5719.
2005. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Wang Z, Ahmad A, Li Y, Kong D, Azmi AS,
Banerjee S and Sarkar FH: Emerging roles of PDGF-D signaling
pathway in tumor development and progression. Biochim Biophys Acta.
1806:122–130. 2010.PubMed/NCBI
|
|
18
|
Yoshida T, Zhang G and Haura EB: Targeting
epidermal growth factor receptor: Central signaling kinase in lung
cancer. Biochem Pharmacol. 80:613–623. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Cohen RB: Epidermal growth factor receptor
as a therapeutic target in colorectal cancer. Clin Colorectal
Cancer. 2:246–251. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Hrustanovic G, Lee BJ and Bivona TG:
Mechanisms of resistance to EGFR targeted therapies. Cancer Biol
Ther. 14:304–314. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Jänne PA, Engelman JA and Johnson BE:
Epidermal growth factor receptor mutations in non-small-cell lung
cancer: Implications for treatment and tumor biology. J Clin Oncol.
23:3227–3234. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Martinez-Marti A, Navarro A and Felip E:
Epidermal growth factor receptor first generation tyrosine-kinase
inhibitors. Transl Lung Cancer Res. 8 (Suppl 3):S235–S246. 2019.
View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Elbaz M, Nasser MW, Ravi J, Wani NA,
Ahirwar DK, Zhao H, Oghumu S, Satoskar AR, Shilo K, Carson WE 3rd
and Ganju RK: Modulation of the tumor microenvironment and
inhibition of EGF/EGFR pathway: Novel anti-tumor mechanisms of
cannabidiol in breast cancer. Mol Oncol. 9:906–919. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Arienti C, Pignatta S and Tesei A:
Epidermal growth factor receptor family and its role in gastric
cancer. Front Oncol. 9:13082019. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Maennling AE, Tur MK, Niebert M,
Klockenbring T, Zeppernick F, Gattenlöhner S, Meinhold-Heerlein I
and Hussain AF: Molecular targeting therapy against EGFR family in
breast cancer: Progress and future potentials. Cancers (Basel).
11:E18262019. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Fu Q, Cheng J, Zhang J, Zhang Y, Chen X,
Luo S and Xie J: MiR-20b reduces 5-FU resistance by suppressing the
ADAM9/EGFR signaling pathway in colon cancer. Oncol Rep.
37:123–130. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Wu H, Wang A, Zhang W, Wang B, Chen C,
Wang W, Hu C, Ye Z, Zhao Z, Wang L, et al: Ibrutinib selectively
and irreversibly targets EGFR (L858R, Del19) mutant but is
moderately resistant to EGFR (T790M) mutant NSCLC cells.
Oncotarget. 6:31313–31322. 2015.PubMed/NCBI
|
|
28
|
Wang A, Yan XE, Wu H, Wang W, Hu C, Chen
C, Zhao Z, Zhao P, Li X, Wang L, et al: Ibrutinib targets
mutant-EGFR kinase with a distinct binding conformation.
Oncotarget. 7:69760–69769. 2016.PubMed/NCBI
|
|
29
|
Chen J, Kinoshita T, Sukbuntherng J, Chang
BY and Elias L: Ibrutinib inhibits ERBB receptor tyrosine kinases
and HER2-amplified breast cancer cell growth. Mol Cancer Ther.
15:2835–2844. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Saito Y, Haendeler J, Hojo Y, Yamamoto K
and Berk BC: Receptor heterodimerization: Essential mechanism for
platelet-derived growth factor-induced epidermal growth factor
receptor transactivation. Mol Cell Biol. 21:6387–6394. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Kuo HP, Ezell SA, Schweighofer KJ, Cheung
LWK, Hsieh S, Apatira M, Sirisawad M, Eckert K, Hsu SJ, Chen CT, et
al: Combination of ibrutinib and ABT-199 in diffuse large B-cell
lymphoma and follicular lymphoma. Mol Cancer Ther. 16:1246–1256.
2017. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Kanehisa M: Toward understanding the
origin and evolution of cellular organisms. Protein Sci.
