Introduction
Inflammation occurs in defense against various
external insults and metabolic products, and results in erythema,
edema, fever, pain and dysfunction due to the activation of
numerous inflammatory mediators (1). It is a local protective response to
injury and infection; however, excessive or persistent inflammation
leads to chronic inflammatory diseases, including periodontitis
(2). Periodontitis is a common
disease of the oral cavity that involves chronic inflammation of
the supporting tissue around the teeth; it is characterized by
alveolar bone destruction and high concentrations of periodontal
bacteria (3). Therefore,
regulation of the inflammatory response in host cells has been
proposed as a method for controlling the progression of
periodontitis (4). Gram-negative
bacteria, including Aggregatibacter actinomycetemcomitans,
Porphyromonas gingivalis and Tannerella forsythia, are
well-known periodontitis-associated pathogens (5); they stimulate periodontal cells to
produce various inflammatory cytokines, such as interleukin (IL)1β
and IL6, and induce cellular immune inflammatory responses, thereby
destroying periodontal tissue (6).
In particular, A. actinomycetemcomitans, a facultative
anaerobic, gram-negative, rod-shaped bacterium, is a major
causative agent of localized aggressive periodontitis (7,8). It
expresses various virulence factors, including a powerful
leukotoxin, lipopolysaccharide (LPS), cell surface-associated
materials, enzymes and less well-defined virulence factors that
modulate the activity of host defenses (9–12).
Among immune cells, macrophages play a pivotal role
in inflammation by releasing inflammatory mediators, including
proinflammatory cytokines (13).
Macrophages are activated by periodontal pathogens and induce
inflammatory responses through the NF-κB pathway (14). NF-κB, a transcription factor, is
normally sequestered in the cytoplasm in a complex with NF-κB
inhibitor α (IκBα) (15).
Periodontal pathogens activate IκBα kinase (IKK)α and IKKβ via
phosphorylation, allowing them to phosphorylate IκBα. This disrupts
the stable complex between NF-κB and IκBα, enabling the
translocation of the NF-κB p65 subunit into the nucleus to activate
the transcription of proinflammatory genes (15). Consequently, inducible nitric oxide
synthase (iNOS) is expressed, and nitric oxide (NO) and
proinflammatory cytokines (16),
including IL1β and IL6, are secreted to induce inflammatory
responses (17). Thus, inhibiting
NF-κB activation is an important therapeutic goal for various
inflammatory diseases.
Inflammation is involved in the pathological
processes of a number of diseases. Recently, periodontal pathogens
have been implicated in systemic conditions, including
cardiovascular diseases, premature birth and Alzheimers disease
(18–20). Therefore, substances capable of
modulating the expression of various inflammatory mediators in
response to these pathogens are promising candidate treatments to
prevent and suppress not only periodontitis but also systemic
disease. Various antibiotics, including minocycline, doxycycline,
metronidazole and tetracycline, have been used to treat periodontal
inflammation (21). However, as
these drugs have a number of adverse effects, including
hypersensitivity, and can result in antibiotic resistance, the
development of alternative therapeutic agents is actively under way
(22).
Over the last few years, researchers have reported
the benefits of using probiotics to maintain oral health (23,24).
Probiotics are living microorganisms that confer health benefits on
their host organisms when consumed in appropriate amounts (25). Effects on oral conditions, such as
dental caries, periodontitis and bad breath, have been reported for
a few probiotics, including Streptococcus salivarius,
Lactobacillus reuteri and Weissella cibaria (26–28).
In particular, W. cibaria was first classified in a
taxonomic study in 2002 and has been denoted as the dominant
species in fermented foods, including kimchi (29). Notably, some W. cibaria
strains are reported to possess stronger immunomodulatory activity
than the commercially available strain (30,31).
The W. cibaria Chonnam Medical University
(CMU) strain (oraCMU) was isolated from the oral cavity and is an
effective oral care probiotic (32). OraCMU inhibits the growth of
periodontal pathogens and the production of proinflammatory
cytokines, including IL6 and IL8, in oral epithelial cells
(33). Furthermore, oraCMU was
recently reported to reduce periodontal tissue destruction by
regulating the production of inflammatory cytokines in a
periodontitis mouse model (34).
However, the mechanism by which oraCMU inhibits
inflammation caused by periodontal pathogens has yet to be
elucidated. The purpose of the present study was to investigate the
effects of oraCMU on the production of inflammatory mediators in
response to the periodontal pathogen A.
actinomycetemcomitans in RAW 264.7 macrophages and explore its
molecular mechanism of action.
Materials and methods
Bacterial strains and sample
preparation
A. actinomycetemcomitans (ATCC 33384,
American Type Culture Collection) was provided by the Laboratory of
Oral Biochemistry (School of Dentistry, Wonkwang University, Korea)
and oraCMU was provided by OraPharm Inc. A.
actinomycetemcomitans was grown anaerobically (85%
N2, 10% H2 and 5% CO2) in tryptic
soy broth supplemented with yeast extract (1 mg/ml) and 10% horse
serum (HyClone; Cytiva) at 37°C. Bacteria in the logarithmic growth
phase were used in the experiments. The bacteria were harvested,
washed three times with phosphate-buffered saline (PBS) and
resuspended in PBS in 10-fold concentrated volumes. To obtain
inactivated A. actinomycetemcomitans, the bacteria were
exposed to 0.5% formalin for 30 min at 4°C and inactivation was
confirmed by agar plating. The optical density (OD) of the
formalin-inactivated bacterial suspension was measured at 600 nm on
a microplate reader (Sunrise™; Tecan Group, Ltd.) and the
suspension was diluted to an OD of 1, which corresponded to
1×108 colony forming units (CFUs)/ml. To evaluate its
potential anti-inflammatory effects, oraCMU was grown in DeMan,
Rogosa and Sharpe (MRS) broth (Difco; BD Biosciences) at 37°C for
16 h under aerobic conditions. OraCMU was subcultured twice in MRS
broth before each experiment. Bacterial cultures were harvested,
washed twice with PBS and then resuspended in antibiotic-free
Dulbeccos modified Eagles medium (DMEM; Gibco; Thermo Fisher
Scientific, Inc.) containing 10% fetal bovine serum (FBS; Gibco;
Thermo Fisher Scientific, Inc.).
Cell culture
The RAW 264.7 macrophage line (TIB-71, ATCC) was
maintained in DMEM supplemented with 10% FBS and 1%
antibiotic-antimycotic solution (Gibco; Thermo Fisher Scientific,
Inc.) at 37°C in 5% CO2. The cells were subcultured and
plated at 80% confluency. Antibiotic-free DMEM medium was used for
the coculture of RAW 264.7 macrophages and live oraCMU.
Bacterial infection
To prepare live oraCMU, bacterial cultures were
harvested, washed twice with PBS and resuspended in antibiotic-free
DMEM medium. The OD was measured at 600 nm and the suspension was
diluted to obtain an OD of 0.5, which corresponded to
5×108 CFU/ml. For each experiment, RAW 264.7 macrophages
were seeded in 24-well plates at 5×105 cells/well. After
24 h, the medium was removed and the macrophages were incubated
with various doses of live oraCMU [multiplicities of infection
(MOIs) of 0.1, 1 and 10] in antibiotic-free DMEM medium and
1×107 CFU/ml A. actinomycetemcomitans cells, and
the cocultures were incubated at 37°C in 5% CO2. For
mRNA analysis, oraCMU and A. actinomycetemcomitans were
added for 4 or 6 h. For western blotting, 1×106 cells/ml
were seeded in 60-mm dishes and oraCMU and A.
actinomycetemcomitans were added for 10 min, 30 min, 1 h or 16
h.
