Introduction
Uric acid (UA) is a product of mammalian purine
metabolism. UA is synthesized in the liver and ~70% is secreted
into kidney tubules; notably, the liver and kidney are the
principal organs responsible for the regulation of serum UA (SUA)
concentration (1). High SUA
concentrations are associated with cardiovascular and
cerebrovascular diseases, metabolic syndrome and renal
insufficiency (2). The prevalence
of hyperuricemia (HUA) and its complications have increased
worldwide in recent decades (3).
The levels of UA in serum are partly regulated by
urate transport-related proteins; among them, urate transporter 1
(URAT1) and glucose transporter 9 (GLUT9) serve an important role
in renal UA reabsorption (4,5). It
has been reported that the expression of GLUT9 and URAT1 is related
to renal inflammation in an animal model of fructose-induced HUA
(6). In the inflammation system,
Toll-like receptor 4 (TLR4) and NOD-like receptor family, pyrin
domain-containing 3 (NLRP3) serve an important role in regulating a
cascade of events in the body (7–9).
Resveratrol (RSV) is a polyphenol, non-flavonoid
antitoxin that is abundant in plant materials, including grapes,
peanuts and Polygonum cuspidatum. RSV has been reported to exert
anti-inflammatory, anti-oxidative, anti-tumor and
anti-cardiovascular disease effects, to inhibit lipid peroxidation
and to extend life in mice (10).
Effects of RSV on amelioration of IR, and liver and kidney
pathologies have been shown in several animal models (11,12).
The present study investigated the effects and
mechanisms of RSV on high-fat diet (HFD)-induced HUA and renal
inflammation in mice. The present study aimed to verify whether RSV
could affect the expression of GLUT9 and URAT1 in the kidney.
Additionally, whether the effects of RSV were associated with
suppression of the TLR4/myeloid differentiation factor 88
(MyD88)/nuclear factor-κB (NF-κB) signaling pathway and NLRP3
inflammasome were investigated.
Materials and methods
Animal model
A total of 30 6-week-old male C57BL/6J mice (weight,
21.0–23.0 g) were purchased from Beijing Weitong Lihua Experimental
Animal Center [license no. SCXK (Beijing, China) 2016-0006] and
housed in the Clinical Medical Research Center of Hebei General
Hospital. The room temperature was controlled at 23–25°C and the
relative humidity was ~60%; the mice were maintained under a 12-h
light/dark cycle and had free access to food and water. The present
study was approved by the Ethics Committee of Hebei General
Hospital Ethical Committee (approval no. 2019E367; Shijiazhuang,
China) and was conducted in accordance with the international rules
for the use of experimental animals.
After 1 week of adaptation, the mice were allocated
to two groups: A control group (Con, n=10), which was fed a
standard diet (D12450J: 20% protein, 70% carbohydrate, 10% fat,
3.85 kcal/g); and a HFD group (n=20), which was fed a HFD (D12492:
20% protein, 20% carbohydrate, 60% fat, 5.24 kcal/g). Both diets
were purchased from Beijing Huafukang Biotechnology Co., Ltd. The
body mass and food intake of the mice were recorded weekly during
the study. After 12 weeks, intraperitoneal glucose tolerance
testing (IPGTT) (13) was performed
after 12 h of fasting, and the area under the curve (AUC) was
calculated to confirm the development of glucose intolerance in the
HFD group. AUC is calculated using the approximate trapezoidal area
(The side length is the blood glucose value at different time
points, the height is the time, and the AUC value is the sum of the
four trapezoidal areas under the curve. AUC does not have a
specific value that proves impaired glucose tolerance. It is based
on the difference between the groups to compare the abnormalities
of glucose tolerance.) Blood was taken from the tail vein at 0, 15,
30, 60 and 120 min to test glucose levels by an Accuchek Active
Meter (ACCU-CHEK Roche; Roche Diagnostics GmbH).A total of 10 mice
from the HFD group were then randomly selected to form a HFD + RSV
group, which was administered 100 mg/kg RSV (Sigma-Aldrich; Merck
KGaA) (14). The drug concentration
of RSV intervention was obtained according to the reference
(10). Our research group explored
the concentration of RSV in a previous study (15), and 100 mg/kg RSV was revealed to
exert beneficial effect on insulin sensitivity without toxicity.
RSV was administered daily by gavage for 6 weeks, and the remaining
mice in the HFD group and those in the Con group were administered
vehicle (0.9% sodium chloride solution containing 0.1% DMSO) by
gavage over the same time period.
At the end of this period, the mice were fasted for
12 h overnight, euthanized by cervical dislocation and blood was
collected by cardiac puncture. The right kidneys were quickly
removed and rinsed with pre-cooled 0.9% sodium chloride solution. A
small piece of each kidney was fixed in 4% paraformaldehyde (6 h;
4°C), in addition, small pieces of kidney (1×1×2 mm) were fixed in
transmission electron microscopy (TEM) fixing solution (TEM
fixative; Wuhan Servicebio Technology Co., Ltd.), and the remaining
tissue was frozen in liquid nitrogen for western blotting or ELISA
and then stored at −80°C.
Measurement of the serum
concentrations of key substances
The serum concentrations of triglyceride (TG; cat.
no. A110-1-1), total cholesterol (TC; cat. no. A111-1-1),
high-density lipoprotein-cholesterol (HDL-C; cat. no. A112-1-1) and
low-density lipoprotein-cholesterol (LDL-C; cat. no. A113-1-1) were
measured using kits from the Nanjing Jiancheng Bioengineering
Institute according to manufacturer's protocol. SUA was measured
using kit from Beijing Solarbio Science & Technology Co., Ltd.
(cat. no. BC1365) and insulin concentrations were determined using
ELISA kits from Alpco (cat. no. 80-INSMSU-E01) according to
manufacturer's protocol. The Qualitative Insulin Sensitivity Check
Index (QUICKI) was determined according to the equation: QUICKI =
1/[log(I0) + log(G0)]) (16), in which I0 is fasting
insulin and G0 is fasting glucose.
Histological assessment of kidney
tissue
The kidney samples fixed in 4% paraformaldehyde (6
h; 4°C) were dehydrated using an alcohol gradient, clarified using
xylene, embedded in paraffinand sectioned at 5-µm thickness.
Sections were then stained with H&E (Beijing Solarbio Science
& Technology Co., Ltd.; cat. no. G1005, hematoxylin staining
for 5 min, staining in eosin solution for 5 min, both at room
temperature), periodic acid-Schiff (PAS; Beijing Solarbio Science
& Technology Co., Ltd.; cat. no. G1008, PAS dye solution B for
10–15 min, staining with PAS A for 25–30 min in the dark, staining
with PAS C for 30 sec, all at room temperature) or Masson's
trichrome(Beijing Solarbio Science & Technology Co., Ltd.; cat.
no. G1006, Masson D for 6 min, Masson E for 1 min, both at room
temperature) according to manufacturer's protocol, and examined
under a light microscope (magnification, ×400).
Electron microscopy
Kidney samples to be used for electron microscopy
were, post-fixed in 1% osmium tetroxide, dehydrated and embedded in
Epon (1 h; 4°C) in the longitudinal orientation. After an initial
low-power screening of semithin (300 nm) sections stained with
toluidine blue to optimize the plane of sectioning, ultrathin (60
nm) longitudinal sections were cut for each sample. Briefly, the
sections were mounted on copper grids, and stained with lead
citrate and uranyl acetate (8 min; 37°C). For each biopsy, ≥10
longitudinal sections were examined by TEM (HT7800/HT7700; Hitachi
Ltd.) at an accelerating voltage of 80 kV. A minimum of 10
micrographs were taken at ×2,500 magnification.
Measurement of the renal
concentrations of interleukin (IL)-6, IL-18, IL-1β and tumor
necrosis factor-α (TNF-α)
Briefly, ~0.3 g kidney tissue was added to 2.7 ml
0.9% sodium chloride and the tissue was homogenized on ice. The
homogenate was then centrifuged at 1,500 × g at 4°C for 10 min and
the supernatant was removed. The renal concentrations of IL-6 (cat.
no. 88-7064; Thermo Fisher Scientific, Inc.), IL-18 (cat. no.
