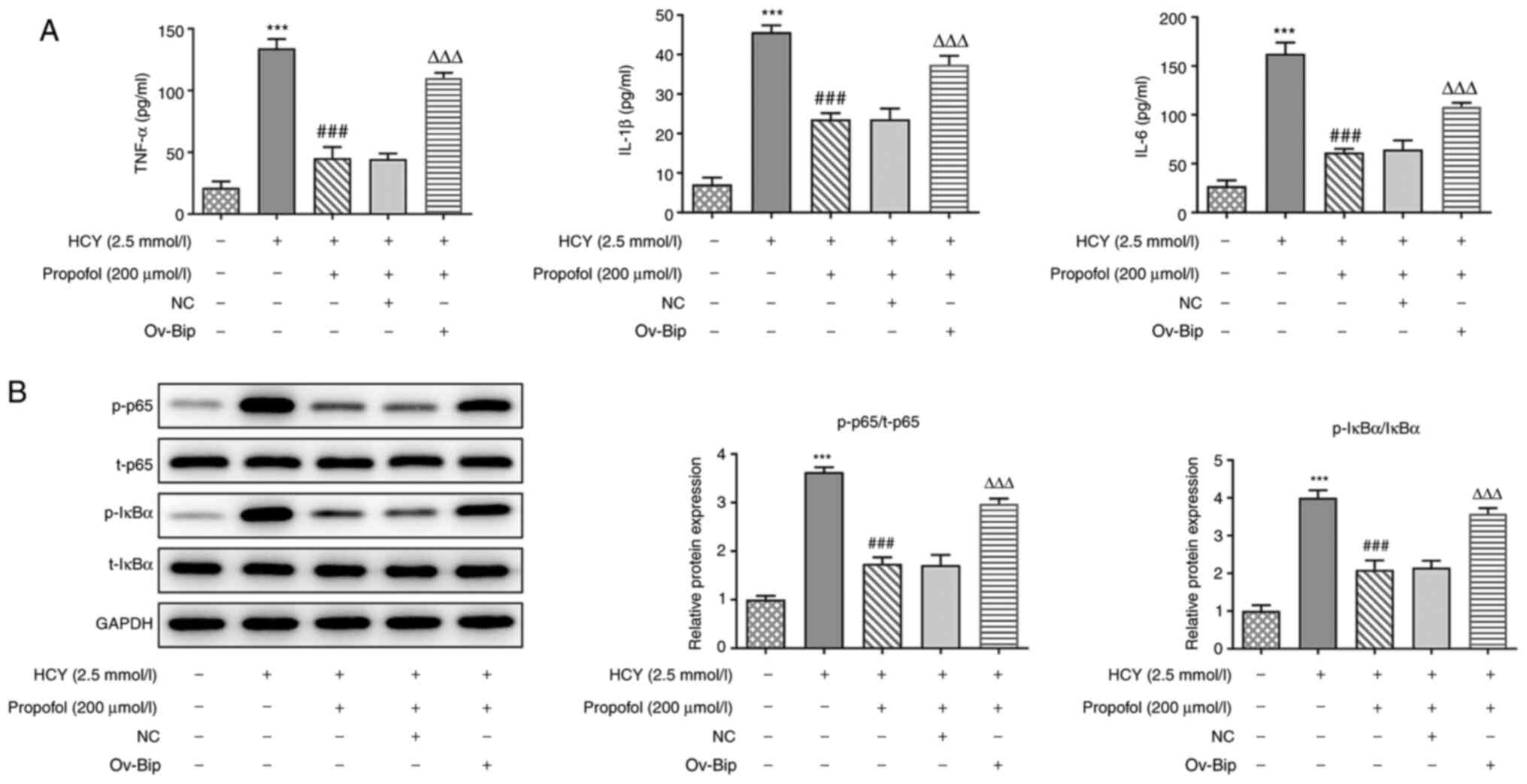
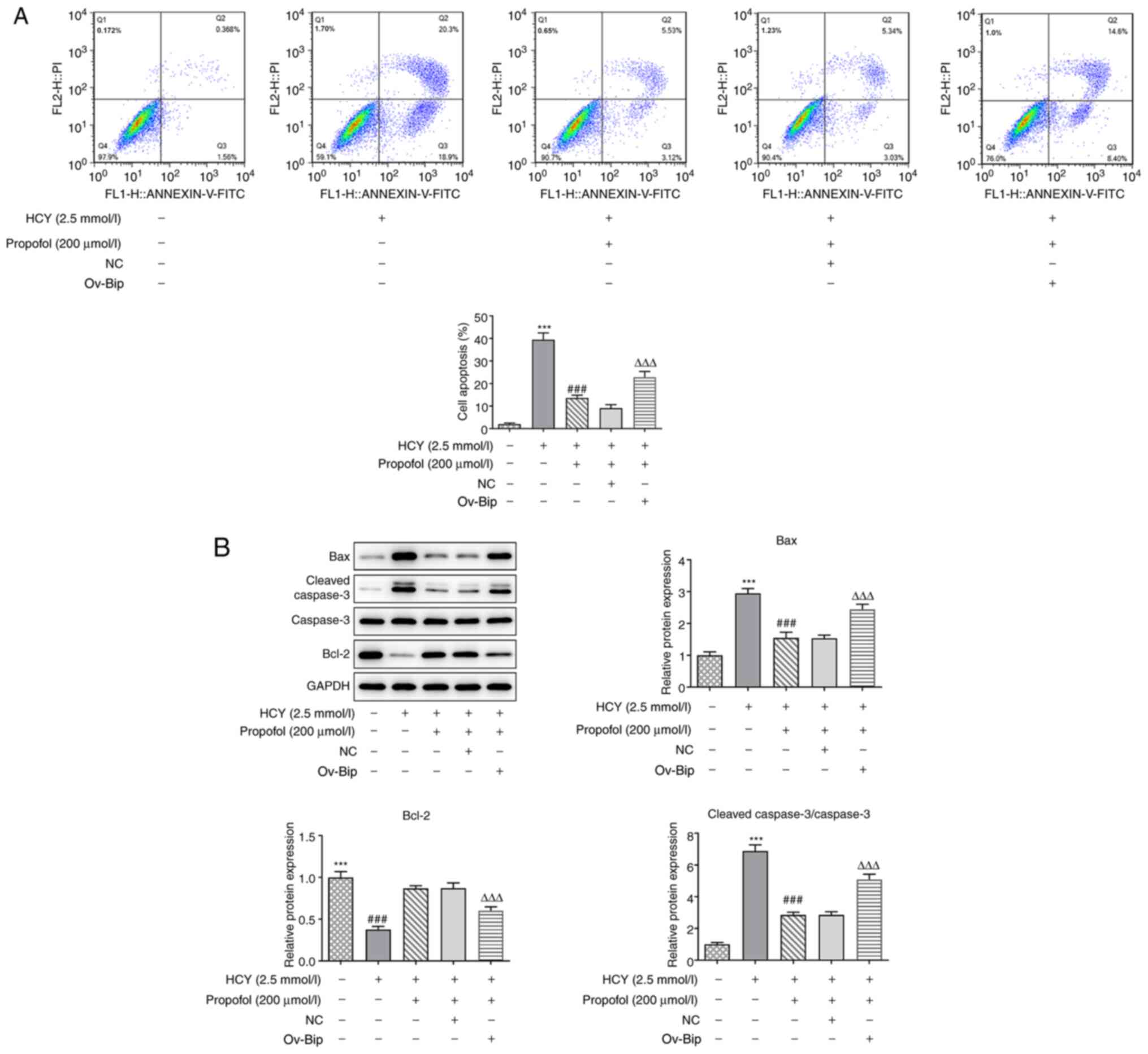
|
1
|
Pang X, Liu J, Zhao J, Mao J, Zhang X,
Feng L, Han C, Li M, Wang S and Wu D: Homocysteine induces the
expression of C-reactive protein via NMDAr-ROS-MAPK-NF-κB signal
pathway in rat vascular smooth muscle cells. Atherosclerosis.
236:73–81. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Li J, Luo M, Xie N, Wang J and Chen L:
Curcumin protects endothelial cells against homocysteine induced
injury through inhibiting inflammation. Am J Transl Res.
8:4598–4604. 2016.PubMed/NCBI
|
|
3
|
Hales TG and Lambert JJ: The actions of
propofol on inhibitory amino acid receptors of bovine
adrenomedullary chromaffin cells and rodent central neurones. Br J
Pharmacol. 104:619–628. 1991. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Mickey BJ, White AT, Arp AM, Leonardi K,
Torres MM, Larson AL, Odell DH, Whittingham SA, Beck MM, Jessop JE,
et al: Propofol for treatment-resistant depression: A pilot study.