Cell viability assays
The cytotoxicity of live oraCMU was measured using
the MTS assay (CellTiter 96® Aqueous One Solution Cell
Proliferation Assay kit; Promega Corporation). RAW 264.7
macrophages were seeded in a 96-well culture plate at
1×105 cells/well and incubated overnight at 37°C in 5%
CO2. Then, cells were treated with various
concentrations of oraCMU (MOIs =0.1, 1, 10, 100 and 1,000). After
incubation for 24 h at 37°C, the media was changed to remove almost
all oraCMU and only macrophages were left in each well. After
adding only DMEM, MTS was added to each well at a 1:5 ratio and the
plate was incubated at 37°C and 5% CO2 for 2 h. The
absorbance was measured at 490 nm on a microplate reader.
NO quantification assays
RAW 264.7 cells were seeded at 5×105
cells/well in 24-well culture plates. After 24 h at 37°C, the cells
were treated with A. actinomycetemcomitans and various
concentrations of oraCMU (MOIs =0.1, 1 and 10). Following
incubation for 24 h at 37°C, the supernatants were assessed by
mixing with the same volume of Griess reagent (Promega
Corporation). The absorbance was measured at 540 nm on a microplate
reader and the nitrite concentration was calculated using a sodium
nitrite calibration curve.
Reverse transcription-quantitative
(RTq)-PCR
Total RNA was extracted with TRIzol®
reagent (Invitrogen; Thermo Fisher Scientific, Inc.), according to
the manufacturers protocol, and quantified spectrophotometrically.
First-strand cDNA was synthesized from 1 µg of RNA using
PrimeScript RT Reagent kit (Takara Bio, Inc.). RT-qPCR was
performed on a GeneAmp PCR system 2400 (Applied Biosystems; Thermo
Fisher Scientific, Inc.) using the AccuPower PCR PreMix kit
(Bioneer Corporation). Each RT-PCR reaction used 0.5 µM of each
primer. Each cycle consisted of denaturation at 94°C (30 sec),
annealing at 55°C (30 sec) and extension at 72°C (60 sec). The
primer sequences were as follows: IL6 forward,
5′-GATGGATGCTACCAAACTGGA-3′ and reverse, 5′-TCTGAAGGACTCTGGCTTTG-3′
(142 bp); IL1β forward, 5′-GAAAGACGGCACACCCACCCT-3′ and reverse,
5′-GCTCTGCTTGTGAGGTGCTGATGTA-3′ (166 bp); and β-actin forward,
5′-CATCACTATTGGCAACGAGC-3′ and reverse, 5′-GACAGCACTGTGTTGGCATA-3′
(159 bp). The number of PCR cycles for IL6, IL1β and β-actin were
25, 25 and 28, respectively. β-actin was used as an internal
control. The amplified cDNA products were resolved on 1.5% agarose
gels. The sizes of the amplified DNA fragments were identified by
comparison with a SolGent 100 bp Plus DNA Ladder (SolGent Co.,
Ltd.). Bands were detected using an Azure cSeries (Azure
Biosystems, Inc.). Densitometry was performed using ImageJ 1.52a
software (National Institutes of Health) and normalized to the
untreated control group. Quantitative amplification of cDNA was
conducted in a StepOnePlus™ Real-Time PCR System (Applied
Biosystems; Thermo Fisher Scientific, Inc.) with PowerSYBR Green
PCR Master Mix (Applied Biosystems; Thermo Fisher Scientific,
Inc.). The RT-qPCR conditions were as follows: Incubation for 5 min
at 95°C, followed by 30 cycles of denaturation for 15 sec at 95°C,
annealing for 15 sec at 60°C and extension for 15 sec at 72°C.
Relative mRNA levels were calculated using a standard curve
generated from cDNA dilutions. The 2−ΔΔCq method was
used to calculate relative gene expression using quadruplicate
measurements, with β-actin as an internal control (35).
Western blot analysis
Cytosolic protein extracts from RAW 264.7 cells were
prepared with PhosphoSafe Protein Extraction Reagent (Novagen,
Inc.), according to the manufacturers protocol. Isolation of
nuclear fractions from RAW 264.7 cells was performed using a
nuclear extraction kit (Cayman Chemical Company). Total protein was
quantified using a bicinchoninic acid protein assay (Pierce; Thermo
Fisher Scientific, Inc.) at a wavelength of 562 nm, and 15 µg
cytosolic protein/lane and 10 µg nuclear protein/lane were resolved
by SDS-PAGE on 10% gels and transferred to Protran nitrocellulose
membranes (Whatman plc; Cytiva). Membranes were blocked with 10 mM
Tris-buffered saline with 0.1% Tween-20 (TBST) containing 5%
skimmed milk for 1 h at 25°C, followed by incubation with primary
antibodies overnight at 4°C with gentle shaking. The antibodies
used were as follows: phosphorylated (p-)IκBα kinase (IKK)α/β (cat.
no. 2697; 1:1,000; Cell Signaling Technology, Inc.), IKKα (cat. no.
2682; 1:1,000; Cell Signaling Technology, Inc.), IKKβ (cat. no.
2678; 1:1,000; Cell Signaling Technology, Inc.), p-IκBα (cat. no.
2859; 1:1,000; Cell Signaling Technology, Inc.), IκBα (cat. no.
9242; 1:1,000; Cell Signaling Technology, Inc.), NF-κB p65 subunit
(cat. no. 8242; 1:1,000; Cell Signaling Technology, Inc.), iNOS
(cat. no. 13120; 1:1,000; Cell Signaling Technology, Inc.) and
proliferating cell nuclear antigen (PCNA; cat. no. sc-56; 1:1,000;
Santa Cruz Biotechnology, Inc.). A mouse monoclonal primary
antibody against β-actin (cat. no. A5441; 1:5,000; Sigma-Aldrich;
Merck KGaA) was used as a loading control. The blots were washed in
TBST and then incubated for 2 h at room temperature with
horseradish peroxidase (HRP)-conjugated anti-rabbit (cat. no. 7074;
1:2,500; Cell Signaling Technology, Inc.) or anti-mouse IgG (cat.
no. sc-516102; 1:2,500; Santa Cruz Biotechnology, Inc.). The blots
were subsequently washed with TBST and protein bands were
visualized with HRP Substrate Luminol Reagent (EMD Millipore) and
imaged on a Chemiluminescent Western Blot Imaging System (Azure
Biosystems, Inc.). Densitometry of western blot bands was performed
using ImageJ 1.52a software (National Institutes of Health). The
detected bands were quantified using ImageJ and normalized to the
untreated control group.
