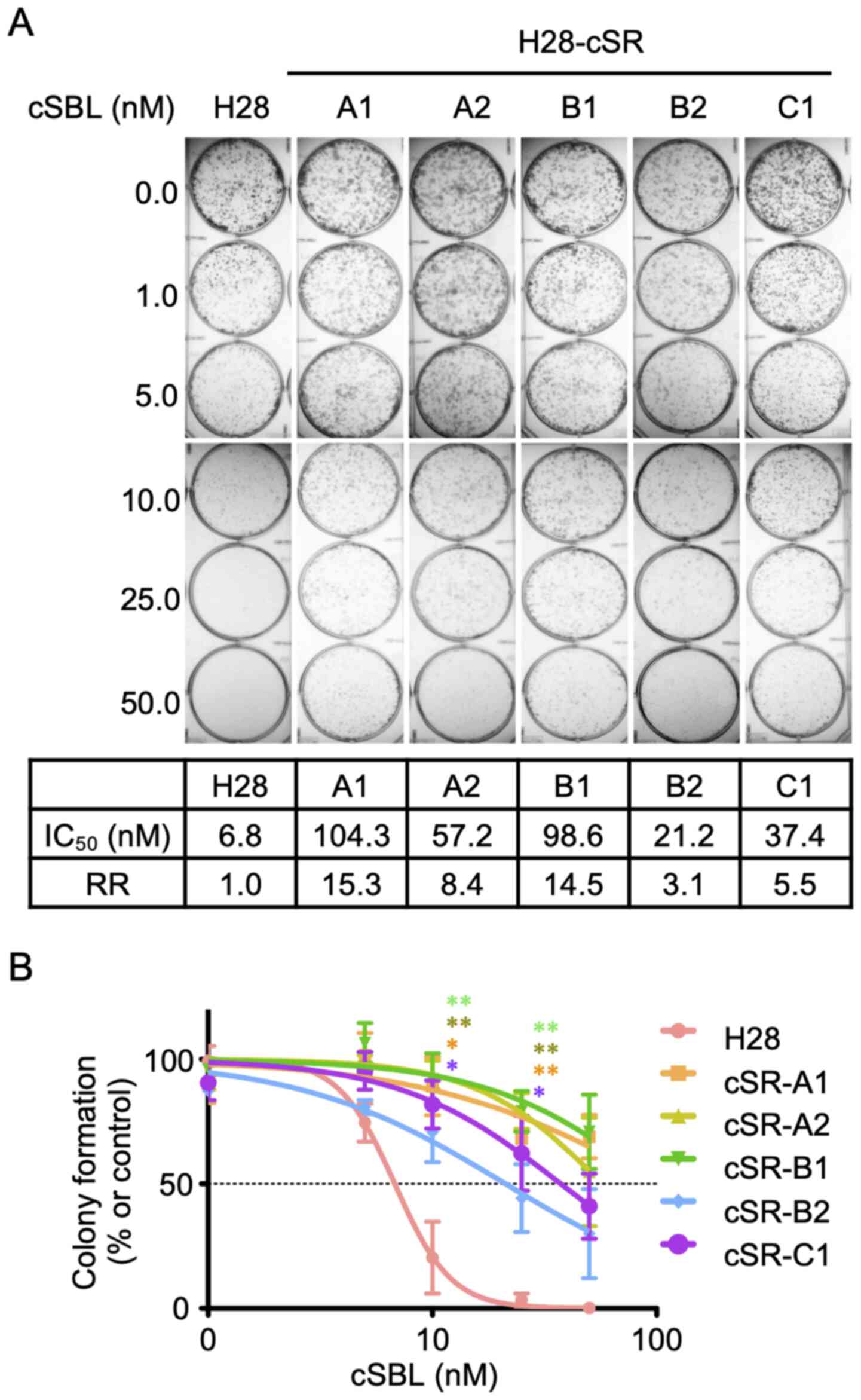
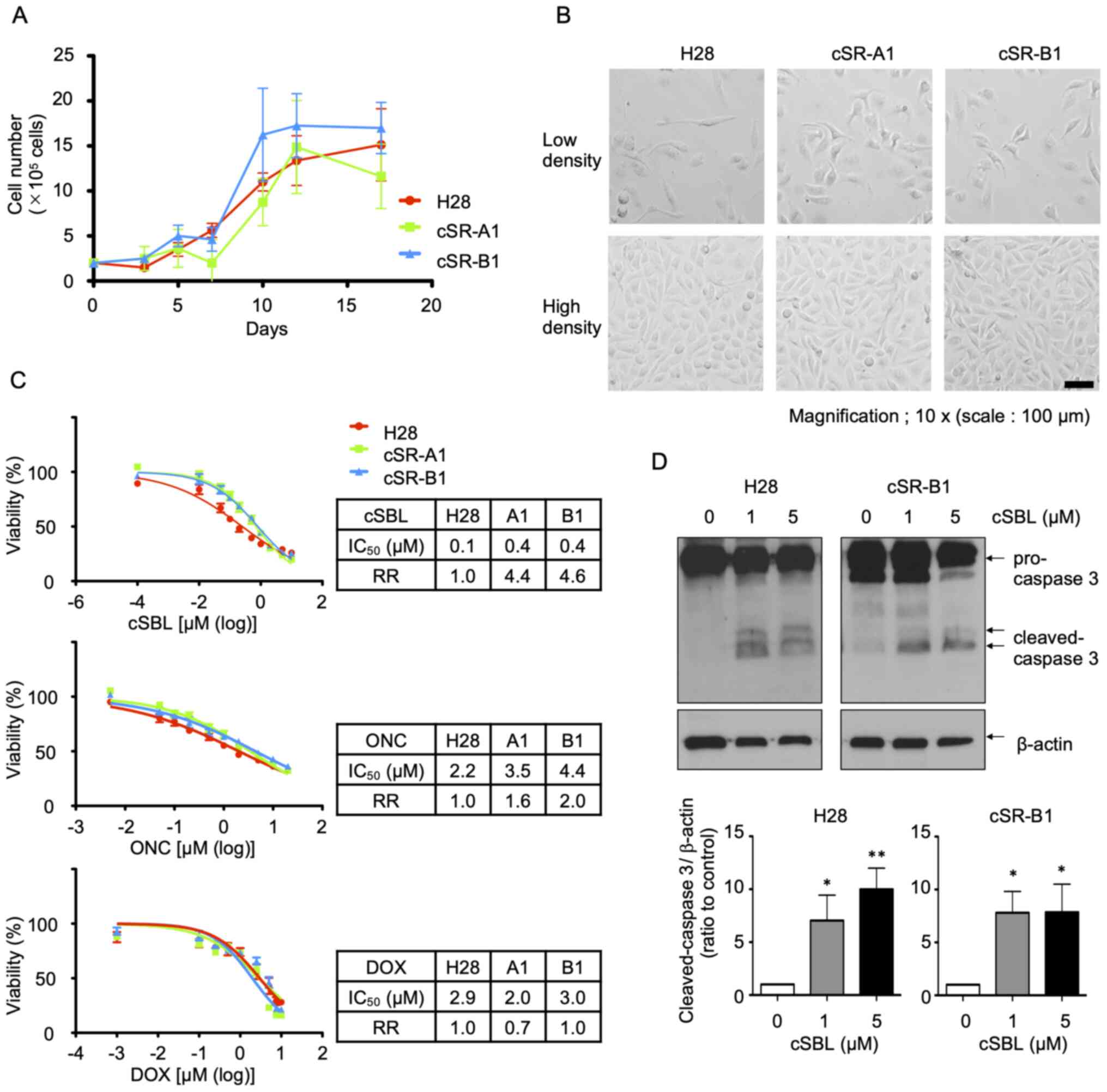
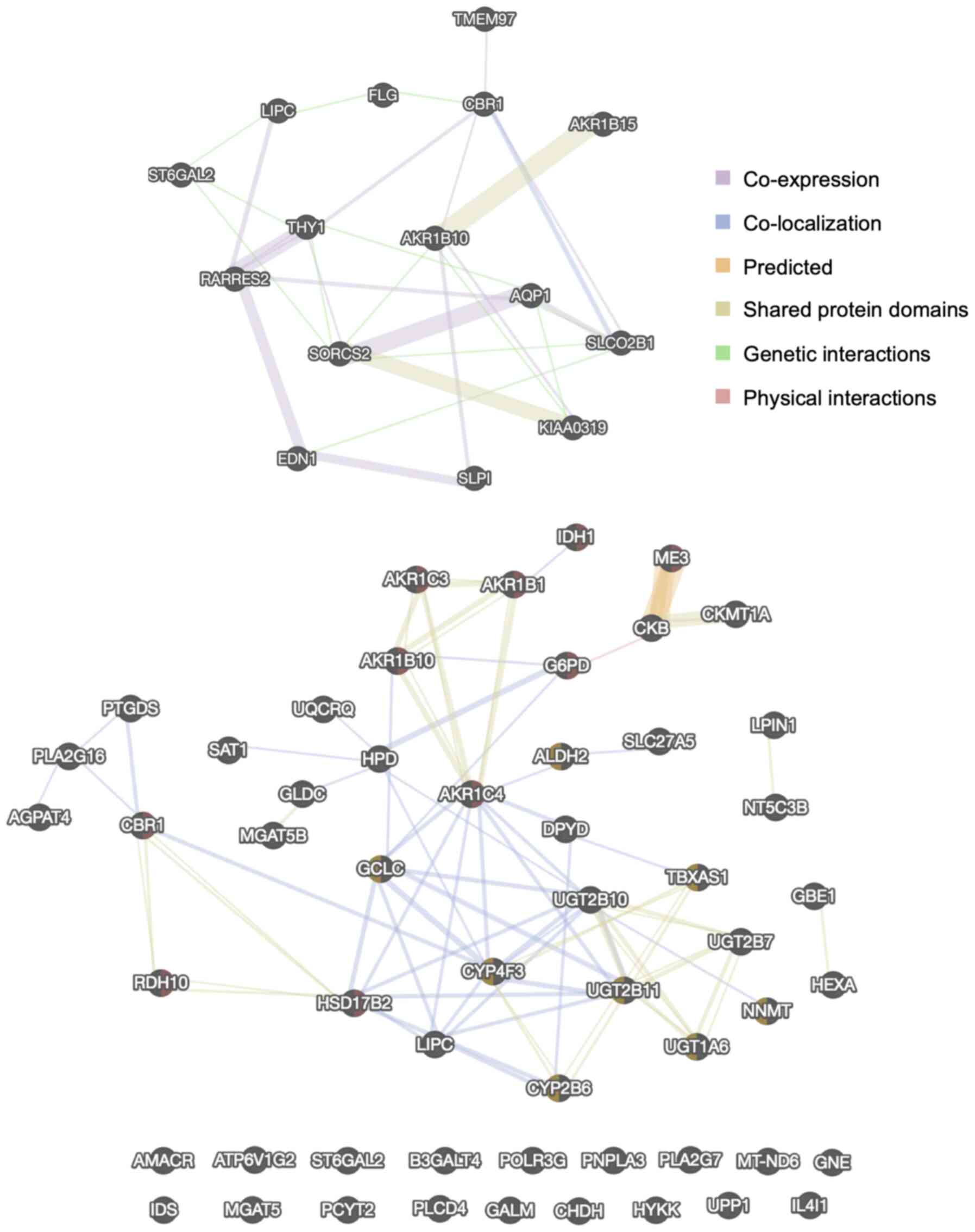
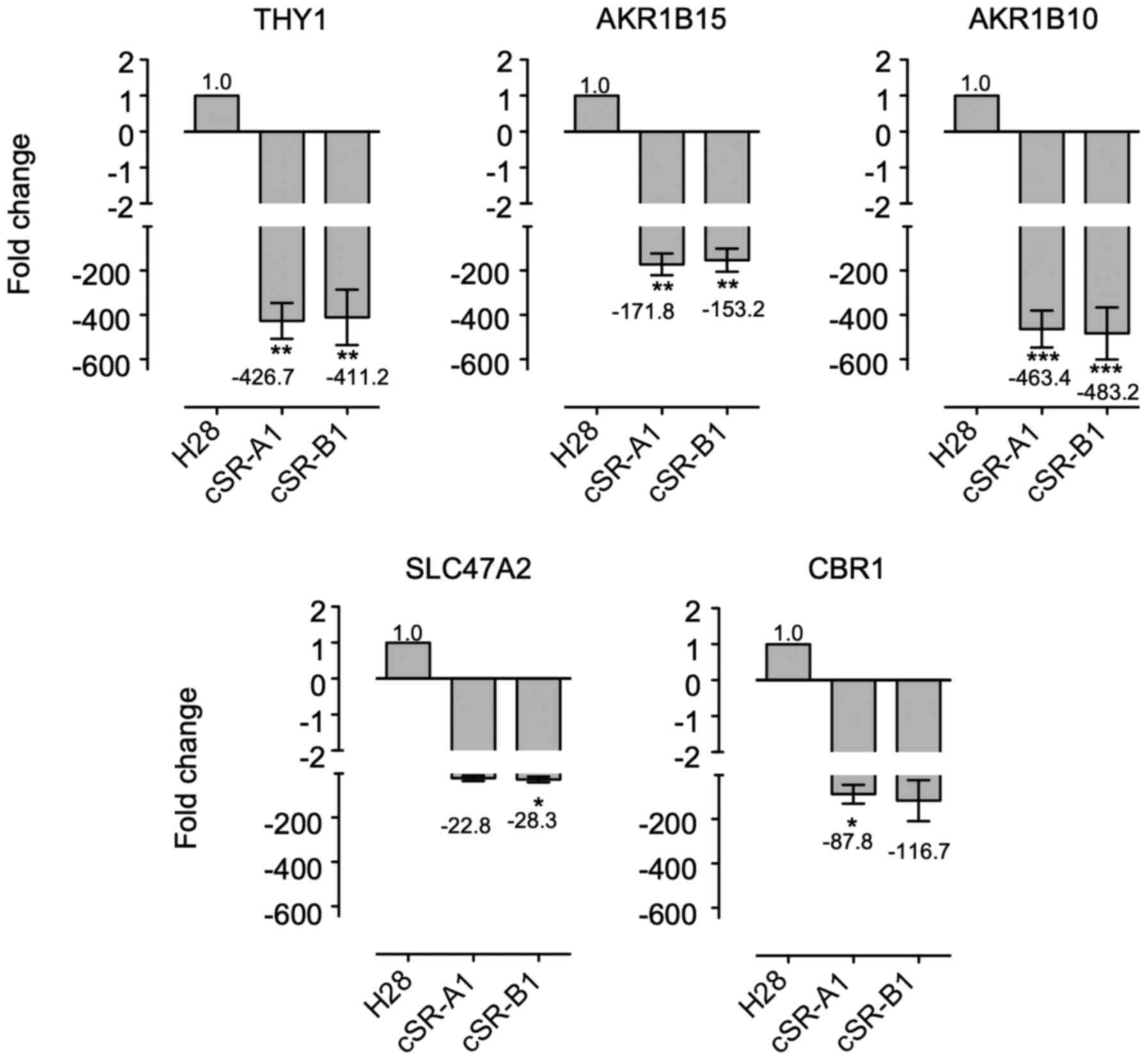
