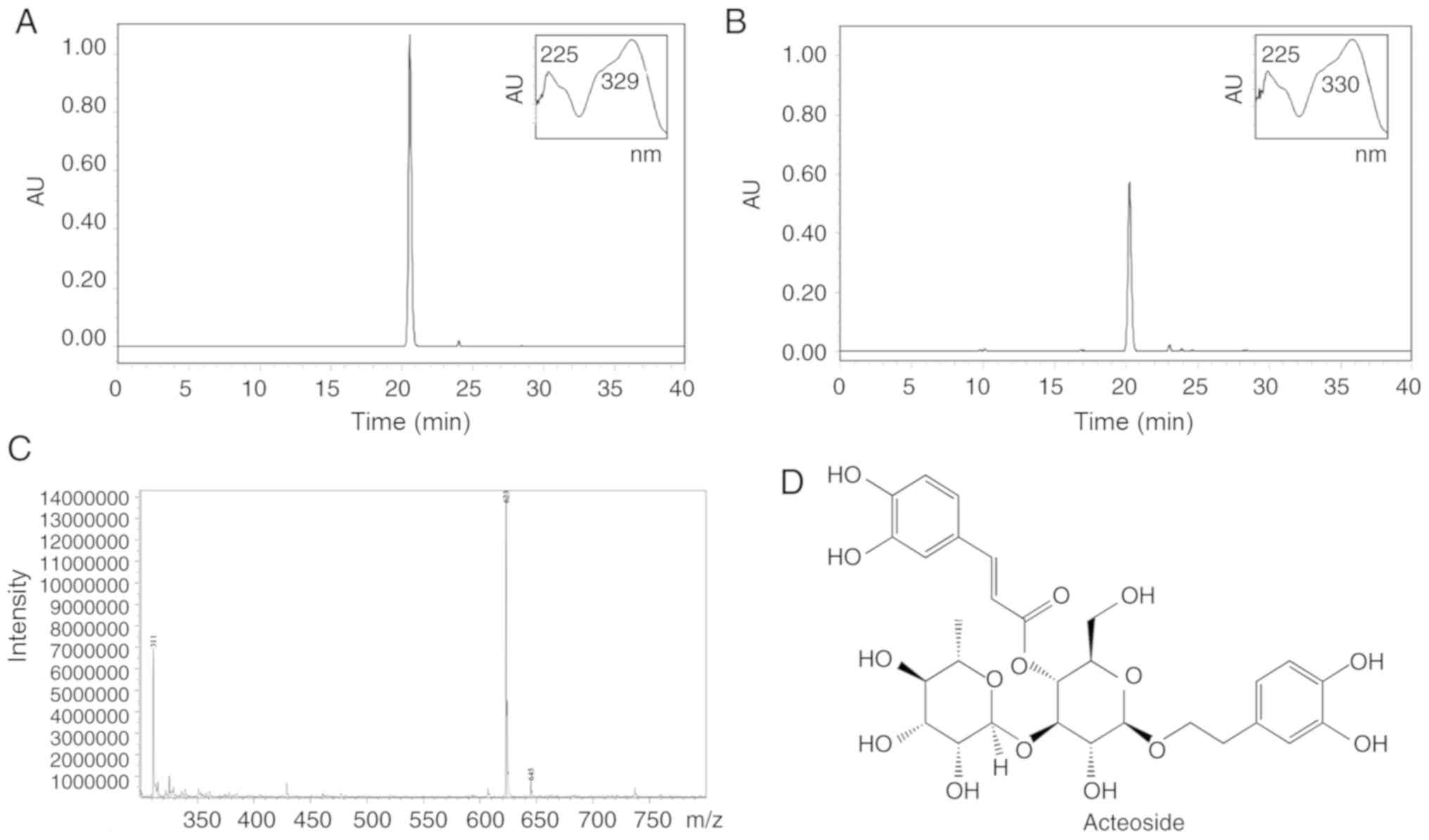
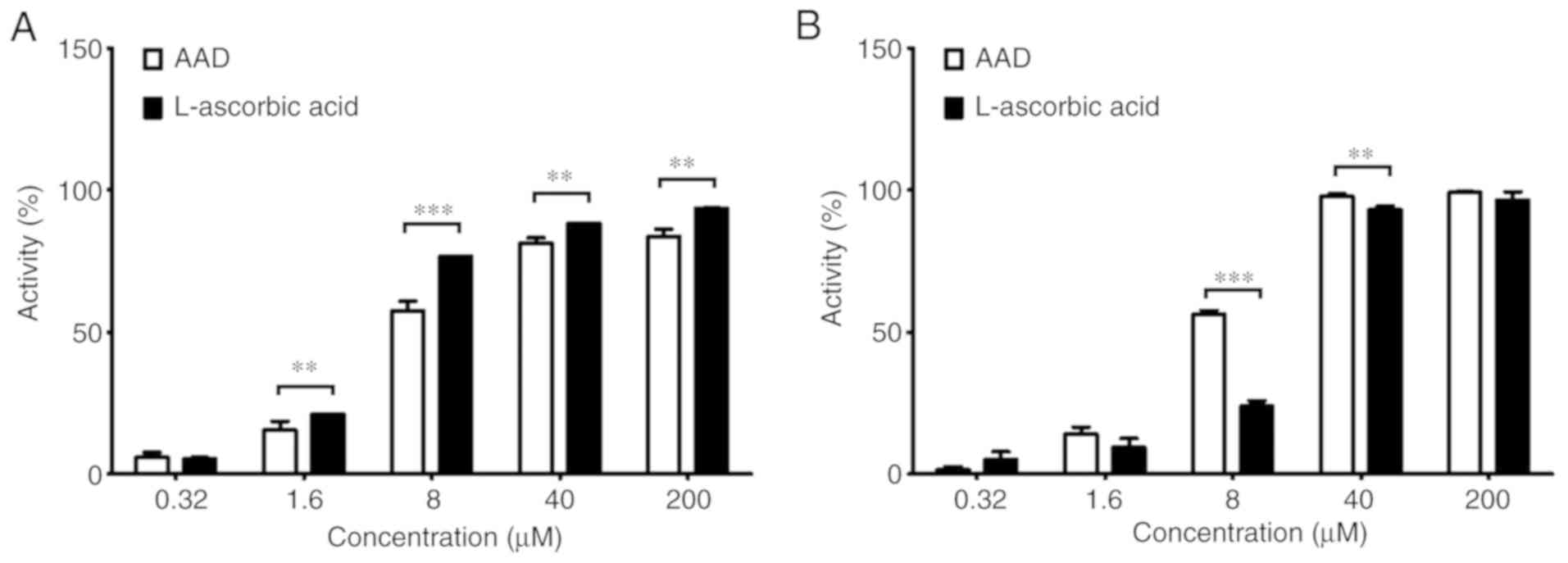