Statistical analysis
Statistical analyses were performed using SPSS v17.0
(SPSS, Inc.). Data are presented as the mean ± standard deviation
of the mean. The Kruskal-Wallis test followed by Dunns post hoc
test was used to compare the different groups. P<0.05 was
considered to indicate a statistically significant difference. Each
experiment was performed three times.
Results
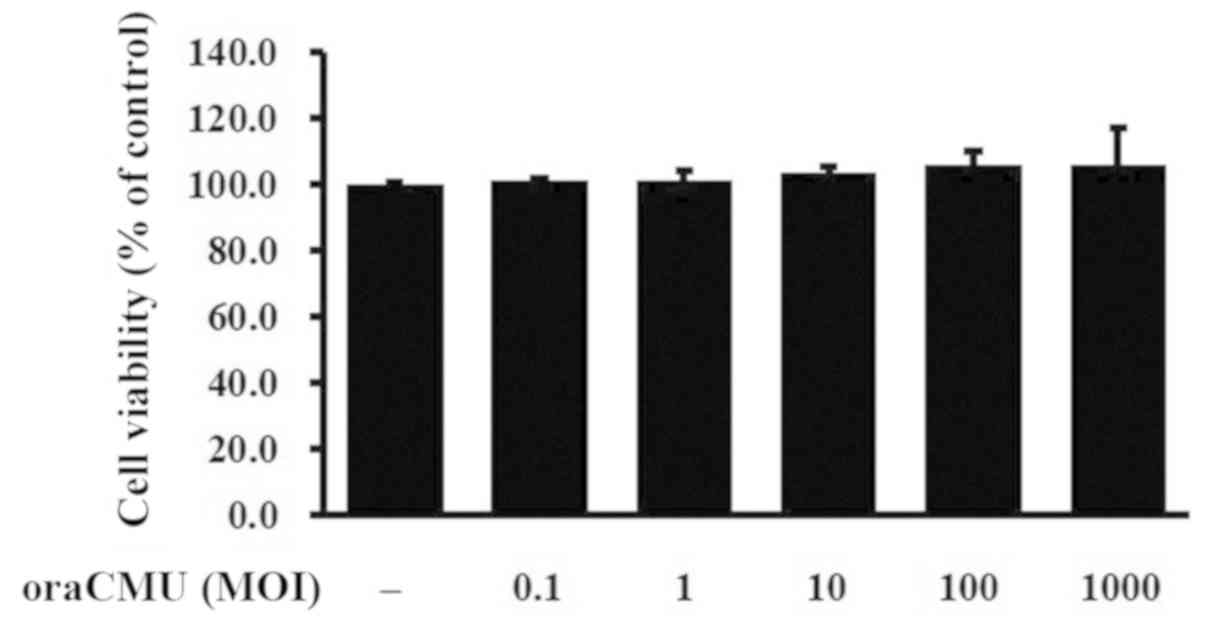
Cytotoxic effects of oraCMU on RAW
264.7 macrophages
To assess the cytotoxicity of live oraCMU, its
effects on the viability of RAW 264.7 macrophages were examined. No
cytotoxic effects were detected after 24 h of treatment at various
concentrations (Fig. 1).
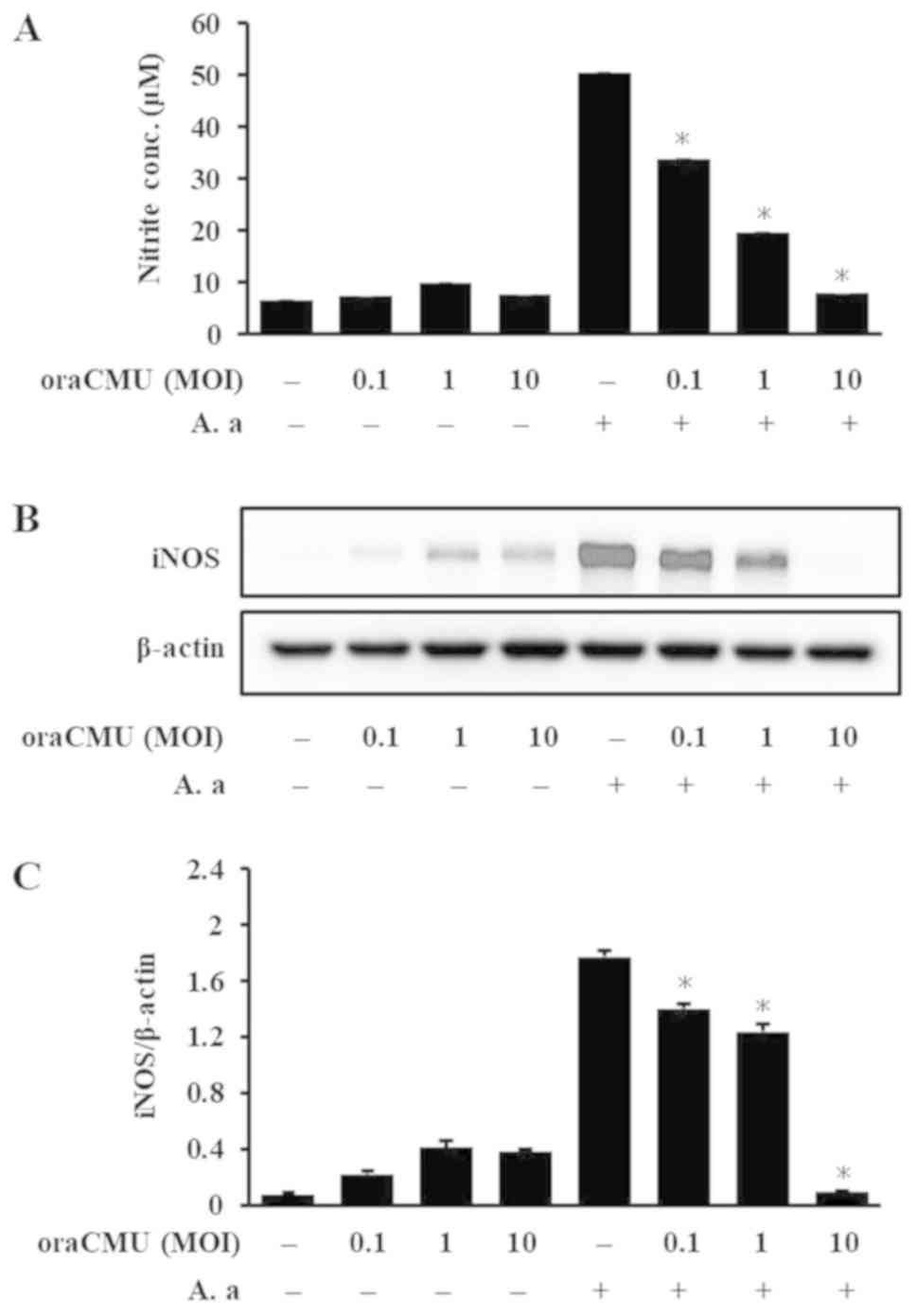
Inhibitory effects of oraCMU on NO
production and iNOS expression
To investigate whether oraCMU possesses
anti-inflammatory effects against A. actinomycetemcomitans
in RAW 264.7 macrophages, its effects on NO production were
examined. After 24 h of A. actinomycetemcomitans treatment,
NO release was higher compared with that in the untreated controls
(Fig. 2A). Treatment with oraCMU
significantly decreased A. actinomycetemcomitans-induced NO
production in a dose-dependent manner. Changes in NO production can
be attributed to changes in iNOS expression (30). Treatment with A.
actinomycetemcomitans significantly increased iNOS expression
in RAW 264.7 macrophages and oraCMU treatment significantly
decreased iNOS expression in a dose-dependent manner (Fig. 2B and C).