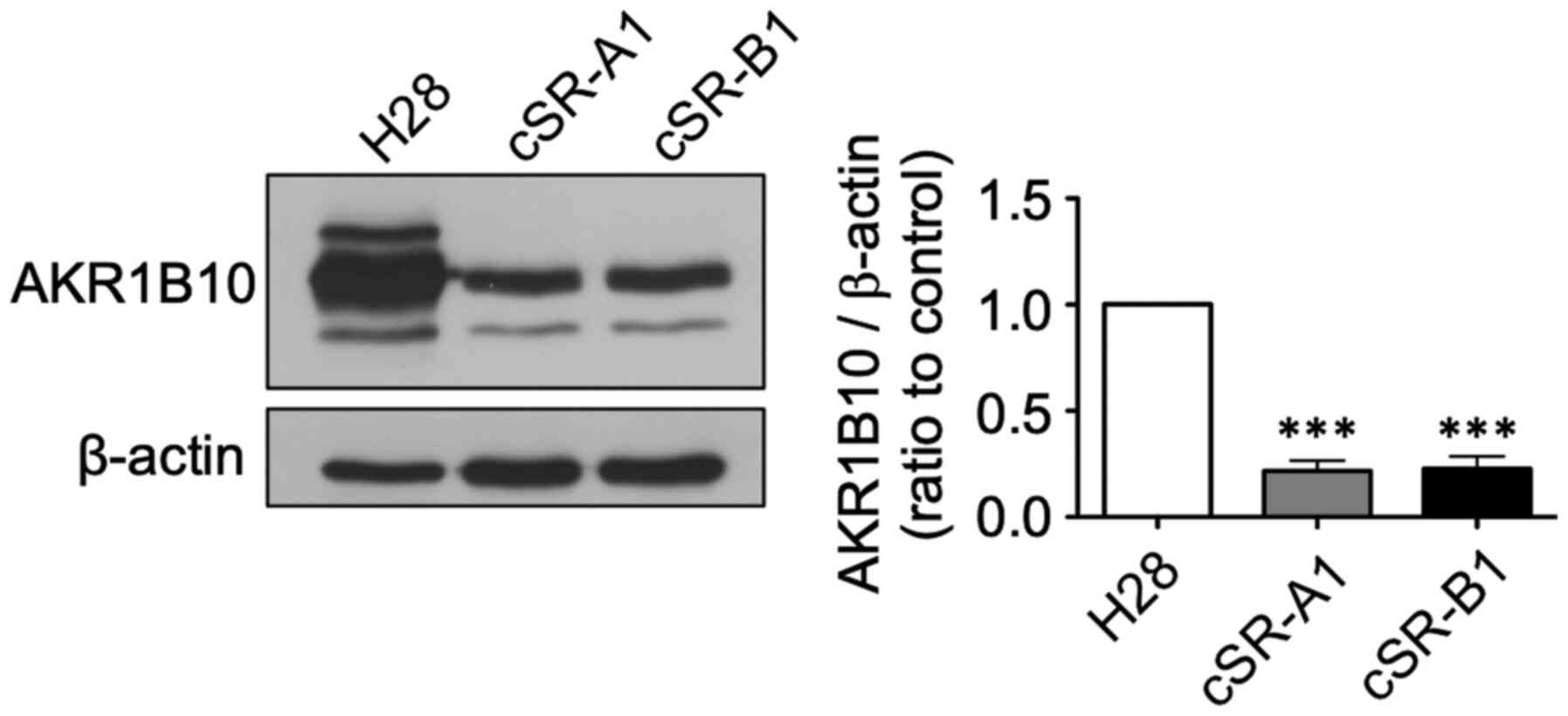
|
1
|
Cakiroglu E and Senturk S: Genomics and
functional genomics of malignant pleural mesothelioma. Int J Mol
Sci. 21:63422020. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Cugell DW and Kamp DW: Asbestos and the
pleura: A review. Chest. 125:1103–1117. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Thanh TD, Van Tho N, Lam NS, Dung NH,
Tabata C and Nakano Y: Simian virus 40 may be associated with
developing malignant pleural mesothelioma. Oncol Lett.
