Introduction
Gliomas are the most common type of primary brain
tumors in adults and represent >50% of all primary brain tumors.
In adults, glioblastoma is the most common histological type
(1). It is characterized by
aggressive growth, rapid progression of the disease and poor
prognosis. In children, high-grade gliomas represent only a
minority of primary brain tumors, while the low-grade tumors
predominate (2). Compared with
adults, pediatric tumor types are generally more sensitive to
irradiation and chemotherapy (3).
In all age groups current treatments for high-grade gliomas
(surgery, radiotherapy and chemotherapy) very rarely yield
long-term control of the disease.
Several rodent glioma models have been established
and utilized for malignant glioma research. These models generally
involve implantation of cultured human or rodent glioma cells into
the brains of nude mice or syngeneic mice or rats, respectively
(4,5). Previously, spontaneously arising
tumors or tumors arising after exposure of animals to carcinogenic
chemicals, such as N-ethylnitrosourea, were used for the
establishment of malignant glioma cell lines (6–9). More
recently, development of gene transfer techniques has provided
novel tools for creating cancer models with desired genetic
profiles. Lentivirus vectors are useful tools for stable long-term
expression of transgenes. They have been utilized for oncogene
transfer to induce malignant transformation and to study the role
of different oncogenes in malignant transformation (10,11).
Akt, H-Ras and p53 are all integral components of signaling
pathways (PI3K-Akt-mTOR and EGFR-Ras-MEK/ERK pathways) or cell
cycle regulation (p53). Due to the central roles of these genes in
glioblastoma pathogenesis they were selected for transformation in
the present study (12).
Translation of promising experimental therapies from
rodent models to clinical success has been complicated as the novel
therapies often fail in clinical trials. Rodent glioma models
generally do not allow for preclinical evaluation of the efficiency
of novel therapies in combination with surgical resection, which is
an essential part of current therapeutic protocols (13–15).
Utilizing a larger laboratory animal model, such as rabbit, enables
the testing of novel treatments and local administration techniques
in combination with surgery. For example, preclinical evaluation of
endovascular therapies require larger animal models, such as rabbit
(16). However, no rabbit glioma
cell lines exist. The aim of the present study was to investigate
the utility of lentivirus vector-mediated oncogenic transformation
for establishing rabbit glioma tumors and to create a novel rabbit
model for malignant glioma. In the present study, a malignant
glioma rabbit model was not created. However, the study produced
unexpected, but noteworthy findings deciphering the pathogenesis of
a rare benign brain tumor, ganglioglioma.
Materials and methods
Animals
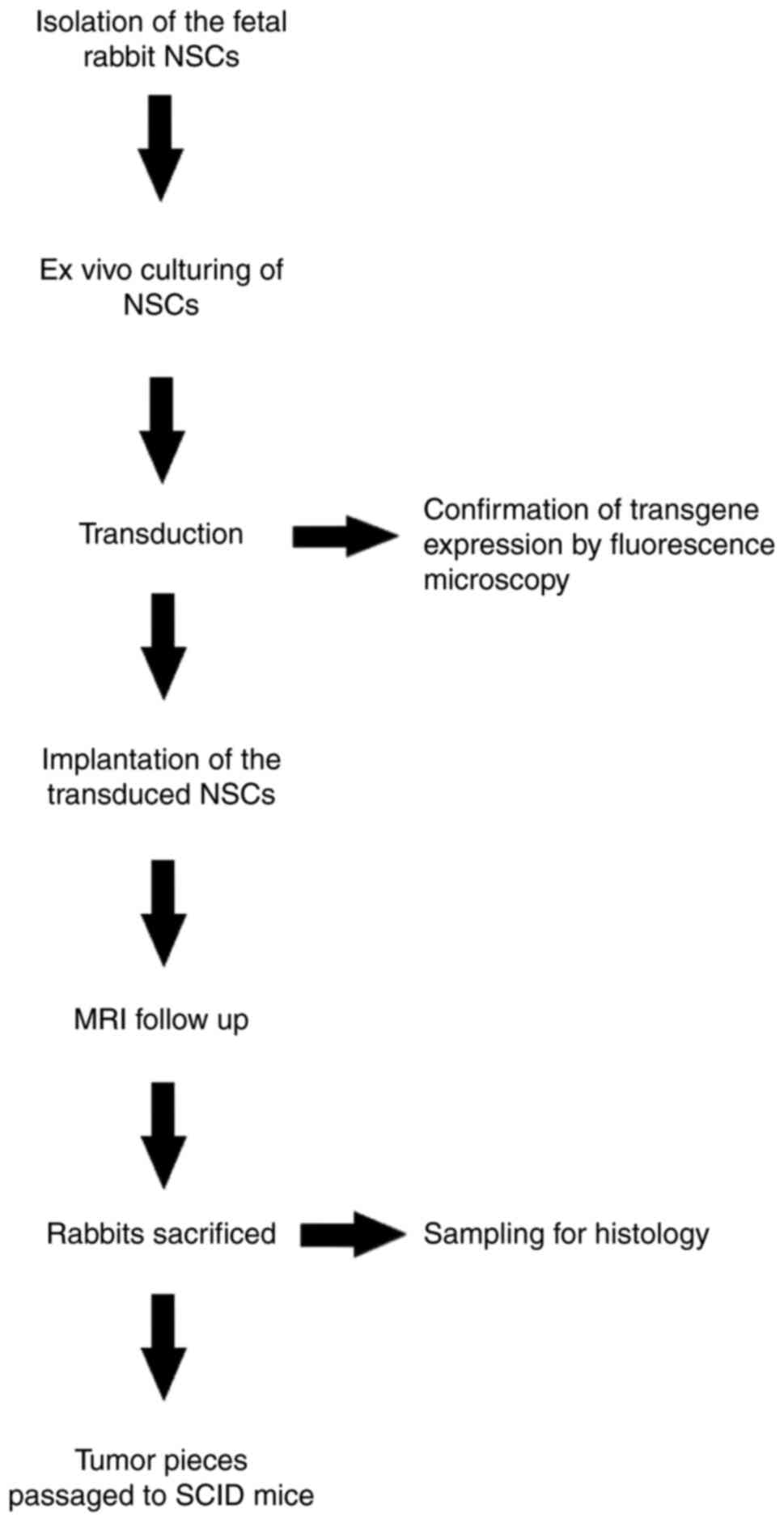
The experimental design is presented in the Fig. 1. New Zealand White (NZW, Harlan
Sprague Dawley, Inc.) rabbits were used for this study. For
implantation of the transduced neural stem cells (NSCs), female NZW
rabbits (age, 5.5 months) that weighed between 2.5–3.5 kg were
used. The study was carried out according to Finnish National and
European Union legislation and guidelines. The permission for
animal studies was acquired from the Project Authorization Board of
the Regional State Administrative Agency. All animals were
maintained at the Lab Animal Center of the University of Eastern
Finland (Kuopio, Finland). Housing temperatures for mice and
rabbits were 22° and 18°C, respectively. Moreover, animals were
housed under a normal atmosphere with 55±15% relative humidity and
12 h light-dark cycle. The animals had free access to food and
water. Animal welfare was monitored twice a day, once a day by the
research group members and once a day by the personnel of the lab
animal center. MRI imaging was carried out in the Nuclear Magnetic
Resonance Imaging Laboratory at the A. I. Virtanen Institute for
Molecular Sciences, University of Eastern Finland, with facilities
suitable for experimental animal imaging. Excel (version 2011,
Microsoft Corporation) was used to create the graph. For
anesthesia, Domitor® (medetomidine 1 mg/ml; Orion
Corporation) 0.25–0.5 mg/kg subcutaneous (s.c.) and
Ketalar® (ketamine 50 mg/ml; Pfizer, Inc.) 15–25 mg/kg
s.c. for rabbits, and 75 mg/kg ketamine and 1.0 mg/kg medetomidine
for s.c. mice. Before use, 1 ml Hypnorm (containing 0.315 mg
fentanyl/ml and 10 mg fluanisone/ml) was mixed with 1 ml Dormicum
(containing 5 mg midazolam) and 2 ml sterile water and the solution
was used for anesthesia of the mice at a dose of 0.1 ml/10 g,
intraperitoneal (0.7875 mg/kg fentanyl, 25 mg/kg fluanison and 12.5
mg/kg midazolam) (17). Duration of
the experiment was 141 days and at the end rabbits were sacrificed
by decapitation under deep overdosing terminal anesthesia. One
rabbit died under anesthesia during MRI imaging at day 141. The
exact reason for the death of this animal remains unknown since the
animal was asymptomatic before anesthesia induction. The criteria
for euthanasia included neurological deterioration or poor physical
condition. No control rabbits receiving Mock transduced NSCs were
used to avoid unnecessary use of animals and no statistical
analyses were performed. The severe combined immunodeficient (SCID)
mice (Harlan Sprague Dawley, Inc.) were sacrificed with
CO2 (gas flow 41% of the chamber volume/min) and death
was confirmed by cervical dislocation. For this experiment, two
male (age, 33 weeks; weight, 30 g) mice were used.
