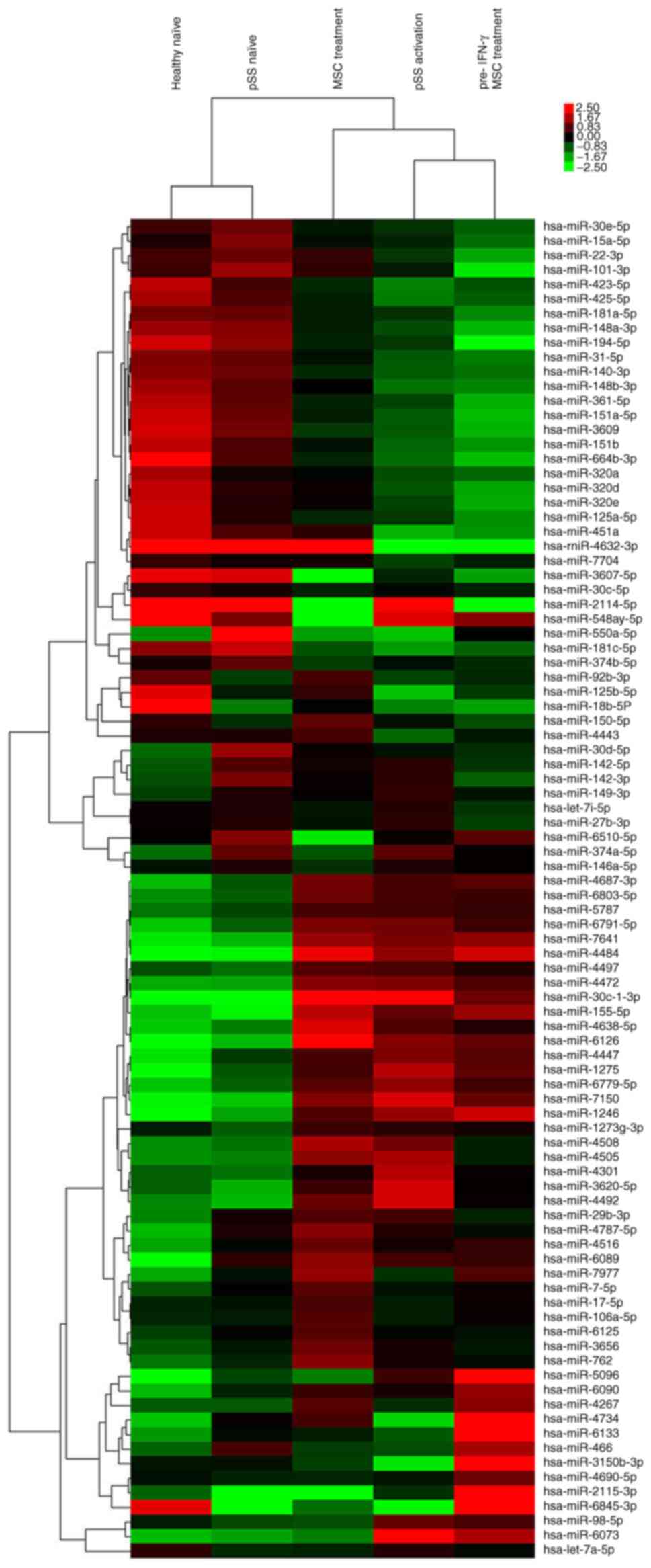
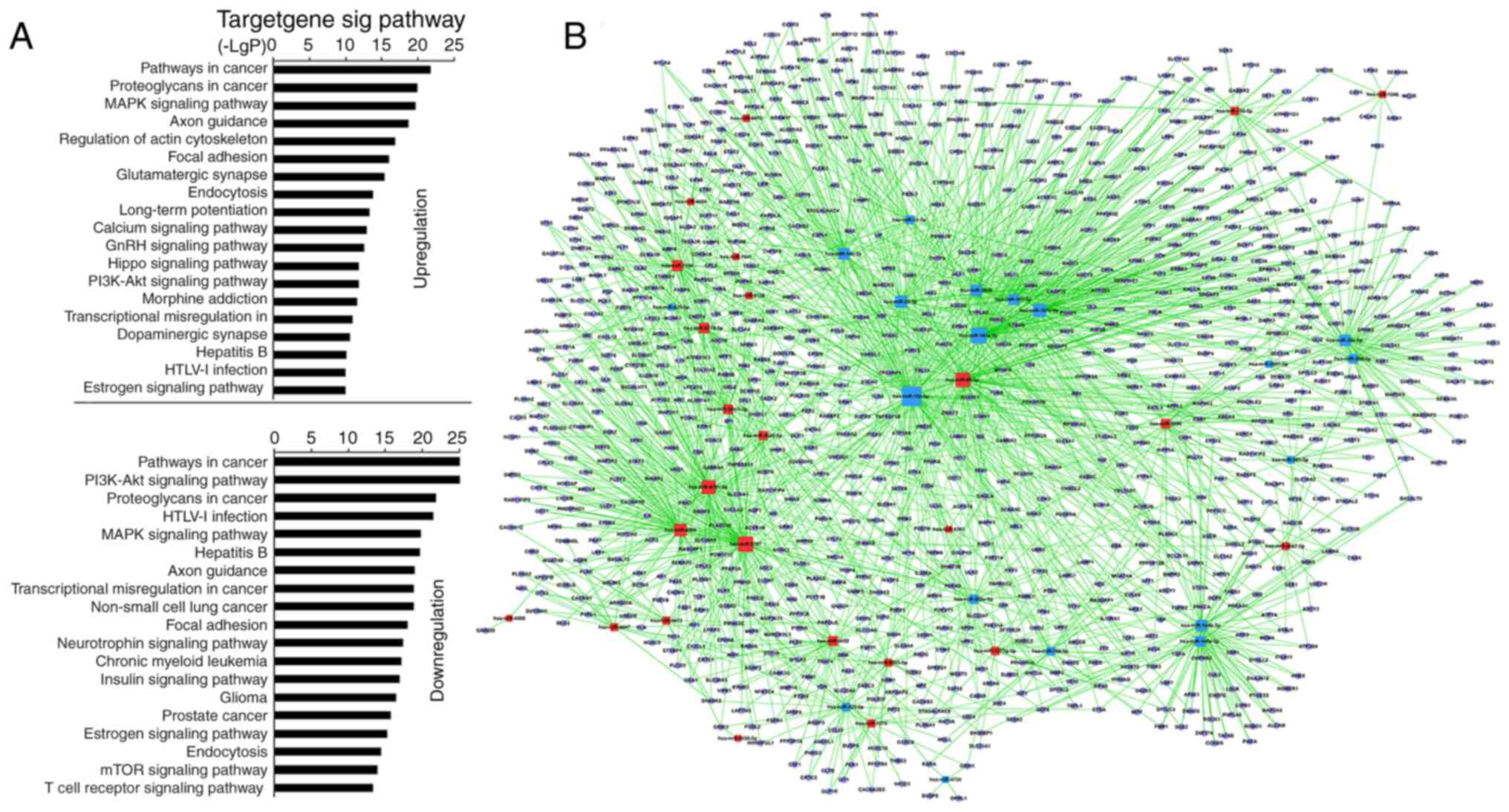
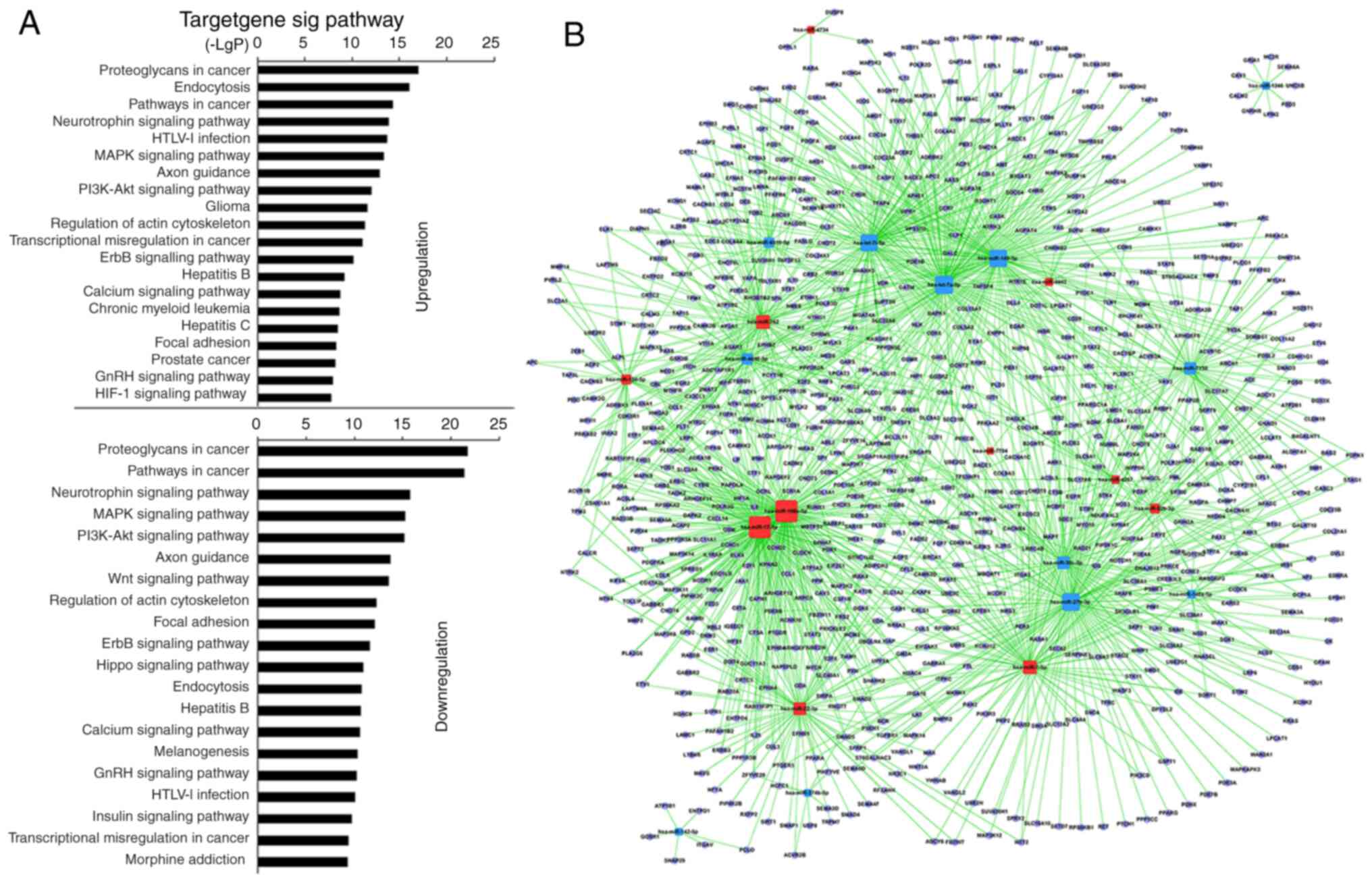
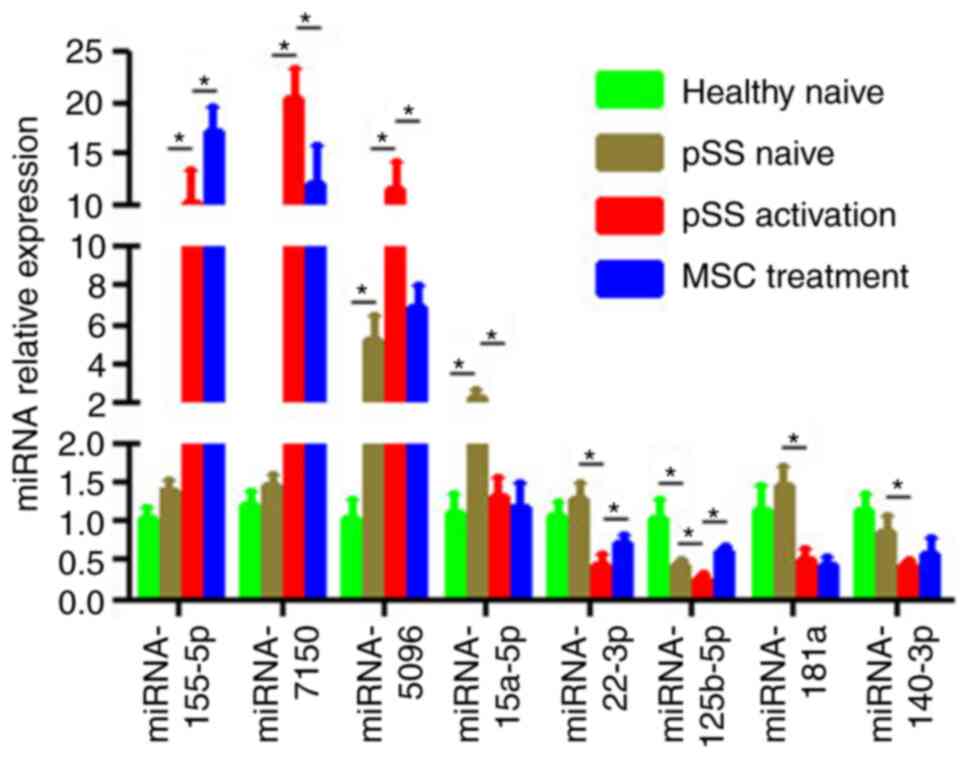
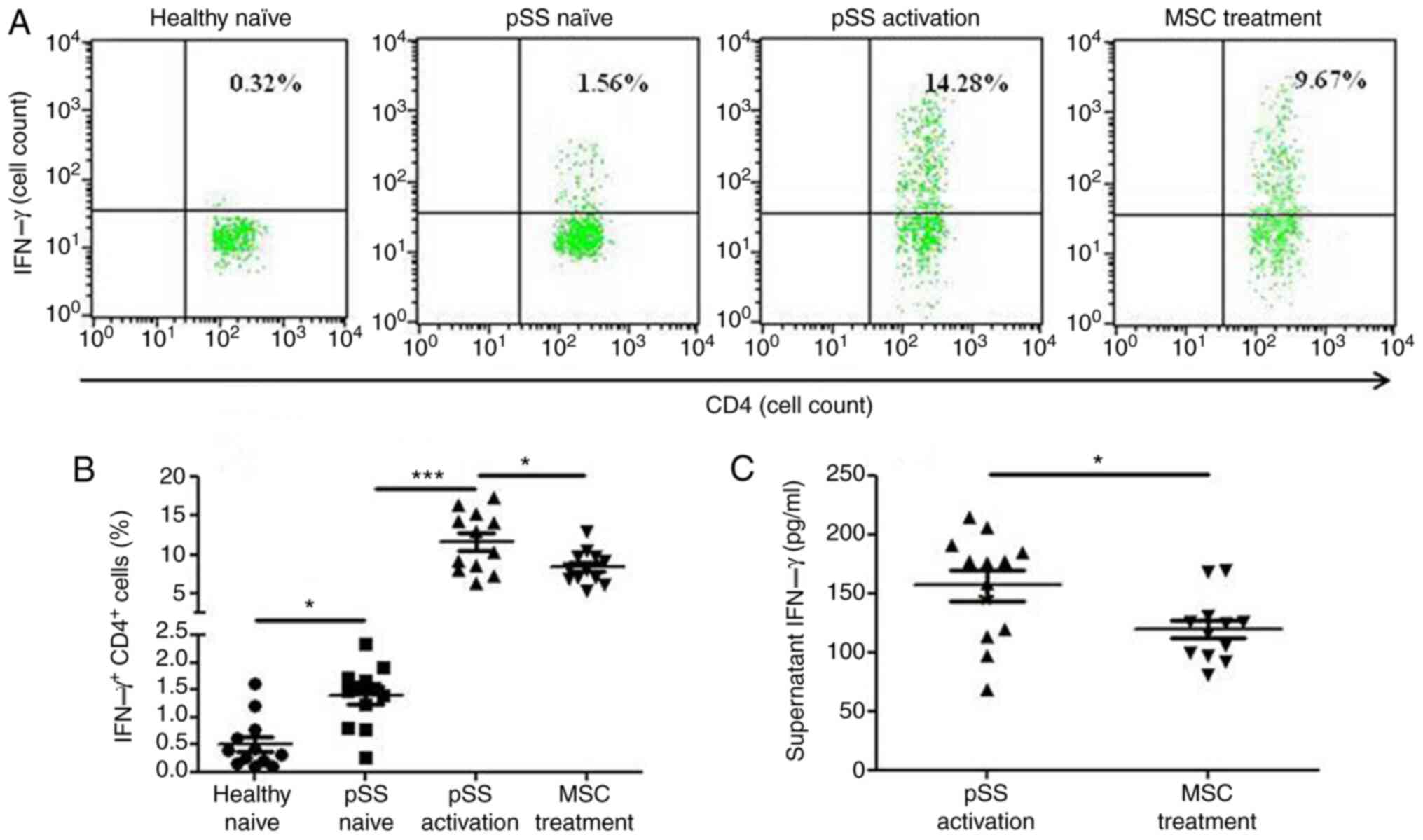
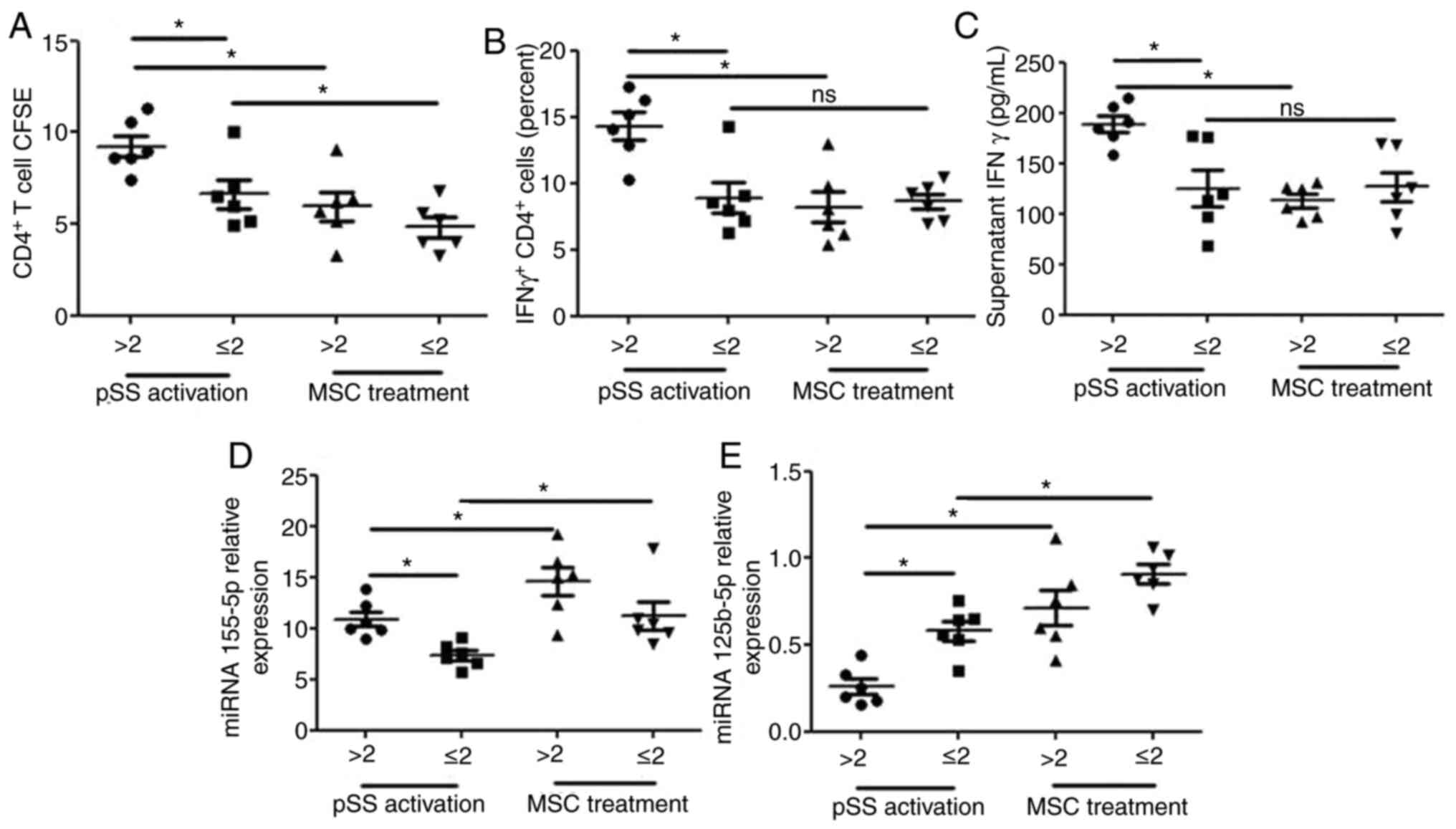
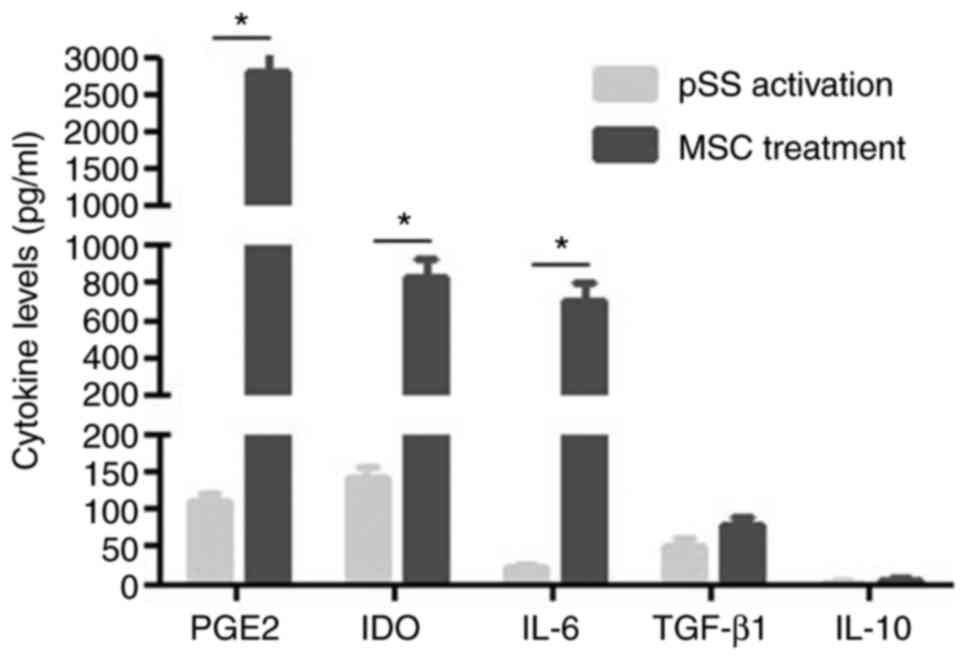
|
1
|
