Introduction
Breast cancer (BCa) is one of the most common types
of malignant tumor in women (1,2). There
were 2.26 million new cases of BCa worldwide in 2019, accounting
for 24.5% of all female-related cancers worldwide. BCa also
accounted for 680,000 deaths in 2019, ranking as the fifth most
common cause of cancer-related mortality worldwide, and the
incidence rate of BCa increases with age (2). Hereditary factors, infertility, an
unhealthy lifestyle and excess mental stress are common risk
factors for BCa (3). The global
incidence rate of BCa is 24.2%, ranking first among cancers
affecting women (4). In China,
~300,000 women are diagnosed with BCa each year. Following the
development and implementation of novel treatment strategies, the
mortality rate of BCa has gradually decreased (4,5).
However, in vast rural areas of China, the decreasing trend of BCa
mortality is not significant, and still accounts for 120,000 deaths
each year (6). Symptoms of early
BCa are not obvious, whereas distant metastasis and multiple organ
disease often occur in advanced late-stage disease, which pose a
significant threat to the lives of affected patients (7). Therefore, the molecular mechanisms
underlying the occurrence and development of BCa must be identified
to provide novel treatment options for BCa.
Mammalian STE20-like protein kinase 1 (MST1) is a
class II GC kinase belonging to the sterile (STE)-20 protein kinase
family; it comprises 487 amino acid residues and has a molecular
weight of 59 kDa (8). MST1 is
widely expressed in most cells of the human body and is a core
component of the Hippo signaling pathway, which serves an important
role in cell differentiation, adhesion, migration, apoptosis and
other physiological activities through the phosphorylation,
dimerization and nuclear localization of target proteins (9,10).
MST1 was reported to promote the differentiation of tumor cells and
to regulate trophoblast differentiation and placental morphogenesis
(11). Moreover, MST1 was
previously reported to serve regulatory roles in cell adhesion,
endocytosis and migration speed by modulating C-C motif chemokine
receptor 7 (CCR7). Downregulated expression levels of MST1 also
inhibited the phosphorylation of CCR7 in mature dendritic cells and
reduced the migration speed of cells by downregulating the
expression of curl protein, myosin l light chain and myosin l light
chain phosphatase (12). Another
previous study found that MST1 promoted the apoptosis of
cardiomyocytes and islet cells (13). In diabetes, MST1 induced apoptotic
cell death by upregulating the expression levels of the BCL2-like
11 protein in β cells, and disrupting normal cardiac function
(14).
The Hippo signaling pathway comprises a group of
conserved kinases and is a key signaling pathway regulating cell
proliferation (15). In mammals,
the membrane protein receptor upstream of the Hippo signaling
pathway senses the growth inhibition signal of the extracellular
environment and then acts on downstream effectors, YY1-associated
protein 1 (YAP) and tafazzin (TAZ) through a series of
kinase-phosphorylation reactions (16). In the Hippo signaling pathway, MST1
binds to and activates the phosphorylation of large tumor
suppressor kinase (LATS)1/2. YAP is the main downstream effector of
this signaling pathway, and it is directly phosphorylated by
LATS1/2, which subsequently inhibits the expression of YAP/TAZ. YAP
can bind to the 14-3–3 protein in the phosphorylated state,
translocate from the nucleus to the cytoplasm, and lose the
transcriptional coactivator activity (17). Therefore, MST1 controls LATS1/2 and
YAP activity by regulating YAP subcellular localization and protein
stability. Long-term activation of YAP, such as its transgenic
expression in the mouse liver, was found to lead to cell
transformation and tumor development, indicating a role for the
Hippo signaling pathway in tumorigenesis and development (18). YAP was found to upregulate
zinc-finger E-box-binding homeobox 1 expression and promote
epithelial-mesenchymal transition (EMT), a key step in tumor
metastasis (19). In addition,
YAP-induced genome instability, and its homologous protein TAZ was
necessary to maintain BCa stem cell self-renewal and tumorigenesis
(20). It was previously reported
that YAP/TAZ activity was also associated with drug resistance and
tumor recurrence, as BCa cells with high YAP/TAZ activity were
resistant to paclitaxel, 5-fluorouracil and Adriamycin (21). Tamoxifen is an important drug
commonly used to treat estrogen receptor (ER)-positive BCa;
however, some ER-positive BCas are not sensitive to tamoxifen
(21). Tamoxifen was shown to
activate YAP/TAZ by stimulating the membrane ER, G protein-coupled
ER (22). Therefore, YAP may play
an important role in the malignant development of BCa.
The present study aimed to investigate whether MST1
was an important biological indicator of the prognosis of BCa and
to determine whether MST1 affected the biological functions of BCa
through the Hippo signaling pathway. This research may provide
novel insights and a basis for the further study of the molecular
mechanisms of BCa.
Materials and methods
Patient studies
A total of 75 BCa and paired adjacent normal (>5
cm from the boundary of the tumor) tissues were obtained from
female patients (age range, 27–79 years) with BCa who underwent
surgical resection at The First People's Hospital of Yibin (Yibin,
China) between October 2012 and May 2014. Patients were followed up
to evaluate the survival rate of patients with BCa at 10 years
post-surgery. Cases lost to follow-up or cases in which death
occurred by causes other than BCa were censored in the survival
analysis. The collected clinical BCa samples were stored at −80°C.
The present study was approved by the Ethics Committee of The First
People's Hospital of Yibin (approval no. 20121011). Written
informed consent was obtained from all patients prior to
participation in the study. The following exclusion criteria were
used: i) Patients with incomplete pathological and clinical
information; ii) patients with recurrent disease; iii) patients
that had received preoperative radiation or chemotherapy, or any
other treatment; iv) patients with other preoperative
complications; v) patients with cognitive or mental disorders; and
vi) pregnant women. The following inclusion criteria were used: i)
BCa tissue samples were all pathologically and confirmed as
BCa.
Cell lines, culture and treatment
The human BCa cell lines, MCF-7, MDA-MB-231 and
SKBR3, and the normal mammary epithelial cell line, MCF-10A, were
obtained from the American Type Culture Collection. All cell lines
were cultured in DMEM (Gibco; Thermo Fisher Scientific, Inc.)
supplemented with 10% FBS (Gibco; Thermo Fisher Scientific, Inc.),
and maintained in a humidified incubator at 37°C with 5%
CO2..
XMU-MP-1 (cat. no. S8334; Selleck Chemicals), which
is an inhibitor of MST1, was dissolved in DMSO and added to the
medium at a final concentration of 0.1%. Cell lines were cultured
in the medium for 1 h at 37°C.
Cell transfection
Lentivirus (LV) containing the MST1 sequence and
negative control (NC; non-targeting) were purchased from Shanghai
Gene Pharma Co., Ltd. Cells were seeded into six-well plates and
cultured to 30–50% confluence. Subsequently, lentiviral
transduction (1 µg/ml) was performed using
Lipofectamine® 3000 reagent (Invitrogen; Thermo Fisher
Scientific, Inc.) according to the manufacturer's instructions.
Following 48 h of transfection, reverse transcription-quantitative
PCR (RT-qPCR) and western blotting were performed to analyze MST1
expression levels. Subsequent experiments were performed 24–72 h
after transfection.
Immunohistochemistry staining
Briefly, tissues were fixed in 10% formalin for 12 h
at room temperature and embedded in paraffin blocks for 8 h at room
temperature. Tissue microarray (TMA) cores (1.5-mm diameter) were
constructed from formalin-fixed paraffin-embedded BCa sections
(4-µm thick). TMA slides were deparaffinized twice with xylene for
15 min each at room temperature and rehydrated three times in a
descending series of ethanol solutions (100, 100, 95 and 80%).
Antigen retrieval was performed with citric acid buffer (10 mM; pH
6.0) in a microwave (800 W) for 15 min, and samples were then
cooled at room temperature for 30 min. All slides were incubated
with 5% goat serum (OriGene Technologies, Inc.) for 15 min at room
temperature to block non-specific binding at room temperature,
followed by an incubation with a rabbit monoclonal anti-MST1
antibody (1:200; cat. no. ab231138; Abcam) at 4°C overnight.
Following the primary antibody incubation, slides were incubated
with an anti-rabbit secondary IgG antibody (1:100; cat. no.