Cell cultures
NSCs were generated from the hippocampus and lateral
ventricle wall of the newborn rabbit pups as described by Clarke
et al (18) and Supporting
Materials and Methods from Castrén et al (19) and plated in culture medium
containing 2 mM L-glutamine, 15 mM Hepes, 100 U/ml penicillin, 100
mg/ml streptomycin, B27 supplement, 20 ng/ml epideral growth factor
(EGF; Thermo Fisher Scientific, Inc.), and 10 ng/ml fibroblast
growth factor (FGF)-2 (PeproTech EC Ltd.) in DMEM/F12 medium
(Thermo Fisher Scientific, Inc.). EGF (20 ng/ml) and FGF-2 (10
ng/ml) were added every 3rd day, and half of the medium was
replaced with new medium every 3–4 day. The cells were incubated at
+37°C. The stem cell isolation procedure was repeated twice.
Plasmid and viral vectors for AKT and H-Ras were generated as
previously described (11,20). Briefly, the pTomo H-RasV12 and pTomo
HA-AKT Cre-loxP-controlled lentiviral vectors were constructed
containing a human cytomegalovirus immediate-early promoter
(CMV)-loxP-red fluorescent protein (RFP)-loxP-Flag-H-RasV12/HA-RAS
followed by internal ribosomal entry site (IRES)-green fluorescent
protein (GFP). The stuffer fragment, RFP, keeps the translation of
Flag H-RasV12 in an ‘off’ state. The excision of the stuffer
sequence by Cre recombinase results in the expression of Flag
H-RasV12 or HA-AKT (11,20). Constitutively active AKT and H-Ras
oncogenes were introduced into NSCs using lentiviral vectors
(provided by Dr Inder Verma, Salk Institute, La Jolla, California,
USA) at MOI 3. The same lentivirus vector without a transgene
(Tomo-MOCK) was used for transduction of the negative control
cells. Additionally, NSCs were also transfected with lentivirus
vectors carrying mouse small interfering (si)RNA p53
(5′-CTGTCTAGACAAAAAACAAGTACATGTGTAATAGCTCCTCGATGTCTCTTGAACATGCAGGAGCTATTACACATGTACTTGTGGGATCTGTGGGTCTCATACA-3′)
constructed using CMV–IE promoter in pLV1-hPGK-GFP-WPRE plasmid
(provided by Dr Inder Verma, Salk Institute) (21) at MOI 30. Adeno-Cre was used for the
activation of those vectors at MOI 30 (22). For the transductions, the
neurospheres were trypsinized to yield a single cell suspension to
improve the exposure of each individual cell to the transducing
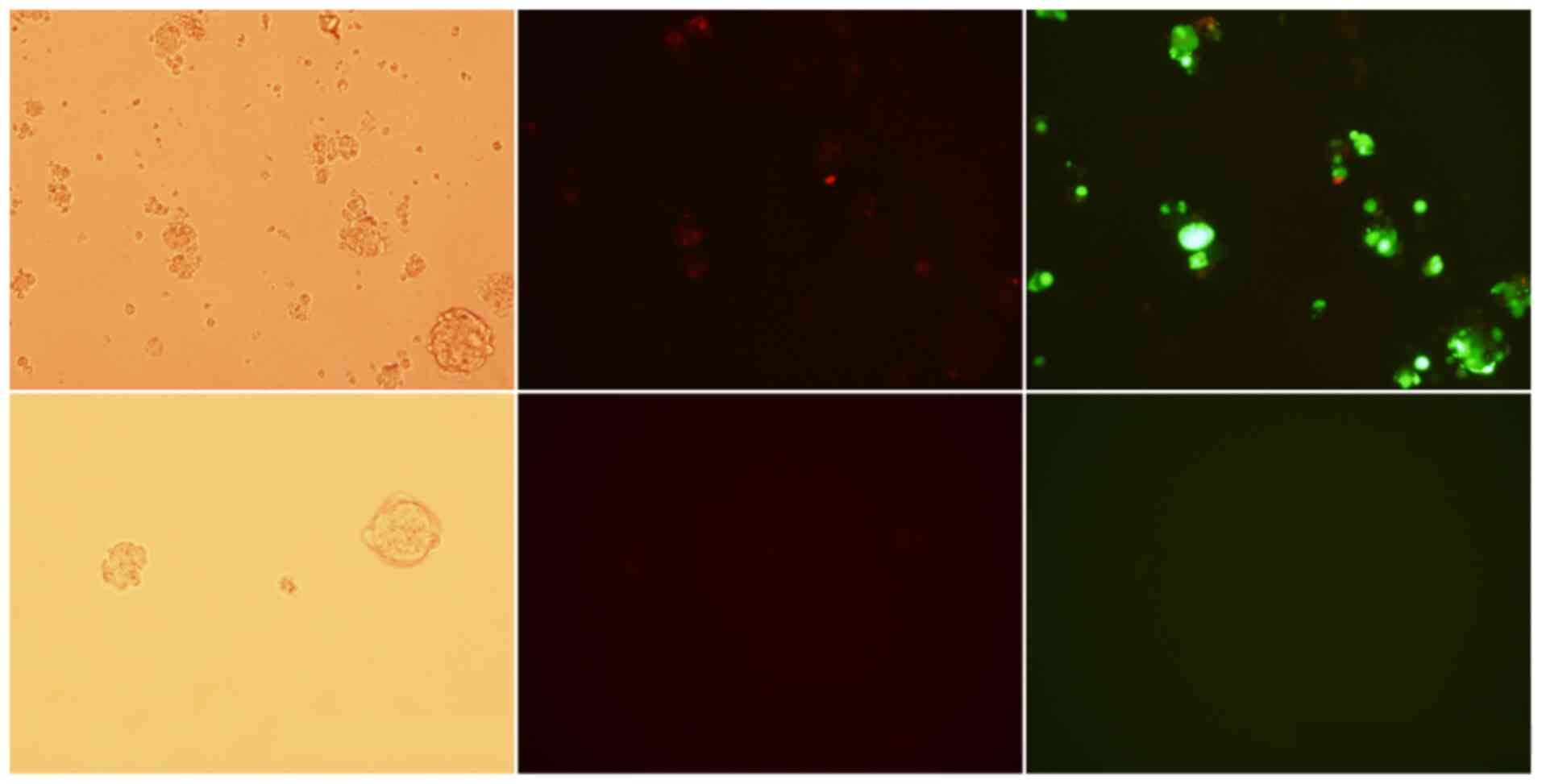
vectors. For demonstration of the transgene expression in
vitro, the cells were cultured for 7 days in the EGF- and
FGF-containing medium to allow the transgene expression to start
before fluorescence microscopy. However, for subcortical in
vivo implantation, the NSCs were used 1 day after transduction.
All transductions were carried out at the same time and cultured
overnight at 37°C before being implanted into animals the next day
(Fig. 2). The proportions of
fluorescent marker gene expressing cells were detected with bright
field and fluorescence microscopy with ×10 magnification after the
transductions.
Stem cell implantation
A total of 10 adult female rabbits were anesthetized
by s.c. injections of 0.25–0.5 mg/kg ketamine and 15–25 mg/kg
medetomidine. Anesthesia was maintained with 0.02–0.05 mg
intravenous boluses of ketamine and medetomidine, respectively, via
cannulated marginal ear vein (23).
After shaving and cleaning the heads of the rabbits, a 3-cm
incision was made along the midline and the periosteum was opened,
before a small 3–4 mm craniotomy was made using a dentist drill. A
total of 3.0×104 viable cells suspended in 15 µl
Opti-MEM (Invitrogen; Thermo Fisher Scientific, Inc.) were then
injected via a stereotactic injection at 5 mm caudally and 6 mm
right from the sagittal suture into 2 mm depth from the dura
surface. For the stereotactic injections, a stereotactic frame with
a custom-made rabbit head holder and a Hamilton syringe were used.
After the injection was made, the needle was slowly drawn out to
prevent backflow of the injected cell suspension. The scalp was
then sutured and all rabbits received 4 mg/kg Rimadyl®
(carprofen; Pfizer, Inc.) s.c. For infection prophylaxis, all
rabbits received 125 mg kefuroxime (GlaxoSmithKline) s.c. during
the operation and on the first postoperative day.
Magnetic resonance imaging (MRI)
MRI scanning was performed every week or 2 weeks to
monitor tumor growth. Anesthesia for MRI scanning was carried out
as aforementioned. MRI data was acquired using a horizontal 4.7 T
magnet (Magnex Scientific; Varian, Inc.) interfaced to a Varian
UNITY INOVA™ console (Varian Medical Systems, Inc.) using an in-lab
built surface RF coil in transmit/receive mode with loop diameter
2.5 cm. T2-weighted images were measured using a spin echo sequence
(echo time, 40 msec; repetition time, 2 sec). A total of 11 slices,
each of 1.5-mm thickness, were imaged. The matrix size was 128×128,
with field of view 50 mm2 yielding an in-plane
resolution of 390 µm. Total tumor volume was processed and analyzed
using MATLAB® version 7.04 software (The MathWorks,
Inc.).