EK218-24; Multi Sciences; www.liankebio.com), IL-1β (cat. no. 88-7013; Thermo
Fisher Scientific, Inc.) and TNF-α (cat. no. 88-7324, Thermo Fisher
Scientific, Inc.) were measured using ELISA kits, according to the
manufacturer's instructions.
Western blotting
Kidney samples (~50 mg) were weighed on an
electronic balance, then pre-cooled 0.5-ml aliquots of RIPA buffer
(Beijing Solarbio Science & Technology Co., Ltd.; cat. no.
G2002) were added, and the samples were homogenized and left at 4°C
overnight. Subsequently, the homogenates were centrifuged at 4°C
and 16,200 × g for 15 min and the supernatants were collected.
Lysates containing 50 µg protein were separated by SDS-PAGE
(stacking gel, 10%; resolving gel, 5%) and wet electro-transferred
to nitrocellulose membranes, which were blocked in buffer
containing 5% skimmed milk powder at room temperature for 4 h. The
membranes were then incubated with the following primary antibodies
(dilution, 1:1,000) at 4°C overnight: Anti-GLUT9 (cat. no.
26486-1-AP), anti-URAT1 (cat. no. 14937-1-AP), anti-monocyte
chemotactic protein-1 (MCP-1; cat no. 25542-1-AP), anti-NLRP3 (cat.
no. a5652; ABclonal Biotech Co., Ltd.), anti-apoptosis-associated
speck-like protein (ASC; cat. no. 67494-1-Ig), anti-MyD88 (cat. no.
BS-1047R; BIOSS), anti-TNF receptor-associated factor 6 (TRAF6;
cat. no. 66498-1-Ig), anti-TGF-β-activated kinase 1 (TAK1; cat. no.
12330-2-AP), anti-NF-κB p65 (cat. no. 66535-1-Ig), anti-caspase-1
(cat. no. 22915-1-AP), anti-TLR4 (cat. no. 19811-1-AP) and
anti-β-actin (cat. no. 20536-1-AP) (Remaining antibodies except
anti-NLRP3 and anti-MyD8 all purchased from ProteinTech Group,
Inc.). After three washes with Tris-buffered saline-Tween-20 (20%)
solution, the membranes were incubated with horseradish
peroxidase-conjugated secondary antibodies (anti-rabbit secondary
antibody; dilution,1:8,000; cat. no. SA00001-2; ProteinTech Group,
Inc.; anti-mouse secondary antibodies; dilution, 1:8,000; cat. no.
GB23301; Beijing Solarbio Science & Technology Co., Ltd.) for 2
h at room temperature. Finally, enhanced chemiluminescence (Beijing
Solarbio Science & Technology Co., Ltd.; cat. no. G2014) and
X-ray film exposure were used to identify specific protein bands.
The optical densities of the target bands were measured using the
Alpha software processing system (AlphaEaseFC 4.0;
ProteinSimple).
Statistical analysis
All experiments are repeated three times. All data
were analyzed using SPSS 22.0 software (IBM, Inc.) and are
expressed as the mean ± SD. Normally distributed data were analyzed
using the independent sample t-test or one-way ANOVA. One-way ANOVA
was used for statistical analysis followed by Bonferroni's multiple
comparison test or Tamhane's multiple comparison test. P<0.05
was considered to indicate a statistically significant
difference.
Results
Establishment of a mouse model of
HFD-induced insulin resistance (IR)
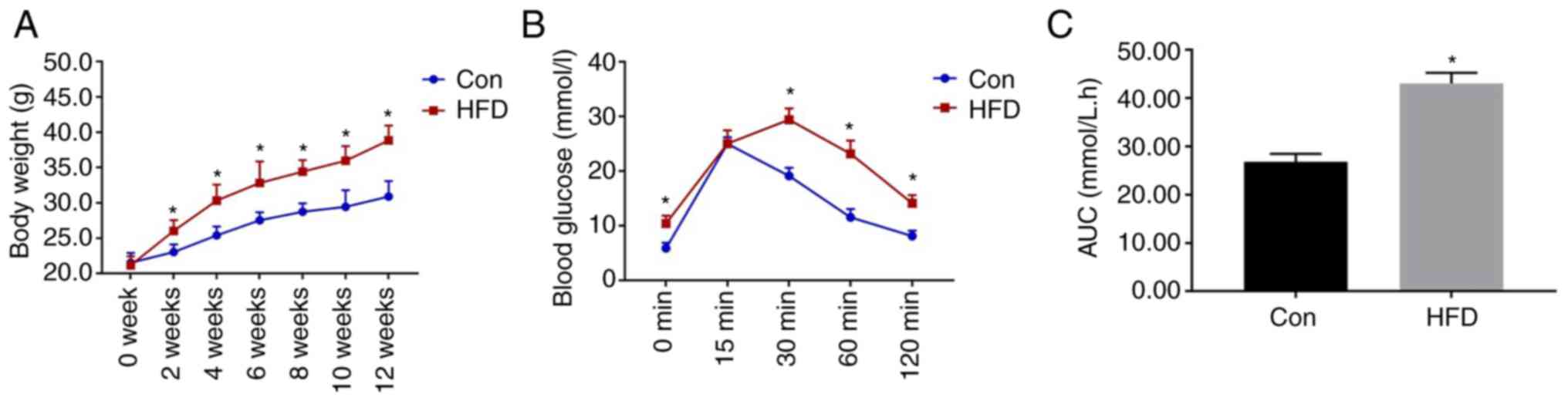
As shown in Fig. 1,
the mice that consumed the HFD weighed significantly more than
those in the Con group from week 2. IPGTT was performed after 12
weeks of diet consumption, and the results revealed that the blood
glucose concentrations of the HFD group were significantly higher
than those of the Con group at 0, 30, 60 and 120 min. In addition,
the area under the glucose curve for the HFD group was
significantly higher (Fig. 1C).
These data confirmed that glucose intolerance had been successfully
induced in the mice.
Effects of RSV administration on
general physiological parameters
The body mass was significantly higher in the HFD
group compared with the Con group at all time points, but after 4
weeks of RSV administration, the weight of mice in the HFD + RSV
group was significantly reduced compared with that of mice in the
HFD group (Fig. 2A). IPGTT was
performed in all three groups of mice at the end of the 6th week of
RSV administration. Compared with the Con group, the HFD group had
significantly higher blood glucose concentrations at the 0, 15, 30,
60 and 120-min time points. In addition, compared with the HFD
group, the HFD + RSV group had significantly lower blood glucose
concentrations at the 0, 30, 60 and 120-min time points (Fig. 2B). Furthermore, the area under the
glucose curve for the HFD group was significantly higher than that
for the Con group, but this was significantly lower for the HFD +
RSV group (Fig. 2C). These data
indicated that RSV may ameliorate the glucose intolerance of
HFD-fed mice.
