Introduction
Stroke, a cerebrovascular disease with high
morbidity, mortality, disability rate and recurrence, is a severe
threat to human health and life and remains the second leading
cause of death worldwide (1,2).
Ischemic strokes account for ~87% of all stroke cases, with the
remainder caused mainly by primary hemorrhage (2–5).
Ischemic heart disease and stroke together accounted for 15.2
million mortalities worldwide in 2015 (1), while the clinically effective
treatments are limited to thrombolytic therapy and symptom
management (6–8). Therefore, it is essential to
elucidate the pathogenic molecular mechanism of ischemic stroke to
construct a novel strategy for clinical diagnosis and treatment of
ischemic cerebrovascular disease.
Non-coding RNAs (ncRNAs), including microRNAs
(miRNAs or miRs), circular RNAs (circRNAs) and long non-coding RNAs
(lncRNAs), are RNAs that are transcribed from the genome; however,
they are not translated into proteins (9,10).
ncRNAs serve critical roles as transcriptional and
post-transcriptional regulators (9). Several previous studies on the
central nervous system have demonstrated that the ncRNA expression
profiles are altered during a stroke, including those of various
miRNAs, Piwi-interacting RNAs and lncRNAs (11,12).
Previous studies on the pathophysiological changes following
strokes have mainly focused on miRNAs. For instance, miR-15b has
been reported to inhibit the proliferation of neural progenitor
cells by downregulating Tet methylcytosine dioxygenase 3 expression
(13) and miR-140 was revealed to
inhibit the proliferation of astrocytes and downregulate the
expression and secretion of brain-derived neurotrophic factor
(14). However, compared with
miRNAs, the role of lncRNAs in the pathogenesis of strokes remains
unclear.
Emerging evidence has demonstrated that the
dysregulation of lncRNA expression is closely associated with the
pathogenesis of ischemic stroke (2,4,15).
lncRNAs can act as competitive endogenous RNAs (ceRNAs) in ischemic
stroke by competing with miRNAs to regulate specific RNA
transcription (16). miR-181a-5p
is a critical member of the miRNA family and is mainly involved in
the regulation of the proliferation and apoptosis of cancer cells
(17). A previous study reported
that miR-181a-5p levels were significantly increased in the serum
of patients with ischemic stroke (18). However, few studies have been
conducted on the role of miR-181a-5p in ischemic stroke and its
regulatory mechanism (19). It has
been demonstrated that a lncRNA, small nucleolar RNA host gene 12
(SNHG12), serves a critical role in ischemic stroke by acting as a
ceRNA and competing with various miRNAs to regulate specific RNA
transcription (20,21). SNHG12 has been revealed to
downregulate the level of miR-181a-5p and enhance the sensitivity
of cancer cells to cisplatin (22). Furthermore, SNHG12 was revealed to
inhibit endothelial cell injury induced by oxygen-glucose
deprivation and repair vascular injury following stroke (23). However, it remains unclear whether
SNHG12 serves a protective role by regulating miR-181a-5p in
oxygen-deprived neurons. Thus the present study aimed to
investigate the regulatory function of SNHG12 and miR-181a-5p in
neuronal apoptosis, which may provide novel strategies for ischemic
stroke therapy.
Materials and methods
Cell culture, cell transfection and
OGD treatment
The SH-SY5Y cell line was obtained from the American
Type Culture Collection and cultured in DMEM (Gibco; Thermo Fisher
Scientific, Inc.) supplemented with 10% FBS, 2 mmol/l glutamine
(Gibco; Thermo Fisher Scientific, Inc.), 100 U/ml penicillin and
100 mg/ml streptomycin (Gibco; Thermo Fisher Scientific, Inc.) at
37°C with humidified air at 5% CO2. miR-181a-5p mimics,
miR-181a-5p inhibitors, small interfering RNAs (siRNAs: siControl
and siNHG12), control mimics, SNHG12-expressing plasmids
(pcDNA3.1-SNHG12) and corresponding negative controls (pcDNA3.1-NC)
were transfected into cells using Lipofectamine® 2000
(Invitrogen; Thermo Fisher Scientific, Inc.), according to the
manufacturer's protocol. The sequences were: miR-181a-5p mimics:
5′-AACAUUCAACGCUGUCGGUGAGU-3′; mimics negative control:
5′-UUUGUACUACACAAAAGUACUG-3′ siControl:
5′-GTTCTCCGAACGTGTCACGTA-3′; siNHG12: 5′-GACCAGAAGATAATGTTCAAG-3′.
At 48 h post-transfection, the cells were split for subsequent
experiments
For OGD treatment in vitro, cells were washed
three times with warm, glucose-free DMEM and placed in glucose-free
DMEM, following, cells were cultured in full culture medium with
normoxic oxygen at 37°C and were maintained for 0, 3, 6, 12 and 24
h. Control cells were cultured in DMEM/high glucose at normoxia.
All cells were incubated in an anaerobic chamber containing 5%
CO2 and 95% N2 at 37°C for the indicated
time-points (24).
Brain RNA-seq database
The expression level of NEGR1 in neurons,
astrocytes, oligodendrocyte precursor cells, newly formed
oligodendrocytes, myelinating oligodendrocytes, microglia, and
endothelial cells were obtained from a transcriptome database
(http://web.stanford.edu/group/barres_lab/brain_rnaseq.html:
September 2014 release) generated by RNA sequencing and a sensitive
algorithm to detect alternative splicing events in each cell
type.
RNA extraction and reverse
transcription-quantitative PCR (RT-qPCR)
A total of 58 patients, n=29 ischemic stroke
patients, and n=29 controls at Department of Neurology, Wenling
Hospital of Traditional Chinese Medicine, between July 2013 and
December 2015 were included. blood samples (10 ml) were collected
from arm veins of the 29 ischemic stroke patients and 29 controls
and written informed consent was obtained from all participants.
The total RNA of blood samples and SH-SY5Y cell lines were
extracted using TRIzol® reagent (Invitrogen; Thermo
Fisher Scientific, Inc). Extracted RNA was reverse-transcribed into
cDNA using a Takara reverse transcription kit (Takara Bio, Inc.),
according to the manufacturer's protocol. RT-qPCR was performed
using IQ SYBR-Green Supermix (Bio-Rad Laboratories, Inc.) and a
real-time PCR detection system (Light Cycler 480; Roche
Diagnostics). Thermocycling conditions were: 95°C for 30 sec,
followed by 95°C for 10 sec and 60°C for 30 sec for 40 cycles. The
relative expression level was calculated using the
2−ΔΔCq method (25).
The sequences of the primers used were: SNHG12 forward,
5′-CGGATTTTTCCGTCTGGTCC-3′ and reverse,
5′-TCTGGTCTCCCTCCTCACAAT-3′; miR-181a-5p forward,
5′-AACATTCAACGCTGTCG-3′ and reverse, 5′-AACTGTGTCGTGGAG-3′;
neuronal growth regulator 1 (NEGR1) forward,
5′-TGAAGCAGCGTGGGATACAAT-3′ and reverse,
5′-CCAGCGATTCCACAGACAAA-3′; GAPDH forward,
5′-GTCAACGGATTTGGTCTGTATT-3′ and reverse,
5′-AGTCTTCTGGGTGGCAGTGAT-3′; and U6 forward,
5′-GCTTCGGCAGCACATATACTAA-3′ and reverse,
5′-AACGCTTCACGAATTTGCGT-3′. GAPDH was used as an internal control
for mRNA and U6 small nuclear RNA was used as an internal control
for lncRNA and miRNA.