Histology
Immediately after euthanasia, the animals were
perfused with 1% paraformaldehyde pH 7.4. Brains were
immersion-fixed in 4% paraformaldehyde/0.15 M sodium phosphate
buffer (pH 7.4) for 48 h at 4°C. After fixation, the samples were
dehydrated, embedded into paraffin, sectioned into 4 µm sections
and stained for light microscopy (×4, ×10, ×20 and ×40
magnifications) with standard hematoxylin and eosin (H&E)
staining, including staining with hematoxylin (Hematoxylin solution
according to Delafield; Sigma-Aldrich; Merck KGaA; cat. no. 03971;
250 ml) for 10 min and eosin (Eosin Y solution alcoholic;
Sigma-Aldrich; Merck KGaA; cat. no. HT110132-1 L) for 30 sec, at
room temperature.
Before the addition of the primary antibody, the
sections were treated with normal serum (Vector Laboratories, Inc.)
diluted 1:20 in PBS. After exposure to the primary antibody
overnight at 4°C the sections were washed with PBS twice for 2 min
each time and then exposed to the secondary antibody (Vectastain
IgG horse anti-mouse antibody, Vector Laboratories, Inc.; 1:200)
for 30 min at room temperature. The staining was carried out with
Vectastain ABC Method based on avidin-biotin-HRP reaction (Vector
Laboratories, Inc.) with DAB substrate. For counterstaining,
Harris-hematoxyline was used. After counterstaining, the sections
were dehydrated with EtOH dilutions with rising concentrations (50,
70, 95, 100 and 100%; 3 min each), followed by incubation with
xylene two times for 5 min at room temperature.
Primary antibodies used for immunostainings included
mouse-anti-HA tag monoclonal antibody to detect H-Ras (1:50;
monoclonal HA11; clone no. 16B12, Nordic BioSite), anti-flag M2
monoclonal antibody to detect AKT (1:2,000; cat. no. F1804,
Sigma-Aldrich; Merck KGaA), CD31 mouse anti-human monoclonal
antibody (1:50; Dako; Agilent Technologies, Inc.; cat. no. M0823),
Ki-67 mouse anti-human monoclonal antibody (1:100; Dako; Agilent
Technologies, Inc.; cat. no. M7240), GFAP mouse monoclonal antibody
(1:250; Abcam; cat. no. ab4648) and p53 mouse monoclonal antibody
(1:100; Santa Cruz Biotechnology, Inc.; cat. no. SC-55476).
Antibody diluent with background (Dako; Agilent Technologies, Inc.;
cat. no. S3022) was used for dilution of the primary antibodies and
for negative control staining. Incubation with primary antibodies
was carried out overnight at 4°C. VECTASTAIN® Elite ABC
Kit (Vector Laboratories, Inc.) was then used for detection.
Double staining for detecting H-Ras and AKT
localization/co-localization was performed using mouse-anti-HA tag
and anti-FLAG® M2 monoclonal antibodies. Briefly, for
the first sequence, slides were incubated with primary antibody
anti-flag tag overnight at 4°C, biotinylated secondary antibody
(Vectastain IgG horse anti-mouse antibody; Vector Laboratories,
Inc.; cat. no. BA-2000-1.5) 1:200 for 30 min at room temperature,
Avidin-Biotin-HRP (Vector Laboratories, Inc.) for 30 min and
Peroxidase Substrate Solution (DAB; Invitrogen; Thermo Fisher
Scientific, Inc.) for 1–5 min under microscopic observation. For
the second staining, slides were incubated with mouse-anti-HA tag
overnight at 4°C, biotinylated secondary antibody (horse
anti-mouse) for 30 min, Avidin-Biotin-AP (Vector Laboratories,
Inc.) for 30 min and Alkaline Phosphatase Solution (Vector Blue)
(Vector Laboratories, Inc.) for 5–30 min under microscopic
observation. No nuclear counterstain was used in order to avoid
difficulties in interpreting three-color tissue preparations. After
staining, sections were air dried and mounted in aqueous medium
(VWR International, LLC). Single-positive cells were stained brown
for AKT, blue for H-Ras and dark blue-brown for colocalization of
both.
Periodic acid-Schiff (PAS) and CD31 dual staining
was performed to validate the existence of vasculogenic mimicry or
vascular co-option in the tumor tissue. Slides were initially
stained with general protocols for CD31 mouse anti-human monoclonal
antibody. Following incubation with DAB, slides were immersed in
PAS reagents for 5 min, rinsed in distilled water three times for 1
min each, immersed in Schiff's reagents for 15 min at room
temperature, washed under running tap water for 5 min, and finally
counterstained with hematoxylin solution for 90 sec at room
temperature. Slides were dehydrated under a series of alcohol
concentrations and xylene, and were then mounted with Permount™
Mounting Medium. PAS reagents were purchased from Sigma-Aldrich
(Merck KGaA).
Re-passaging of the tumor s.c. into
SCID mice
To determine the reproducibility of the tumor, a
small piece of the tumor was excised via an operation after day
141, cut into several pieces and s.c. implanted into
immunodeficient SCID mice. SCID mice were anesthetized by s.c.
injections of 75 mg/kg ketamine and 1.0 mg/kg medetomidine. After
cleaning, a small incision was made on the back of the mice using a
surgical scalpel. The tumor pieces were then inserted under the
skin, one tumor piece/mouse, further from the midline incision. The
incision was then sutured and the mice received 4 mg/kg carprofen
s.c. For infection prophylaxis, the mice received 125 mg kefuroxime
s.c. during operation and on the first postoperative day. For
histological analysis (see above), tumor growth was monitored until
the proper size was reached, and then samples were taken 107 days
after tumor implantation.
Results
Transductions of the NSCs
Fluorescence microscopy showed EGFP expression in
38% and co-expression of EGFP and RFP in ~6% of the transduced
NCSs. Expression of RFP without simultaneous EGFP expression was
not detected.
MRI and tumor growth
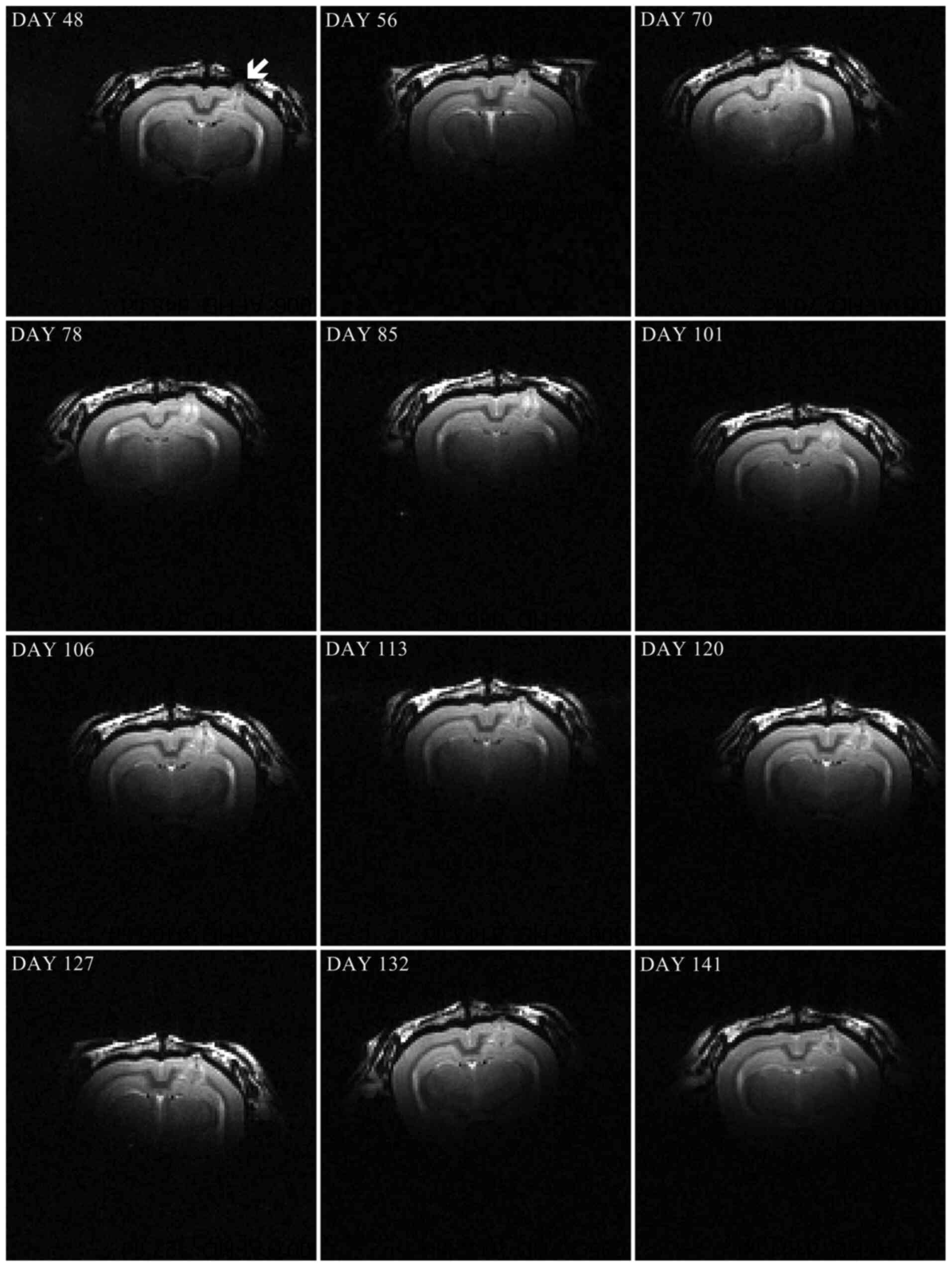
MRI data showed tumor growth after 48 days of NSC
implantation (Fig. 3) in one of the
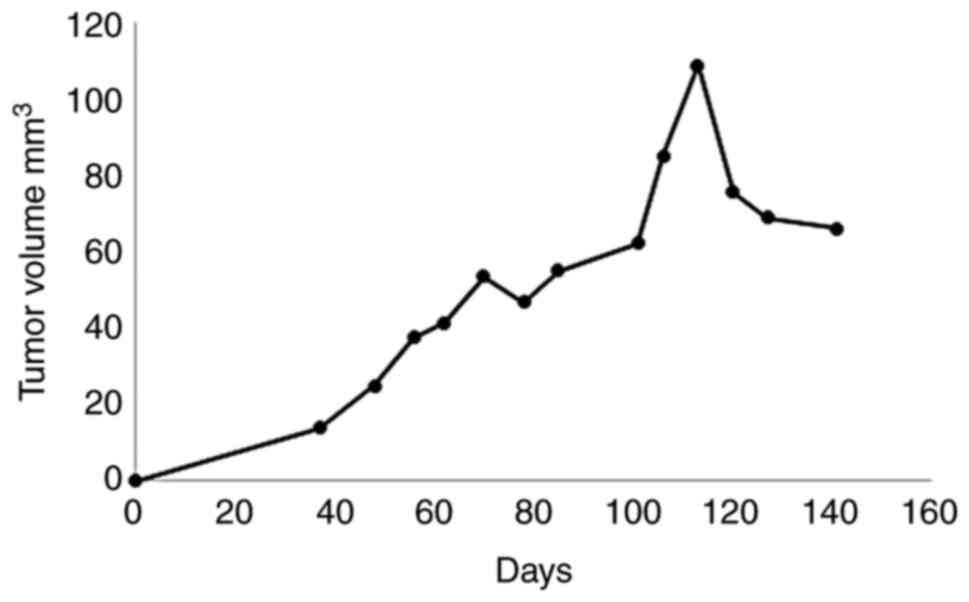
ten rabbits (10%). MRI follow-up showed slow growth of the tumor up
to day 141, when the animal was sacrificed (Fig. 4). Notably, the highest tumor volume
was measured at day 113, followed by some reduction at the later
time points.