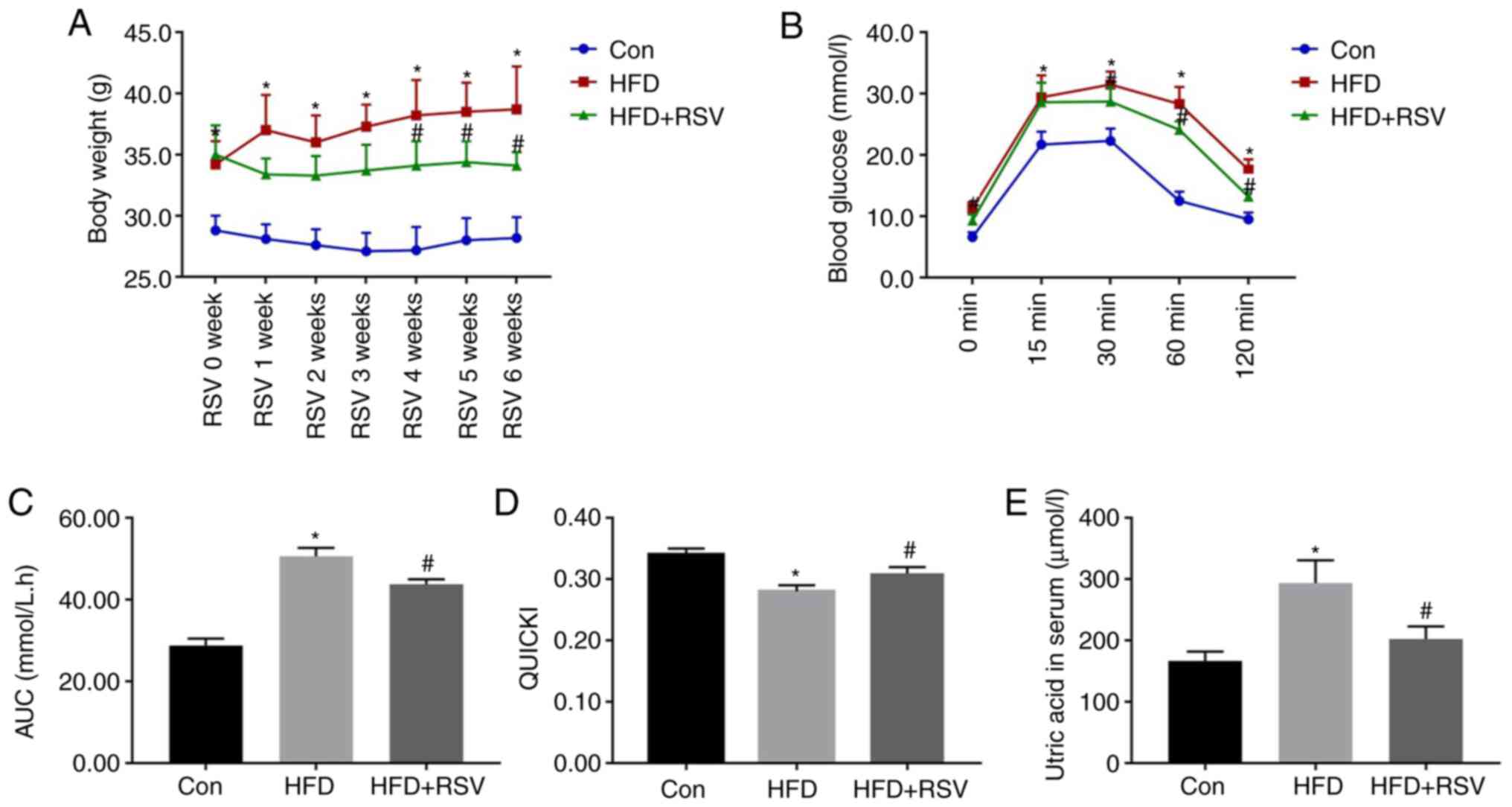 | Figure 2.Body mass, serum uric acid
concentration, glucose tolerance and insulin sensitivity after RSV
administration. (A) Body mass of Con, HFD-fed and HFD +
RSV-administered groups. (B) Blood glucose concentrations 0, 15,
30, 60 and 120 min after the intraperitoneal injection of glucose.
(C) Area under the glucose curve. (D) Quantitative insulin
sensitivity check index. (E) Serum uric acid concentrations in the
Con, HFD and HFD + RSV groups. Data are presented as the mean ± SD
(n=10). One-way ANOVA, followed by Bonferroni's post hoc, or
Tamhane's multiple comparison post hoc tests, was used for
statistical analysis. *P<0.05 vs. Con group;
#P<0.05 vs. HFD group. AUC, area under the curve;
Con, control; HFD, high-fat diet; RSV, resveratrol. |
The fasting blood glucose and insulin concentrations
of the mice in the HFD group were significantly higher than those
in the Con group, but the concentrations in the HFD + RSV group
were significantly lower than those in the HFD group (Table I). Therefore, the QUICKI value
(QUICKI = 1/[log(I0) + log(G0)]) (16) for the HFD group was significantly
lower than that for the Con group, whereas RSV administration
significantly increased this value, which indicated that RSV
ameliorated the IR of HFD-fed mice (Fig. 2D). With the improvement of IR, the
increase in blood UA caused by the HFD was also improved (Fig. 2E). In addition, the serum
concentrations of TG, TC and LDL-C were significantly higher in the
HFD group than in the Con group, whereas those of TG, and LDL-C
were significantly lower, and HDL-C was significantly higher, in
the HFD + RSV group compared with those in the HFD group (Table I).
 | Table I.Fasting circulating glucose, INS and
lipid concentrations after RSV administration. |
Table I.
Fasting circulating glucose, INS and
lipid concentrations after RSV administration.
| Variable | Con, n=10 | HFD, n=10 | HFD + RSV,
n=10 |
|---|
| FBG, mmol/l | 6.36±1.07 |
11.21±1.61a |
8.18±1.01b |
| INS, ng/ml | 0.35±0.06 |
0.82±0.18a |
0.53±0.11b |
| TC, mmol/l | 5.61±0.73 |
7.68±1.04a | 6.28±0.95 |
| TG, mmol/l | 0.51±0.08 |
1.10±0.26a |
0.75±0.11b |
| HDL-C, mmol/l | 3.60±0.52 | 3.23±0.46 |
4.00±0.36b |
| LDL-C, mmol/l | 0.26±0.05 |
0.95±0.22a |
0.55±0.13b |
Effects of RSV administration on
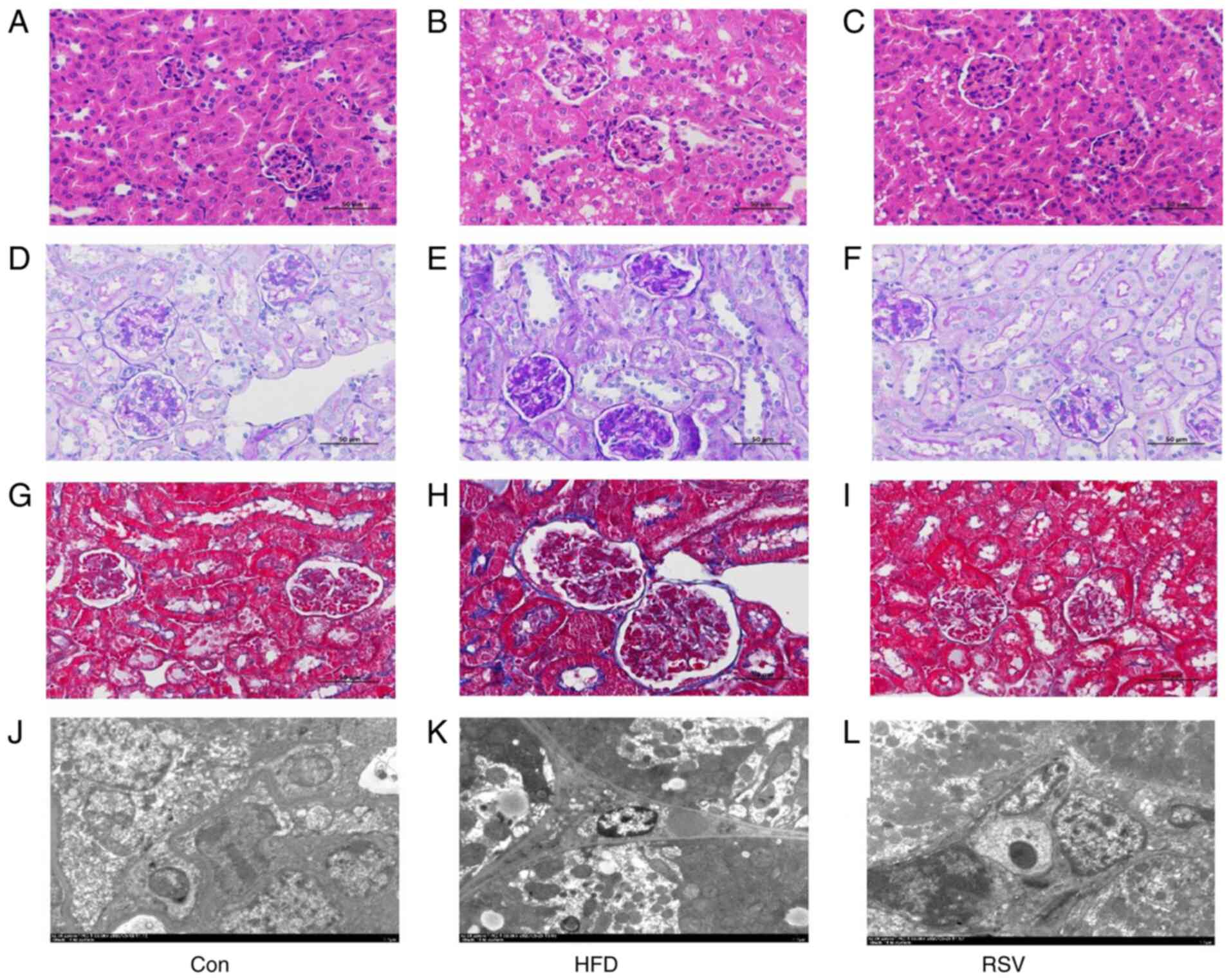
kidney histology
H&E staining revealed normal renal histology in
the Con group. However, in the HFD group, glomerular swelling was
observed and the renal tubules were atrophic and dilated (Fig. 3A and B). PAS staining revealed that
there was carbohydrate deposition in the kidneys of the HFD group
compared with that in the Con group (Fig. 3D and E), and Masson's trichrome
staining demonstrated that there was greater deposition of collagen
fibers in the renal interstitium of the HFD group compared with
that in the Con group (Fig. 3G and
H). In addition, electron microscopy revealed numerous lipid
droplets in the kidneys of mice in the HFD group compared with that
in the Con group (Fig. 3J and K).