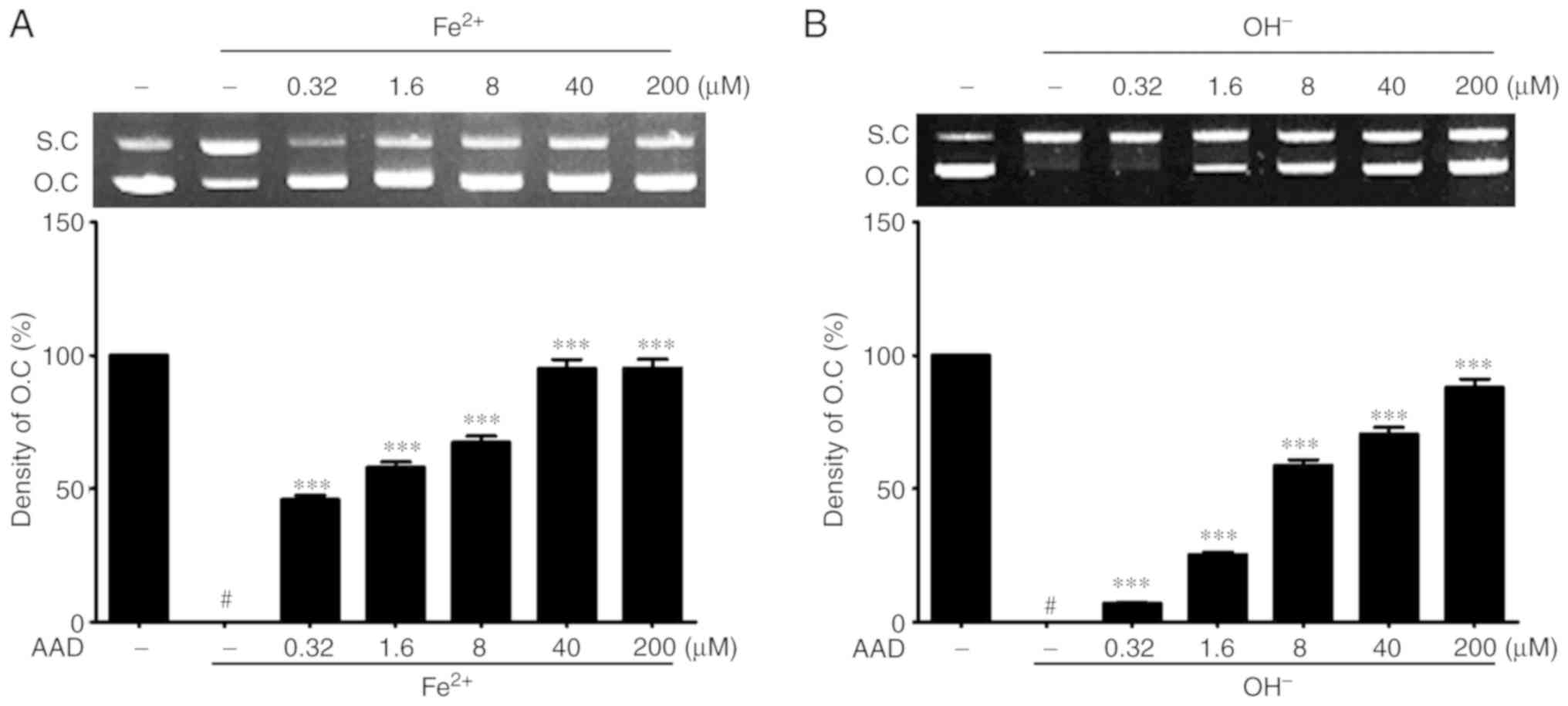
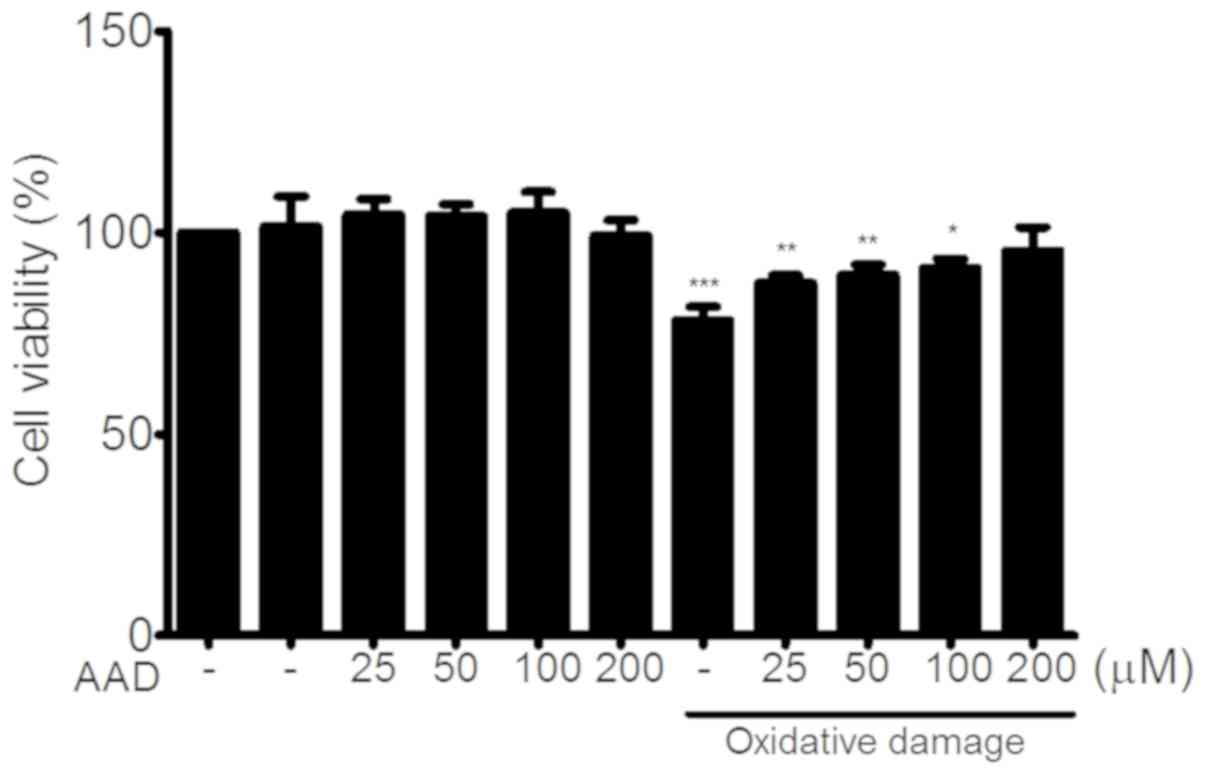
|
1
|
Akerele O: Nature's medicinal bounty:
Don't throw it away. World Health Forum. 14:390–395.
