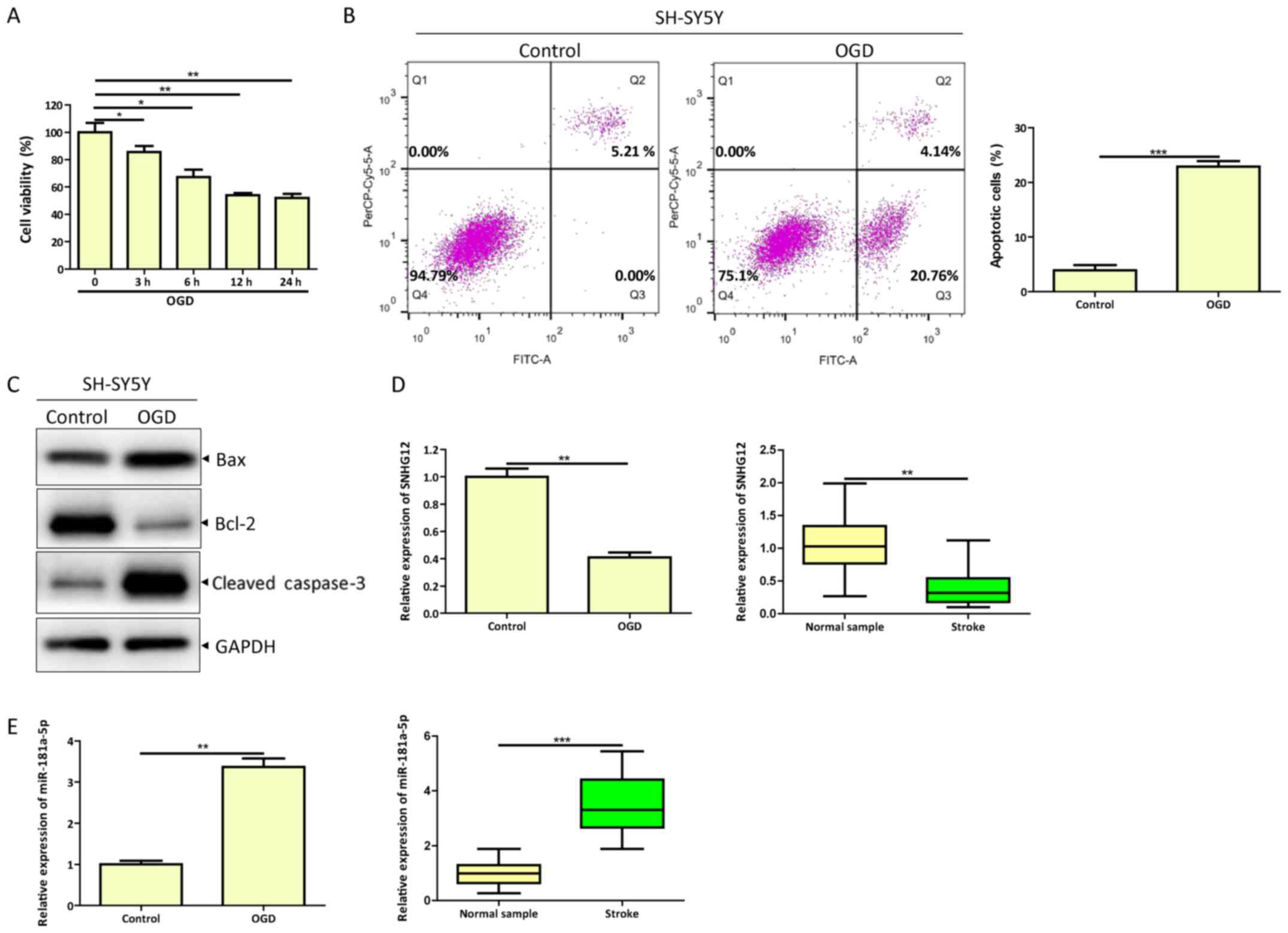
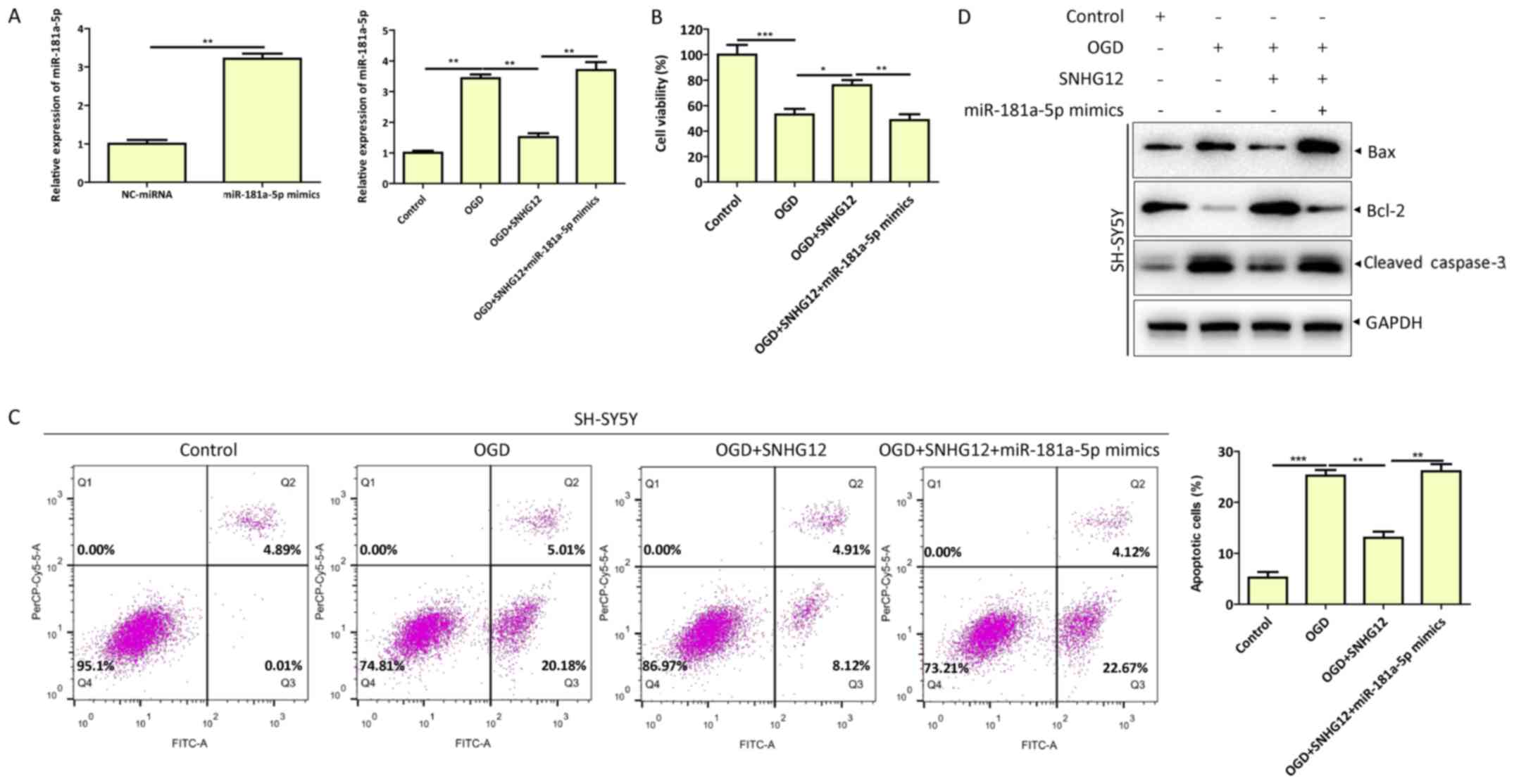
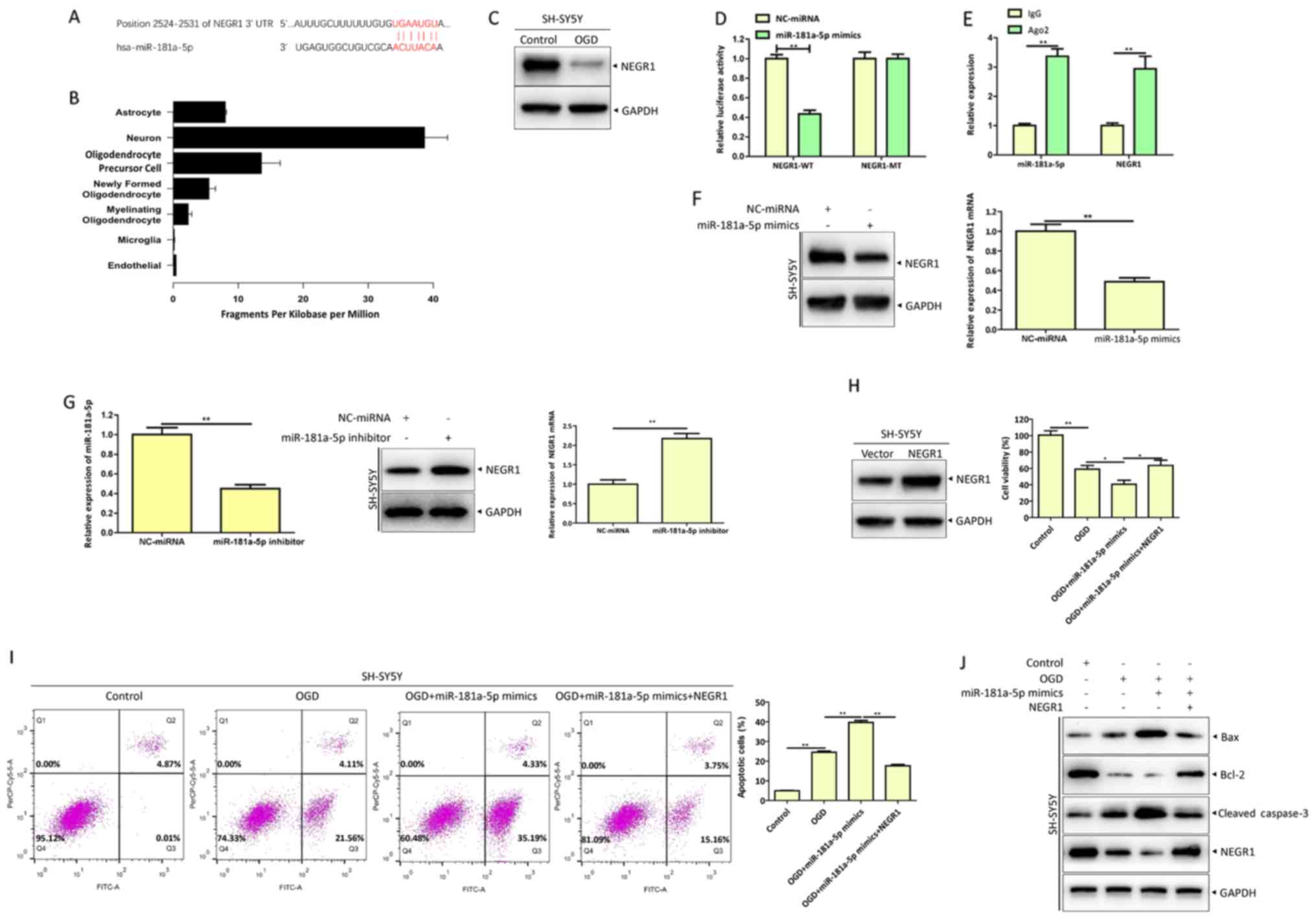
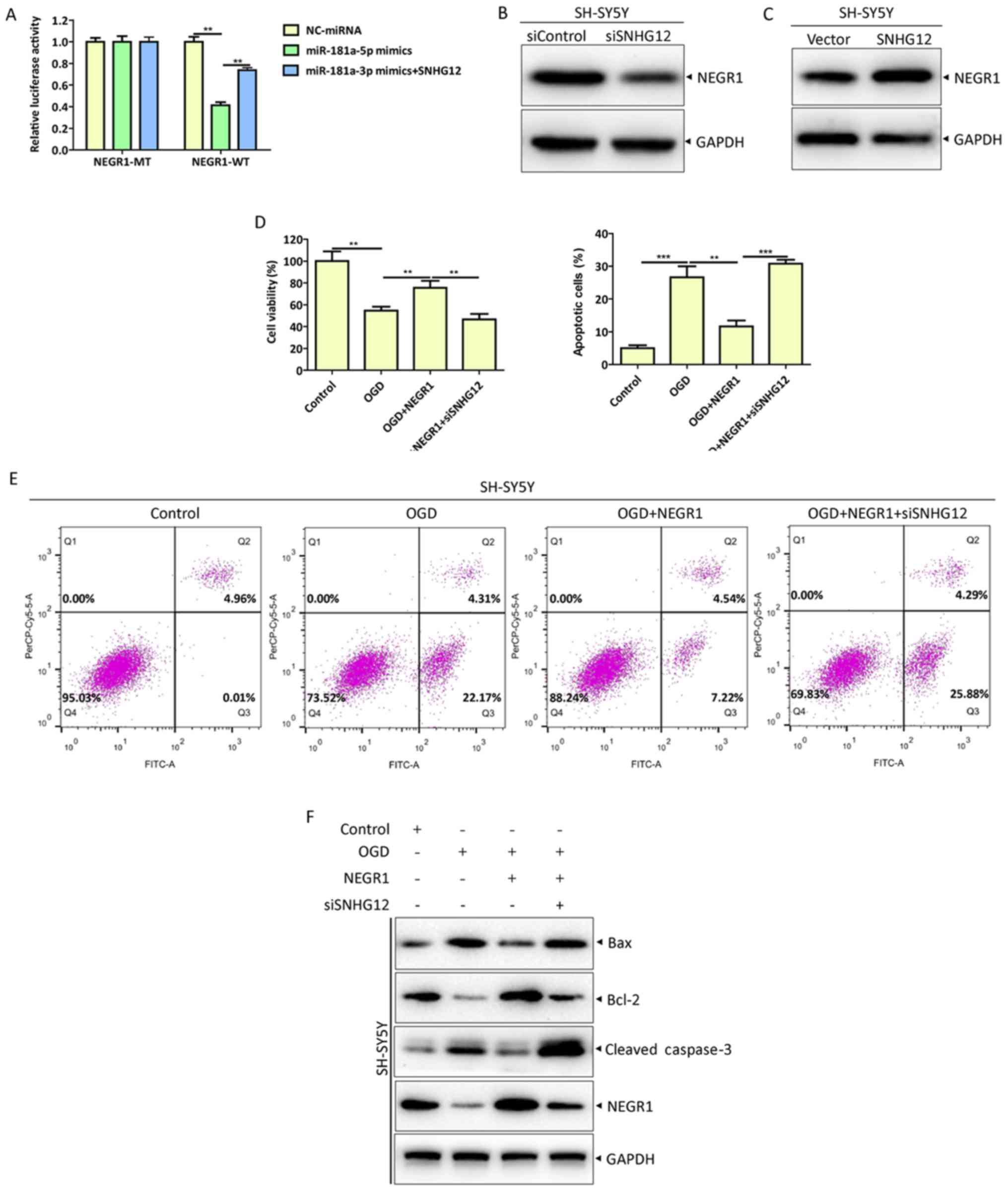
|
1
|
Katan M and Luft A: Global burden of
stroke. Semin Neurol. 38:208–211. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Wu Z, Wu P, Zuo X, Yu NA, Qin Y, Xu Q, He
S, Cen B, Liao W and Ji A: LncRNA-N1LR enhances neuroprotection
against ischemic stroke probably by inhibiting p53 phosphorylation.
Mol Neurobiol. 54:7670–7685. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Yuh WT, Alexander MD and Beauchamp NJ:
Intraarterial treatment for acute ischemic stroke. N Engl J Med.
372:1176–1179. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Ren W and Yang X: Pathophysiology of long
non-coding RNAs in ischemic stroke. Front Mol Neurosci. 11:962018.
View Article : Google Scholar : PubMed/NCBI
|
|
5
|
GBD 2016 Lifetime Risk of Stroke
Collaborators, ; Feigin VL, Nguyen G, Cercy K, Johnson CO, Alam T,
Parmar PG, Abajobir AA, Abate KH, Abd-Allah F, et al: Global,
regional, and country-specific lifetime risks of stroke, 1990 and
2016. N Engl J Med. 379:2429–2437. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Li G, Morris-Blanco KC, Lopez MS, Yang T,
Zhao H, Vemuganti R and Luo Y: Impact of microRNAs on ischemic
stroke: From pre- to post-disease. Prog Neurobiol. 163-164:59–78.
