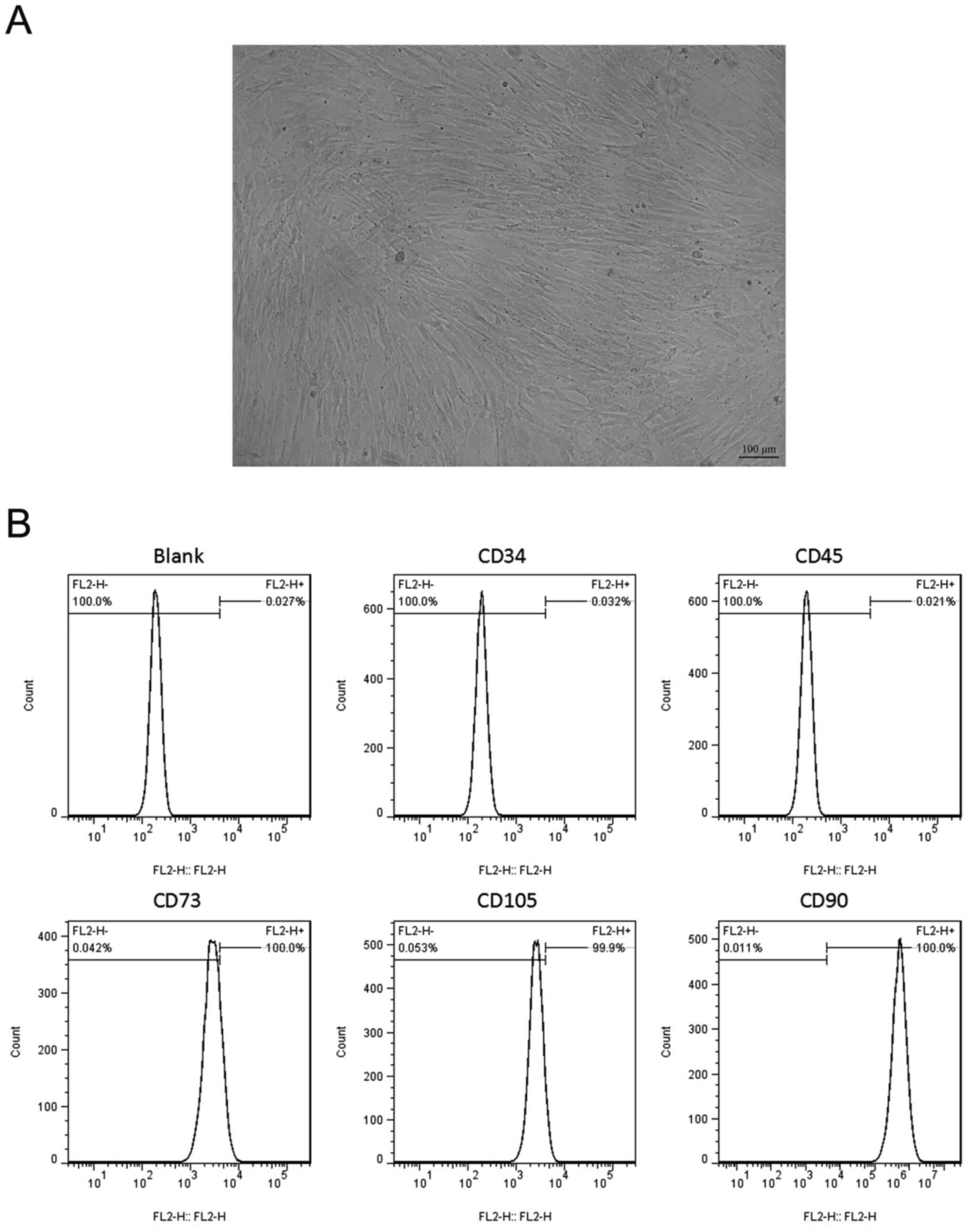
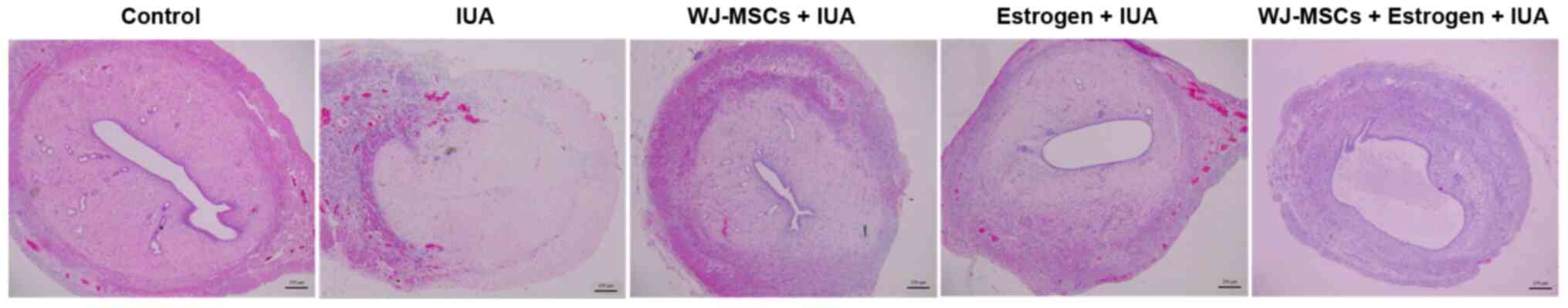
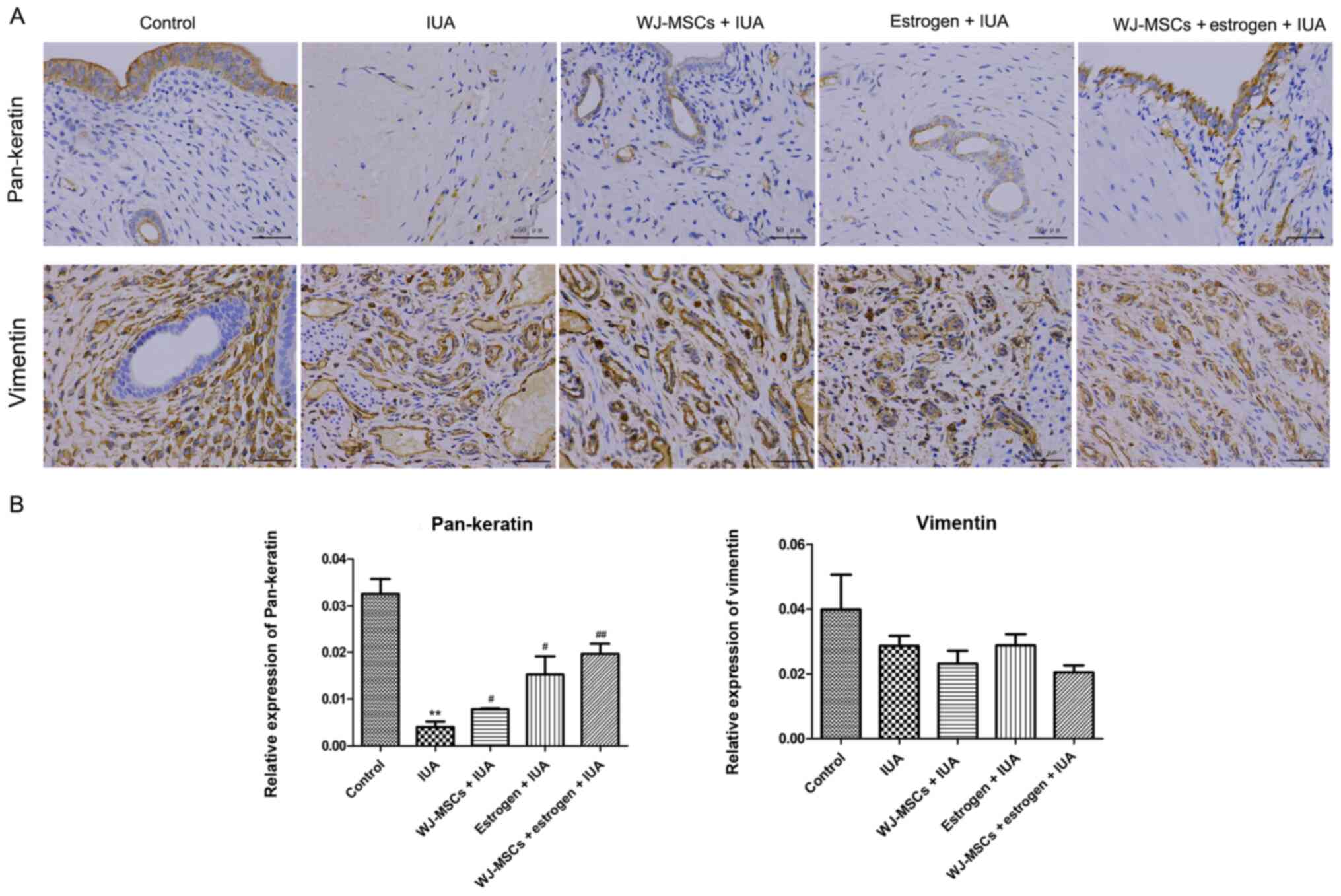
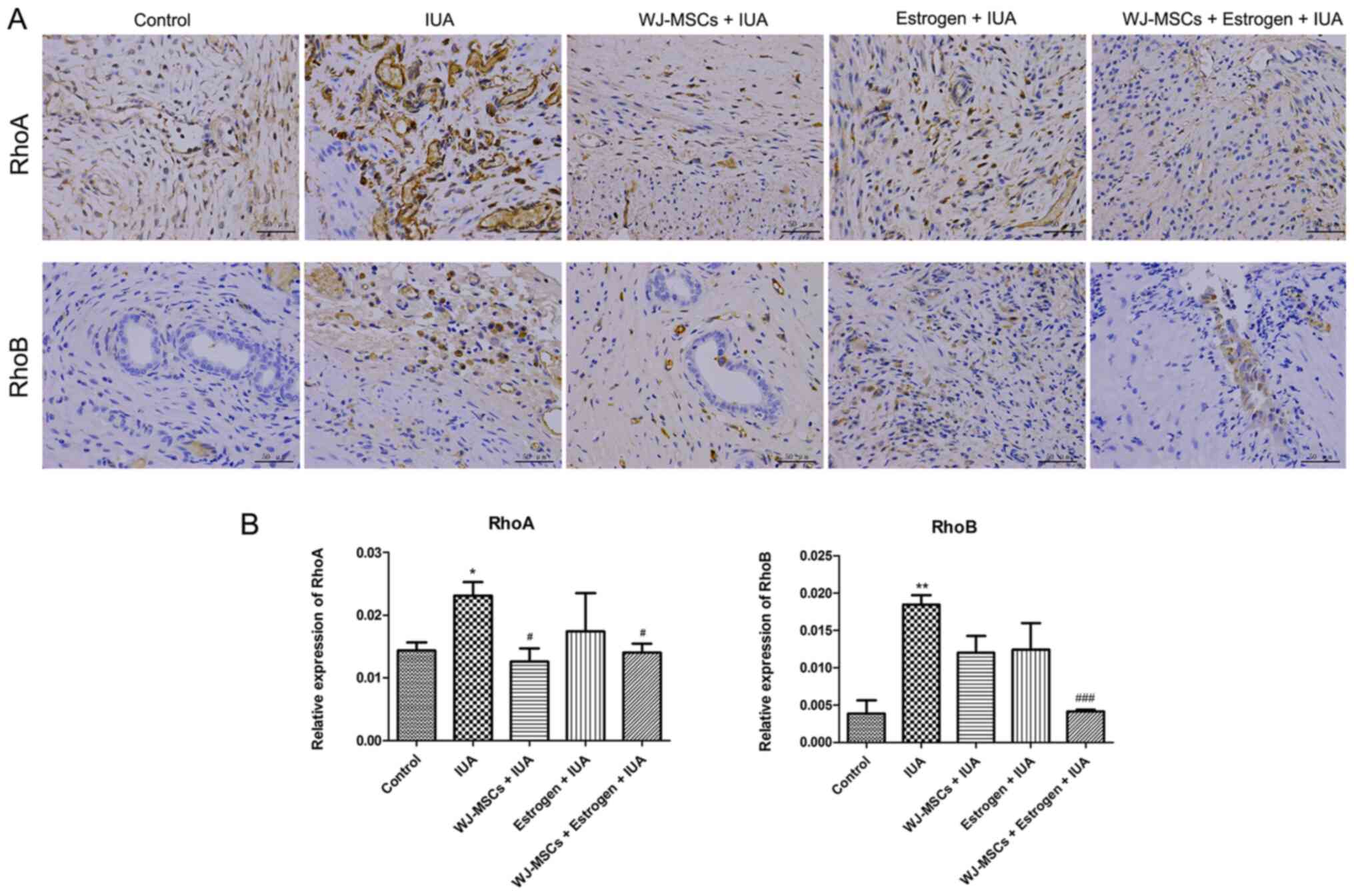
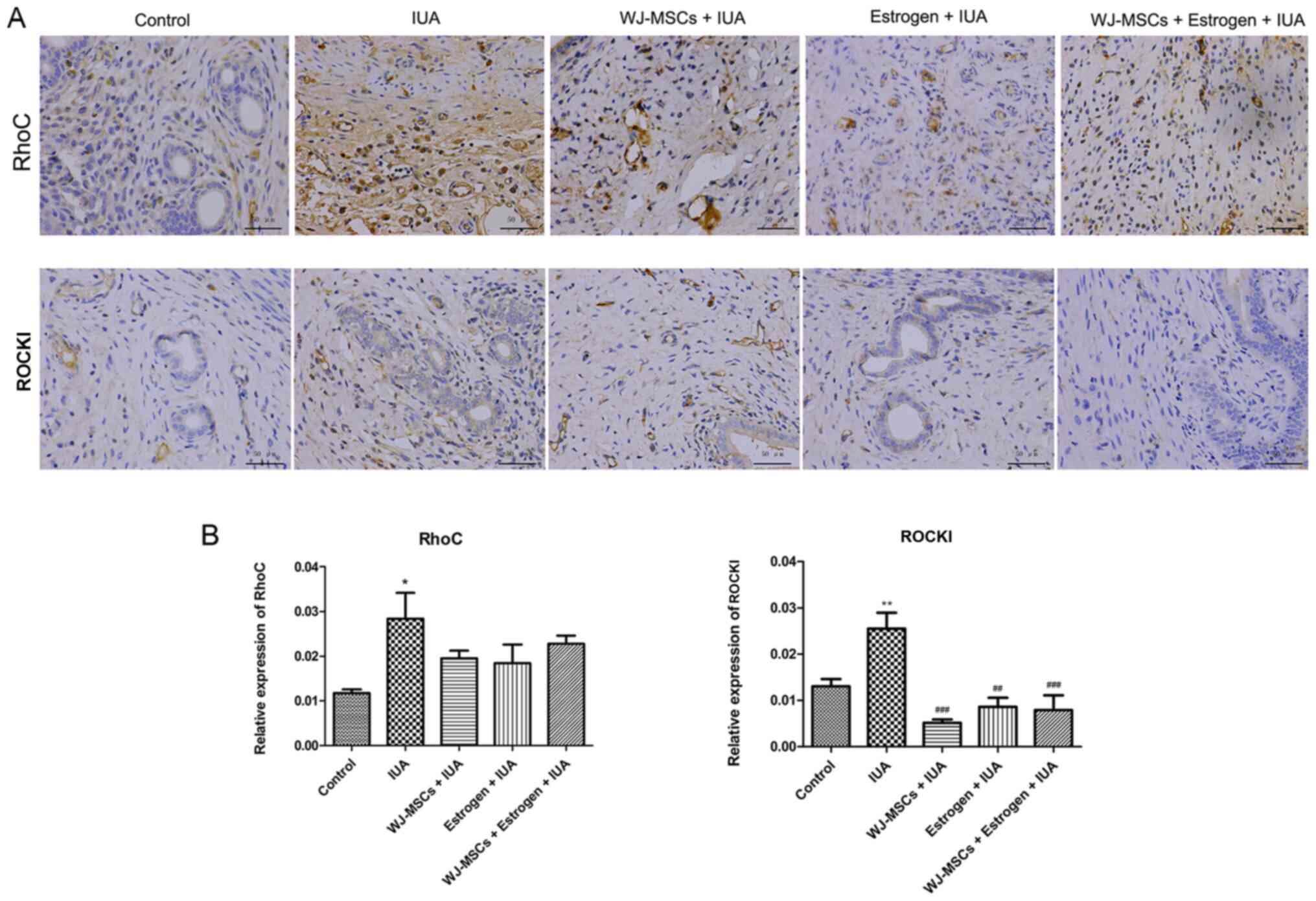
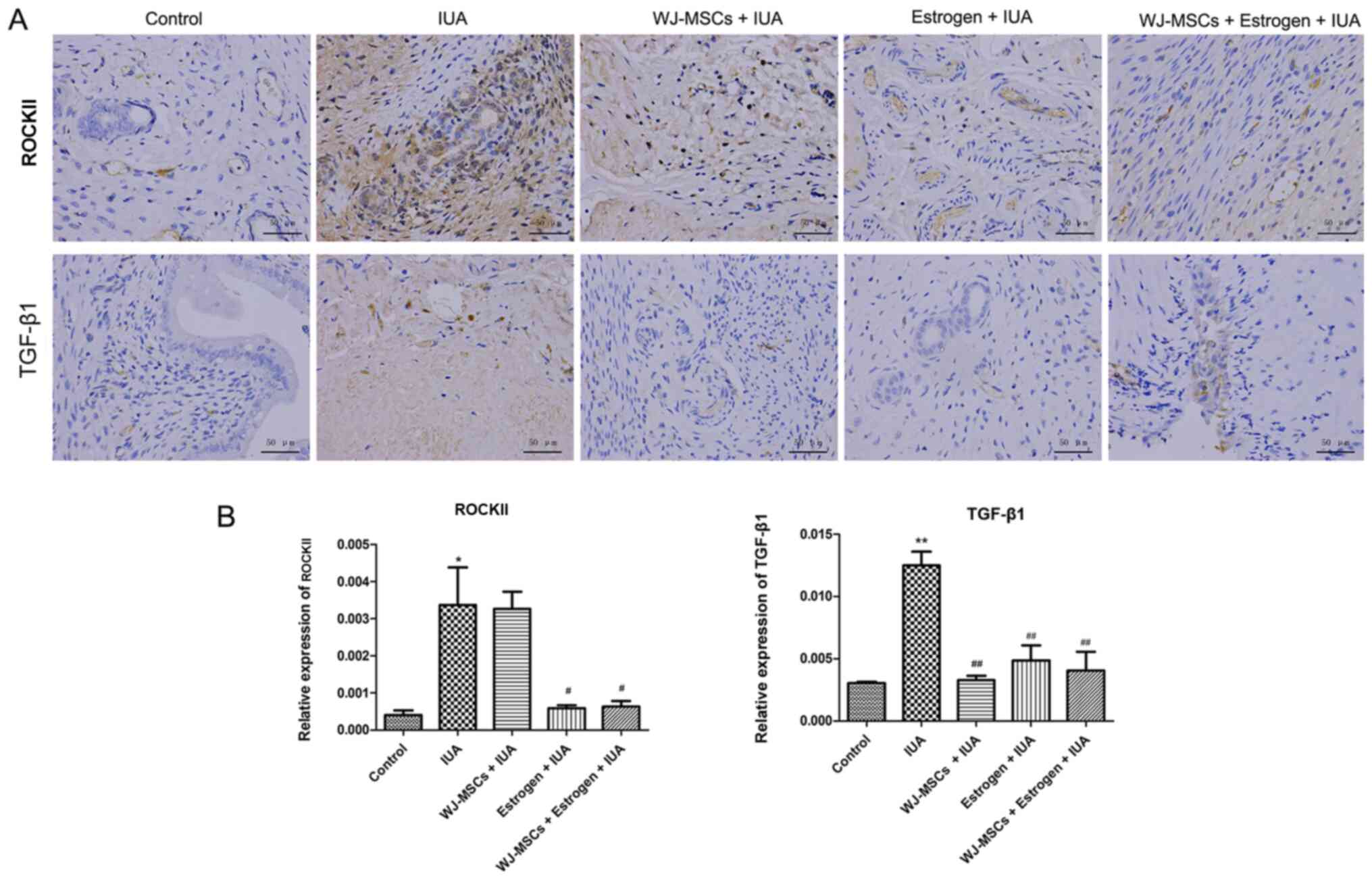
|
1
|
Berman JM: Intrauterine adhesions. Semin