Int J Neuropsychopharmacol. 21:1079–1089. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Allaouchiche B, Debon R, Goudable J,
Chassard D and Duflo F: Oxidative stress status during exposure to
propofol, sevoflurane and desflurane. Anesth Analg. 93:981–985.
2001. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Nesseler N, Launey Y, Isslame S, Flécher
E, Lebouvier T, Mallédant Y and Seguin P: Is extracorporeal
membrane oxygenation for severe acute respiratory distress syndrome
related to intra-abdominal sepsis beneficial? Intensive Care Med.
41:943–945. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Lin C, Sui H, Gu J, Yang X, Deng L, Li W,
Ding W, Li D and Yang Y: Effect and mechanism of propofol on
myocardial ischemia reperfusion injury in type 2 diabetic rats.
Microvasc Res. 90:162–168. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Wang S and Smith JD: ABCA1 and nascent HDL
biogenesis. Biofactors. 40:547–554. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Cao SS, Luo KL and Shi L: Endoplasmic
reticulum stress interacts with inflammation in human diseases. J
Cell Physiol. 231:288–294. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Oakes SA and Papa FR: The role of
endoplasmic reticulum stress in human pathology. Annu Rev Pathol.
10:173–194. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Yang X, Xu H, Hao Y, Zhao L, Cai X, Tian
J, Zhang M, Han X, Ma S, Cao J and Jiang Y: Endoplasmic reticulum
oxidoreductin 1α mediates hepatic endoplasmic reticulum stress in
homocysteine-induced atherosclerosis. Acta Biochim Biophys Sin
(Shanghai). 46:902–910. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Yang S, Wu M, Li X, Zhao R, Zhao Y, Liu L
and Wang S: Role of endoplasmic reticulum stress in atherosclerosis
and its potential as a therapeutic target. Oxid Med Cell Longev.
2020:92701072020. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Ivanova EA and Orekhov AN: The role of
endoplasmic reticulum stress and unfolded protein response in
atherosclerosis. Int J Mol Sci. 17:1932016. View Article : Google Scholar
|
|
14
|
Hamczyk MR, Villa-Bellosta R, Quesada V,
Gonzalo P, Vidak S, Nevado RM, Andrés-Manzano MJ, Misteli T,
López-Otín C and Andrés V: Progerin accelerates atherosclerosis by
inducing endoplasmic reticulum stress in vascular smooth muscle
cells. EMBO Mol Med. 11:e97362019. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Alberts-Grill N, Denning TL, Rezvan A and
Jo H: The role of the vascular dendritic cell network in
atherosclerosis. Am J Physiol Cell Physiol. 305:C1–C21. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Suwaidi JA, Hamasaki S, Higano ST,
Nishimura RA, Holmes DR Jr and Lerman A: Long-term follow-up of
patients with mild coronary artery disease and endothelial
dysfunction. Circulation. 101:948–954. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Reddy KG, Nair RN, Sheehan HM and Hodgson
JM: Evidence that selective endothelial dysfunction may occur in
the absence of angiographic or ultrasound atherosclerosis in
patients with risk factors for atherosclerosis. J Am Coll Cardiol.
23:833–843. 1994. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Esper RJ, Nordaby RA, Vilariño JO,
Paragano A, Cacharron JL and Machado RA: Endothelial dysfunction: A
comprehensive appraisal. Cardiovasc Diabetol. 5:42006. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Verma S, Buchanan MR and Anderson TJ:
Endothelial function testing as a biomarker of vascular disease.
Circulation. 108:2054–2059. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Canpolat U, Kocyigit D and Yildirim A:
Role of endothelial dysfunction and endocan in atherosclerosis:
Point of origin or end point? Angiology. 71:4772020. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Lai WK and Kan MY: Homocysteine-induced
endothelial dysfunction. Ann Nutr Metab. 67:1–12. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Rius J, Guma M, Schachtrup C, Akassoglou
K, Zinkernagel AS, Nizet V, Johnson RS, Haddad GG and Karin M:
NF-kappaB links innate immunity to the hypoxic response through
transcriptional regulation of HIF-1alpha. Nature. 453:807–811.
2008. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Tak PP and Firestein GS: NF-kappaB: A key
role in inflammatory diseases. J Clin Invest. 107:7–11. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Holgate ST: Cytokine and anti-cytokine
therapy for the treatment of asthma and allergic disease. Cytokine.