1993.PubMed/NCBI
|
|
2
|
Omale J and Okafor PN: Comparative
antioxidant capacity, membrane stabilization, polyphenol
composition and cytotoxicity of the leaf and stem of Cissus
multistriata. Afr J Biotechnol. 7:172008.
|
|
3
|
Hill AF: Economic Botany. McGraw-Hill.
(New York, NY). 5601952.
|
|
4
|
Edeoga HO, Okwu D and Mbaebie B:
Phytochemical constituents of some Nigerian medicinal plants. Afr J
Biotechnol. 4:685–688. 2005. View Article : Google Scholar
|
|
5
|
Valko M, Morris H and Cronin MT: Metals,
toxicity and oxidative stress. Curr Med Chem. 12:1161–1208. 2005.
View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Schärer OD: Chemistry and biology of DNA
repair. Angew Chem Int Ed Engl. 42:2946–2974. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Han HM, Kwon YS and Kim MJ: Antioxidant
and antiproliferative activity of extracts from water chestnut
(Trapa japonica Flerow). Hanguk Yakyong Changmul Hakhoe Chi.
24:14–20. 2016. View Article : Google Scholar
|
|
8
|
Roos WP, Thomas AD and Kaina B: DNA damage
and the balance between survival and death in cancer biology. Nat
Rev Cancer. 16:20–33. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Gilgun-Sherki Y, Rosenbaum Z, Melamed E
and Offen D: Antioxidant therapy in acute central nervous system
injury: Current state. Pharmacol Rev. 54:271–284. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Hamanaka RB and Chandel NS: Mitochondrial
reactive oxygen species regulate cellular signaling and dictate
biological outcomes. Trends Biochem Sci. 35:505–513. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Simon HU, Haj-Yehia A and Levi-Schaffer F:
Role of reactive oxygen species (ROS) in apoptosis induction.
Apoptosis. 5:415–418. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Fernandez-Capetillo O, Lee A, Nussenzweig
M and Nussenzweig A: H2AX: The histone guardian of the genome. DNA
Repair (Amst). 3:959–967. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Stucki M, Clapperton JA, Mohammad D, Yaffe
MB, Smerdon SJ and Jackson SP: MDC1 directly binds phosphorylated
histone H2AX to regulate cellular responses to DNA double-strand
breaks. Cell. 123:1213–1226. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Cook PJ, Ju BG, Telese F, Wang X, Glass CK
and Rosenfeld MG: Tyrosine dephosphorylation of H2AX modulates
apoptosis and survival decisions. Nature. 458:591–596. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Liu B, Chen Y and St Clair DK: ROS and
p53: A versatile partnership. Free Radic Biol Med. 44:1529–1535.
2008. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Polyak K, Xia Y, Zweier JL, Kinzler KW and
Vogelstein B: A model for p53-induced apoptosis. Nature.
389:300–305. 1997. View
Article : Google Scholar : PubMed/NCBI
|
|
17
|
Sablina AA, Budanov AV, Ilyinskaya GV,
Agapova LS, Kravchenko JE and Chumakov PM: The antioxidant function
of the p53 tumor suppressor. Nat Med. 11:1306–1313. 2005.
View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Shibata A and Jeggo PA: DNA double-strand
break repair in a cellular context. Clin Oncol (R Coll Radiol).