Apoptosis assay
SH-SY5Y cells were treated with the indicated
stimulating factors shown in Figs.
1B, 3C, 4I and 5E
and harvested for staining using an Annexin V-FITC/propidium iodide
apoptosis detection kit (BD Biosciences), according to the
manufacturer's protocol. Briefly, harvested cells were treated with
10 µg unlabeled Annexin V for 10 min followed by Annexin V-FITC/PI
staining. Cells at different apoptotic periods were detected by
flow cytometry using Annexin V probe conjugated to FITC (a green
fluorescent; FL1 channel) and PI (a dye for nucleic acid with red
fluorescence that cannot penetrate the intact cell membrane of
healthy or early apoptotic cells; FL3 channel). The proportion of
all apoptotic cells, including early and late apoptotic cells, was
measured and analyzed by flow cytometry using a FACS verse flow
cytometer (BD Biosciences) and CELLquest software (v5.1, Becton
Dickinson).
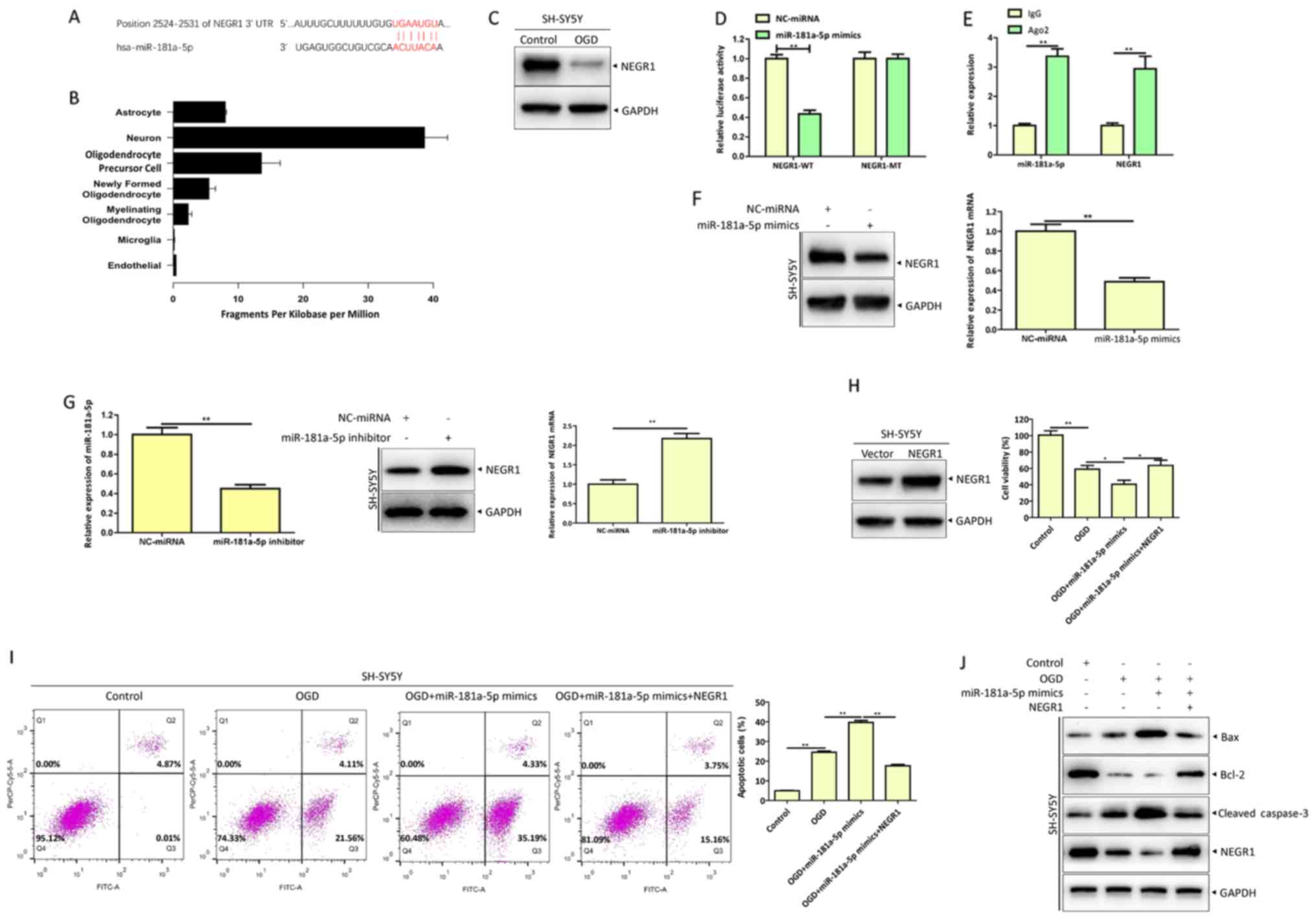 | Figure 4.miR-181a-5p inhibits NEGR1 expression
and promotes apoptosis of SH-SY5Y cells. (A) Binding site of NEGR1
to miR-181a-5p as predicted by TargetScan. (B) RNA-seq analysis of
the expression profile of NEGR1 in various cells. (C) SH-SY5Y cells
were treated with OGD for 12 h and NEGR1 expression was detected by
western blotting. (D) NEGR1-WT or NEGR1-MT reporter plasmids were
co-transfected with NC-miRNA or miR-181a-5p mimics, respectively,
into SH-SY5Y cells. The activities of luciferase were measured
using a Dual Luciferase Reporter Assay System. (E) SH-SY5Y cells
were harvested and lysed, and the lysates were used for RNA
immunoprecipitations assays with Ago2 antibodies. IgG was used as
the control. (F) SH-SY5Y cells were transfected with NC-miRNA or
miR-181a-5p mimics, and at 48 h post-transfection NEGR1 protein
levels were detected by western blotting. (G) Expression levels of
NEGR1 and miR-181a-5p were detected by reverse
transcription-quantitative PCR. (H) NC-miRNA or miR-181a-5p mimics
were transfected into SH-SY5Y cells with an overexpressed NEGR1
plasmids or controls, respectively. At 48 h post-transfection,
cells were treated with OGD for 12 h and cell viability was
measured via Cell Counting Kit-8 assays. (I) Apoptosis rate was
determined by flow cytometry. (J) Protein levels of Bcl-2, Bax and
cleaved caspase-3 were analyzed by western blotting. *P<0.05 and
**P<0.01. miR or miRNA, microRNA; NEGR1, neuronal growth
regulator 1; OGD, oxygen-glucose deprivation; WT, wild-type; MT,
mutant; NC, negative control; Ago2, argonaute 2; IgG,
immunoglobulin G; Bcl-2, B-cell lymphoma 2; Bax, Bcl-2 associated X
protein; UTR, untranslated region. |
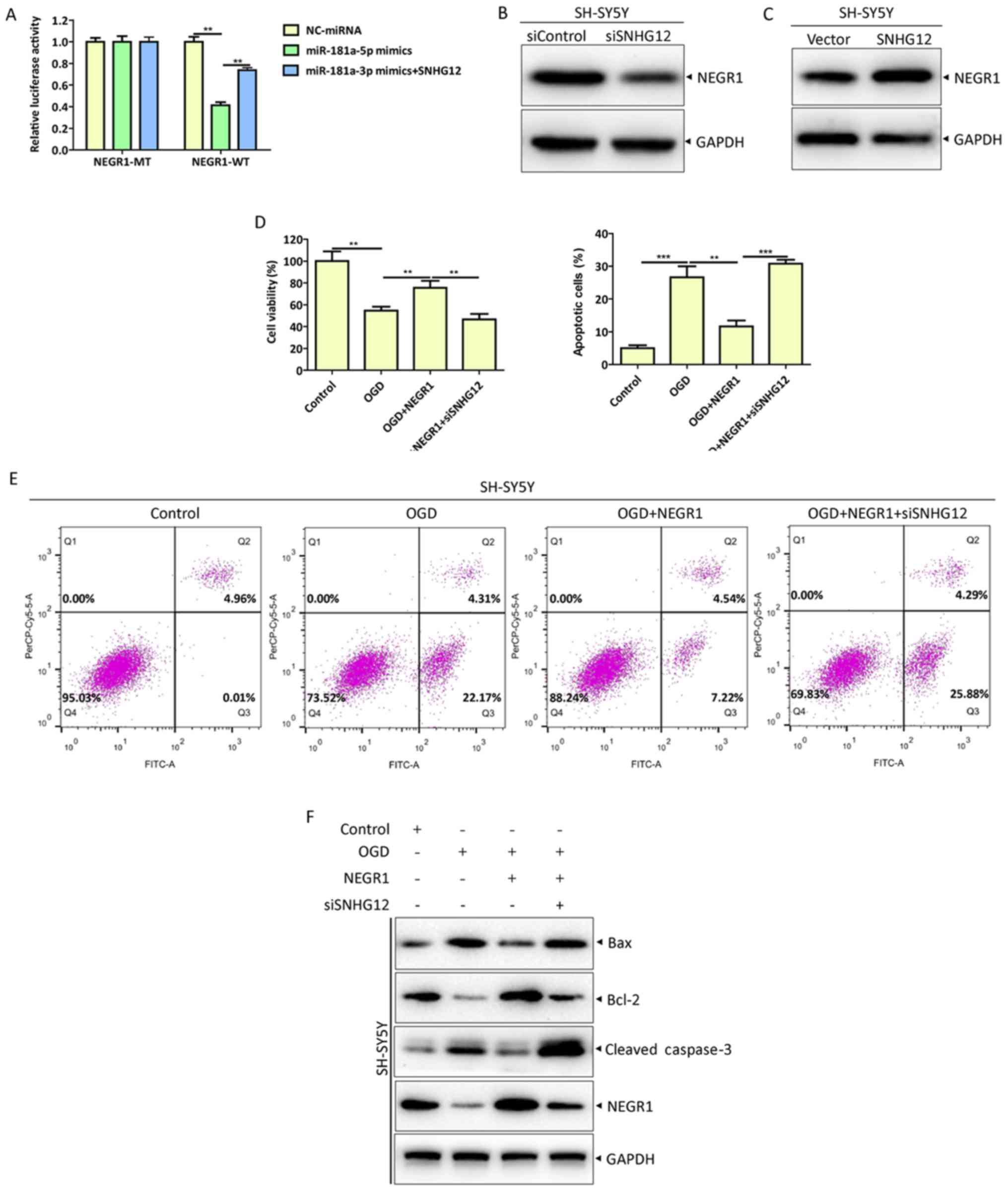 | Figure 5.SNHG12 inhibits OGD-induced apoptosis
in SH-SY5Y cells through the miR-181a-5p/NEGR1 axis. (A) Cells were
co-transfected with the indicated plasmids or miRNAs and luciferase
activity was measured using a Dual-Luciferase Reporter Assay
System. Protein levels of NEGR1 were analyzed by western blotting
in (B) SNHG12 knockdown or (C) overexpressed SH-SY5Y cells. NEGR1
overexpression and internal control plasmids were co-transfected
with siSNHG12 or siControl into SH-SY5Y cells, respectively. At 48