SAP-9100; OriGene Technologies, Inc.) at 37°C for 60 min. The
membranes were then washed with PBS and stained with
3,3′-diaminobenzidine (cat. no. K5007; Dako; Agilent Technologies,
Inc.) for 30 sec at room temperature. Slides were visualized using
a light microscope (magnification, ×100; Zeiss AG).
MST1 immunostaining results were evaluated by two
blinded pathologists. MST1 staining was scored according to the
percentage of positive cells and staining intensity. The scoring
parameters for staining intensity were as follows: 0, negative; 1,
weak; 2, moderate; and 3, strong. The scoring parameters for the
percentage of positive cells were as follows: Negative, 0–5%; 1,
6–25%; 2, 26–50%; 3, 51–75%; and 4, 76–100%. Sections with a total
combined score of <4 were characterized as low MST1 expression,
while sections with a score of ≥4 were characterized as high MST1
expression.
RT-qPCR
Total RNA was extracted from tissues and cell lines
using TRIzol® reagent (Invitrogen; Thermo Fisher
Scientific, Inc.). Total RNA was reverse transcribed into cDNA
using a PrimeScript RT Reagent kit (Takara Bio, Inc.) at 35°C for
10 min, 40°C for 30 min and 76°C for 10 min. qPCR was subsequently
performed using SYBR Premix Ex Taq II (Takara Bio, Inc.) on an ABI
Prism 7500 Real-Time PCR system (Applied Biosystems; Thermo Fisher
Scientific, Inc.). The following primer sequences were used for the
qPCR: MST1 forward, 5′-ATTCGGCTACGGAACAAG-3′ and reverse,
5′-AAAACGGGGTCCCTATTA-3′; and GAPDH forward,
5′-GGAGCGACATCCGTCCAAAAT-3′ and reverse,
5′-GGCTGTTGTCAATCTTCTCATGG-3′. The following thermocycling
conditions were used for the qPCR: Initial denaturation at 90°C for
2 min; followed by 40 cycles at 93°C for 15 sec, 60°C for 30 sec
and 72°C for 30 sec. mRNA expression levels of MST1 were quantified
using the 2−ΔΔCq method (23).
Bioinformatics analysis
Gene Expression Profiling Interactive Analysis
(http://gepia.cancer-pku.cn/index.html), Clinical
Proteomic Tumor Analysis Consortium (CPTAC; http://proteomics.cancer.gov/programs/cptac) and
The Cancer Genome Atlas (TCGA; www.cancer.gov/about-nci/organization/ccg/research/structural-genomics/tcga)
public databases were used to analyze MST1 expression in BCa and
the prognostic value of MST1.
Western blotting
Total protein was extracted using RIPA lysis buffer
(Beyotime Institute of Biotechnology). Total protein was quantified
using the Bradford protein assay (Bio-Rad Laboratories, Inc.) and
50 µg protein/lane was separated via 10% SDS-PAGE. The separated
proteins were subsequently transferred onto PVDF membranes and
blocked with 5% skim milk powder at room temperature for 1 h. The
membranes were then incubated with the following primary antibodies
at 4°C overnight: Anti-MST1 (1:3,000; cat. no. ab231138; Abcam),
anti-LATS1 (1:5,000; cat. no. ab70562; Abcam), anti-YAP (1:5,000;
cat. no. ab205270; Abcam), anti-Bax (1:5,000; cat. no. ab32503;
Abcam), anti-Bcl-2 (1:1,000; cat. no. ab182858; Abcam), anti-Ki-67
(1:1,000; cat. no. ab92742; Abcam) and anti-β-actin (1:5,000; cat.
no. ab8226; Abcam). Following the primary antibody incubation, the
membranes were incubated with secondary antibodies (1:5,000; cat.
nos. ab6721 and ab6789; Abcam) at room temperature for 1 h. Protein
bands were visualized using enhanced chemiluminescence solution
(EMD Millipore) and a ChemiDoc Imaging system (Bio-Rad
Laboratories, Inc.). Protein expression was quantified using
Quantity One version 4.6.5 software (Bio-Rad Laboratories,
Inc.).
Cell Counting Kit-8 (CCK-8) assay
A total of 2×103 MCF-7 and SKBR3
cells/well were transfected with LV for 48 h. Subsequently, 100 µl
cell suspension (1×103 cells/well) was plated into
96-well plates and incubated for 24, 48 and 72 h at 37°C. Following
each incubation, 10 µl CCK-8 solution (Dojindo Molecular
Technologies, Inc.) was added to each well and incubated for 1 h at
37°C. The optical density of each well was measured at a wavelength
of 450 nm using a microplate reader.
Wound healing assay
Briefly, 2 ml cell suspension (1×105
cells/well) was plated into six-well plates and cultured in DMEM
supplemented with 10% FBS at 37°C. After reaching 100% confluence,
the cell monolayer was scratched with a 1-ml pipette tip and then
rinsed twice with PBS to remove nonadherent cells. Cells were
subsequently cultured in serum-free DMEM at 37°C. Following 0, 24
and 48 h of incubation, the distance between the two sides of the
wound was visualized and photographed using a light microscope
(magnification, ×200). The cell migration rate (%) was calculated
using the following equation: [(Original gap distance-gap distance
at each time point)/original gap distance] ×100. ImageJ software
(v2.1.4.7; National Institutes of Health) was used for
analysis.
Flow cytometric analysis of cell
apoptosis and the cell cycle
For cell apoptosis, 2 ml cell suspension
(1×105 cells/well) was plated into six-well plates.
After reaching 100% confluence, cells were centrifuged at 200 × g
for 5 min at room temperature. BCa cells were subsequently
incubated with 5 µl Annexin V-FITC and 5 µl propidium iodide (PI)
solution (Bioworld Technology, Inc.) for 15 min at 37°C. Then,
cells were suspended in 400 µl binding buffer (Bioworld Technology,
Inc.).
For analysis of cell cycle distribution, cells were
cultured as described for cell apoptosis, centrifuged (200 × g; 5
min; room temperature) and fixed in 75% ethanol at −20°C overnight.
The fixed cells were then washed with PBS and incubated with RNase
A for 20 min at room temperature prior to being stained with PI in
the dark for 30 min at 4°C. Cell apoptosis and cell cycle
distribution were analyzed using a FACSCalibur flow cytometer (BD
Biosciences) and FACStation™ 6.1 software (BD Biosciences).
5-Ethynyl-2′-deoxyuridine (EdU)
incorporation assay
A total of 1×105 cells were plated into
96-well plates and cultured until they reached 30–50% confluence.
Cells were then fixed with 4% paraformaldehyde at room temperature
for 30 min, and nuclei were permeabilized with 0.5% Triton X-100
solution for 30 min at room temperature. The cells were
subsequently incubated with 50 µM EdU, 1X ApolloR reaction cocktail
(100 µl) and 1X Hoechst 33342 (100 µl) for 30 min at room
temperature. Cell proliferation was measured by determining the
mean number of the cells in three randomly selected fields of view
from each sample using a fluorescence microscope (BioTek
Instruments, Inc.; magnification, ×100).
Statistical analysis
Statistical analysis was performed using SPSS
version 22.0 software (SPSS, Inc.) and GraphPad Prism version 6.0
software (GraphPad Software, Inc.). Data are presented as the mean
± SD. All experiments were repeated in triplicate. Statistical
differences between two groups were analyzed using an unpaired or
paired Student's t-test. Unpaired Student's t-tests
were used to determine statistical in TCGA and CPTAC data.
Associations between clinicopathological parameters and MST1
expression levels were analyzed using a χ2 test for
small sample sizes. Kaplan-Meier survival analysis, followed by a
log-rank test, and Cox univariate and multivariate regression
analyses were used to analyze the prognostic significance of MST1.
Statistical differences between multiple groups were determined
using a one-way ANOVA followed by a Tukey's post hoc test.
P<0.05 was considered to indicate a statistically significant
difference.
Results
MST1 expression levels are
downregulated in BCa tissues and cell lines
The expression levels of MST1 were analyzed in BCa
tissues obtained from TCGA database. MST1 expression levels were
significantly downregulated in BCa tissues compared with the normal
tissues (Fig. 1A). The protein
expression levels of MST1 in BCa specimens from the Clinical
Proteomic Tumor Analysis Consortium (CPTAC) (https://proteomics.cancer.gov/programs/cptac)
public database were also analyzed, and the results showed that the
expression levels of MST1 were downregulated in BCa tissues
compared with normal tissues (Fig.
