Introduction
Liver cancer is a malignant tumor that usually
involves the digestive system, and consists of primary and
secondary types. Primary liver cancer is divided into
hepatocellular carcinoma and intrahepatic cholangiocarcinoma
(1,2). Notably, liver cancer is associated
with a high incidence and mortality rate worldwide (3). According to the World Health
Organization, it is predicted that there will be >1,000,000
liver cancer-related deaths by 2030, and of the newly confirmed
cases, those in mainland China will account for 46.6%.
Oxaliplatin (OXA) has been reported to exert
inhibitory effects on the growth of liver cancer with tolerable
toxicity in clinical settings. Nevertheless, the overall efficiency
of platinum-based drug therapy is hampered by tumor cell resistance
(4). It is well acknowledged that
liver cancer exhibits lower sensitivity to chemotherapy compared
with other types of cancer. The multidrug resistance of liver
cancer has contributed to its resistance to numerous therapeutic
agents (5). In a previous study,
Xie and Zhong (6) reported that
HepG2 cells exhibited poor sensitivity to adriamycin,
5-fluorouracil and cisplatin under hypoxic conditions. Despite the
fact that platinum-based chemotherapy agents are the major
treatment options for cancer, resistance to these drugs does exist
among patients. Furthermore, the prognosis of patients with liver
cancer remains poor (7,8). To date, extensive efforts have been
made to investigate drug resistance and to improve drug sensitivity
in patients with liver cancer.
Under hypoxic conditions, revascularization occurs
in cancer cells, which can lead to epithelial-mesenchymal
transition (EMT) and vascular mimicry (VM). EMT and VM may
subsequently promote invasion and distant metastasis. Moreover, EMT
has been speculated to serve a role as the driving force for cancer
progression (9). In addition,
hypoxia-inducible factors (HIFs) are involved in neovessel
formation, energy metabolism, cellular proliferation, invasion and
metastasis (10).
Lysyl oxidase (LOX)-like 2 (LOXL2) protein is a
member of the LOX family, and is closely related to the covalent
cross-linking of collagen and elastin, which can result in fibrosis
and is crucial for the integrity of the extracellular matrix
(11). In a previous study, LOXL2
was considered to be closely related to the metastasis of cancer
cells (12). Furthermore, LOXL2 has
been shown to modulate the pathogenesis and progression of numerous
types of malignant cancer though extra- and intra-cellular
pathways, which were important indices for the evaluation of poor
prognosis (13,14).
Herba Cistanche is a tonic herb commonly distributed
in desert regions, which has been frequently used in traditional
Chinese medicine (15,16). Cistanche tubulosa (C.
tubulosa) is a natural herbal medicine commonly planted in
Xinjiang Autonomous Region. The phenylethanol glycosides from Herba
Cistanche (CPhGs) serve as one of the major active components of
Herba Cistanche. Previously, Hu et al (17) indicated that CPhGs could attenuate
liver injury in H22 tumor-bearing mice and inhibit the growth of
cancer cells. It was suggested that the underlying mechanism may be
related to a reduction in serum α-fetoprotein and could enhance
immunity in the mice.
In the present study, a hypoxic model of HepG2 liver
cancer cells was induced using CoCl2. On this basis, the
present study aimed to investigate the effects of OXA on the
proliferation, apoptosis, migration and invasion of cancer cells in
the presence of CPhGs under hypoxic conditions. In addition, the
mRNA and protein expression levels of HIF-1α, LOXL2, E-cadherin and
Twist were detected. Moreover, the exact mechanisms underlying the
effects of CPhGs on the pathogenesis of liver cancer were
investigated.
Materials and methods
Cell line
The liver cancer cell line HepG2, as identified
using the STR method, was provided by the Clinical Research
Institution, First Affiliated Hospital of Xinjiang Medical
University (Urumqi, China). The cells were cultured in high-glucose
DMEM (HyClone; Cytiva) containing 10% fetal bovine serum (FBS;
Hyclone; Cytiva) at 37°C in an incubator containing 5%
CO2.
Preparation of CPhGs
C. tubulosa extraction (CPhGs) was obtained
from Hetian Dichen Biotech Co., Ltd.. The content of CPhGs was
>80%, among which the content of echinacoside and verbascose was
44.5 and 16.1%, respectively. Stems of C. tubulosa (Schrenk)
Wight were collected in October 2016 from Xinjiang, China. The
plant was identified by Dr. Junping Hu. All these voucher specimens
(no. 201610) have been deposited at the Plant Herbarium, School of
Pharmacy, Xinjiang Medical University, Xinjiang, China.
Experimental design
HepG2 cells (5×104/ml) were treated with
various concentrations of CPhGs (5, 25, 50, 100, 200 and 500 µg/ml)
for 48 h at 37°C (24 h after seeding) to screening CPhGs-L/M/H
dose. HepG2 cells (5×104/ml) were divided into the
following groups: i) Control group, cultured in high-glucose DMEM;
ii) DMSO group, cultured in 0.1% DMSO (v/v); iii) CoCl2
group (hypoxia model group), cultured in serum-free DMEM containing
100 µM CoCl2; iv) OXA group (positive control group),
cultured in 5 µM OXA (Beijing Solarbio Science & Technology
Co., Ltd.); v) OXA + CoCl2 group, cultured in 5 µM OXA
combined with 100 µM CoCl2; and vi) CPhGs groups,
treated with CPhGs-L/M/H (25, 50 and 100 µg/ml, respectively)
combined with 5 µM OXA and 100 µM CoCl2.
Cell viability assay
Cell viability was determined using a Cell Counting
Kit-8 (CCK-8) assay according to the manufacturer's instructions
(Beijing Solarbio Science & Technology Co., Ltd.) and as
previously described (18). Cells
(2.0×103 cells/well) were seeded in 96-well plates.
Subsequently, ~24 h after seeding, cells were treated for 48 h
according to the treatment conditions in each group. The medium was
then replaced with 100 µl high-glucose DMEM, followed by the
addition of 10 µl CCK-8 reagent; the cells were incubated for 1 h
at 37°C. Optical density was measured using a multi-detection
microplate reader (Thermo Fisher Scientific, Inc.) at 450 nm. Six
replicates were prepared for each condition.
Determination of apoptotic rate
Apoptotic rate was determined using an Annexin V/PI
Apoptotic Detection Kit (Beijing Solarbio Science & Technology
Co., Ltd.). HepG2 cells (5×105) were inoculated in
6-well plates at 37°C in 5% CO2. Subsequently, ~24 h
after treatment, the cells were cultured in serum-free DMEM for 4
h. The cells were then digested using 0.25% trypsinized (Gibco;
Thermo Fisher Scientific, Inc.), followed by at least three washes
with pre-cooled PBS. Upon centrifugation at 167.7 × g for 5 min at
4°C, the cells were resuspended with 1X binding buffer and the
concentration was adjusted to 1~5×106/ml, and then
stained with 5 µl Annexin V-FITC and 5 µl PI for 15 min at room
temperature. The cells then underwent flow cytometry using a BD
LSRFortessa flow cytometer (BD Biosciences) and FlowJo 10.6.2
software (Tree Star, Inc.).
