Introduction
Cervical cancer has been the second most common
gynecological malignancy in women since the 1990s (1) and results in >300,000 deaths per
year worldwide (2). Radiotherapy is
the most frequently used therapeutical modality for cervical cancer
(3); however, due to the occurrence
of radioresistance (RR), the therapeutic efficacy of radiotherapy
is far from satisfactory (4).
Therefore, exploring effective and alternative methods to enhance
radiosensitivity (RS) is crucial to the clinical radiotherapy of
cervical cancer.
Intratumoral hypoxia is a typical characteristic of
numerous invasive solid tumors (5).
It has been proposed that hypoxic tumor cells achieve RR more
easily compared with those in a normoxic environment, and
therefore, tumor hypoxia has been associated with poor survival
outcomes (6,7). Although the application of novel
radiosensitizers, including Ro 90–7501, cisplatin (or nedaplatin)
and curcumin, have enhanced the radiotherapy efficacy of cervical
cancer (8–11), the toxic effects, such as acute
hematological and gastrointestinal toxicity, induced by these
chemicals are difficult to avoid.
Most tumors contain O2- and
nutrient-deprived compartments (12). The sterilization of tumor cells
under hypoxic conditions requires radiation doses that are three
times higher than those for cells under normoxic conditions
(13). Hyperbaric O2
therapy is an efficient method to cope with the phenomenon of
hypoxia by enhancing the O2 load in tumor areas and
enhance the response to ionizing radiation (IR) (14–16).
Nordsmark et al (17)
demonstrated that increased tumor partial O2 pressure
enhanced IR response in mammary carcinoma. Pietrofesa et al
(18) reported that exposure to the
combination of IR + O2 increased DNA damage and cell
death compared with the individual exposures to IR or O2
alone. However, how a high concentration of O2
pretreatment affects the efficacy of cervical cancer radiotherapy
has not been fully elucidated.
Tumor cells in a hypoxic region adapt to low
O2 tension conditions by activating survival factors and
cell signaling pathways, such as hypoxia-inducible factor 1α
(HIF-1α) and VEGF (19). HIF-1α is
one of the most recognized transcription factors used by hypoxic
cells in the harsh tumor microenvironment, and it activates >100
downstream genes required for tumor survival and progression,
including VEGF, erythropoietin and c-MYC (20). The Akt and ERK signaling pathways
are two major upstream regulators of HIF-1α. Previous studies have
reported that there is an association between the Akt/ERK signaling
pathways and HIF-1α/VEGF expression, which may affect the cellular
reaction to radiation, O2 tension and chemical stimuli
(21–23). However, the roles that HIF-1α, VEGF
and the Akt/ERK signaling pathways serve in cervical cancer
radiotherapy remain unclear.
The aim of present study was to investigate how
hypoxia, normoxia and hyperoxia affect the efficacy of radiotherapy
for cervical cancer and whether hyperoxia can reverse
hypoxia-induced IR.
Materials and methods
Cell line and culture
Human cervical cancer HeLa cells were purchased from
The Cell Bank of Type Culture Collection of the Chinese Academy of
Sciences and stored in the laboratory of Department of Obstetrics
and Gynecology in the First Hospital of Jilin University. Cells
were cultured in DMEM (Thermo Fisher Scientific, Inc.) supplemented
with 10% FBS (HyClone; Cytiva) and 1% antibiotics
(penicillin/streptomycin; Invitrogen; Thermo Fisher Scientific,
Inc.) in a humidified 37°C incubator with 5% CO2.
Normoxia, hypoxia, reoxygenation and
hyperoxia
For the normoxic culture (Normo), HeLa cells were
maintained at 37°C in a humidified O2 (20%),
CO2 (5%) and N2 (75%) atmosphere (Thermo
Fisher Scientific, Inc.). For the hypoxic culture (Hypo), cells
were cultured at 37°C in a humidified O2 (1%),
CO2 (5%) and N2 (94%) incubator (Thermo
Fisher Scientific, Inc.) for 12 h. For reoxygenation (Reoxy), cells
were cultured under hypoxic conditions at 37°C for 12 h followed by
normoxic conditions at 37°C for 2 h. For the hyperoxic culture
(Hyper), cells were placed in a CO2 (5%) and
O2 (95%) chamber (ProOX P360 O2 controller
and chamber; BioSpherix, Ltd.) at 37°C for 2 h (24). In order to investigate the effects
of hypoxia, reoxygenation and hyperoxia on IR, HeLa cells were
divided into the following treatment groups: i) IR-alone (IR); ii)
hypoxia followed by IR (Hypo + IR); iii) hyperoxia followed by IR
(Hyper + IR); iv) hypoxia followed by reoxygenation and IR (Hypo +
Reoxy + IR); and v) hypoxia followed by hyperoxia and IR (Hypo +
Hyper + IR).
IR
Monolayer HeLa cells were seeded into six-well
plates (1×105 cells per well) or 10-cm dishes
(6×105 cells per dish) to achieve 70% confluency and
were then exposed to X-rays at a dose rate of 1 Gy/min in a X-RAD
320 (Precision X-RAD; Precision X-Ray, Inc.). A moderate X-ray dose
of 6 Gy was applied, according to previous studies (25,26).
Following IR, cells were cultured for 24 h at 37°C in a humidified
O2 (20%), CO2 (5%) and N2 (75%)
atmosphere until cell proliferation or apoptosis tests were
conducted. For other experiments (e.g. western blotting), cells
were harvested immediately. Control groups were treated similarly;
however, these cells did not undergo IR.
Plasmid and cell transfection
Coding sequence of VEGF165 was
synthesized (Sangon Biotech Co., Ltd.) and cloned into pcDNA3.1
vector (Invitrogen; Thermo Fisher Scientific, Inc.) to form a
pCDNA3.1-VEGF plasmid. For the forced expression of
VEGF165 (feVEGF) in HeLa cells, 2 µg plasmid DNA and
blank control vectors were transfected using
Lipofectamine® 3000 (Invitrogen), according to the
manufacturer's protocol. Western blotting or IR (feVEGF + IR) were
conducted at 48 h post-transfection.
VEGF/VEGFR stimulation and
neutralization tests
For the VEGF stimulation test, 10 ng/ml recombinant
human VEGF165 (reVEGF, cat. no. ab9571; Abcam) (27) was added to cell culture media (37°C,
5% CO2) at 2 h prior to IR (reVEGF + IR). For the
ligand-receptor neutralization test, 25 µg/ml VEGF, 30 µg/ml VEGFR1
or 500 ng/ml VEGFR2 neutralization antibodies (cat. nos. AF293,
AF321 and MAB3572; R&D Systems China Co., Ltd.) were added to
cell culture media (37°C, 5% CO2) at 2 h prior to IR, as
previously described, and the manufacturer's protocol (28,29).
Human IgG1 κ (cat. no. ab206200; Abcam) was used as the isotype
control (IgG1 κ + Hypo + IR).
Inhibitors
To inhibit HIF-1α function, 100 nM echinomycin
(30) (Merck KGaA) was added to
cell culture media (37°C, 5% CO2) 2 h prior to hypoxic
exposure. To block the Akt and ERK signaling pathways, 50 µM
LY294002 and 20 µM U0126 (Beyotime Institute of Biotechnology) were
added to cell culture media (37°C, 5% CO2) 2 h prior to
IR as previously described (31,32).
ELISA
The level of secreted VEGF in the medium was
quantified using a human VEGF Quantikine ELISA kit (cat. no. DVE00;
R&D Systems, Inc.). According to the manufacturer's protocol,
the supernatant of ≥1×105 HeLa cells (or treated HeLa
cells) were collected and centrifuged at 4°C and 1,000 × g for 15
min to remove debris and particulates. The absorbance at 450 nm
(A450), which is proportional to VEGF concentration, was
determined using an ELISA reader (Synergy H1 Hybric Multi-Mode
Reader; BioTek Instruments, Inc.).