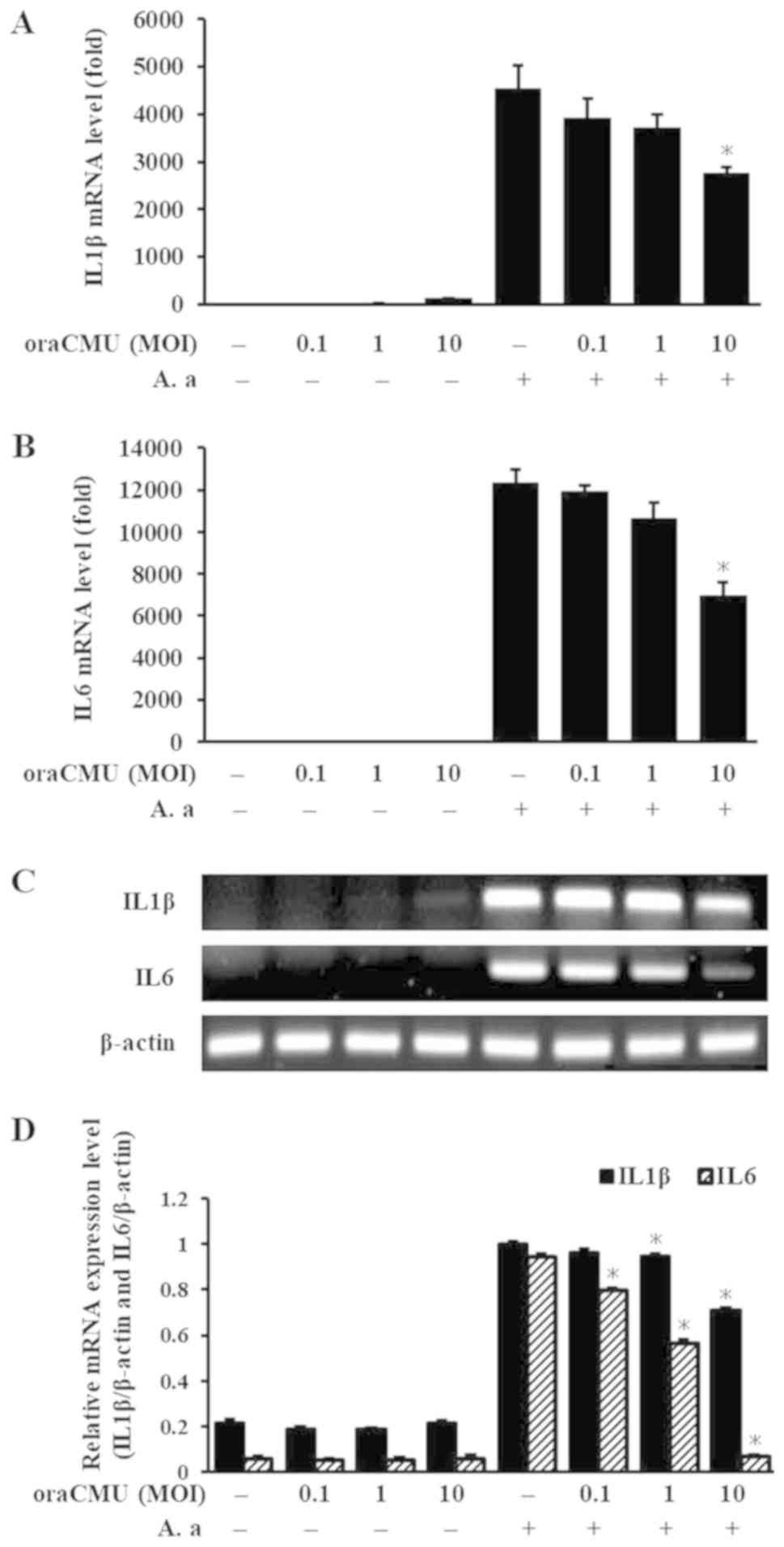
Inhibitory effects of oraCMU on the
mRNA expression of proinflammatory cytokines
To determine whether oraCMU modulates the mRNA
expression of proinflammatory cytokines, RAW 264.7 macrophages were
incubated with A. actinomycetemcomitans and various
concentrations of oraCMU. IL6 and IL1β increased significantly with
A. actinomycetemcomitans treatment and were significantly
decreased at higher doses of oraCMU (Fig. 3).
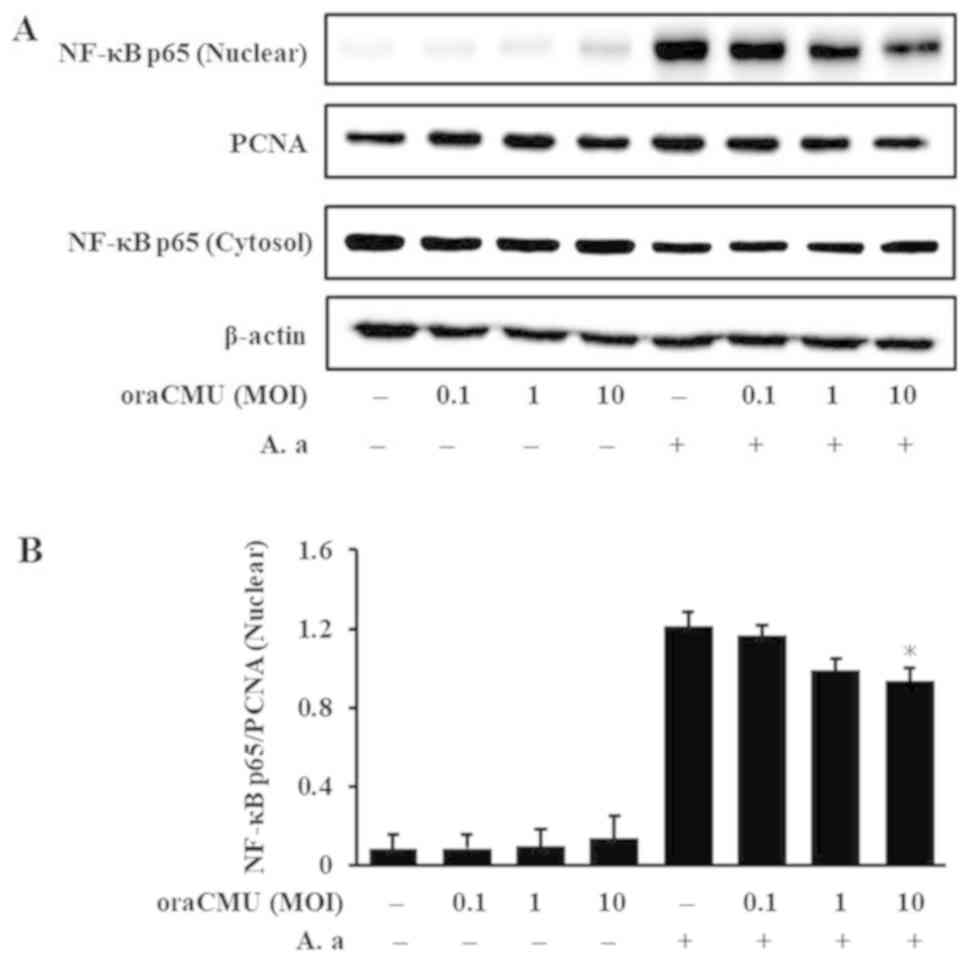
Inhibitory effects of oraCMU on NF-κB
activation
As the NF-κB pathway plays an important role in the
transcriptional activation of proinflammatory factors, the effects
of oraCMU on NF-κB activation were next assessed by examining p65
levels in cytosolic and nuclear extracts from macrophages treated
with A. actinomycetemcomitans and oraCMU for 30 min or 1 h.
