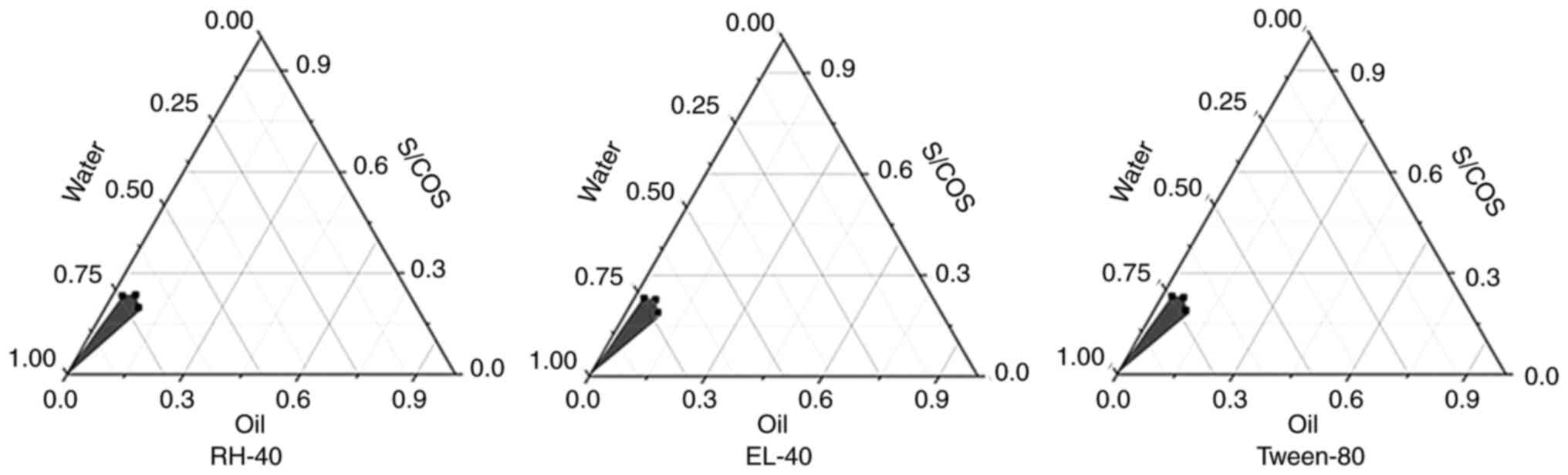
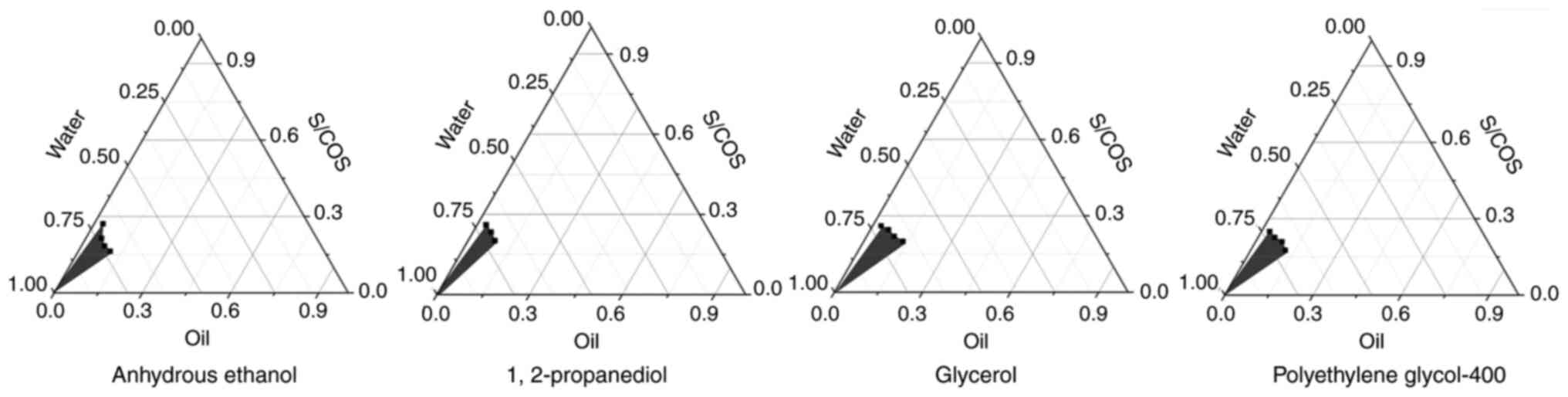