VEGFR2 was quantified using a human VEGF R2/KDR
Quantikine ELISA kit (cat. no. DVR200; R&D Systems, Inc.).
According to the manufacturer's protocol, ≥1×105 cells
were washed 3 times in cold PBS and resuspended in 1 ml lysis
buffer. The lysate was incubated at RT for 1 h with gentle
agitation and centrifuged at 1,000 × g for 15 min at RT.
A450 was determined as aforementioned.
Cell proliferation inhibition
assay
Cell proliferation was determined using a standard
spectrophotometric MTT assay. HeLa cells were seeded at a density
of 5,000 cells/well into a 96-well plate and underwent Hypo, Reoxy,
Hyper and/or IR, as aforementioned. Subsequently, cells were
cultured at 37°C for another 24 h in normal O2 condition
and incubated at 37°C for 4 h with MTT solution (5 mg/ml,
Sigma-Aldrich; Merck KGaA). The cell supernatant was carefully
aspirated, and the precipitate was dissolved using 200 µl DMSO
(Sigma-Aldrich; Merck KGaA) at 37°C for 10 min. A490 was
determined as aforementioned. The cell proliferation inhibition
rate (%) of each group was defined as follows: (A490 of
the experimental group-A490 of the control
group)/A490 of the control group.
Apoptosis assay
Apoptosis was determined using an Annexin V-FITC
Apoptosis Detection kit (cat. no. 556547; BD Biosciences).
According to the manufacturer's protocol, 1×105 HeLa
cells were washed with cold PBS and centrifuged at RT and 200 × g
for 5 min. The cell pellet was resuspended and incubated in 50 µl
Annexin V solution containing 5 µl PI and 5 µl Annexin V-FITC at RT
for 15 min. Data acquisition and analysis were performed using flow
cytometry (FACScan; BD Biosciences) and FlowJo software (version
10.0; FlowJo LLC). Results for early and late apoptosis were
combined as the total amount of apoptosis.
Western blotting
Total protein was extracted from HeLa or transfected
HeLa cells using RIPA lysis buffer (Beyotime Institute of
Biotechnology) supplemented with cocktail protease inhibitors
[Roche Diagnostics (Shanghai) Co., Ltd.] and quantified using a
Bradford dye binding assay kit (Beyotime Institute of
Biotechnology) according to the manufacturer's protocol. Each
sample was loaded with 20 µg protein/lane on a 12% gel and
separated via SDS-PAGE. Samples were electrophoretically
transferred to a PVDF membrane (EMD Millipore), which was then
blocked with 5% skimmed milk in PBS at RT for 1 h and incubated
with the following primary antibodies against VEGF (1:1,500; cat.
no. AF-293-SP; R&D Systems, Inc.), HIF-1α (1:1,000; cat. no.
ab1; Abcam), ERK1/2 (1:1,000; cat. no. 4696; Cell Signaling
Technology, Inc.), phosphorylated (p-)ERK1/2 (1:1,000; cat. no.
4370; Cell Signaling Technology, Inc.), Akt (1:1,000; cat. no.
4685; Cell Signaling Technology, Inc.), p-Akt (1:1,000; cat. no.
4060; Cell Signaling Technology, Inc.) and β-actin (1:1,500; cat.
no. sc-47778; Santa Cruz Biotechnology, Inc.) at 37°C for 1 h.
Membranes were then incubated with goat anti-mouse, goat
anti-rabbit or rabbit anti-goat (1:3,000; cat. nos. sc-2005,
sc-2004 and sc-2768, respectively; Santa Cruz Biotechnology, Inc.)
IgG peroxidase-conjugated secondary antibodies at 37°C for 45 min.
Immunoreactivity was visualized using an ECL detection kit (EMD
Millipore). Densitometric analysis was performed using Quantity One
software (version 4.6; Bio-Rad Laboratories, Inc.).
Statistical analysis
All experiments were repeated ≥3 times. Data were
analyzed using SPSS software (version 17.0; SPSS, Inc.) and
presented as the mean ± SD. Statistical analysis was performed
using Student's unpaired t-test or one-way ANOVA followed by
Fisher's Least Significant Difference, Bonferroni or Sidak post-hoc
tests. P<0.05 was considered to indicate a statistically
significant difference.
Results
Hyperoxic treatment enhances RS in
hypoxic HeLa cells
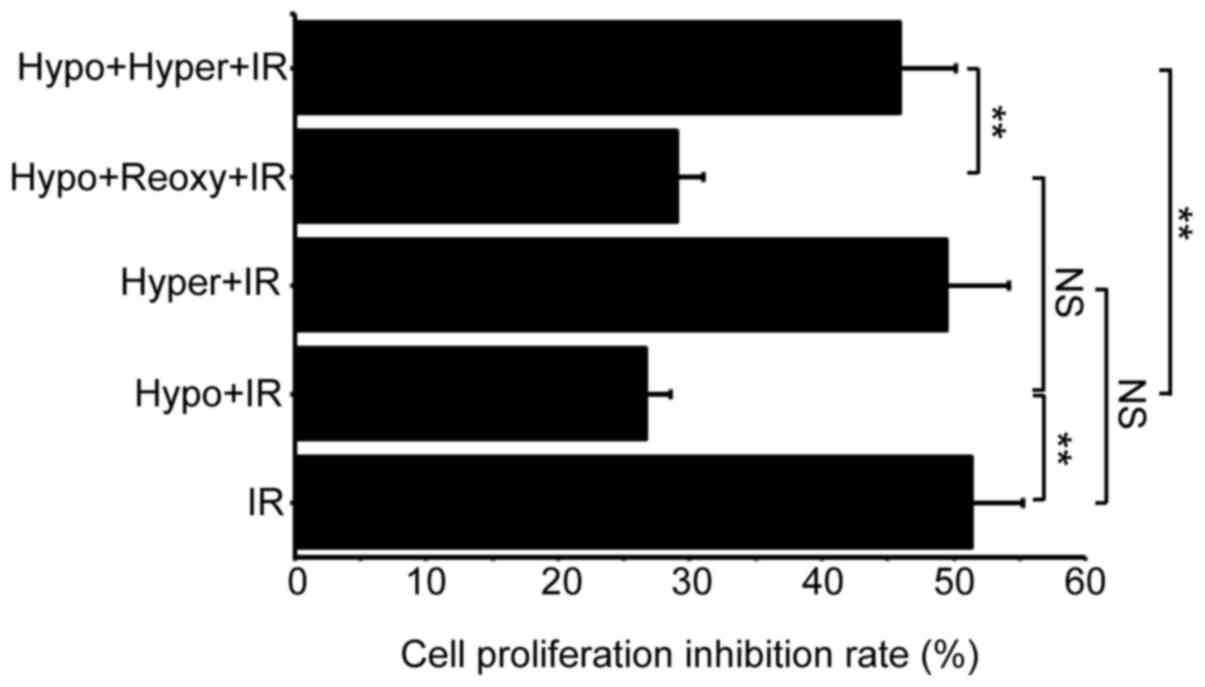
The cellular inhibition rates were as follows: IR,
51.36%; Hypo + IR, 26.61%; Hyper + IR, 49.42%; Hypo + Reoxy + IR,
28.94%; and Hypo + Hyper + IR,45.96% (Fig. 1). These data indicated that hypoxia
promoted RR in HeLa cells and that hyperoxia sensitized the hypoxic
HeLa cells to IR; however, reoxygenation did not sensitize cells to
IR. There is a possibility that extending the reoxygenation time
may achieve the same effect as the cells being exposed to hyperoxia
for 2 h. However, this hypothesis requires further experimental
verification.
