Introduction
Chronic infection by hepatitis B virus (HBV), or
chronic HBV infection (CHBVI), is a major global health problem
despite the introduction of effective anti-viral therapies and
vaccines (1). According to
statistics released by the United Nations, there were >250
million CHBVI patients in 2015 who were positive for hepatitis B
surface antigen (HBsAg). Moreover, >50% of patients with CHBVI
are located in the Pacific region countries, including Vietnam,
Philippines, Japan, Korea and China. For example, the incidence of
CHBVI was ~10% in Korea during 2009–2017, although it has been
gradually decreasing to a rate of <3% (2,3). DM is
associated with the progression of severe liver outcomes in adults
with HBV (3).
There is a complex community of intestinal
microbiotas, including Bacteroides, Escherichia coli,
Enterococcus faecalis and Bifidobacteria, living in the
human digestive system, including the gut and intestines (4). These intestinal microbiotas are
crucial for human health, although the change in the composition of
intestinal microbiotas may occur in certain pathological situations
to cause bacterial translocation and pathological symptoms
(4). In particular, since the liver
and intestinal microbiotas are connected by the blood circulation
system, the abnormality in intestinal microbiotas can easily affect
the liver (5). A previous study
showed reduced levels of Lactobacillus and
Bifidobacteria infection in patients with liver cirrhosis
and CHBVI, thus weakening the protection provided by the intestinal
barrier and resulting in bacterial translocation, microbiota
imbalance and endotoxemia (6). As a
result, promoting the growth of intestinal bacteria, including
Bifidobacterium and Lactobacillus, while suppressing
the reproduction of potential intestinal pathogens, including
Enterobacteriaceae, are of great significance in improving the
intestinal microbiota imbalance as well as delaying and preventing
liver cirrhosis (6).
MicroRNAs (miRNAs/miRs) are a type of non-coding RNA
implicated in a number of diseases and the serum levels of certain
miRNAs are utilized as markers to aid the prognosis and
classification of tumors (7).
miR-192-5p, a miRNA related to inflammatory bowel disease, can
suppress the expression of nucleotide-binding oligomerization
domain-containing protein 2 while regulating inflammatory reactions
in epithelial cells in the colon (8). In addition, miR-215-5p can be utilized
as a prognostic biomarker to predict the transformation of
non-penetrating Crohn's disease (CD) into penetrating CD (9). Moreover, the expression levels of
miR-215-5p and miR-192-5p have been found to be reduced during the
progression of colon cancer (10–12).
It has also been demonstrated that the miRNAs released from the
epithelial cells in the intestine can regulate the composition of
local microbiota. Another study also investigated the effect of
stomach microbiota on the expression of miRNAs in epithelial cells
in the intestine (13). It has
previously been demonstrated that the microbiota regulates gene
expression in the colon of mice (14). Therefore, it has been further
suggested that the miRNAs synthesized by epithelial cells in the
intestine are involved in the growth of stomach microbiota
(15). Detection of miRNAs isolated
from the feces of rodents may help to monitor the relationship
between stomach microbiota and miRNA expression in live animals in
a time-dependent manner (13).
Moreover, miR-192-5p expression is increased in diabetes mellitus
(DM) to inhibit insulin production by promoting the apoptosis of
β-cells in the pancreas. Moreover, miR-192-5p can promote type 1 DM
by reducing the expression of glucagon-like peptide-1 (GLP-1)
(16).
It has been shown that HBV infection may cause
intestinal microbiota imbalance and deregulation of miR-192-5p
expression (17–19). Furthermore, intestinal microbiota
imbalance and deregulation of miR-192-5p may elevate the risk of DM
by altering the expression of GLP-1 (16,20).
In the present study, samples from HBV-infected subjects with or
without high alanine transaminase (ALT) levels were collected and
the association among HBV infection, liver function and the
expression of miR-192-5p and GLP-1 were investigated.