1B). In addition, patients with low expression levels of MST1
had a poor prognosis compared with patients with high MST1
expression levels (Fig. 1C). The
expression levels of MST1 were subsequently analyzed in 75 tumor
and non-tumoral tissues from patients with BCa from The First
People's Hospital of Yibin to verify its expression in BCa. The
results demonstrated that MST1 mRNA expression levels were
significantly downregulated in BCa tissues compared with normal
tissues (Fig. 1D). The results of
the western blotting analysis also showed that MST1 protein
expression levels were notably downregulated in BCa tissues
compared with normal tissues (Fig.
1E). The mRNA expression levels of MST1 were also downregulated
in BCa cell lines compared with the normal mammary epithelial cell
line, MCF-10A (Fig. 1F).
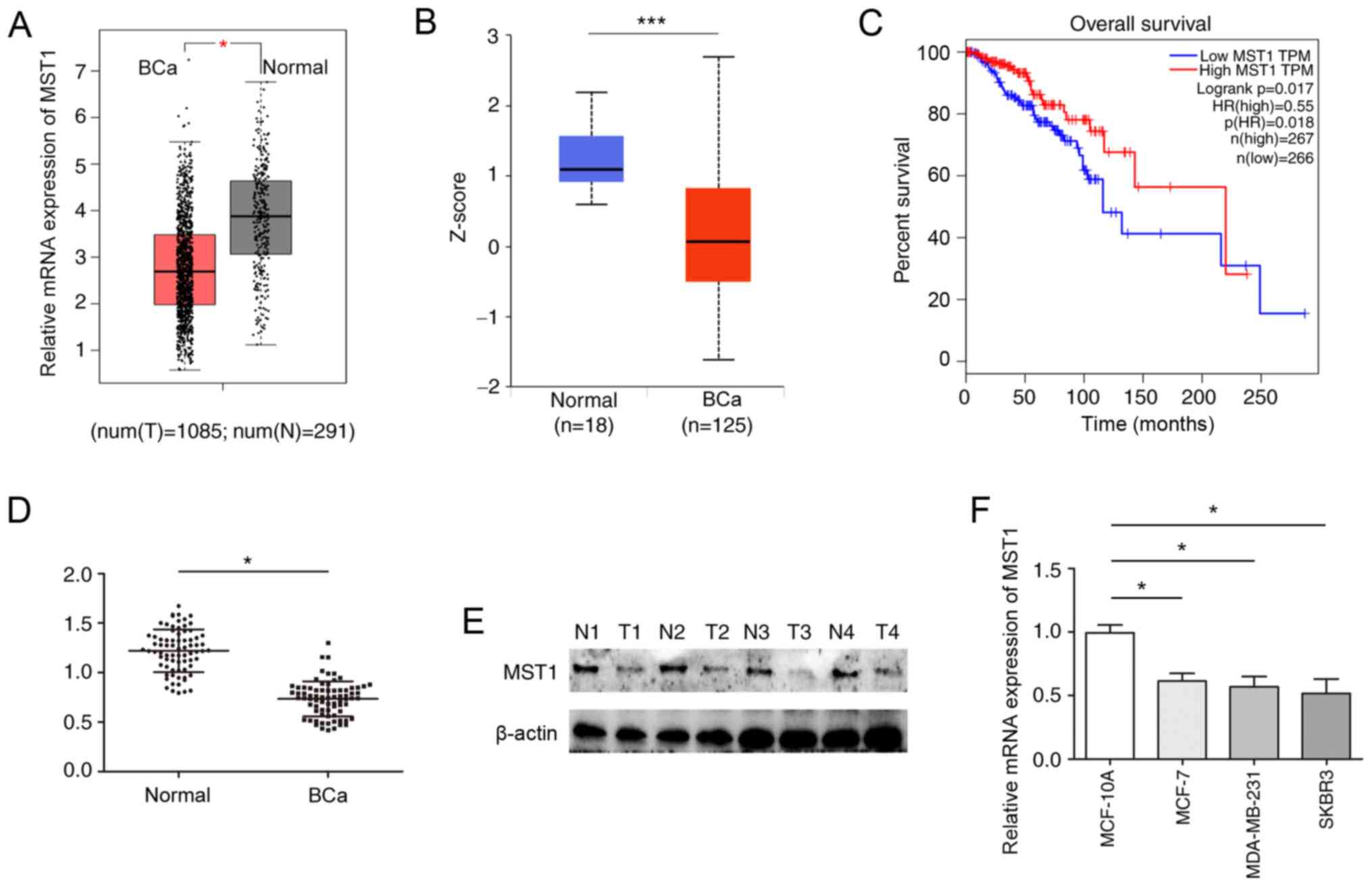 | Figure 1.MST1 expression levels are
downregulated in BCa tissues and cell lines. (A) Expression levels
of MST1 in BCa and adjacent normal tissues were analyzed in a BCa
dataset from TCGA (T=1,085; N=291). (B) Expression levels of MST1
in BCa and normal tissues obtained from the Clinical Proteomic
Tumor Analysis Consortium public database. (C) Kaplan-Meier
survival analysis and a log-rank test were performed to analyze the
relationship between MST1 expression levels and the overall
survival of patients using data from TCGA database (low MST1
expression, 266 patients; high MST1 expression, 267 patients). (D)
RT-qPCR was used to analyze the mRNA expression levels of MST1 in
BCa and adjacent normal tissues (n=75). (E) Western blotting was
performed to determine the protein expression levels of MST1 in BCa
and adjacent normal tissues. (F) RT-qPCR was performed to analyze
the expression levels of MST1 in BCa cell lines. *P<0.05,
***P<0.001. BCa, breast cancer; MST1, mammalian STE20-like
protein kinase 1; N, normal; TCGA, The Cancer Genome Atlas;
RT-qPCR, reverse transcription-quantitative PCR; T, tumor. |
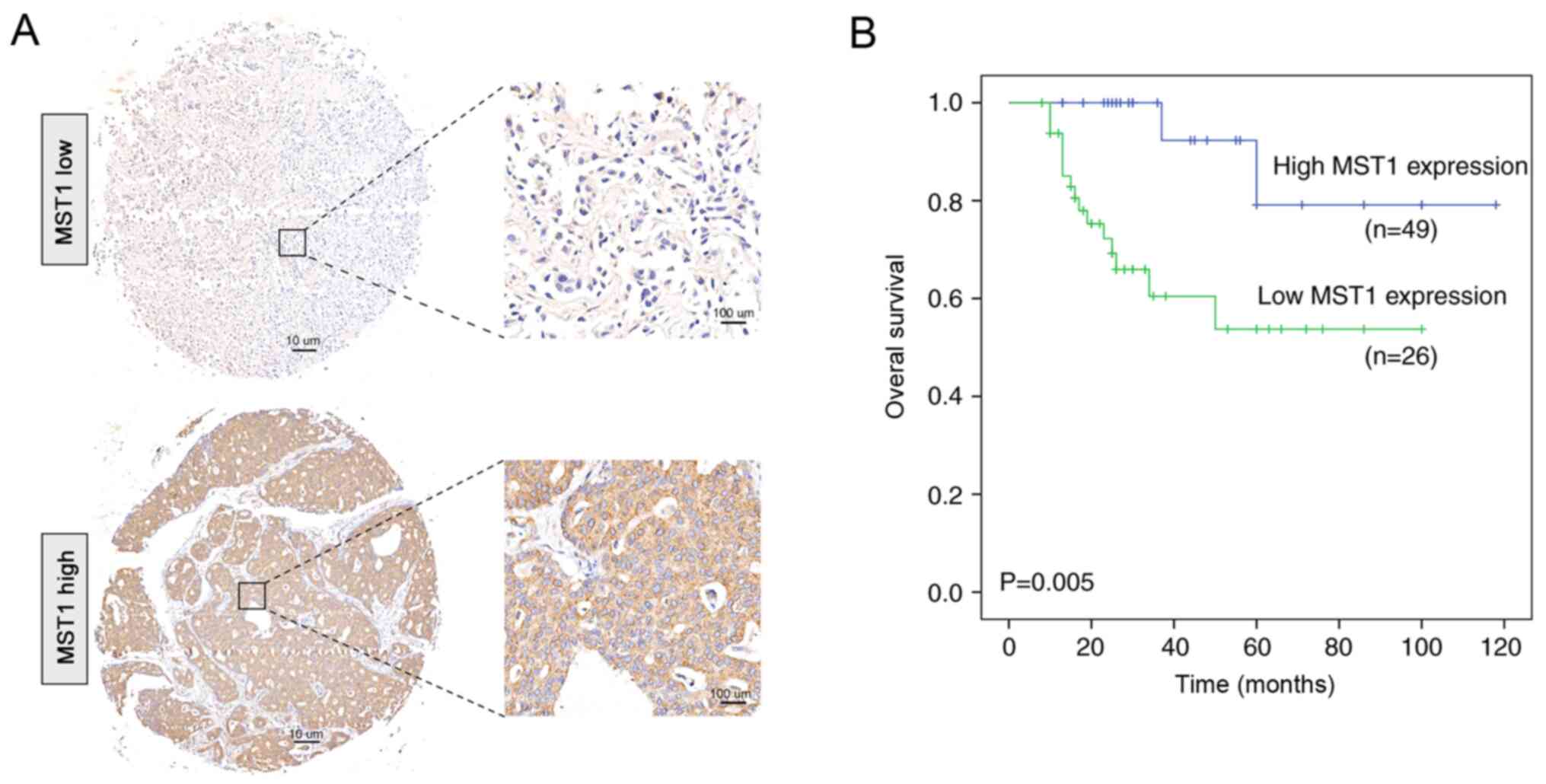
Downregulated expression levels of
MST1 are associated with worse clinicopathological features in
patients with BCa
According to the immunohistochemical staining,
patients with BCa were divided into two groups: MST1 low expression
and MST1 high expression (Fig. 2A).