Wound-healing assay
The inhibitory effects of CPhGs on cell migration
were examined by wound-healing assay (19). The cells were seeded into 6-well
plates until a 100% confluent monolayer was obtained. Subsequently,
the cells were wounded using a 200-µl pipette tip, washed with PBS
and incubated with treatments in serum-free medium. After drug
treatment, the wound-healing speed was measured at 0, 12, 24 and 48
h, respectively. Wound images were obtained using a fluorescent
microscope (Nikon Ti-S, Japan) under a magnification of ×10. Wound
closure was measured by the wound distance in each period and
expressed as percentage of the initial wound distance at 0 h.
Transwell assays
Cell invasion was assessed by Transwell assays.
Cells (1×105 cells/ml) were suspended in 200 µl
high-glucose DMEM without FBS. The cells were then seeded onto
Matrigel-coated upper wells covered with a polyethylene
terephthalate filter membrane (pore size, 8.0 µm). A total of 500
µl high-glucose DMEM containing 10% FBS was placed in the lower
chamber. Cotton swabs were used to remove the cells on the upper
surface of the filter after 48 h at 37°C. The cells that invaded
through the membrane were fixed with 4% paraformaldehyde for 30
min. Subsequently, cells were stained with 0.1% crystal violet for
15 min at room temperature. Invading cells were observed under a
fluorescent microscope (Nikon Ti-S) at a magnification of ×100.
Reverse transcription-quantitative PCR
(RT-qPCR)
Total RNA was extracted from HepG2 cells using
TRIzol® reagent (Invitrogen; Thermo Fisher Scientific,
Inc.) and cDNA synthesis was carried out using the PrimeScript RT
reagent kit (Takara Bio, Inc.) according to manufacturer's
protocol. qPCR was performed on a 7500 Real-Time PCR system
(Applied Biosystems; Thermo Fisher Scientific, Inc.) using TB
green™ Premix Ex Taq™ (Takara Bio, Inc.) according to
manufacturer's protocol. The primers used for qPCR are listed in
Table I. PCR conditions consisted
of denaturation at 95°C for 30 sec, followed by 40 cycles of
denaturation at 95°C for 5 sec and annealing at 60°C for 30 sec.
Finally, the amplification results were analyzed using the
2−ΔΔCq method (20).
 | Table I.Primer sequences. |
Table I.
Primer sequences.
| Gene | Sequence
(5′-3′) |
|---|
| β-actin | F:
TGGCACCCAGCACAATGAA |
|
| R:
CTAAGTCATAGTCCGCCTAGAAGCA |
| HIF-1α | F:
CAAGAAACCACCCATGAC |
|
| R:
GGCTCATAACCCATCAAC |
| LOXL2 | F:
ACAGAATGTGAAGGAGACATCC |
|
| R:
TGATGTTGTTGGAGTAATCGGA |
| E-cadherin | F:
GACAGGCTGGCTGAAAGTG |
|
| R:
TGGCTGACGATGGTGTAGG |
| Twist1 | F:
GTACATCGACTTCCTCTACCAG |
|
| R:
CATCCTCCAGACCGAGAAG |
Western blot analysis
Proteins were extracted from cells treated for 48 h
by homogenization in RIPA lysis buffer (Thermo Fisher Scientific,
Inc.) containing protease and phosphatase inhibitors. Cell protein
content was determined using the BCA method. Proteins (40 µg) were
then separated by SDS-PAGE on a 10% gel and transferred to a PVDF
membrane. The membrane was blocked in 5% nonfat milk for 1 h at
4°C, and incubated with the following primary antibodies: β-actin
(1:5,000; cat. no. bs-0061R; BIOSS), HIF-1α (1:1,000; cat. no.
ab179483; Abcam), LOXL2 (1:500; cat. no. ab179810; Abcam),
E-cadherin (1:1,000; cat. no. bs-10009R; BIOSS) and Twist1 (1:500;
cat. no. bs-2441R; BIOSS) overnight at 4°C. The membrane was then
incubated with goat anti-rabbit IgG H&L secondary antibodies
(1:2,000; cat. no. ab205718; Abcam) for 4 h at room temperature.
After washing with TBS-0.05% Tween-20, the blots were visualized
using the Enhanced Chemiluminescence system (Amersham; Cytiva). The
relative intensity of the bands was semi-quantified by
densitometric analysis using ImageJ2× software (version 2.1.4.7;
Rawak Software Inc.), and densitometric plots of the results were
normalized to the intensity of β-actin.
Statistical analysis
SPSS 19.0 software (SPSS, Inc.) was utilized for
data analysis. Data are presented as the mean ± standard deviation
and were analyzed by one-way ANOVA followed by Tukey's post hoc
test. P<0.05 was considered to indicate a statistically
significant difference. All experiments were performed at least in
triplicate.
Results
Effects of CPhGs on cell
viability
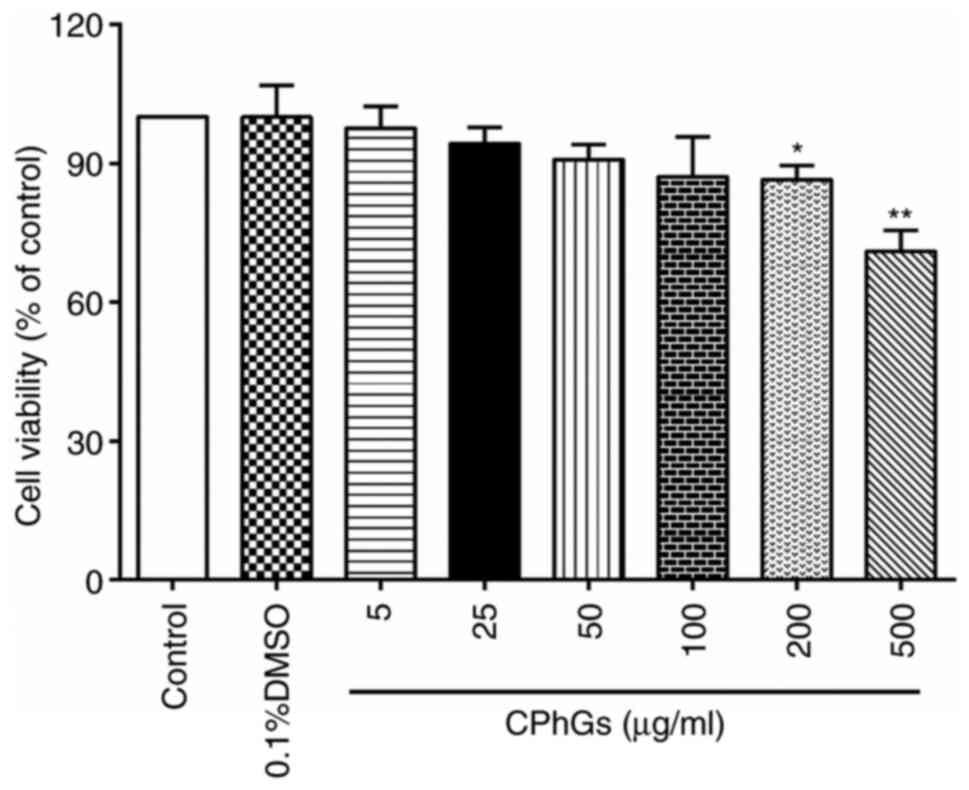
HepG2 cells were treated with various concentrations
of CPhGs (5, 25, 50, 100, 200 and 500 µg/ml) for 48 h (24 h after
seeding). As shown in Fig. 1, there
was significant decline in the viability of cells treated with 200
and 500 µg/ml CPhGs compared with that of the control group
(P<0.05). These findings indicated that CPhGs could modulate
cell viability in a dose-dependent manner.