Reprod Med. 26:349–355. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Deans R and Abbott J: Review of
intrauterine adhesions. J Minim Invasive Gynecol. 17:555–569. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Luo S, Shen L and Zhang X: Clearing heat
and activating blood method for intrauterine adhesion. J Changchun
Univ Tradit Chin Med. 1:78–80. 2016.
|
|
4
|
Panayotidis C, Weyers S, Bosteels J and
Herendael BV: Intrauterine adhesions (IUA): Has there been progress
in understanding and treatment over the last 20 years? Gynecol
Surg. 6:197–211. 2009. View Article : Google Scholar
|
|
5
|
Johary J, Xue M, Zhu X, Xu D and Velu PP:
Efficacy of estrogen therapy in patients with intrauterine
adhesions: Systematic review. J Minim Invasive Gynecol. 21:44–54.
2014. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Ebrahim N, Mostafa O, El Dosoky RE, Ahmed
IA, Saad AS, Mostafa A, Sabry D, Ibrahim KA and Farid AS: Human
mesenchymal stem cell-derived extracellular vesicles/estrogen
combined therapy safely ameliorates experimentally induced
intrauterine adhesions in a female rat model. Stem Cell Res Ther.
9:1752018. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Gargett CE and Ye L: Endometrial
reconstruction from stem cells. Fertil Steril. 98:11–20. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Fong CY, Chak LL, Biswas A, Tan JH,
Gauthaman K, Chan WK and Bongso A: Human Wharton's jelly stem cells
have unique transcriptome profiles compared to human embryonic stem
cells and other mesenchymal stem cells. Stem Cell Rev Rep. 7:1–16.
2011. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Taghizadeh RR, Cetrulo KJ and Cetrulo CL:
Wharton's Jelly stem cells: Future clinical applications. Placenta.