Materials and methods
Human sample collection
This study included subjects undergoing HBsAg
testing and stool sample testing at The Third Hospital of Hebei
Medical University (Shijiazhuang, China). A total of 60 DM patients
with HBV infections were recruited in the research between December
2015 and August 2016. A total of 5 ml peripheral blood was
collected from each subject after they were informed that those
samples would be used for academic study. These patients were
further divided into the following three groups according to their
levels of HBsAg and ALT: i) Group A, HBsAg-negative control (NC)
group (n=20); ii) group B, HBsAg-positive group with a normal level
of ALT (n=20); and iii) group C, HBsAg-positive group with a high
level of ALT (n=20). The demographic and clinicopathological
characteristics of all participants, such as their age, gender,
body mass index (BMI), fat content, systolic blood pressure (BP),
diastolic BP, glucose, total cholesterol (C), high-density
lipoprotein (HDL)-C, low-density lipoprotein (LDL)-C,
triglycerides, total bilirubin, aspartate transaminase (AST), ALT,
white blood cell (WBC), platelet, homeostatic model assessment
(HOMA)-insulin resistance (IR) and fatty liver symptoms, were
collected, summarized and compared, as presented in Table I. Mucosal biopsy samples were
obtained from the jejunum or descending colon by dissection under a
microscope, removing the overlying smooth muscle and associated
myenteric innervation, and then they were directly snap frozen in
liquid nitrogen and stored at −18°C until further analysis. All
procedures were approved by the ethics committee of The Third
Hospital of Hebei Medical University (approval no. CTHHMU2015037)
and written informed consent was obtained from participants before
the initiation of this study.
 | Table I.Demographic and clinical data of the
participants included in the present study. |
Table I.
Demographic and clinical data of the
participants included in the present study.
|
Characteristics | Group A (n=20) | Group B (n=20) | Group C (n=20) | P-value |
|---|
| Age, years | 42.5±5.3 | 43.1±6.2 | 43.0±5.8 | 0.842 |
| Male, n (%) | 12 (0.6) | 10 (0.5) | 12 (0.6) | 0.846 |
| BMI,
kg/m2 | 25.6±3.4 | 25.4±3.8 | 25.9±3.4 | 0.326 |
| Fat percent, % | 24.1±5.2 | 23.4±3.5 | 23.9±3.1 | 0.645 |
| SBP, mmHg | 104.5±5.1 | 105.8±6.5 | 105.4±5.2 | 0.624 |
| DBP, mmHg | 68.1±5.3 | 66.9±5.2 | 69.3±5.6 | 0.415 |
| Glucose, mg/dl | 93.5±11.2 | 94.3±7.3 | 114.6±9.3 | <0.01 |
| Total-C, mg/dl | 194.2±25.1 | 199.8±31.6 | 189.5±25.5 | 0.342 |
| HDL-C, mg/dl | 52.3±3.8 | 52.8±6.4 | 53.8±5.8 | 0.727 |
| LDL, mg/dl | 124.5±23.8 | 124.5±21.8 | 113±25.2 | 0.854 |
| Triglycerides,
mg/dl | 125.6±17.7 | 128.6±12.4 | 133.7±6.8 | 0.251 |
| Total bilirubin,
mg/dl | 0.91±0.17 | 0.92±0.13 | 0.88±0.16 | 0.532 |
| AST, IU/l | 20.3±4.2 | 21.2±6.8 | 54.8±7.4 | <0.01 |
| ALT, IU/l | 20.8±7.2 | 21.5±6.7 | 65.8±9.1 | <0.01 |
| WBC,
×103/mm3 | 6.3±1.2 | 6.6±0.5 | 5.9±0.9 | 0.335 |
| Platelet,
×103/mm3 | 225.6±58.4 | 242.1±37.9 | 237±6.5 | 0.242 |
| HOMA-IR | 1.2±0.4 | 1.5±0.4 | 1.2±0.2 | 0.224 |
| Fatty liver, n
(%) | 8 (0.4) | 8 (0.4) | 10 (0.5) | 0.425 |
Criteria for enrollment
This research used a case vs. control design as
described in a previous article (13). Among all candidates who showed
positive reactions in the HBsAg testing, the subjects with the
following exclusion criteria were excluded: i) A history of
antibiotics usage; ii) a history of probiotics use; iii) a history
of treatment using C-lowering drugs; iv) a history of diabetes
mellitus; and v) patients with liver cirrhosis. All patients
included in the present study who showed positive reactions in the
HBsAg testing were matched by control subjects of the same gender
and a similar age. All procedures were approved by the
institutional ethics committee.