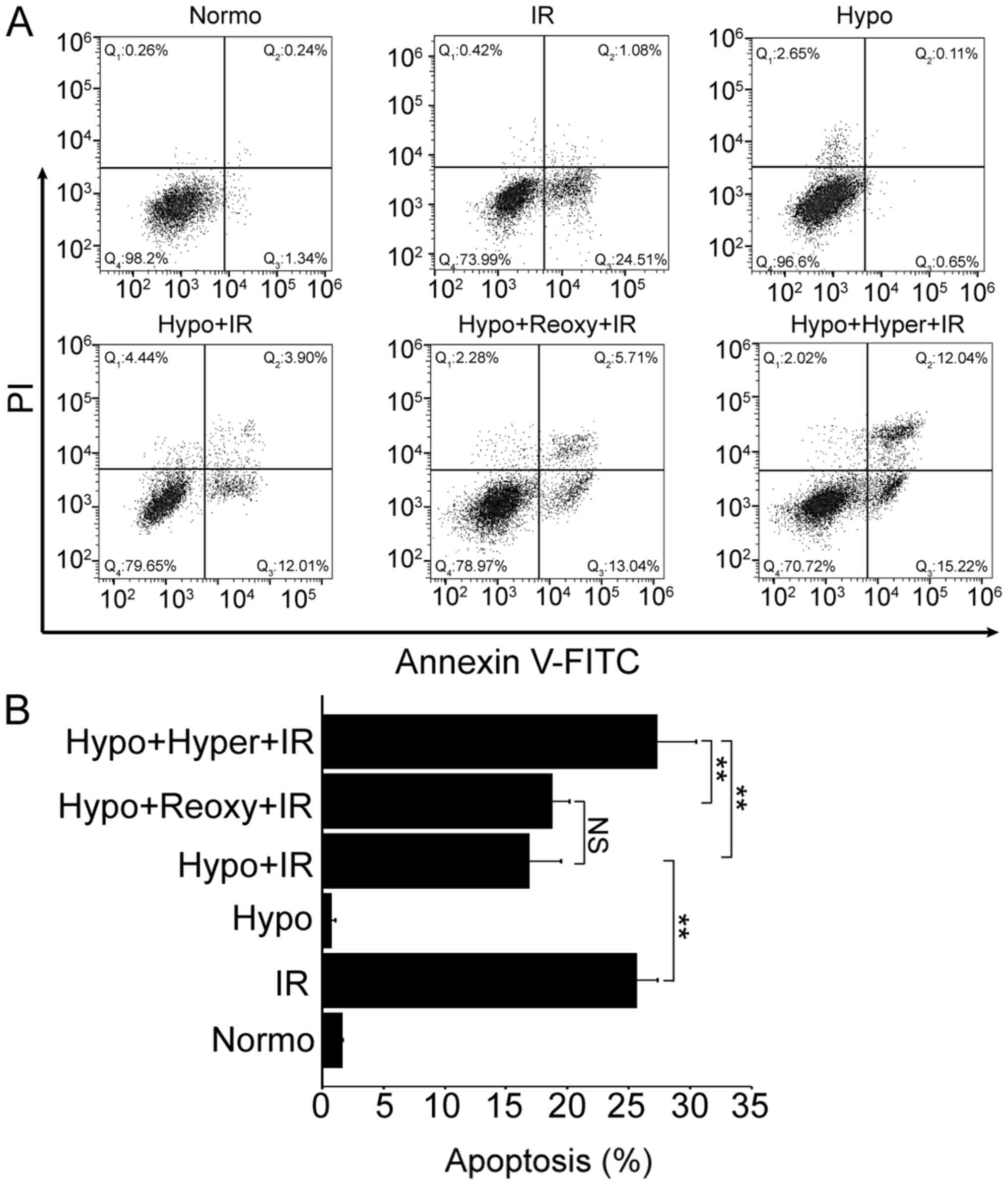
Hypoxia pretreatment significantly decreased the
cellular apoptosis rates compared with the IR-alone (15.91% vs.
25.59%; Fig. 2). However, hypoxic
cells which subsequently underwent hyperoxia exhibited a
significant increase in RS and cellular apoptosis rate (27.26%)
compared with cells which underwent reoxygenation (18.75%) or
hypoxia-alone (15.91%).
Hyperoxia downregulates
hypoxia-stimulated HIF-1α and VEGF expression
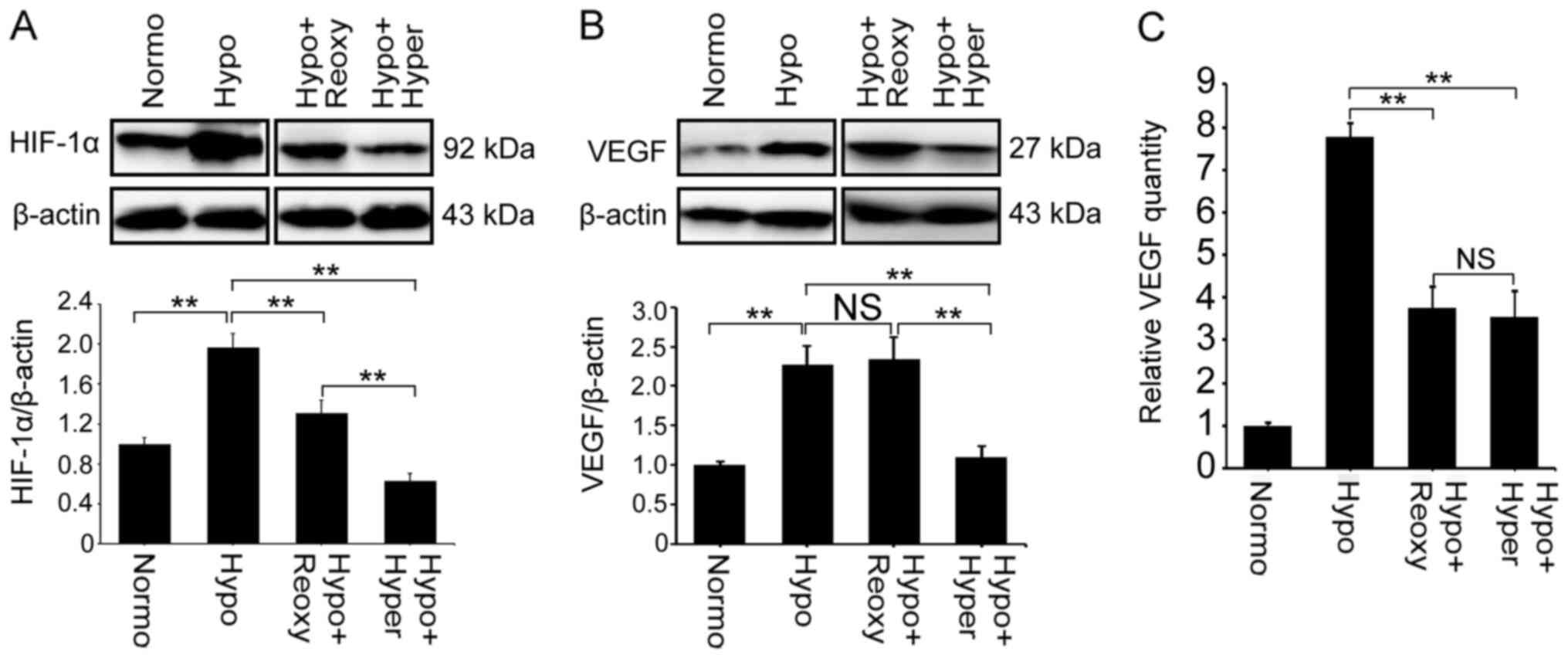
Hypoxia has been reported to stimulate VEGF
expression, particularly in cancer cells (33). Western blotting showed that hypoxia
treatment upregulated HIF-1α levels 2-fold and VEGF levels 2.3-fold
compared with cells cultured in normal O2 (Fig. 3A and B). Reoxygenation decreased
HIF-1α levels in hypoxic cells to a similar level as the untreated
cells but did not affect VEGF levels (Fig. 3A and B). By contrast, hyperoxia
decreased both the HIF-1α and VEGF levels in hypoxic cells
(Fig. 3A and B). ELISA indicated
that reoxygenation or hyperoxia decreased secretory VEGF levels in
hypoxic cells (Fig. 3C).