A. actinomycetemcomitans alone resulted in increased nuclear
p65, whereas oraCMU treatment significantly inhibited the nuclear
accumulation of p65 at high doses (Fig. 4).
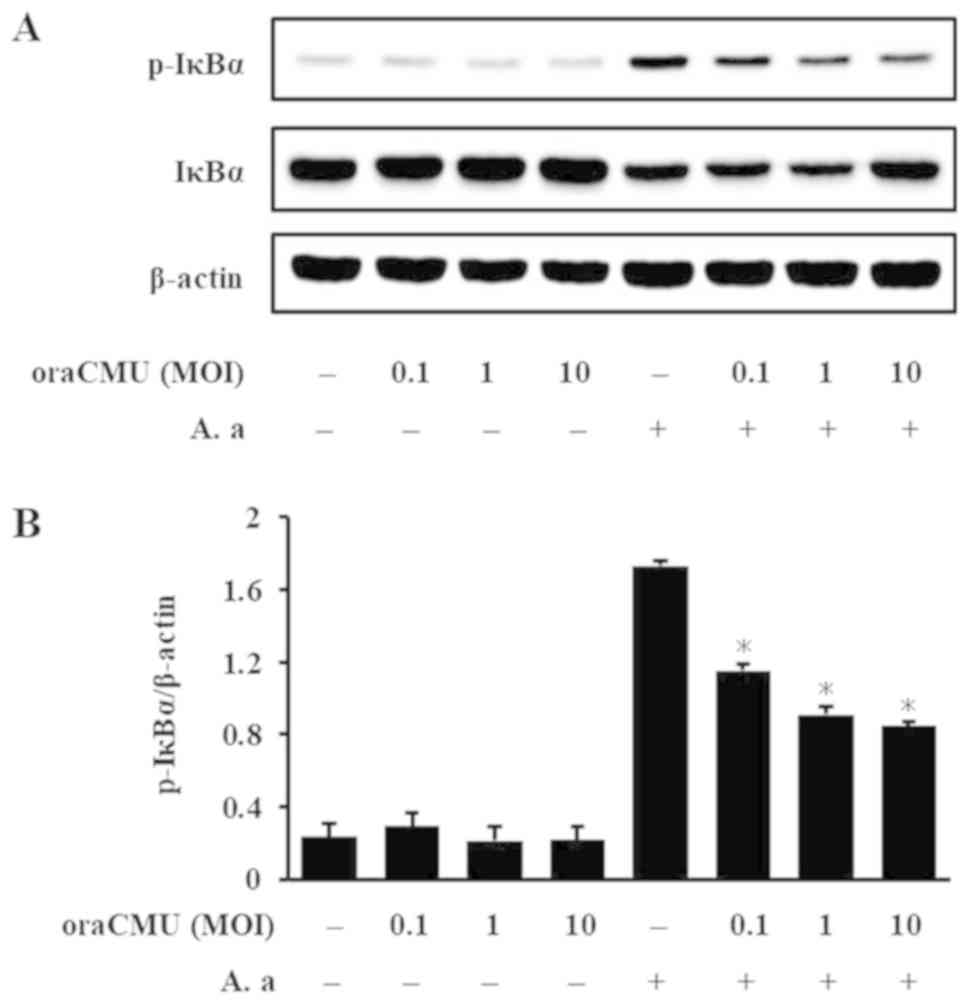
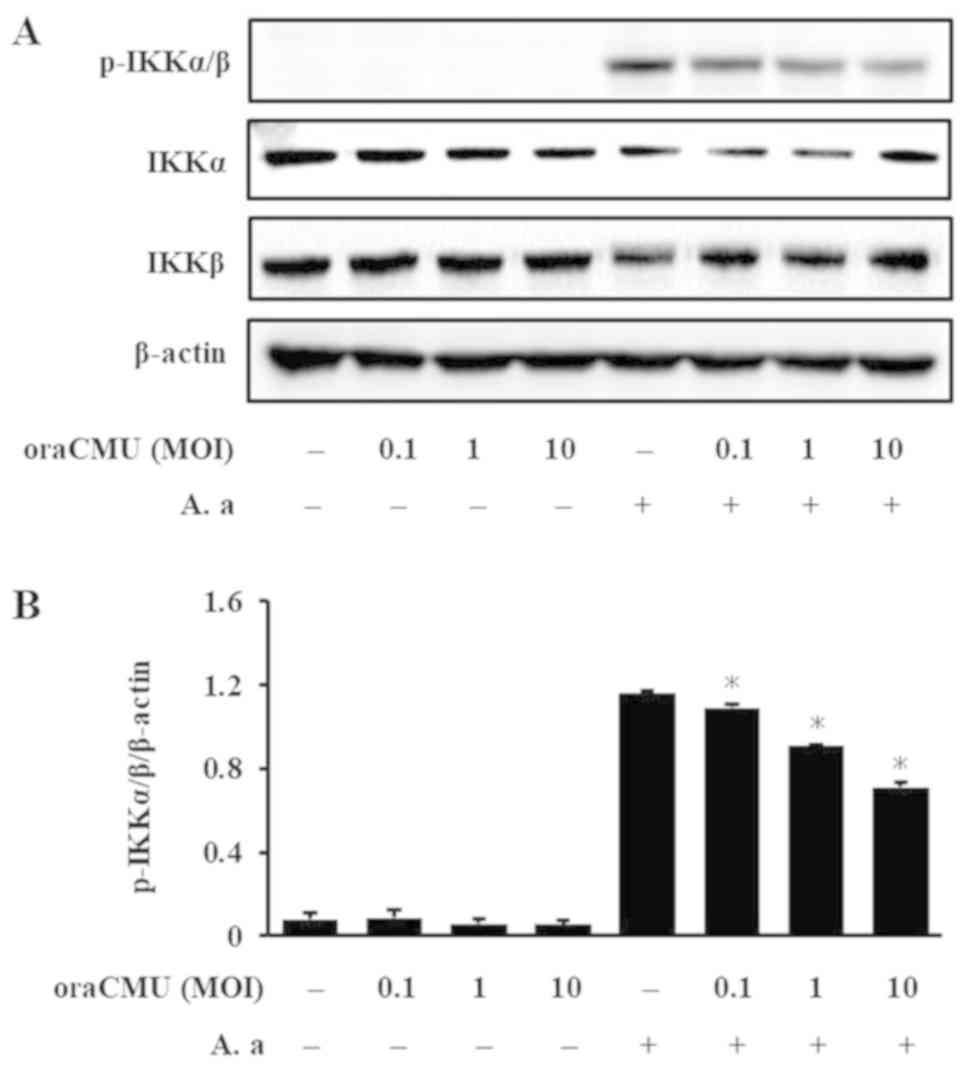
Inhibitory effects of oraCMU on A.
actinomycetemcomitans-induced IκBα and IKKα/β phosphorylation
As IκBα and IKKα/β are important regulators of NF-κB
activation, their activation after oraCMU treatment of A.
actinomycetemcomitans-stimulated macrophages were evaluated.