Bacterial enumeration using
quantitative (q)PCR
A sample of ~1.0 g feces collected from each
participant was directly put in a test tube holding 2 ml RNA later
buffer (Ambion; Thermo Fisher Scientific, Inc.) to achieve RNA
stabilization. Then, the composition of stomach microbiota in all
samples was evaluated with a YIF-SCAN instrument (Yakult Honsha
Co., Ltd.) in conjunction with a SYBR Green kit (Thermo Fisher
Scientific, Inc.) to measure the expression of bacterial 23S and
16S ribosomal RNA (rRNA) (21–25).
It is known that the bacterial species in the digestive system
includes six anaerobic species of bacteria, including
Prevotella, the Bacteroides fragilis group, the
Atopobium cluster, Bifidobacterium, the
Clostridium leptum subgroup and the Clostridium
coccoides group. In addition, there were five potential
pathogens, including Pseudomonas, Staphylococcus,
Enterococcus, Enterobacteriaceae and Clostridium
perfringens, along with eight types of Lactobacilli,
including the L. sakei group, the L. reuteri group,
the L. ruminis group, the L. plantarum group, the
L. casei group, L. fermentum, L. brevis and the L.
gasseri group (26,27). The bacteria strains investigated in
this research were chosen according to their high rate of incidence
in the human digestive system. To test the presence of various
bacterial strains in the patient samples, qPCR was performed to
confirm the number of 23S and 16S rRNA gene copies related to the
quantity of cultured bacteria following a protocol described in
previous studies (26,27). Since the YIF-SCAN instrument can be
used to quantify the accurate numbers of cells in each of the
bacterial species described above, the broad spectrum of bacteria
in the gut and intestine, including rare bacteria, can be
accurately detected.
Reverse transcription (RT)-qPCR
Total RNA in the feces, peripheral blood (serum
samples) and intestinal mucosal tissue samples collected from the
patients in various groups was extracted using TRIzol (Invitrogen;
Thermo Fisher Scientific, Inc.). Then, RT was carried out using a
RT assay kit (Thermo Fisher Scientific, Inc.) according to the
manufacture's protocol and then amplified using qPCR carried out on
a CFX Real-Time PCR machine (Bio-Rad Laboratories, Inc.) with a
GoTaq® Master Mix assay kit (Promega Corporation)
following the manufacturer's instructions. The thermocycling
conditions were 10 min at 95°C, 30 sec at 95°C (40 cycles), 30 sec
at 60°C and 30 sec at 72°C. The relative expression of miR-192-5p
(Forward: 5′-CTGACCTATGAATTGACAG-3′; Reverse:
5′-GAACATGTCTGCGTATCTC-3′) and GLP-1 (Forward:
5′-CGTTCCCTTCAAGACACAGAGG-4′; Reverse:
5′-ACGCCTGGAGTCCAGATACTTG-3′) was quantified using the
2−ΔΔCq method (28). U6
(Forward: 5′-GTGCTCGCTTCGGCAGCA-3′ Reverse:
5′-CAAAATATGGAACGCTTC-3′) and GAPDH mRNA (Forward:
5′-GTCTCCTCTGACTTCAACAGCG-3′; Reverse:
5′-ACCACCCTGTTGCTGTAGCCAA-3′) were utilized as the internal
standard for miR-192-5p and GLP-1, respectively.
Cell culture and treatment
CAPAN-1 and HPAC cells were acquired from American
Type Culture Collection, and then cultured in high-glucose DMEM
(HyClone; Cytiva) with 100 g/ml streptomycin, 2 mM/ml L-glutamine,
100 U/ml penicillin and 10% FBS (HyClone; Cytiva). Then, the cells
were divided into the following three groups: i) Negative control
(NC) group; ii) a group of miR-192-5p precursors; and iii) a group
of miR-192-5p inhibitors. Cells were cultured at 37°C in a
humidified tissue culture incubator containing 5% CO2.