CPhGs enhances the effects of OXA on
liver cancer
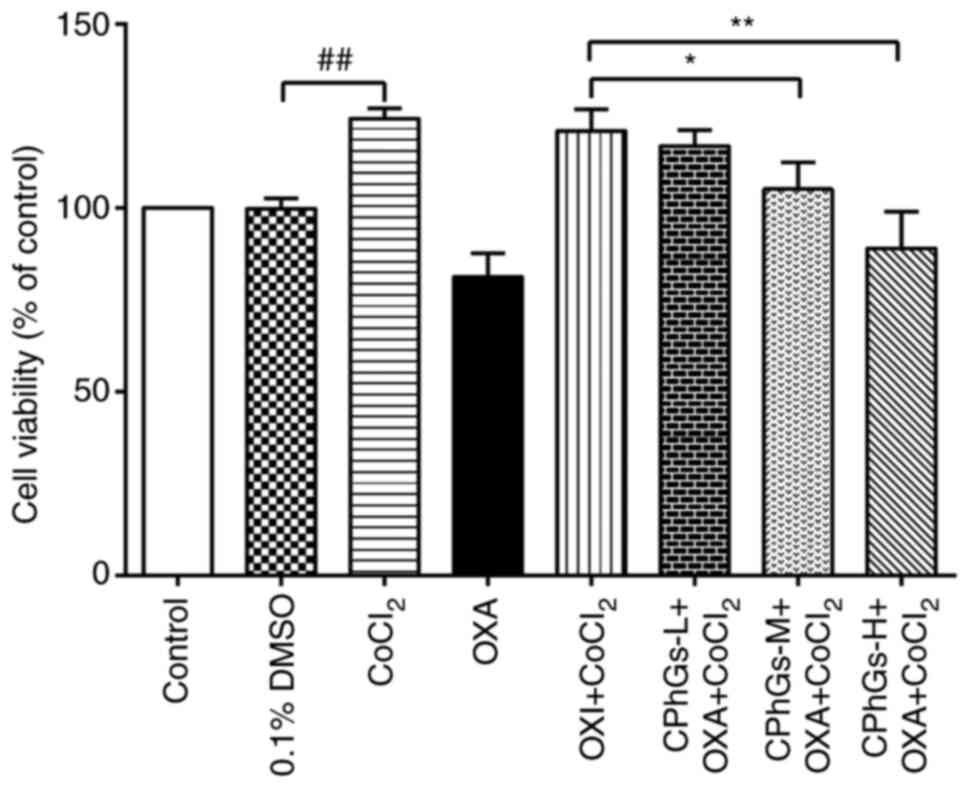
The effects of CPhGs on OXA-modulated HepG2 cell
viability were subsequently assessed (Fig. 2). After ~48 h, the combination of
CPhGs and OXA significantly decreased the viability of HepG2 cells
compared with that in the OXA + CoCl2 group.
Specifically, CPhGs-M + OXA + CoCl2 and CPhGs-H + OXA +
CoCl2 significantly inhibited the viability of HepG2
cells compared with in the OXA + CoCl2 group (P<0.05
and P<0.01, respectively).
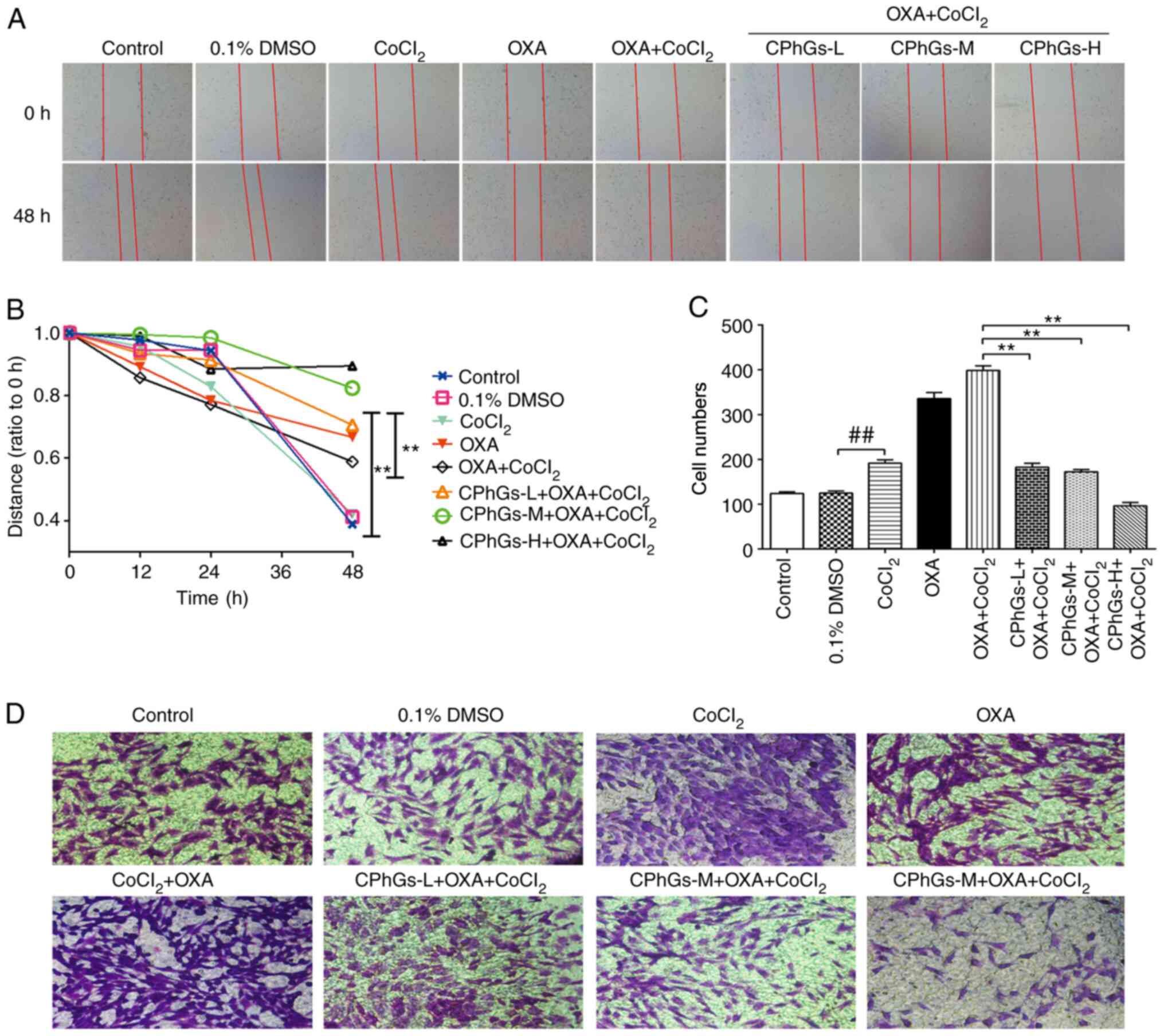
CPhGs inhibit migration and invasion
of liver cancer cells
To further study the invasive potential and
migratory ability of liver cancer cells following treatment with
CPhGs and OXA, wound-healing and Transwell assays were performed.