The results of the immunohistochemical staining for MST1 expression
is presented in Fig. S1. The
prognosis of patients with BCa with low MST1 expression was worse
compared with patients with high expression (Fig. 2B). Furthermore, by analyzing the
clinicopathological features of the two groups of patients, MST1
expression was found to be significantly associated with tumor size
(P<0.001) and clinical stage (P=0.003) (Table I). Through Cox univariate analysis,
MST1 expression (P=0.005), tumor size (P 0.004) and clinical stage
(P=0.019) were discovered to be significantly associated with
overall survival (OS) (Table II).
Cox multivariate analysis revealed that OS was significantly
associated with MST1 expression (P=0.009), tumor size (P=0.010) and
clinical stage (P=0.013) (Table
II), suggesting that MST1 expression may be an independent
prognostic factor for OS in BCa.
 | Table I.Association between MST1 expression
levels and clinicopathological features of patients with breast
cancer. |
Table I.
Association between MST1 expression
levels and clinicopathological features of patients with breast
cancer.
|
|
| MST1
expression |
|
|---|
|
|
|
|
|
|---|
| Clinicopathological
feature | Cases (n=75) | High (n=26) | Low (n=49) | P-value |
|---|
| Age, years |
|
|
| 0.228 |
|
<60 | 39 | 16 | 23 |
|
|
≥60 | 36 | 10 | 26 |
|
| Tumor size, cm |
|
|
| <0.001 |
| ≤2 | 38 | 21 | 17 |
|
|
>2 | 37 | 5 | 32 |
|
| Clinical stage |
|
|
| 0.003 |
|
I/II | 39 | 20 | 19 |
|
|
III/IV | 36 | 6 | 30 |
|
| Lymph node
metastasis |
|
|
| 0.110 |
| ≤3
metastatic sites | 38 | 15 | 23 |
|
| ≥3
metastatic sites | 37 | 11 | 26 |
|
| Estrogen receptor
status |
|
|
| 0.128 |
|
Positive | 40 | 17 | 23 |
|
|
Negative | 35 | 9 | 26 |
|
| Progesterone
receptor status |
|
|
| 0.089 |
|
Positive | 48 | 20 | 28 |
|
|
Negative | 27 | 6 | 21 |
|
| HER2 status |
|
|
| 0.592 |
|
Positive | 43 | 16 | 27 |
|
|
Negative | 32 | 10 | 22 |
|
 | Table II.Cox univariate and multivariate
analyses of different prognostic variables influencing overall
survival in patients with breast cancer. |
Table II.
Cox univariate and multivariate
analyses of different prognostic variables influencing overall
survival in patients with breast cancer.
|
|
| Univariate
analysis | Multivariate
analysis |
|---|
|
|
|
|
|
|---|
| Clinicopathological
feature | Cases (n=75) | HR (95% CI) | P-value | HR (95% CI) | P-value |
|---|
| Age, years |
| 0.630
(0.448–1.064) | 0.736 |
|
|
|
<60 | 39 |
|
|
|
|
|
≥60 | 36 |
|
|
|
|
| Tumor size, cm |
| 1.397
(0.655–3.845) | 0.004 | 1.048
(0.596–3.674) | 0.010 |
| ≤2 | 38 |
|
|
|
|
|
>2 | 37 |
|
|
|
|
| Clinical stage |
| 1.554
(1.078–3.994) | 0.019 | 1.484
(0.980–3.481) | 0.013 |
|
I/II | 39 |
|
|
|
|
|
III/IV | 36 |
|
|
|
|
| Lymph node
metastasis |
| 1.073
(0.761–2.663) | 0.166 |
|
|
| ≤3
metastatic sites | 38 |
|
|
|
|
| >3
metastatic sites | 37 |
|
|
|
|
| Estrogen receptor
status |
| 1.886
(1.840–3.617) | 0.189 |
|
|
|
Positive | 40 |
|
|
|
|
|
Negative | 35 |
|
|
|
|
| Progesterone
receptor status |
| 1.073 (0.647–1.
747) | 0.307 |
|
|
|
Positive | 48 |
|
|
|
|
|
Negative | 27 |
|
|
|
|
| HER2 status |
| 0.843
(0.663–1.850) | 0.647 |
|
|
|
Positive | 43 |
|
|
|
|
|
Negative | 32 |
|
|
|
|
| MST1 |
| 1.308
(0.537–2.478) | 0.005 | 1.186
(0.634–2.705) | 0.009 |
|
Low | 49 |
|
|
|
|
|
High | 26 |
|
|
|
|
Overexpression of MST1 inhibits BCa
cell proliferation, migration and apoptosis
MCF-7 cells had the highest expression of MST1 in
the three tumor cell lines, while SKBR3 cells had the lowest
expression of MST1 in the three tumor cell lines. Therefore, MCF-7
and SKBR3 cells were selected for use in subsequent experiments.