11:2051–2056. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Farioli A, Ottone M, Morganti AG,
Compagnone G, Romani F, Cammelli S, Mattioli S and Violante FS:
Radiation-induced mesothelioma among long-term solid cancer
survivors: A longitudinal analysis of SEER database. Cancer Med.
5:950–959. 2016. View
Article : Google Scholar : PubMed/NCBI
|
|
5
|
Yap TA, Aerts JG, Popat S and Fennell DA:
Novel insights into mesothelioma biology and implications for
therapy. Nat Rev Cancer. 17:475–488. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Van Meerbeeck JP, Gaafar R, Manegold C,
Van Klaveren RJ, Van Marck EA, Vincent M, Legrand C, Bottomley A,
Debruyne C, Giaccone G, et al: Randomized phase III study of
cisplatin with or without raltitrexed in patients with malignant
pleural mesothelioma: An intergroup study of the European
organisation for research and treatment of cancer lung cancer group
and the National Cancer Institute. J Clin Oncol. 23:6881–6889.
2005. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Vogelzang NJ, Rusthoven JJ, Symanowski J,
Denham C, Kaukel E, Ruffie P, Gatzemeier U, Boyer M, Emri S,
Manegold C, et al: Phase III study of pemetrexed in combination
with cisplatin versus cisplatin alone in patients with malignant
pleural mesothelioma. J Clin Oncol. 21:2636–2644. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Wright K: FDA approves nivolumab plus
ipilimumab for the treatment of advanced HCC. Oncology (Willist
Park). 34:6936062020.
|
|
9
|
Shukla A, Hillegass JM, MacPherson MB,
Beuschel SL, Vacek PM, Pass HI, Carbone M, Testa JR and Mossman BT:
Blocking of ERK1 and ERK2 sensitizes human mesothelioma cells to
doxorubicin. Mol Cancer. 9:3142010. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Pouliquen DL, Nawrocki-Raby B, Nader J,
Blandin S, Robard M, Birembaut P and Grégoire M: Evaluation of
intracavitary administration of curcumin for the treatment of
sarcomatoid mesothelioma. Oncotarget. 8:57552–57573. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Makarov AA and Ilinskaya ON: Cytotoxic
ribonucleases: Molecular weapons and their targets. FEBS Lett.
540:15–20. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Fang EF and Ng TB: Ribonucleases of
different origins with a wide spectrum of medicinal applications.
Biochim Biophys Acta. 1815:65–74. 2011.PubMed/NCBI
|
|
13
|
Wus Y, Mikulskiq SM, Ardeltll W, Rybakt SM
and Youlet RJ: A cytotoxic ribonuclease. J Biol Chem.
268:10686–10693. 1993. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Bosch M, Benito A, Ribó M, Puig T,
Beaumelle B and Vilanova M: A nuclear localization sequence endows
human pancreatic ribonuclease with cytotoxic activity.
Biochemistry. 43:2167–2177. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Balandin TG, Edelweiss E, Andronova NV,
Treshalina EM, Sapozhnikov AM and Deyev SM: Antitumor activity and
toxicity of anti-HER2 immunoRNase scFv 4D5-dibarnase in mice
bearing human breast cancer xenografts. Invest New Drugs. 29:22–32.
2011. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Nitta K, Ozaki K, Tsukamoto Y, Hosono M,
Ogawakonno Y, Kawauchi H, Takayanagi Y, Tsuiki S and Hakomori S:
Catalytic lectin (leczyme) from bullfrog (Rana catesbeiana)
eggs: Mechanism of tumoricidal activity. Int J Oncol. 9:19–23.
1996.PubMed/NCBI
|
|
17
|
Nitta K, Takayanagi G, Kawauchi H and
Hakomori S: Isolation and characterization of Rana
catesbeiana lectin and demonstration of the lectin-binding
glycoprotein of rodent and human tumor cell membranes. Cancer Res.
47:4877–83. 1987.PubMed/NCBI
|
|
18
|
Nitta K, Ozaki K, Tsukamoto Y, Furusawa S,
Ohkubo Y, Takimoto H, Murata R, Hosono M, Hikichi N, Sasaki K, et
al: Characterization of a Rana catesbeiana lectin-resistant
mutant of leukemia P388 cells. Cancer Res. 54:928–934.