h post-transfection, cells were treated with OGD for 12 h. (D) Cell
viability was measured by Cell Counting Kit-8 assays. (E) Apoptosis
rates were determined by flow cytometry. (F) Protein levels of
Bcl-2, Bax and cleaved caspase-3 were analyzed by western blotting.
**P<0.01 and ***P<0.001. SNHG12, small nucleolar RNA host
gene 12; OGD, oxygen-glucose deprivation; miR or miRNA, microRNA;
NEGR1, neuronal growth regulator 1; si, small interfering; Bcl-2,
B-cell lymphoma 2; Bax, Bcl-2 associated X protein; MT, mutant; WT,
wild-type; NC, negative control |
RNA immunoprecipitation (RIP)
assay
RIP experiments were performed using a Magna RIP kit
(EMD Millipore), according to the manufacturer's protocol. Briefly,
SH-SY5Y cells were washed with cold PBS and lysed in RIP lysis
buffer. Cells were then incubated with RIP buffer and magnetic
beads conjugated with anti-argonaute 2 (Ago2) antibodies (EMD
Millipore; cat. no. LINC00980) or NC mouse immunoglobulin G (IgG;
EMD Millipore). Following this, proteins were digested with
protease K buffer and immunoprecipitated RNA was isolated.
Corresponding RNA was purified and detected using RT-PCR and
RT-qPCR as described above.
Western blotting
SH-SY5Y cells were harvested and lysed in RIPA lysis
buffer (Beyotime Institute of Biotechnology) supplemented with
phosphatase inhibitors and protease inhibitors (Roche Diagnostics;
cat. no. 04693132001) and centrifuged at 12,000 × g at 4°C for 25
min. A BCA Protein Assay kit (Generay Biotech Co., Ltd.) was used
to measure the concentrations of extracted protein, according to
the manufacturer's protocol. A total of 30 µg of protein/lane was
separated by 8–12% SDS-PAGE gel and transferred to PVDF membranes
(EMD Millipore). Membranes were blocked in TBST buffer with 5%
non-fat skimmed milk for 45 min at room temperature and washed
three times with TBST buffer. Following this, membranes were
incubated overnight with primary antibodies at 4°C and with
horseradish peroxidase-labeled secondary antibodies (Santa Cruz
Biotechnology, Inc.; cat. nos. sc-2357 and sc-516102; 1:5,000) at
room temperature for 1 h. Bands were visualized using commercially
ECL reagents.
The following antibodies were used: B-cell lymphoma
2 (Bcl-2; product no. 15071; 1:1,000) Bcl-2 associated X protein
(Bax; cat. no. 5023; 1:1,000), cleaved caspase-3 (cat. no. 9654;
1:1,000; all from Cell Signaling Technology, Inc.), NEGR1 (cat. no.
sc-137625; 1:1,000; Santa Cruz Biotechnology, Inc.) and GAPDH (cat.
no. 5174; 1:2,000 Cell Signaling Technology, Inc.).
Cell Counting Kit-8 (CCK-8) assay
A total of 1×105 cells were plated in
96-well plates in triplicate for 24 h and treated with the
indicated stimulating factors for the indicated time-points shown
in Figs. 1A, 3B, 4H
and 5D, then CCK-8 reagent was
added into each well and further incubated for 2 h at 37°C, The
cell viability under various conditions was measured using a CCK-8
assay (Beyotime Institute of Biotechnology), according to the
manufacturer's protocol and the optical density at 450 nm
(OD450) value in each well was measured using a
microplate reader (Quant BioTek Instruments). Cell viability rates
were calculated as follows: Cell viability rate (%)=(experimental
OD/control group OD) ×100%.