The wound-healing assay indicated that, compared with that in the
DMSO group, the migration of HepG2 cells was reduced following
treatment with CoCl2, OXA and CPhGs (200 or 500 µg/ml)
(P<0.05; Fig. 3A and B). For the
Transwell assay, the invasive ability of cells was markedly
inhibited in the co-treatment groups (CPhGs + OXA +
CoCl2) compared with that in the OXA + CoCl2
group (P<0.01; Fig. 3C and D).
Conversely, in the co-treatment groups (CPhGs + OXA +
CoCl2), the invasive potential and migratory ability of
cells was markedly inhibited.
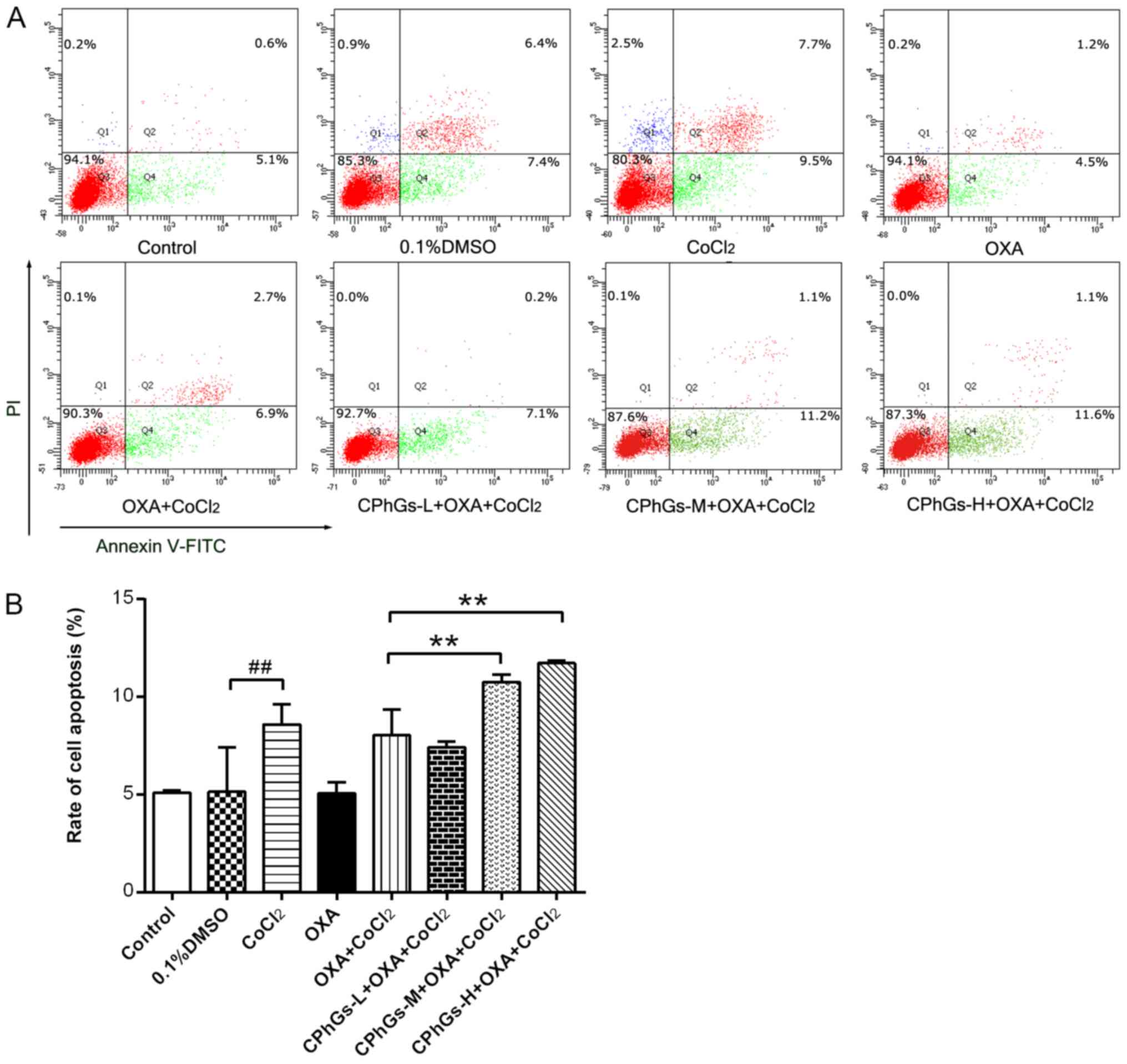
Effects of CPhGs and OXA on
apoptosis
Following treatment with the combination of CPhGs
and OXA for 48 h, the HepG2 cells were stained with Annexin V-FITC
and PI, followed by flow cytometry to determine cellular apoptosis.
As shown in Fig. 4A, the
co-treatment groups (CPhGs-L/M/H + OXA + CoCl2)
exhibited a gradual elevation in the proportion of apoptotic cells
with the increase in CPhGs concentration. Most of the cells treated
with CPhGs and OXA were localized in the Q4 region, which indicated
that the combination of CPhGs and OXA induced apoptosis at the
early stage. Compared with in the OXA + CoCl2 group, a
significant elevation in the apoptotic rate of cells was detected
in the groups treated with OXA, CoCl2 and moderate or
high doses of CPhGs (P<0.01; Fig.