32 (Suppl 4):S311–S315. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Chen X, Yang X, Wu R, Chen W, Xie H, Qian
X and Zhang Y: Therapeutic effects of Wharton jelly-derived
mesenchymal stem cells on rat abortion models. J Obstet Gynaecol
Res. 42:972–982. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Yu D, Wong YM, Cheong Y, Xia E and Li TC:
Asherman syndrome - one century later. Fertil Steril. 89:759–779.
2008. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Li J, Du S, Sheng X, Liu J, Cen B, Huang F
and He Y: MicroRNA-29b inhibits endometrial fibrosis by regulating
the Sp1-TGF-β1/Smad-CTGF Axis in a Rat Model. Reprod Sci.
23:386–394. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Liu X, Duan H, Zhang HH, Gan L and Xu Q:
Integrated Data Set of microRNAs and mRNAs Involved in Severe
Intrauterine Adhesion. Reprod Sci. 23:1340–1347. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Defert O and Boland S: Rho kinase
inhibitors: A patent review (2014–2016). Expert Opin Ther Pat.
27:507–515. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Ji H, Tang H, Lin H, Mao J, Gao L, Liu J
and Wu T: Rho/Rock cross-talks with transforming growth
factor-β/Smad pathway participates in lung fibroblast-myofibroblast
differentiation. Biomed Rep. 2:787–792. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Kohno M, Watanabe M, Goto T, et al:
Attenuation Lung Ischemia-Reperfusion Inj Rho-Associated Kinase
Inhib Rat Model Lung Transplant. 20:359–364. 2014.PubMed/NCBI
|
|
17
|
Ivaska J, Pallari H-M, Nevo J and Eriksson
JE: Novel functions of vimentin in cell adhesion, migration, and
signaling. Exp Cell Res. 313:2050–2062. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Magin TM, Vijayaraj P and Leube RE:
Structural and regulatory functions of keratins. Exp Cell Res.
313:2021–2032. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Clark JD, Gebhart GF, Gonder JC, Keeling
ME and Kohn DF: Special Report: The 1996 Guide for the Care and Use
of Laboratory Animals. ILAR J. 38:41–48. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Ren H, Sang Y, Zhang F, Liu Z, Qi N and
Chen Y: Comparative analysis of human mesenchymal stem cells from
umbilical cord, dental pulp, and menstrual blood as sources for
cell therapy. Stem Cells Int. 2016:35165742016. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Zhang L, Li Y, Guan CY, Tian S, Lv XD, Li
JH, Ma X and Xia HF: Therapeutic effect of human umbilical
cord-derived mesenchymal stem cells on injured rat endometrium
during its chronic phase. Stem Cell Res Ther. 9:362018. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Wang J, Ju B, Pan C, Gu Y and Zhang Y, Sun
L, Zhang B and Zhang Y: Application of Bone Marrow-Derived
Mesenchymal Stem Cells in the Treatment of Intrauterine Adhesions
in Rats. Cell Physiol Biochem. 39:1553–1560. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Zheng SX, Wang J, Wang XL, Ali A, Wu LM
and Liu YS: Feasibility analysis of treating severe intrauterine
adhesions by transplanting menstrual blood-derived stem cells. Int
J Mol Med. 41:2201–2212. 2018.PubMed/NCBI
|
|
24
|
Pan X: Regulation of skin keratin
intermediate filament proteins by phosphorylation. Dissertations
& Theses - Gradworks. 2012.
|
|
25
|
Fuchs E and Weber K: Intermediate
filaments: Structure, dynamics, function, and disease. Annu Rev
Biochem. 63:345–382. 1994. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
McIntosh PB, Laskey P, Sullivan K, Davy C,
Wang Q, Jackson DJ, Griffin HM and Doorbar J: E1--E4-mediated
keratin phosphorylation and ubiquitylation: A mechanism for keratin
depletion in HPV16-infected epithelium. J Cell Sci. 123:2810–2822.