Dual-luciferase reporter assay
Putative miR-181a-5p binding sites in SNHG12 and
binding sites in the 3′-untranslated region (UTR) of NEGR1 to
miR-181a-5p were predicted using StarBase (http://starbase.sysu.edu.cn/starbase2/, v2.0;
September 2013 release) and TargetScan (www.targetscan.org, v7.2; March 2018 release)
(26). Sequence fragments
SHNG12-wild-type (WT) and SHNG12-mutant (MT) and WT and MT 3′-UTR
fragments of NEGR1 containing putative miR-181a-5p binding sites
were cloned into pmirGLO dual-luciferase reporter vectors (Promega
Corporation) and confirmed by sequencing using Illumina MiSeq
MiSeqdxDNA (Illumina, Inc.) according to the manufacturer's
protocol. SH-SY5Y cells were then co-transfected with the
corresponding plasmids as described above (Cell culture, cell
transfection and OGD treatment). At 48 h post-transfection, a
Dual-Luciferase Reporter Assay System (Promega Corporation) was
used to measure luciferase activity. Relative luciferase activity
was normalized to the Renilla luciferase activity.
Statistical analysis
Experiments were performed in triplicate. Data are
presented as the mean ± standard error of the mean. GraphPad Prism
software (version 6.01; GraphPad Software, Inc.) was used for
statistical analysis of experimental data. Statistical differences
were assessed using unpaired two-tailed Student's t-tests for
comparison between two groups or ANOVA followed by Tukey's multiple
comparisons post hoc test for comparison between >2 groups.
P<0.05 was considered to indicate a statistically significant
difference.
Results
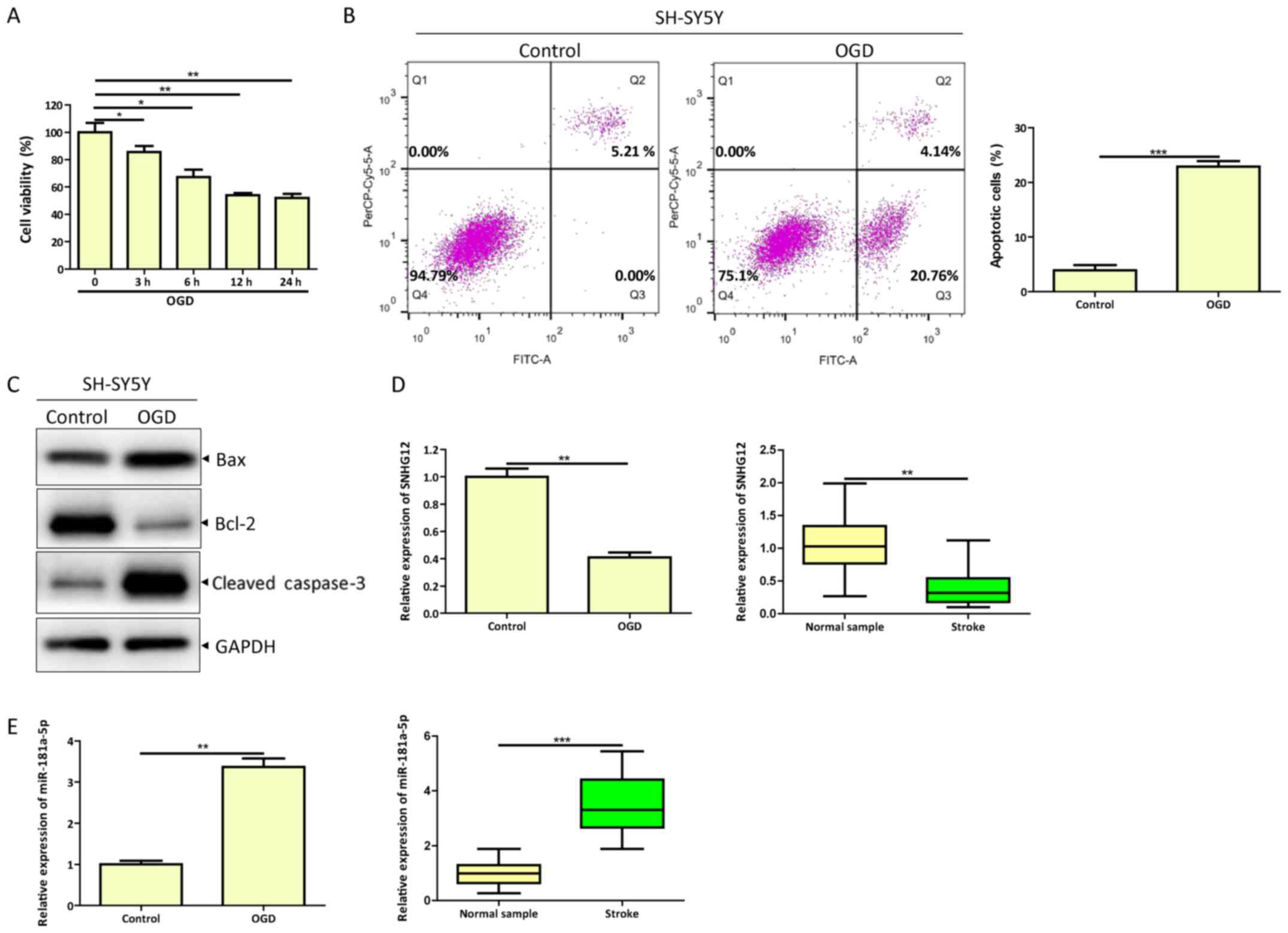
OGD induces apoptosis in SH-SY5Y
cells, reduction in SNHG12 expression and upregulation of
miR-181a-5p
To create an in vitro model of ischemia,
SH-SY5Y cells were treated with OGD (24). Viability of SH-SY5Y cells was
gradually decreased in the OGD group following 0, 3, 6 12 and 24 h
of treatment (Fig. 1A).
Furthermore, OGD promoted apoptosis in SH-SY5Y cells compared with
the control as indicated by apoptosis assays (Fig. 1B). Western blotting results for
apoptosis-related proteins revealed increased expression of cleaved
caspase-3 and Bax and decreased expression of Bcl-2 (Fig. 1C). To determine whether the
expression levels of SNHG12 and miR-181a-5p were altered following
OGD, RT-qPCR analysis was performed and the results indicated that
SNHG12 expression was significantly decreased and miR-181a-5p
expression was significantly increased compared with their
respective controls (Fig. 1D and
E). Consistent with these results, SNHG12 expression in the
serum of patients with cerebral ischemia was significantly
decreased and miR-181a-5p expression was significantly increased
compared with healthy controls (Fig.
1D and E). These results indicated that SNHG12 and miR-181a-5p
may be associated with OGD-induced apoptosis in SH-SY5Y cells.
