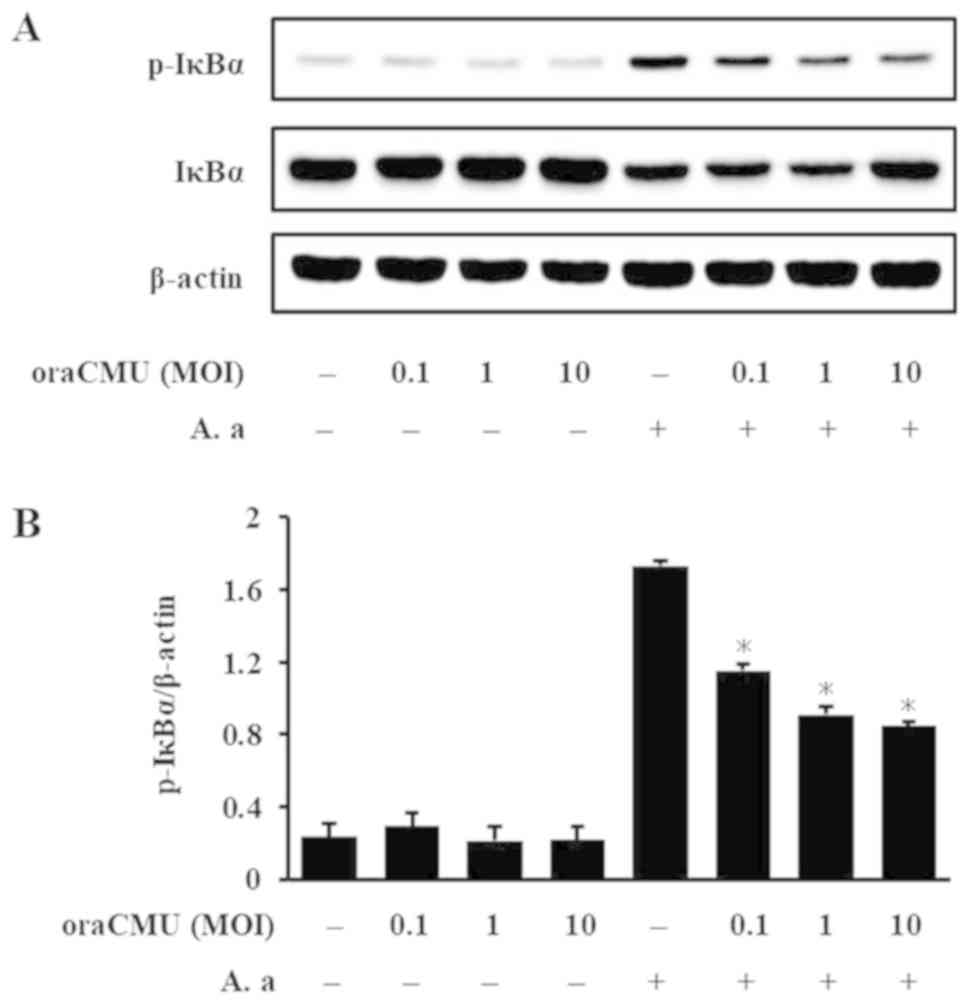
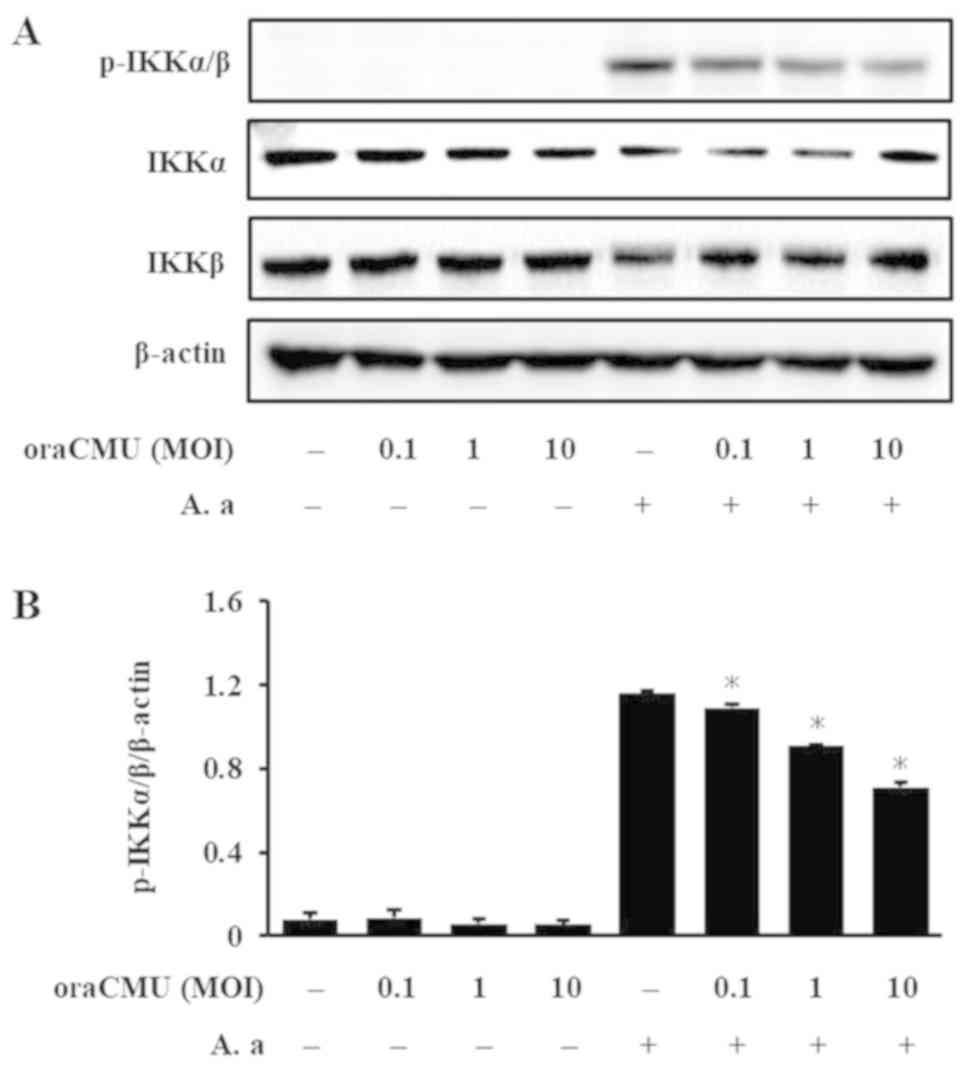
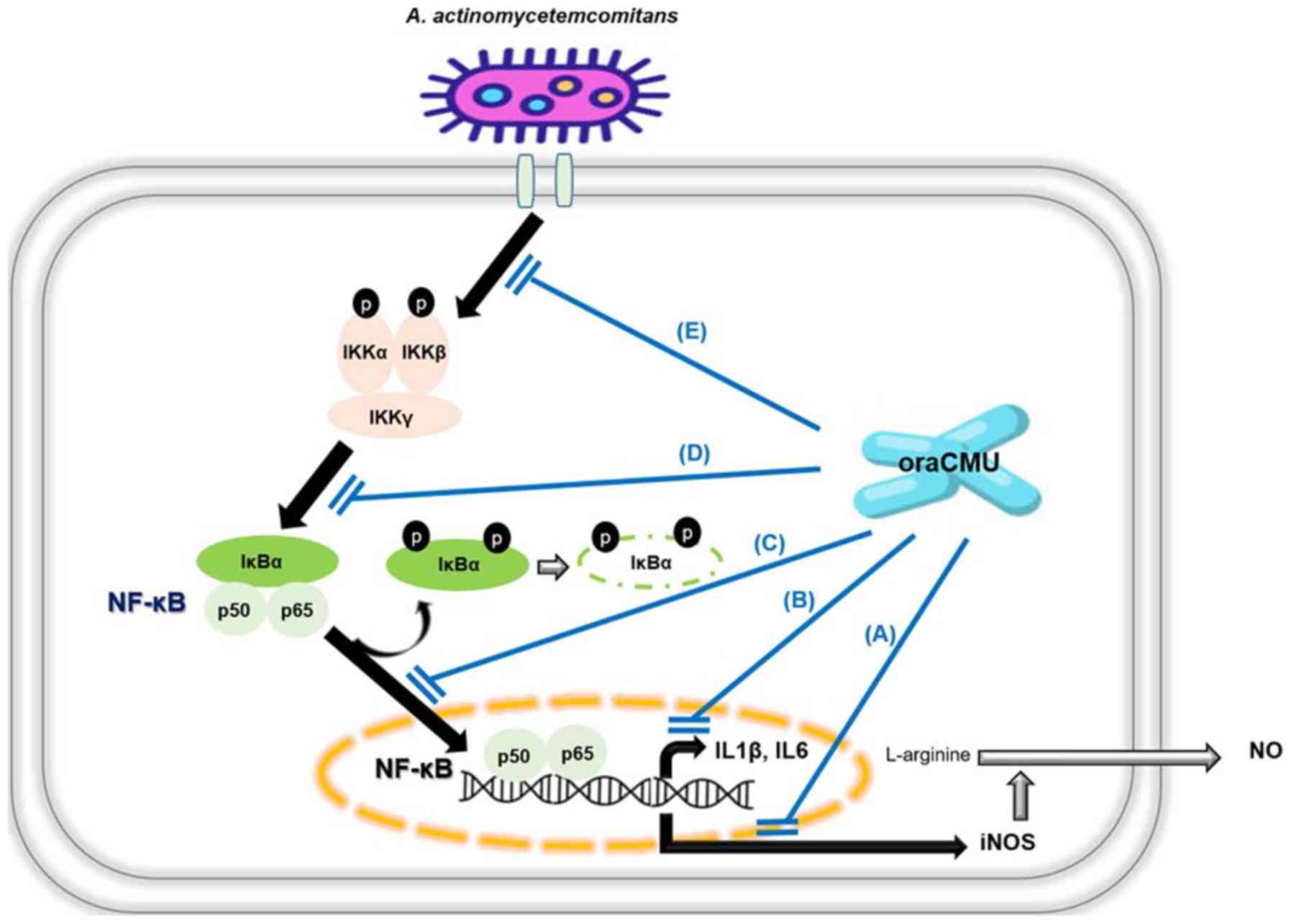
|
1
|
Wojdasiewicz P, Poniatowski ŁA and
Szukiewicz D: The role of inflammatory and anti-inflammatory
cytokines in the pathogenesis of osteoarthritis. Mediators Inflamm.
2014:5614592014. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Kinane DF, Stathopoulou PG and Papapanou
PN: Periodontal diseases. Nat Rev Dis Primers. 3:170382017.
View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Cochran DL: Inflammation and bone loss in
periodontal disease. J Periodontol. 79 (Suppl):1569–1576. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Silva N, Abusleme L, Bravo D, Dutzan N,
Garcia-Sesnich J, Vernal R, Hernández M and Gamonal J: Host
response mechanisms in periodontal diseases. J Appl Oral Sci.
23:329–355. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Sánchez GA, Acquier AB, De Couto A, Busch
L and Mendez CF: Association between Aggregatibacter
actinomycetemcomitans and Porphyromonas gingivalis in subgingival
plaque and clinical parameters, in Argentine patients with
aggressive periodontitis. Microb Pathog. 82:31–36. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Graves D: Cytokines that promote
periodontal tissue destruction. J Periodontol. 79
(Suppl):1585–1591. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Gholizadeh P, Pormohammad A, Eslami H,
Shokouhi B, Fakhrzadeh V and Kafil HS: Oral pathogenesis of
Aggregatibacter actinomycetemcomitans. Microb Pathog. 113:303–311.