LV-MST1 was used to successfully overexpress MST1 in BCa cells
(Fig. 3A). The results of the CCK-8
assay revealed that MST1 overexpression significantly inhibited the
proliferation of BCa cells compared with the NC group (Fig. 3B). The results of the EdU assay
further verified that the overexpression of MST1 significantly
inhibited cell proliferation compared with the NC group (Fig. 3C). The migration of BCa cells was
analyzed using a wound healing assay, and the results revealed that
cell migration was also significantly decreased following MST1
overexpression compared with the NC group at both 24 and 48 h
(Fig. 3D). Furthermore, the effect
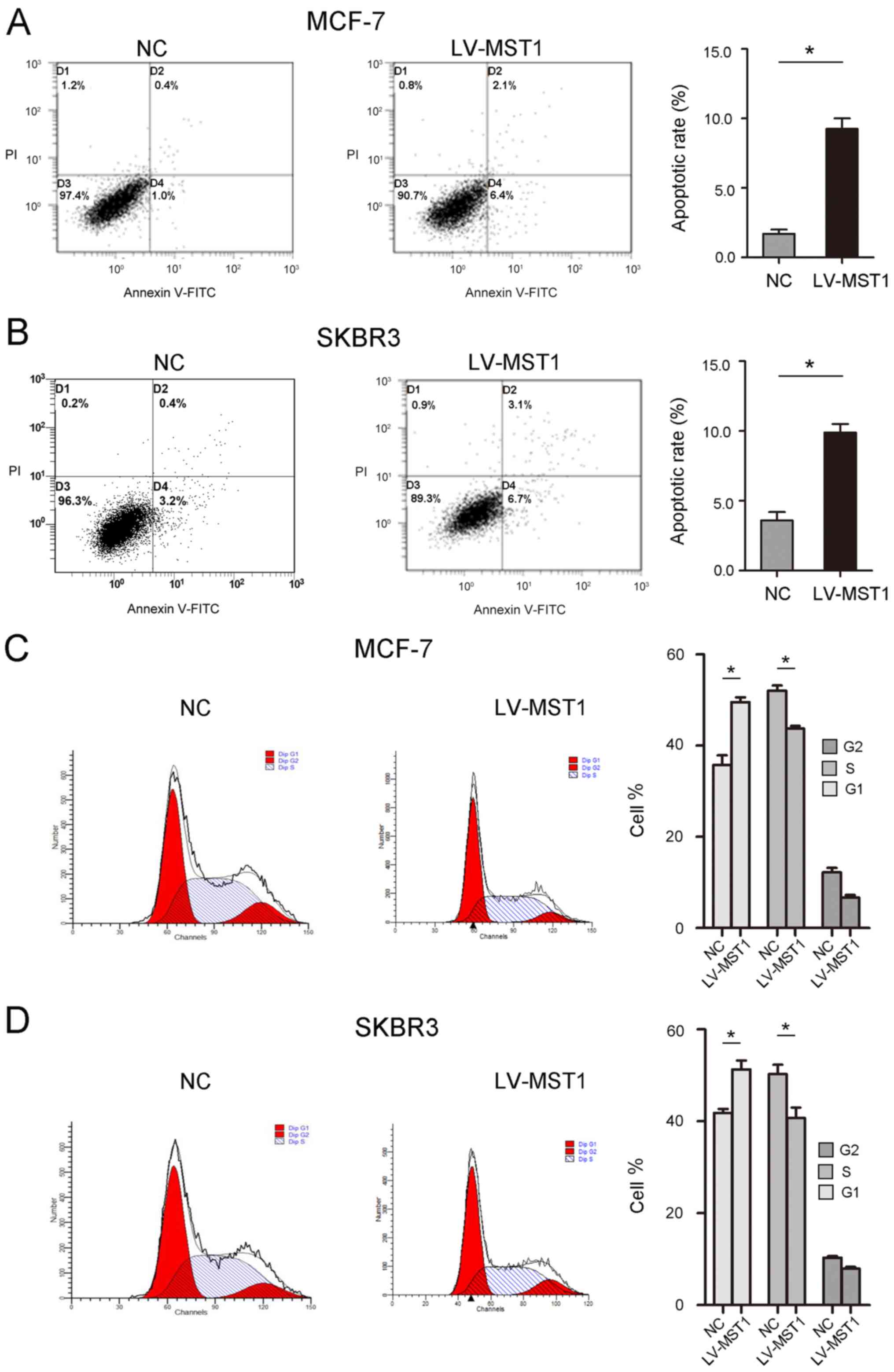
of MST1 on the apoptosis of BCa cells was determined. Flow
cytometric analysis revealed a significant increase in the
apoptotic rate of both MCF-7 (8.5 vs. 1.4%) and SKBR3 (9.8 vs.
3.6%,) cells transfected with LV-MST1 compared with the NC group
(Fig. 4A and B). In addition, the
number of cells arrested in the G1 phase was
significantly increased, and the number of cells arrested in the S
phase was significantly decreased in LV-MST1-transfected cells
compared with NC-transfected cells (Fig. 4C and D).
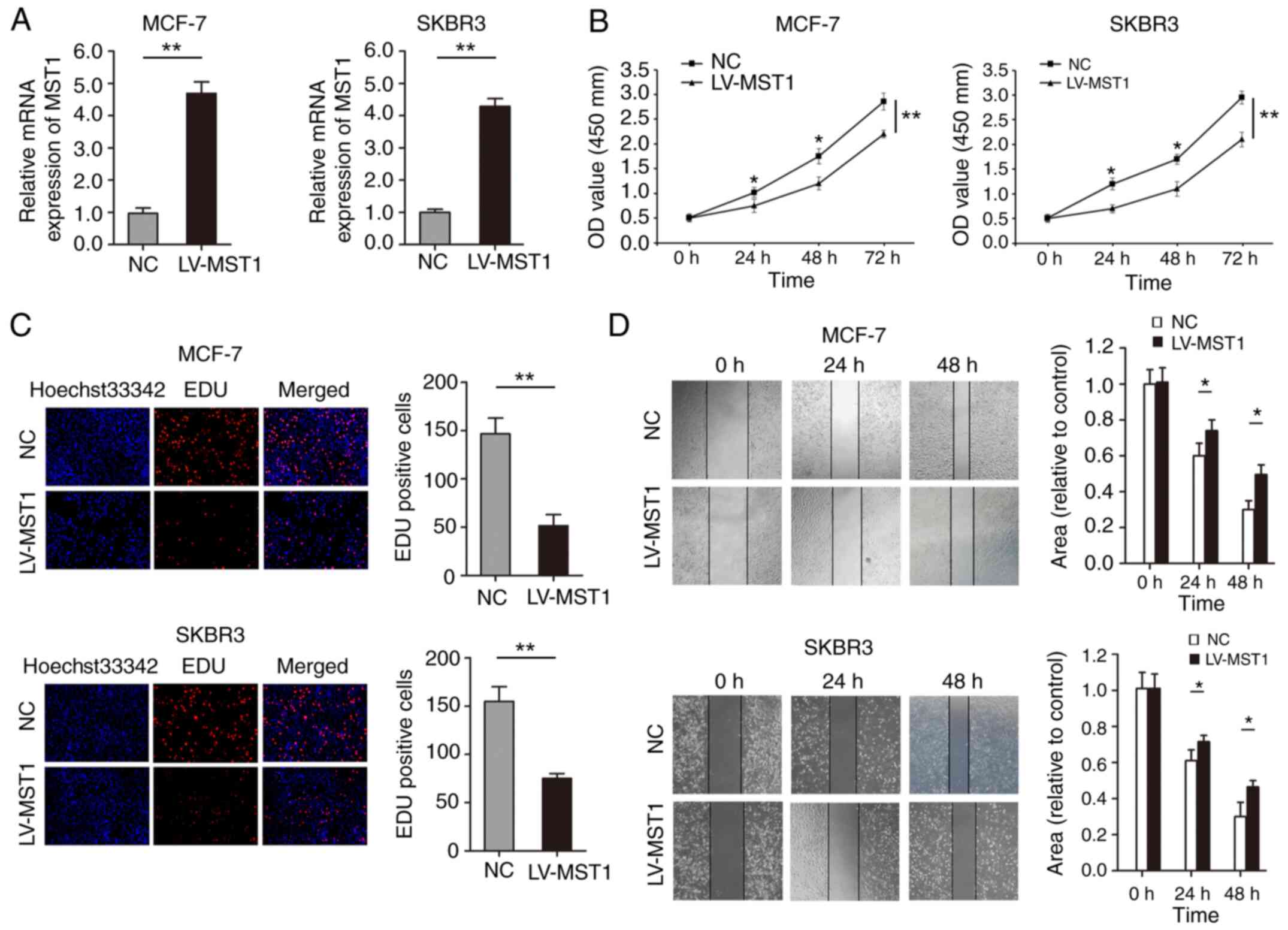 | Figure 3.Overexpression of MST1 regulates the
proliferation, migration and apoptosis of BCa cell lines. (A)
Transfection efficiency of MCF-7 and SKBR3 cells transfected with
LV-MST1 was determined using reverse transcription-quantitative
PCR. (B) Cell Counting Kit-8 and (C) EdU incorporation assays
(magnification, ×200) were performed to determine the effects of
MST1 overexpression on the proliferation of BCa cells. (D) Wound
healing assays were conducted to determine the effects of MST1
overexpression on BCa cell migration (magnification, ×100).