VEGF enhances RR
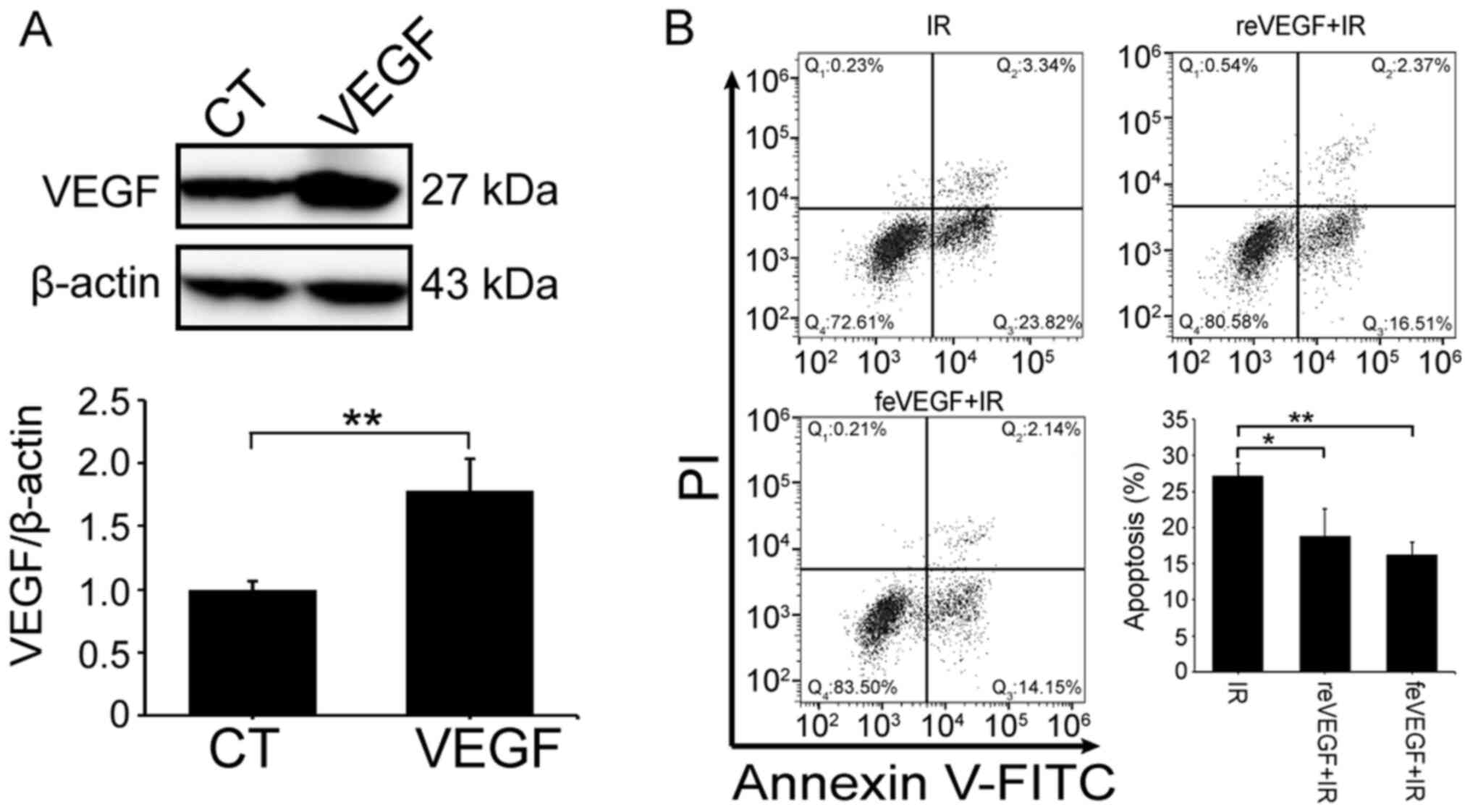
VEGF has been reported to be involved in
hypoxia-induced RR in HeLa cells (34). Therefore, the current study treated
HeLa cells with 10 ng/ml reVEGF or feVEGF to HeLa cells. Western
blotting showed that VEGF levels increased 1.62-fold in HeLa
transfectants (Fig. 4A). Cell
apoptosis tests were conducted at 24 h post-IR; the apoptosis rates
of the reVEGF and feVEGF treatment groups were 18.88% and 16.29%,
respectively, compared with 27.16% in the untreated group
(IR-alone) (Fig. 4B). These results
suggested that reVEGF and feVEGF elevated the RR of HeLa cells.
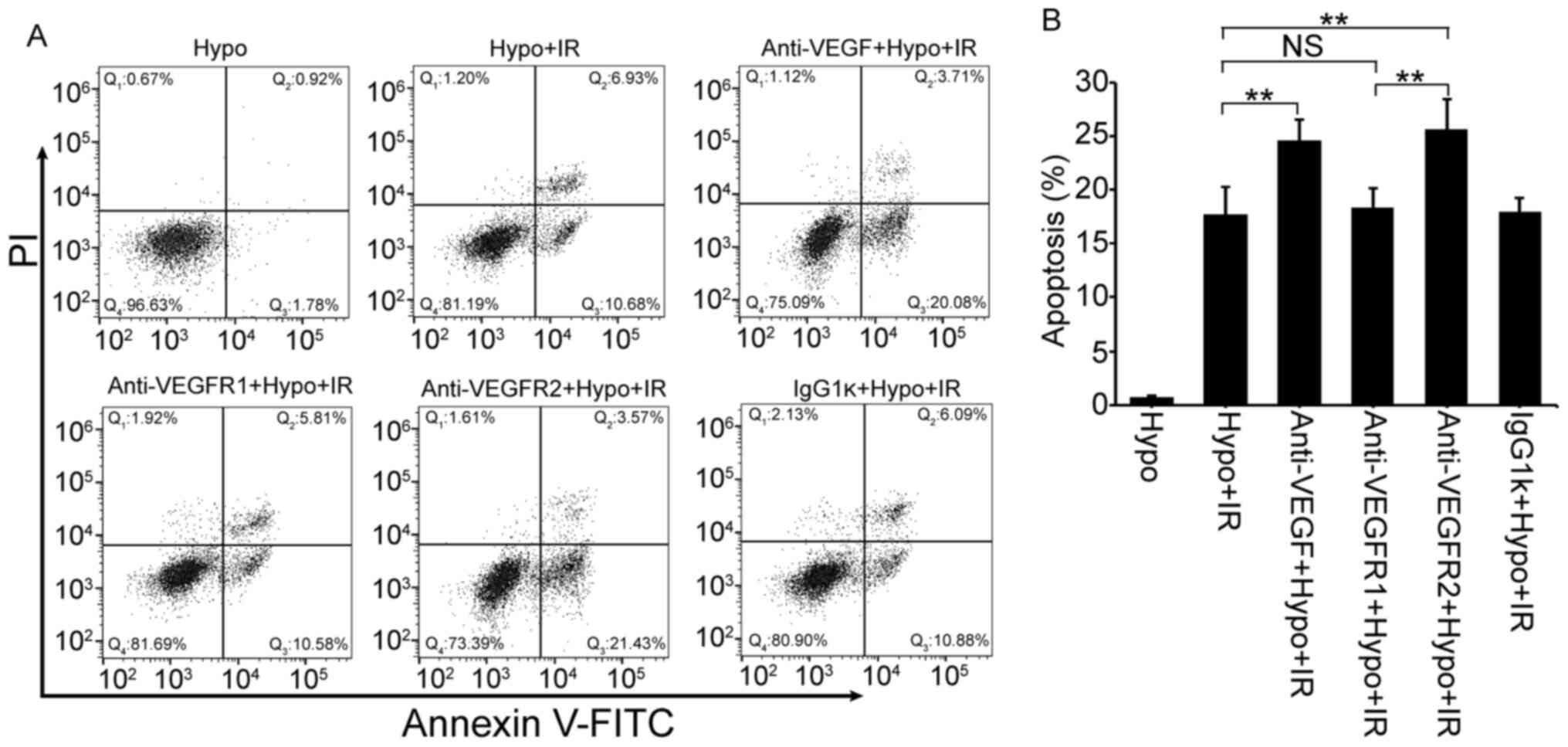
Blocking the VEGF-VEGFR2 interaction
decreases hypoxia-induced RR
VEGFRs are required for VEGF-based signal transfer
(35). The current study used VEGF,
VEGFR1 and VEGFR2 neutralization antibodies to verify whether the
VEGF-VEGFR interaction is implicated in the hypoxia-induced RR. As
presented in Fig. 5, treatment with
VEGF and VEGFR2 neutralization antibodies significantly promoted
IR-induced apoptosis (23.79 and 25.00%) compared with the Hypo + IR
group (17.61%). However, administration of VEGFR1 neutralization
antibodies did not significantly affect IR-induced apoptosis. These
data indicated that VEGF may induce RR through interaction with
VEGFR2, but not VEGFR1.