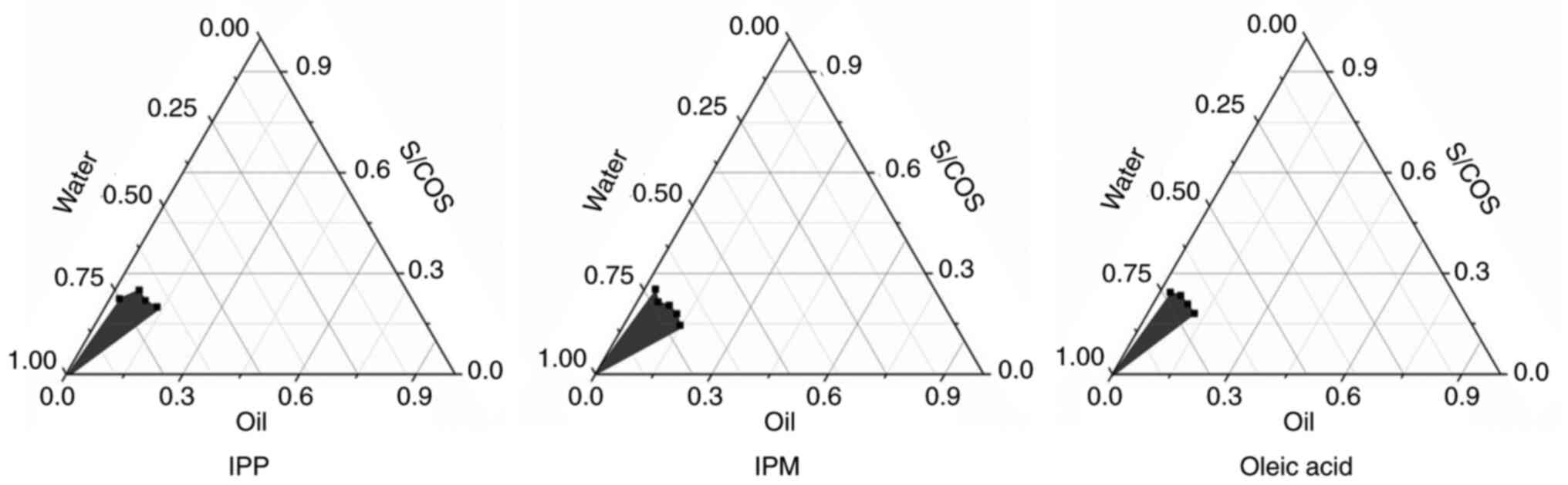
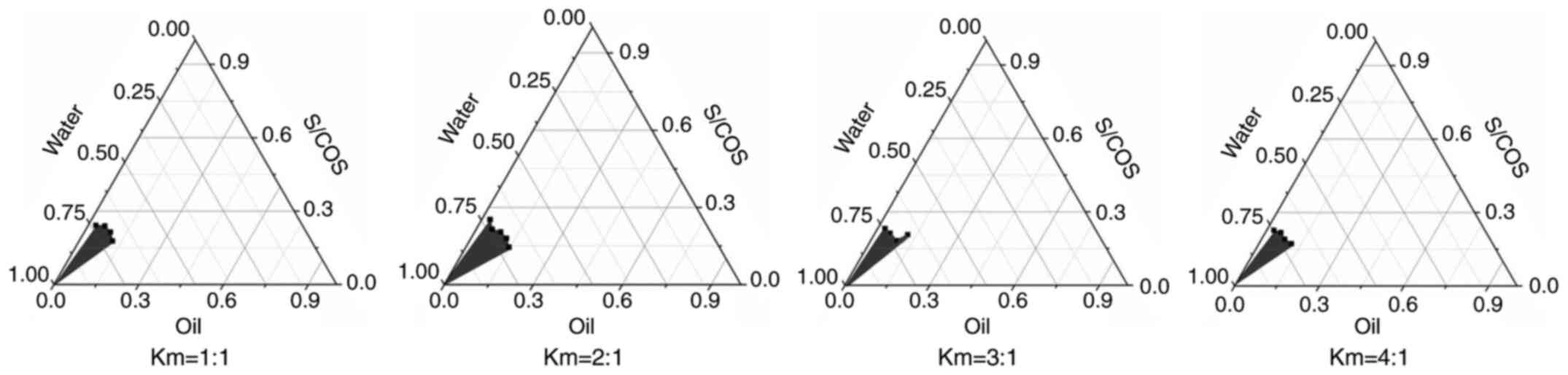
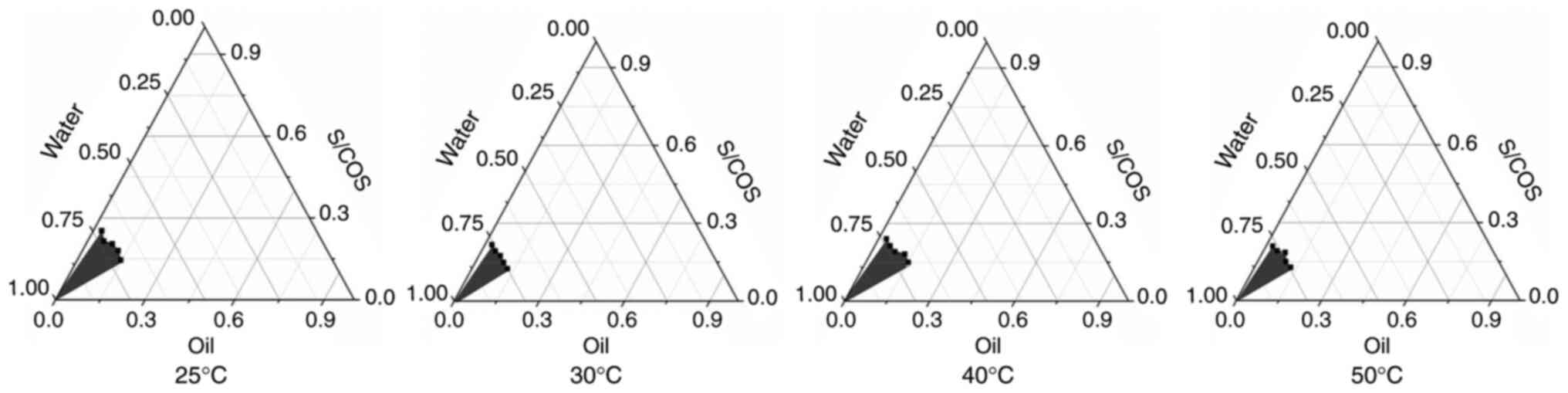