However, RSV administration markedly ameliorated the glomerular
swelling, tubular atrophy and dilatation, renal interstitial
fibrosis, and glycogen and lipid deposition in the kidneys of
HFD-fed mice (Fig. 3C, F, I and
L).
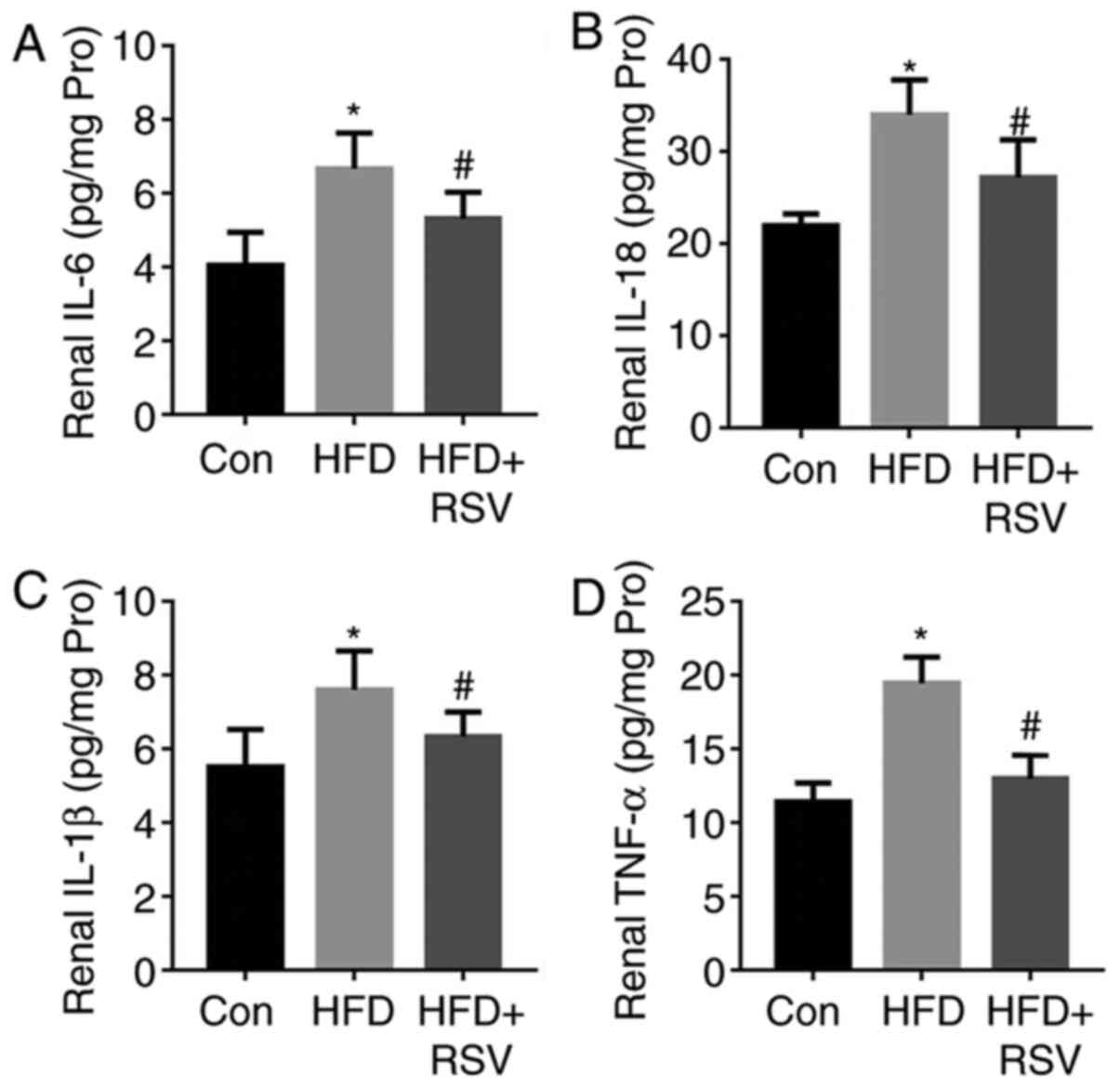
RSV administration reduces renal
inflammation in HFD-fed, insulin-resistant mice
As shown in Fig. 4,
HFD-feeding significantly increased the concentrations of the
pro-inflammatory cytokines IL-6, IL-18, IL-1β and TNF-α in the
kidneys of the mice. By contrast, RSV administration reduced their
concentrations and ameliorated renal inflammation.