Tumor histology and
immunostainings
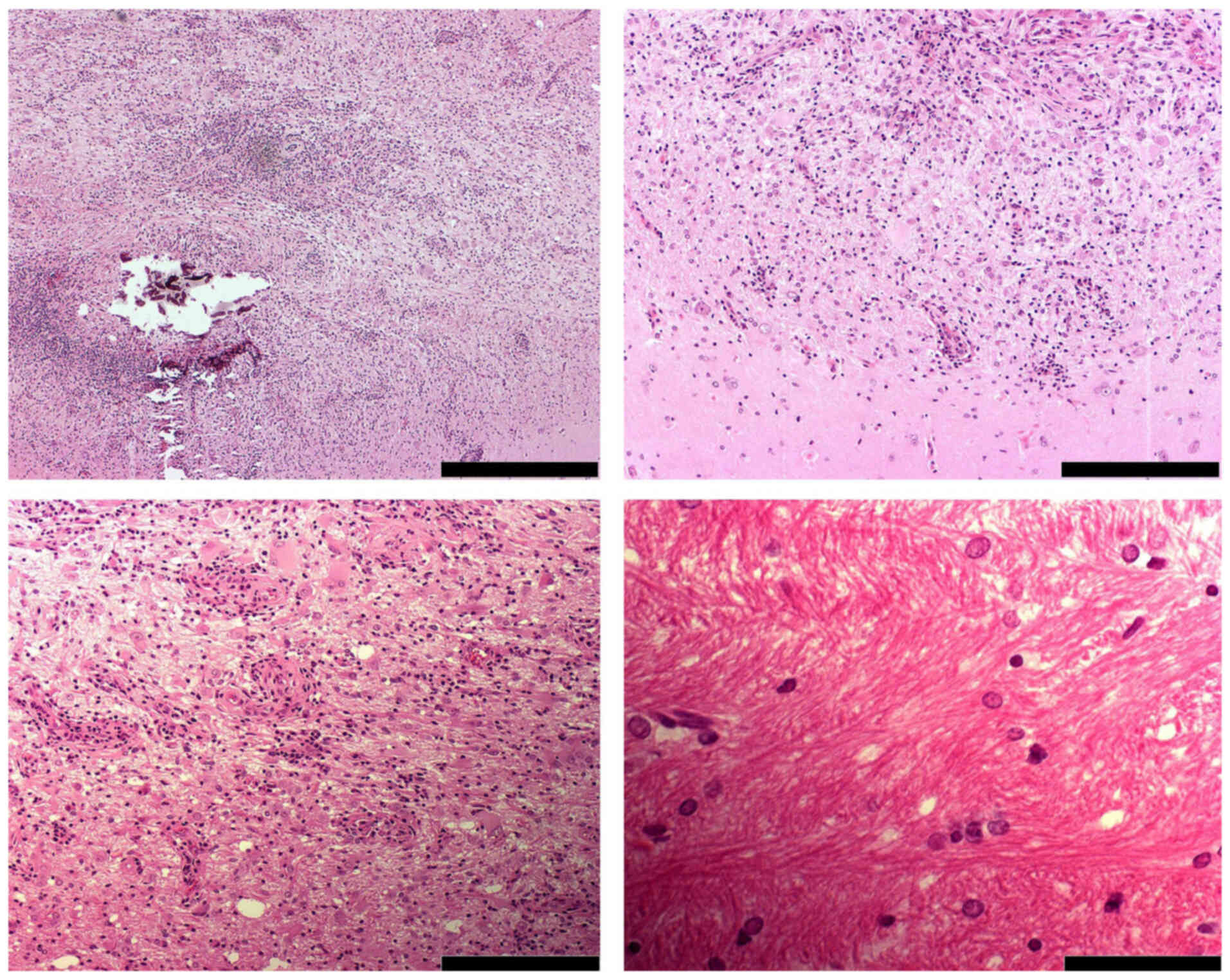
Histological analysis showed that the tumor was
similar to a benign low grade ganglioglioma, according to World
Health Organization classification of tumors of the central nervous
system (24,25). H&E staining revealed that the
tumor recapitulates the emergence of ganglion and glial cells,
which are generally characterized as large, rounded with nuclear
atypia and multinucleated cells. The presence of microcalcification
within the tumor area also marks one of the important features of
gangliogliomas (Fig. 5). Similar
calcification has been seen in a number of human ganglioglioma
cases in cystic lesions (26). The
tumor borders were ill-defined, with tumor cells scattered within
the surrounding brain tissue, and peritumoral edema was detected.
Additionally, within the tumor area, a network-like pattern was
observed.
The constitutively active AKT and H-Ras transgenes
contain Flag M2 and HA tags, respectively, which act as markers
that allow the recognition of proteins. Staining results
demonstrated the expression of both proteins in the tumor cells
(Fig. 6A and B). In addition,
immunostaining of a glial marker in the resultant tumor showed the
strong expression of GFAP (Fig.
6C), which is seen in the majority of ganglioglioma subtypes
(26,27). p53 protein expression was low in the
tumor area. However, we were not able to conclusively show that the
activity of the tumor suppressor gene may have been blocked by the
introduction of the p53 siRNA (Fig.
6D) since the control healthy brain also showed a low
expression of the protein. A dense network of small branching
capillaries and highly proliferative tumor cells were identified in
immunohistochemistry with CD31 and Ki-67 markers, respectively
(Fig. 6E and F). Proliferation
figures were found in this tumor more frequently compared with a
typical grade 1 ganglioglioma but not as frequently as in
anaplastic gangliogliomas (28).
Furthermore, the slow growth of the tumor during the follow up did
not suggest malignant transformation to anaplastic grade 3
ganglioglioma. Therefore, the tumor was defined as a benign grade 2
ganglioglioma. Usually <5% of tumor cells express Ki-67 in Grade
I gangliogliomas (29).