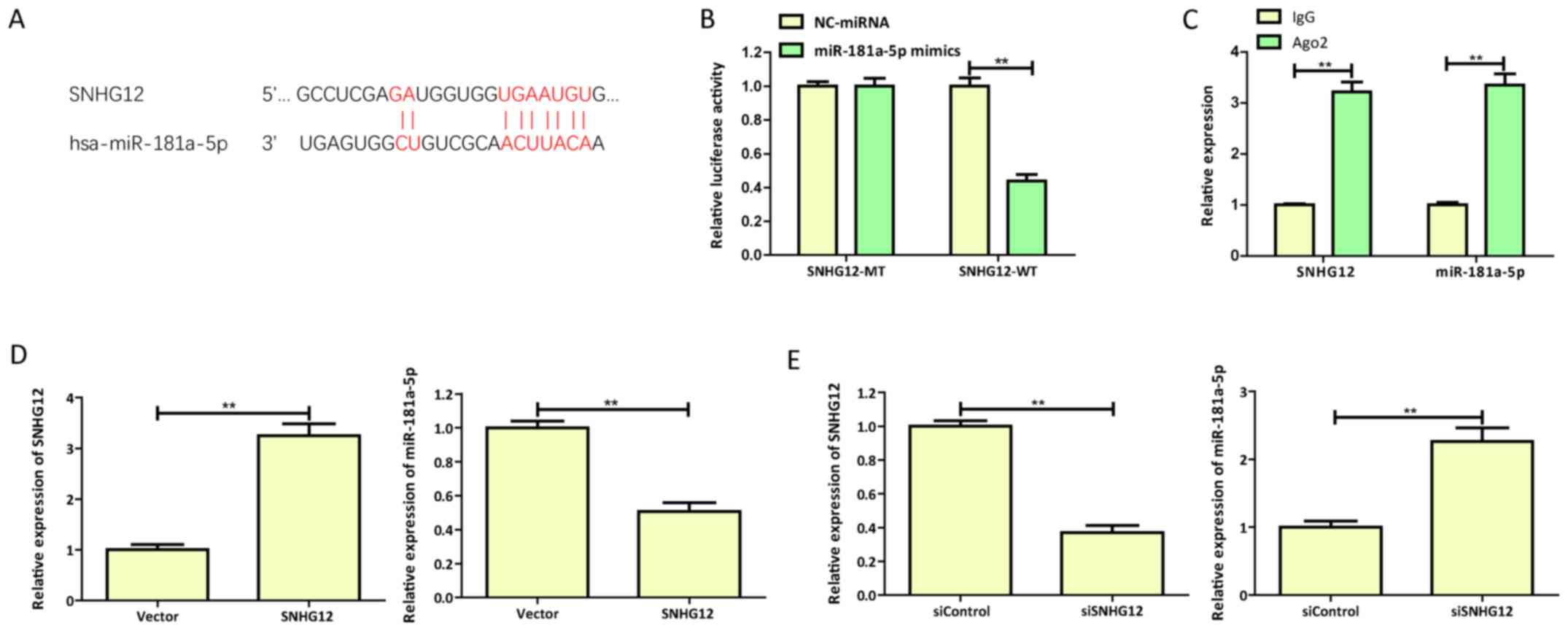
SNHG12 targets miR-181a-5p and
negatively regulates its expression
Increasing evidence has demonstrated that lncRNAs
act as ceRNAs by competing with miRNAs to regulate transcription
(12,16). The present study found that the
expression of SNHG12 and miR-181a-5p exhibited a negative
correlation in OGD-treated SH-SY5Y cells (Fig. 1D and E). Therefore, the possible
association between SNHG12 and miR-181a-5p was investigated. To
explore whether SNHG12 acts as a ceRNA of miR-181a-5p, the
bioinformatics analysis tools StarBase (http://starbase.sysu.edu.cn/starbase2/) and TargetScan
(http://www.targetscan.org) were used to
predict putative SNHG12 binding sites in miR-181a-5p (27). SNHG12 contained a conserved binding
site for miR-181a-5p (Fig.
2A).
To directly verify whether miR-181a-5p is a target
of SNHG12, dual-luciferase reporter assays were performed in
SH-SY5Y cells co-transfected with SNHG12 reporters (SNHG12-WT or
SNHG12-MT) and miR-181a-5p mimics or NC-miRNA. The results
demonstrated that miR-181a-5p mimics significantly reduced the
activity of the SNHG12-WT reporter; however, the luciferase
activity of the SNHG12-MT reporter was not affected (Fig. 2B). These results indicated that
miR-181a-5p binds to SNHG12 at the predicted binding site.
Furthermore, RIP assays confirmed the physical association between
miR-181a-5p and SNHG12 since SNHG12 and miR-181a were enriched in
Ago2 pellets compared with controls (Fig. 2C). Additionally, the exogenous
overexpression of SNHG12 significantly decreased the expression of
miR-181a-5p in OGD-injured SH-SY5Y cells, while siRNA-mediated
downregulation of SNHG12 (siSNHG12) demonstrated the opposite
effect (Fig. 2D and E).
In summary, these results confirmed that SNHG12 acts
as a ceRNA via direct inhibition of miR-181a-5p expression in
OGD-injured SH-SY5Y cells.
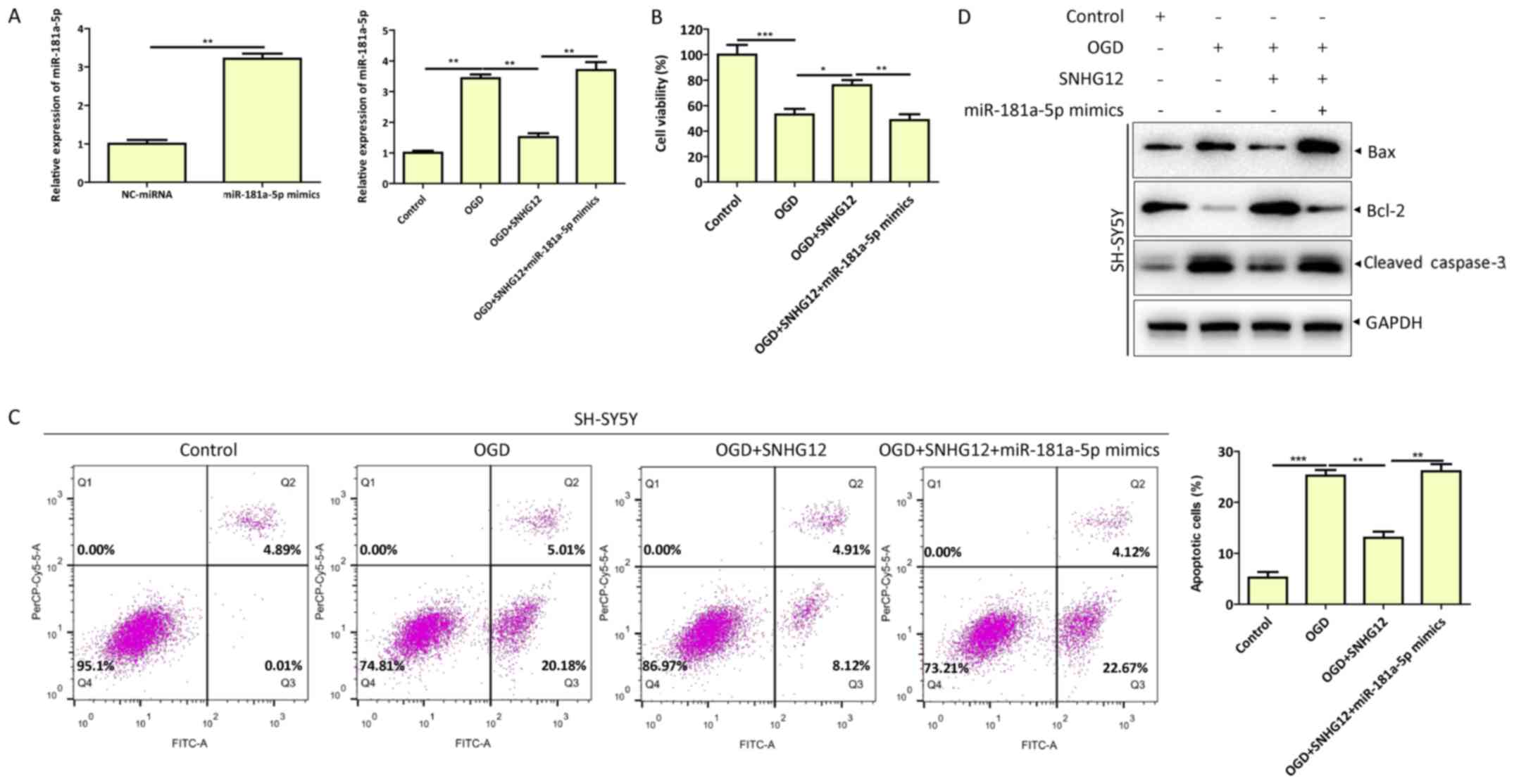
SNHG12 downregulates miR-181a-5p and
inhibits OGD-induced apoptosis of SH-SY5Y cells
To determine the function of SNHG12 and miR-181a-5p
in the apoptosis of OGD-injured SH-SY5Y cells, miR-181a-5p mimics
or NC-miRNA were transfected into SH-SY5Y cells, along with
pcDNA3.1-SNHG12 or pcDNA3.1-NC. Overexpression and downregulation
of miR-181a-5p was confirmed by RT-qPCR (Fig. 3A). Consistent with these results,
SNHG12 overexpression reduced miR-181a-5p expression, increased
cell viability and decreased apoptotic cell death compared with
control group (Fig. 3A-C). Western
blotting of apoptosis-related proteins Bax, Bcl-2 and cleaved
caspase-3 confirmed the effects of SNHG12 overexpression on
OGD-induced cell apoptosis (Fig.
3D). Furthermore, SNHG12 overexpression with co-transfected
miR-181a-5p mimics restored OGD-induced SNHG12-mediated inhibition
of SH-SY5Y cell apoptosis (Fig.
3C-D). These results demonstrated that SNHG12 regulated
apoptosis in OGD-injured SH-SY5Y cells by downregulating
miR-181a-5p expression.
miR-181a-5p inhibits NEGR1 expression
and promotes apoptosis of SH-SY5Y cells
Increasing evidence has indicated that miRNAs
function by binding to the 3′UTR of downstream target mRNAs to
regulate target genes (27,28).
Therefore, possible target genes of miR-181a-5p were predicted
using the web-based miRNA database TargetScan. Bioinformatics
analysis revealed that NEGR1 was a potential target gene of
miR-181a-5p. Identified target sites for miR-181a-5p in the 3′UTR
of NEGR1 are presented in Fig. 4A.
