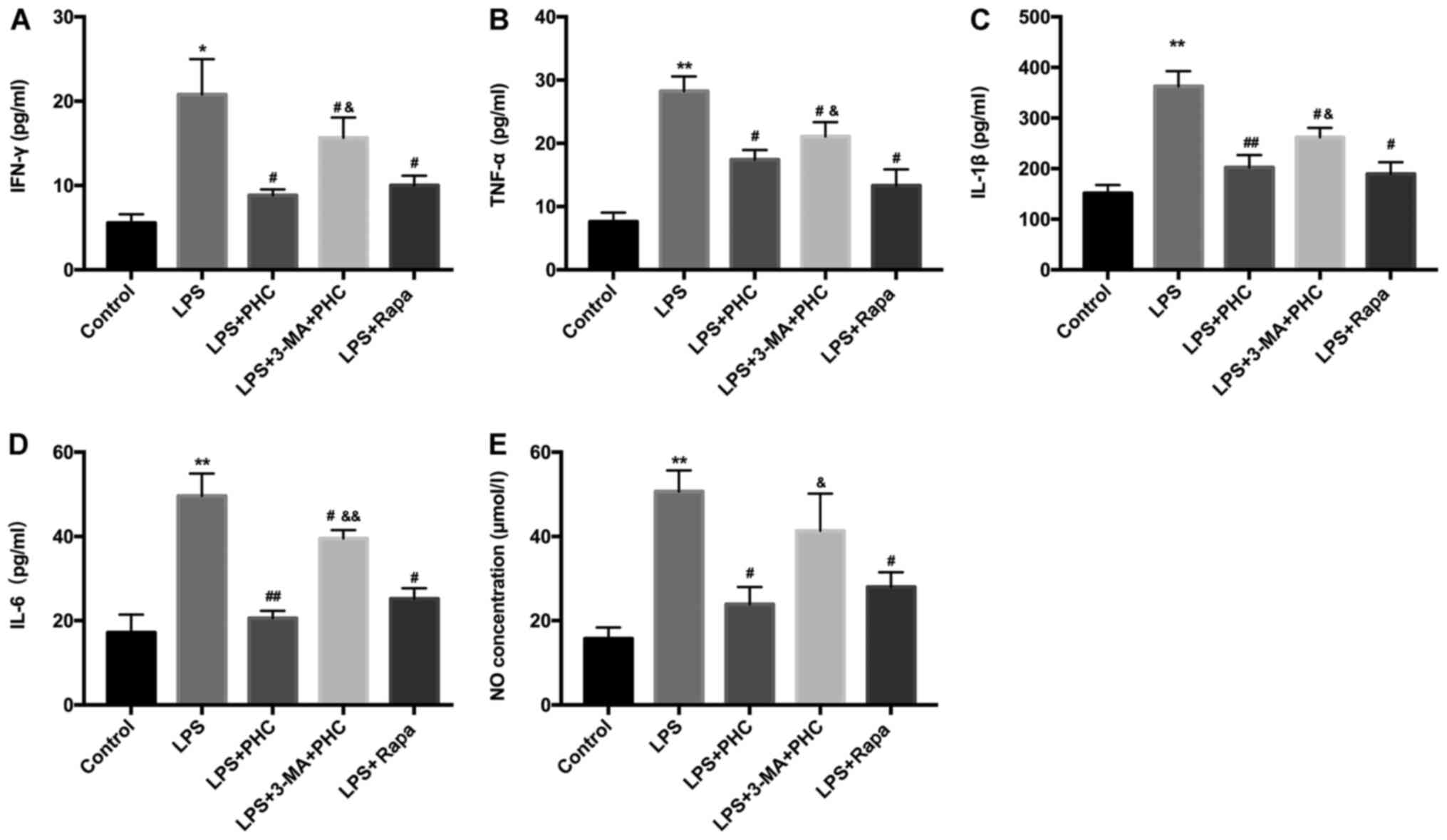
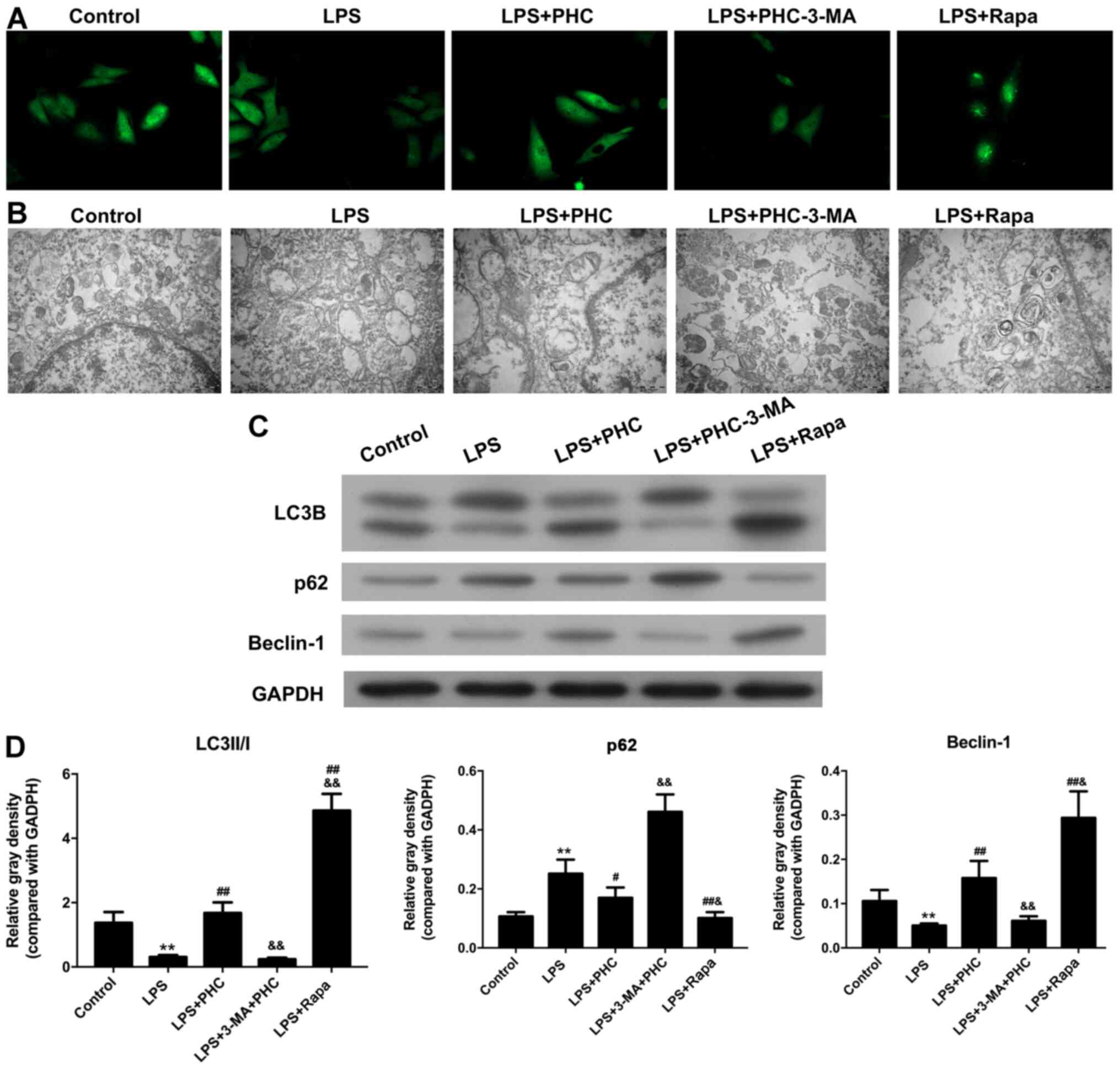
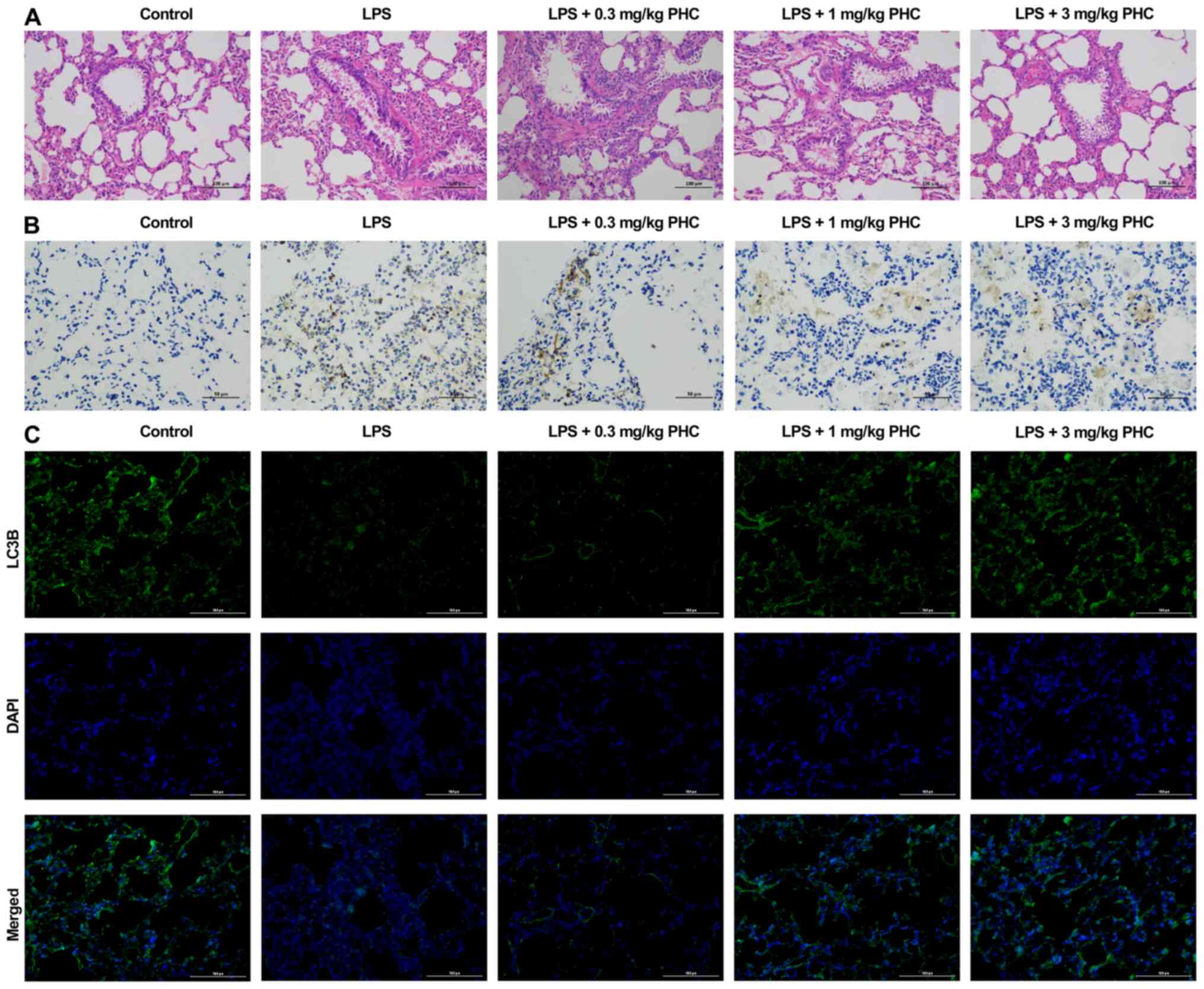
|
1
|
Matthay MA and Zemans RL: The acute
respiratory distress syndrome: Pathogenesis and treatment. Annu Rev
Pathol. 6:147–163. 2011. View Article : Google Scholar
|
|
2
|
Li BQ, Sun HC, Nie SN, Shao DB, Liu HM and
Qian XM: Effect of penehyclidine hydrochloride on patients with
acute lung injury and its mechanisms. Chin J Traumatol. 13:329–335.
2010.
|
|
3
|
Saguil A and Fargo M: Acute respiratory
distress syndrome: Diagnosis and management. Am Fam Physician.
85:352–358. 2012.
|
|
4
|
Lee-Chiong T Jr and Matthay RA:
Drug-induced pulmonary edema and acute respiratory distress
syndrome. Clin Chest Med. 25:95–104. 2004. View Article : Google Scholar
|
|
5
|
Shah J and Rana SS: Acute respiratory
distress syndrome in acute pancreatitis. Indian J Gastroenterol.