2018. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Snow SJ: Stroke and t-PA-triggering new
paradigms of care. N Engl J Med. 374:809–811. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Henderson SJ, Weitz JI and Kim PY:
Fibrinolysis: Strategies to enhance the treatment of acute ischemic
stroke. J Thromb Haemost. 16:1932–1940. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Adams BD, Parsons C, Walker L, Zhang WC
and Slack FJ: Targeting noncoding RNAs in disease. J Clin Invest.
127:761–771. 2017. View
Article : Google Scholar : PubMed/NCBI
|
|
10
|
Wang M, Yu F, Wu W, Zhang Y, Chang W,
Ponnusamy M, Wang K and Li P: Circular RNAs: A novel type of
non-coding RNA and their potential implications in antiviral
immunity. Int J Biol Sci. 13:1497–1506. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Bao MH, Szeto V, Yang BB, Zhu SZ, Sun HS
and Feng ZP: Long non-coding RNAs in ischemic stroke. Cell Death
Dis. 9:2812018. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Mehta SL, Kim T and Vemuganti R: Long
noncoding RNA FosDT promotes ischemic brain injury by interacting
with REST-associated chromatin-modifying proteins. J Neurosci.
35:16443–16449. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Lv X, Jiang H, Liu Y, Lei X and Jiao J:
MicroRNA-15b promotes neurogenesis and inhibits neural progenitor
proliferation by directly repressing TET3 during early neocortical
development. EMBO Rep. 15:1305–1314. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Tu Z, Li Y, Dai Y, Li L, Lv G, Chen I and
Wang B: MiR-140/BDNF axis regulates normal human astrocyte
proliferation and LPS-induced IL-6 and TNF-α secretion. Biomed
Pharmacother. 91:899–905. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Deng Y, Chen D, Wang L, Gao F, Jin B, Lv
H, Zhang G, Sun X, Liu L, Mo D, et al: Silencing of long noncoding
RNA nespas aggravates microglial cell death and neuroinflammation
in ischemic stroke. Stroke. 50:1850–1858. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Wei R, Zhang L, Hu W, Wu J and Zhang W:
Long non-coding RNA AK038897 aggravates cerebral
ischemia/reperfusion injury via acting as a ceRNA for miR-26a-5p to
target DAPK1. Exp Neurol. 314:100–110. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Lu Z, Luo T, Pang T, Du Z, Yin X, Cui H,
Fang G and Xue X: MALAT1 promotes gastric adenocarcinoma through
the MALAT1/miR-181a-5p/AKT3 axis. Open Biol. 9:1900952019.
View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Moon J, Xu L and Giffard RG: Inhibition of
microRNA-181 reduces forebrain ischemia-induced neuronal loss. J
Cereb Blood Flow Metab. 33:1976–1982. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Wu J, Fan CL, Ma LJ, Liu T, Wang C, Song
JX, Lv QS, Pan H, Zhang CN and Wang JJ: Distinctive expression
signatures of serum microRNAs in ischaemic stroke and transient
ischaemic attack patients. Thromb Haemost. 117:992–1001. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Cheng Y, Jiang Y, Sun Y and Jiang H: The
role of long non-coding RNA SNHG12 in neuroprotection following
cerebral ischemic injury. Neuroreport. 30:945–952. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Yin WL, Yin WG, Huang BS and Wu LX: LncRNA
SNHG12 inhibits miR-199a to upregulate SIRT1 to attenuate cerebral
ischemia/reperfusion injury through activating AMPK signaling
pathway. Neurosci Lett. 690:188–195. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Wang P, Chen D, Ma H and Li Y: LncRNA
SNHG12 contributes to multidrug resistance through activating the
MAPK/Slug pathway by sponging miR-181a in non-small cell lung
cancer. Oncotarget. 8:84086–84101. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Zhao M, Wang J, Xi X, Tan N and Zhang L:
SNHG12 promotes angiogenesis following ischemic stroke via
regulating miR-150/VEGF pathway. Neuroscience. 390:231–240. 2018.