RNA-seq results obtained from the Brain RNA-seq database
(http://web.stanford.edu/group/barres_lab/brain_rnaseq.html)
demonstrated that NEGR1 was highly expressed in neurons (Fig. 4B). Furthermore, NEGR1 expression
was decreased in OGD-injured SH-SY5Y cells (Fig. 4C). Accordingly, dual-luciferase
reporter and RIP assays confirmed that NEGR1 is a target of
miR-181a-5p (Fig. 4D and E). To
further confirm the regulatory effect of miR-181a-5p on NEGR1, the
results for RT-qPCR and western blotting demonstrated that
miR-181a-5p overexpression significantly decreased the mRNA and
protein levels of NEGR1, while miR-181a-5p inhibition upregulated
the mRNA and protein levels of NEGR1 (Fig. 4F and G).
Previous studies have reported that NEGR1 is a
cellular adhesion molecule involved in neurite outgrowth during
neuronal development (29,30). The present study demonstrated that
NEGR1 was associated with OGD-induced neuronal apoptosis.
miR-181a-5p overexpression resulted in decreased cell viability
(Fig. 4H) and increased apoptotic
cell death (Fig. 4I) compared with
the control group. NEGR1 overexpression reversed the effect of
miR-181a-5p by exhibiting increased cell activity, decreased cell
apoptosis, increased expression of Bcl-2 and decreased expression
of cleaved caspase-3 and Bax (Fig.
4J).
In summary, these results revealed that NEGR1 is a
target of miR-181a-5p and that miR-181a-5p functions as a negative
modulator of NEGR1 to regulate OGD-induced neuronal apoptosis.
SNHG12 inhibits OGD-induced apoptosis
in SH-SY5Y cells through the miR-181a-5p/NEGR1 axis
To further confirm the regulation of the
SNHG12/miR-181a-5p/NEGR1 axis in OGD-induced neuronal apoptosis,
dual-luciferase, CCK-8 and apoptosis assays, and western blotting
were performed in SH-SY5Y cells transfected with NEGR1-MT or
NEGR1-WT in combination with NC-miRNA or miR-181a-5p mimics, or
co-transfected with siSNHG12 or PCDNA3.1-SNHG12. SNHG12
overexpression significantly increased NEGR1-WT luciferase activity
(Fig. 5A). Moreover, SNHG12
knockdown decreased the protein level of NEGR1 (Fig. 5B), while SNHG12 overexpression had
the opposite effect (Fig. 5C).
Additionally, SNHG12 siRNA notably abolished the effect of NEGR1
overexpression on cell viability (Fig.
5D) and apoptosis (Fig. 5E and
F) of OGD-injured SH-SY5Y cells.
Collectively, these results strongly confirmed the
hypothesis that SNHG12 functions as a ceRNA for miR-181a-5p to
regulate the expression of NEGR1 and to inhibit the OGD-induced
apoptosis of SH-SY5Y cells.
Discussion
There is ample evidence indicating that ischemic
stroke is caused by intracranial thrombosis or extracranial
embolism, resulting in a lack of blood flow and supply of oxygen
and nutrients (4,19,31).
Subsequently, multiple complex factors lead to the destruction of
the blood-brain barrier following ischemic stroke, which further
aggravates brain damage and a series of neurological events,
including oxidative stress, toxicity of excitatory amino acids,
excessively high calcium ion concentrations and increased apoptosis
(4,31,32).
Due to this, it is crucial to understand the molecular mechanism
underlying neuronal apoptosis in ischemic stroke to establish a
novel strategy for the clinical diagnosis and treatment of ischemic
cerebrovascular disease.
Emerging evidence has demonstrated that abnormal
expression of ncRNA, including miRNAs, circRNAs and lncRNAs, is
closely associated with the pathogenesis of ischemic stroke
(6,9,20).