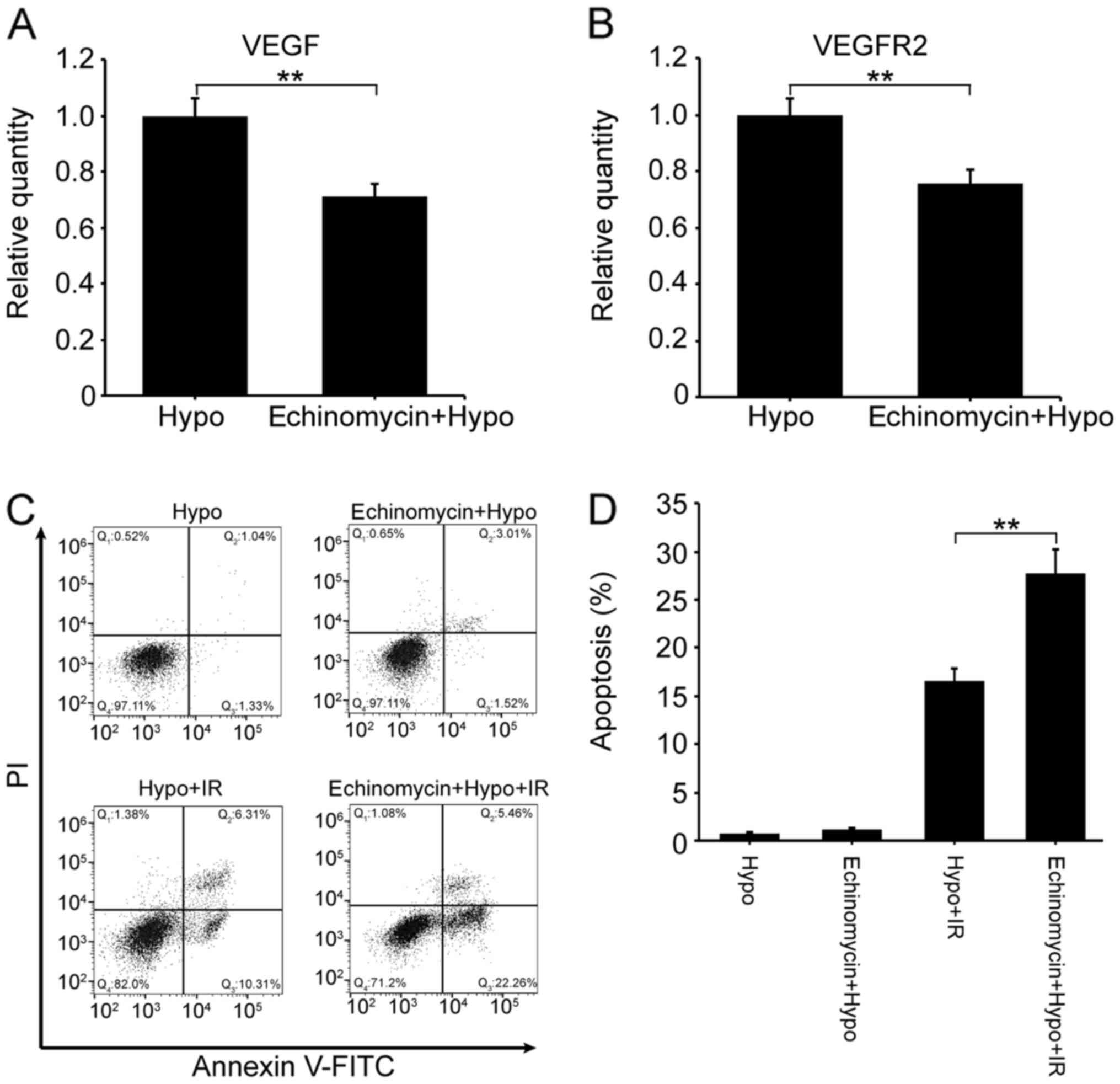
HIF-1α inactivation suppresses VEGF
and VEGFR2 expression, and increases RS in hypoxic HeLa cells
Since the aforementioned results demonstrated that
HIF-1α levels paralleled those of VEGF following hypoxic and
hyperoxic treatments, it was hypothesized that HIF-1α may interact
with VEGF and function collectively to induce RR in hypoxic HeLa
cells. Therefore, the HIF-1α inhibitor echinomycin was added to the
cell media prior to hypoxia treatment. The results demonstrated
that the levels of VEGF (Fig. 6A)
and VEGFR2 (Fig. 6B) in cells
pretreated with echinomycin were significantly decreased compared
with those in hypoxic cells. Furthermore, the results revealed that
the administration of echinomycin promoted IR-induced apoptosis in
hypoxic HeLa cells (Fig. 6C). The
cellular apoptosis rate in the Hypo + echinomycin + IR group
increased by 1.7-fold compared with the Hypo + IR group (27.72% vs.
16.62%, respectively; Fig. 6D).