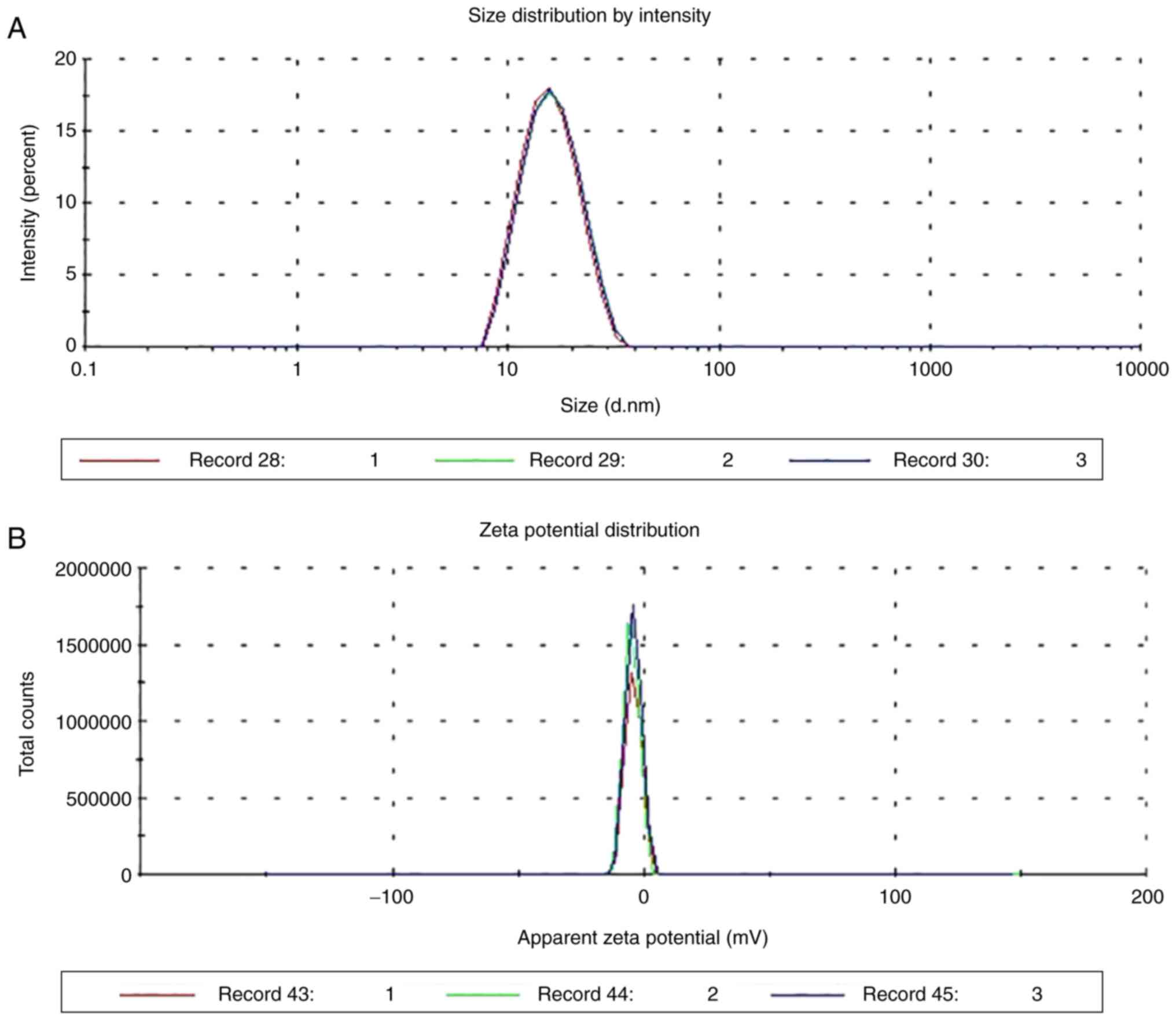
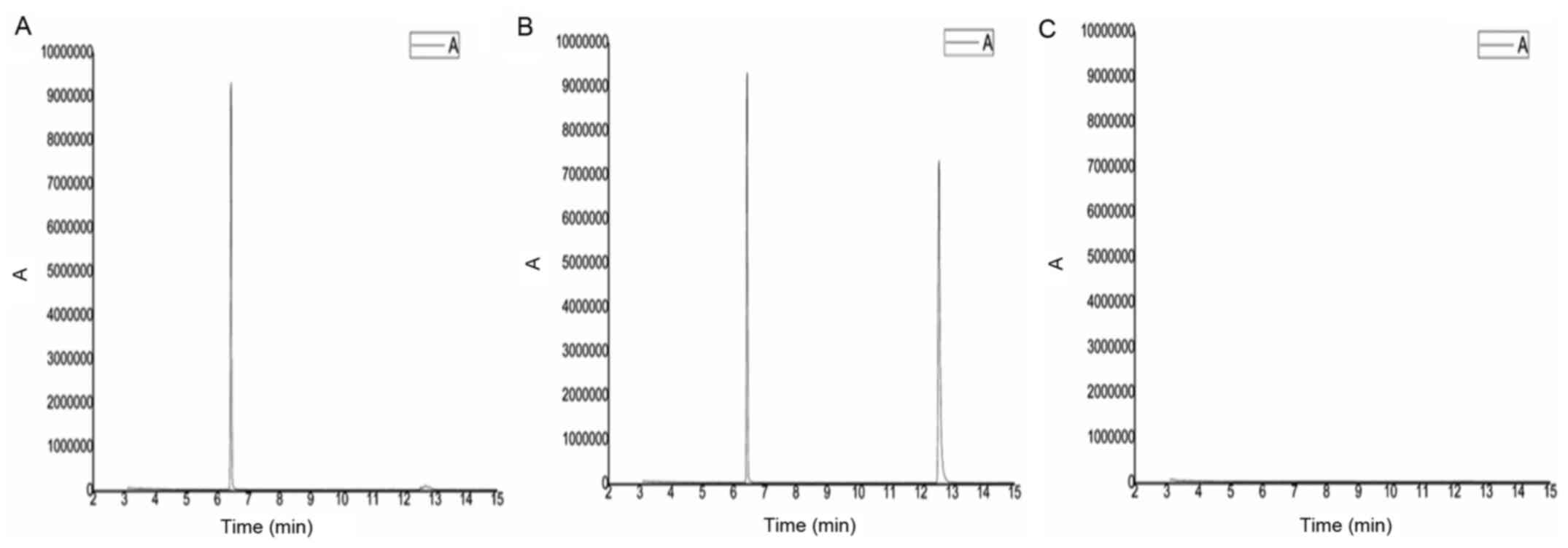