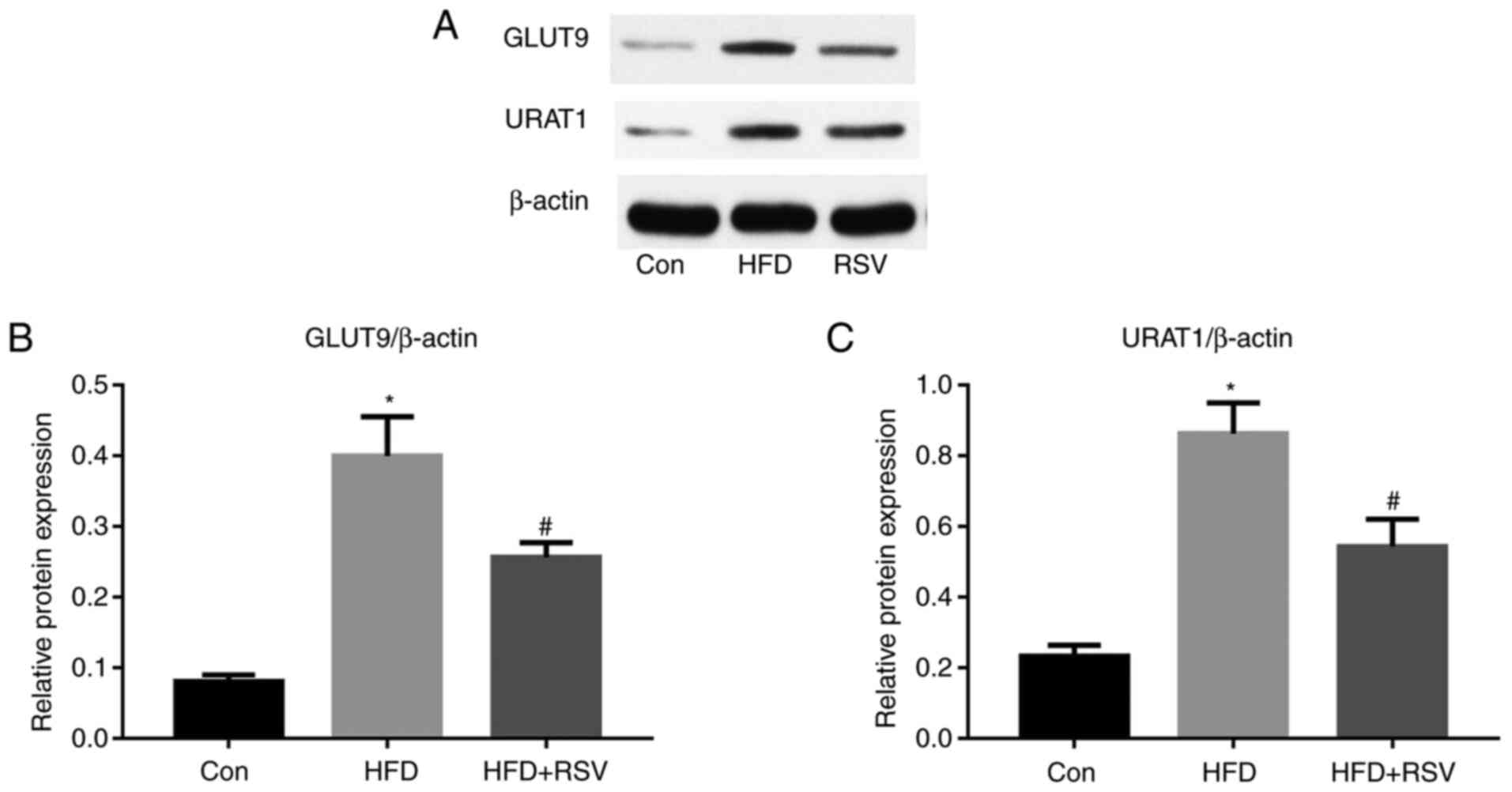
Effects of RSV on the renal expression
of GLUT9 and URATI
A previous study (6)
revealed that high-fructose diet-feeding increased the renal
expression of GLUT9 and URAT1, and SUA concentration in rats. The
present study revealed that long-term HFD-feeding significantly
increased the protein expression levels of GLUT9 and URAT1 in the
kidneys of mice (Fig. 5). However,
compared with those in the HFD group, the protein expression levels
of GLUT9 and URAT1 were significantly lower in the HFD + RSV
group.
RSV administration reduces the renal
expression of NLRP3, ASC, and caspase-1
As shown in Fig. 6,
long-term HFD-feeding significantly increased the protein
expression levels of NLRP3, ASC and caspase-1 in the kidneys,
whereas the administration of RSV reduced the expression levels of
these proteins compared with those in the HFD group. These results
indicated that RSV may induce its effects by inhibiting the
NLRP3-ASC-caspase-1 pathway.
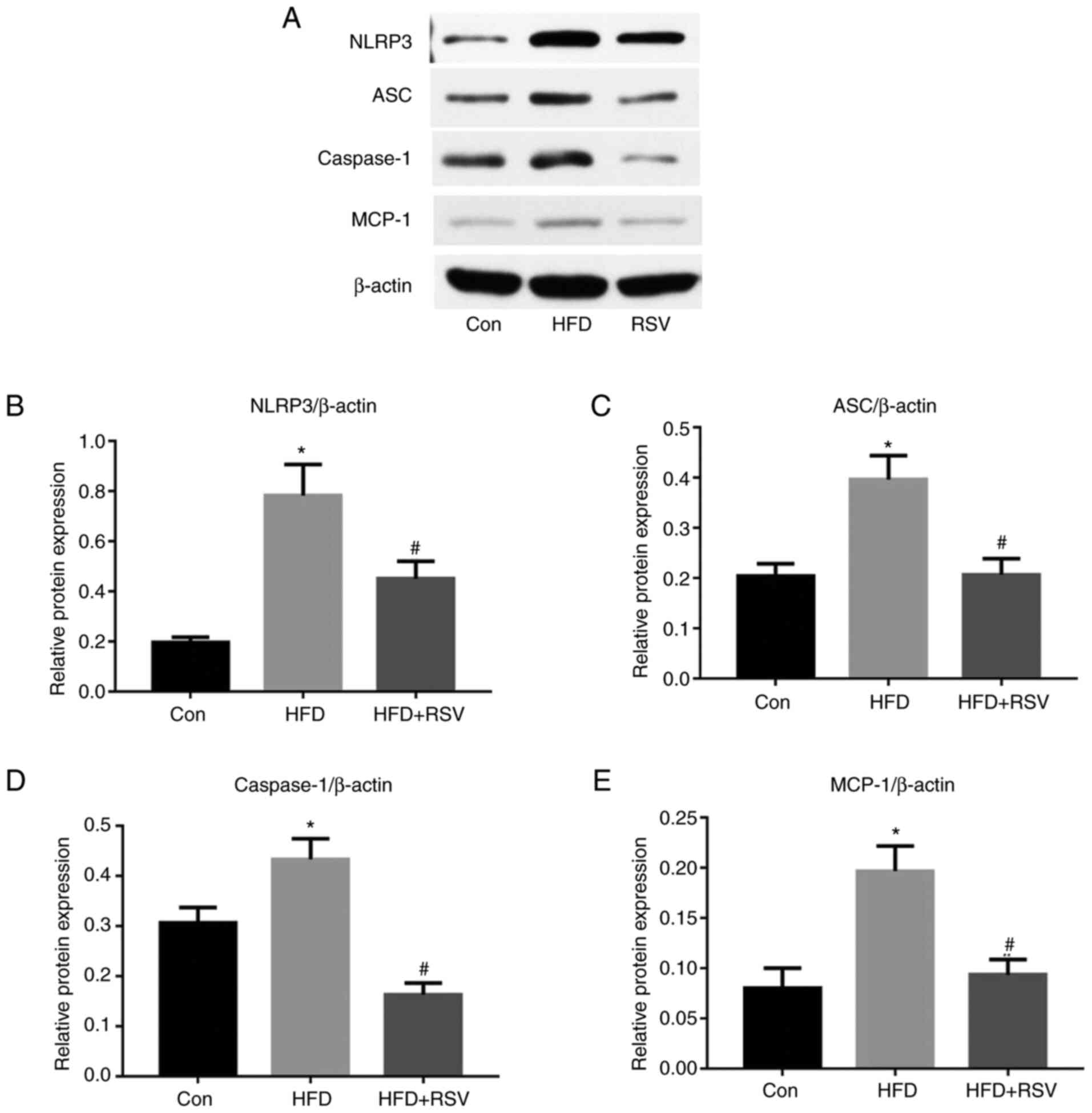 | Figure 6.Effect of RSV administration on the
protein expression levels of MCP-1, NLRP3, ASC and caspase-1 in the
kidneys of HFD-fed mice. (A) Western blot analysis of NLRP3, ASC,
caspase-1, and MCP-1 expression in the kidney. Relative protein
expression levels of (B) NLRP3, (C) ASC, (D) caspase-1 and (E)
MCP-1, normalized to β-actin. Data are presented as the mean ± SD
(n=10). *P<0.05 vs. Con group; #P<0.05 vs. HFD
group. ASC, apoptosis-associated speck-like protein; Con, control;
HFD, high-fat diet; MCP-1, monocyte chemotactic protein-1; NLRP3,
NOD-like receptor family, pyrin domain-containing 3; RSV,
resveratrol. |
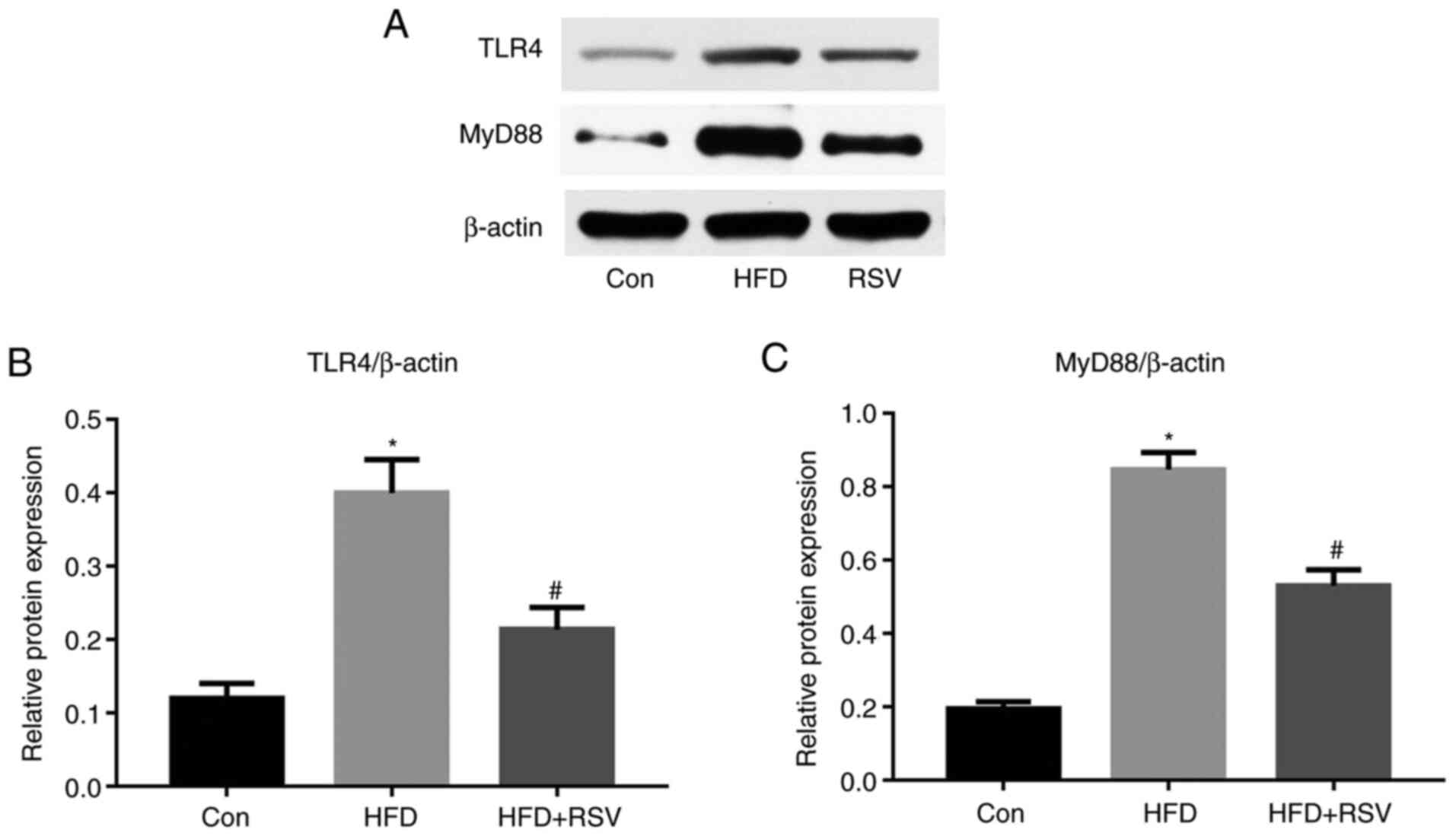
RSV administration reduces renal TLR4
and MyD88 expression
It has previously been reported that TLR4 may serve
an important role in the immune response (17). In the present study, long-term
HFD-feeding significantly increased the protein expression levels
of TLR4 in the kidney, whereas this was reduced by RSV
administration (Fig. 7A and B).
Previous studies have also shown that MyD88 is involved in signal
transduction from most TLRs, including TLR4 (18); therefore, MyD88 protein expression
was also measured and the results revealed that RSV administration
significantly reduced the protein expression levels of MyD88 in
HFD-fed mice (Fig. 7A and C).