4B). These findings indicated that the combination of OXA and
CPhGs may contribute to the apoptosis of HepG2 cells.
mRNA expression levels of HIF-1α,
LOXL2, E-cadherin and Twist following CPhGs and OXA
co-incubation
There were no statistical differences in the mRNA
expression levels of HIF-1α, LOXL2, E-cadherin and Twist between
the control and DMSO groups (P>0.05; Fig. 5A-D). Conversely, CoCl2
induced a significant increase in the mRNA expression levels of
LOXL2, HIF-1α and Twist compared with those in the DMSO group
(P<0.01; Fig. 5A, B and D).
Compared with those in the OXA + CoCl2 group, the mRNA
expression levels of LOXL2, HIF-1α and Twist were significantly
enhanced in the CPhGs-H + OXA + CoCl2 groups (P<0.01;
Fig. 5A, B and D). By contrast,
CoCl2 induced a significant downregulation in the mRNA
expression levels of E-cadherin compared with those in the DMSO
group (P<0.01; Fig. 5C). All
concentrations of CPhGs combined with OXA and CoCl2 were
able to upregulate the mRNA expression levels of E-cadherin
compared with those in the OXA + CoCl2 group (P<0.01;
Fig. 5C). These results indicated
that the combination of CPhGs and OXA effectively inhibited the EMT
under hypoxic conditions.
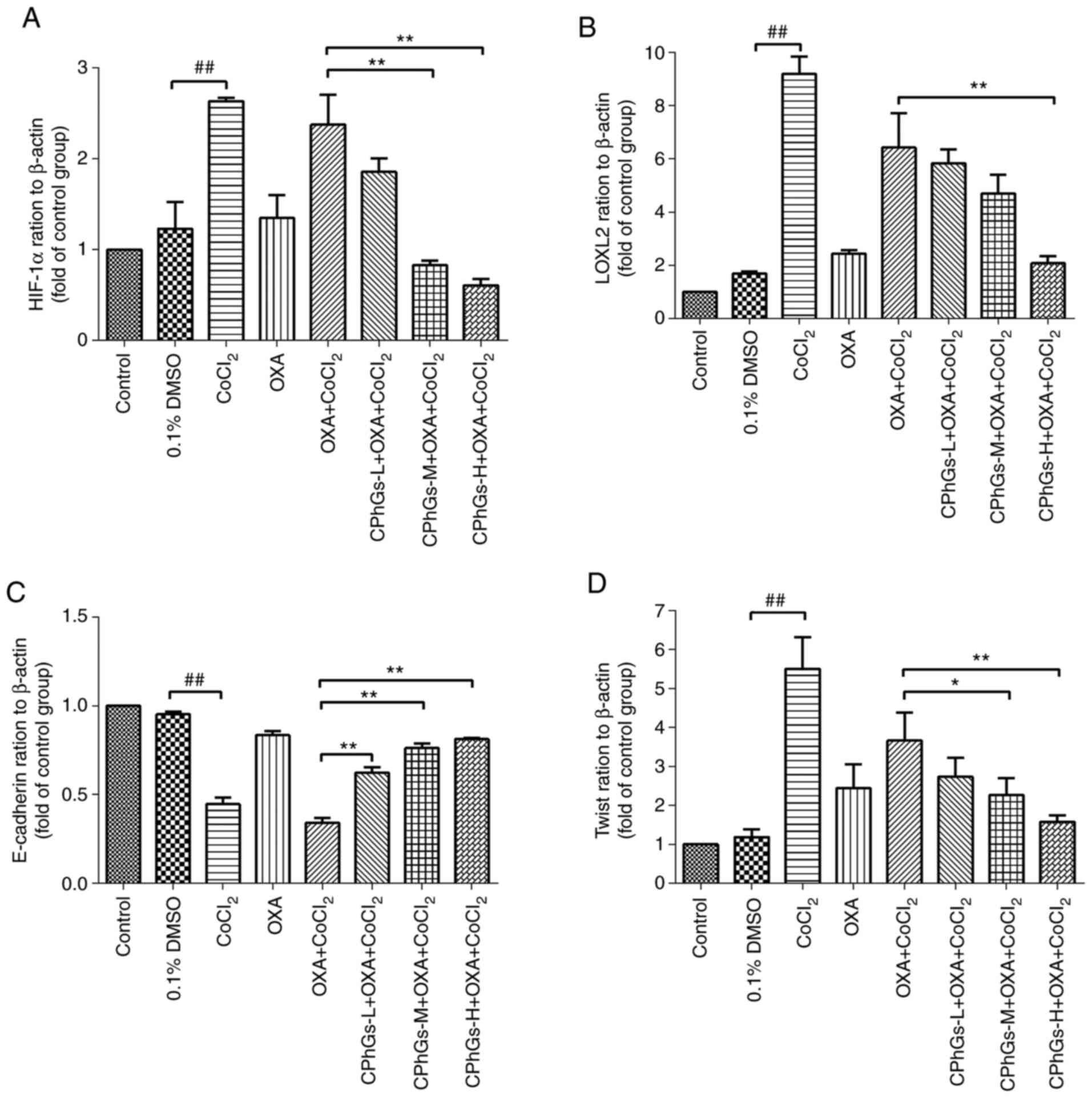 | Figure 5.Effect of CPhGs combined with OXA on
the mRNA expression levels of HIF-1α, LOXL2, E-cadherin and Twist
in HepG2 cells. Effect of CPhGs combined with OXA on (A) HIF-1α,
(B) LOXL2, (C) E-cadherin and (D) Twist gene expression in HepG2
cells. ##P<0.01; *P<0.05, **P<0.05. CPhGs,
phenylethanol glycosides from Herba Cistanche; H, high; HIF-1α,
hypoxia-inducible factor 1α; L, low; LOXL2, lysyl oxidase-like 2;
M, moderate; OXA, oxaliplatin. |
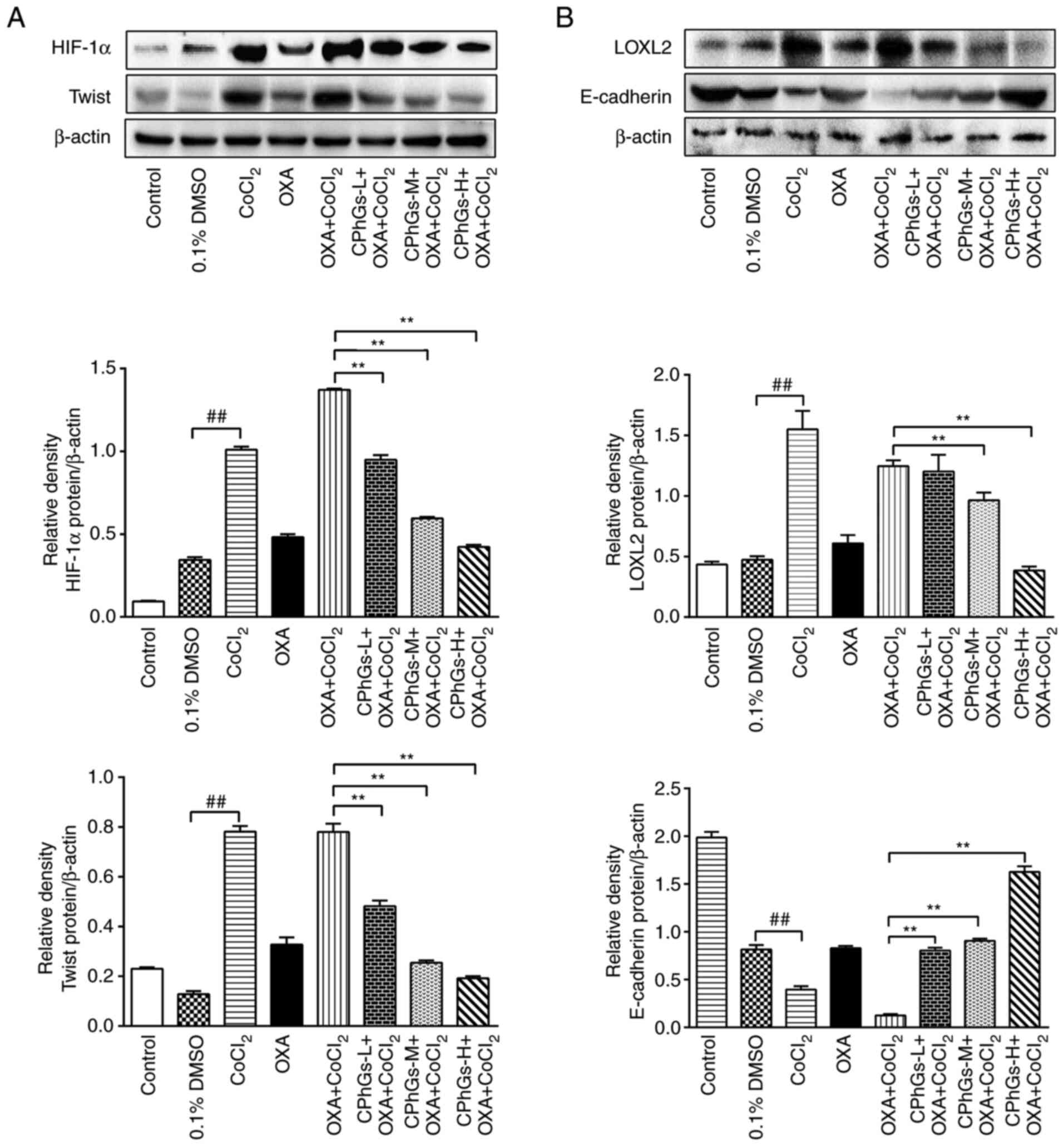
Protein expression levels of HIF-1α,
LOXL2, E-cadherin and Twist following CPhGs and OXA
co-incubation
The results of western blotting revealed that the
protein expression levels of HIF-1α, LOXL2 and Twist were
upregulated under hypoxic conditions compared with those in the
DMSO group. By contrast, the protein expression levels of
E-cadherin were downregulated under hypoxic conditions (P<0.01;
Fig. 6A and B). Notably, the
protein expression levels of HIF-1α, LOXL2 and Twist were
significantly decreased in the CPhGs-M + OXA + CoCl2 or
CPhGs-H + OXA + CoCl2 groups compared with those in the
OXA + CoCl2 group (P<0.01; Fig. 6A and B). Compared with DMSO group,
CoCl2 treatment significantly decreased the expression
level of E-cadherin. In the CPhGs groups, the protein expression
levels of E-cadherin were significantly increased compared with
those in the OXA + CoCl2 group (P<0.01; Fig. 6B). These findings indicated that
CPhGs treatment could effectively inhibit the downregulation of
E-cadherin, and the upregulation of HIF-1α, LOXL2 and Twist induced