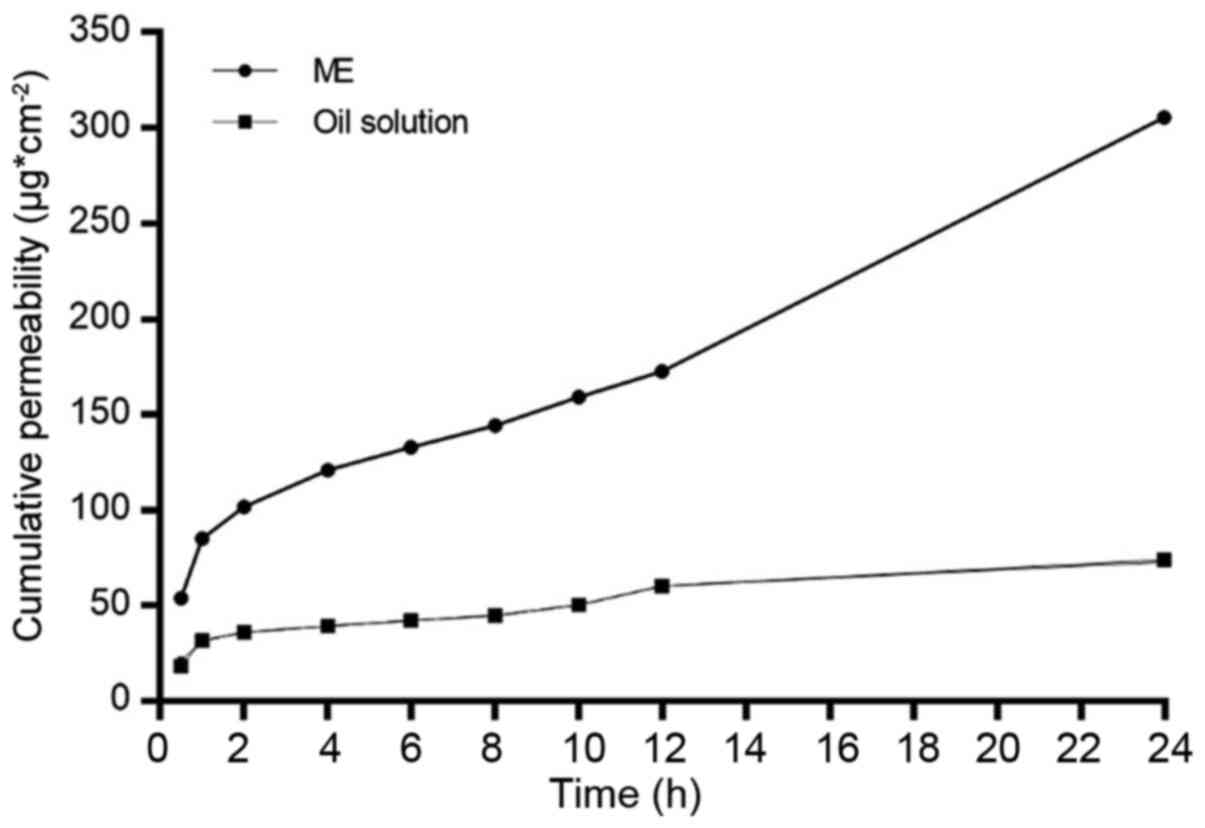
|
1
|
Lam HY, Tergaonkar V and Ahn KS:
Mechanisms of allergen-specific immunotherapy for allergic rhinitis
and food allergies. Biosci Rep. Apr 30–2020.(Epub ahead of print).