On the day of the experiment, 1×104 cells/well of
CAPAN-1 and HPAC cells were transfected with 30 nM NC miRNA
(5′-CAGUACUUUUGUGUAGUACAA-3′), miR-192-5p precursors (sense,
5′-CUGACCUAUGAAUUGACAGCC-3′ and anti-sense,
5′-CUGUCAAUUCAUAGGUCAGUU-3′) or miR-192-5p inhibitors (sense,
5′-GGCUGUCAAUUCAUAGGUCAG-3′ and anti-sense,
5′-GGCUGUCAAUUCAUAGGUCAG-3′) manufactured by Guangzhou RiboBio Co.,
Ltd., using FuGENE® 6 transfection reagent (Promega
Corporation) according to the manufacturer's instructions.
Scrambled miRNA was used as the NC. The transfected cells were
harvested after 48 h of transfection for subsequent assays.
Vector construction, mutagenesis and luciferase
assay. The potential targets of miR-192-5p were identified using
TargetScan (http://www.targetscan.org/vert_72/) (29). Then, GLP-1 was identified as a
potential miR-192-5p target, and the 3′ UTR fragment of GLP-1
containing the miR-192-5p binding site was inserted into a pcDNA3.1
luciferase vector (Promega Corporation) to generate the wild-type
3′ UTR plasmid of the GLP-1 gene. In the next step, site-directed
mutagenesis was carried out using a QuikChange mutagenesis kit
(Stratagene) following the manufacturer's instructions, and the
mutant 3′ UTR fragment of GLP-1 containing the mutated miR-192-5p
binding site was inserted into another pcDNA3.1 vector to generate
the mutant plasmid of GLP-1 3′ UTR. Then, CAPAN-1 and HPAC cells
were transfected with wild-type or mutant plasmid of GLP-1 3′ UTR
in conjunction with miR-192-5p precursors using FuGENE 6
transfection reagent. At 48 h after the start of the transfection,
the activity of firefly luciferase was normalized to Renilla
luciferase activity and measured using a Luciferase assay kit
(Cytiva) in conjunction with a Synergy plate reader (BioTek
Instruments, Inc.).
Western blotting
Cell lysate containing 1% NP-40, 0.1% sodium dodecyl
sulfate, 50 mM Tris-HCl, 150 mM NaCl and protease inhibitors (Roche
Diagnostics) was prepared in an ice-cold lysis buffer (pH 7.4). The
protein concentration was measured with a BCA assay kit (Bio-Rad
Laboratories, Inc.) and total protein (50 µg/lane) was separated
via SDS-PAGE on 6% gel, which was then electrotransferred onto a
nitrocellulose membrane. After being blocked in phosphate buffered
saline (PBS) and 5% non-fat dry milk at room temperature for 1 h,
the membrane was incubated at 4°C for 12 h in PBS containing
primary anti-GLP-1 antibody (cat. no. ab240494; 1:10,000; Abcam)
and β-actin (cat. no. ab8226; Abcam) as the internal control
(1:10,000; Abcam). After being washed twice in PBS and incubated at
room temperature for 2 h with secondary antibodies conjugated to
horseradish peroxidase (cat. no. ab6721; 1:15,000; Abcam), the
protein level of GLP-1 was visualized by SuperSignal West Pico
Chemiluminescent Substrate (Thermo Fisher Scientific, Inc.) and
subsequently semi-quantified using Quantity One software (v4.6.6,
Bio-Rad Laboratories, Inc.).
Immunohistochemistry
The immunohistochemistry staining of feces,
peripheral blood and intestinal mucosal tissue samples (5 µm
sections blocked in 3% hydrogen peroxide for 15 min and incubated
at room temperature for 120 min) collected from various groups of
patients was performed using an anti-GLP-1 staining kit including
the corresponding primary and secondary antibodies (cat. no.
AB3244, EMD Millipore), following the manufacturer's instructions,
to determine the GLP-1 protein expression in various samples. The
samples were counterstained with hematoxylin at 37°C for 2 h and
observed under an Olympus light microscope (magnification, ×200;
Olympus Corporation).