26:243–249. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Paull TT, Rogakou EP, Yamazaki V,
Kirchgessner CU, Gellert M and Bonner WM: A critical role for
histone H2AX in recruitment of repair factors to nuclear foci after
DNA damage. Curr Biol. 10:886–895. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Ji K, Jang NY and Kim YT: Isolation of
lactic acid bacteria showing antioxidative and probiotic activities
from kimchi and infant feces. J Microbiol Biotechnol. 25:1568–1577.
2015. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Nakai T: Genus novum Oleacearum in Corea
media inventum. Shokubutsugaku Zasshi. 33:153–154. 1919. View Article : Google Scholar
|
|
22
|
Oh H, Kang DG, Kwon TO, Jang KK, Chai KY,
Yun YG, Chung HT and Lee HS: Four glycosides from the leaves of
Abeliophyllum distichum with inhibitory effects on
angiotensin converting enzyme. Phytother Res. 17:811–813. 2003.
View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Xiong Q, Hase K, Tezuka Y, Tani T, Namba T
and Kadota S: Hepatoprotective activity of phenylethanoids from
Cistanche deserticola. Planta Med. 64:120–125. 1998.
View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Schlesier K, Harwat M, Böhm V and Bitsch
R: Assessment of antioxidant activity by using different in vitro
methods. Free Radic Res. 36:177–187. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Xie J: Effect of ethanolic extract of
Cistanche deserticola on the contents of monoamine
neurotransmitters in rat brain. Chin Tradit Herbal Drugs.
24:417–419. 1993.
|
|
26
|
He ZD, Lau KM, Xu H-X, Li PC and Pui-Hay
But P: Antioxidant activity of phenylethanoid glycosides from
Brandisia hancei. J Ethnopharmacol. 71:483–486. 2000.
View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Li J, Wang PF, Zheng R, Liu ZM and Jia Z:
Protection of phenylpropanoid glycosides from Pedicularis
against oxidative hemolysis in vitro. Planta Med. 59:315–317. 1993.
View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Xiong Q, Kadota S, Tani T and Namba T:
Antioxidative effects of phenylethanoids from Cistanche
deserticola. Biol Pharm Bull. 19:1580–1585. 1996. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Zong G, He W, Wu G, Chen M, Shen X and Shi
M: Comparisons between Cistanche deserticola Y.C. Ma and
C. tubulosa (Schenk) Wight on Some Pharmacological Actions.
Zhongguo Zhong Yao Za Zhi. 21:436–437. 1996.(In Chinese).
PubMed/NCBI
|
|
30
|
Bondet V, Brand-Williams W and Berset C:
Kinetics and mechanisms of antioxidant activity using the DPPH.
free radical method. Lebensm Wiss Technol. 30:609–615. 1997.
View Article : Google Scholar
|
|
31
|
van den Berg R, Haenen GR, van den Berg H
and Bast A: Applicability of an improved Trolox equivalent
antioxidant capacity (TEAC) assay for evaluation of antioxidant
capacity measurements of mixtures. Food Chem. 66:511–517. 1999.
View Article : Google Scholar
|
|
32
|
Jung Y and Surh Y: Oxidative DNA damage
and cytotoxicity induced by copper-stimulated redox cycling of
salsolinol, a neurotoxic tetrahydroisoquinoline alkaloid. Free
Radic Biol Med. 30:1407–1417. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2−ΔΔCT method. Methods. 25:402–408. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Otterbein LE, Hedblom A, Harris C,
Csizmadia E, Gallo D and Wegiel B: Heme oxygenase-1 and carbon
monoxide modulate DNA repair through ataxia-telangiectasia mutated
(ATM) protein. Proc Natl Acad Sci USA. 108:14491–14496. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Kang U, Chang CS and Kim YS: Genetic
structure and conservation considerations of rare endemic
Abeliophyllum distichum Nakai (Oleaceae) in Korea. J Plant
Res. 113:127–138. 2000. View Article : Google Scholar
|
|
36
|
Kim NY and Lee HY: Effect of antioxidant
and skin whitening of ethanol extracts from ultrasonic pretreated
Abeliophyllum distichum Nakai. Hanguk Yakyong Changmul
Hakhoe Chi. 23:155–160. 2015. View Article : Google Scholar
|
|
37
|
Kim HW, Lee HL, Lee DK and Kim KJ:
Complete plastid genome sequences of Abeliophyllum distichum
Nakai (Oleaceae), a Korea endemic genus. Mitochondrial DNA B
Resour. 1:596–598. 2016. View Article : Google Scholar
|
|
38
|
Park J, Kim Y, Xi H, Jang T and Park JH:
The complete chloroplast genome of Abeliophyllum distichum
Nakai (Oleaceae), cultivar Ok Hwang 1ho: Insights of cultivar
specific variations of A. distichum. Mitochondrial DNA B
Resour. 4:1640–1642. 2019. View Article : Google Scholar
|
|
39
|
Min J, Kim Y, Xi H, Jang T, Kim G, Park J
and Park JH: The complete chloroplast genome of a new candidate
cultivar, Sang Jae, of Abeliophyllum distichum Nakai
(Oleaceae): Initial step of A. distichum intraspecies
variations atlas. Mitochondrial DNA B Resour. 4:3716–3718. 2019.