doi: 10.1042/BSR20200256. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Mitsias DI, Dimou MV, Lakoumentas J,
Alevizopoulos K, Sousa-Pinto B, Fonseca JA, Bousquet J and
Papadopoulos NG: Effect of nasal irrigation on allergic rhinitis
control in children; complementarity between CARAT and MASK
outcomes. Clin Transl Allergy. 10:92020. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Rawls M, Thiele J, Adams DE, Steacy LM and
Ellis AK: Clinical symptoms and biomarkers of Bermuda grass-induced
allergic rhinitis using the nasal allergen challenge model. Ann
Allergy Asthma Immunol. 124:608–615.e2. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Wang GA: Efficacy of desloratadine citrate
disodium combined with mometasone furoate in the treatment of
allergic rhinitis and its effect on patients' complications.
Chinese Journal of Rational Drug Use. 16:179–181. 2019.(In
Chinese).
|
|
5
|
Yang CS, Xu J and Zhang YY: Determination
of 1,8-cineole in bitongning spray by gas chromatography. Guizhou
Med J. 35:929–930. 2011.(In Chinese).
|
|
6
|
Li XB and Wang P: Determination of
volatile oil content in bitongning drops by GC method. Heilongjiang
Med J. 24:16–17. 2011.
|
|
7
|
Cai XZ: Rudimentary research on
identification of Herba Centipedae and screening of its
active material anti-Allergic Rhinitis. Southern Medical
University; 2008, (In Chinese).
|
|
8
|
Lin YC and Gao M: Research progress on
chemical constituents and pharmacology of Centipeda minima.
J Zhejiang Chin Med Univ. 35:303–304. 2011.
|
|
9
|
Yang XX and Zhuang ZQ: Research progress
on chemical constituents and pharmacological action of Flos
magnoliae. Chin Tradit Herbal Drugs. 7:490–492. 1998.
|
|
10
|
Yuan JM, Huang XY and Zhou YT: Preparation
of Flos magnoliae-Herba Centipedae thermosensitive nasal in
situ gels. Zhong Cheng Yao. 40:2, 656–662. 2018.
|
|
11
|
Liang YL, Zhang XF and Zou JB:
Pharmacology mechanism of and for treating allergic rhinitis based
on pharmacology network. Drug Dev Ind Pharm. 45:547–555. 2019.
View Article : Google Scholar
|
|
12
|
Okińczyc P, Szumny A, Szperlik J, Kulma A,
Franiczek R, Żbikowska B, Krzyżanowska B and Sroka Z: Profile of
polyphenolic and essential oil composition of polish propolis,
black poplar and Aspens buds. Molecules. 23:E12622018. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Katdare A, Khunt D, Thakkar S, Polaka SN
and Misra M: Comparative evaluation of fish oil and butter oil in
modulating delivery of galantamine hydrobromide to brain via
intranasal route: Pharmacokinetic and oxidative stress studies.
Drug Deliv Transl Res. 10:1136–1146. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Mitsou E, Pletsa V, Sotiroudis GT, Panine
P, Zoumpanioti M and Xenakis A: Development of a microemulsion for
encapsulation and delivery of gallic acid. The role of chitosan.
Colloids Surf B Biointerfaces. 190:1109742020. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Xu T, Ge SM, Deng LH, Wei MY, Xu YH and Wu
CB: Preparation of pseudolaric acid B microemulsion and its
transdermal penetration. Chin Tradit Herbal Drugs. 43:683–689.
