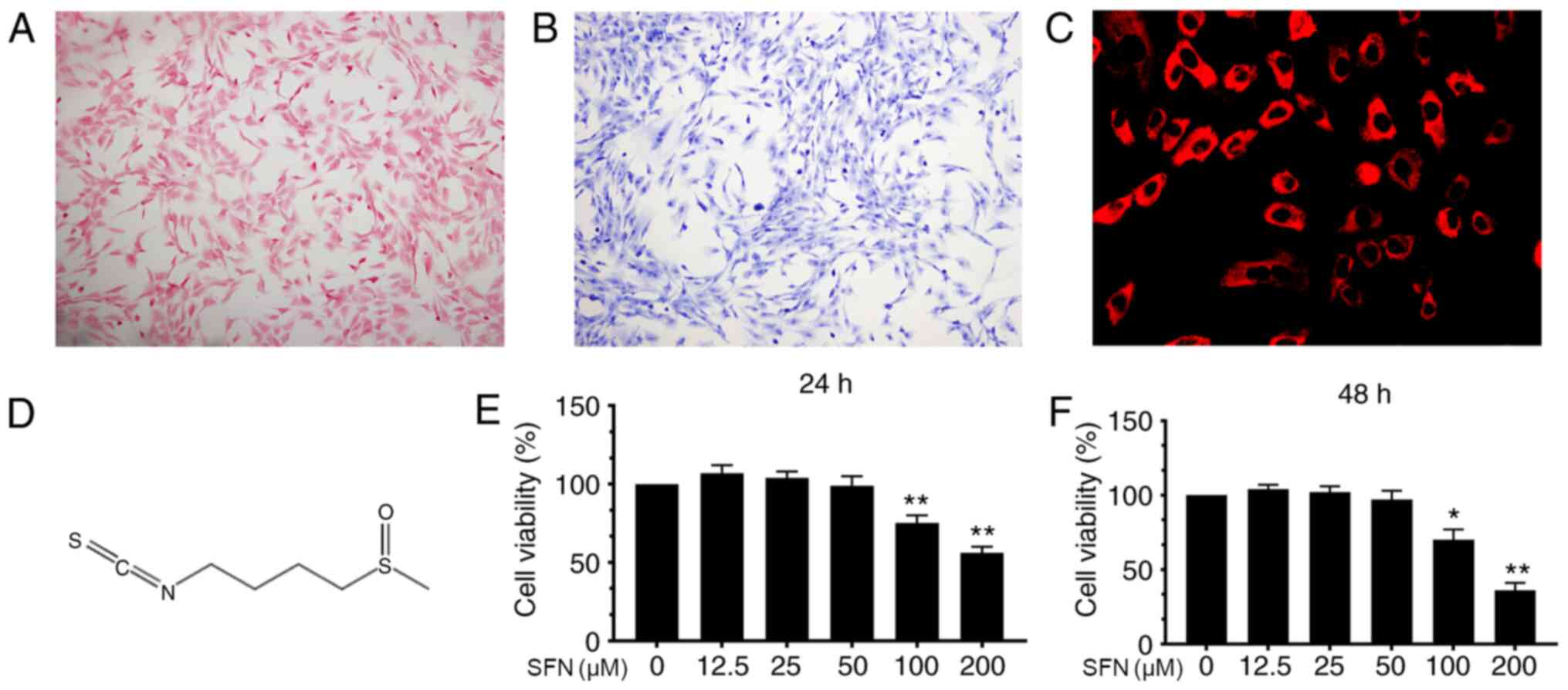
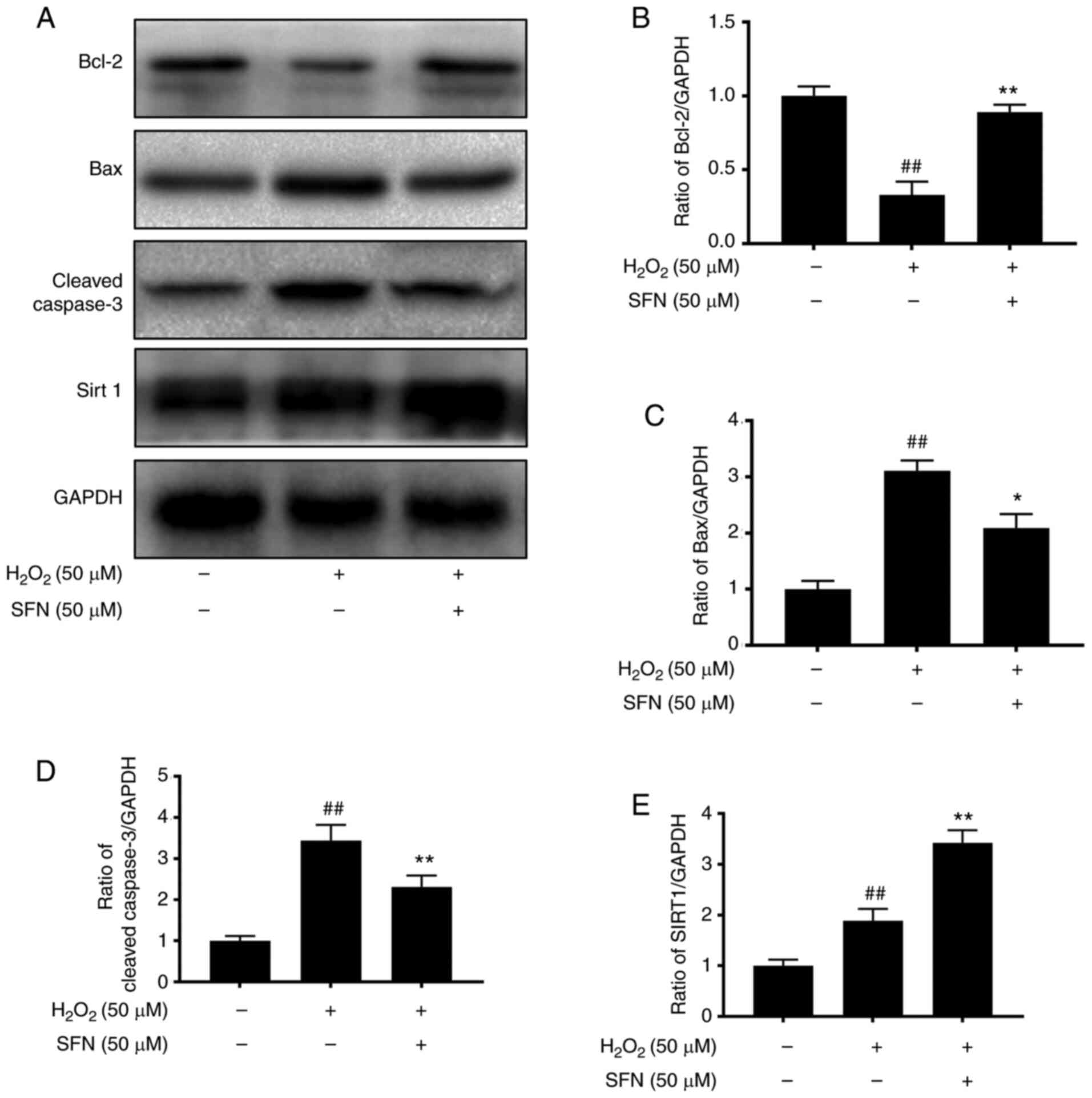
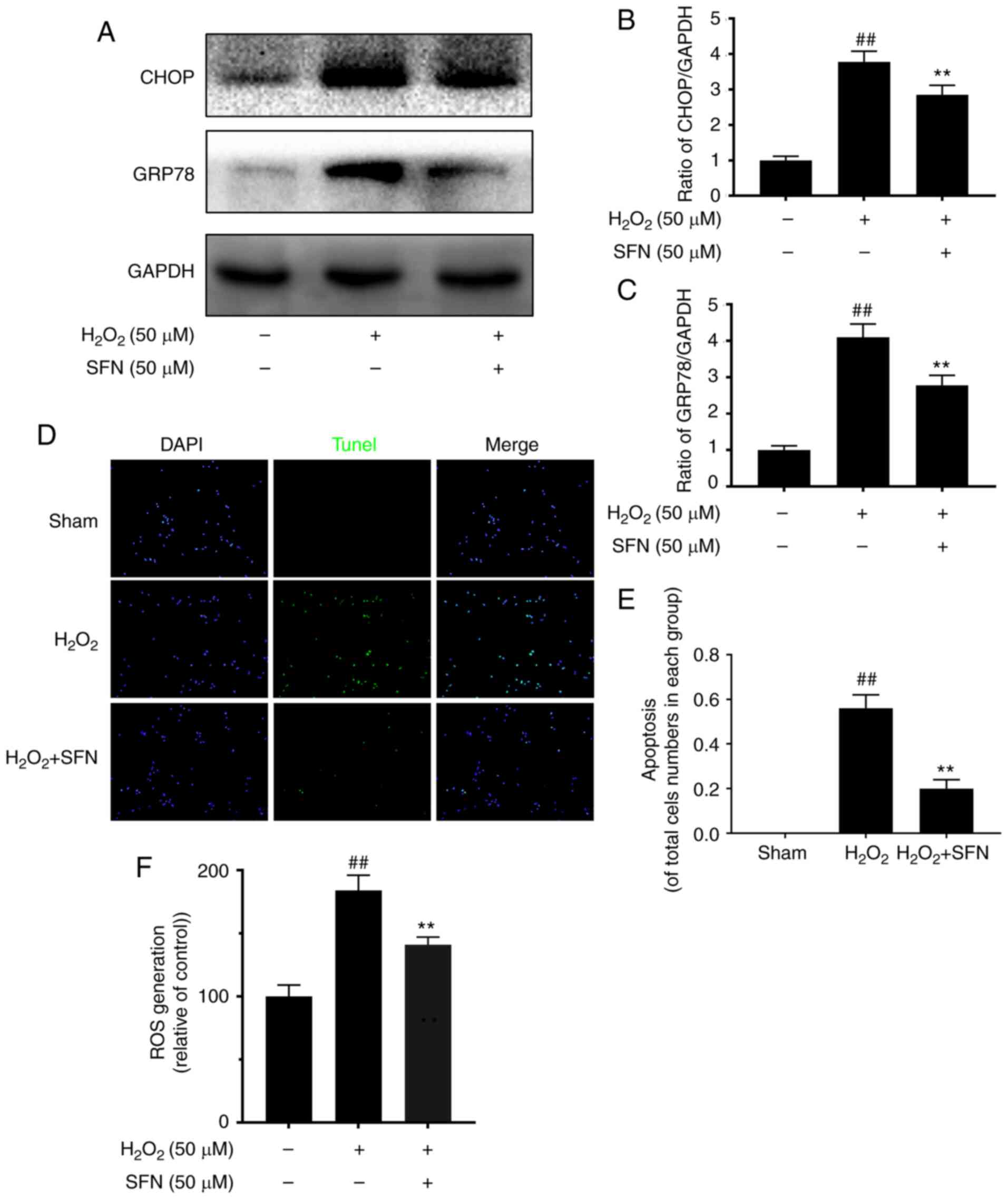
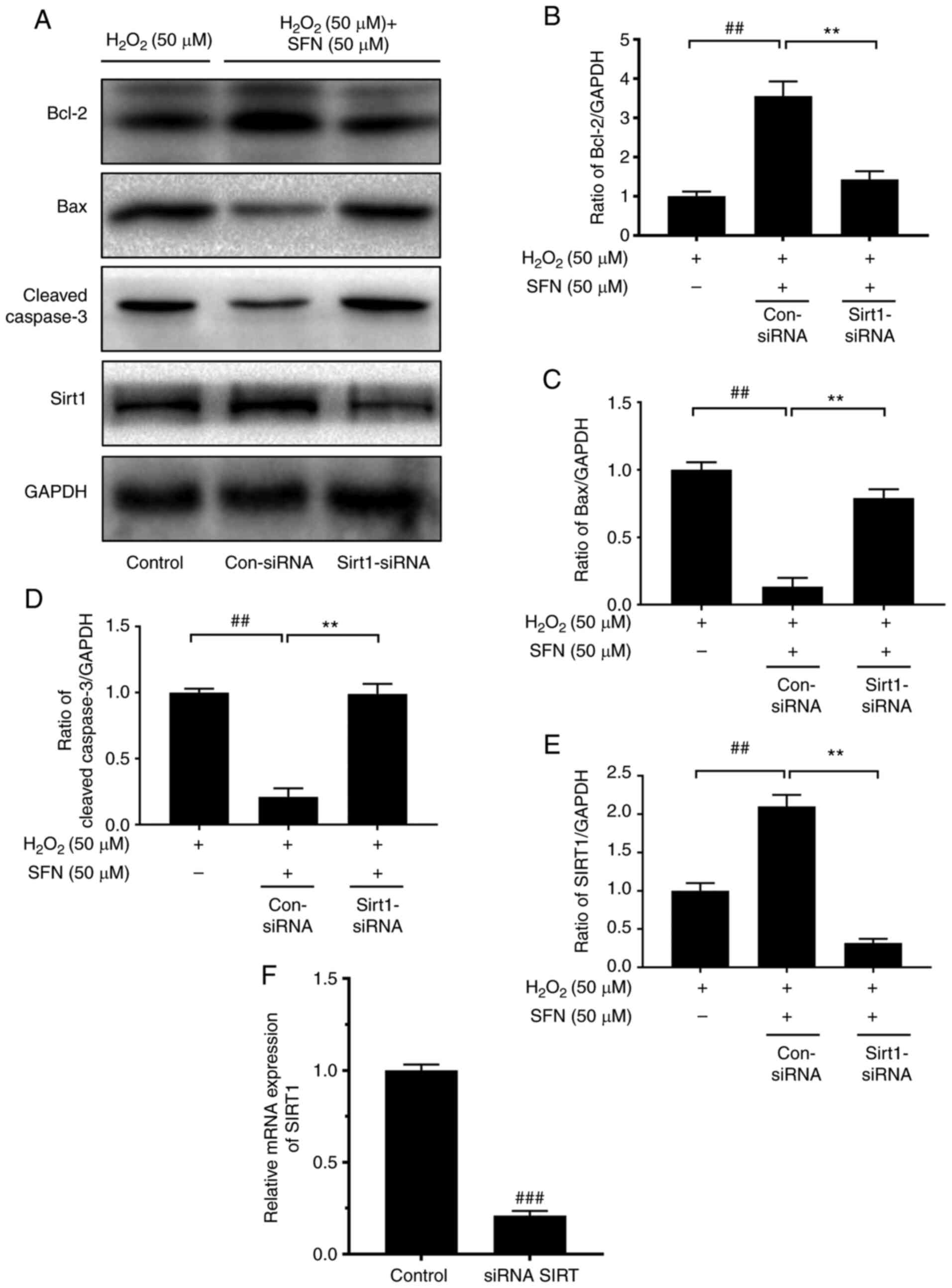
|
1
|
Xing D, Xu Y, Liu Q, Ke Y, Wang B, Li Z
and Lin J: Osteoarthritis and all-cause mortality in worldwide
populations: Grading the evidence from a meta-analysis. Sci Rep.
6:243932016. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Hunter DJ and Felson DT: Osteoarthritis.
BMJ. 332:639–642. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Lane NE, Shidara K and Wise BL:
Osteoarthritis year in review 2016: Clinical. Osteoarthritis
Cartilage. 25:209–215. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Reginster JY: The prevalence and burden of
arthritis. Rheumatology (Oxford). 41 (Suppl 1):S3–S6. 2002.