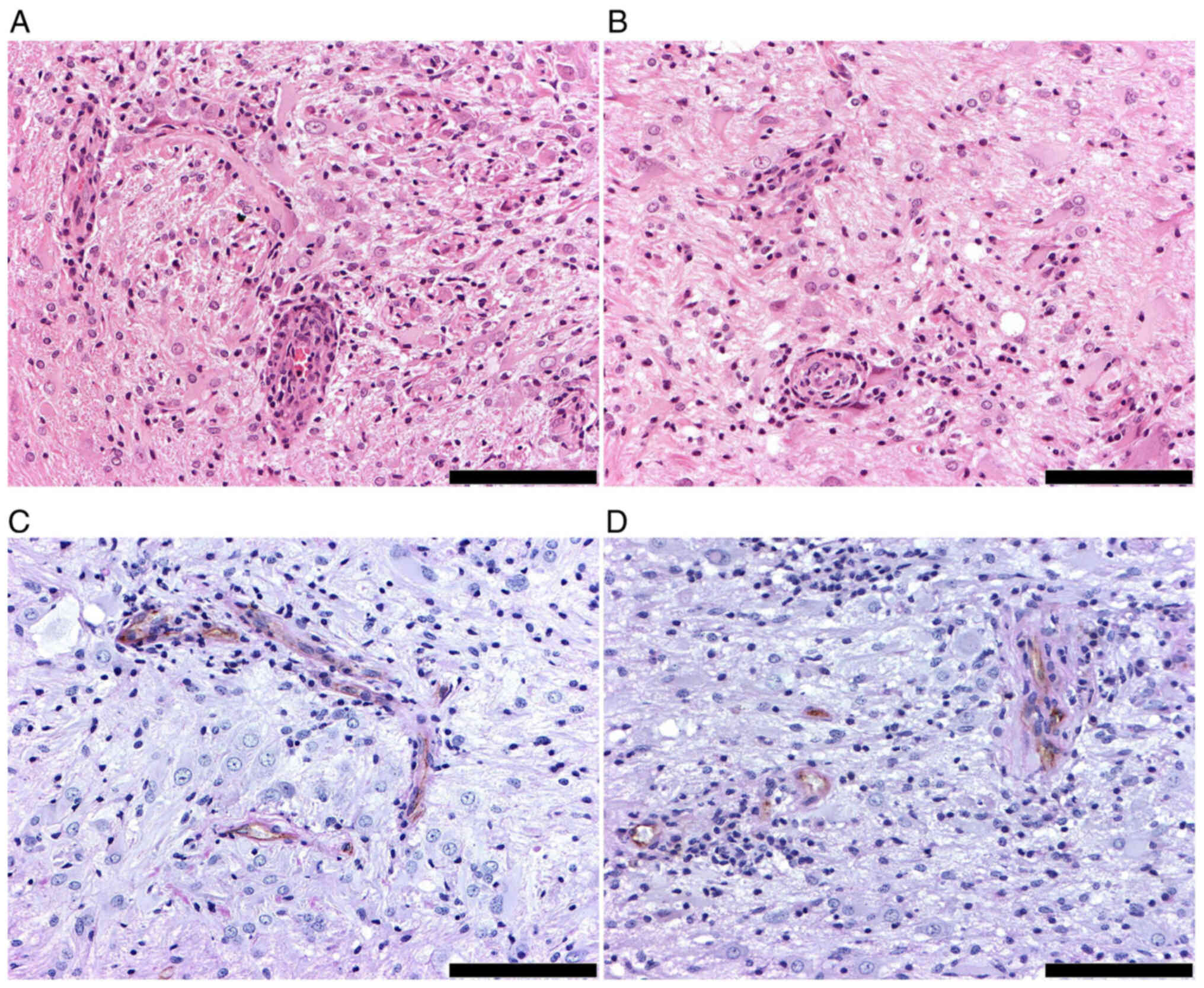
 | Figure 6.Immunohistochemistry staining. Both
(A) AKT and (B) H-Ras transgenes were expressed in the tumor cells
(magnification, ×20; scale bar, 100 µm). (C) The tumor was
characterized by strong GFAP expression (magnification, ×10; scale
bar, 200 µm). However, (D) low expression of p53 protein was
detected (magnification, ×20; scale bar, 100 µm). Actively
proliferating tumor cells were observed using (E) Ki-67
immunostaining (brown) and hematoxylin counterstain (blue)
(magnification, ×20; scale bar, 100 µm), and (F) a dense network of
small branching capillaries with CD31 (magnification, ×10; scale
bar, 200 µm). (G and H) Double staining for detecting H-Ras (blue)
and AKT (brown) localization/co-localization showed that the tumor
was composed of heterogeneous cell populations, which expressed
either H-Ras or AKT or both in the same cells (magnification, ×40;
scale bar, 50 µm). |
When investigating the co-localization of AKT and
RAS proteins, double staining showed that the tumor composed of
heterogeneous cell populations expressed either AKT or H-Ras or
both in the same cells (Fig. 6G and
H).
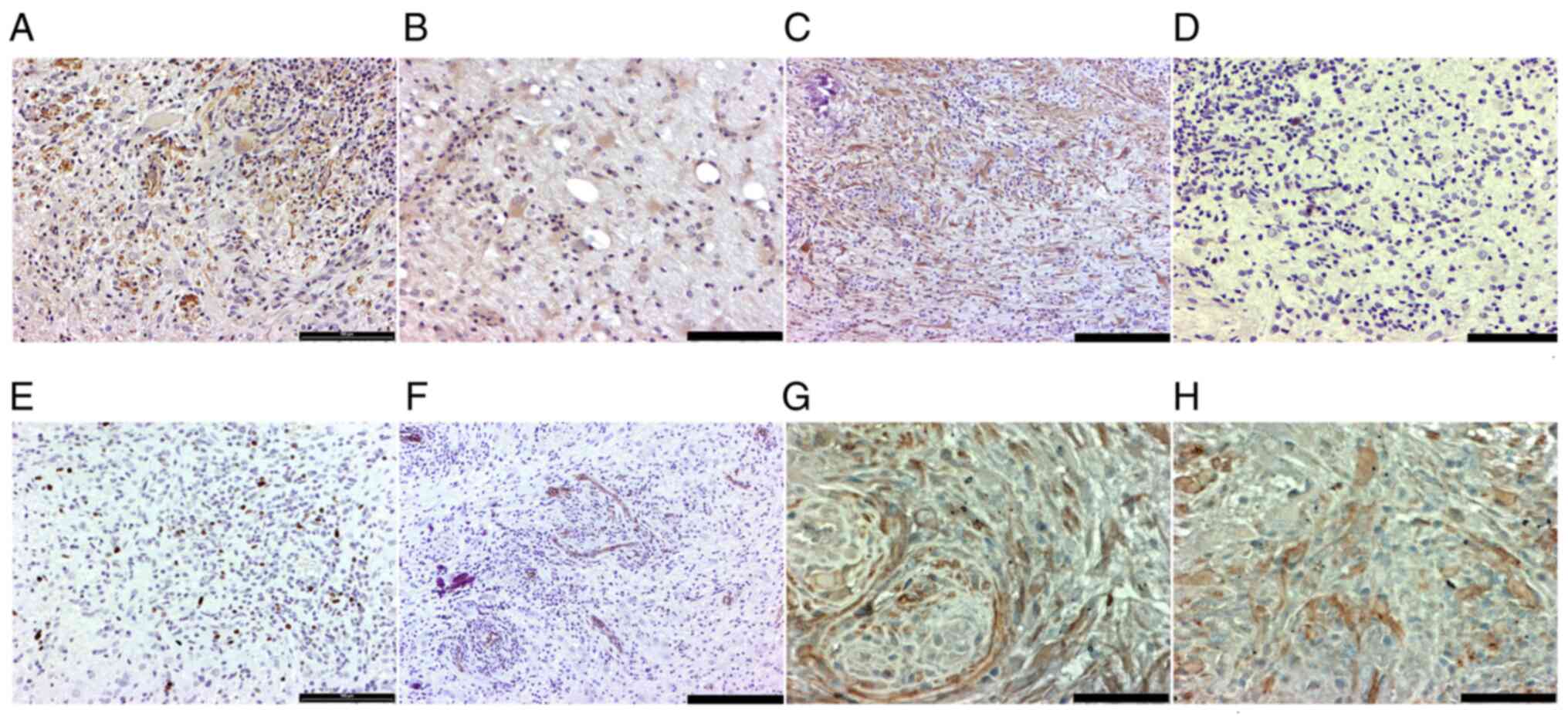
H&E staining showed the presence of tumor cells
around the vessels within the tumor area (Fig. 7A and B). Of note, PAS-CD31 dual
staining demonstrated the positive expression of PAS lining nearly
all CD31-positive channels (Fig. 7C and
D). CD31-positive staining was found on the luminal surface of
the vessels, whereas a PAS-positive pattern was found on the vessel
wall of the intratumoral vessels, which suggested that vascular
co-option was primarily in the peritumoral area (30,31).
Likewise, vascular co-option occurrence in human astrocytoma has
also been reported previously (32). No vasculogenic mimicry was
observed.