2012.
|
|
16
|
Lu SS, Qian YH and Xu BB: Application of
microemulsification technology in transdermal penetration of
volatile oil from Chinese material medical. Sci Technol Vis.
25:237–239. 2018.
|
|
17
|
Wu JY, Li YJ, Han M, Hu XB, Yang L, Wang
JM and Xiang DX: A microemulsion of puerarin-phospholipid complex
for improving bioavailability: Preparation, in vitro and in vivo
evaluations. Drug Dev Ind Pharm. 44:1336–1341, 336–341. 2018.
View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Chen Y, Cheng G, Hu R, Chen S, Lu W, Gao
S, Xia H, Wang B, Sun C, Nie X, et al: A nasal temperature and pH
dual-responsive in situ gel delivery system based on microemulsion
of huperzine A: Formulation, evaluation, and in vivo
pharmacokinetic study. AAPS PharmSciTech. 20:3012019. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Mishra R, Prabhavalkar KS and Bhatt LK:
Preparation, optimization, and evaluation of Zaltoprofen-loaded
microemulsion and microemulsion-based gel for transdermal delivery.
J Liposome Res. 26:297–306. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Cai H, Ma KY and Li HL: Study on
extraction technology of volatile oil from Flos magnoliae.
Jiujiang Xueyuan Xuebao Ziran Kexue Ban. 32:8–12. 2017.(In
Chinese).
|
|
21
|
Cho YH, Kim S, Bae EK, Mok CK and Park J:
Formulation of a cosurfactant-free O/W microemulsion using nonionic
surfactant mixtures. J Food Sci. 73:E115–E121. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Shah A, Thool P, Sorathiya K, Prajapati H,
Dalrymple D and Serajuddin AT: Effect of different polysorbates on
development of self-microemulsifying drug delivery systems using
medium chain lipids. Drug Dev Ind Pharm. 44:215–223. 2018.
View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Song WJ and Gu W: Pharmacology study and
prospect of aromatic Chinese herbs. Zhonghua Zhongyiyao Zazhi.
32:2, 609–611. 2017.
|
|
24
|
Yang XY and Yi L: Preparation and in vitro
transdermal study of zolmitriptan-diclofenac microemulsion. China
Pharmacy. 28:1841–1844. 2017.
|
|
25
|
Golmohammadzadeh S, Farhadian N, Biriaee
A, Dehghani F and Khameneh B: Preparation, characterization and in
vitro evaluation of microemulsion of raloxifene hydrochloride. Drug
Dev Ind Pharm. 43:1619–1625, 619–625. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Kotmakçı M, Akbaba H, Erel G, Ertan G and
Kantarcı G: Improved method for solid lipid nanoparticle
preparation based on hot microemulsions: Preparation,
characterization, cytotoxicity, and hemocompatibility evaluation.
AAPS PharmSciTech. 18:1355–1365. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Akram A, Rasul A, Waqas MK, Irfan M,
Khalid SH, Aamir MN, Murtaza G, Ur Rehman K, Iqbal M and Khan BA:
Development, characterization and evaluation of in-vitro
anti-inflammatory activity of ginger extract based micro emulsion.
Pak J Pharm Sci. 32:1327–1332. 2019.PubMed/NCBI
|
|
28
|
Lee J, Lee Y, Kim J, Yoon M and Choi YW:
Formulation of microemulsion systems for transdermal delivery of
aceclofenac. Arch Pharm Res. 28:1097–1102. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Golwala P, Rathod S, Patil R, Joshi A, Ray
D, Aswal VK, Bahadur P and Tiwari S: Effect of cosurfactant
addition on phase behavior and microstructure of a water dilutable
microemulsion. Colloids Surf B Biointerfaces. 186:1107362020.
View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Zhang JJ, Cui SF and Tang XL: Study on
preparation technology and in vitro transdermal permeability of
compound clove microemulsion. Zhong Yao Cai. 608–611. 2007.
|
|
31
|
Liu X, Zhang ZH and Chen Y: Preparation of
colchicine microemulsion and its transdermal permeation in vitro.