*P<0.05, **P<0.01. MST1, mammalian STE20-like protein kinase
1; BCa, breast cancer; LV, lentivirus; OD, optical density; NC,
negative control; EdU, 5-Ethynyl-2′-deoxyuridine. |
Inhibitor of the Hippo signaling
pathway reverses MST1-induced effects in BCa
To determine whether the Hippo signaling pathway
mediated the biological functions of MST1 in BCa, the Hippo
signaling pathway inhibitor, XMU-MP-1 (an inhibitor of MST1/2), was
used to inhibit the function of the Hippo signaling pathway in BCa
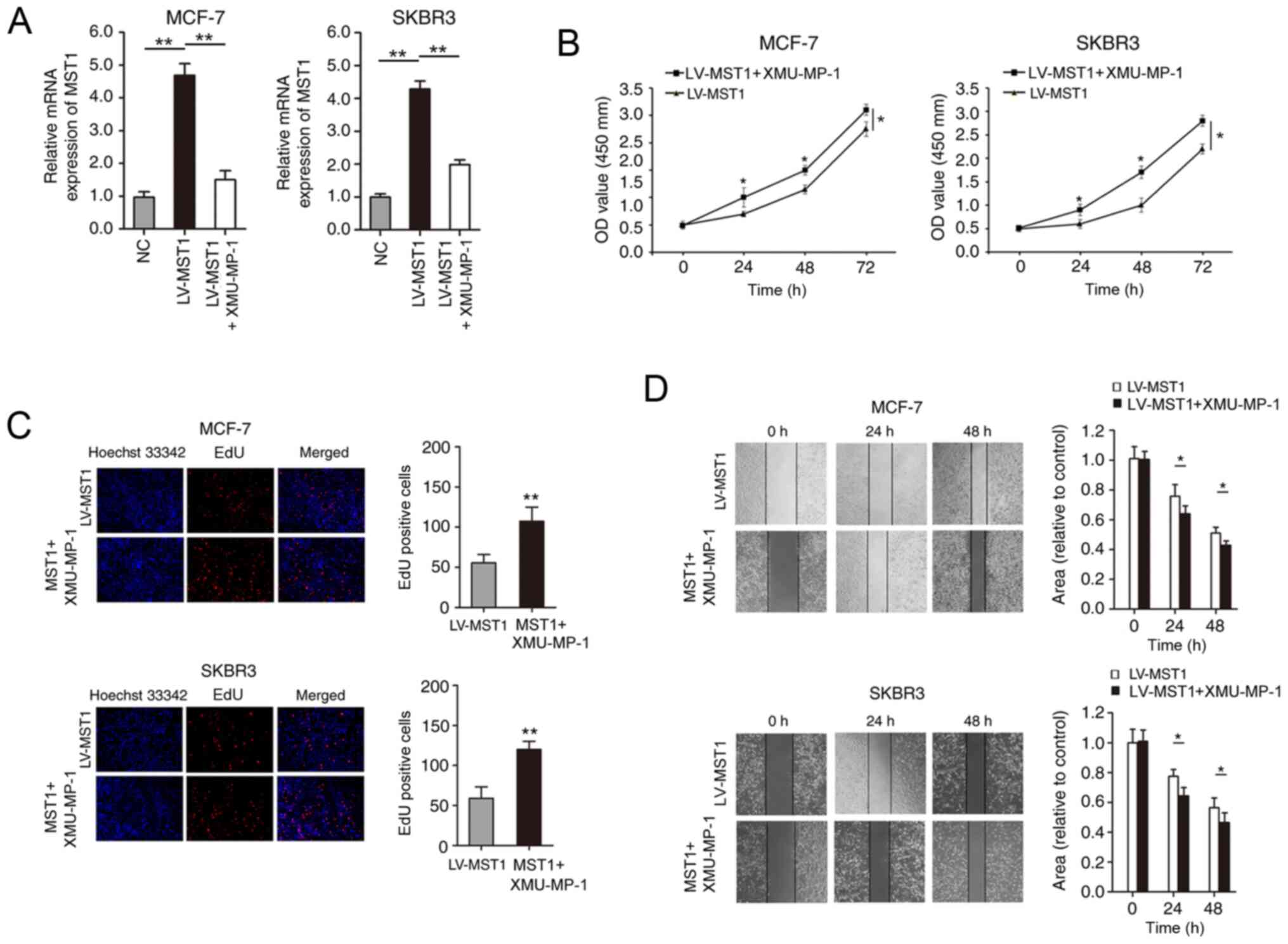
cells overexpressing MST1. RT-qPCR analysis demonstrated that the
treatment of MST1-overexpressing cells with the inhibitor
downregulated the expression levels of MST1 (Fig. 5A). The results of the CCK-8 and EdU
incorporation assays revealed that the inhibited proliferative
ability in the LV-MST1 group was partially restored in the LV-MST1
+ XMU-MP-1 group (Fig. 5B and C).
BCa cell migration was analyzed using wound healing assays; the
results revealed that cell migration was also significantly
increased in the MST1 + XMU-MP-1 cell group compared with the
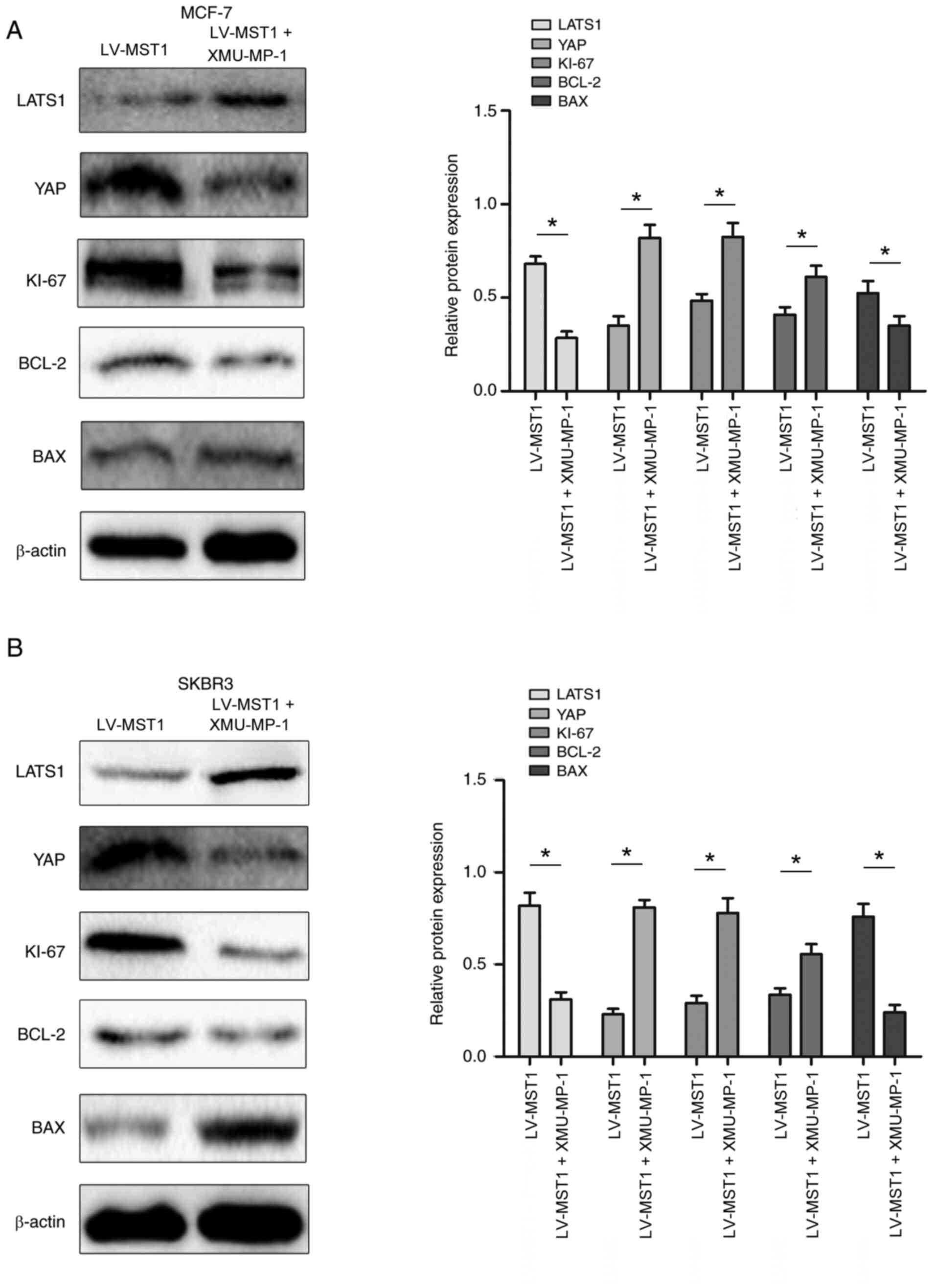
LV-MST1 group (Fig. 5D). Finally,
western blotting analysis was performed to analyze the expression
levels of key proteins in the Hippo signaling pathway. The
expression levels of LATS1 and Bax were significantly
downregulated, while the expression levels of YAP, Bcl-2 and Ki-67
were significantly upregulated in the LV-MST1 + XMU-MP-1 group
compared with the LV-MST1 group in both cell lines (Fig. 6A and B).