Re-passaging of the tumor
subcutaneously into SCID mice
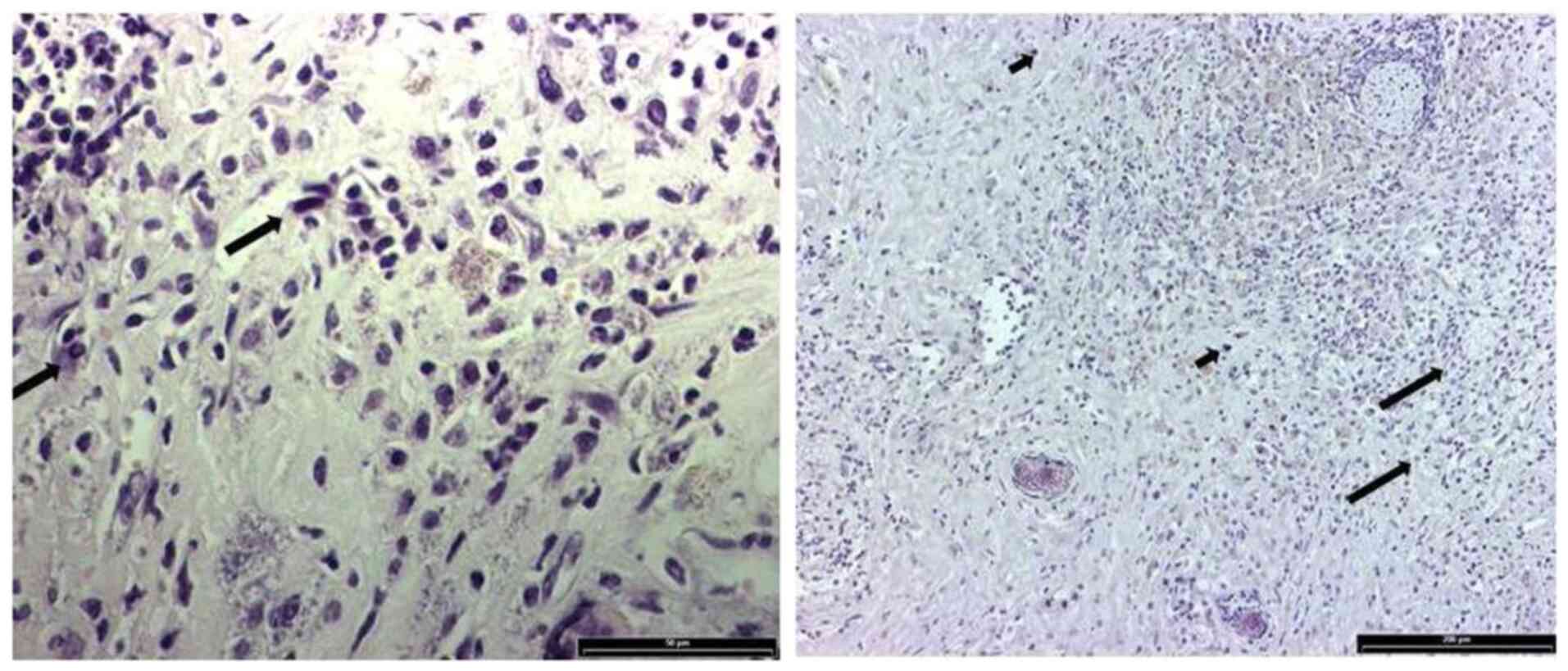
H&E staining demonstrated the presence of a
mixed cell population of reactive ganglion and glial cells in the
tumor tissue. Some oligodendroglia cells were also detected, which
were characterized by pale nuclei and clear perinuclear area due to
inflammation. Additionally, reactive astrocytes showed dark nuclei
and atypia (Fig. 8).
Discussion
In a previous study by Marumoto et al
(11) AKT and H-Ras oncogenes were
transferred using Cre-LoxP controlled lentivirus vectors into NSCs
containing brain regions (subventricular zone or hippocampus) of
GFAP-Cre transgenic mice. These mice expressed Cre recombinase in
GFAP expressing central nervous system cells, such as mature
astrocytes and multipotent NSCs. Gene transfer resulted in the
formation of glioblastoma multiforme tumors with frequencies of 10
or 30% after vector injections into the subventricular zone or
hippocampus, respectively. The tumorigenicity was notably enhanced
when the mice were also heterozygous for the p53 tumor suppressor
gene. In p53 heterozygous mice, vector injections into the
subventricular zone and hippocampus yielded glioblastoma formation
in 80 and 100% of the mice, respectively. However, transfer of AKT
and H-Ras oncogenes into the cerebral cortex of p53 heterozygote
mice resulted in tumor formation only in 1 out of 15 mice. Most of
the histological characteristics of the glioblastoma-like tumors
found in the hippocampus or subventricular zone were also present
in this cortical tumor (11).
A previous study has reported that NSCs can be
isolated and cultured ex vivo followed by gene transfer and
implantation in vivo (33).
In the present study, newborn NZW rabbit stem cells were isolated
and transduced ex vivo with Cre-LoxP controlled lentivirus
vectors encoding AKT and H-Ras oncogenes, adenovirus vector
carrying Cre recombinase gene and lentivirus vector with p53 siRNA
expression cassette. The transduced NSCs were then implanted into
the cerebral cortex of NZW rabbits, which is a location that allows
surgical resection of tumors (34).
Still, with this experimental setting the tumorigenicity was low in
the present study. According to histological analysis the tumor
represented low grade ganglioglioma.
Ganglioglioma is mostly found in patients younger
than 30 years (30,31). In pediatric patients, gangliogliomas
represent ~10% of all primary brain tumors (27). Gangliogliomas are characterized by
biphasic histological architecture involving transformed neuronal
and glial components (35).
Clonality studies suggest that the two different tumor cell
populations originate from a single neuroglial precursor cell
(36). Gangliogliomas are typically
well-differentiated and characterized by slow growth (37,38).
Occasionally malignant transformation of the glial component can
occur (37,39,40).
It is noteworthy that the use of AKT, H-Ras and p53 siRNA
transduced newborn rabbit NSCs for tumor induction resulted in
ganglioglioma formation, whereas in the previous study
glioblastoma-like tumors arose after injection of the H-Ras and AKT
encoding vectors into NSC containing areas in adult p53
heterozygote mouse brains (11).
Several potential reasons for the different
pathology of the arising tumor exist. Age of both donor
(age-dependent properties of the cells) and recipient animals
(growth factors and the tumor microenvironment) may influence
tumorigenic properties of NSCs during the malignant transformation
process, as well as in resulting histological tumor types. Marumoto
et al (11) injected the
vectors into the NSC-containing areas of the adult mice, whereas
the present study used ex vivo transduction of precultured
newborn rabbit NSCs. In newborn mammals, the development of the
brains is still ongoing and the NSCs located in the hippocampus and
subventricular zone play a key role in the process. The NSC
population gives rise to radial glial cells (RGCs), which are
progenitors for neural and glia cell differentiation that migrate
to different areas of the cortical brain and form the neurons and
glia cells, including astrocytes and oligodendroglia. Analysis of
326 gangliogliomas by Blümcke and Wiestler (35) revealed that ~80% of the
gangliogliomas expressed CD34-positive staining in immunostaining
experiments. CD34 is a stem cell epitope that is not expressed in
the normal brain. The majority of these tumors occur in the
temporal lobe, which is also the site of the hippocampus, a major
NSC-containing region in the brain. Blümcke and Wiestler (35) pointed out that their results
supported the idea that ganglioglioma arises from a glioneuronal
precursor lesion.
In the present study, the newborn rabbit NSCs were
cultured in vitro as neurospheres floating in medium
containing EGF and FGF, which have shown to be crucial for
maintenance of the NCS phenotype. In absence of these growth
factors the NCSs start to differentiate via precursor cells to
neurons and glial cells. Without EGF and FGF exposure the cells
start to lose their ability to grow as neurospheres (typical
feature of the NSCs) and attach to the cell culture plate
relatively quickly, within 24 h. The subcortical area is not an
anatomical site containing a NSC niche as are the hippocampus or
subventricular zone. Instead, the subcortical microenvironment is
likely to lack the growth factors and extracellular signals to
maintain the NSC phenotype of the implanted cells and rather
support the differentiation to neurons and glia cells, likewise the
RGCs migrating from the NSC-containing areas of the animal's brain.
The present data supported the hypothesis that ganglioglioma arises
from a single transformed RGC or NSC committed to differentiate to
RGC that then divides and differentiates further, giving rise to
two different tumor cell populations, neuronal and glial components
of the ganglioglioma.
AKT and H-Ras transgene expression of the tumor was
verified with immunostaining. Little is known about the molecular
pathogenesis of ganglioglioma, largely because this tumor type is
relatively rare. However, there is previous evidence that the
PI3K-mTOR signaling pathway, including AKT, is activated in
ganglioglioma (41). The present
data also supported a previously presented hypothesis, which states
that the Ras pathway can play a role in the molecular pathogenesis
of ganglioglioma (42).
However, the present study has several limitations.
When planning the experiment, it was speculated that the ex
vivo culturing of the NSCs may be a process that could
potentially change the properties of the cultured cells, especially
if the culture is prolonged and continued for multiple passages.
Therefore, fluorescence microscopy was selected to count the
enhanced (E) GFP or RFP expressing and co-expressing cells for the
verification of transgene expression (Fig. 1) After activation with the
cre-recombinase, the vectors did not express RFP, but only EGFP in
addition to Akt or H-Ras transgene (11). To yield a sufficient number of
cultured NSCs for both in vivo implantation and simultaneous
flow cytometry analysis would have required significantly longer
ex vivo passaging. For the same reason, western blot
analysis for confirmation of the p53 knockdown was not carried out
with the ex vivo cultured and transduced rabbit NSCs of the
same batch that was used for the in vivo experiment.