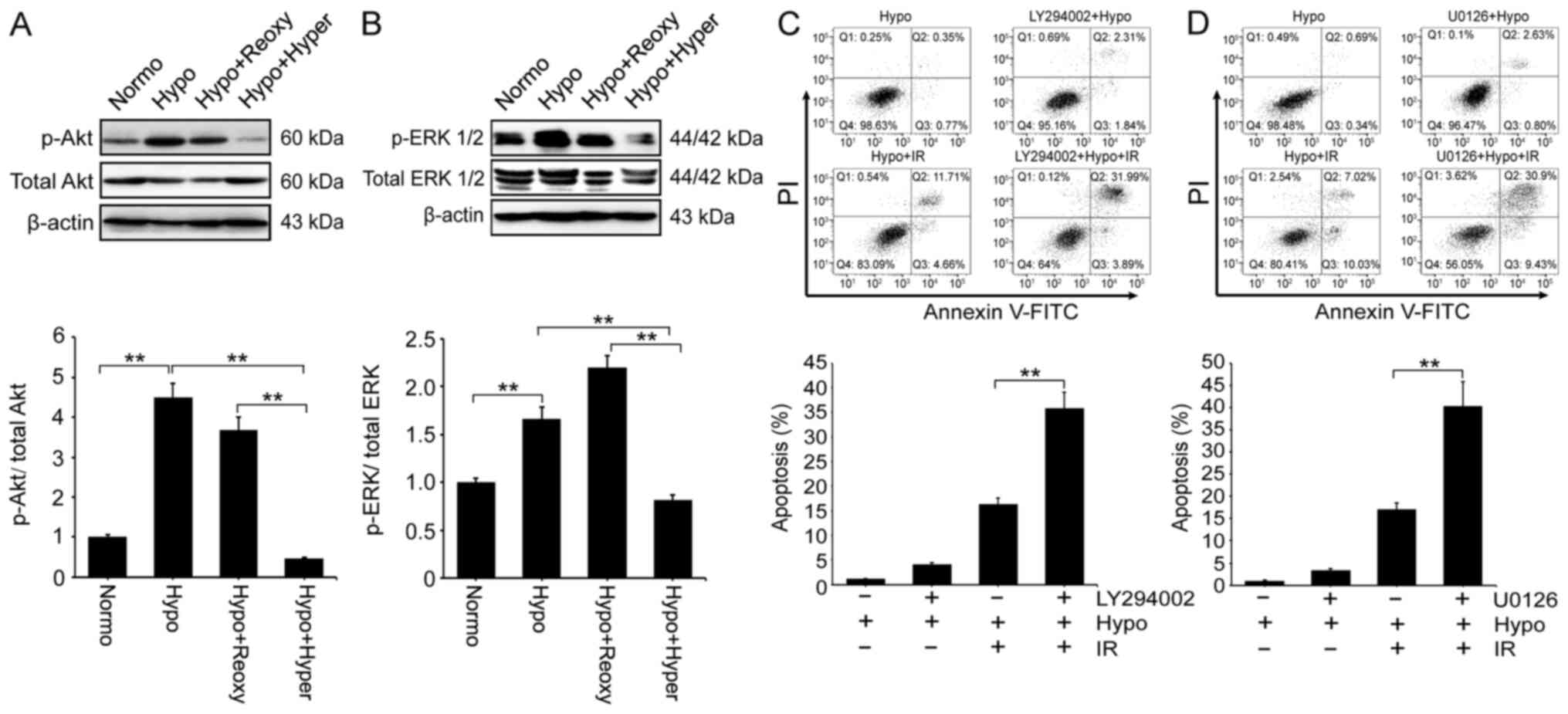
Hyperoxia enhances RS by suppressing
the Akt and ERK signaling pathways
It has been reported that hyperactivation of Akt and
ERK is associated with RR (36–38).
The results of the current model detected the activation of Akt and
ERK1/2. Western blotting results indicated that the levels of p-Akt
(Fig. 7A) and p-ERK1/2 (Fig. 7B) were significantly increased in
hypoxic HeLa cells compared with those in cells cultured in normal
O2, whereas hyperoxia exposure significantly suppressed
the phosphorylation of Akt and ERK1/2 induced by hypoxia.
Furthermore, LY294002 (Fig. 7C) and
U0126 (Fig. 7D) were used to block
the PI3K/Akt and MAPK/ERK pathways, respectively, and the results
demonstrated that hypoxia-induced RR was decreased and the
IR-induced apoptosis was increased using the inhibitors.
Discussion
Most, if not all, solid tumors exist in an anoxic
microenvironment due to insufficient blood supply (39). To survive in hypoxic conditions,
tumor cells gradually adapt to hypoxic stress through regulation of
numerous genes, such as HIF-1α, VEGF, insulin-like growth factor α
and transforming growth factor α, which are associated with tumor
cell proliferation, apoptosis, chemotherapy resistance and
radiotherapy resistance (40).
Tumor hypoxia is a major contributing factor for the
failure of anticancer therapies, including radiotherapy (7). It has been proposed that cells under
hypoxic stress are more likely to generate RR due to the lack of
O2 as a source of radiation-induced radicals and DNA
damage (41). By contrast,
increasing O2 pressure within the tumor can overcome
hypoxia-induced RR (42).
Pietrofesa et al (18)
demonstrated that the combination of hyperoxia and IR increased
cell death and DNA damage compared with hyperoxia or IR alone in
mouse lung cells.
Mammalian cells respond to O2 deprivation
by mobilizing multiple intracellular signal transduction pathways,
including HIF family-depending pathways (5). HIF-VEGF axis-mediated cell survival
and angiogenesis are implicated in the radiation response in
tumors. Fu et al (34)
reported that hypoxic conditions enhance the RR of HeLa cells
dependent on HIF-1α by elevating VEGF expression and inhibiting p53
expression. Gorski et al (43) indicated that IR-induced VEGF
expression may contribute to the protection of tumor blood vessels
from cytotoxicity-mediated radiation and, thereby, to tumor RR. The
results of the current study confirmed that hypoxia induced RR in
cervical cancer cells via the activation of HIF-1α and VEGF.
Administration of recombinant VEGF or introduction of exogenous
VEGF increased RR in HeLa cells. By contrast, inactivation of a
HIF-1α or blockage of the VEGF-VEGFR interaction abrogated
hypoxia-induced RR. According to the present results, it was
hypothesized that tumor cells may secrete VEGF to build blood
vessels and increase O2 and nutrients supply in order to
survive IR-mediated cytotoxicity.
VEGFR1 and VEGFR2 have highly homologous structures;
however, their biological functions differ (44). VEGFR2 is implicated in pathological
and physiological angiogenesis, whereas VEGFR1 acts as a decoy
receptor that limits VEGFR2 activation (45,46).
Ding et al (47) reported
that silencing of HIF-1α promoted cell apoptosis and inhibited cell
invasion by downregulating VEGF and VEGFR2 expression in thyroid
cancer. The results of the current study demonstrated that VEGF may
function as an IR-resistant factor through interaction with VEGFR2.
Additionally, inhibition of HIF-1α may abrogate hypoxia-induced RR
through the downregulation of VEGF and VEGFR2 expression. The
present findings indicated that the HIF-1α-VEGF axis may serve an
important role in regulating the response of hypoxic cervical
cancer cells to IR.
The results of the present study demonstrated that
hyperoxia treatment significantly sensitized hypoxic HeLa cells to
IR and promoted cellular apoptosis. To gain further insight into
the molecular mechanism contributing to this phenomenon, the
activation of the Akt and ERK signaling pathways was detected, and
the results revealed that hypoxia treatment increased the protein
expression levels of p-Akt and p-ERK, whereas hyperoxia abrogated
hypoxia-activated p-Akt and p-ERK. Additionally, the administration
of Akt and ERK pathway inhibitors significantly decreased
hypoxia-induced RR. Gupta et al (48) demonstrated that VEGF significantly
enhanced the survival of vein endothelial cells in radiotherapy via
the activation of MEK and ERK. Additionally, Chen et al
(49) reported that antrocin
synergistically induced cell apoptosis and inhibited cell
proliferation in radioresistant prostate cancer cells by
suppressing the Akt and ERK signaling pathways. The current study
indicated that hyperoxia may function as a radiosensitizer by
suppressing the hypoxia-induced hyperactivation of the Akt and ERK
signaling pathways. However, to validate IR-induced cellular
apoptosis, detection of the changes of additional apoptosis
markers, including p53, poly (ADP-ribose) polymerase, the caspase
cascade and the Bcl-2 family (50–52),
should be investigated in future work.