RSV administration reduces the protein
expression of TRAF6, TAK1 and NF-κB
TRAF6, TAK1 and NF-κB are some of the numerous
proteins that lie downstream of the MyD88 signaling pathway
(19). As shown in Fig. 8A-C, long-term HFD-feeding
significantly increased the protein expression levels of TRAF6 and
TAK1 in the kidney, whereas RSV significantly reduced their
expression. Notably, NF-κB protein expression exhibited the same
trend (Fig. 8A and D). Thus, RSV
significantly inhibited the expression of TRAF6, TAK1 and NF-κB in
the kidney of HFD-fed mice.
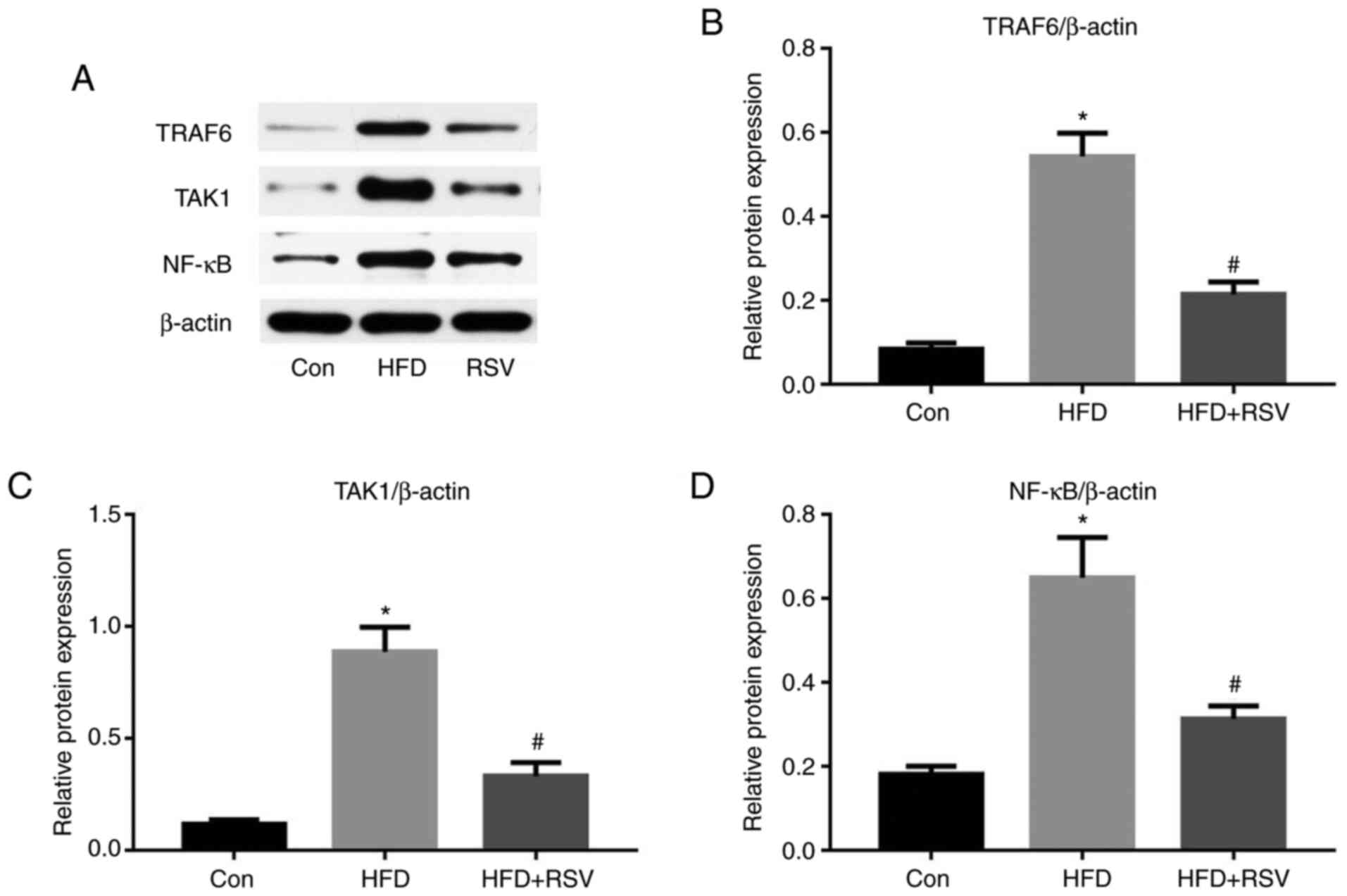 | Figure 8.Effect of RSV on the protein
expression levels of TRAF6, TAK1 and NF-κB in the kidneys of
HFD-fed mice. (A) Western blot analysis of TRAF6, TAK1 and NF-κB in
the kidney. Relative protein expression levels of (B) TRAF6, (C)
TAK1 and (D) NF-κB, normalized to β-actin. Data are presented as
the mean ± SD (n=10). *P<0.05 vs. Con group;
#P<0.05 vs. HFD group. Con, control; HFD, high-fat
diet; NF-κB, nuclear factor-κB; RSV, resveratrol; TAK1,
TGF-β-activated kinase 1; TRAF6, tumor necrosis factor
receptor-associated factor 6. |
Discussion
The present study demonstrated that: i) Long-term
HFD-feeding induced IR, and increased the blood glucose, lipid and
UA concentrations in mice. ii) RSV administration could improve
HFD-induced IR and SUA concentration. iii) Long-term HFD affected
the protein expression levels of GLUT9 and URAT1 in the kidneys,
which could lead to increased reabsorption of UA, thereby
increasing blood UA levels, whereas RES intervention reversed this
process. iv) Long-term HFD-feeding led to increased kidney
inflammation indicators, and the intervention of RES reduced kidney
inflammation. v) Insulin-resistant mice established by long-term
HFD feeding exhibited lipid deposition, glycogen accumulation,
inflammatory damage, renal fibrosis, significantly increased
expression of UA transport-related proteins, and increased blood UA
levels. Notably, RES intervention, by improving IR, reduced kidney
inflammation, reversed the pathological damage of the kidney,
regulated the expression of renal UA transport-related proteins,
reduced the reabsorption of UA, and thus reduced the blood UA
level. vi) The effect of RSV may involve inhibition of the NLRP3
inflammasome and the TLR4/MyD88/NF-κB signaling pathway.