Therefore, one of the major limitations of this study was that we
were unable to indisputably show p53 expression suppression by p53
siRNA. The immunostaining experiment showed very low p53 expression
within the tumor area; however, the finding was similar in the
surrounding normal brain tissue. In a previous study by Marumoto
et al (11) with AKT and
H-Ras transgene-induced tumors, the proportion of Ki-67-positive
tumor cells increased from ~1 to 5.5% when p53 heterozygous mice
were used instead of mice who expressed p53 normally. In the
present study, a high number of Ki-67 expressing cells were
detected, which indicated tumor cell proliferation and the reduced
activity or inactivation of p53. Still, the MRI follow-up indicated
a reduction in tumor size post-day 120. The reason for this remains
to be elucidated. However, gradual weakening of siRNA-mediated p53
blockade during the relatively long follow-up cannot be excluded.
Due to these limitations of the experimental design, future
studies, especially on the role of p53 in the pathogenesis of
ganglioglioma, are warranted. Since the aim of the present study
was to develop a new rabbit model for malignant glioma, the study
was not repeated to avoid using animals for such experiments that
were unlikely to serve this purpose. The lack of data supporting
the reproducibility of these findings also remains a major
limitation of the present study.
A few case reports with molecular genetic analysis
indicate that in rare occasions malignant transformation of
ganglioglioma to high grade glioma can occur. These reports suggest
the role of p53 inactivation in this process (39,43).
In the present study, the rabbit ganglioglioma tumor showed a
relatively high number of Ki-67-positive cells, which suggested the
malignant transformation to anaplastic ganglioglioma.
Previous results demonstrated that injection of AKT
and H-Ras encoding vectors into the mouse cerebral cortex resulted
in tumor formation, but tumorigenicity was low (11). Lower tumorigenicity and different
histological characteristics of the resulting tumor compared with a
previous study in mice (11) could
be the result of several factors. It seems likely that this could
be related to the absence or low number of NSCs in the cerebral
cortex compared with the hippocampus or subventricular zone, the
known areas that contain NSCs in the brain. Instead of direct
vector injection, ex vivo transduced NSCs (isolated from
newborn NZW rabbit hippocampus and subventricular zone) were
injected into the cerebral cortex of NZW rabbits into a location
favorable for tumor resection surgery. The tumorigenicity remained
low despite the use of NSCs cultured ex vivo. This may be
related to injection site-dependent factors, such as growth
factors, which are absent in the cortical brain tissue compared
with the hippocampus. Microenvironment and growth factors may be
important in the early phase of tumor formation. The properties of
NSCs may have changed, perhaps due to the differentiation to RGCs
before the start of the transgene expression. Different cell types
may present variation in the sensitivity to oncogenic stress, such
as lentivirus vector-driven overexpression of AKT and H-Ras genes.
Both AKT/mTOR and FGF/Ras/MAPK/ETV signaling pathways are known to
play roles in the natural process of RGC differentiation from NSCs
(44), and this may explain the
relative resistance of NSCs for the tumorigenic effects of AKT and
H-Ras transgenes in the present study. When the cell is committed
to a differentiation process involving activation of these
signaling pathways, it may have turned on its inherent protective
mechanisms for the adverse oncogenic effects of activation of these
particular signaling pathways. There is evidence that cells have
inherent mechanisms to protect themselves against the oncogenic
effects of the signaling pathways needed for their normal actions
in tissue regeneration (45–47).
In a previous study by Marumoto et al (11), the NCSs were transduced with
oncogenes in their natural environment, the stem cell niche located
in the hippocampus or subventricular zone, without simultaneously
taking them to an environment less favorable for maintaining the
stemness of the cells. We hypothesize that the already ongoing
process of differentiation to RGCs may protect the NSCs from
developing into glioblastoma multiforme, and instead lead to the
formation of a far more benign ganglioglioma or in most cases
completely prevent tumor formation.
The animal studies were conducted between June 2008
and August 2010. At that time, we were unable to understand the
findings, i.e. the emergence of a ganglioglioma tumor, instead of
the expected glioblastoma. However, the recently published findings
on the molecular mechanisms of the NSC differentiation processes
have now enabled us to interpret the results of the present study
(41,42,44).
In conclusion, lentiviral vectors provide a tool for
the manipulation of oncogenic molecular pathways to induce tumor
growth in the rabbit brain. Thereby, in addition to their benefits
for the development of rodent cancer models and investigating the
roles of different oncogenes, they could be useful for establishing
larger animal models, such as resectable malignant glioma in
rabbits. Moreover, it is hoped that these findings on ganglioglioma
formation will stimulate further research on NSC differentiation,
interferences in the differentiation process and pathogenesis of
ganglioglioma, a rare tumor type associated with drug-resistant
focal epilepsy.
Acknowledgements
The authors would like to thank Dr Inder Verma (Salk
Institute, USA) for providing the lentiviral vectors.
Funding
This study was supported by grants from Finnish
Academy (grant no. 124222) and Sigrid Juselius Foundation.
Availability of data and materials
All data generated or analyzed during this study are
included in this published article.
Authors' contributions
FA was involved in acquisition of the data,
especially the in vitro and animal studies, MRI and
histologic data, as well as analysis and interpretation of the
data. AH was involved in study design, acquisition of the data,
especially the in vitro and animal studies, MRI and
histologic data, and analysis and interpretation of the data. AP
was involved in acquisition of the data, especially the in
vitro and animal studies and MRI data, and analysis and
interpretation of the data. VO was involved in acquisition of the
data, especially the animal studies and histology, as well as the
analysis and interpretation of the data. JR was involved in the
study design and interpretation of the histology of the tumor. AI
was involved in the study design, development of the operative
technique and instrumentation suitable for rabbits together with
AH, and the interpretation of the data. JN was involved in study
design, design of the MRI sequences and instructions on the MRI
imaging, and interpretation of the data. PT was involved in study
design, design of the MRI sequences and instructions on the MRI
imaging, and interpretation of the data. TL was involved in study
design, design of the MRI sequences and instructions on the MRI
imaging, and interpretation of the data. VK was involved in study
design, stem cell isolations together with AH, FA and AP, and
interpretation of the data. JK was involved in study design and
interpretation of the data. SYH was involved in study design and
interpretation of the data. FA, AH and VO confirm the authenticity
of the raw data. All authors read and approved the final
manuscript.
Ethics approval and consent to
participate
The study was carried out according to Finnish
National and European Union legislation and guidelines. The
permission for animal studies was acquired from the Project
Authorization Board of the Regional State Administrative
Agency.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Lönn S, Klaeboe L, Hall P, Mathiesen T,
Auvinen A, Christensen HC, Johansen C, Salminen T, Tynes T and
Feychting M: Incidence trends of adult primary intracerebral tumors
in four Nordic countries. Int J Cancer. 108:450–455. 2004.
View Article : Google Scholar
|
|
2
|
Houben MP, Aben KK, Teepen JL,
Schouten-Van Meeteren AY, Tijssen CC, Van Duijn CM and Coebergh JW:
Stable incidence of childhood and adult glioma in the Netherlands,
1989–2003. Acta Oncol. 45:272–279. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Merchant TE, Pollack IF and Loeffler JS:
Brain tumors across the age spectrum: Biology, therapy, and late
effects. Semin Radiat Oncol. 20:58–66. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Candolfi M, Curtin JF, Nichols WS,
Muhammad AG, King GD, Pluhar GE, McNiel EA, Ohlfest JR, Freese AB,
Moore PF, et al: Intracranial glioblastoma models in preclinical
neuro-oncology: Neuropathological characterization and tumor
progression. J Neurooncol. 85:133–148. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Huse JT and Holland EC: Genetically
engineered mouse models of brain cancer and the promise of
preclinical testing. Brain Pathol. 19:132–143. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Laerum OD and Rajewsky MF: Neoplastic
transformation of fetal rat brain cells in culture after exposure
to ethylnitrosourea in vivo. J Natl Cancer Inst. 55:1177–1187.
1975. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Laerum OD, Rajewsky MF, Schachner M,
Stavrou D, Haglid KG and Haugen A: Phenotypic properties of
neoplastic cell lines developed from fetal rat brain cells in
culture after exposure to ethylnitrosourea in vivo. Z Krebsforsch
Klin Onkol Cancer Res Clin Oncol. 89:273–295. 1977. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Sampson JH, Ashley DM, Archer GE, Fuchs
HE, Dranoff G, Hale LP and Bigner DD: Characterization of a
spontaneous murine astrocytoma and abrogation of its tumorigenicity
by cytokine secretion. Neurosurgery. 41:1365–1373. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Serano RD, Pegram CN and Bigner DD:
Tumorigenic cell culture lines from a spontaneous VM/Dk murine
astrocytoma (SMA). Acta Neuropathol. 51:53–64. 1980. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
de Vries NA, Bruggeman SW, Hulsman D, de
Vries HI, Zevenhoven J, Buckle T, Hamans BC, Leenders WP, Beijnen
JH, van Lohuizen M, et al: Rapid and robust transgenic high-grade
glioma mouse models for therapy intervention studies. Clin Cancer
Res. 16:3431–3441. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Marumoto T, Tashiro A, Friedmann-Morvinski
D, Scadeng M, Soda Y, Gage FH and Verma IM: Development of a novel
mouse glioma model using lentiviral vectors. Nat Med. 15:110–116.