After 30 min, OraCMU significantly inhibited IκBα phosphorylation
in a dose-dependent manner (Fig.
5). In addition, after 10 min, oraCMU dose-dependently
inhibited IKKα/β phosphorylation (Fig.
6).
Discussion
OraCMU is the first commercialized oral care
probiotic in Korea (32) that can
help prevent bad breath and dental caries (28,36,37).
It inhibits the Fusobacterium nucleatum-induced increase of the
proinflammatory cytokines IL6 and IL8 in oral epithelial cells
(33) and has antimicrobial
activity against various representative periodontal pathogens,
including A. actinomycetemcomitans (38). Therefore, oraCMU may aid in
preventing periodontal disease. However, its inhibitory effects on
proinflammatory cytokine expression in macrophages stimulated with
A. actinomycetemcomitans, a periodontal pathogen, have not
yet been reported. To the best of our knowledge, the present study
is the first to elucidate the mechanism by which W. cibaria
inhibits inflammatory cytokine expression after infection with
periodontal pathogens.
The present study evaluated whether live oraCMU had
inhibitory effects on the inflammation induced by
formalin-inactivated A. actinomycetemcomitans in RAW 264.7
cells. The A. actinomycetemcomitans-induced inflammatory
response was characterized by increased NO production and increased
iNOS, IL1β and IL6 expression; oraCMU decreased the levels of these
proinflammatory mediators. To exclude the possibility that
cytotoxicity caused by live oraCMU infection was responsible for
the inhibition of the proinflammatory mediators, the viability of
oraCMU-infected cells was tested. No obvious cytotoxic effects were
detected at any MOI used, consistent with the results of our
previous study (33).
NO is an important biomarker of the inflammatory
response and is regulated by iNOS (16). The iNOS enzyme cannot be detected
under normal conditions but is induced through NF-κB activation,
leading to excessive NO production. Excessive NO leads to the
upregulation of other proinflammatory cytokines and can cause
malfunctions ranging from severe cellular damage to inflammatory
disorders (39). Thus, regulating
iNOS expression is an important strategy in the development of
inflammatory disease therapies. The present study found that oraCMU
significantly decreased A. actinomycetemcomitans-induced NO
production and downregulated iNOS expression in a dose-dependent
manner, suggesting that oraCMU acts as an anti-inflammatory
regulator.
Madeira et al (40), found that A.
actinomycetemcomitans LPS plays an important role in alveolar
bone loss. They also demonstrated that it can induce NO production
in murine macrophages. The present study used inactivated A.
actinomycetemcomitans as a trigger instead of its LPS and found
that live oraCMU decreased A. actinomycetemcomitans-induced
NO production in RAW 264.7 cells by inhibiting iNOS at the mRNA
level. Similarly, Yu et al (31), reported that treatment with
heat-inactivated W. cibaria JW15 decreased NO production in
RAW 264.7 cells upon LPS stimulation, which was attributable to
downregulated iNOS expression.
NF-κB regulates the expression of iNOS and other
proinflammatory factors (16,17).
In the present study, A. actinomycetemcomitans stimulation
increased p65 levels in the nucleus; this was inhibited by high
doses of oraCMU, suggesting that it can inactivate NF-κB. In
addition, it inhibited IκBα and IKKα/β phosphorylation in a
dose-dependent manner. These results suggested that oraCMU blocks
the expression of proinflammatory mediators by inhibiting the
classical NF-κB pathway.
The proinflammatory cytokines IL1β and IL6 are
representative diagnostic markers that provide information about
the progression of periodontal disease (17). Their expression is higher at sites
of periodontal inflammation and is closely associated with the
clinical severity of periodontitis. IL6 secretion, stimulated by
exposure to IL1β, is involved in the periodontal tissue destruction
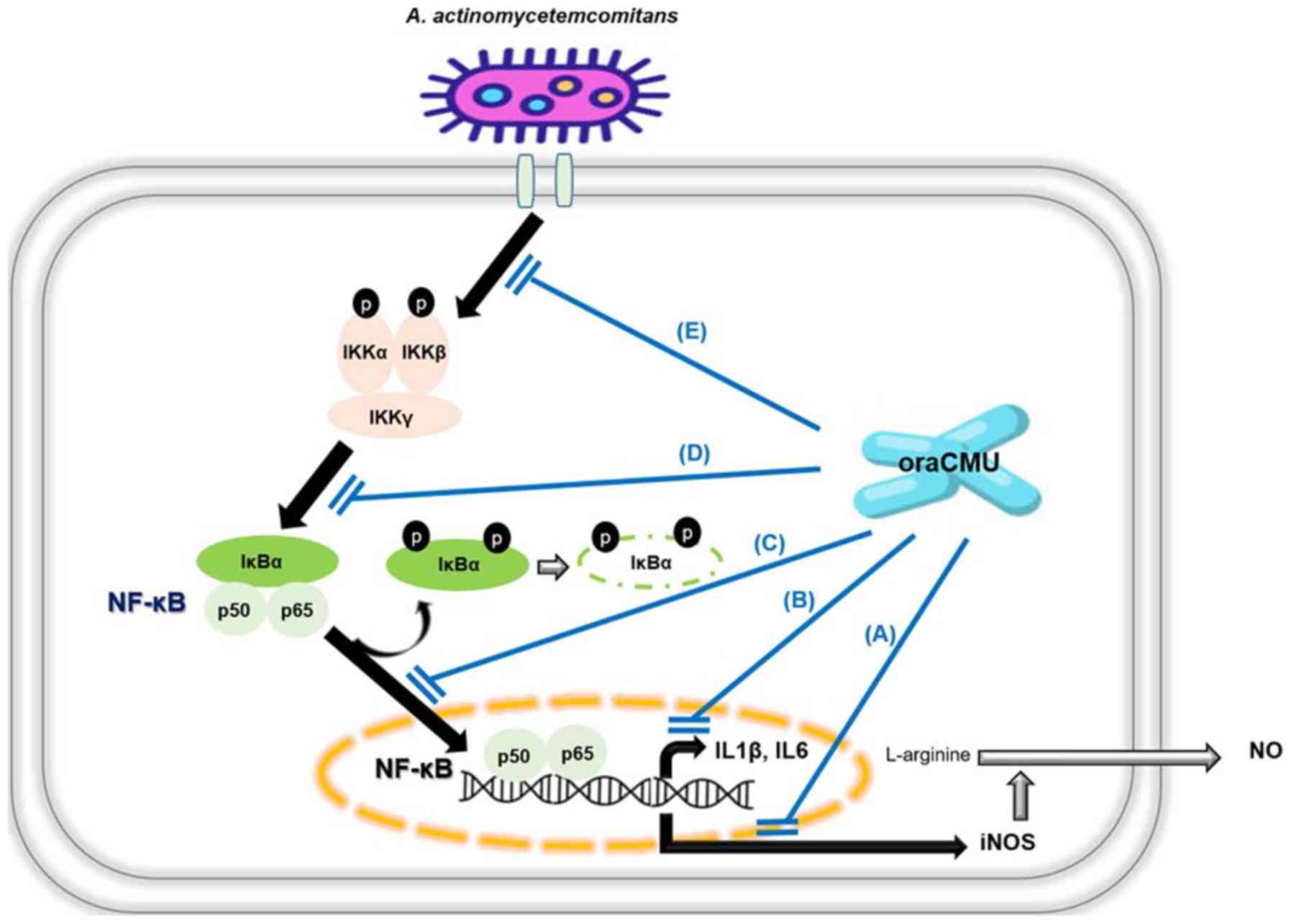
that occurs in periodontitis (17). In the present study, live oraCMU
displayed dose-dependent anti-inflammatory activities in
macrophages activated by A. actinomycetemcomitans by
inhibiting NF-κB signaling (Fig.