SNHG12 is one of numerous lncRNAs involved in the progression of
ischemic stroke. Long et al (33) reported that SNHG12 suppressed brain
microvascular endothelial cell injury induced by oxygen-glucose
deprivation/reoxygenation by targeting miR-199a. Yin et al
(21) reported that SNHG12
interacted with miR-199a to attenuate apoptosis and cerebral
ischemia/reperfusion injury via the AMP-activated protein kinase
(AMPK) signaling pathway under oxygen-glucose
deprivation/reoxygenation conditions. Furthermore, silencing SNHG12
enhanced the effects of mesenchymal stem cells by reducing
apoptosis and autophagy of brain microvascular endothelial cells by
activating the phosphoinositide 3-kinase/protein kinase B/mammalian
target of rapamycin signaling pathway (34). SNHG12 functions as a ceRNA for
miR-150 to mediate VEGF expression and promote angiogenesis and
alleviate ischemic stroke (23).
Additionally, miR-181a-5p is reportedly involved in ischemia
(35). As miR-181a-5p is a
brain-enriched miRNA that exhibits elevated expression levels in
the cerebrospinal fluid of patients with acute ischemic stroke
(36), miR-181a-5p inhibition
enhanced estradiol-mediated stroke protection in females partly by
augmenting estrogen receptor α production in mouse and human
astrocytes (37). Moreover,
previous studies have demonstrated that miR-181a-5p inhibition has
a protective effect in male rodent models of stroke and forebrain
ischemia by targeting stress proteins and apoptotic regulators
(18,35).
Recently, Wang et al (22) demonstrated a regulatory association
between SNHG12 and miR-181a-5p by revealing that SNHG12
downregulated the level of miR-181a-5p and enhanced the sensitivity
of cancer cells to cisplatin (22). However, the specific function of
SNHG12 and miR-181a-5p in the process of OGD-induced neuronal
apoptosis and their therapeutic effects remain unclear. The present
study demonstrated that OGD leads to an induction of apoptosis in
SH-SY5Y cells, reduction in SNHG12 expression and upregulation of
miR-181a-5p. This indicated that the expression of SNHG12 and
miR-181a-5p were negatively associated in OGD-treated SH-SY5Y
cells. Therefore, the possible association between SNHG12 and
miR-181a-5p was investigated. The results reported that miR-181a-5p
was a target of SNHG12 and negatively regulated its expression.
Furthermore, miR-181a-5p functioned as a negative modulator of
NEGR1 to regulate OGD-induced neuronal apoptosis. Previous studies
have demonstrated that NEGR1 is a cellular adhesion molecule
involved in neurite outgrowth during neuronal development (29,30).
The present study reported, to the best of our knowledge, for the
first time that NEGR1 inhibited OGD-induced cell apoptosis. While
the present study only discussed the mechanism at one time-point,
it is not necessary to discuss the same mechanism at different
time-points. Different time-points under OGD treatment may
influence on the proportion of apoptosis and the expression level
of SNHG12, miR-181a-5p and NEGR1, not the association between
SNHG12, miR-181a-5p and NEGR1. RIP assays, western blotting,
dual-luciferase reporter assays and other experimental results
fully supported the conclusion that SNHG12 functioned as a ceRNA
for miR-181a-5p and regulated the expression of NEGR1 thus
inhibiting OGD-induced apoptosis in SH-SY5Y cells. Similarly,
previous studies (21,38) have discussed the interaction
mechanisms of SNHG12/miR-199a/SIRT1 and lncRNA
CRNDE/miR-181a-5p/Wnt/β-catenin axis at a single time-point.
In summary, the present study revealed that SNHG12
inhibited OGD-induced neuronal apoptosis via the miR-181a-5p/NEGR1
axis. This indicated that SNHG12 and NEGR1 serve anti-apoptotic
roles and miR-181a-5p serves a pro-apoptotic role in neurons
following OGD injury. Therefore, the inhibition of miR-181a-5p and
upregulation of SNHG12 and NEGR1 may be potential therapeutic
strategies for the treatment of cerebral ischemic injury. Future
studies are required to determine whether SNHG12 functions as a
ceRNA for miR-181a-5p to regulate the expression of NEGR1 in
vivo.
Acknowledgements
Not applicable.
Funding
The present study was supported by grant from the
Health Science and Technology Project of the Health and Family
Planning Commission of Hangzhou Municipality, China (grant no.
2017A45).
Availability of data and materials
The datasets used and/or analyzed during the present
study are available from the corresponding author on reasonable
request.
Authors' contributions
YY conducted experiments, acquired and analyzed the
data, and drafted the manuscript. LC conducted experiments and
acquired data. JZ analyzed and interpreted data, and revised the
manuscript. LX conceptualized and designed the data, analyzed and
interpreted data, and drafted and revised the manuscript. All
authors read and approved the final manuscript.
Ethics approval and consent to
participate
Blood samples were collected from patients with
cerebral ischemia and healthy controls, and all participants
provided written informed consent. The present study was approved
by the Ethics Committee of the Wenling Hospital of Traditional
Chinese Medicine, Wenling, Zhejiang 317500, China (Project ID:
H-I-2012-011).
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
Glossary
Abbreviations
Abbreviations:
|
lncRNAs
|
long non-coding RNAs
|
|
SNHG12
|
small nucleolar RNA host gene 12
|
|
ceRNAs
|
competitive endogenous RNAs
|
|
OGD
|
oxygen-glucose deprivation
|
References
|
1
|
Katan M and Luft A: Global burden of
stroke. Semin Neurol. 38:208–211. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Wu Z, Wu P, Zuo X, Yu NA, Qin Y, Xu Q, He
S, Cen B, Liao W and Ji A: LncRNA-N1LR enhances neuroprotection
against ischemic stroke probably by inhibiting p53 phosphorylation.
Mol Neurobiol. 54:7670–7685. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Yuh WT, Alexander MD and Beauchamp NJ:
Intraarterial treatment for acute ischemic stroke. N Engl J Med.
372:1176–1179. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Ren W and Yang X: Pathophysiology of long
non-coding RNAs in ischemic stroke. Front Mol Neurosci. 11:962018.
View Article : Google Scholar : PubMed/NCBI
|
|
5
|
GBD 2016 Lifetime Risk of Stroke
Collaborators, ; Feigin VL, Nguyen G, Cercy K, Johnson CO, Alam T,
Parmar PG, Abajobir AA, Abate KH, Abd-Allah F, et al: Global,
regional, and country-specific lifetime risks of stroke, 1990 and
2016. N Engl J Med. 379:2429–2437. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Li G, Morris-Blanco KC, Lopez MS, Yang T,
Zhao H, Vemuganti R and Luo Y: Impact of microRNAs on ischemic
stroke: From pre- to post-disease. Prog Neurobiol. 163-164:59–78.
2018. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Snow SJ: Stroke and t-PA-triggering new
paradigms of care. N Engl J Med. 374:809–811. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Henderson SJ, Weitz JI and Kim PY:
Fibrinolysis: Strategies to enhance the treatment of acute ischemic
stroke. J Thromb Haemost. 16:1932–1940. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Adams BD, Parsons C, Walker L, Zhang WC
and Slack FJ: Targeting noncoding RNAs in disease. J Clin Invest.
127:761–771. 2017. View
Article : Google Scholar : PubMed/NCBI
|
|
10
|
Wang M, Yu F, Wu W, Zhang Y, Chang W,
Ponnusamy M, Wang K and Li P: Circular RNAs: A novel type of
non-coding RNA and their potential implications in antiviral
immunity. Int J Biol Sci. 13:1497–1506. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Bao MH, Szeto V, Yang BB, Zhu SZ, Sun HS
and Feng ZP: Long non-coding RNAs in ischemic stroke. Cell Death
Dis. 9:2812018. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Mehta SL, Kim T and Vemuganti R: Long
noncoding RNA FosDT promotes ischemic brain injury by interacting
with REST-associated chromatin-modifying proteins. J Neurosci.