Discussion
BCa is one of the most common types of malignant
tumor in women, and its incidence continues to increase annually.
In women, there were 2.26 million new cases of breast cancer
worldwide in 2019, and BCa ranks second in incidence after uterine
cancer, thus it poses a significant threat to the health of women,
especially as the age of onset is steadily decreasing (2,20).
Currently, the primary prevention of BCa has not been achieved;
therefore, strengthening secondary prevention, early detection,
early diagnosis and early treatment methods may serve a vital role
in reducing the mortality rates of patients with BCa, thereby
improving the survival rates (24).
Despite recent significant advances in understanding the mechanisms
of BCa development and the improvement of treatment strategies, BCa
remains the second leading cause of cancer-related mortality in
women, and death is almost always due to metastasis (25). The prognosis of patients with
metastatic BCa is poor, with an average 5-year survival rate of
only 27% (26). Although the
expression of certain molecular markers in the different
histological subtypes of BCa have strong prognostic value (such as
HER2, Ras and K-Ras), the primary treatment of BCa still comprises
chemotherapy or radiotherapy, and surgical resection for isolated
lesions; however, the long-term effects of these treatments on
survival is limited (27). Although
chemotherapy, radiotherapy and surgical resection control the
growth of primary tumors, they are not effective in preventing
recurrence. Therefore, it remains a priority to identify the
underlying molecular mechanism of BCa progression to help devise
more precise, targeted treatments for BCa and to discover novel
prognostic markers.
MST1, which belongs to the human serine/threonine
kinase family, shares considerable homology with budding yeast
kinase sps1 and ste2 in its kinase domain (28). MST1 was reported to render cells
highly sensitive to death receptor-mediated apoptosis by
accelerating the activation of caspase-3, thus promoting apoptosis
(29). Another study revealed that
MST1 was a strong predictor of BCa prognosis (30). In pancreatic cancer, TNF
receptor-associated factor 6 promoted the degradation of MST1
through the ubiquitination degradation pathway, upregulated the
expression levels of YAP and regulated the malignant proliferation
and metastasis of tumor cells (31). In addition, the knockdown of
microRNA (miR)-135b induced the apoptosis of gastric cancer cells
by inhibiting the MAPK signaling pathway and upregulating the
expression levels of MST1, thus inhibiting the proliferation of
gastric cancer cells and increasing their resistance to cisplatin
(32). Notably, in human
HER2-positive BCa cells, the knockout of fibroblast growth factor
receptor 4 led to the activation of MST1 and increased the nuclear
localization of MST1, resulting in N-terminal cleavage and
autophosphorylation of MST1, which subsequently induced BCa cell
apoptosis (33).
The results of the present study were similar to
those reported previously. A large number of clinical BCa tissues
were collected and the association between MST1 expression and the
clinicopathological characteristics and the poor prognosis of
patients with BCa were analyzed. In most cases, mRNA expression
levels are strongly associated with the protein expression levels;
however, due to gene post-transcriptional modifications or
regulation, the balance between mRNA and protein expression levels
can be disrupted. All the biological functions of MST1 depend on
the expression of the protein (34); therefore, the present study analyzed
the association between MST1 protein expression and prognosis using
immunohistochemistry staining instead of determining mRNA
expression levels. The results of the current study revealed that
MST1 expression levels were significantly downregulated in BCa
tissues and were significantly associated with the poor prognosis
of patients. The expression levels of MST1 were also analyzed in
BCa cell lines, in which MST1 was overexpressed using lentiviral
vectors. MST1 overexpression significantly inhibited the
proliferation and migration of BCa cells, and promoted apoptosis,
indicating that MST1 may act as a tumor suppressor. The Hippo
signaling pathway inhibits cell growth (35). In mammals, the upstream membrane
protein receptor of the Hippo signaling pathway senses
extracellular growth inhibition signals (36). Once the extracellular growth
inhibition signal is sensed, a series of kinase cascade
phosphorylation reactions are activated to result in the
phosphorylation of the downstream effectors, YAP and TAZ.
Subsequently, cytoskeletal proteins bind to phosphorylated YAP and
TAZ, inducing their retention in the cytoplasm and a decrease in
their nuclear activity to regulate organ size and cell growth
(37). In hepatocellular carcinoma,
miR-29c-3p was found to inhibit the invasion and metastasis of
hepatocellular carcinoma cells through the Hippo signaling pathway
(38). In addition, the interaction
between WWC family member 3 (WWC3) and disheveled reduced the
interaction between WWC3 and LATS1, which subsequently reduced the
phosphorylation of LATS1, increased the nuclear import of YAP, and
weakened the role of Hippo signaling pathway in lung cancer cells
(39). MST1 is a key factor in the
Hippo signaling pathway (39).
XMU-MP-1 is an inhibitor of the sterile 20-like kinases MST1, which
can inhibit MST1 activity, activate the downstream effector YAP and
promote cell growth (40). In the
present study, the XMU-MP-1 pathway inhibitor was used to further
verify whether MST1 inhibited the progression of BCa through the
Hippo signaling pathway. XMU-MP-1 successfully reversed the
inhibitory effect of MST1 on BCa cell functions (proliferation,
migration and apoptosis). Subsequently, the expression levels of
protein involved in the Hippo signaling pathway were altered, that
is, the expression levels of LATS1 were downregulated, while the
expression of YAP was upregulated, indicating that MST1 may play a
role in BCa through the Hippo signaling pathway. However, there are
some limitations to the study. For example, further investigations
into how MST1 affected the Hippo signaling pathway and how it
altered the expression levels of proliferation-related proteins and
the apoptotic process (except for the expression levels of Bax and
Bcl-2) were not performed. Future studies will aim to determine
other influencing factors that may affect the role of MST1 in BCa,
which may provide further evidence that MST1 could be used as a
prognostic biomarker in BCa.
In conclusion, the findings of the present study
revealed that MST1 expression levels were significantly
downregulated in BCa tissues and cells, and were closely associated
with tumor size and clinical stage. The downregulated expression
was also closely associated with the poor prognosis of patients
with BCa. Mechanistically, the present findings suggested that MST1
may inhibit BCa cell proliferation and migration and promote its
apoptosis by regulating the Hippo signaling pathway.
Supplementary Material
Supporting Data
Acknowledgements
Not applicable.
Funding
The present study was supported by grants from The
Key Research Projects on Application Foundation of Sichuan Science
and Technology Department (grant no. 2017JY0030) and Yibin Health
and Family Planning Commission (grant no. 2017ZB031).
Availability of data and materials
The datasets used and/or analyzed during the current
study are available from the corresponding author on reasonable
request.
Authors' contributions
XJ and MX conceptualized and designed the study. XJ,
LZ and SX made substantial contributions to the conception and
design of the study, performed the experiments, and analyzed and
interpreted the data. XJ and MX wrote the manuscript. ZC, JT and JY
analyzed the data and performed the statistical analysis. All
authors read and approved the manuscript, and take public
responsibility for appropriate portions of the content. XJ and MX
confirm the authenticity of all the raw data.
Ethics approval and consent to
participate
The present study was approved by the Ethics
Committee of The First People's Hospital of Yibin (Yibin, China;
approval no. 20121011). Written informed consent was obtained from
all patients prior to participation in the study.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Harbeck N and Gnant M: Breast cancer.
Lancet. 18:1134–1150. 2017. View Article : Google Scholar
|
|
2
|
Britt KL, Cuzick J and Phillips KA: Key
steps for effective breast cancer prevention. Nat Rev Cancer.