39:123–132. 2020. View Article : Google Scholar
|
|
6
|
Ibadov RA, Arifjanov AS, Ibragimov SK and
Abdullajanov BR: Acute respiratory distress-syndrome in the general
complications of severe acute pancreatitis. Ann Hepatobiliary
Pancreat Surg. 23:359–364. 2019. View Article : Google Scholar
|
|
7
|
Iriyama H, Abe T, Kushimoto S, Fujishima
S, Ogura H, Shiraishi A, Saitoh D, Mayumi T, Naito T, Komori A, et
al: Risk modifiers of acute respiratory distress syndrome in
patients with non-pulmonary sepsis: A retrospective analysis of the
FORECAST study. J Intensive Care. 8:72020. View Article : Google Scholar
|
|
8
|
Rambaud J, Lidouren F, Sage M, Kohlhauer
M, Nadeau M, Fortin-Pellerin É, Micheau P, Zilberstein L, Mongardon
N, Ricard JD, et al: Hypothermic total liquid ventilation after
experimental aspiration-associated acute respiratory distress
syndrome. Ann Intensive Care. 8:572018. View Article : Google Scholar
|
|
9
|
Tambe A, Gentile T, Ramadas P, Tambe V and
Badrinath M: Cytomegalovirus pneumonia causing acute respiratory
distress syndrome after brentuximab vedotin therapy. Am J Ther.
26:e794–e795. 2019. View Article : Google Scholar
|
|
10
|
Killien EY, Huijsmans RLN, Ticknor IL,
Smith LS, Vavilala MS, Rivara FP and Watson RS: Acute respiratory
distress syndrome following pediatric trauma: Application of
pediatric acute lung injury consensus conference criteria. Crit
Care Med. 48:e26–e33. 2020. View Article : Google Scholar
|
|
11
|
Fan E, Brodie D and Slutsky AS: Acute
respiratory distress syndrome: Advances in diagnosis and treatment.
JAMA. 319:698–710. 2018. View Article : Google Scholar
|
|
12
|
Fanelli V and Ranieri VM: Mechanisms and
clinical consequences of acute lung injury. Ann Am Thorac Soc. 12
(Suppl 1):S3–S8. 2015. View Article : Google Scholar
|
|
13
|
Su BC, Huang HN, Lin TW, Hsiao CD and Chen
JY: Epinecidin-1 protects mice from LPS-induced endotoxemia and
cecal ligation and puncture-induced polymicrobial sepsis. Biochim
Biophys Acta Mol Basis Dis. 1863:3028–3037. 2017. View Article : Google Scholar
|
|
14
|
Chen M, Chen C, Yuan X, Chen X, Zheng F,
Shao L and Zhang Z: Angiotensin II aggravates lipopolysaccharide
induced human pulmonary microvascular endothelial cells
permeability in high glucose status. Endocr J. 65:717–725. 2018.
View Article : Google Scholar
|
|
15
|
Wang H, Wang T, Yuan Z, Cao Y, Zhou Y, He
J, Shen Y, Zeng N, Dai L, Wen F and Chen L: Role of receptor for
advanced glycation end products in regulating lung fluid balance in
lipopolysaccharide-induced acute lung injury and infection-related
acute respiratory distress syndrome. Shock. 50:472–482. 2018.
View Article : Google Scholar
|
|
16
|
Wu DQ, Wu HB, Zhang M and Wang JA: Effects
of zinc finger protein A20 on lipopolysaccharide (LPS)-induced
pulmonary inflammation/anti-inflammatory mediators in an acute lung
injury/acute respiratory distress syndrome rat model. Med Sci
Monit. 23:3536–3545. 2017. View Article : Google Scholar
|
|
17
|
Shi L, Dong N, Ji D, Huang X, Ying Z, Wang
X and Chen C: Lipopolysaccharide-induced CCN1 production enhances
interleukin-6 secretion in bronchial epithelial cells. Cell Biol
Toxicol. 34:39–49. 2018. View Article : Google Scholar
|
|
18
|
Wang Y, Lin D, Tan H, Gao Y and Ma J:
Penehyclidine hydrochloride preconditioning provides pulmonary and
systemic protection in a rat model of lung ischaemia reperfusion
injury. Eur J Pharmacol. 839:1–11. 2018. View Article : Google Scholar
|
|
19
|
Xiao HT, Liao Z and Tong RS: Penehyclidine
hydrochloride: A potential drug for treating COPD by attenuating
Toll-like receptors. Drug Des Devel Ther. 6:317–22. 2012.