2017. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Cheng Z, Meade J, Mankia K, Emery P and
Devine DA: Periodontal disease and periodontal bacteria as triggers
for rheumatoid arthritis. Best Pract Res Clin Rheumatol. 31:19–30.
2017. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Brage M, Holmlund A and Johansson A:
Humoral immune response to Aggregatibacter actinomycetemcomitans
leukotoxin. J Periodontal Res. 46:170–175. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Singh S, Bhatia R, Singh A, Singh P, Kaur
R, Khare P, Purama RK, Boparai RK, Rishi P, Ambalam P, et al:
Probiotic attributes and prevention of LPS-induced pro-inflammatory
stress in RAW264.7 macrophages and human intestinal epithelial cell
line (Caco-2) by newly isolated Weissella cibaria strains. Food
Funct. 9:1254–1264. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Weidenmaier C, Kristian SA and Peschel A:
Bacterial resistance to antimicrobial host defenses--an emerging
target for novel antiinfective strategies? Curr Drug Targets.
4:643–649. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Nau GJ, Richmond JF, Schlesinger A,
Jennings EG, Lander ES and Young RA: Human macrophage activation
programs induced by bacterial pathogens. Proc Natl Acad Sci USA.
99:1503–1508. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Turner MD, Nedjai B, Hurst T and
Pennington DJ: Cytokines and chemokines: At the crossroads of cell
signalling and inflammatory disease. Biochim Biophys Acta.
1843:2563–2582. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Choi EY, Jin JY, Lee JY, Choi JI, Choi IS
and Kim SJ: Anti-inflammatory effects and the underlying mechanisms
of action of daidzein in murine macrophages stimulated with
Prevotella intermedia lipopolysaccharide. J Periodontal Res.
47:204–211. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Liu T, Zhang L, Joo D and Sun SC: NF-κB
signaling in inflammation. Signal Transduct Target Ther. doi:
10.1038/sigtrans.2017.23.
|
|
16
|
Aktan F: iNOS-mediated nitric oxide
production and its regulation. Life Sci. 75:639–653. 2004.
View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Chen CC, Chang KL, Huang JF, Huang JS and
Tsai CC: Correlation of interleukin-1 beta, interleukin-6, and
periodontitis. Kaohsiung J Med Sci. 13:609–617. 1997.PubMed/NCBI
|
|
18
|
Carrizales-Sepúlveda EF, Ordaz-Farías A,
Vera-Pineda R and Flores-Ramírez R: Periodontal disease, systemic
inflammation and the risk of cardiovascular disease. Heart Lung
Circ. 27:1327–1334. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Parihar AS, Katoch V, Rajguru SA, Rajpoot
N, Singh P and Wakhle S: Periodontal disease: A possible
risk-factor for adverse pregnancy outcome. J Int Oral Health.
7:137–142. 2015.PubMed/NCBI
|
|
20
|
Ganesh P, Karthikeyan R, Muthukumaraswamy
A and Anand J: A potential role of periodontal inflammation in
Alzheimers disease: A Review. Oral Health Prev Dent. 15:7–12.
2017.PubMed/NCBI
|
|
21
|
Da Rocha HA, Silva CF, Santiago FL,
Martins LG, Dias PC and De Magalhães D: Local drug delivery systems
in the treatment of periodontitis: A literature review. J Int Acad
Periodontol. 17:82–90. 2015.PubMed/NCBI
|
|
22
|
Blumenthal KG, Peter JG, Trubiano JA and
Phillips EJ: Antibiotic allergy. Lancet. 393:183–198. 2019.
