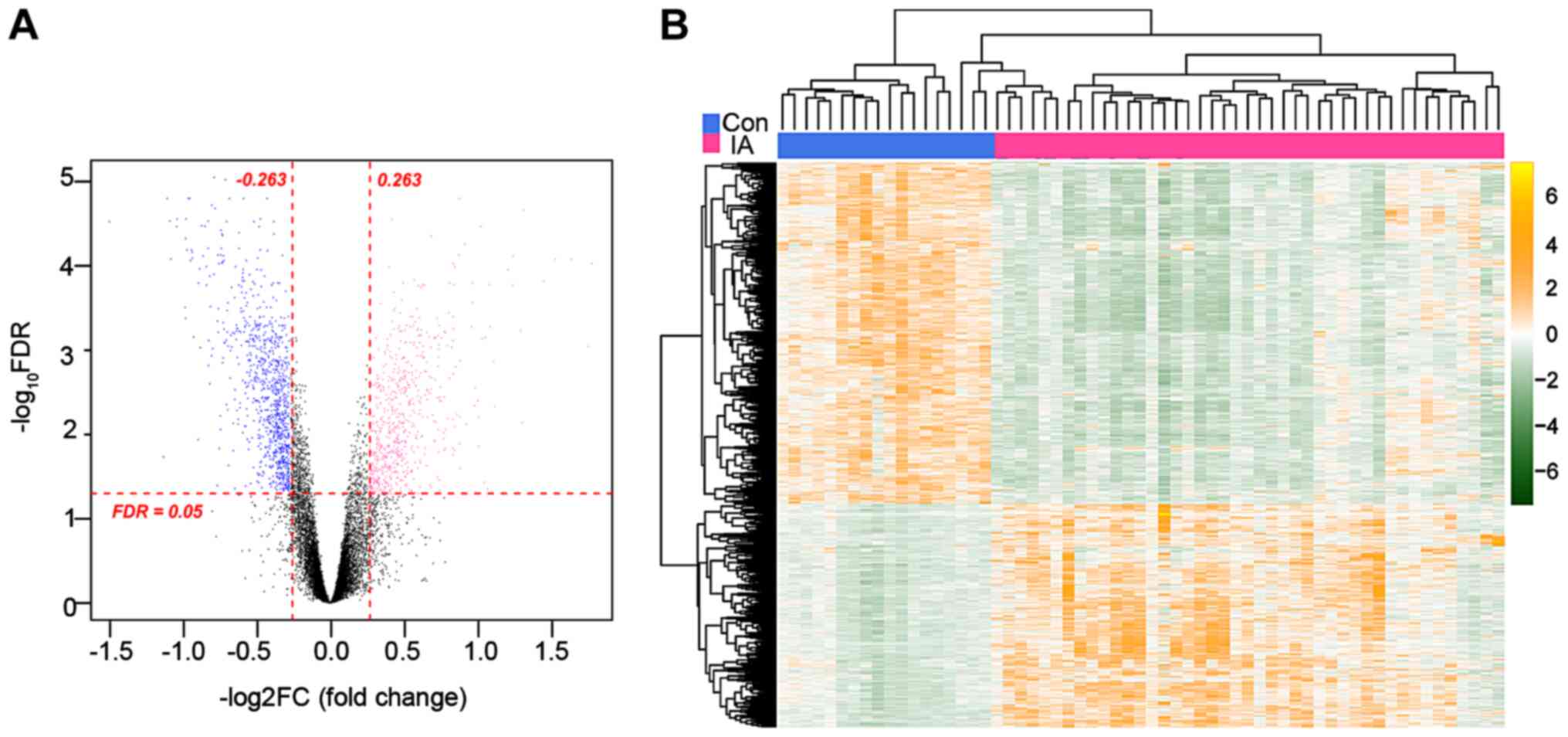
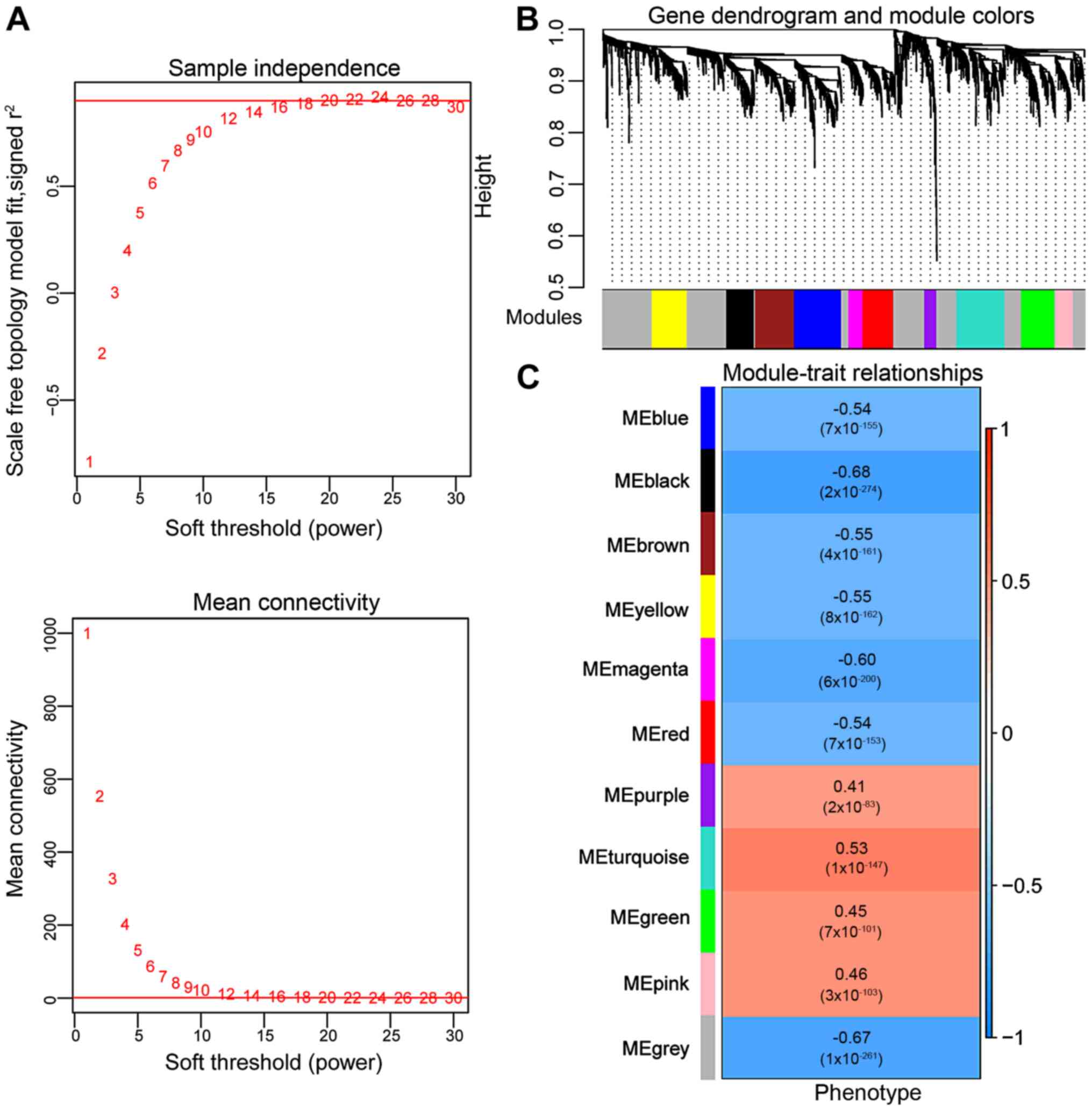
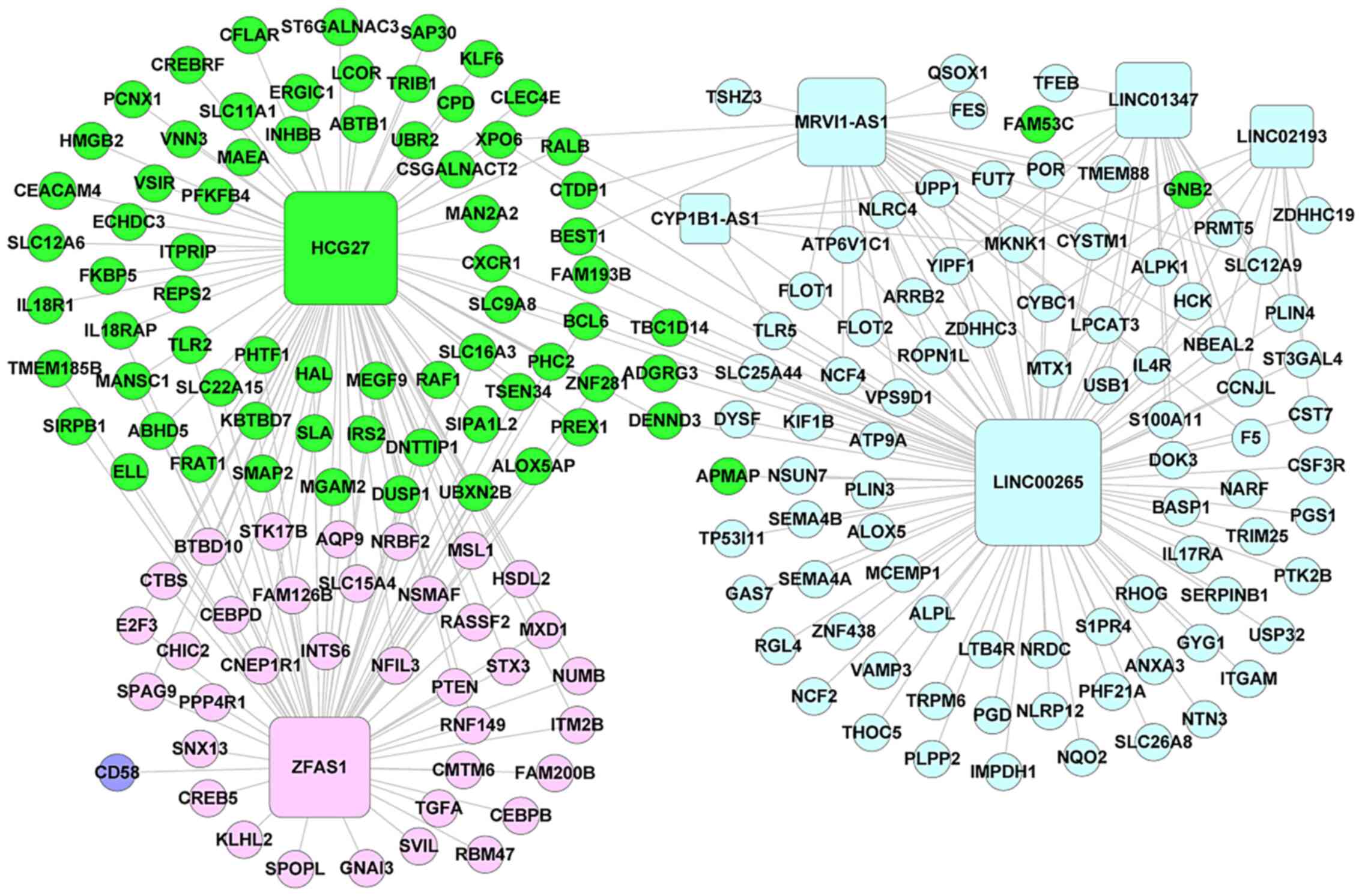
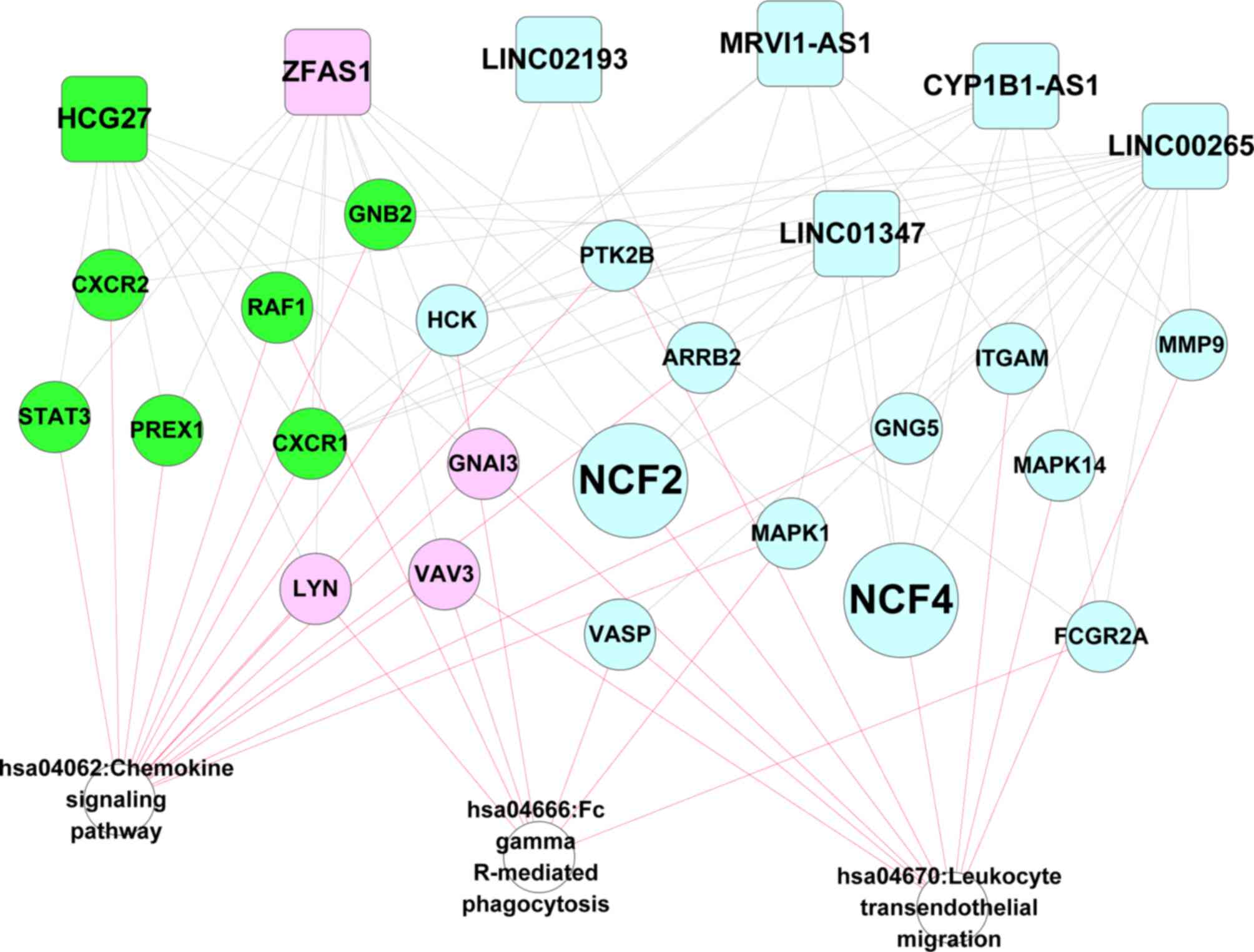
|
1
|
Garg K, Sinha S, Kale SS, Chandra PS, Suri
A, Singh MM, Kumar R, Sharma MS, Pandey RM, Sharma BS and Mahapatra
AK: Role of simvastatin in prevention of vasospasm and improving
functional outcome after aneurysmal sub-arachnoid hemorrhage: A
prospective, randomized, double-blind, placebo-controlled pilot
trial. Br J Neurosurg. 27:181–186. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Wesali S, Persson HC, Cederin B and
Sunnerhagen KS: Improved survival after non-traumatic subarachnoid
haemorrhage with structured care pathways and modern intensive
care. Clin Neurol Neurosurg. 138:52–58. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Banki NM, Alexander K, Dae MW, Miss J,
Tung P, Lawton MT, Drew BJ, Foster E, Smith W, Parmley WW and
Zaroff JG: Acute neurocardiogenic injury after subarachnoid
hemorrhage. Circulation. 112:3314–3319. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Johnston SC, Selvin S and Gress DR: The
burden, trends, and demographics of mortality from subarachnoid
hemorrhage. Neurology. 50:1413–1418. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Suarez JI, Tarr RW and Selman WR:
Aneurysmal subarachnoid hemorrhage. N Engl J Med. 354:387–396.