by CoCl2.
Discussion
Hypoxia is a common feature in the cancer
microenvironment; this is mainly associated with the fact that
proliferation of cancer cells is more rapid compared with vascular
formation of aberrant neovessels. In addition, other biological
processes, including proliferation, metastasis and drug sensitivity
are affected by hypoxia (21). The
tumor hypoxic microenvironment is crucial for the pathogenesis and
progression of cancer, and it is also important in the drug
resistance and vascularization of liver cancer (22).
In the present study, HepG2 cells were treated with
various concentrations of CPhGs, among which CPhGs (200 µg/ml)
could significantly induce a decrease in cell viability. Notably,
CPhGs could modulate cellular viability in a dose-dependent manner.
Under hypoxic conditions, the combination of OXA and CPhGs (50 or
100 µg/ml) significantly inhibited the viability of HepG2 cells
compared with OXA treatment alone. A similar dose-dependent trend
was observed in the migration and invasion assays of liver cancer
cells. Currently, extensive studies have been conducted to
investigate the roles of cancer cell apoptosis in the pathogenesis
of liver disease (23–26). Several strategies have been
developed for treating liver cancer by promoting apoptosis
(27–29); therefore, interference in HepG2 cell
apoptosis may serve as a promising candidate for the prevention and
treatment of liver cancer. The apoptosis of HepG2 cells was
significantly enhanced following treatment with the combination of
CPhGs-M/-H and OXA compared with that in cells treated with OXA
alone. Therefore, it was indicated that CPhGs could significantly
enhance the antitumor affects of OXA.
HIF-1α can upregulate the expression levels of
E-cadherin, N-cadherin and Vimentin, as well as some transcription
factors, such as Snail1/2, Zeb1 and Twist1. Subsequently, this
might lead to a loss of cellular polarity, loosening of cell-cell
junctions, alterations in cytoskeletal protein, and the migration
and invasion of cancer cells, which could result in the
translocation of cancer cells to the circulatory system through the
basilar membrane and subsequent metastasis (30). In the present study,
CoCl2 was used to induce a model of hypoxia, which
triggered an increase in the viability of HepG2 cells, as well as
cell migration and invasion. Furthermore, the mRNA and protein
expression levels of HIF-1α were significantly increased,
indicating that CoCl2 induced the generation of a
hypoxic microenvironment. Treatment with the combination of CPhGs
and OXA markedly inhibited the mRNA and protein expression levels
of HIF-1α induced by hypoxia. These findings suggested that CPhGs
could attenuate the microenvironment of liver cancer in a
dose-dependent manner.