1994.PubMed/NCBI
|
|
19
|
Titani K, Takio K, Kuwada M, Nitta K,
Sakakibara F, Kawauchi H, Takayanagi G and Hakomori S: Amino acid
sequence of sialic acid binding lectin from frog (Rana
catesbeiana) eggs. Biochemistry. 26:2189–2194. 1987. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Nitta K, Ozaki K, Ishikawa M, Furusawa S,
Hosono M, Kawauchi H, Sasaki K, Takayanagi Y, Tsuiki S and Hakomori
S: Inhibition of cell proliferation by Rana catesbeiana and
Rana japonica lectins belonging to the ribonuclease
superfamily. Cancer Res. 54:920–927. 1994.PubMed/NCBI
|
|
21
|
Tatsuta T, Sugawara S, Takahashi K, Ogawa
Y, Hosono M and Nitta K: Cancer-selective induction of apoptosis by
leczyme. Front Oncol. 4:1392014. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Tatsuta T, Hosono M, Ogawa Y, Inage K,
Sugawara S and Nitta K: Downregulation of Hsp70 inhibits apoptosis
induced by sialic acid-binding lectin (leczyme). Oncol Rep.
31:13–18. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Tatsuta T, Sugawara S, Takahashi K, Ogawa
Y, Hosono M and Nitta K: Leczyme: A new candidate drug for cancer
therapy. Biomed Res Int. 2014:4214152014. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Chen JN, Yiang GT, Lin YF, Chou PL, Wu TK,
Chang WJ, Chen C and Yu YL: Rana catesbeiana ribonuclease
induces cell apoptosis via the caspase-9/-3 signaling pathway in
human glioblastoma DBTRG, GBM8901 and GBM8401 cell lines. Oncol
Lett. 9:2471–2476. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Tatsuta T, Sato S, Sato T, Sugawara S,
Suzuki T, Hara A and Hosono M: Sialic acid-binding lectin from
bullfrog eggs exhibits an anti-tumor effect against breast cancer
cells including triple-negative phenotype cells. Molecules.
23:27142018. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Tatsuta T, Satoh T, Sugawara S, Hara A and
Hosono M: Sialic acid-binding lectin from bullfrog eggs inhibits
human malignant mesothelioma cell growth in vitro and in vivo. PLoS
One. 13:e01906532018. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Tatsuta T, Hosono M, Sugawara S, Kariya Y,
Ogawa Y, Hakomori S and Nitta K: Sialic acid-binding lectin
(leczyme) induces caspase-dependent apoptosis-mediated
mitochondrial perturbation in Jurkat cells. Int J Oncol.
43:1402–1412. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Tatsuta T, Hosono M, Miura Y, Sugawara S,
Kariya Y, Hakomori S and Nitta K: Involvement of ER stress in
apoptosis induced by sialic acid-binding lectin (leczyme) from
bullfrog eggs. Int J Oncol. 43:1799–1808. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Kariya Y, Tatsuta T, Sugawara S, Kariya Y,
Nitta K and Hosono M: RNase activity of sialic acid-binding lectin
from bullfrog eggs drives antitumor effect via the activation of
p38 MAPK to caspase3/7 signaling pathway in human breast cancer
cells. Int J Oncol. 49:1334–1342. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Tatsuta T, Hosono M, Takahashi K, Omoto T,
Kariya Y, Sugawara S, Hakomori S and Nitta K: Sialic acid-binding
lectin (leczyme) induces apoptosis to malignant mesothelioma and
exerts synergistic antitumor effects with TRAIL. Int J Oncol.
44:377–384. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Satoh T, Tatsuta T, Sugawara S, Hara A and
Hosono M: Synergistic anti-tumor effect of bullfrog sialic
acid-binding lectin and pemetrexed in malignant mesothelioma.
Oncotarget. 8:42466–42477. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Altomare DA, Rybak SM, Pei J, Maizel JV,
Cheung M, Testa JR and Shogen K: Onconase responsive genes in human
mesothelioma cells: Implications for an RNA damaging therapeutic
agent. BMC Cancer. 10:342010. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Vert A, Castro J, Ribó M, Benito A and
Vilanova M: Activating transcription factor 3 is crucial for
antitumor activity and to strengthen the antiviral properties of
Onconase. Oncotarget. 8:11692–11707. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Vert A, Castro J, Ribó M, Benito A and
Vilanova M: A nuclear-directed human pancreatic ribonuclease (PE5)
targets the metabolic phenotype of cancer cells. Oncotarget.
7:18309–18324. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Bolstad BM, Irizarry RA, Astrand M and
Speed TP: A comparison of normalization methods for high density
oligonucleotide array data based on variance and bias.
Bioinformatics. 19:185–193. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Gentleman R, Carey V, Bates D, Bolstad B,
Dettling M, Dudoit S, Ellis B, Gautier L, Ge Y, Gentry J, et al:
Bioconductor: Open software development for computational biology
and bioinformatics. Genome Biol. 5:R802004. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Dudoit S, Gentleman RC and Quackenbush J:
Open source software for the analysis of microarray data.