View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Gupta G: Probiotics and periodontal
health. J Med Life. 4:387–394. 2011.PubMed/NCBI
|
|
24
|
Saha S, Tomaro-Duchesneau C, Tabrizian M
and Prakash S: Probiotics as oral health biotherapeutics. Expert
Opin Biol Ther. 12:1207–1220. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Reid G, Jass J, Sebulsky MT and McCormick
JK: Potential uses of probiotics in clinical practice. Clin
Microbiol Rev. 16:658–672. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Burton JP, Drummond BK, Chilcott CN, Tagg
JR, Thomson WM, Hale JDF and Wescombe PA: Influence of the
probiotic Streptococcus salivarius strain M18 on indices of dental
health in children: A randomized double-blind, placebo-controlled
trial. J Med Microbiol. 62:875–884. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Szkaradkiewicz AK, Stopa J and Karpiński
TM: Effect of oral administration involving a probiotic strain of
Lactobacillus reuteri on pro-inflammatory cytokine response in
patients with chronic periodontitis. Arch Immunol Ther Exp (Warsz).
62:495–500. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Kang MS, Kim BG, Chung J, Lee HC and Oh
JS: Inhibitory effect of Weissella cibaria isolates on the
production of volatile sulphur compounds. J Clin Periodontol.
33:226–232. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Björkroth KJ, Schillinger U, Geisen R,
Weiss N, Hoste B, Holzapfel WH, Korkeala HJ and Vandamme P:
Taxonomic study of Weissella confusa and description of
Weissella cibaria sp. nov., detected in food and clinical
samples. Int J Syst Evol Microbiol. 52:141–148. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Park HE, Kang KW, Kim BS, Lee SM and Lee
WK: Immunomodulatory potential of Weissella cibaria in aged
C57BL/6J mice. J Microbiol Biotechnol. 27:2094–2103. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Yu HS, Lee NK, Choi AJ, Choe JS, Bae CH
and Paik HD: Anti-inflammatory potential of probiotic strain
Weissella cibaria JW15 isolated from Kimchi through regulation of
NF-κB and MAPKs pathways in LPS-induced RAW 264.7 cells. J
Microbiol Biotechnol. 29:1022–1032. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Jang HJ, Kang MS, Yi SH, Hong JY and Hong
SP: Comparative study on the characteristics of Weissella cibaria
CMU and probiotic strains for oral care. Molecules. 21:212016.
View Article : Google Scholar
|
|
33
|
Kang MS, Lim HS, Kim SM, Lee HC and Oh JS:
Effect of Weissella cibaria on Fusobacterium
nucleatum-induced interleukin-6 and interleukin-8 production in KB
cells. J Bacteriol Virol. 41:9–18. 2011. View Article : Google Scholar
|
|
34
|
Kim JW, Jung BH, Lee JH, Yoo KY, Lee H,
Kang MS and Lee JK: Effect of Weissella cibaria on the reduction of
periodontal tissue destruction in mice. J Periodontol.
doi:10.1002/JPER.19-0288.
|
|
35
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(−ΔΔC(T)) method. Methods. 25:402–408. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Kang MS, Chung J, Kim SM, Yang KH and Oh
JS: Effect of Weissella cibaria isolates on the formation of
Streptococcus mutans biofilm. Caries Res. 40:418–425. 2006.
View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Park HR, Kim HJ and Kang MS: Clinical
studies on the dental caries prevention effects of the ability of
Weissella cibaria CMU to adhere to the oral cavity. Indian J Public
Health Res Dev. 9:1163–1169. 2018. View Article : Google Scholar
|
|
38
|
Asok A, Bhandary R, Shetty M and Shenoy
MS: Probiotics and periodontal disease. Int J Oral Health Sci.
8:68–72. 2018. View Article : Google Scholar
|
|
39
|
Baron VT, Pio R, Jia Z and Mercola D:
Early growth response 3 regulates genes of inflammation and
directly activates IL6 and IL8 expression in prostate cancer. Br J
Cancer. 112:755–764. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Madeira MF, Queiroz-Junior CM, Cisalpino
D, Werneck SM, Kikuchi H, Fujise O, Ryffel B, Silva TA, Teixeira MM
and Souza DG: MyD88 is essential for alveolar bone loss induced by
Aggregatibacter actinomycetemcomitans lipopolysaccharide in mice.
Mol Oral Microbiol. 28:415–424. 2013. View Article : Google Scholar : PubMed/NCBI
|