Statistical analysis
All data was tested for heterogeneity using the
I2 statistical method. Inter-group comparisons were
performed using one-way analysis of variance (ANOVA), followed by
the Scheffe post hoc test. All statistical calculations were
carried out using Stata software version 12.0 (StataCorp LP).
P<0.05 was considered to indicate a statistically significant
difference. All data are presented as the mean ± standard deviation
and each experiment was repeated in triplicate.
Results
Demographic and clinicopathological
characteristics of the participants recruited in this study
A total of 60 patients with HBV infections were
recruited in this research. These patients were further divided
into three groups according to their levels of HBsAg and ALT: Group
A, B and C. The demographic and clinicopathological characteristics
of all participants, including their age, gender, BMI, fat content,
systolic BP, diastolic BP, glucose, total-C, HDL-C, LDL-C,
triglycerides, total bilirubin, AST, ALT, WBC, platelet, HOMA-IR
and fatty liver symptoms, were collected, summarized and compared
in Table I. One-way ANOVA was
utilized to compare the differences among the three groups. The
results showed that only the glucose level and liver function
(indicated by AST and ALT level) in the HBsAg-positive group with
high ALT was much higher than that in the other two groups, whereas
the other characteristics showed no obvious differences among the
three groups.
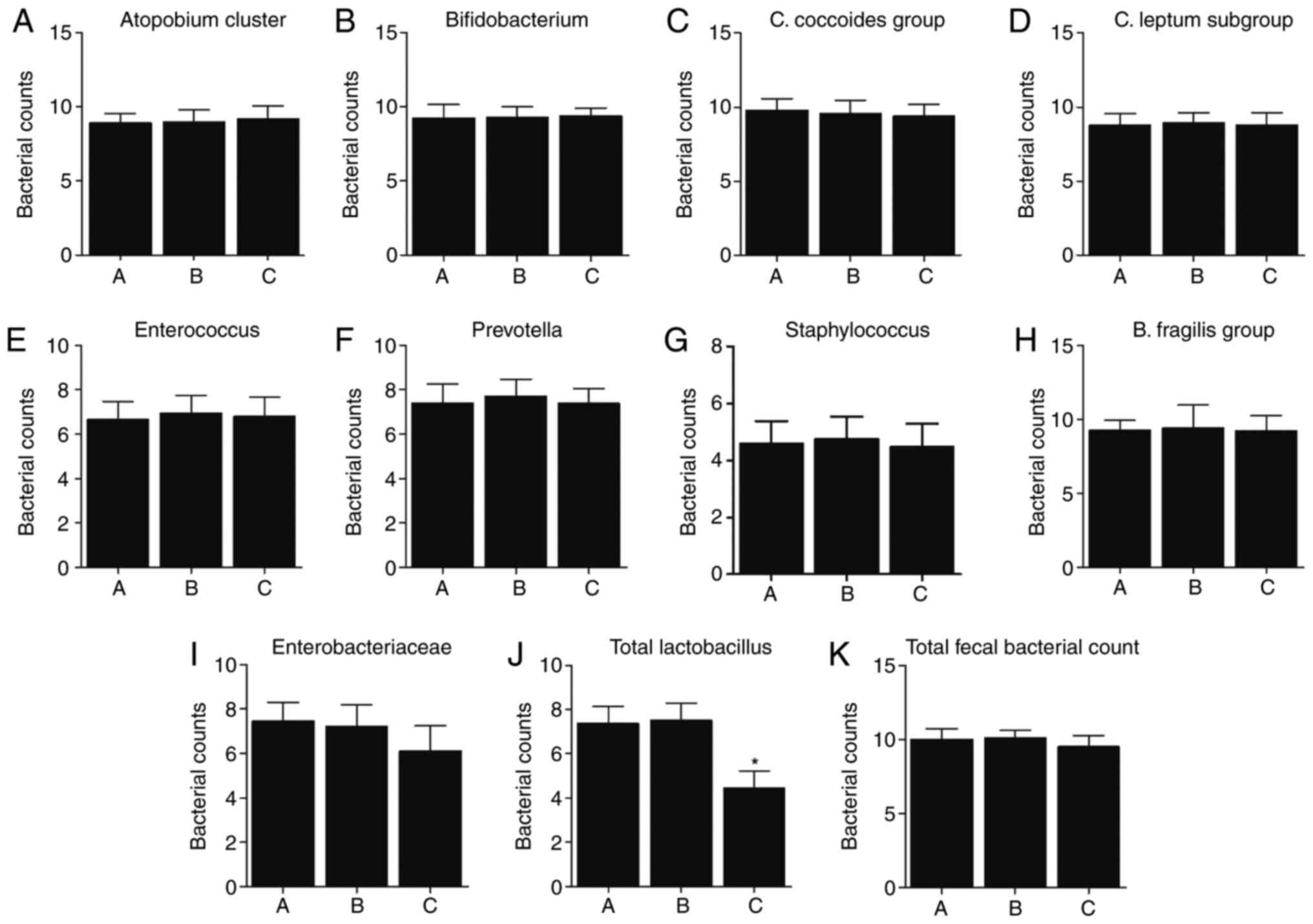
Reduced total Lactobacillus in
HBsAg-positive patients with high ALT
Fecal sampling was carried out to analyze intestinal
microbiotas in the patients. No obvious difference was found in
terms of the number of majority of bacteria among the three groups,
except that the counts of total Lactobacillus were
significantly lower in Group C compared with that in Groups A and B
(Fig. 1A-K).