View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Liang J, Yu Y, Wang B, Lu B, Zhang J,
Zhang H and Ge P: Ginsenoside Rb1 attenuates oxygen-glucose
deprivation-induced apoptosis in SH-SY5Y cells via protection of
mitochondria and inhibition of AIF and cytochrome c release.
Molecules. 18:12777–12792. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) method. Methods. 25:402–408. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Shang D, Xie C, Hu J, Tan J, Yuan Y, Liu Z
and Yang Z: Pancreatic cancer cell-derived exosomal microRNA-27a
promotes angiogenesis of human microvascular endothelial cells in
pancreatic cancer via BTG2. J Cell Mol Med. 24:588–604. 2020.
View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Wang X, Lan Z, He J, Lai Q, Yao X, Li Q,
Liu Y, Lai H, Gu C, Yan Q, et al: LncRNA SNHG6 promotes
chemoresistance through ULK1-induced autophagy by sponging
miR-26a-5p in colorectal cancer cells. Cancer Cell Int. 19:2342019.
View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Liu K, Xie F, Gao A, Zhang R, Zhang L,
Xiao Z, Hu Q, Huang W, Huang Q, Lin B, et al: SOX2 regulates
multiple malignant processes of breast cancer development through
the SOX2/miR-181a-5p, miR-30e-5p/TUSC3 axis. Mol Cancer. 16:622017.
View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Kaur P, Tan JR, Karolina DS, Sepramaniam
S, Armugam A, Wong PTH and Jeyaseelan K: A long non-coding RNA,
BC048612 and a microRNA, miR-203 coordinate the gene expression of
neuronal growth regulator 1 (NEGR1) adhesion protein. Biochim
Biophys Acta. 1863:533–543. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Kaur P, Tan JR, Karolina DS, Sepramaniam
S, Armugam A, Wong THP and Jeyaseelan K: Supporting data for
characterization of non-coding RNAs associated with the neuronal
growth regulator 1 (NEGR1) adhesion protein. Data Brief. 7:381–385.
2016. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Sweeney MD, Zhao Z, Montagne A, Nelson AR
and Zlokovic BV: Blood-brain barrier: From physiology to disease
and back. Physiol Rev. 99:21–78. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Liu YC, Tsai YH, Tang SC, Liou HC, Kang
KH, Liou HH, Jeng JS and Fu WM: Cytokine MIF enhances blood-brain
barrier permeability: Impact for therapy in ischemic stroke. Sci
Rep. 8:7432018. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Long FQ, Su QJ, Zhou JX, Wang DS, Li PX,
Zeng CS and Cai Y: LncRNA SNHG12 ameliorates brain microvascular
endothelial cell injury by targeting miR-199a. Neural Regen Res.
13:1919–1926. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Li Y, Guo S, Liu W, Jin T, Li X, He X,
Zhang X, Su H, Zhang N and Duan C: Silencing of SNHG12 enhanced the
effectiveness of MSCs in alleviating ischemia/reperfusion injuries
via the PI3K/AKT/mTOR signaling pathway. Front Neurosci. 13:1–12.
2019. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Ouyang YB, Lu Y, Yue S, Xu LJ, Xiong XX,
White RE, Sun X and Giffard RG: miR-181 regulates GRP78 and
influences outcome from cerebral ischemia in vitro and in vivo.
Neurobiol Dis. 45:555–563. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Sørensen SS, Nygaard AB, Carlsen AL,
Heegaard NHH, Bak M and Christensen T: Elevation of brain-enriched
miRNAs in cerebrospinal fluid of patients with acute ischemic
stroke. Biomark Res. 5:242017. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Stary CM, Xu L, Li L, Sun X, Ouyang YB,
Xiong X, Zhao J and Giffard RG: Inhibition of miR-181a protects
female mice from transient focal cerebral ischemia by targeting
astrocyte estrogen receptor-α. Mol Cell Neurosci. 82:118–125. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Han P, Li JW, Zhang BM, Lv JC, Li YM, Gu
XY, Yu ZW, Jia YH, Bai XF, Li L, et al: The lncRNA CRNDE promotes
colorectal cancer cell proliferation and chemoresistance via
miR-181a-5p-mediated regulation of Wnt/β-catenin signaling. Mol
Cancer. 16:92017. View Article : Google Scholar : PubMed/NCBI
|