View Article : Google Scholar
|
|
20
|
Wang Y, Gao Y and Ma J: Pleiotropic
effects and pharmacological properties of penehyclidine
hydrochloride. Drug Des Devel Ther. 12:3289–3299. 2018. View Article : Google Scholar
|
|
21
|
Lin D, Ma J, Xue Y and Wang Z:
Penehyclidine hydrochloride preconditioning provides
cardioprotection in a rat model of myocardial ischemia/reperfusion
injury. PLoS One. 10:e01380512015. View Article : Google Scholar
|
|
22
|
Shen W, Gan J, Xu S, Jiang G and Wu H:
Penehyclidine hydrochloride attenuates LPS-induced acute lung
injury involvement of NF-kappaB pathway. Pharmacol Res. 60:296–302.
2009. View Article : Google Scholar
|
|
23
|
Wang YA, Zhou WX, Li JX, Liu YQ, Yue YJ,
Zheng JQ, Liu KL and Ruan JX: Anticonvulsant effects of
phencynonate hydrochloride and other anticholinergic drugs in soman
poisoning: neurochemical mechanisms. Life Sci. 78:210–223. 2005.
View Article : Google Scholar
|
|
24
|
Guoshou Z, Chengye Z, Zhihui L and Jinlong
L: Effects of high dose of anisodamine on the respiratory function
of patients with traumatic acute lung injury. Cell Biochem Biophys.
66:365–369. 2013. View Article : Google Scholar
|
|
25
|
Zhan J, Xiao F, Li JJ, Zhang ZZ, Chen K,
Wang YP and Wang YL: Penehyclidine hydrochloride decreases
pulmonary microvascular permeability by upregulating beta arrestins
in a murine cecal ligation and puncture model. J Surg Res.
193:391–398. 2015. View Article : Google Scholar
|
|
26
|
Krenn K, Lucas R, Croizé A, Boehme S,
Klein KU, Hermann R, Markstaller K and Ullrich R: Inhaled AP301 for
treatment of pulmonary edema in mechanically ventilated patients
with acute respiratory distress syndrome. A phase IIa randomized
placebo-controlled trial. 21:1942017.
|
|
27
|
Zheng F, Xiao F, Yuan QH, Liu QS, Zhang
ZZ, Wang YL and Zhan J: Penehyclidine hydrochloride decreases
pulmonary microvascular endothelial inflammatory injury through a
beta-arrestin-1-dependent mechanism. Inflammation. 41:1610–1620.
2018. View Article : Google Scholar
|
|
28
|
Dong L, Yu WM, Zheng H, Loh ML, Bunting
ST, Pauly M, Huang G, Zhou M, Broxmeyer HE, Scadden DT and Qu CK:
Leukaemogenic effects of Ptpn11 activating mutations in the stem
cell microenvironment. Nature. 539:304–308. 2016. View Article : Google Scholar
|
|
29
|
Huang X, Kong G, Li Y, Zhu W, Xu H, Zhang
X, Li J, Wang L, Zhang Z, et al: Decitabine and 5-azacitidine both
alleviate LPS induced ARDS through anti-inflammatory/antioxidant
activity and protection of glycocalyx and inhibition of MAPK
pathways in mice. Biomedicine & Pharmacotherapy. 84:447–453.
2016. View Article : Google Scholar
|
|
30
|
Kellner M..Noonepalle S..Lu Q..Srivastava
A..Zemskov E..Black S. M: ROS Signaling in the Pathogenesis of
Acute Lung Injury (ALI) and Acute Respiratory Distress Syndrome
(ARDS). Wang YX: Pulmonary Vasculature Redox Signaling in Health
and Disease. Advances in Experimental Medicine and Biology.