28:152–157. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Williams RO, Paleolog E and Feldmann M:
Cytokine inhibitors in rheumatoid arthritis and other autoimmune
diseases. Curr Opin Pharmacol. 7:412–417. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Baldwin AS Jr: The NF-kappa B and I kappa
B proteins: New discoveries and insights. Annu Rev Immunol.
14:649–683. 1996. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Du QH, Xu YB, Zhang MY, Yun P and He CY:
Propofol induces apoptosis and increases gemcitabine sensitivity in
pancreatic cancer cells in vitro by inhibition of nuclear factor-κB
activity. World J Gastroenterol. 19:5485–5492. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Peng X, Li C, Yu W, Liu S, Cong Y, Fan G
and Qi S: Propofol attenuates hypoxia-induced inflammation in BV2
microglia by inhibiting oxidative stress and NF-κB/Hif-1α
signaling. Biomed Res Int. 2020:89787042020. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Zhang K and Kaufman RJ: From
endoplasmic-reticulum stress to the inflammatory response. Nature.
454:455–462. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Shamu CE, Cox JS and Walter P: The
unfolded-protein-response pathway in yeast. Trends Cell Biol.
4:56–60. 1994. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Ron D and Walter P: Signal integration in
the endoplasmic reticulum unfolded protein response. Nat Rev Mol
Cell Biol. 8:519–529. 2007. View
Article : Google Scholar : PubMed/NCBI
|
|
32
|
Cox JS, Shamu CE and Walter P:
Transcriptional induction of genes encoding endoplasmic reticulum
resident proteins requires a transmembrane protein kinase. Cell.
73:1197–1206. 1993. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Shi Y, Vattem KM, Sood R, An J, Liang J,
Stramm L and Wek RC: Identification and characterization of
pancreatic eukaryotic initiation factor 2 alpha-subunit kinase,
PEK, involved in translational control. Mol Cell Biol.
18:7499–7509. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Zhu L, Jia F, Wei J, Yu Y, Yu T, Wang Y,
Sun J and Luo G: Salidroside protects against homocysteine-induced
injury in human umbilical vein endothelial cells via the regulation
of endoplasmic reticulum stress. Cardiovasc Ther. 35:33–39. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Zhou X, Wei Y, Qiu S, Xu Y, Zhang T and
Zhang S: Propofol decreases endoplasmic reticulum stress-mediated
apoptosis in retinal pigment epithelial cells. PLoS One.
11:e01575902016. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Kassan M, Galán M, Partyka M, Saifudeen Z,
Henrion D, Trebak M and Matrougui K: Endoplasmic reticulum stress
is involved in cardiac damage and vascular endothelial dysfunction
in hypertensive mice. Arterioscler Thromb Vasc Biol. 32:1652–1661.
2012. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Tabas I: The role of endoplasmic reticulum
stress in the progression of atherosclerosis. Circ Res.
107:839–850. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Xue X, Piao JH, Nakajima A, Sakon-Komazawa
S, Kojima Y, Mori K, Yagita H, Okumura K, Harding H and Nakano H:
Tumor necrosis factor alpha (TNFalpha) induces the unfolded protein
response (UPR) in a reactive oxygen species (ROS)-dependent
fashion, and the UPR counteracts ROS accumulation by TNFalpha. J
Biol Chem. 280:33917–33925. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Sprenkle NT, Sims SG, Sánchez CL and
Meares GP: Endoplasmic reticulum stress and inflammation in the
central nervous system. Mol Neurodegener. 12:422017. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Wu GJ, Lin YW, Chuang CY, Tsai HC and Chen
RM: Liver nitrosation and inflammation in septic rats were
suppressed by propofol via downregulating TLR4/NF-κB-mediated iNOS
and IL-6 gene expressions. Life Sci. 195:25–32. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Ding XW, Sun X, Shen XF, Lu Y, Wang JQ,
Sun ZR, Miao CH and Chen JW: Propofol attenuates TNF-α-induced
MMP-9 expression in human cerebral microvascular endothelial cells
by inhibiting Ca2+/CAMK II/ERK/NF-κB signaling pathway.
Acta Pharmacol Sin. 40:1303–1313. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Cheng L, Chen Z, Wang L, Lan Y, Zheng L
and Wu F: Propofol partially attenuates complete freund's
adjuvant-induced neuroinflammation through inhibition of the
ERK1/2/NF-κB pathway. J Cell Biochem. 120:9400–9408. 2019.
View Article : Google Scholar : PubMed/NCBI
|