2006. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Zumofen DW, Roethlisberger M, Achermann R,
Bawarjan S, Stienen MN, Fung C, D'Alonzo D, Maldaner N, Ferrari A,
Corniola MV, et al: Factors associated with clinical and
radiological status on admission in patients with aneurysmal
subarachnoid hemorrhage. Neurosurg Rev. 41:1059–1069. 2018.
View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Hendrix P, Foreman PM, Harrigan MR, Fisher
WS Rd, Vyas NA, Lipsky RH, Lin M, Walters BC, Tubbs RS, Shoja MM,
et al: The role of endothelial nitric oxide synthase-786 T/C
polymorphism in cardiac instability following aneurysmal
subarachnoid hemorrhage. Nitric Oxide. 71:52–56. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Liang Z, Chi YJ, Lin GQ, Xiao LF, Su GL
and Yang LM: LncRNA MEG3 participates in neuronal cell injury
induced by subarachnoid hemorrhage via inhibiting the Pi3k/Akt
pathway. Eur Rev Med Pharmacol Sci. 22:2824–2831. 2018.PubMed/NCBI
|
|
9
|
Santiago-Sim T, Fang X, Hennessy ML,
Nalbach SV, DePalma SR, Lee MS, Greenway SC, McDonough B,
Hergenroeder GW, Patek KJ, et al: THSD1 (thrombospondin type 1
domain containing protein 1) mutation in the pathogenesis of
intracranial aneurysm and subarachnoid hemorrhage. Stroke.
47:3005–3013. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Pawlowska E, Szczepanska J, Wisniewski K,
Tokarz P, Jaskólski DJ and Blasiak J: NF-κB-mediated inflammation
in the pathogenesis of intracranial aneurysm and subarachnoid
hemorrhage. Does autophagy play a role? Int J Mol Sci.
19:12452018.
|
|
11
|
Zhao L, Liu H, Yue L, Zhang J, Li X, Wang
B, Lin Y and Qu Y: Melatonin attenuates early brain injury via the
melatonin receptor/Sirt1/NF-κB signaling pathway following
subarachnoid hemorrhage in mice. Mol Neurobiol. 54:1612–1621. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Ma C, Zhou W, Yan Z, Qu M and Bu X:
Toll-like receptor 4 (TLR4) is associated with cerebral vasospasm
and delayed cerebral ischemia in aneurysmal subarachnoid
hemorrhage. Neurol Med Chir (Tokyo). 55:878–884. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Okada T and Suzuki H: Toll-like receptor 4
as a possible therapeutic target for delayed brain injuries after
aneurysmal subarachnoid hemorrhage. Neural Regen Res. 12:193–196.
2017. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Pera J, Korostynski M, Golda S, Piechota
M, Dzbek J, Krzyszkowski T, Dziedzic T, Moskala M, Przewlocki R,
Szczudlik A and Slowik A: Gene expression profiling of blood in
ruptured intracranial aneurysms: In search of biomarkers. J Cereb
Blood Flow Metab. 33:1025–1031. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Ritchie ME, Phipson B, Wu D, Hu Y, Law CW,
Shi W and Smyth GK: limma powers differential expression analyses
for RNA-sequencing and microarray studies. Nucleic Acids Res.
43:e472015. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Yates B, Braschi B, Gray KA, Seal RL,
Tweedie S and Bruford EA: Genenames.org: The HGNC and VGNC
resources in 2017. Nucleic Acids Res. 45(D1): D619–D625. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Szekely GJ and Rizzo ML: Hierarchical
clustering via joint between-within distances: Extending ward's
minimum variance method. J Classif. 22:151–183. 2005. View Article : Google Scholar
|
|
18
|
Wright RM, Aglyamova GV, Meyer E and Matz
MV: Variance stabilized gene expression data for WGCNA. Comput
Graph. 29:805–817. 2015.
|
|
19
|
Langfelder P and Horvath S: WGCNA: An R
package for weighted correlation network analysis. BMC
Bioinformatics. 9:5592008. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Ashburner M, Ball CA, Blake JA, Botstein
D, Butler H, Cherry JM, Davis AP, Dolinski K, Dwight SS, Eppig JT,
et al: Gene ontology: Tool for the unification of biology. The gene
ontology consortium. Nat Genet. 25:25–29. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
The Gene Ontology Consortium: The gene
ontology resource: 20 Years and still GOing strong. Nucleic Acids
Res. 47(D1): D330–D338. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Huang da W, Sherman BT and Lempicki RA:
Bioinformatics enrichment tools: Paths toward the comprehensive
functional analysis of large gene lists. Nucleic Acids Res.