20:417–436. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
McGuire A, Brown JA, Malone C, McLaughlin
R and Kerin MJ: Effects of age on the detection and management of
breast cancer. Cancers (Basel). 7:908–29. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Heer E, Harper A, Escandor N, Sung H,
McCormack V and Fidler-Benaoudia MM: Global burden and trends in
premenopausal and postmenopausal breast cancer: A population-based
study. Lancet Glob Health. 8:e1027–e1037. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Anastasiadi Z, Lianos GD, Ignatiadou E,
Harissis HV and Mitsis M: Breast cancer in young Women: An
overview. Updates Surg. 69:313–317. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Li Q, Liu J, Jiang Z and Liu Q: CSCO
breast cancer guideline: Precise, economical and oriental. Sci
China Life Sci. 63:1–3. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Winters S, Martin C, Murphy D and Shokar
NK: Breast cancer epidemiology, prevention, and screening. Prog Mol
Biol Transl Sci. 151:1–32. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Galan JA and Avruch J: MST1/MST2 Protein
Kinases: Regulation and physiologic roles. Biochemistry.
55:5507–5519. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Hui OY, Zhou EX and Wang H: Mst1-Hippo
pathway triggers breast cancer apoptosis via inducing mitochondrial
fragmentation in a manner dependent on JNK-Drp1 axis. Onco Targets
Ther. 12:1147–1159. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Harvey KF, Zhang X and Thomas DM: The
Hippo pathway and human cancer. Nat Rev Cancer. 13:246–257. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Hong AW, Meng Z and Guan KL: The Hippo
pathway in intestinal regeneration and disease. Nat Rev
Gastroenterol Hepatol. 13:324–337. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Torres-Bacete J, Delgado-Martin C,
Gomez-Moreira C, Simizu S and Rodríguez-Fernández JL: The mammalian
sterile 20-like 1 kinase controls selective CCR7-dependent
functions in human dendritic cells. J Immunol. 195:973–81. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Zhang M, Zhang L, Hu J, Lin J, Wang T,
Duan Y, Man W, Feng J, Sun L, Jia H, et al: MST1 coordinately
regulates autophagy and apoptosis in diabetic cardiomyopathy in
mice. Diabetologia. 59:2435–2447. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Zhao WB, Lu Q, Nguyen MN, Su Y, Ziemann M,
Wang LN, Kiriazis H, Puthalakath H, Sadoshima J, Hu HY and Du XJ:
Stimulation of β-adrenoceptors up-regulates cardiac expression of
galectin-3 and BIM through the hippo signalling pathway. Br J
Pharmacol. 176:2465–2481. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Singh A, Ramesh S, Cibi DM, Yun LS, Li J,
Li L, Manderfield LJ, Olson EN, Epstein JA and Singh MK: Hippo
signaling mediators yap and taz are required in the epicardium for
coronary vascµl ature development. Cell Rep. 15:1384–1393. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Yu FX, Zhao B and Guan KL: Hippo pathway
in organ size control, tissue homeostasis, and cancer. Cell.
163:811–828. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Nallet-Staub F, Marsaud V, Li L, Gilbert
C, Dodier S, Bataille V, Sudol M, Herlyn M and Mauviel A:
Pro-invasive activity of the Hippo pathway effectors YAP and TAZ in
cutaneous melanoma. J Invest Dermatol. 134:123–132. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
18
|
He M, Zhou Z, Shah AA, Hong Y, Chen Q and
Wan Y: New insights into posttranslational modifications of Hippo
pathway in carcinogenesis and therapeutics. Cell Div. 11:42016.
View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Vincent-Mistiaen Z, Elbediwy A, Vanyai H,
Cotton J, Stamp G, Nye E, Spencer-Dene B, Thomas GJ, Mao J and
Thompson B: YAP drives cutaneous squamous cell carcinoma formation
and progression. Elife. 7:e333042018. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Maugeri-Saccà M and De Maria R: Hippo
pathway and breast cancer stem cells. Crit Rev Oncol Hematol.
99:115–122. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Rodriguez D, Ramkairsingh M, Lin X, Kapoor
A, Major P and Tang D: The central contributions of breast cancer
stem cells in developing resistance to endocrine therapy in
estrogen receptor (ER)-positive breast cancer. Cancers (Basel).
11:10282019. View Article : Google Scholar
|
|
22
|
Cortes E, Lachowski D, Rice A, Thorpe SD,
Robinson B, Yeldag G, Lee DA, Ghemtio L, Rombouts K and Del Río
Hernández AE: Tamoxifen mechanically deactivates hepatic stellate
cells via the G protein-coupled estrogen receptor. Oncogene.
38:2910–2922. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) method. Methods. 25:402–408. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Kolak A, Kamińska M, Sygit K, Budny A,
Surdyka D, Kukiełka-Budny B and Burdan F: Primary and secondary
prevention of breast cancer. Ann Agric Environ Med. 24:549–553.
2017. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Hosonaga M, Saya H and Arima Y: Molecular
and cellular mechanisms underlying brain metastasis of breast
cancer. Cancer Metastasis Rev. 39:711–720. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Siegel RL, Miller KD and Jemal A: Cancer
statistics. 2016. CA Cancer J Clin. 66:7–30. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
McDonald ES, Clark AS, Tchou J, Zhang P
and Freedman GM: Clinical diagnosis and management of breast
cancer. J Nucl Med. 57 (Suppl 1):9S–16S. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Kim MK, Jang JW and Bae SC: DNA binding
partners of YAP/TAZ. BMB Rep. 51:126–133. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
He Z, Zhao TT, Jin F, Li JG, Xu YY, Dong
HT, Liu Q, Xing P, Zhu GL, Xu H and Miao ZF: Downregulation of
RASSF6 promotes breast cancer growth and chemoresistance through
regulation of Hippo signaling. Biochem Biophys Res Commun.
503:2340–2347. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Li JA, Kuang T, Pu N, Fang Y, Han X, Zhang
L, Xu X, Wu W, Wang D, Lou W and Rong Y: TRAF6 Regulates YAP
signaling by promoting the ubiquitination and degradation of MST1
in pancreatic cancer. Clin Exp Med. 19:211–218. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Lin XY, Cai FF, Wang MH, Pan X, Wang F,
Cai L, Cui RR, Chen S and Biskup E: Mammalian sterile 20-like
kinase 1 expression and its prognostic significance in patients
with breast cancer. Oncol Lett. 14:5457–5463. 2017.PubMed/NCBI
|
|
32
|
Zhou J and Che Q: Poor expression of
microRNA-135b results in the inhibition of cisplatin resistance and
proliferation and induces the apoptosis of gastric cancer cells
through MST1-mediated MAPK signaling pathway. FASEB J.
33:3420–3436. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Turunen SP, Nandelstadh PV, Öhman T,
Gucciardo E, Seashore-Ludlow B, Martins B, Rantanen V, Li H,
Höpfner K, Östling P, et al: FGFR4 phosphorylates MST1 to confer
breast cancer cells resistance to MST1/2-dependent apoptosis. Cell
Death Differ. 26:2577–2593. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Buccitelli C and Selbach M: mRNAs,
proteins and the emerging principles of gene expression control.
Nat Rev Genet. 21:630–644. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Kulkarni A, Chang MT, Vissers JHA, Dey A
and Harvey KF: The hippo pathway as a driver of select human
cancers. Trends Cancer. 6:781–796. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Zheng Y and Pan D: The hippo signaling
pathway in development and disease. Dev Cell. 50:264–282. 2019.
View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Tao Y, Cai F, Shan L, Jiang H, Ma L and Yu
Y: The hippo signaling pathway: An emerging anti-cancer drug
target. Discov Med. 24:7–18. 2017.PubMed/NCBI
|
|
38
|
Wu H, Zhang W, Wu Z, Liu Y, Shi Y, Gong J,
Shen W and Liu C: miR-29c-3p regulates DNMT3B and LATS1 methylation
to inhibit tumor progression in hepatocellular carcinoma. Cell
Death Dis. 10:482019. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Han Q, Lin X, Zhang X, Jiang G, Zhang Y,
Miao Y, Rong X, Zheng X, Han Y, Han X, et al: WWC3 regulates the
Wnt and hippo pathways via dishevelled proteins and large tumour
suppressor 1, to suppress lung cancer invasion and metastasis. J
Pathol. 242:435–447. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Fan F, He Z, Kong LL, Chen Q, Yuan Q,
Zhang S, Ye J, Liu H, Sun X, Geng J, et al: Pharmacological
targeting of kinases MST1 and MST2 augments tissue repair and
regeneration. Sci Transl Med. 8:352ra1082016. View Article : Google Scholar : PubMed/NCBI
|