2009. View
Article : Google Scholar : PubMed/NCBI
|
|
12
|
Kanu OO, Hughes B, Di C, Lin N, Fu J,
Bigner DD, Yan H and Adamson C: Glioblastoma multiforme
oncogenomics and signaling pathways. Clin Med Oncol. 2009:39–52.
2009.
|
|
13
|
Barker FG, Chang SM, Larson DA, Sneed PK,
Wara WM, Wilson CB and Prados MD: Age and radiation response in
glioblastoma multiforme. Neurosurgery. 49:1288–1298. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Ng WH, Wan GQ and Too HP: Higher
glioblastoma tumour burden reduces efficacy of chemotherapeutic
agents: In vitro evidence. J Clin Neurosci. 14:261–266. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Stewart LA: Chemotherapy in adult
high-grade glioma: A systematic review and meta-analysis of
individual patient data from 12 randomised trials. Lancet.
359:1011–1018. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Katayama N, Sugimoto K, Okada T, Ueha T,
Sakai Y, Akiyoshi H, Mie K, Ueshima E, Sofue K, Koide Y, et al:
Intra-arterially infused carbon dioxide-saturated solution for
sensitizing the anticancer effect of cisplatin in a rabbit VX2
liver tumor model. Int J Oncol. 51:695–701. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Flecknell P: Laboratory animal
anaesthesia, Third edition. Acad Press; 2009
|
|
18
|
Clarke DL, Johansson CB, Wilbertz J,
Veress B, Nilsson E, Karlström H, Lendahl U and Frisén J:
Generalized potential of adult neural stem cells. Science.
288:1660–1663. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Castrén M, Tervonen T, Kärkkäinen V,
Heinonen S, Castrén E, Larsson K, Bakker CE, Oostra BA and Akerman
K: Altered differentiation of neural stem cells in fragile X
syndrome. Proc Natl Acad Sci USA. 102:17834–17839. 2005. View Article : Google Scholar
|
|
20
|
Ikawa M, Tanaka N, Kao WW and Verma IM:
Generation of transgenic mice using lentiviral vectors: A novel
preclinical assessment of lentiviral vectors for gene therapy. Mol
Ther. 8:666–673. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Follenzi A and Naldini L: HIV-based
vectors. Preparation and use. Methods Mol Med. 69:259–274.
2002.PubMed/NCBI
|
|
22
|
Leppänen P, Kholová I, Mähönen AJ, Airenne
K, Koota S, Mansukoski H, Närväinen J, Wirzenius M, Alhonen L,
Jänne J, et al: Short and long-term effects of hVEGF-A(165) in
Cre-activated transgenic mice. PLoS One. 1:e132006. View Article : Google Scholar
|
|
23
|
Close B, Banister K, Baumans V, Bernoth
EM, Bromage N, Bunyan J, Erhardt W, Flecknell P, Gregory N,
Hackbarth H, et al: Recommendations for euthanasia of experimental
animals: Part 1. DGXI of the European Commission. Lab Anim.
30:293–316. 1996. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Louis DN, Ohgaki H, Wiestler OD, Cavenee
WK, Burger PC, Jouvet A, Scheithauer BW and Kleihues P: The 2007
WHO classification of tumours of the central nervous system. Acta
Neuropathol. 114:97–109. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Louis DN, Perry A, Reifenberger G, von
Deimling A, Figarella-Branger D, Cavenee WK, Ohgaki H, Wiestler OD,
Kleihues P and Ellison DW: The 2016 World Health Organization
Classification of Tumors of the Central Nervous System: A summary.
Acta Neuropathol. 131:803–820. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Zhang D, Henning TD, Zou LG, Hu LB, Wen L,
Feng XY, Dai SH, Wang WX, Sun QR and Zhang ZG: Intracranial
ganglioglioma: Clinicopathological and MRI findings in 16 patients.
Clin Radiol. 63:80–91. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Karremann M, Pietsch T, Janssen G, Kramm
CM and Wolff JE: Anaplastic ganglioglioma in children. J
Neurooncol. 92:157–163. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Rogojan L and Olinici CD: Ganglioglioma
with glioblastoma component. Rom J Morphol Embryol. 49:403–406.
2008.PubMed/NCBI
|
|
29
|
Rousseau A, Kujas M, Bergemer-Fouquet AM,
Effenterre R and Hauw JJ: Survivin expression in ganglioglioma. J
Neurooncol. 77:153–159. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Miller DC, Lang FF and Epstein FJ: Central
nervous system gangliogliomas. Part 1: Pathology. J Neurosurg.
79:859–866. 1993. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Mpairamidis E, Alexiou GA, Stefanaki K,
Sfakianos G and Prodromou N: Brainstem ganglioglioma. J Child
Neurol. 23:1481–1483. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Yue WY and Chen ZP: Does vasculogenic
mimicry exist in astrocytoma? J Histochem Cytochem. 53:997–1002.
2005. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Jandial R, Singec I, Ames CP and Snyder
EY: Genetic modification of neural stem cells. Mol Ther.
16:450–457. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Ahmad F, Pacholska A, Tuppurainen V,
Ylä-Herttuala S and Hyvärinen A: Resectable VX-2 rabbit brain tumor
model for development of intraoperative local administration of
drugs. Acta Neurochir (Wien). 153:1979–1981. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Blümcke I and Wiestler OD: Gangliogliomas:
An intriguing tumor entity associated with focal epilepsies. J
Neuropathol Exp Neurol. 61:575–584. 2002. View Article : Google Scholar
|
|
36
|
Zhu JJ, Leon SP, Folkerth RD, Guo SZ, Wu
JK and Black PM: Evidence for clonal origin of neoplastic neuronal
and glial cells in gangliogliomas. Am J Pathol. 151:565–571.
1997.PubMed/NCBI
|
|
37
|
Im SH, Chung CK, Cho BK, Wang KC, Yu IK,
Song IC, Cheon GJ, Lee DS, Kim NR and Chi JG: Intracranial
ganglioglioma: Preoperative characteristics and oncologic outcome
after surgery. J Neurooncol. 59:173–183. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Luyken C, Blümcke I, Fimmers R, Urbach H,
Wiestler OD and Schramm J: Supratentorial gangliogliomas:
Histopathologic grading and tumor recurrence in 184 patients with a
median follow-up of 8 years. Cancer. 101:146–155. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Pandita A, Balasubramaniam A, Perrin R,
Shannon P and Guha A: Malignant and benign ganglioglioma: A
pathological and molecular study1. Neuro Oncol. 9:124–134. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Zentner J, Wolf HK, Ostertun B, Hufnagel
A, Campos MG, Solymosi L and Schramm J: Gangliogliomas: Clinical,
radiological, and histopathological findings in 51 patients. J
Neurol Neurosurg Psychiatry. 57:1497–1502. 1994. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Boer K, Troost D, Timmermans W, van Rijen
PC, Spliet WG and Aronica E: Pi3K-mTOR signaling and AMOG
expression in epilepsy-associated glioneuronal tumors. Brain
Pathol. 20:234–244. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Pekmezci M, Villanueva-Meyer JE, Goode B,
Van Ziffle J, Onodera C, Grenert JP, Bastian BC, Chamyan G, Maher
OM, Khatib Z, et al: The genetic landscape of ganglioglioma. Acta
Neuropathol Commun. 6:472018. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Kim NR, Wang KC, Bang JS, Choe G, Park Y,
Kim SK, Cho BK and Chi JG: Glioblastomatous transformation of
ganglioglioma: Case report with reference to molecular genetic and
flow cytometric analysis. Pathol Int. 53:874–882. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Penisson M, Ladewig J, Belvindrah R and
Francis F: Genes and mechanisms involved in the generation and
amplification of basal radial glial cells. Front Cell Neurosci.
13:3812019. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Wang H, Lafdil F, Wang L, Park O, Yin S,
Niu J, Miller AM, Sun Z and Gao B: Hepatoprotective versus
oncogenic functions of STAT3 in liver tumorigenesis. Am J Pathol.
179:714–724. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Guadagno E, Vitiello M, Francesca P, Calì
G, Caponnetto F, Cesselli D, Camorani S, Borrelli G, Califano M,
Cappabianca P, et al: PATZ1 is a new prognostic marker of
glioblastoma associated with the stem-like phenotype and enriched
in the proneural subtype. Oncotarget. 8:59282–59300. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Vitiello M, Palma G, Monaco M, Bello AM,
Camorani S, Francesca P, Rea D, Barbieri A, Chiappetta G, Vita G,
et al: Dual oncogenic/anti-oncogenic role of PATZ1 in FRTL5 rat
thyroid cells transformed by the Ha-Rasv12 oncogene.
Genes (Basel). 10:1272019. View Article : Google Scholar : PubMed/NCBI
|