Chin Tradit Herbal Drugs. 42:963–967. 2011.
|
|
32
|
Yang F, Zhou J, Hu X, Yu SK, Liu C, Pan R,
Chang Q, Liu X and Liao Y: Preparation and evaluation of
self-microemulsions for improved bioavailability of ginsenoside-Rh1
and Rh2. Drug Deliv Transl Res. 7:731–737. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Yang J, Xu H, Wu S, Ju B, Zhu D, Yan Y,
Wang M and Hu J: Preparation and evaluation of microemulsion based
transdermal delivery of Cistanche tubulosa phenylethanoid
glycosides. Mol Med Rep. 15:109–116. 2017. View Article : Google Scholar
|
|
34
|
Zhai XY: Effect of volatile oil from
magnolia on Th cells of Guinea pigs with allergic rhinitis. Shaanxi
J Tradit Chin Med. 31:116–118. 2010.
|
|
35
|
Liu ZG, Yu HM, Wen SL and Liu YL:
Histopathological study on allergic rhinitis treated with
Centipeda minima. Zhongguo Zhong Yao Za Zhi. 30:292–294.
2005.(In Chinese). PubMed/NCBI
|
|
36
|
Wu M, Zhang J and Zhang X: Clinical
observation of Flos magnoliae volatile oil nano-liposome
nasal drops in treating pediatric allergic rhinitis. Zhong guo
Zhong Xi Yi Jie He Za Zhi. 29:740–742. 2009.
|
|
37
|
Jia XS and Zhang YY: Research progress on
essential oils of Cenpiteda minima (L.) A. Br. Aschers.
Shandong Chem Ind. 47:66–68. 2018.
|
|
38
|
Guan Z, Ma XZ and Lv GY: Effects of VOM bp
on IL-12, IFN-γ and histamine levels of rats with allergic
rhinitis. Pharmacol Clin Chin Materia Med. 27:70–72. 2011.
|
|
39
|
Wang YQ, Yang YZ, Wu ZF, Xiong YK and Yang
M: Traditional function and modern research progress on volatile
oil in Chinese materia medica. Chin Tradit Herbal Drugs.
49:455–461. 2018.
|
|
40
|
Das S, Lee SH, Chia VD, Chow PS, Macbeath
C, Liu Y and Shlieout G: Development of microemulsion based topical
ivermectin formulations: Pre-formulation and formulation studies.
Colloids Surf B Biointerfaces. 189:1108232020. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Li SL, Duan Q, Zhao ZD, Xia L, Liu WH,
Wang XG and Shen XZ: Preparation and quality evaluation of Acori
Tatarinowii Rhizoma-volatile oil microemulsion. Chin Tradit Herbal
Drugs. 50(1): 935–941. 2019.(In Chinese).
|
|
42
|
Xu WT: Study on the regularity of the
formation of microemulsion by nonionic surfactans. PhD
dissertation. Jiangnan University; 2009, (In Chinese).
|
|
43
|
Paulo BB, Alvim ID, Reineccius G and Prata
AS: Performance of oil-in-water emulsions stabilized by different
types of surface-active components. Colloids Surf B Biointerfaces.
190:1109392020. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Su J, Wu CH and Jiang F: Preparation of
Paeonol self-microemulsion transdermal delivery system with
peppermint oil as carrier. Zhongguo Shiyan Fangjixue Zazhi.
23:11–16. 2017.(In Chinese).
|
|
45
|
Yuan ZZ, Yin SY and Jin Y: Preparation and
quality evaluation of Eugenia oleifera oil bano-emulsion.
Lishizhen Med Materia Med Res. 28:620–622. 2017.
|
|
46
|
Chen JJ, Xia J, Song J, Luo MF and Huang
Z: Effect of Eugenol in the microemulsion as an oil excipient and
enhancer on percutaneous absorption of Baicalin. Xiandai Shengwu
Yixue Jinzhan. 16:410–413. 2016.(In Chinese).
|
|
47
|
Lu SS, Zhao YR, Yao JH, Ren L, Li Y and
Chen J: Preparation technology on essential oil from Chinese
materia medica as penetration enhancers. Chin Tradit Herbal Drugs.
49:2477–2481. 2018.(In Chinese).
|