37:1–13. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
R Core Team: R: A language and environment
for statistical computing. R Foundation for Statistical Computing;
Vienna, Austria: 2015
|
|
24
|
Shannon P, Markiel A, Ozier O, Baliga NS,
Wang JT, Ramage D, Amin N, Schwikowski B and Ideker T: Cytoscape: A
software environment for integrated models of biomolecular
interaction networks. Genome Res. 13:2498–2504. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Kanehisa M and Goto S: KEGG: Kyoto
encyclopedia of genes and genomes. Nucleic Acids Res. 28:27–30.
2000. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Davis AP, Grondin CJ, Johnson RJ, Sciaky
D, McMorran R, Wiegers J, Wiegers TC and Mattingly CJ: The
comparative toxicogenomics database: Update 2019. Nucleic Acids
Res. 47(D1): D948–D954. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Kurki MI, Häkkinen SK, Frösen J, Tulamo R,
von und zu Fraunberg M, Wong G, Tromp G, Niemelä M, Hernesniemi J,
Jääskeläinen JE and Ylä-Herttuala S: Upregulated signaling pathways
in ruptured human saccular intracranial aneurysm wall: An emerging
regulative role of Toll-like receptor signaling and nuclear
factor-κB, hypoxia-inducible factor-1A, and ETS transcription
factors. Neurosurgery. 68:1667–1676. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Guo J, Qi J, He K, Wu J, Bai S, Zhang T,
Zhao J and Wang Z: The Asian corn borer Ostrinia furnacalis feeding
increases the direct and indirect defense of mid-whorl stage
commercial maize in the field. Plant Biotechnol J. 17:88–102. 2019.
View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Diringer MN and Zazulia AR: Aneurysmal
subarachnoid hemorrhage: Strategies for preventing vasospasm in the
intensive care unit. Semin Respir Crit Care Med. 38:760–767. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Leclerc JL, Blackburn S, Neal D, Mendez
NV, Wharton JA, Waters MF and Doré S: Haptoglobin phenotype
predicts the development of focal and global cerebral vasospasm and
may influence outcomes after aneurysmal subarachnoid hemorrhage.
Proc Natl Acad Sci USA. 112:1155–1160. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Li YC, Zhao L, Wu JP, Qu CX, Song QK and
Wang RB: Cytokine-induced killer cell infusion combined with
conventional treatments produced better prognosis for
hepatocellular carcinoma patients with barcelona clinic liver
cancer B or earlier stage: A systematic review and meta-analysis.
Cytotherapy. 18:1525–1531. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Denson LA, Jurickova I, Karns R, Shaw KA,
Cutler DJ, Okou DT, Dodd A, Quinn K, Mondal K, Aronow BJ, et al:
Clinical and genomic correlates of neutrophil reactive oxygen
species production in pediatric patients with Crohn's disease.
Gastroenterology. 154:2097–2110. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Armstrong DL, Miriam E, Raphael Z and
Jacob CO: Systemic lupus erythematosus-associated neutrophil
cytosolic factor 2 mutation affects the structure of NADPH oxidase
complex. J Biol Chem. 290:12595–12602. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Winter S, Hultqvist Hopkins M, Laulund F
and Holmdahl R: A reduction in intracellular reactive oxygen
species due to a mutation in NCF4 promotes autoimmune arthritis in
mice. Antioxid Redox Signal. 25:983–996. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Gauss KA, Bunger PL, Larson TC, Young CJ,
Nelson-Overton LK, Siemsen DW and Quinn MT: Identification of a
novel tumor necrosis factor alpha-responsive region in the NCF2
promoter. J Leukoc Biol. 77:267–278. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Cunninghame Graham DS, Morris DL, Bhangale
TR, Criswell LA, Syvänen AC, Rönnblom L, Behrens TW, Graham RR and
Vyse TJ: Association of NCF2, IKZF1, IRF8, IFIH1, and TYK2 with
systemic lupus erythematosus. PLoS Genet. 7:e10023412011.