RSV is a polyphenolic plant antitoxin that has
effects on metabolism, and has been reported to ameliorate IR and
related defects, including dyslipidemia, hyperglycemia, HUA and
hyperinsulinemia (10). A previous
study revealed that RSV exerted protective effects via activation
of the silent information regulator factor 2-related enzyme 1
signaling pathway (20). In
addition, RSV has been shown to reduce inflammation in the kidney
by inhibiting the synthesis of pro-inflammatory cytokines IL-6 and
TNF-α, or reducing the release of nitric oxide (11). To the best of our knowledge, it has
not previously been shown that RSV ameliorates HUA and affects the
expression of UA transporter proteins in such a model. The effects
of RSV on renal GLUT9 and URAT1 expression may also be mediated
through its anti-inflammatory effects.
The NLRP3 inflammasome and the TLR4 signaling
pathway serve key roles in HUA-associated kidney injury. The NLRP3
inflammasome is a multimeric protein complex that comprises the
adaptor molecule ASC, caspase-1 and NLRP3 (21,22),
and activation of the TLR4 pathway promotes inflammation (23). Inhibition of NLRP3 and the TLR4
signaling pathway are promising approaches for the treatment of
metabolic diseases (23). The
findings of the present study suggested that RSV ameliorated renal
inflammation induced by HFD-feeding via these pathways and thereby
downregulated the expression levels of GLUT9 and URAT1, inhibited
UA reabsorption, and subsequently ameliorated HUA and IR in the
mice. Specifically, RSV reduced the renal expression levels of
NLRP3/ASC/caspase-1 and of TLR4/MyD88 in the HFD-fed mice. This was
associated with a reduction in the expression of downstream
proteins, including TRAF6, TAK1 and NF-κB, which mediate renal
inflammation. It was hypothesized that this may cause a reduction
in the expression levels of GLUT9 and URAT1, reduce SUA and thus
ameliorate IR.
HUA is a chronic metabolic disease that is mainly
caused by greater production of and/or lower excretion of UA
(24,25). In recent years, it has been reported
that high SUA is not only the main cause of gout, but also has a
close relationship with components of metabolic syndrome, such as
hyperinsulinemia, hypertension and diabetes (26). IR can also promote HUA because
hyperinsulinemia reduces the activity of 3-phosphoglycerate
dehydrogenase, a key glycolytic enzyme, which leads to the
conversion of glycolytic intermediates to 5-phosphate ribose and
phosphoribose pyrophosphate, thereby promoting UA generation. In
addition, IR promotes the accumulation of liver fat, which impairs
purine metabolism, leading to the production of UA (27,28).
These data linking HUA to IR suggest that SUA may be an independent
predictor of HUA. Both IR and HUA can induce kidney pathology,
metabolic abnormalities and cardiovascular diseases, and they
interact to cause pathological progression. However, the effects of
HFD-feeding and the associated IR on UA turnover in the kidney are
not yet fully understood.
Glucose, lipid and UA metabolism disorders are
features of numerous primary and secondary kidney diseases, and
although there have been a number of studies regarding the kidney
pathology associated with metabolic disorders (29,30),
the mechanism involved remains unclear (30). Previous studies have shown that high
SUA concentrations are associated with inflammation and renal
insufficiency (6,31,32),
and the present study revealed that long-term HFD-feeding caused
renal lipid deposition and increased the renal concentrations of
IL-1β, IL-6, IL-18 and TNF-α, which may lead to renal dysfunction.
RSV can affect the expression of UA transporter proteins by
improving kidney inflammation and can reduce blood UA levels. GLUT9
and URAT1 may be the key proteins linking lipid metabolism and UA
metabolism. The present study provides novel theoretical evidence
for RSV to improve metabolic syndrome and kidney inflammation.
In conclusion, the present study demonstrated that
RSV improved SUA and reduced UA reabsorption. These effects of RSV
may be achieved by reducing renal inflammation in HFD-fed
insulin-resistant mice. The mechanisms underlying the improvement
in renal inflammation may involve a reduction in the expression of
the NLRP3 inflammasome and TLR4 signaling pathway components.
Acknowledgements
Not applicable.
Funding
The present study was supported by a grant from the
Natural Science Foundation of Hebei Province (grant no.
H2018307071).
Availability of data and materials
The datasets used and/or analyzed during the current
study are available from the corresponding author on reasonable
requests.
Authors' contributions
XZ and GS conceived and designed the study. XZ, QN,
ZZ, JZ, FZ, CW and XW acquired and analyzed the data. XZ, CW and GS
confirmed the authenticity of all the raw data. XZ prepared the
draft of the manuscript, including the figures. All authors read
and approved the final manuscript.
Ethics approval and consent to
participate
All animals received humane care in compliance with
the Guide for the Care and Use of Laboratory Animals in China. The
present study was approved by Hebei General Hospital Ethical
Committee (approval no. 2019E389).
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
Glossary
Abbreviations
Abbreviations:
|
RSV
|
resveratrol
|
|
IR
|
insulin resistance
|
|
HFD
|
high-fat diet
|
|
UA
|
uric acid
|
|
SUA
|
serum uric acid
|
|
HUA
|
hyperuricemia
|
|
IPGTT
|
intraperitoneal glucose tolerance
testing
|
|
TG
|
triglyceride
|
|
TC
|
total cholesterol
|
|
HDL-C
|
high-density
lipoprotein-cholesterol
|
|
LDL-C
|
low-density
lipoprotein-cholesterol
|
|
H&E
|
hematoxylin and eosin
|
|
PAS
|
periodic acid-Schiff
|
|
IL-6
|
interleukin-6
|
|
IL-18
|
interleukin-18
|
|
IL-1β
|
interleukin-1β
|
|
TNF-α
|
tumor necrosis factor-α
|
|
GLUT9
|
glucose transporter 9
|
|
URAT1
|
urate transporter 1
|
|
MCP-1
|
monocyte chemotactic protein-1
|
|
NLRP3
|
NOD-like receptor family, pyrin
domain-containing 3
|
|
ASC
|
apoptosis-associated speck-like
protein
|
|
MyD88
|
myeloid differentiation factor 88
|
|
TRAF6
|
TNF receptor-associated factor 6
|
|
TAK1
|
TGF-β-activated kinase 1
|
|
NF-κB
|
nuclear factor-κB
|
|
TLR4
|
Toll-like receptor 4
|
References
|
1
|
El Ridi R and Tallima H: Physiological
functions and pathogenic potential of uric acid: A review. J Adv
Res. 8:487–493. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Billiet L, Doaty S, Katz JD and Velasquez
MT: Review of hyperuricemia as new marker for metabolic syndrome.