View Article : Google Scholar
|
|
5
|
Mobasheri A: The future of osteoarthritis
therapeutics: Emerging biological therapy. Curr Rheumatol Rep.
15:3852013. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Portal-Núñez S, Esbrit P, Alcaraz MJ and
Largo R: Oxidative stress, autophagy, epigenetic changes and
regulation by miRNAs as potential therapeutic targets in
osteoarthritis. Biochem Pharmacol. 108:1–10. 2016. View Article : Google Scholar
|
|
7
|
Xia H, Cao D and Yang F, Yang W, Li W, Liu
P, Wang S and Yang F: Jiawei Yanghe decoction ameliorates cartilage
degradation in vitro and vivo via Wnt/β-catenin signaling pathway.
Biomed Pharmacother. 122:1097082020. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Dai M, Sui B, Xue Y, Liu X and Sun J:
Cartilage repair in degenerative osteoarthritis mediated by squid
type II collagen via immunomodulating activation of M2 macrophages,
inhibiting apoptosis and hypertrophy of chondrocytes. Biomaterials.
180:91–103. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Lee AS, Ellman MB, Yan D, Kroin JS, Cole
BJ, van Wijnen AJ and Im HJ: A current review of molecular
mechanisms regarding osteoarthritis and pain. Gene. 527:440–447.
2013. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Musumeci G, Castrogiovanni P, Trovato FM,
Weinberg AM, Al-Wasiyah MK, Alqahtani MH and Mobasheri A:
Biomarkers of chondrocyte apoptosis and autophagy in
osteoarthritis. Int J Mol Sci. 16:20560–20575. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Bondeson J, Wainwright S, Hughes C and
Caterson B: The regulation of the ADAMTS4 and ADAMTS5 aggrecanases
in osteoarthritis: A review. Clin Exp Rheumatol. 26:139–145.
2008.PubMed/NCBI
|
|
12
|
Sharif M, Whitehouse A, Sharman P, Perry M
and Adams M: Increased apoptosis in human osteoarthritic cartilage
corresponds to reduced cell density and expression of caspase-3.
Arthritis Rheum. 50:507–515. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Claudio N, Dalet A, Gatti E and Pierre P:
Mapping the crossroads of immune activation and cellular stress
response pathways. EMBO J. 32:1214–1224. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Feng K, Chen Z, Pengcheng L, Zhang S and
Wang X: Quercetin attenuates oxidative stress-induced apoptosis via
SIRT1/AMPK-mediated inhibition of ER stress in rat chondrocytes and
prevents the progression of osteoarthritis in a rat model. J Cell
Physiol. 234:18192–18205. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Bernales S, Papa FR and Walter P:
Intracellular signaling by the unfolded protein response. Annu Rev
Cell Dev Biol. 22:487–508. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Wictome M, Henderson I, Lee AG and East
JM: Mechanism of inhibition of the calcium pump of sarcoplasmic
reticulum by thapsigargin. Biochem J. 283:525–529. 1992. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Hetz C: The unfolded protein response:
Controlling cell fate decisions under ER stress and beyond. Nat Rev
Mol Cell Biol. 13:89–102. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Huang L, Xie H and Liu H: Endoplasmic
reticulum stress, diabetes mellitus, and tissue injury. Curr
Protein Pept Sci. 15:812–818. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Chen D, Wang Y and Chin ER: Activation of
the endoplasmic reticulum stress response in skeletal muscle of
G93A*SOD1 amyotrophic lateral sclerosis mice. Front Cell Neurosci.
9:1702015. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Rasmussen ML, Kline LA, Park KP, Ortolano
NA, Romero-Morales AI, Anthony CC, Beckermann KE and Gama V: A
non-apoptotic function of MCL-1 in promoting pluripotency and
modulating mitochondrial dynamics in stem cells. Stem Cell Reports.