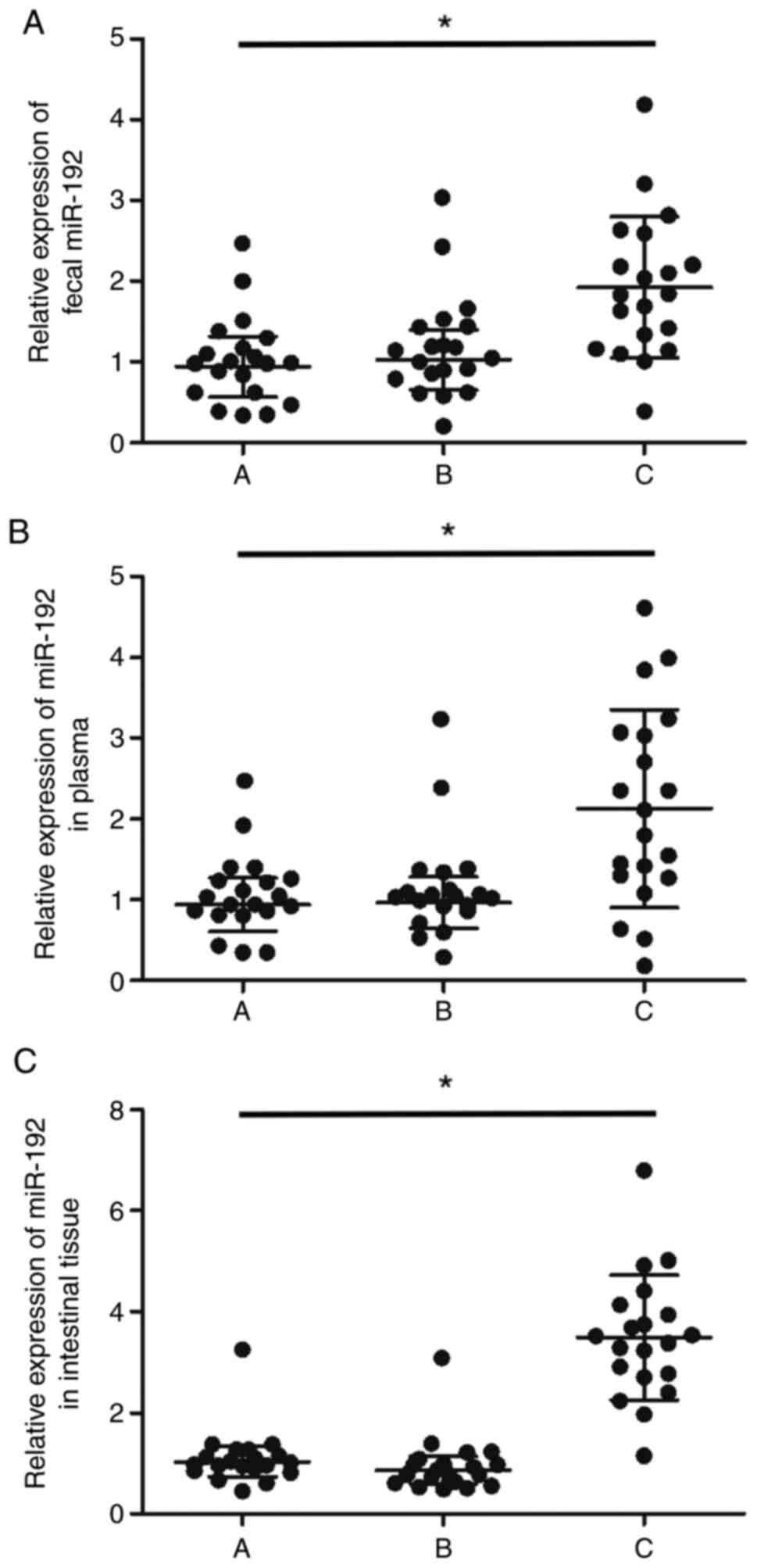
Upregulated miR-192-5p expression in
the feces, peripheral blood and intestinal mucosal tissue samples
of HBsAg-positive patients with high ALT
Then, the peripheral blood, intestinal mucosal
tissue and fecal samples were collected from every patient to
measure their miR-192-5p expression. It was found that the
expression of miR-192-5p was significantly upregulated in the feces
(Fig. 2A), peripheral blood
(Fig. 2B) and intestinal mucosal