Mariette X and Criswell LA: Primary
sjögren's syndrome. N Engl J Med. 378:931–939. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Brito-Zerón P, Sisó-Almirall A, Bové A,
Kostov BA and Ramos-Casals M: Primary Sjögren syndrome: An update
on current pharmacotherapy options and future directions. Expert
Opin Pharmacother. 14:279–289. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Xu J, Wang D, Liu D, Fan Z, Zhang H, Liu
O, Ding G, Gao R, Zhang C, Ding Y, et al: Allogeneic mesenchymal
stem cell treatment alleviates experimental and clinical sjögren
syndrome. Blood. 120:3142–3151. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Weiss AR and Dahlke MH: Immunomodulation
by mesenchymal stem cells (MSCs): Mechanisms of action of living,
apoptotic, and dead MSCs. Front Immunol. 10:11912019. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
De Miguel MP, Fuentes-Julián S,
Blázquez-Martínez A, Pascual CY, Aller MA, Arias J and
Arnalich-Montiel F: Immunosuppressive properties of mesenchymal
stem cells: Advances and applications. Curr Mol Med. 12:574–591.
2012. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Podshivalova K and Salomon DR: MicroRNA
regulation of T-lymphocyte immunity: Modulation of molecular
networks responsible for T-cell activation, differentiation, and
development. Crit Rev Immunol. 33:435–476. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Alevizos I, Alexander S, Turner RJ and
Illei GG: MicroRNA expression profiles as biomarkers of minor
salivary gland inflammation and dysfunction in sjögren's syndrome.