28:1947–1951. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Kanehisa M and Goto S: KEGG: Kyoto
encyclopedia of genes and genomes. Nucleic Acids Res. 28:27–30.
2000. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Kanehisa M, Sato Y, Furumichi M, Morishima
K and Tanabe M: New approach for understanding genome variations in
KEGG. Nucleic Acids Res. 47:D590–D595. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Ashburner M, Ball CA, Blake JA, Botstein
D, Butler H, Cherry JM, Davis AP, Dolinski K, Dwight SS, Eppig JT,
et al: Gene ontology: Tool for the unification of biology. The gene
ontology consortium. Nat Genet. 25:25–29. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
The Gene Ontology Consortium, . The gene
ontology resource: 20 Years and still GOing strong. Nucleic Acids
Res. 47:D330–D338. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Szklarczyk D, Santos A, von Mering C,
Jensen LJ, Bork P and Kuhn M: STITCH 5: Augmenting protein-chemical
interaction networks with tissue and affinity data. Nucleic Acids
Res. 44:D380–D384. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Huang da W, Sherman BT and Lempicki RA:
Systematic and integrative analysis of large gene lists using DAVID
bioinformatics resources. Nat Protoc. 4:44–57. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Szklarczyk D, Gable AL, Lyon D, Junge A,
Wyder S, Huerta-Cepas J, Simonovic M, Doncheva NT, Morris JH, Bork
P, et al: STRING v11: Protein-protein association networks with
increased coverage, supporting functional discovery in genome-wide
experimental datasets. Nucleic Acids Res. 47:D607–D613. 2019.
View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) method. Methods. 25:402–408. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Wu ZH, Tao ZH, Zhang J, Li T, Ni C, Xie J,
Zhang JF and Hu XC: MiRNA-21 induces epithelial to mesenchymal
transition and gemcitabine resistance via the PTEN/AKT pathway in
breast cancer. Tumour Biol. 37:7245–7254. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Sukswai N, Lyapichev K, Khoury JD and
Medeiros LJ: Diffuse large B-cell lymphoma variants: An update.
Pathology. 52:53–67. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Camicia R, Winkler HC and Hassa PO: Novel
drug targets for personalized precision medicine in
relapsed/refractory diffuse large B-cell lymphoma: A comprehensive
review. Mol Cancer. 14:2072015. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Walewski J: Aggressive B-cell lymphoma:
Chasing the target. J Investig Med. 68:331–334. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Cabanillas F and Shah B: Advances in
diagnosis and management of diffuse large B-cell lymphoma. Clin
Lymphoma Myeloma Leuk. 17:783–796. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Maddocks K, Christian B, Jaglowski S,
Flynn J, Jones JA, Porcu P, Wei L, Jenkins C, Lozanski G, Byrd JC
and Blum KA: A phase 1/1b study of rituximab, bendamustine, and
ibrutinib in patients with untreated and relapsed/refractory
non-Hodgkin lymphoma. Blood. 125:242–248. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Sagiv-Barfi I, Kohrt HE, Czerwinski DK, Ng
PP, Chang BY and Levy R: Therapeutic antitumor immunity by
checkpoint blockade is enhanced by ibrutinib, an inhibitor of both
BTK and ITK. Proc Natl Acad Sci USA. 112:E966–E972. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Grassilli E, Pisano F, Cialdella A, Bonomo
S, Missaglia C, Cerrito MG, Masiero L, Ianzano L, Giordano F,
Cicirelli V, et al: A novel oncogenic BTK isoform is overexpressed
in colon cancers and required for RAS-mediated transformation.
Oncogene. 35:4368–4378. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Wu L, Zhang YZ, Xia B, Li XW, Yuan T, Tian
C, Zhao HF, Yu Y and Sotomayor E: Ibrutinib inhibits mesenchymal
stem cells-mediated drug resistance in diffuse large B-cell
lymphoma. Zhonghua Xue Ye Xue Za Zhi. 38:1036–1042. 2017.(In
Chinese; Abstract available in Chinese from the publisher).