In our previous work, different tumor cell lines
were investigated; these cell lines exhibited different sensitivity
to IR and O2 (data not shown). Therefore, there may be a
tumor type-specific association among RR, the HIF-1α-VEGF axis and
hyperoxic sensitization. The present study only reported the data
from HeLa cells, as HeLa cells are the most commonly used cell
model for the research of cervical cancer. However, to obtain a
more comprehensive and solid conclusion, further experiments using
more cervical cancer cell lines and in vivo assays are
required.
In conclusion, the current investigation
demonstrated that hyperoxia may be considered as a radiotherapeutic
sensitizer for hypoxic HeLa cells.
Acknowledgements
Not applicable.
Funding
This study was supported by Jilin Patent
Biotechnology Development Co., Ltd. (grant no. 2020YX016).
Availability of data and materials
The datasets used and/or analyzed during the current
study are available from the corresponding author on reasonable
request.
Authors' contributions
JW and DD wrote the manuscript and performed hypoxia
and hyperoxia culturing, cell irradiation, cell proliferation and
apoptosis detection. YF contributed to the experimental design. FC
and JZ performed plasmid construction experiments. HJ and JL
performed the ELISA and western blotting experiments. HW performed
the antibody neutralization tests and contributed to statistical
analysis. All authors read and approved the final manuscript.
Ethics approval and consent to
participate
Not applicable.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Wu WJ, Shen Y, Sui J, Li CY, Yang S, Xu
SY, Zhang M, Yin LH, Pu YP and Liang GY: Integrated analysis of
long noncoding RNA competing interactions revealed potential
biomarkers in cervical cancer: Based on a public database. Mol Med
Rep. 17:7845–7858. 2018.PubMed/NCBI
|
|
2
|
Cohen PA, Jhingran A, Oaknin A and Denny
L: Cervical cancer. Lancet. 393:169–182. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Lin CL, Lee CH, Chen CM, Cheng CW, Chen
PN, Ying TH and Hsieh YH: Protodioscin induces apoptosis through
ROS-mediated endoplasmic reticulum stress via the JNK/p38
activation pathways in human cervical cancer cells. Cell Physiol
Biochem. 46:322–334. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Pranatharthi A, Thomas P, Udayashankar AH,
Bhavani C, Suresh SB, Krishna S, Thatte J, Srikantia N, Ross CR and
Srivastava S: RhoC regulates radioresistance via crosstalk of ROCK2
with the DNA repair machinery in cervical cancer. J Exp Clin Cancer
Res. 38:3922019. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Liu C, Lin Q and Yun Z: Cellular and
molecular mechanisms underlying oxygen-dependent radiosensitivity.
Radiat Res. 183:487–496. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Hennessey D, Martin LM, Atzberger A, Lynch
TH, Hollywood D and Marignol L: Exposure to hypoxia following
irradiation increases radioresistance in prostate cancer cells.
Urol Oncol. 31:1106–1116. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Hong BJ, Kim J, Jeong H, Bok S, Kim YE and
Ahn GO: Tumor hypoxia and reoxygenation: The yin and yang for
radiotherapy. Radiat Oncol J. 34:239–249. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Candelaria M, Garcia-Arias A, Cetina L and
Duenas-Gonzalez A: Radiosensitizers in cervical cancer. Cisplatin
and beyond. Radiat Oncol. 1:152006. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Tamari K, Sano K, Li Z, Seo Y, Otani K,
Tatekawa S, Toratani M, Takaoka Y, Takahashi Y, Minami K, et al: Ro
90–7501 is a novel radiosensitizer for cervical cancer cells that
inhibits ATM phosphorylation. Anticancer Res. 39:4805–4810. 2019.
View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Mabuchi S and Kimura T: Nedaplatin: A
radiosensitizing agent for patients with cervical cancer. Chemother
Res Pract. 2011:9631592011.PubMed/NCBI
|
|
11
|
Javvadi P, Segan AT, Tuttle SW and
Koumenis C: The chemopreventive agent curcumin is a potent
radiosensitizer of human cervical tumor cells via increased
reactive oxygen species production and overactivation of the
mitogen-activated protein kinase pathway. Mol Pharmacol.
73:1491–1501. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Vaupel P: Oxygenation of human tumors.
Strahlenther Onkol. 166:377–386. 1990.PubMed/NCBI
|
|
13
|
Mayer R, Hamilton-Farrell MR, van der
Kleij AJ, Schmutz J, Granström G, Sicko Z, Melamed Y, Carl UM,
Hartmann KA, Jansen EC, et al: Hyperbaric oxygen and radiotherapy.
Strahlenther Onkol. 181:113–123. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Becker A, Kuhnt T, Liedtke H, Krivokuca A,
Bloching M and Dunst J: Oxygenation measurements in head and neck
cancers during hyperbaric oxygenation. Strahlenther Onkol.
178:105–108. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Brizel DM, Hage WD, Dodge RK, Munley MT,
Piantadosi CA and Dewhirst MW: Hyperbaric oxygen improves tumor
radiation response significantly more than carbogen/nicotinamide.
Radiat Res. 147:715–720. 1997. View
Article : Google Scholar : PubMed/NCBI
|
|
16
|
Hartmann KA, van der Kleij AJ, Carl UM,
Hulshof MC, Willers R and Sminia P: Effects of hyperbaric oxygen
and normobaric carbogen on the radiation response of the rat
rhabdomyosarcoma R1H. Int J Radiat Oncol Biol Phys. 51:1037–1044.
2001. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Nordsmark M, Maxwell RJ, Horsman MR,
Bentzen SM and Overgaard J: The effect of hypoxia and hyperoxia on
nucleoside triphosphate/inorganic phosphate, pO2 and radiation
response in an experimental tumour model. Br J Cancer.
76:1432–1439. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Pietrofesa RA, Velalopoulou A, Lehman SL,
Arguiri E, Solomides P, Koch CJ, Mishra OP, Koumenis C, Goodwin TJ
and Christofidou-Solomidou M: Novel double-hit model of radiation
and hyperoxia-induced oxidative cell damage relevant to space
travel. Int J Mol Sci. 17:9532016. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Hofer T, Desbaillets I, Hopfl G, Wenger RH
and Gassmann M: Characterization of HIF-1 alpha overexpressing HeLa
cells and implications for gene therapy. Comp Biochem Physiol C
Toxicol Pharmacol. 133:475–481. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Masoud GN and Li W: HIF-1α pathway: Role,
regulation and intervention for cancer therapy. Acta Pharm Sin B.
5:378–389. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Park JH, Yoon J and Park B: Pomolic acid
suppresses HIF1α/VEGF-mediated angiogenesis by targeting p38-MAPK
and mTOR signaling cascades. Phytomedicine. 23:1716–1726. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Chen JY, Gu ZJ, Wu MX, Yang Y, Zhang JH,
Ou JS, Zuo ZY, Wang JF and Chen YX: C-reactive protein can
upregulate VEGF expression to promote ADSC-induced angiogenesis by
activating HIF-1α via CD64/PI3k/Akt and MAPK/ERK signaling
pathways. Stem Cell Res Ther. 7:1142016. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Miyasaka A, Oda K, Ikeda Y, Sone K, Fukuda
T, Inaba K, Makii C, Enomoto A, Hosoya N, Tanikawa M, et al:
PI3K/mTOR pathway inhibition overcomes radioresistance via
suppression of the HIF1-α/VEGF pathway in endometrial cancer.
Gynecol Oncol. 138:174–180. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Fechner G, Dederichs F, Schmidt D, Muller
S, Vaupel P and Albers P: Hyperoxia-induced improvement of the in
vitro response to gemcitabine in transitional cell carcinoma.