10:684–692. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Zhao CQ, Zhang YH, Jiang SD, Jiang LS and
Dai LY: Both endoplasmic reticulum and mitochondria are involved in
disc cell apoptosis and intervertebral disc degeneration in rats.
Age (Dordr). 32:161–177. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Lin J, Zhuge J, Zheng X, Wu Y, Zhang Z, Xu
T, Meftah Z, Xu H, Wu Y, Tian N, et al: Urolithin A-induced
mitophagy suppresses apoptosis and attenuates intervertebral disc
degeneration via the AMPK signaling pathway. Free Radic Biol Med.
150:109–119. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Shah SA, Khan M, Jo MH, Jo MG, Amin FU and
Kim MO: Melatonin stimulates the SIRT1/Nrf2 signaling pathway
counteracting lipopolysaccharide (LPS)-induced oxidative stress to
rescue postnatal rat brain. CNS Neurosci Ther. 23:33–44. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Morris BJ: Seven sirtuins for seven deadly
diseases of aging. Free Radic Biol Med. 56:133–171. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Rajendran R, Garva R, Krstic-Demonacos M
and Demonacos C: Sirtuins: Molecular traffic lights in the
crossroad of oxidative stress, chromatin remodeling, and
transcription. J Biomed Biotechnol. 2011:3682762011. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Li YP, Wang SL, Liu B, Tang L, Kuang RR,
Wang XB, Zhao C, Song XD, Cao XM, Wu X, et al: Sulforaphane
prevents rat cardiomyocytes from hypoxia/reoxygenation injury in
vitro via activating SIRT1 and subsequently inhibiting ER stress.
Acta Pharmacol Sin. 37:344–353. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Katto J, Engel N, Abbas W, Herbein G and
Mahlknecht U: Transcription factor NFκB regulates the expression of
the histone deacetylase SIRT1. Clin Epigenetics. 5:112013.
View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Feng K, Ge Y, Chen Z, Li X, Liu Z, Li X,
Li H, Tang T, Yang F and Wang X: Curcumin inhibits the PERK-eIF2
α-CHOP pathway through promoting SIRT1 expression in oxidative
stress-induced rat chondrocytes and ameliorates osteoarthritis
progression in a rat model. Oxid Med Cell Longev. 2019:85743862019.
View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Danilov CA, Chandrasekaran K, Racz J,
Soane L, Zielke C and Fiskum G: Sulforaphane protects astrocytes
against oxidative stress and delayed death caused by oxygen and
glucose deprivation. Glia. 57:645–656. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Nguyen B, Luong L, Naase H, Vives M, Jakaj
G, Finch J, Boyle J, Mulholland JW, Kwak JH, Pyo S, et al:
Sulforaphane pretreatment prevents systemic inflammation and renal
injury in response to cardiopulmonary bypass. J Thorac Cardiovasc
Surg. 148:690–697. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Forster T, Rausch V, Zhang Y, Isayev O,
Heilmann K, Schoensiegel F, Liu L, Nessling M, Richter K, Labsch S,
et al: Sulforaphane counteracts aggressiveness of pancreatic cancer
driven by dysregulated Cx43-mediated gap junctional intercellular
communication. Oncotarget. 5:1621–1634. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Ho JN, Yoon HG, Park CS, Kim S, Jun W,
Choue R and Lee J: Isothiocyanates ameliorate the symptom of heart
dysfunction and mortality in a murine AIDS model by inhibiting
apoptosis in the left ventricle. J Med Food. 15:781–787. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Davidson RK, Jupp O, de Ferrars R, Kay CD,
Culley KL, Norton R, Driscoll C, Vincent TL, Donell ST, Bao Y and
Clark IM: Sulforaphane represses matrix-degrading proteases and
protects cartilage from destruction in vitro and in vivo. Arthritis
Rheum. 65:3130–3140. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Davidson RK, Green J, Gardner S, Bao Y,
Cassidy A and Clark IM: Identifying chondroprotective diet-derived
bioactives and investigating their synergism. Sci Rep. 8:171732018.