View Article : Google Scholar
|
|
40
|
Park J, Min J, Kim Y, Xi H, Kwon W, Jang
T, Kim G and Park JH: The complete chloroplast genome of a new
candidate cultivar, Dae Ryun, of Abeliophyllum distichum
Nakai (Oleaceae). Mitochondrial DNA B Resour. 4:3713–3715. 2019.
View Article : Google Scholar
|
|
41
|
Bremer B, Bremer K, Heidari N, Erixon P,
Olmstead RG, Anderberg AA, Källersjö M and Barkhordarian E:
Phylogenetics of asterids based on 3 coding and 3 non-coding
chloroplast DNA markers and the utility of non-coding DNA at higher
taxonomic levels. Mol Phylogenet Evol. 24:274–301. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Pisoschi AM, Cheregi MC and Danet AF:
Total antioxidant capacity of some commercial fruit juices:
Electrochemical and spectrophotometrical approaches. Molecules.
14:480–493. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Prasad KN, Chew LY, Khoo HE, Kong KW,
Azlan A and Ismail A: Antioxidant capacities of peel, pulp, and
seed fractions of Canarium odontophyllum Miq. fruit. J
Biomed Biotechnol. 2010:8713792010. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Sies H: Strategies of antioxidant defense.
Eur J Biochem. 215:213–219. 1993. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Devasagayam TP, Tilak JC, Boloor KK, Sane
KS, Ghaskadbi SS and Lele RD: Free radicals and antioxidants in
human health: Current status and future prospects. J Assoc
Physicians India. 52:794–804. 2004.PubMed/NCBI
|
|
46
|
Alfadda AA and Sallam RM: Reactive oxygen
species in health and disease. J Biomed Biotechnol.
2012:9364862012. View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Asif M: Chemistry and antioxidant activity
of plants containing some phenolic compounds. Chem Int. 1:35–52.
2015.
|
|
48
|
Blazics B, Alberti Á, Kursinszki L, Kéry
Á, Béni S and Tölgyesi L: Identification and LC-MS-MS determination
of acteoside, the main antioxidant compound of Euphrasia
rostkoviana, using the isolated target analyte as external
standard. J Chromatogr Sci. 49:203–208. 2011. View Article : Google Scholar
|
|
49
|
Ahn J and Park JH: Effects of
Abeliophyllum distichum Nakai flower extracts on
antioxidative activities and inhibition of DNA damage. Korean J
Plant Resour. 26:355–361. 2013. View Article : Google Scholar
|
|
50
|
Park JH: Antioxidant activities and
inhibitory effect on oxidative DNA damage of extracts from
Abeliophylli distichi Folium. Korea J Herbology. 26:95–99.
2011.
|
|
51
|
Sieniawska E, Baj T and Głowniak K:
Influence of the preliminary sample preparation on the tannins
content in the extracts obtained from Mutellina purpurea.
Ann UMCS Sect. 1500:47–54. 2010.
|
|
52
|
Luís Â, Domingues F, Gil C and Duarte AP:
Antioxidant activity of extracts of Portuguese shrubs:
Pterospartum tridentatum, Cytisus scoparius and Erica spp. J
Med Plants Res. 3:886–893. 2009.
|
|
53
|
Venkatasubramanian L and Maruthamuthu P:
Kinetics and mechanism of formation and decay of
2,2-azinobis-(3-ethylbenzothiazole-6-sulphonate) radical cation in
aqueous solution by inorganic peroxides. Int J Chem Kinet.