tissue (Fig. 2C) samples from the
HBsAg-positive patients with a high level of ALT compared with
other patients.
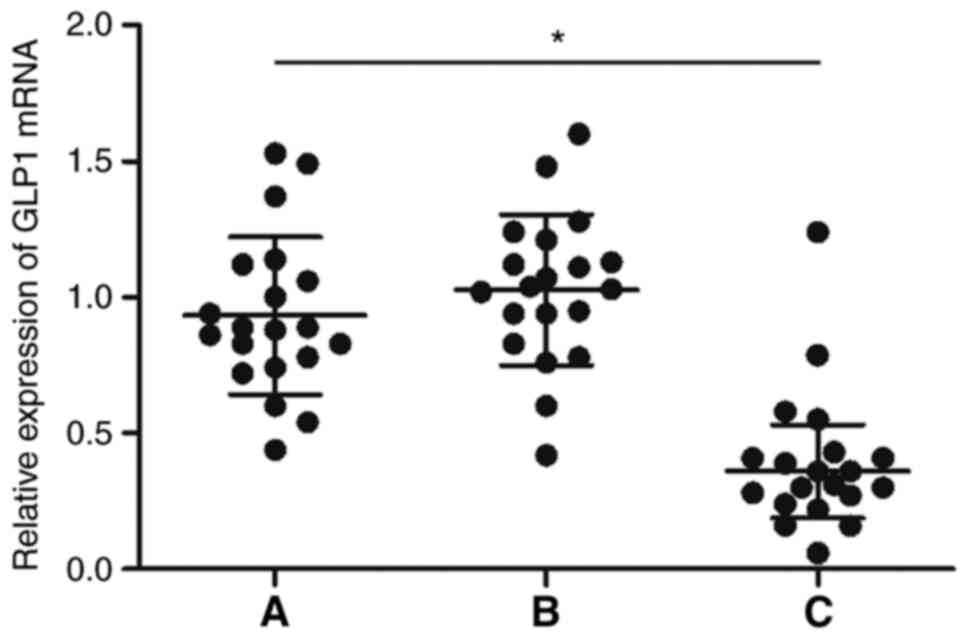
GLP-1 expression is suppressed in the
intestinal mucosal tissue samples of HBsAg-positive patients with
high ALT
RT-qPCR was performed to analyze the differential
expression of GLP-1 mRNA in the intestinal mucosal tissue samples
collected from patients of the three groups. The results clearly