Springer; Cham: 967. pp. 105–137. 2017, View Article : Google Scholar
|
|
31
|
Kartlaşmış K, Kökbaş U and Kayrin L:
Biochemistry of apoptosis. Arch Med Rev J. 25:52–69. 2016.
|
|
32
|
Redza-Dutordoir M and Averill-Bates DA:
Activation of apoptosis signalling pathways by reactive oxygen
species. Biochim Biophys Acta. 1863:2977–2992. 2016. View Article : Google Scholar
|
|
33
|
Ekizceli G, Uluer ET and Inan S:
Investigation of the effects of rapamycin on the mTOR pathway and
apoptosis in metastatic and non-metastatic human breast cancer cell
lines. Bratisl Lek Listy. 121:308–315. 2020.
|
|
34
|
Diwanji N and Bergmann A: An unexpected
friend-ROS in apoptosis-induced compensatory proliferation:
Implications for regeneration and cancer. Semin Cell Dev Biol.
80:74–82. 2018. View Article : Google Scholar
|
|
35
|
El-Shafey ES and Elsherbiny ES: The role
of apoptosis and autophagy in the insulin-enhancing activity of
oxovanadium(IV) bipyridine complex in streptozotocin-induced
diabetic mice. Biometals. 33:123–135. 2020. View Article : Google Scholar
|
|
36
|
Liu J, Liu W and Yang H: Balancing
apoptosis and autophagy for parkinson's disease therapy: Targeting
BCL-2. ACS Chem Neurosci. 10:792–802. 2019. View Article : Google Scholar
|
|
37
|
Yang Y, Gao H, Zhou H, Liu Q, Qi Z, Zhang
Y and Zhang J: The role of mitochondria-derived peptides in
cardiovascular disease: Recent updates. Biomed Pharmacother.
117:1090752019. View Article : Google Scholar
|
|
38
|
Kiraz Y, Adan A, Kartal Yandim M and Baran
Y: Major apoptotic mechanisms and genes involved in apoptosis.
Tumour Biol. 37:8471–8486. 2016. View Article : Google Scholar
|
|
39
|
Wang J, Oppenheimer L, Fata P, Pintin J,
Stimpson R and Mantsch HH: Spectroscopic approach to
capillary-alveolar membrane damage induced acute lung injury. Can
Respir J. 6:499–506. 1999. View Article : Google Scholar
|
|
40
|
Kiser TH, Burnham EL, Clark B, Ho PM,
Allen RR, Moss M and Vandivier RW: Half-dose versus full-dose
alteplase for treatment of pulmonary embolism. Crit Care Med.
46:1617–1625. 2018. View Article : Google Scholar
|
|
41
|
Li W, Qiu X, Jiang H, Han Y, Wei D and Liu
J: Downregulation of miR-181a protects mice from LPS-induced acute
lung injury by targeting Bcl-2. Biomed Pharmacother. 84:1375–1382.
2016. View Article : Google Scholar
|
|
42
|
Li B, Zeng M, Zheng H, Huang C, He W, Lu
G, Li X, Chen Y and Xie R: Effects of ghrelin on the apoptosis of
human neutrophils in vitro. Int J Mol Med. 38:794–802. 2016.
View Article : Google Scholar
|
|
43
|
Wang L, Wang T, Li H, Liu Q, Zhang Z, Xie
W, Feng Y, Socorburam T, Wu G, Xia Z and Wu Q: Receptor interacting
protein 3-mediated necroptosis promotes lipopolysaccharide-induced
inflammation and acute respiratory distress syndrome in mice. PLoS
One. 11:e01557232016. View Article : Google Scholar
|
|
44
|
Zhan J, Wang Y, Wang C, Li J, Zhang Z and
Jia B: Protective effects of penehyclidine hydrochloride on septic
mice and its mechanism. Shock. 28:727–732. 2007.
|
|
45
|
Yuan Q, Xiao F, Liu Q, Zheng F, Shen S, He
Q, Chen K, Wang Y, Zhang Z and Zhan J: M3 receptor is
involved in the effect of penehyclidine hydrochloride reduced
endothelial injury in LPS-stimulated human pulmonary microvascular
endothelial cell. Pulm Pharmacol Ther. 48:144–150. 2018. View Article : Google Scholar
|
|
46
|
Feng M, Wang L, Chang S and Yuan P:
Penehyclidine hydrochloride regulates mitochondrial dynamics and
apoptosis through p38MAPK and JNK signal pathways and provides
cardioprotection in rats with myocardial ischemia-reperfusion
injury. Eur J Pharm Sci. 121:243–250. 2018. View Article : Google Scholar
|
|
47
|
Lin D, Cui B, Ren J and Ma J: Regulation
of VDAC1 contributes to the cardioprotective effects of
penehyclidine hydrochloride during myocardial ischemia/reperfusion.