PubMed/NCBI
|
|
50
|
Erdmann T and Lenz G: Approaching
resistance to ibrutinib in diffuse large B-cell lymphoma. Leuk
Lymphoma. 57:1254–1255. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
51
|
Kim JH, Kim WS, Ryu K, Kim SJ and Park C:
CD79B limits response of diffuse large B cell lymphoma to
ibrutinib. Leuk Lymphoma. 57:1413–1422. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
52
|
Chen JG, Liu X, Munshi M, Xu L, Tsakmaklis
N, Demos MG, Kofides A, Guerrera ML, Chan GG, Patterson CJ, et al:
BTK(Cys481Ser) drives ibrutinib resistance via ERK1/2 and protects
BTK(wild-type) MYD88-mutated cells by a paracrine mechanism. Blood.
131:2047–2059. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
53
|
Olsen RS, Dimberg J, Geffers R and
Wågsäter D: Possible role and therapeutic target of PDGF-D
signalling in colorectal cancer. Cancer Invest. 37:99–112. 2019.
View Article : Google Scholar : PubMed/NCBI
|
|
54
|
Bartoschek M and Pietras K: PDGF family
function and prognostic value in tumor biology. Biochem Biophys Res
Commun. 503:984–990. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
55
|
Wang JC, Li GY, Wang B, Han SX, Sun X,
Jiang YN, Shen YW, Zhou C, Feng J, Lu SY, et al: Metformin inhibits
metastatic breast cancer progression and improves chemosensitivity
by inducing vessel normalization via PDGF-B downregulation. J Exp
Clin Cancer Res. 38:2352019. View Article : Google Scholar : PubMed/NCBI
|
|
56
|
Wang Y, Appiah-Kubi K, Wu M, Yao X, Qian
H, Wu Y and Chen Y: The platelet-derived growth factors (PDGFs) and
their receptors (PDGFRs) are major players in oncogenesis, drug
resistance, and attractive oncologic targets in cancer. Growth
Factors. 34:64–71. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
57
|
Zhang M, Liu T, Xia B, Yang C, Hou S, Xie
W and Lou G: Platelet-derived growth factor D is a prognostic
biomarker and is associated with platinum resistance in epithelial
ovarian cancer. Int J Gynecol Cancer. 28:323–331. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
58
|
Wang R, Li Y, Hou Y, Yang Q, Chen S, Wang
X, Wang Z, Yang Y, Chen C, Wang Z and Wu Q: The
PDGF-D/miR-106a/twist1 pathway orchestrates epithelial-mesenchymal
transition in gemcitabine resistance hepatoma cells. Oncotarget.
6:7000–7010. 2015.PubMed/NCBI
|
|
59
|
Nogi H, Kobayashi T, Suzuki M, Tabei I,
Kawase K, Toriumi Y, Fukushima H and Uchida K: EGFR as paradoxical
predictor of chemosensitivity and outcome among triple-negative
breast cancer. Oncol Rep. 21:413–417. 2009.PubMed/NCBI
|
|
60
|
Nielsen TO, Hsu FD, Jensen K, Cheang M,
Karaca G, Hu Z, Hernandez-Boussard T, Livasy C, Cowan D, Dressler
L, et al: Immunohistochemical and clinical characterization of the
basal-like subtype of invasive breast carcinoma. Clin Cancer Res.
10:5367–5374. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
61
|
Yue C, Niu M, Shan QQ, Zhou T, Tu Y, Xie
P, Hua L, Yu R and Liu X: High expression of bruton's tyrosine
kinase (BTK) is required for EGFR-induced NF-kB activation and
predicts poor prognosis in human glioma. J Exp Clin Cancer Res.
36:1322017. View Article : Google Scholar : PubMed/NCBI
|
|
62
|
Tanaka K, Babic I, Nathanson D, Akhavan D,
Guo D, Gini B, Dang J, Zhu S, Yang H, De Jesus J, et al: Oncogenic
EGFR signaling activates an mTORC2-NF-kB pathway that promotes
chemotherapy resistance. Cancer Discov. 1:524–538. 2011. View Article : Google Scholar : PubMed/NCBI
|