Biotechniques. 34:496–501. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Edgar R, Domrachev M and Lash AE: Gene
Expression Omnibus: NCBI gene expression and hybridization array
data repository. Nucleic Acids Res. 30:207–210. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Ashburner M, Ball CA, Blake JA, Botstein
D, Butler H, Cherry JM, Davis AP, Dolinski K, Dwight SS, Eppig JT,
et al: Gene ontology: Tool for the unification of biology. The Gene
Ontology Consortium. Nat Genet. 25:25–29. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
The Gene Ontology Consortium, . The Gene
Ontology Resource: 20 years and still GOing strong. Nucleic Acids
Res. 47:D330–D338. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Kanehisa M: KEGG: Kyoto encyclopedia of
genes and genomes. Nucleic Acids Res. 28:27–30. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Dennis G Jr, Sherman BT, Hosack DA, Yang
J, Gao W, Lane HC and Lempicki RA: DAVID: Database for annotation,
visualization, and integrated discovery. Genome Biol. 4:R602003.
View Article : Google Scholar
|
|
43
|
Warde-Farley D, Donaldson SL, Comes O,
Zuberi K, Badrawi R, Chao P, Franz M, Grouios C, Kazi F, Lopes CT,
et al: The GeneMANIA prediction server: Biological network
integration for gene prioritization and predicting gene function.
Nucleic Acids Res. 38((Web Server issue)): W214–W220. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Balendiran GK, Martin HJ, El-Hawari Y and
Maser E: Cancer biomarker AKR1B10 and carbonyl metabolism. Chem
Biol Interact. 178:134–137. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Thuren T: Hepatic lipase and HDL
metabolism. Curr Opin Lipidol. 11:277–283. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Takashima S, Tsuji S and Tsujimoto M:
Characterization of the second type of human beta-galactoside alpha
2,6-sialyltransferase (ST6Gal II), which sialylates Galβ1,4GlcNAc
structures on oligosaccharides preferentially: Genomic analysis of
human sialyltransferase genes. J Biol Chem. 277:45719–45728. 2002.
View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Jez JM, Flynn TG and Penning TM: A
nomenclature system for the aldo-keto reductase superfamily. Adv
Exp Med Biol. 414:579–589. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Barski OA, Tipparaju SM and Bhatnagar A:
The aldo-keto reductase superfamily and its role in drug metabolism
and detoxification. Drug Metab Rev. 40:553–624. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Mindnich RD and Penning TM: Aldo-keto
reductase (AKR) superfamily: Genomics and annotation. Hum Genomics.
3:362–370. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Fukumoto SI, Yamauchi N, Moriguchi H,
Hippo Y, Watanabe A, Shibahara J, Taniguchi H, Ishikawa S, Ito H,
Yamamoto S, et al: Overexpression of the aldo-keto reductase family
protein AKR1B10 is highly correlated with smokers' non-small cell
lung carcinomas. Clin Cancer Res. 11:1776–1785. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
51
|
Cao D, Fan ST and Chung SSM:
Identification and characterization of a novel human aldose
reductase- like gene. J Biol Chem. 273:11429–11435. 1998.
View Article : Google Scholar : PubMed/NCBI
|
|
52
|
Distefano JK and Davis B: Diagnostic and
prognostic potential of akr1b10 in human hepatocellular carcinoma.
Cancers (Basel). 11:4862019. View Article : Google Scholar : PubMed/NCBI
|
|
53
|
Chung YT, Matkowskyj KA, Li H, Bai H,
Zhang W, Tsao MS, Liao J and Yang GY: Overexpression and oncogenic
function of aldo-keto reductase family 1B10 (AKR1B10) in pancreatic
carcinoma. Mod Pathol. 25:758–766. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
54
|
Fang CY, Lin YH and Chen CL:
Overexpression of AKR1B10 predicts tumor recurrence and short
survival in oral squamous cell carcinoma patients. J Oral Pathol
Med. 48:712–719. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
55
|
Huang P, Chandra V and Rastinejad F:
Retinoic acid actions through mammalian nuclear receptors. Chem
Rev. 114:233–254. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
56
|
Huang L, He R, Luo W, Zhu YS, Li J, Tan T,
Zhang X, Hu Z and Luo D: Aldo-Keto reductase family 1 member B10
inhibitors: Potential drugs for cancer treatment. Recent Pat
Anticancer Drug Discov. 11:184–196. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
57
|
Ruiz FX, Porté S, Parés X and Farrés J:
Biological role of aldo-keto reductases in retinoic acid
biosynthesis and signaling. Front Pharmacol. 3:582012. View Article : Google Scholar : PubMed/NCBI
|
|
58
|
Wang C, Yan R, Luo D, Watabe K, Liao DF
and Cao D: Aldo-keto reductase family 1 member B10 promotes cell
survival by regulating lipid synthesis and eliminating carbonyls. J
Biol Chem. 284:26742–26748. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
59
|
Shen Y, Zhong L, Johnson S and Cao D:
Human aldo-keto reductases 1B1 and 1B10: A comparative study on
their enzyme activity toward electrophilic carbonyl compounds. Chem
Biol Interact. 191:192–198. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
60
|
Matsunaga T, Suzuki A, Kezuka C, Okumura
N, Iguchi K, Inoue I, Soda M, Endo S, El-Kabbani O, Hara A and
Ikari A: Aldo-keto reductase 1B10 promotes development of cisplatin
resistance in gastrointestinal cancer cells through down-regulating
peroxisome proliferator-activated receptor-γ-dependent mechanism.