View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Li SL, Valente AJ, Wang L, Gamez MJ and
Clark RA: Transcriptional regulation of the p67phox gene: Role of
AP-1 in concert with myeloid-specific transcription factors. J Biol
Chem. 276:39368–39378. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Gupta JW, Kubin M, Hartman L, Cassatella M
and Trinchieri G: Induction of expression of genes encoding
components of the respiratory burst oxidase during differentiation
of human myeloid cell lines induced by tumor necrosis factor and
gamma-interferon. Cancer Res. 52:2530–2537. 1992.PubMed/NCBI
|
|
39
|
Wisdom R: AP-1: One switch for many
signals. Exp Cell Res. 253:180–185. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Cohen M, Meisser A, Haenggeli L and
Bischof P: Involvement of MAPK pathway in TNF-alpha-induced MMP-9
expression in human trophoblastic cells. Mol Hum Reprod.
12:225–232. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Robinson SC, Scott KA and Balkwill FR:
Chemokine stimulation of monocyte matrix metalloproteinase-9
requires endogenous TNF-alpha. Eur J Immunol. 32:404–412. 2002.
View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Soumyarani VS and Jayakumari N:
Oxidatively modified high density lipoprotein promotes inflammatory
response in human monocytes-macrophages by enhanced production of
ROS, TNF-α, MMP-9, and MMP-2. Mol Cell Biochem. 366:277–285. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Chen J, Yang X, Zhang W, Peng D, Xia Y, Lu
Y, Han X, Song G, Zhu J and Liu R: Therapeutic effects of
resveratrol in a mouse model of LPS and cigarette smoke-induced
COPD. Inflammation. 39:1949–1959. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Higgins KA, Park D, Lee GY, Curran WJ and
Deng X: Exercise-induced lung cancer regression: Mechanistic
findings from a mouse model. Cancer. 120:3302–3310. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Li BS, Zuo QF, Zhao YL, Xiao B, Zhuang Y,
Mao XH, Wu C, Yang SM, Zeng H, Zou QM and Guo G: MicroRNA-25
promotes gastric cancer migration, invasion and proliferation by
directly targeting transducer of ERBB2, 1 and correlates with poor
survival. Oncogene. 34:2556–2565. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Qian Y, Cao L, Guan T, Chen L, Xin H, Li
Y, Zheng R and Yu D: Protection by genistein on cortical neurons
against oxidative stress injury via inhibition of NF-kappaB, JNK
and ERK signaling pathway. Pharm Biol. 53:1124–1132. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Wu JS, Tsai HD, Cheung WM, Hsu CY and Lin
TN: PPAR-γ ameliorates neuronal apoptosis and ischemic brain injury
via suppressing NF-κB-driven p22phox transcription. Mol Neurobiol.
53:3626–3645. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Pan W, Lin L, Zhang N, Yuan F, Hua X, Wang
Y and Mo L: Neuroprotective effects of dexmedetomidine against
hypoxia-induced nervous system injury are related to inhibition of
NF-κB/COX-2 pathways. Cell Mol Neurobiol. 36:1179–1188. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Joosten LA, Helsen MM, Saxne T, van De Loo
FA, Heinegard D and van Den Berg WB: IL-1 alpha beta blockade
prevents cartilage and bone destruction in murine type II
collagen-induced arthritis, whereas TNF-alpha blockade only
ameliorates joint inflammation. J Immunol. 163:5049–5055.
1999.PubMed/NCBI
|
|
50
|
Charrad R, Berraïes A, Hamdi B, Ammar J,
Hamzaoui K and Hamzaoui A: Anti-inflammatory activity of IL-37 in
asthmatic children: Correlation with inflammatory cytokines TNF-α,
IL-β, IL-6 and IL-17A. Immunobiology. 221:182–187. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
51
|
Su Y, Hu J, Lan H, Chen Y and Wei X: Long
non-coding RNA LINC00265 promotes inflammation via sponging
miR-let-7a in abdominal aortic aneurysm. Aging. 11:4463–4477. 2019.