21:399–421. 1989. View Article : Google Scholar
|
|
54
|
Branchi B, Galli C and Gentili P: Kinetics
of oxidation of benzyl alcohols by the dication and radical cation
of ABTS. Comparison with laccase-ABTS oxidations: An apparent
paradox. Org Biomol Chem. 3:2604–2614. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
55
|
Gates KS: An overview of chemical
processes that damage cellular DNA: Spontaneous hydrolysis,
alkylation, and reactions with radicals. Chem Res Toxicol.
22:1747–1760. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
56
|
Sancar A and Sancar GB: DNA repair
enzymes. Annu Rev Biochem. 57:29–67. 1988. View Article : Google Scholar : PubMed/NCBI
|
|
57
|
Cowan JA: Chemical nucleases. Curr Opin
Chem Biol. 5:634–642. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
58
|
Vacek J, Mozga T, Cahová K, Pivoňková H
and Fojta M: Electrochemical sensing of chromium-induced DNA
damage: DNA strand breakage by intermediates of chromium (VI)
electrochemical reduction. Electroanalysis. Int J Devoted Fundam
Pract Asp Electroanalysis. 19:2093–2102. 2007.
|
|
59
|
Kanvah S and Schuster GB: One-electron
oxidation of DNA: The effect of replacement of cytosine with
5-methylcytosine on long-distance radical cation transport and
reaction. J Am Chem Soc. 126:7341–7344. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
60
|
Demple B and Harrison L: Repair of
oxidative damage to DNA: Enzymology and biology. Annu Rev Biochem.
63:915–948. 1994. View Article : Google Scholar : PubMed/NCBI
|
|
61
|
Poulsen HE, Prieme H and Loft S: Role of
oxidative DNA damage in cancer initiation and promotion. Eur J
Cancer Prev. 7:9–16. 1998.PubMed/NCBI
|
|
62
|
Hasty P and Vijg J: Aging. Genomic
priorities in aging. Science. 296:1250–1251. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
63
|
Hutchinson F: Chemical changes induced in
DNA by ionizing radiation. Prog Nucleic Acid Res Mol Biol.
32:115–154. 1985. View Article : Google Scholar : PubMed/NCBI
|
|
64
|
Bennett CB, Lewis AL, Baldwin KK and
Resnick MA: Lethality induced by a single site-specific
double-strand break in a dispensable yeast plasmid. Proc Natl Acad
Sci USA. 90:5613–5617. 1993. View Article : Google Scholar : PubMed/NCBI
|
|
65
|
Mills KD, Ferguson DO and Alt FW: The role
of DNA breaks in genomic instability and tumorigenesis. Immunol
Rev. 194:77–95. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
66
|
Rogakou EP, Pilch DR, Orr AH, Ivanova VS
and Bonner WM: DNA double-stranded breaks induce histone H2AX
phosphorylation on serine 139. J Biol Chem. 273:5858–5868. 1998.
View Article : Google Scholar : PubMed/NCBI
|
|
67
|
Celeste A, Fernandez-Capetillo O, Kruhlak
MJ, Pilch DR, Staudt DW, Lee A, Bonner RF, Bonner WM and
Nussenzweig A: Histone H2AX phosphorylation is dispensable for the
initial recognition of DNA breaks. Nat Cell Biol. 5:675–679. 2003.
View Article : Google Scholar : PubMed/NCBI
|
|
68
|
Sedelnikova OA, Pilch DR, Redon C and
Bonner WM: Histone H2AX in DNA damage and repair. Cancer Biol Ther.
2:233–235. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
69
|
Phillips ER and McKinnon PJ: DNA
double-strand break repair and development. Oncogene. 26:7799–7808.
2007. View Article : Google Scholar : PubMed/NCBI
|
|
70
|
Kastan MB, Onyekwere O, Sidransky D,
Vogelstein B and Craig RW: Participation of p53 protein in the
cellular response to DNA damage. Cancer Res. 51:6304–6311.
1991.PubMed/NCBI
|