E-cadherin is a Ca2+-dependent adhesion
molecule, which has a key role in cell-cell adhesion, maintenance
of integrity of tissue structure and signaling transmission. In
cases of downregulation or even loss of adhesion function, cancer
cells may exhibit uncontrolled proliferation and dedifferentiation,
which may promote increased invasion of cancer cells and subsequent
metastasis (31). In addition,
E-cadherin is crucial for inhibiting the EMT of cancer cells, which
is closely associated with the differentiation, invasion,
metastasis and prognosis of multiple epithelial malignancies.
EMT-inducing transcription factors (EMT-TFs), such as Twist, Snail
and Zeb, are crucial for EMT. Hypoxia has been reported to activate
signaling pathways that induce EMT-TF expression; notably, it could
directly promote EMT via the transcriptional activation of these
factors (32). Twist is a highly
conserved helix-ring-helix transcription factor that has been newly
identified in recent years. High Twist expression has been detected
in numerous types of cancer cells (33). Therefore, it is essential to
investigate the association between Twist expression and the
migration or metastasis of cancer cells, as well as clinical
prevention and treatment of metastasis (34). In the present study, it was revealed
that the mRNA and protein expression levels of E-cadherin were
downregulated in the presence of hypoxia, whereas the mRNA and
protein expression levels of Twist were elevated. These findings
were consistent with the results of invasion and migration assays,
which implied that hypoxia may contribute to the occurrence of EMT.
Following treatment with OXA, the protein expression levels of
E-cadherin were downregulated; this indicated that OXA exhibited
poor efficiency in inhibiting the growth of liver cancer cells,
whereas it could promote the EMT. However, in combination with
CPhGs, the sensitivity of HepG2 cells to OXA exhibited marked
improvement in the presence of hypoxia. Furthermore, cotreatment
with CPhGs and OXA could inhibit cellular viability, migration and
invasion of HepG2 cells. The present study only detected Twist and
E-cadherin expression; therefore, future studies aim to focus on
more EMT-related markers, in order to evaluate the inhibitory
effect of CPhGs on hypoxia-induced EMT in liver cancer.
HIF-1α has been reported to promote the expression
of LOXL2, and to enhance the migration and invasion of hepatic
cancer cells, which may be closely related to the poor prognosis of
liver cancer (30). In a previous
study, the expression levels of LOXL2 in adjacent liver cancer
tissues were markedly increased compared with those in the cancer
tissues (35). In addition, it was
closely related to the invasion and metastasis of liver cancer.
LOXL2 gene silencing by small interfering RNA inhibited the
proliferation of HepG2 and SMCC-7721 cells, which resulted in cell
cycle arrest of cancer cells and increased apoptosis (36). Shao et al (35) investigated the correlation between
LOXL2 in liver cancer samples, and clinicopathological factors, VM
and prognosis among 201 cases that received surgery for treatment.
It was hypothesized that LOXL2 served important roles in the
pathogenesis and progression of liver cancer, which may serve as a
target for drug development. Furthermore, Peng et al
(37) demonstrated that LOXL2 could
activate the Snail/E-cadherin and Src kinase/Focal adhesion kinase
signaling pathways, which may contribute to the pathogenesis and
progression of EMT of gastric cancer cells. In the present study,
under hypoxic conditions, the mRNA and protein expression levels of
LOXL2 were increased, whereas its expression was downregulated
following treatment with OXA. Furthermore, treatment with a
combination of CPhGs and OXA resulted in obvious downregulation of
LOXL2 expression, which may effectively aid the antitumor effects
of OXA on liver cancer.
There are some limitations to the present study. The
present study should have used two more liver cancer cell lines,
including the SMCC-7721 cell line, but these could not be used as
it was not possible to purchase these cell lines because they were
misidentified and were derived from HeLa cells. In addition, the
present study did not analyze the antioxidant effects of different
concentrations of CPhGs in the cells.
In conclusion, CPhGs could alternate the hypoxic
tumor microenvironment of liver cancer cells through modulating the
HIF-1α signaling pathway. In addition, the sensitivity of liver
cancer cells to OXA was significantly elevated in response to
treatment with a combination of CPhGs and OXA. These findings may
provide a novel treatment strategy to improve the sensitivity of
liver cancer to chemotherapy.
Acknowledgements
Not applicable.
Funding
The present study was supported by the Xinjiang Key
Laboratory of Natural Drug Active Components and Drug Release
Technology (grant no. XJDX1713), the Reserve Candidate Project for
the Leader of Scientific and Technological Innovation in Xinjiang
Uygur Autonomous Region (grant no. 2019XS14), the National Natural
Science Foundation of China (grant no. 81860735) and the Bethune
Charitable Foundation ‘Bethune·Quest-construction of pharmaceutical
scientific research capacity’ (grant no. B-19-H-20200622).
Availability of data and materials
The datasets used and/or analyzed during the current
study are available from the corresponding author on reasonable
request.
Authors' contributions
LMW and JWZ performed the experiments, drafted the
manuscript and confirmed the authenticity of all the raw data. JPH
and JHY designed the present study. All authors read and approved
the final manuscript.
Ethics approval and consent to
participate
Not applicable.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Hepatocellular carcinoma. Nat Rev Dis
Primers. 2:160192016. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Gingold JA, Zhu D, Lee DF, Kaseb A and
Chen J: Genomic profiling and metabolic homeostasis in primary
liver cancers. Trends Mol Med. 24:395–411. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
McGuire S: World cancer report 2014.
Geneva, Switzerland: World health organization, international
agency for research on cancer, WHO press, 2015. Adv Nutr.
7:418–419. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Gholamreza K, Jadidi-Niaragh F, Jahromi
AS, Zandi K and Hojjat-Farsangi M: Mechanisms of tumor cell
resistance to the current targeted-therapy agents. Tumour Biol.
37:10021–10039. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Dong X and Mumper RJ: Nanomedicinal
strategies to treat multidrug-resistant tumors: Current progress.
Nanomedicine (Lond). 5:597–615. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Xie Y and Zhong DW: AEG-1 is associated
with hypoxia-induced hepatocellular carcinoma chemoresistance via
regulating PI3K/AKT/HIF-1alpha/MDR-1 pathway. EXCLI J. 15:745–757.
2016.PubMed/NCBI
|
|
7
|
Xiong H, Ni Z, He J, Jiang S, Li X, He J,
Gong W, Zheng L, Chen S, Li B, et al: LncRNA HULC triggers
autophagy via stabilizing Sirt1 and attenuates the chemosensitivity
of HCC cells. Oncogene. 36:3528–3540. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Gade TPF, Tucker E, Nakazawa MS, Hunt SJ,
Wong W, Krock B, Weber CN, Nadolski GJ, Clark TWI, Soulen MC, et
al: Ischemia induces quiescence and autophagy dependence in
hepatocellular carcinoma. Radiology. 283:702–710. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Siegel RL, Miller KD and Jemal A: Cancer
statistics, 2017. CA Cancer J Clin. 67:7–30. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Dong LQ, Shen BQ and Ma Y: Research
progress of hypoxia microenvironment in hepatocellular carcinoma.