View Article : Google Scholar : PubMed/NCBI
|
|
52
|
Malaplate-Armand C, Ferrari L, Masson C,
Siest G and Batt AM: Astroglial CYP1B1 up-regulation in
inflammatory/oxidative toxic conditions: IL-1beta effect and
protection by N-acetylcysteine. Toxicol Lett. 138:243–251. 2003.
View Article : Google Scholar : PubMed/NCBI
|
|
53
|
Smerdová L, Šmerdová J, Kabátková M,
Kohoutek J, Blažek D, Machala M and Vondráček J: Upregulation of
CYP1B1 expression by inflammatory cytokines is mediated by the p38
MAP kinase signal transduction pathway. Carcinogenesis.
35:2534–2543. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
54
|
Fan W, Fnag R, Feng M, Dai G and Wu G:
Microarray expression profile of long non-coding RNAs in human
endothelial cells exposed to atheroprotective shear stress. J Am
Coll Cardiol. 66:C67–C68. 2015. View Article : Google Scholar
|
|
55
|
Leng L, Zhang C, Ren L and Li Q:
Construction of a long non-coding RNA-mediated competitive
endogenous RNA network reveals global patterns and regulatory
markers in gestational diabetes. Int J Mol Med. 43:927–935.
2019.PubMed/NCBI
|
|
56
|
Ye Y, Gao X and Yang N: LncRNA ZFAS1
promotes cell migration and invasion of fibroblast-like
synoviocytes by suppression of miR-27a in rheumatoid arthritis.
Human Cell. 31:14–21. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
57
|
Marcos V, Zhou Z, Yildirim AO, Bohla A,
Hector A, Vitkov L, Wiedenbauer EM, Krautgartner WD, Stoiber W,
Belohradsky BH, et al: CXCR2 mediates NADPH oxidase-independent
neutrophil extracellular trap formation in cystic fibrosis airway
inflammation. Nat Med. 16:1018–1023. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
58
|
Bertini R, Allegretti M, Bizzarri C,
Moriconi A, Locati M, Zampella G, Cervellera MN, Di Cioccio V,
Cesta MC, Galliera E, et al: Noncompetitive allosteric inhibitors
of the inflammatory chemokine receptors CXCR1 and CXCR2: Prevention
of reperfusion injury. Proc Natl Acad Sci USA. 101:11791–11796.
2004. View Article : Google Scholar : PubMed/NCBI
|
|
59
|
Wuyts A, Van Osselaer N, Haelens A, Samson
I, Herdewijn P, Ben-Baruch A, Oppenheim JJ, Proost P and Van Damme
J: Characterization of synthetic human granulocyte chemotactic
protein 2: Usage of chemokine receptors CXCR1 and CXCR2 and in vivo
inflammatory properties. Biochemistry. 36:2716–2723. 1997.
View Article : Google Scholar : PubMed/NCBI
|
|
60
|
De Paola M, Buanne P, Biordi L, Bertini R,
Ghezzi P and Mennini T: Chemokine MIP-2/CXCL2, acting on CXCR2,
induces motor neuron death in primary cultures.
Neuroimmunomodulation. 14:310–316. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
61
|
Du SH, Zhang W, Yue X, Luo XQ, Tan XH, Liu
C, Qiao DF and Wang H: Role of CXCR1 and interleukin-8 in
methamphetamine-induced neuronal apoptosis. Front Cell Neurosci.
12:2302018. View Article : Google Scholar : PubMed/NCBI
|
|
62
|
Zhou Y, Li RJ, Li M, Liu X, Zhu HY, Ju Z,
Miao X and Xu GY: Overexpression of GRK6 attenuates neuropathic
pain via suppression of CXCR2 in rat dorsal root ganglion. Mol
Pain. 12:17448069166463812016. View Article : Google Scholar : PubMed/NCBI
|
|
63
|
Lopes AH, Brandolini L, Aramini A,
Bianchini G, Silva RL, Zaperlon AC, Verri WA Jr, Alves-Filho JC,
Cunha FQ, Teixeira MM, et al: DF2755A, a novel non-competitive
allosteric inhibitor of CXCR1/2, reduces inflammatory and
post-operative pain. Pharmacol Res. 103:69–79. 2016. View Article : Google Scholar : PubMed/NCBI
|