Exp Cell Res. 367:257–263. 2018. View Article : Google Scholar
|
|
48
|
Liu ZY, Guo L, Xiao G, Dong GY, Zhang YX,
Cheng H, Wang XY and Yang C: Significance and expression of
c-erBb-2, p53, and caspase-3 in breast cancer tissue in different
age groups. Eur J Gynaecol Oncol. 39:430–432. 2018.
|
|
49
|
Bosmann M and Ward PA: The inflammatory
response in sepsis. Trends Immunol. 34:129–236. 2013. View Article : Google Scholar
|
|
50
|
Johnson CL, Soeder Y and Dahlke MH:
Concise review: Mesenchymal stromal cell-based approaches for the
treatment of acute respiratory distress and sepssis syndromes. Stem
Cells Transl Med. 6:1141–1151. 2017. View Article : Google Scholar
|
|
51
|
Monteleon CL, Agnihotri T, Dahal A, Liu M,
Rebecca VW, Beatty GL, Amaravadi RK and Ridky TW: Lysosomes support
the degradation, signaling, and mitochondrial metabolism necessary
for human epidermal differentiation. J Invest Dermatol.
138:1945–1954. 2018. View Article : Google Scholar
|
|
52
|
Ko S, Gu MJ, Kim CG, Kye YC, Lim Y, Lee
JE, Park BC, Chu H, Han SH and Yun CH: Rapamycin-induced autophagy
restricts porcine epidemic diarrhea virus infectivity in porcine
intestinal epithelial cells. Antiviral Res. 146:86–95. 2017.
View Article : Google Scholar
|
|
53
|
Lv C, Wang L, Zhu X, Lin W, Chen X, Huang
Z, Huang L and Yang S: Glucosamine promotes osteoblast
proliferation by modulating autophagy via the mammalian target of
rapamycin pathway. 99:271–277. 2018.
|
|
54
|
Zeng M, Sang W, Chen S, Chen R, Zhang H,
Xue F, Li Z, Liu Y and Gong Y: 4-PBA inhibits LPS-induced
inflammation through regulating ER stress and autophagy in acute
lung injury models. Toxicol. 271((5)): 26–37. 2017.
|
|
55
|
Ren J and Zhang Y: Targeting autophagy in
aging and aging-related cardiovascular diseases. Trends Pharmacol
Sci. 39:1064–1076. 2018. View Article : Google Scholar
|
|
56
|
Decuypere JP, Ceulemans LJ, Agostinis P,
Monbaliu D, Naesens M, Pirenne J and Jochmans I: Autophagy and the
kidney: Implications for ischemia-reperfusion injury and therapy.
Am J Kidney Dis. 66:699–709. 2015. View Article : Google Scholar
|
|
57
|
Gao Y, Wang N, Liu L, Liu Y and Zhang J:
Relationship between mammalian target of rapamycin and autophagy in
lipopolysaccharide-induced lung injury. J Surg Res. 201:356–363.
2016. View Article : Google Scholar
|
|
58
|
Zhao H and Luo F: Rapamycin reverse
lipopolysaccharide-induced acute lung injury through activating
autophagy flux. J Pharm Univ. 2015.
|
|
59
|
Liu Y and Zhang J: Saturated hydrogen
saline ameliorates lipopolysaccharide-induced acute lung injury by
reducing excessive autophagy. Exp Ther Med. 13:2609–2615. 2017.
|
|
60
|
Zhu Q, Wang H, Wang H, Luo Y, Yu Y, Du Q,
Fei A and Pan S: Protective effects of ethyl pyruvate on
lipopolysaccharide-induced acute lung injury through inhibition of
autophagy in neutrophils. Mol Med Rep. 15:1272–1278. 2017.
View Article : Google Scholar
|