View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Zheng G, Zhan Y, Tang Q, Chen T, Zheng F,
Wang H, Wang J, Wu D, Li X, Zhou Y, et al: Monascin inhibits IL-1β
induced catabolism in mouse chondrocytes and ameliorates murine
osteoarthritis. Food Funct. 9:1454–1464. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) method. Methods. 25:402–408. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
37
|
The Guide for the Care and Use of
Laboratory Animals of the National Institutes of Health published
by the National Institutes of Health. NIH Publication No. 85-23,
revised 1996.
|
|
38
|
Glasson SS, Blanchet TJ and Morris EA: The
surgical destabilization of the medial meniscus (DMM) model of
osteoarthritis in the 129/SvEv mouse. Osteoarthritis Cartilage.
15:1061–1069. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Yoo IH, Kim MJ, Kim J, Sung JJ, Park ST
and Ahn SW: The anti-inflammatory effect of sulforaphane in mice
with experimental autoimmune encephalomyelitis. J Korean Med Sci.
34:e1972019. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Huo L, Su Y, Xu G, Zhai L and Zhao J:
Sulforaphane protects the male reproductive system of mice from
obesity-induced damage: Involvement of oxidative stress and
autophagy. Int J Environ Res Public Health. 16:37592019. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Glasson SS, Chambers MG, Van Den Berg WB
and Little CB: The OARSI histopathology initiative-recommendations
for histological assessments of osteoarthritis in the mouse.
Osteoarthritis Cartilage. 18 (Suppl 3):S17–S23. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
French HP, Galvin R, Horgan NF and Kenny
RA: Prevalence and burden of osteoarthritis amongst older people in
Ireland: Findings from The Irish LongituDinal Study on Ageing
(TILDA). Eur J Public Health. 26:192–198. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Glyn-Jones S, Palmer AJ, Agricola R, Price
AJ, Vincent TL, Weinans H and Carr AJ: Osteoarthritis. Lancet.
386:376–387. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Johnson VL and Hunter DJ: The epidemiology
of osteoarthritis. Best Pract Res Clin Rheumatol. 28:5–15. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Asada S, Fukuda K, Nishisaka F, Matsukawa
M and Hamanisi C: Hydrogen peroxide induces apoptosis of
chondrocytes; involvement of calcium ion and extracellular
signal-regulated protein kinase. Inflamm Res. 50:19–23. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Kim EN, Lee HS and Jeong GS:
Cudratricusxanthone O Inhibits H2O2-Induced
cell damage by activating Nrf2/HO-1 pathway in human chondrocytes.
Antioxidants (Basel). 9:7882020. View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Sutipornpalangkul W, Morales NP and
Harnroongroj T: Free radicals in primary knee osteoarthritis. J Med
Assoc Thai. 92 (Suppl 6):S268–S274. 2009.PubMed/NCBI
|
|
48
|
Hadjigogos K: The role of free radicals in
the pathogenesis of rheumatoid arthritis. Panminerva Med. 45:7–13.
2003.PubMed/NCBI
|
|
49
|
Russo M, Spagnuolo C, Russo GL,
Skalicka-Woźniak K, Daglia M, Sobarzo-Sánchez E, Nabavi SF and
Nabavi SM: Nrf2 targeting by sulforaphane: A potential therapy for
cancer treatment. Crit Rev Food Sci Nutr. 58:1391–1405. 2018.