Arthritis Rheum. 63:535–544. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Campard D, Lysy PA, Najimi M and Sokal EM:
Native umbilical cord matrix stem cells express hepatic markers and
differentiate into hepatocyte-like cells. Gastroenterology.
134:833–848. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Tondreau T, Lagneaux L, Dejeneffe M,
Delforge A, Massy M, Mortier C and Bron D: Isolation of BM
mesenchymal stem cells by plastic adhesion or negative selection:
Phenotype, proliferation kinetics and differentiation potential.
Cytotherapy. 6:372–379. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Vitali C, Bombardieri S, Jonsson R,
Moutsopoulos HM, Alexander EL, Carsons SE, Daniels TE, Fox PC, Fox
RI, Kassan SS, et al: Classification criteria for sjögren's
syndrome: A revised version of the European criteria proposed by
the American-European consensus group. Ann Rheum Dis. 61:554–558.
2002. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Seror R, Ravaud P, Bowman SJ, Baron G,
Tzioufas A, Theander E, Gottenberg JE, Bootsma H, Mariette X and
Vitali C; EULAR Sjögren's Task Force, : EULAR sjögren's task force.
EULAR sjogren's syndrome disease activity index: Development of a
consensus systemic disease activity index for primary sjogren's
syndrome. Ann Rheum Dis. 69:1103–1109. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Bolstad BM, Irizarry RA, Astrand M and
Speed TP: A comparison of normalization methods for high density
oligonucleotide array data based on variance and bias.
Bioinformatics. 19:185–193. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Pfaffl MW, Lange IG, Daxenberger A and
Meyer HH: Tissue-specific expression pattern of estrogen receptors
(ER): Quantification of ER alpha and ER beta mRNA with real-time
RT-PCR. APMIS. 109:345–355. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
The Gene Ontology Consortium: The gene
ontology resource: 20 years and still GOing strong. Nucleic Acids
Res. 47:D330–D338. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Rouillard AD, Gundersen GW, Fernandez NF,
Wang Z, Monteiro CD, McDermott MG and Ma'ayan A: The harmonizome: A
collection of processed datasets gathered to serve and mine
knowledge about genes and proteins. Database (Oxford).
2016:baw1002016. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Jassal B, Matthews L, Viteri G, Gong C,
Lorente P, Fabregat A, Sidiropoulos K, Cook J, Gillespie M, Haw R,
et al: The reactome pathway knowledgebase. Nucleic Acids Res.
48:D498–D503. 2020.PubMed/NCBI
|
|
17
|
Kanehisa M, Goto S, Kawashima S, Okuno Y
and Hattori M: The KEGG resource for deciphering the genome.
Nucleic Acids Res. 32:D277–D280. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Yi M, Horton JD, Cohen JC, Hobbs HH and
Stephens RM: WholePathwayScope: A comprehensive pathway-based
analysis tool for high-throughput data. BMC Bioinformatics.
19:302006. View Article : Google Scholar
|
|
19
|
Draghici S, Khatri P, Tarca AL, Amin K,
Done A, Voichita C, Georgescu C and Romero R: A systems biology
approach for pathway level analysis. Genome Res. 17:1537–1545.
2007. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Kim JH, Lee YT, Hong JM and Hwang YI:
Suppression of in vitro murine T cell proliferation by human
adipose tissue-derived mesenchymal stem cells is dependent mainly
on cyclooxygenase-2 expression. Anat Cell Biol. 46:262–271. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Schurgers E, Kelchtermans H, Mitera T,
Geboes L and Matthys P: Discrepancy between the in vitro and
in vivo effects of murine mesenchymal stem cells on T-cell
proliferation and collagen-induced arthritis. Arthritis Res Ther.
12:R312010. View
Article : Google Scholar : PubMed/NCBI
|
|
22
|
Ryan JM, Barry F, Murphy JM and Mahon BP:
Interferon-gamma does not break, but promotes the immunosuppressive
capacity of adult human mesenchymal stem cells. Clin Exp Immunol.