Anticancer Res. 25:3413–3418. 2005.PubMed/NCBI
|
|
25
|
Liu Q, He X, Liu Y, Du B, Wang X, Zhang W,
Jia P, Dong J, Ma J, Wang X, et al: NADPH oxidase-mediated
generation of reactive oxygen species: A new mechanism for
X-ray-induced HeLa cell death. Biochem Biophys Res Commun.
377:775–779. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Lu J, Suzuki T, Lu S and Suzuki N:
Involvement of glyceraldehyde-3-phosphate dehydrogenase in the
X-Ray resistance of HeLa cells. Biosci Biotechnol Biochem.
72:2432–2435. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Lake AC, Vassy R, Di Benedetto M, Lavigne
D, Visage CL, Perret GY and Letourneur D: Low molecular weight
fucoidan increases VEGF165-induced endothelial cell migration by
enhancing VEGF165 binding to VEGFR-2 and NRP1. J Biol Chem.
281:37844–37852. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Wu XJ, Zhu JW, Jing J, Xue D, Liu H, Zheng
M and Lu ZF: VEGF165 modulates proliferation, adhesion, migration
and differentiation of cultured human outer root sheath cells from
central hair follicle epithelium through VEGFR-2 activation in
vitro. J Dermatol Sci. 73:152–160. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Zhang H, Han Y, Tao J, Liu S, Yan C and Li
S: Cellular repressor of E1A-stimulated genes regulates vascular
endothelial cell migration by the ILK/AKT/mTOR/VEGF(165) signaling
pathway. Exp Cell Res. 317:2904–2913. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Tsuzuki T, Okada H, Shindoh H, Shimoi K,
Nishigaki A and Kanzaki H: Effects of the hypoxia-inducible
factor-1 inhibitor echinomycin on vascular endothelial growth
factor production and apoptosis in human ectopic endometriotic
stromal cells. Gynecol Endocrinol. 32:323–328. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Walsh CM, Chvanov M, Haynes LP, Petersen
OH, Tepikin AV and Burgoyne RD: Role of phosphoinositides in STIM1
dynamics and store-operated calcium entry. Biochem J. 425:159–168.
2009. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Ma S, Musa T and Bag J: Reduced stability
of mitogen-activated protein kinase kinase-2 mRNA and
phosphorylation of poly(A)-binding protein (PABP) in cells
overexpressing PABP. J Biol Chem. 281:3145–3156. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Lin C, McGough R, Aswad B, Block JA and
Terek R: Hypoxia induces HIF-1alpha and VEGF expression in
chondrosarcoma cells and chondrocytes. J Orthop Res. 22:1175–1181.
2004. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Fu Z, Chen D, Cheng H and Wang F:
Hypoxia-inducible factor-1α protects cervical carcinoma cells from
apoptosis induced by radiation via modulation of vascular
endothelial growth factor and p53 under hypoxia. Med Sci Monitor.
21:318–325. 2015. View Article : Google Scholar
|
|
35
|
Shibuya M: VEGF-VEGFR system as a target
for suppressing inflammation and other diseases. Endocr Metab
Immune Disord Drug Targets. 15:135–144. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Gupta AK, Bakanauskas VJ, Cerniglia GJ,
Cheng Y, Bernhard EJ, Muschel RJ and McKenna WG: The Ras radiation
resistance pathway. Cancer Res. 61:4278–4282. 2001.PubMed/NCBI
|
|
37
|
McKenna WG, Muschel RJ, Gupta AK, Hahn SM
and Bernhard EJ: The RAS signal transduction pathway and its role
in radiation sensitivity. Oncogene. 22:5866–5875. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Shimura T, Kakuda S, Ochiai Y, Kuwahara Y,
Takai Y and Fukumoto M: Targeting the AKT/GSK3beta/cyclin D1/Cdk4
survival signaling pathway for eradication of tumor radioresistance
acquired by fractionated radiotherapy. Int J Radiat Oncol Biol
Phys. 80:540–548. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Vaupel P and Mayer A: Hypoxia in cancer:
Significance and impact on clinical outcome. Cancer Metastasis Rev.
26:225–239. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Liao D and Johnson RS: Hypoxia: A key
regulator of angiogenesis in cancer. Cancer Metastasis Rev.
26:281–290. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Thomlinson RH and Gray LH: The
histological structure of some human lung cancers and the possible
implications for radiotherapy. Br J Cancer. 9:539–549. 1955.
View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Bennett MH, Feldmeier J, Smee R and
Milross C: Hyperbaric oxygenation for tumour sensitisation to
radiotherapy. Cochrane Database Syst Rev. 4:CD0050072018.PubMed/NCBI
|
|
43
|
Gorski DH, Beckett MA, Jaskowiak NT,
Calvin DP, Mauceri HJ, Salloum RM, Seetharam S, Koons A, Hari DM,
Kufe DW and Weichselbaum RR: Blockage of the vascular endothelial
growth factor stress response increases the antitumor effects of
ionizing radiation. Cancer Res. 59:3374–3378. 1999.PubMed/NCBI
|
|
44
|
Ferrara N, Gerber HP and LeCouter J: The
biology of VEGF and its receptors. Nat Med. 9:669–676. 2003.
View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Hiratsuka S, Minowa O, Kuno J, Noda T and
Shibuya M: Flt-1 lacking the tyrosine kinase domain is sufficient
for normal development and angiogenesis in mice. Proc Natl Acad Sci
USA. 95:9349–9354. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Selvaraj D, Gangadharan V, Michalski CW,
Kurejova M, Stösser S, Srivastava K, Schweizerhof M, Waltenberger
J, Ferrara N, Heppenstall P, et al: A functional role for VEGFR1
expressed in peripheral sensory neurons in cancer pain. Cancer
Cell. 27:780–796. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Ding ZY, Huang YJ, Tang JD, Li G, Jiang PQ
and Wu HT: Silencing of hypoxia-inducible factor-1α promotes
thyroid cancer cell apoptosis and inhibits invasion by
downregulating WWP2, WWP9, VEGF and VEGFR2. Exp Ther Med.
12:3735–3741. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Gupta VK, Jaskowiak NT, Beckett MA,
Mauceri HJ, Grunstein J, Johnson RS, Calvin DA, Nodzenski E,
Pejovic M, Kufe DW, et al: Vascular endothelial growth factor
enhances endothelial cell survival and tumor radioresistance.
Cancer J. 8:47–54. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Chen YA, Tzeng DTW, Huang YP, Lin CJ, Lo
UG, Wu CL, Lin H, Hsieh JT, Tang CH and Lai CH: Antrocin sensitizes
prostate cancer cells to radiotherapy through inhibiting PI3K/AKT
and MAPK signaling pathways. Cancers (Basel). 11:342018. View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Mazzatti DJ, Lee YJ, Helt CE, O'Reilly MA
and Keng PC: p53 modulates radiation sensitivity independent of p21
transcriptional activation. Am J Clin Oncol. 28:43–50. 2005.
View Article : Google Scholar : PubMed/NCBI
|
|
51
|
Lee KB, Kim KR, Huh TL and Lee YM: Proton
induces apoptosis of hypoxic tumor cells by the p53-dependent and
p38/JNK MAPK signaling pathways. Int J Oncol. 33:1247–1256.
2008.PubMed/NCBI
|
|
52
|
Yoon TM, Kim SA, Lee DH, Lee JK, Park YL,
Lee KH, Chung IJ, Joo YE and Lim SC: EGR1 regulates
radiation-induced apoptosis in head and neck squamous cell
carcinoma. Oncol Rep. 33:1717–1722. 2015. View Article : Google Scholar : PubMed/NCBI
|