ISRN Rheumatol. 2014:8529542014. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Sharaf El Din UAA, Salem MM and Abdulazim
DO: Uric acid in the pathogenesis of metabolic, renal, and
cardiovascular diseases: A review. J Adv Res. 8:537–548. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Tan PK, Farrar JE, Gaucher EA and Miner
JN: Coevolution of URAT1 and Uricase during Primate Evolution:
Implications for Serum Urate Homeostasis and Gout. Mol Biol Evol.
33:2193–2200. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Ebert K, Ludwig M, Geillinger KE,
Schoberth GC, Essenwanger J, Stolz J, Daniel H and Witt H:
Reassessment of GLUT7 and GLUT9 as Putative Fructose and Glucose
Transporters. J Membr Biol. 250:171–182. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Yang Y, Zhang DM, Liu JH, Hu LS, Xue QC,
Ding XQ and Kong LD: Wuling San protects kidney dysfunction by
inhibiting renal TLR4/MyD88 signaling and NLRP3 inflammasome
activation in high fructose-induced hyperuricemic mice. J
Ethnopharmacol. 169:49–59. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Akira S, Takeda K and Kaisho T: Toll-like
receptors: Critical proteins linking innate and acquired immunity.
Nat Immunol. 2:675–680. 2001. View
Article : Google Scholar : PubMed/NCBI
|
|
8
|
Zeng XZ, Zhang YY, Yang Q, Wang S, Zou BH,
Tan YH, Zou M, Liu SW and Li XJ: Artesunate attenuates LPS-induced
osteoclastogenesis by suppressing TLR4/TRAF6 and
PLCγ1-Ca2+-NFATc1 signaling pathway. Acta Pharmacol Sin.
41:229–236. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Shigeoka AA, Mueller JL, Kambo A, Mathison
JC, King AJ, Hall WF, Correia JS, Ulevitch RJ, Hoffman HM and McKay
DB: An inflammasome-independent role for epithelial-expressed Nlrp3
in renal ischemia-reperfusion injury. J Immunol. 185:6277–6285.
2010. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Thiel G and Rössler OG: Resveratrol
regulates gene transcription via activation of stimulus-responsive
transcription factors. Pharmacol Res. 117:166–176. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Cheng K, Song Z, Chen Y, Li S, Zhang Y,
Zhang H, Zhang L, Wang C and Wang T: Resveratrol protects against
renal damage via attenuation of inflammation and oxidative stress
in high-fat-diet-induced obese mice. Inflammation. 42:937–945.
2019. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Saldanha JF, Leal VO, Stenvinkel P,
Carraro-Eduardo JC and Mafra D: Resveratrol: Why is it a promising
therapy for chronic kidney disease patients? Oxid Med Cell Longev.
2013:9632172013. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Jørgensen MS, Tornqvist KS and Hvid H:
Calculation of glucose dose for intraperitoneal glucose tolerance
tests in lean and obese mice. J Am Assoc Lab Anim Sci. 56:95–97.
2017.
|
|
14
|
Bhatt JK, Thomas S and Nanjan MJ:
Resveratrol supplementation improves glycemic control in type 2
diabetes mellitus. Nutr Res. 32:537–541. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Shu L, Hou G, Zhao H, Huang W, Song G and
Ma H: Resveratrol improves high-fat diet-induced insulin resistance
in mice by downregulating the lncRNA NONMMUT008655.2. Am J Transl
Res. 12:1–18. 2020.PubMed/NCBI
|
|
16
|
Katz A, Nambi SS, Mather K, Baron AD,
Follmann DA, Sullivan G and Quon MJ: Quantitative insulin
sensitivity check index: A simple, accurate method for assessing
insulin sensitivity in humans. J Clin Endocrinol Metab.
85:2402–2410. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Brenner C, Simmonds RE, Wood S, Rose V,
Feldmann M and Turner J: TLR signalling and adapter utilization in
primary human in vitro differentiated adipocytes. Scand J Immunol.
76:359–370. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
O'Neill LA, Dunne A, Edjeback M, Gray P,
Jefferies C and Wietek C: Mal and MyD88: Adapter proteins involved
in signal transduction by Toll-like receptors. J Endotoxin Res.
9:55–59. 2003. View Article : Google Scholar
|
|
19
|
Verstak B, Nagpal K, Bottomley SP,
Golenbock DT, Hertzog PJ and Mansell A: MyD88 adapter-like
(Mal)/TIRAP interaction with TRAF6 is critical for TLR2- and
TLR4-mediated NF-kappaB proinflammatory responses. J Biol Chem.
284:24192–24203. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Lagouge M, Argmann C, Gerhart-Hines Z,
Meziane H, Lerin C, Daussin F, Messadeq N, Milne J, Lambert P,
Elliott P, et al: Resveratrol improves mitochondrial function and
protects against metabolic disease by activating SIRT1 and
PGC-1alpha. Cell. 127:1109–1122. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Zhao J, Wang H, Dai C, Wang H, Zhang H,
Huang Y, Wang S, Gaskin F, Yang N and Fu SM: P2X7 blockade
attenuates murine lupus nephritis by inhibiting activation of the
NLRP3/ASC/caspase 1 pathway. Arthritis Rheum. 65:3176–3185. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Zhou R, Yazdi AS, Menu P and Tschopp J: A
role for mitochondria in NLRP3 inflammasome activation. Nature.
469:221–225. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Roger T, David J, Glauser MP and Calandra
T: MIF regulates innate immune responses through modulation of
Toll-like receptor 4. Nature. 414:920–924. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Albert D, Scudder PN, Bagley P and Saag
KG: Vascular consequences of hyperuricemia and hypouricemia. Rheum
Dis Clin North Am. 45:453–464. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Kostka-Jeziorny K, Widecka K and Tykarski
A: Study of epidemiological aspects of hyperuricemia in Poland.
Cardiol J. 26:241–252. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Zhang S, Wang Y, Cheng J, Huangfu N, Zhao
R, Xu Z, Zhang F, Zheng W and Zhang D: Hyperuricemia and
Cardiovascular Disease. Curr Pharm Des. 25:700–709. 2019.
View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Doualla M, Halle MP, Moutchia J, Tegang S
and Ashuntantang G: Determinants of hyperuricemia in non-dialysed
chronic kidney disease patients in three hospitals in Cameroon. BMC
Nephrol. 19:1692018. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Yang H, Bai W, Gao L, Jiang J, Tang Y, Niu
Y, Lin H and Li L: Mangiferin alleviates hypertension induced by
hyperuricemia via increasing nitric oxide releases. J Pharmacol Sc.
137:154–161. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Sha J, Sui B, Su X, Meng Q and Zhang C:
Alteration of oxidative stress and inflammatory cytokines induces
apoptosis in diabetic nephropathy. Mol Med Rep. 16:7715–7723. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Mount PF and Juncos LA: Obesity-Related
CKD: When kidneys get the munchies. J Am Soc Nephrol. 28:3429–3432.
2017. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Wang MX, Zhao XJ, Chen TY, Liu YL, Jiao
RQ, Zhang JH, Ma CH, Liu JH, Pan Y and Kong LD: Nuciferine
alleviates renal injury by inhibiting inflammatory responses in
fructose-fed rats. J Agric Food Chem. 64:7899–7910. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Hu QH, Wang C, Li JM, Zhang DM and Kong
LD: Allopurinol, rutin, and quercetin attenuate hyperuricemia and
renal dysfunction in rats induced by fructose intake: Renal organic
ion transporter involvement. Am J Physiol Renal Physiol.
297:F1080–F1091. 2009. View Article : Google Scholar : PubMed/NCBI
|