7). Heat-inactivated W. cibaria JW15 has also been shown
to suppress IL1β and IL6 expression; moreover, mechanistically, its
anti-inflammatory properties are mediated by mitogen-activated
protein kinase signaling and result in NF-κB inhibition (31).
Probiotics are viable microorganisms that have a
number of health benefits, which includes regulating intestinal
microbial balance and exerting immune-modulating effects on the
host through colonization of the intestinal microflora (25). Since the composition of bacterial
surface molecules, including amino acid residues, disaccharide
ratio and differences in cross-link type, are different between
microbes, microbe-mediated immune responses are probiotic
strain-specific (23–25). The present study simultaneously
inoculated macrophages with formalin-inactivated A.
actinomycetemcomitans and live bacteria oraCMU, and then
confirmed the mechanism of action of A.
actinomycetemcomitans-induced inflammatory cytokine expression
via cell signaling. Probiotics and periodontal pathogens were
evaluated by direct contact with macrophages.
Metabolites and altered surroundings produced by
probiotics can affect the inhibition of inflammatory cytokine
expression in macrophages, but do not exert inhibitory effects due
to the use of inactivated A. actinomycetemcomitans. Since
oraCMU is commercially used as a living bacterium, the live
bacterium oraCMU was used in this study. As oraCMU is an anaerobic
bacterium, it was expected to be effective because it can grow well
under anaerobic conditions when it enters the periodontal
pocket.
Previous studies on W. cibaria strain derived
from Kimchi, a fermented food, have focused on intestinal immunity
(30,31). However, previous studies have
reported that probiotics work in the oral cavity and have
beneficial effects (24–28). The present study is novel in that
the W. cibaria strain, derived from the saliva of children
with healthy mouths, was investigated for the prevention of
proinflammatory responses by A. actinomycetemcomitans and
for its cellular signaling mechanism.
In conclusion, the present study demonstrated the
anti-inflammatory effects of the oral cavity-derived probiotic
oraCMU, indicating its usefulness as a prophylactic oral probiotic.
OraCMU inhibited proinflammatory signaling in A.
actinomycetemcomitans-induced macrophages by blocking NF-κB
activation, resulting in decreased phosphorylation of IKKs and
IκBα, decreased translocation of p65 to the nucleus and decreased
expression of iNOS, IL1β and IL6. Although further in vivo
research will be required to confirm the anti-inflammatory effects
of this strain, these results provide molecular evidence for the
immunomodulatory effects of oraCMU. Overall, the findings of
present study indicated that oraCMU could be used to develop oral
care probiotics that can aid in the prevention of periodontal
disease. The effects of live probiotics were investigated in the
present study. Comparative studies on live and dead oraCMU in a
further study might be meaningful and should be considered in
further research.
Acknowledgements
Not applicable.
Funding
The present study was supported by the Basic Science
Research Program of the National Research Foundation of Korea (NRF)
funded by the Ministry of Education (grant no.
2017R1D1A1B03030952).
Availability of data and materials
The datasets used and/or analyzed during the present
study are available from the corresponding author on reasonable
request.
Authors contributions
MJK and JYK performed most of the experiments. YOY
and HJK analyzed the data. HJK and MSK designed the study and wrote
the manuscript. All authors read and approved the final manuscript,
and agree to be accountable for all aspects of the research and
ensure that the accuracy or integrity of any part of the work are
appropriately investigated and resolved.
Ethics approval and consent to
participate
Not applicable.
Patient consent for publication
Not applicable.
Competing interests
MSK is employed by OraPharm Inc. The other authors
declare that they have no competing interests.
References
|
1
|
Wojdasiewicz P, Poniatowski ŁA and
Szukiewicz D: The role of inflammatory and anti-inflammatory
cytokines in the pathogenesis of osteoarthritis. Mediators Inflamm.
2014:5614592014. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Kinane DF, Stathopoulou PG and Papapanou
PN: Periodontal diseases. Nat Rev Dis Primers. 3:170382017.
View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Cochran DL: Inflammation and bone loss in
periodontal disease. J Periodontol. 79 (Suppl):1569–1576. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Silva N, Abusleme L, Bravo D, Dutzan N,
Garcia-Sesnich J, Vernal R, Hernández M and Gamonal J: Host
response mechanisms in periodontal diseases. J Appl Oral Sci.
23:329–355. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Sánchez GA, Acquier AB, De Couto A, Busch
L and Mendez CF: Association between Aggregatibacter
actinomycetemcomitans and Porphyromonas gingivalis in subgingival
plaque and clinical parameters, in Argentine patients with
aggressive periodontitis. Microb Pathog. 82:31–36. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Graves D: Cytokines that promote
periodontal tissue destruction. J Periodontol. 79
(Suppl):1585–1591. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Gholizadeh P, Pormohammad A, Eslami H,
Shokouhi B, Fakhrzadeh V and Kafil HS: Oral pathogenesis of
Aggregatibacter actinomycetemcomitans. Microb Pathog. 113:303–311.
2017. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Cheng Z, Meade J, Mankia K, Emery P and
Devine DA: Periodontal disease and periodontal bacteria as triggers
for rheumatoid arthritis. Best Pract Res Clin Rheumatol. 31:19–30.
2017. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Brage M, Holmlund A and Johansson A:
Humoral immune response to Aggregatibacter actinomycetemcomitans
leukotoxin. J Periodontal Res. 46:170–175. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Singh S, Bhatia R, Singh A, Singh P, Kaur
R, Khare P, Purama RK, Boparai RK, Rishi P, Ambalam P, et al:
Probiotic attributes and prevention of LPS-induced pro-inflammatory
stress in RAW264.7 macrophages and human intestinal epithelial cell
line (Caco-2) by newly isolated Weissella cibaria strains. Food
Funct. 9:1254–1264. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Weidenmaier C, Kristian SA and Peschel A:
Bacterial resistance to antimicrobial host defenses--an emerging
target for novel antiinfective strategies? Curr Drug Targets.