Chem Biol Interact. 256:142–153. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
61
|
Matsunaga T, Yamane Y, Iida K, Endo S,
Banno Y, El-Kabbani O and Hara A: Involvement of the aldo-keto
reductase, AKR1B10, in mitomycin-c resistance through reactive
oxygen species-dependent mechanisms. Anticancer Drugs. 22:402–408.
2011. View Article : Google Scholar : PubMed/NCBI
|
|
62
|
Zhong L, Shen H, Huang C, Jing H and Cao
D: AKR1B10 induces cell resistance to daunorubicin and idarubicin
by reducing C13 ketonic group. Toxicol Appl Pharmacol. 255:40–47.
2011. View Article : Google Scholar : PubMed/NCBI
|
|
63
|
Mundt F, Johansson HJ, Forshed J, Arslan
S, Metintas M, Dobra K, Lehtiö J and Hjerpe A: Proteome screening
of pleural effusions identifies galectin 1 as a diagnostic
biomarker and highlights several prognostic biomarkers for
malignant mesothelioma. Mol Cell Proteomics. 13:701–715. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
64
|
Usami N, Fukui T, Kondo M, Taniguchi T,
Yokoyama T, Mori S, Yokoi K, Horio Y, Shimokata K, Sekido Y and
Hida T: Establishment and characterization of four malignant
pleural mesothelioma cell lines from Japanese patients. Cancer Sci.
97:387–394. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
65
|
Suzuki Y, Murakami H, Kawaguchi K,
Tanigushi T, Fujii M, Shinjo K, Kondo Y, Osada H, Shimokata K,
Horio Y, et al: Activation of the PI3K-AKT pathway in human
malignant mesothelioma cells. Mol Med Rep. 2:181–188.
2009.PubMed/NCBI
|
|
66
|
Endo S, Xia S, Suyama M, Morikawa Y, Oguri
H, Hu D, Ao Y, Takahara S, Horino Y, Hayakawa Y, et al: Synthesis
of potent and selective inhibitors of Aldo-Keto reductase 1B10 and
their efficacy against proliferation, metastasis, and cisplatin
resistance of lung cancer cells. J Med Chem. 60:8441–8455. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
67
|
Plebuch M, Soldan M, Hungerer C, Koch L
and Maser E: Increased resistance of tumor cells to daunorubicin
after transfection of cDNAs coding for anthracycline inactivating
enzymes. Cancer Lett. 255:49–56. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
68
|
Shiiba M, Yamagami H, Yamamoto A, Minakawa
Y, Okamoto A, Kasamatsu A, Sakamoto Y, Uzawa K, Takiguchi Y and
Tanzawa H: Mefenamic acid enhances anticancer drug sensitivity via
inhibition of aldo-keto reductase 1C enzyme activity. Oncol Rep.
37:2025–2032. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
69
|
Vasiliou V, Vasiliou K and Nebert DW:
Human ATP-binding cassette (ABC) transporter family. Hum Genomics.
3:281–290. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
70
|
Pasello M, Giudice AM and Scotlandi K: The
ABC subfamily A transporters: Multifaceted players with incipient
potentialities in cancer. Semin Cancer Biol. 60:57–71. 2020.
View Article : Google Scholar : PubMed/NCBI
|
|
71
|
Iwasaki H, Okabe T, Takara K, Yoshida Y,
Hanashiro K and Oku H: Down-regulation of lipids transporter ABCA1
increases the cytotoxicity of nitidine. Cancer Chemother Pharmacol.
66:953–959. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
72
|
Hudson AL, Weir C, Moon E, Harvie R, Klebe
S, Clarke SJ, Pavlakis N and Howell VM: Establishing a panel of
chemo-resistant mesothelioma models for investigating
chemo-resistance and identifying new treatments for mesothelioma.
Sci Rep. 4:61522014. View Article : Google Scholar : PubMed/NCBI
|
|
73
|
Milosevic V, Kopecka J, Salaroglio IC,
Libener R, Napoli F, Izzo S, Orecchia S, Ananthanarayanan P,
Bironzo P, Grosso F, et al: Wnt/IL-1β/IL-8 autocrine circuitries
control chemoresistance in mesothelioma initiating cells by
inducing ABCB5. Int J Cancer. 146:192–207. 2020. View Article : Google Scholar : PubMed/NCBI
|