2010. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Zhou Q, Wu X, Hu J and Yuan R: Abnormal
expression of fibrosis markers, estrogen receptor α and stromal
derived factor 1/chemokine (C-X-C motif) receptor 4 axis in
intrauterine adhesions. Int J Mol Med. 42:81–90. 2018.PubMed/NCBI
|
|
28
|
Zhu HY, Ge TX, Pan YB and Zhang SY:
Advanced role of hippo signaling in endometrial fibrosis:
Implications for intrauterine adhesion. Chin Med J (Engl).
130:2732–2737. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Guarino M, Tosoni A and Nebuloni M: Direct
contribution of epithelium to organ fibrosis:
Epithelial-mesenchymal transition. Hum Pathol. 40:1365–1376. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Verrecchia F and Mauviel A: Transforming
growth factor-beta and fibrosis. World J Gastroenterol.
13:3056–3062. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Salma U, Xue M, Ali Sheikh MS, Guan X, Xu
B, Zhang A, Huang L and Xu D: Role of transforming frowth factor-β1
and smads signaling pathway in intrauterine adhesion. Mediators
Inflamm. 2016:41582872016. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Xue X, Chen Q, Zhao G, Zhao JY, Duan Z and
Zheng PS: The Overexpression of TGF-β and CCN2 in Intrauterine
Adhesions Involves the NF-κB Signaling Pathway. PLoS One.
10:e01461592015. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Ji H, Tang H, Lin H, Mao J, Gao L, Liu J
and Wu T: Rho/Rock cross-talks with transforming growth
factor-β/Smad pathway participates in lung fibroblast-myofibroblast
differentiation. Biomed Rep. 2:787–792. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Wang S, Sun A, Li L, Zhao G, Jia J, Wang
K, Ge J and Zou Y: Up-regulation of BMP-2 antagonizes
TGF-β1/ROCK-enhanced cardiac fibrotic signalling through activation
of Smurf1/Smad6 complex. J Cell Mol Med. 16:2301–2310. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Zhu J, Nguyen D, Ouyang H, Zhang XH, Chen
XM and Zhang K: Inhibition of RhoA/Rho-kinase pathway suppresses
the expression of extracellular matrix induced by CTGF or TGF-β in
ARPE-19. Int J Ophthalmol. 6:8–14. 2013.PubMed/NCBI
|
|
36
|
Adesanya OO, Zhou J, Samathanam C,
Powell-Braxton L and Bondy CA: Insulin-like growth factor 1 is
required for G2 progression in the estradiol-induced mitotic cycle.
Proc Natl Acad Sci USA. 96:3287–3291. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Hu K, Zhong G and He F: Expression of
estrogen receptors ERalpha and ERbeta in endometrial hyperplasia
and adenocarcinoma. Int J Gynecol Cancer. 15:537–541. 2005.
View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Osteen KG and Anderson TL: Effect of
estrogen on human endometrial epithelial cell growth and
differentiation in vitro. Steroids. 56:279–283. 1991. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Zhou Q, Wu X, Dai X, Rui Y and Qi H: The
different dosages of estrogen affect endometrial fibrosis and
receptivity, but not SDF-1/CXCR4 axis in the treatment of
intrauterine adhesions. Gynecol Endocrinol. 34:49–55. 2018.
View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Jiang H and Dai Y: Estrogen attenuates the
TGFβ1-induced conversion of primary penile tunica albuginea
fibroblasts into myofibroblasts and inhibits collagen production
and myofibroblast contraction by modulating the Smad and Rho/ROCK
signaling pathways. Transl Androl Urol. 4:463–472. 2015.
|
|
41
|
Luft FC: ERα on the cell membrane helps
the heart. J Mol Med (Berl). 92:1–3. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Lee TM, Lin SZ and Chang NC: Membrane ERα
attenuates myocardial fibrosis via RhoA/ROCK-mediated actin
remodeling in ovariectomized female infarcted rats. J Mol Med
(Berl). 92:43–51. 2014. View Article : Google Scholar : PubMed/NCBI
|