Zhong Guo Pu Wai Ji Chu Yu Lin Chuang Za Zhi. 25:1254–1258.
2018.(In Chinese).
|
|
11
|
Moon HJ, Finney J, Ronnebaum T and Mure M:
Human lysyl oxidase-like 2. Bioorg Chem. 57:231–241. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Ferreira S, Saraiva N, Rijo P and
Fernandes AS: LOXL2 inhibitors and breast cancer progression.
Antioxidants (Basel). 10:3122021. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Philp CJ, Siebeke I, Clements D, Miller S,
Habgood A, John AE, Navaratnam V, Hubbard RB, Jenkins G and Johnson
SR: Extracellular matrix cross-linking enhances fibroblast growth
and protects against matrix proteolysis in lung fibrosis. Am J
Respir Cell Mol Biol. 58:594–603. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Galván JA, Zlobec I, Wartenberg M, Lugli
A, Gloor B, Perren A and Karamitopoulou E: Expression of E-cadherin
repressors SNAIL, ZEB1 and ZEB2 by tumour and stromal cells
influences tumour-budding phenotype and suggests heterogeneity of
stromal cells in pancreatic cancer. Br J Cancer. 112:1944–1950.
2015. View Article : Google Scholar
|
|
15
|
Gu C, Yang X and Huang L: Cistanches
herba: A neuropharmacology review. Front Pharmacol. 7:2892016.
View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Fu Z, Fan X, Wang X and Gao X: Cistanches
Herba: An overview of its chemistry, pharmacology, and
pharmacokinetics property. J Ethnopharmacol. 219:233–247. 2018.
View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Hu Q, You SP, Liu T, Wang B, Liu X and
Jiang Y: An investigation on the anti-liver cancer effect of
cistanche. Carcinog Teratog Mutagen. 30:194–199. 2018.
|
|
18
|
Mao J, Tian Y, Wang C, Jiang K, Li R, Yao
Y, Zhang R, Sun D, Liang R, Gao Z, et al: CBX2 regulates
proliferation and apoptosis via the phosphorylation of YAP in
hepatocellular carcinoma. J Cancer. 10:2706–2719. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Qin Y, Liu HJ, Li M, Zhai DH, Tang YH,
Yang L, Qiao KL, Yang JH, Zhong WL, Zhang Q, et al: Salidroside
improves the hypoxic tumor microenvironment and reverses the drug
resistance of platinum drugs via HIF-1α signaling pathway.
EBioMedicine. 38:25–36. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) method. Methods. 25:402–408. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Vaupel P: Tumor microenvironmental
physiology and its implications for radiation oncology. Semin
Radiat Oncol. 14:198–206. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Chen C and Lou T: Hypoxia inducible
factors in hepatocellular carcinoma. Oncotarget. 8:46691–46703.
2017. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Schwabe RF and Luedde T: Apoptosis and
necroptosis in the liver: A matter of life and death. Nat Rev
Gastroenterol Hepatol. 15:738–752. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Kanda T, Matsuoka S, Yamazaki M, Shibata
T, Nirei K, Takahashi H, Kaneko T, Fujisawa M, Higuchi T, Nakamura
H, et al: Apoptosis and non-alcoholic fatty liver diseases. World J
Gastroenterol. 24:2661–2672. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Pittala S, Krelin Y and Shoshan-Barmatz V:
Targeting liver cancer and associated pathologies in mice with a
mitochondrial VDAC1-based Peptide. Neoplasia. 20:594–609. 2018.
View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Jing ZT, Liu W, Xue CR, Wu SX, Chen WN,
Lin XJ and Lin X: AKT activator SC79 protects hepatocytes from
TNF-α-mediated apoptosis and alleviates d-Gal/LPS-induced liver
injury. Am J Physiol Gastrointest Liver Physiol. 316:G387–G396.
2019. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Zhu YJ, Zheng B, Wang HY and Chen L: New
knowledge of the mechanisms of sorafenib resistance in liver
cancer. Acta Pharmacol Sin. 38:614–622. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Wei R, Cao J and Yao S: Matrine promotes
liver cancer cell apoptosis by inhibiting mitophagy and
PINK1/Parkin pathways. Cell Stress Chaperones. 23:1295–1309. 2018.
View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Shen L and Zhang G, Lou Z, Xu G and Zhang
G: Cryptotanshinone enhances the effect of arsenic trioxide in
treating liver cancer cell by inducing apoptosis through
downregulating phosphorylated-STAT3 in vitro and in vivo. BMC
Complement Altern Med. 17:1062017. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Wu F, Zhang J, Liu Y, Zheng Y and Hu N:
HIF1α genetic variants and protein expressions determine the
response to platinum based chemotherapy and clinical outcome in
patients with advanced NSCLC. Cell Physiol Biochem. 32:1566–1576.
2013. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Heerboth S, Housman G, Leary M, Longacre
M, Byler S, Lapinska K, Willbanks A and Sarkar S: EMT and tumor
metastasis. Clin Transl Med. 4:62015. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Hapke RY and Haake SM: Hypoxia-induced
epithelial to mesenchymal transition in cancer. Cancer Lett.
487:10–20. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Zhao Z, Rahman MA, Chen ZG and Shin DM:
Multiple biological functions of Twist1 in various cancers.
Oncotarget. 8:20380–20393. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Goossens S, Vandamme N, Van Vlierberghe P
and Berx G: EMT transcription factors in cancer development
re-evaluated: Beyond EMT and MET. Biochim Biophys Acta Rev Cancer.
1868:584–591. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Shao B, Zhao X, Liu T, Zhang Y, Sun R,
Dong X, Liu F, Zhao N, Zhang D, Wu L, et al: LOXL2 promotes
vasculogenic mimicry and tumour aggressiveness in hepatocellular
carcinoma. J Cell Mol Med. 23:1363–1374. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Wu L, Zhang Y, Zhu Y, Cong Q, Xiang Y and
Fu L: The effect of LOXL2 in hepatocellular carcinoma. Mol Med Rep.
14:1923–1932. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Peng L, Ran YL, Hu H, Yu L, Liu Q, Zhou Z,
Sun YM, Sun LC, Pan J, Sun LX, et al: Secreted LOXL2 is a novel
therapeutic target that promotes gastric cancer metastasis via the
Src/FAK pathway. Carcinogenesis. 30:1660–1669. 2009. View Article : Google Scholar : PubMed/NCBI
|