35:16443–16449. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Lv X, Jiang H, Liu Y, Lei X and Jiao J:
MicroRNA-15b promotes neurogenesis and inhibits neural progenitor
proliferation by directly repressing TET3 during early neocortical
development. EMBO Rep. 15:1305–1314. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Tu Z, Li Y, Dai Y, Li L, Lv G, Chen I and
Wang B: MiR-140/BDNF axis regulates normal human astrocyte
proliferation and LPS-induced IL-6 and TNF-α secretion. Biomed
Pharmacother. 91:899–905. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Deng Y, Chen D, Wang L, Gao F, Jin B, Lv
H, Zhang G, Sun X, Liu L, Mo D, et al: Silencing of long noncoding
RNA nespas aggravates microglial cell death and neuroinflammation
in ischemic stroke. Stroke. 50:1850–1858. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Wei R, Zhang L, Hu W, Wu J and Zhang W:
Long non-coding RNA AK038897 aggravates cerebral
ischemia/reperfusion injury via acting as a ceRNA for miR-26a-5p to
target DAPK1. Exp Neurol. 314:100–110. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Lu Z, Luo T, Pang T, Du Z, Yin X, Cui H,
Fang G and Xue X: MALAT1 promotes gastric adenocarcinoma through
the MALAT1/miR-181a-5p/AKT3 axis. Open Biol. 9:1900952019.
View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Moon J, Xu L and Giffard RG: Inhibition of
microRNA-181 reduces forebrain ischemia-induced neuronal loss. J
Cereb Blood Flow Metab. 33:1976–1982. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Wu J, Fan CL, Ma LJ, Liu T, Wang C, Song
JX, Lv QS, Pan H, Zhang CN and Wang JJ: Distinctive expression
signatures of serum microRNAs in ischaemic stroke and transient
ischaemic attack patients. Thromb Haemost. 117:992–1001. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Cheng Y, Jiang Y, Sun Y and Jiang H: The
role of long non-coding RNA SNHG12 in neuroprotection following
cerebral ischemic injury. Neuroreport. 30:945–952. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Yin WL, Yin WG, Huang BS and Wu LX: LncRNA
SNHG12 inhibits miR-199a to upregulate SIRT1 to attenuate cerebral
ischemia/reperfusion injury through activating AMPK signaling
pathway. Neurosci Lett. 690:188–195. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Wang P, Chen D, Ma H and Li Y: LncRNA
SNHG12 contributes to multidrug resistance through activating the
MAPK/Slug pathway by sponging miR-181a in non-small cell lung
cancer. Oncotarget. 8:84086–84101. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Zhao M, Wang J, Xi X, Tan N and Zhang L:
SNHG12 promotes angiogenesis following ischemic stroke via
regulating miR-150/VEGF pathway. Neuroscience. 390:231–240. 2018.
View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Liang J, Yu Y, Wang B, Lu B, Zhang J,
Zhang H and Ge P: Ginsenoside Rb1 attenuates oxygen-glucose
deprivation-induced apoptosis in SH-SY5Y cells via protection of
mitochondria and inhibition of AIF and cytochrome c release.
Molecules. 18:12777–12792. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) method. Methods. 25:402–408. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Shang D, Xie C, Hu J, Tan J, Yuan Y, Liu Z
and Yang Z: Pancreatic cancer cell-derived exosomal microRNA-27a
promotes angiogenesis of human microvascular endothelial cells in
pancreatic cancer via BTG2. J Cell Mol Med. 24:588–604. 2020.
View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Wang X, Lan Z, He J, Lai Q, Yao X, Li Q,
Liu Y, Lai H, Gu C, Yan Q, et al: LncRNA SNHG6 promotes
chemoresistance through ULK1-induced autophagy by sponging
miR-26a-5p in colorectal cancer cells. Cancer Cell Int. 19:2342019.
View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Liu K, Xie F, Gao A, Zhang R, Zhang L,
Xiao Z, Hu Q, Huang W, Huang Q, Lin B, et al: SOX2 regulates
multiple malignant processes of breast cancer development through
the SOX2/miR-181a-5p, miR-30e-5p/TUSC3 axis. Mol Cancer. 16:622017.
View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Kaur P, Tan JR, Karolina DS, Sepramaniam
S, Armugam A, Wong PTH and Jeyaseelan K: A long non-coding RNA,
BC048612 and a microRNA, miR-203 coordinate the gene expression of
neuronal growth regulator 1 (NEGR1) adhesion protein. Biochim
Biophys Acta. 1863:533–543. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Kaur P, Tan JR, Karolina DS, Sepramaniam
S, Armugam A, Wong THP and Jeyaseelan K: Supporting data for
characterization of non-coding RNAs associated with the neuronal
growth regulator 1 (NEGR1) adhesion protein. Data Brief. 7:381–385.
2016. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Sweeney MD, Zhao Z, Montagne A, Nelson AR
and Zlokovic BV: Blood-brain barrier: From physiology to disease
and back. Physiol Rev. 99:21–78. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Liu YC, Tsai YH, Tang SC, Liou HC, Kang
KH, Liou HH, Jeng JS and Fu WM: Cytokine MIF enhances blood-brain
barrier permeability: Impact for therapy in ischemic stroke. Sci
Rep. 8:7432018. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Long FQ, Su QJ, Zhou JX, Wang DS, Li PX,
Zeng CS and Cai Y: LncRNA SNHG12 ameliorates brain microvascular
endothelial cell injury by targeting miR-199a. Neural Regen Res.
13:1919–1926. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Li Y, Guo S, Liu W, Jin T, Li X, He X,
Zhang X, Su H, Zhang N and Duan C: Silencing of SNHG12 enhanced the
effectiveness of MSCs in alleviating ischemia/reperfusion injuries
via the PI3K/AKT/mTOR signaling pathway. Front Neurosci. 13:1–12.
2019. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Ouyang YB, Lu Y, Yue S, Xu LJ, Xiong XX,
White RE, Sun X and Giffard RG: miR-181 regulates GRP78 and
influences outcome from cerebral ischemia in vitro and in vivo.
Neurobiol Dis. 45:555–563. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Sørensen SS, Nygaard AB, Carlsen AL,
Heegaard NHH, Bak M and Christensen T: Elevation of brain-enriched
miRNAs in cerebrospinal fluid of patients with acute ischemic
stroke. Biomark Res. 5:242017. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Stary CM, Xu L, Li L, Sun X, Ouyang YB,
Xiong X, Zhao J and Giffard RG: Inhibition of miR-181a protects
female mice from transient focal cerebral ischemia by targeting
astrocyte estrogen receptor-α. Mol Cell Neurosci. 82:118–125. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Han P, Li JW, Zhang BM, Lv JC, Li YM, Gu
XY, Yu ZW, Jia YH, Bai XF, Li L, et al: The lncRNA CRNDE promotes
colorectal cancer cell proliferation and chemoresistance via
miR-181a-5p-mediated regulation of Wnt/β-catenin signaling. Mol
Cancer. 16:92017. View Article : Google Scholar : PubMed/NCBI
|