4:643–649. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Nau GJ, Richmond JF, Schlesinger A,
Jennings EG, Lander ES and Young RA: Human macrophage activation
programs induced by bacterial pathogens. Proc Natl Acad Sci USA.
99:1503–1508. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Turner MD, Nedjai B, Hurst T and
Pennington DJ: Cytokines and chemokines: At the crossroads of cell
signalling and inflammatory disease. Biochim Biophys Acta.
1843:2563–2582. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Choi EY, Jin JY, Lee JY, Choi JI, Choi IS
and Kim SJ: Anti-inflammatory effects and the underlying mechanisms
of action of daidzein in murine macrophages stimulated with
Prevotella intermedia lipopolysaccharide. J Periodontal Res.
47:204–211. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Liu T, Zhang L, Joo D and Sun SC: NF-κB
signaling in inflammation. Signal Transduct Target Ther. doi:
10.1038/sigtrans.2017.23.
|
|
16
|
Aktan F: iNOS-mediated nitric oxide
production and its regulation. Life Sci. 75:639–653. 2004.
View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Chen CC, Chang KL, Huang JF, Huang JS and
Tsai CC: Correlation of interleukin-1 beta, interleukin-6, and
periodontitis. Kaohsiung J Med Sci. 13:609–617. 1997.PubMed/NCBI
|
|
18
|
Carrizales-Sepúlveda EF, Ordaz-Farías A,
Vera-Pineda R and Flores-Ramírez R: Periodontal disease, systemic
inflammation and the risk of cardiovascular disease. Heart Lung
Circ. 27:1327–1334. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Parihar AS, Katoch V, Rajguru SA, Rajpoot
N, Singh P and Wakhle S: Periodontal disease: A possible
risk-factor for adverse pregnancy outcome. J Int Oral Health.
7:137–142. 2015.PubMed/NCBI
|
|
20
|
Ganesh P, Karthikeyan R, Muthukumaraswamy
A and Anand J: A potential role of periodontal inflammation in
Alzheimers disease: A Review. Oral Health Prev Dent. 15:7–12.
2017.PubMed/NCBI
|
|
21
|
Da Rocha HA, Silva CF, Santiago FL,
Martins LG, Dias PC and De Magalhães D: Local drug delivery systems
in the treatment of periodontitis: A literature review. J Int Acad
Periodontol. 17:82–90. 2015.PubMed/NCBI
|
|
22
|
Blumenthal KG, Peter JG, Trubiano JA and
Phillips EJ: Antibiotic allergy. Lancet. 393:183–198. 2019.
View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Gupta G: Probiotics and periodontal
health. J Med Life. 4:387–394. 2011.PubMed/NCBI
|
|
24
|
Saha S, Tomaro-Duchesneau C, Tabrizian M
and Prakash S: Probiotics as oral health biotherapeutics. Expert
Opin Biol Ther. 12:1207–1220. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Reid G, Jass J, Sebulsky MT and McCormick
JK: Potential uses of probiotics in clinical practice. Clin
Microbiol Rev. 16:658–672. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Burton JP, Drummond BK, Chilcott CN, Tagg
JR, Thomson WM, Hale JDF and Wescombe PA: Influence of the
probiotic Streptococcus salivarius strain M18 on indices of dental
health in children: A randomized double-blind, placebo-controlled
trial. J Med Microbiol. 62:875–884. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Szkaradkiewicz AK, Stopa J and Karpiński
TM: Effect of oral administration involving a probiotic strain of
Lactobacillus reuteri on pro-inflammatory cytokine response in
patients with chronic periodontitis. Arch Immunol Ther Exp (Warsz).
62:495–500. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Kang MS, Kim BG, Chung J, Lee HC and Oh
JS: Inhibitory effect of Weissella cibaria isolates on the
production of volatile sulphur compounds. J Clin Periodontol.
33:226–232. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Björkroth KJ, Schillinger U, Geisen R,
Weiss N, Hoste B, Holzapfel WH, Korkeala HJ and Vandamme P:
Taxonomic study of Weissella confusa and description of
Weissella cibaria sp. nov., detected in food and clinical
samples. Int J Syst Evol Microbiol. 52:141–148. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Park HE, Kang KW, Kim BS, Lee SM and Lee
WK: Immunomodulatory potential of Weissella cibaria in aged
C57BL/6J mice. J Microbiol Biotechnol. 27:2094–2103. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Yu HS, Lee NK, Choi AJ, Choe JS, Bae CH
and Paik HD: Anti-inflammatory potential of probiotic strain
Weissella cibaria JW15 isolated from Kimchi through regulation of
NF-κB and MAPKs pathways in LPS-induced RAW 264.7 cells. J
Microbiol Biotechnol. 29:1022–1032. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Jang HJ, Kang MS, Yi SH, Hong JY and Hong
SP: Comparative study on the characteristics of Weissella cibaria
CMU and probiotic strains for oral care. Molecules. 21:212016.
View Article : Google Scholar
|
|
33
|
Kang MS, Lim HS, Kim SM, Lee HC and Oh JS:
Effect of Weissella cibaria on Fusobacterium
nucleatum-induced interleukin-6 and interleukin-8 production in KB
cells. J Bacteriol Virol. 41:9–18. 2011. View Article : Google Scholar
|
|
34
|
Kim JW, Jung BH, Lee JH, Yoo KY, Lee H,
Kang MS and Lee JK: Effect of Weissella cibaria on the reduction of
periodontal tissue destruction in mice. J Periodontol.
doi:10.1002/JPER.19-0288.
|
|
35
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(−ΔΔC(T)) method. Methods. 25:402–408. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Kang MS, Chung J, Kim SM, Yang KH and Oh
JS: Effect of Weissella cibaria isolates on the formation of
Streptococcus mutans biofilm. Caries Res. 40:418–425. 2006.
View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Park HR, Kim HJ and Kang MS: Clinical
studies on the dental caries prevention effects of the ability of
Weissella cibaria CMU to adhere to the oral cavity. Indian J Public
Health Res Dev. 9:1163–1169. 2018. View Article : Google Scholar
|
|
38
|
Asok A, Bhandary R, Shetty M and Shenoy
MS: Probiotics and periodontal disease. Int J Oral Health Sci.
8:68–72. 2018. View Article : Google Scholar
|
|
39
|
Baron VT, Pio R, Jia Z and Mercola D:
Early growth response 3 regulates genes of inflammation and
directly activates IL6 and IL8 expression in prostate cancer. Br J
Cancer. 112:755–764. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Madeira MF, Queiroz-Junior CM, Cisalpino
D, Werneck SM, Kikuchi H, Fujise O, Ryffel B, Silva TA, Teixeira MM
and Souza DG: MyD88 is essential for alveolar bone loss induced by
Aggregatibacter actinomycetemcomitans lipopolysaccharide in mice.
Mol Oral Microbiol. 28:415–424. 2013. View Article : Google Scholar : PubMed/NCBI
|