View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Sita G, Hrelia P, Graziosi A and Morroni
F: Sulforaphane from cruciferous vegetables: Recent advances to
improve glioblastoma treatment. Nutrients. 10:17552018. View Article : Google Scholar : PubMed/NCBI
|
|
51
|
Corssac GB, Campos-Carraro C, Hickmann A,
da Rosa Araujo AS, Fernandes RO and Belló-Klein A: Sulforaphane
effects on oxidative stress parameters in culture of adult
cardiomyocytes. Biomed Pharmacother. 104:165–171. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
52
|
Wang G, Nie JH, Bao Y and Yang X:
Sulforaphane rescues ethanol-suppressed angiogenesis through
oxidative and endoplasmic reticulum stress in chick embryos. J
Agric Food Chem. 66:9522–9533. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
53
|
Pu D, Zhao Y, Chen J, Sun Y, Lv A, Zhu S,
Luo C, Zhao K and Xiao Q: Protective effects of sulforaphane on
cognitive impairments and AD-like lesions in diabetic mice are
associated with the upregulation of Nrf2 transcription activity.
Neuroscience. 381:35–45. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
54
|
Tubbs E, Axelsson AS, Vial G, Wollheim CB,
Rieusset J and Rosengren AH: Sulforaphane improves disrupted
ER-mitochondria interactions and suppresses exaggerated hepatic
glucose production. Mol Cell Endocrinol. 461:205–214. 2018.
View Article : Google Scholar : PubMed/NCBI
|
|
55
|
Chen Z, Yuan Q, Xu G, Chen H, Lei H and Su
J: Effects of quercetin on proliferation and
H2O2-induced apoptosis of intestinal porcine
enterocyte cells. Molecules. 23:20122018. View Article : Google Scholar : PubMed/NCBI
|
|
56
|
Chandrika BB, Yang C, Ou Y, Feng X, Muhoza
D, Holmes AF, Theus S, Deshmukh S, Haun RS and Kaushal GP:
Endoplasmic reticulum stress-induced autophagy provides
cytoprotection from chemical hypoxia and oxidant injury and
ameliorates renal ischemia-reperfusion injury. PLoS One.
10:e01400252015. View Article : Google Scholar : PubMed/NCBI
|
|
57
|
Tang Q, Zheng G, Feng Z, Chen Y, Lou Y,
Wang C, Zhang X, Zhang Y, Xu H, Shang P and Liu H: Trehalose
ameliorates oxidative stress-mediated mitochondrial dysfunction and
ER stress via selective autophagy stimulation and autophagic flux
restoration in osteoarthritis development. Cell Death Dis.
8:e30812017. View Article : Google Scholar : PubMed/NCBI
|
|
58
|
Deng Z, Li Y, Liu H, Xiao S, Li L, Tian J,
Cheng C, Zhang G and Zhang F: The role of sirtuin 1 and its
activator, resveratrol in osteoarthritis. Biosci Rep.
39:BSR201901892019. View Article : Google Scholar : PubMed/NCBI
|
|
59
|
Guo JY, Li F, Wen YB, Cui HX, Guo ML,
Zhang L, Zhang YF, Guo YJ and Guo YX: Melatonin inhibits
Sirt1-dependent NAMPT and NFAT5 signaling in chondrocytes to
attenuate osteoarthritis. Oncotarget. 8:55967–55983. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
60
|
Hu A, Liu HB, Mlynski R, Plontke S, Zhang
JF, Dai WJ, Duan JL, Fan JP, Zheng HL, Xu WH, et al: Therapeutic
ultrasound potentiates the anti-nociceptive and anti-inflammatory
effects of curcumin to postoperative pain via Sirt1/NF-κB signaling
pathway. Am J Transl Res. 10:3099–3110, eCollection.
2018.PubMed/NCBI
|
|
61
|
Li T, Pang Q, Liu Y, Bai M, Peng Y and
Zhang Z: Sulforaphane protects human umbilical vein endothelial
cells from oxidative stress via the miR-34a/SIRT1 axis by
upregulating nuclear factor erythroid-2-related factor 2. Exp Ther
Med. 21:1862021. View Article : Google Scholar : PubMed/NCBI
|
|
62
|
Sun X, Mi L, Liu J, Song L, Chung LF and
Gan N: Sulforaphane prevents microcystin-LR-induced oxidative
damage and apoptosis in BALB/c mice. Toxicol Appl Pharmacol.
255:9–17. 2011. View Article : Google Scholar : PubMed/NCBI
|