149:353–363. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Krampera M, Cosmi L, Angeli R, Pasini A,
Liotta F, Andreini A, Santarlasci V, Mazzinghi B, Pizzolo G,
Vinante F, et al: Role for interferon-gamma in the immunomodulatory
activity of human bone marrow mesenchymal stem cells. Stem Cells.
24:386–398. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Wang Y, Chen X, Cao W and Shi Y:
Plasticity of mesenchymal stem cells in immunomodulation:
Pathological and therapeutic implications. Nat Immunol.
15:1009–1016. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Teteloshvili N, Smigielska-Czepiel K,
Kroesen BJ, Brouwer E, Kluiver J, Boots AM and van den Berg A:
T-Cell activation induces dynamic changes in miRNA expression
patterns in CD4 and CD8 T-cell subsets. Microrna. 4:117–122. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Lind EF and Ohashi PS: Mir-155, a central
modulator of T-cell responses. Eur J Immunol. 44:11–15. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Smigielska-Czepiel K, van den Berg A,
Jellema P, van der Lei RJ, Bijzet J, Kluiver J, Boots AM, Brouwer E
and Kroesen BJ: Comprehensive analysis of miRNA expression in
T-cell subsets of rheumatoid arthritis patients reveals defined
signatures of naive and memory tregs. Genes Immun. 15:115–125.
2014. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Zhang Y, Feng ZP, Naselli G, Bell F,
Wettenhall J, Auyeung P, Ellis JA, Ponsonby AL, Speed TP, Chong MM
and Harrison LC: MicroRNAs in CD4+ T cell subsets are
markers of disease risk and T cell dysfunction in individuals at
risk for type 1 diabetes. J Autoimmun. 68:52–61. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Schaffert SA, Loh C, Wang S, Arnold CP,
Axtell RC, Newell EW, Nolan G, Ansel KM, Davis MM, Steinman L and
Chen CZ: Mir-181a-1/b-1 modulates tolerance through opposing
activities in selection and peripheral t cell function. J Immunol.
195:1470–1479. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Grigoryev YA, Kurian SM, Hart T,
Nakorchevsky AA, Chen C, Campbell D, Head SR, Yates JR III and
Salomon DR: MicroRNA regulation of molecular networks mapped by
global microRNA, mRNA, and protein expression in activated T
lymphocytes. J Immunol. 187:2233–2243. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Rossi RL, Rossetti G, Wenandy L, Curti S,
Ripamonti A, Bonnal RJ, Birolo RS, Moro M, Crosti MC, Gruarin P, et
al: Distinct microRNA signatures in human lymphocyte subsets and
enforcement of the naive state in CD4+ T cells by the microRNA
miR-125b. Nat Immunol. 12:796–803. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Domingo-Gonzalez R, Katz S, Serezani CH,
Moore TA, Levine AM and Moore BB: Prostaglandin E2-induced changes
in alveolar macrophage scavenger receptor profiles differentially
alter phagocytosis of pseudomonas aeruginosa and Staphylococcus
aureus post-bone marrow transplant. J Immunol. 190:5809–5817. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Oshima H and Oshima M: The role of
PGE2-associated inflammatory responses in gastric cancer
development. Semin Immunopathol. 35:139–150. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Domingo-Gonzalez R, Wilke CA, Huang SK,
Laouar Y, Brown JP, Freeman CM, Curtis JL, Yanik GA and Moore BB:
Transforming growth factor-β induces microRNA-29b to promote murine
alveolar macrophage dysfunction after bone marrow transplantation.
Am J Physiol Lung Cell Mol Physiol. 308:L86–L95. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Davis BN, Hilyard AC, Lagna G and Hata A:
SMAD proteins control DROSHA-mediated microRNA maturation. Nature.
454:56–61. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Curtale G, Mirolo M, Renzi TA, Rossato M,
Bazzoni F and Locati M: Negative regulation of toll-like receptor 4
signaling by IL-10-dependent microRNA-146b. Proc Natl Acad Sci USA.
110:11499–11504. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Schaefer JS, Montufar-Solis D, Vigneswaran
N and Klein JR: Selective upregulation of microRNA expression in
peripheral blood leukocytes in IL-10-/-mice precedes expression in
the colon. J Immunol. 187:5834–5841. 2011. View Article : Google Scholar : PubMed/NCBI
|