showed that the expression of GLP-1 mRNA was significantly lower in
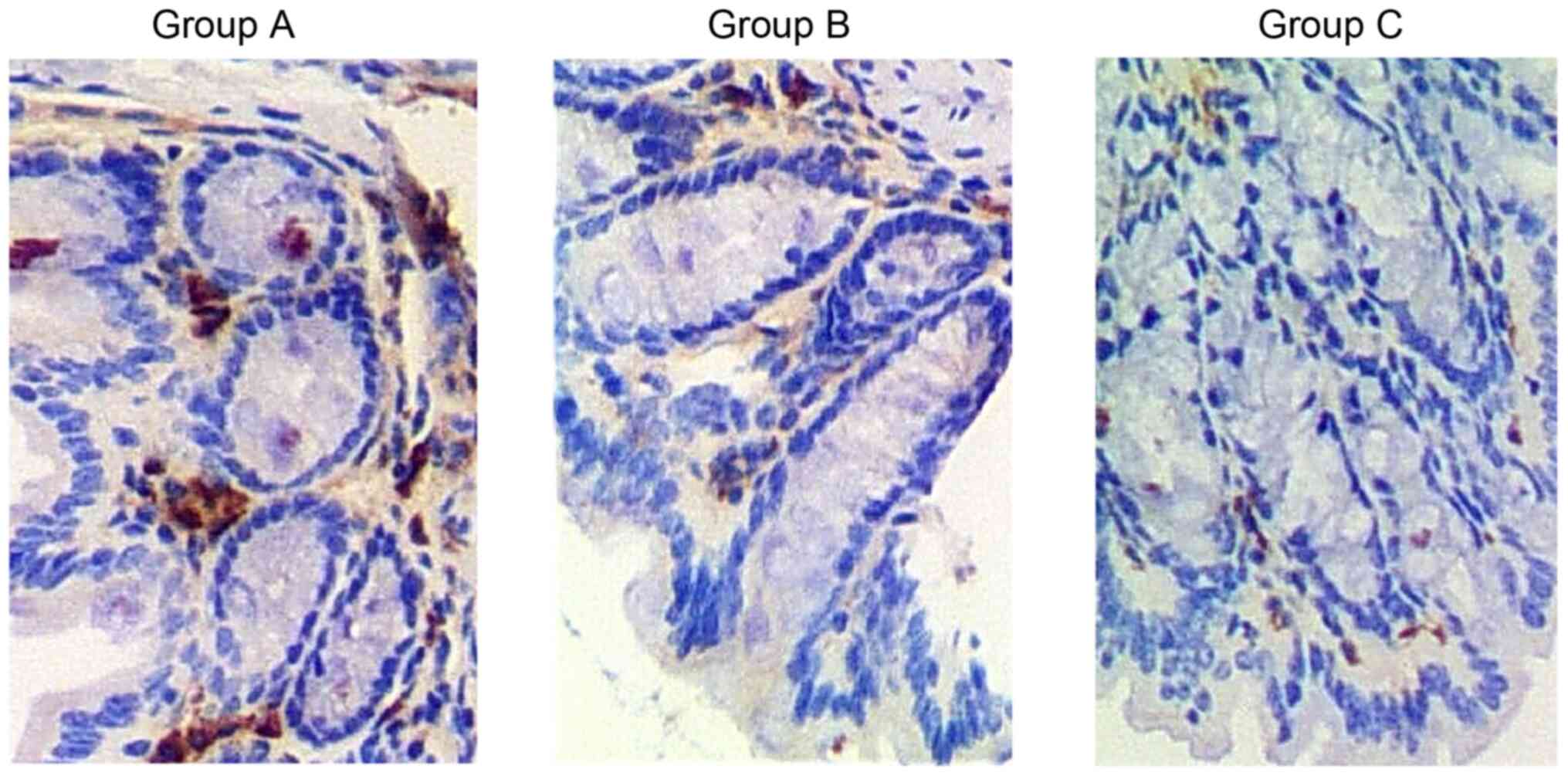
patients in Group C compared with that in Groups A and B (Fig. 3). Furthermore, immunohistochemistry
was carried out to explore the differential expression of GLP-1
proteins in a case representative of each of the three groups of
patients. Consistently, GLP-1 protein was evidently downregulated
in patients in Group C (Fig.
4).
miR-192-5p inhibits GLP-1 expression
by binding to the 3′ UTR of GLP-1
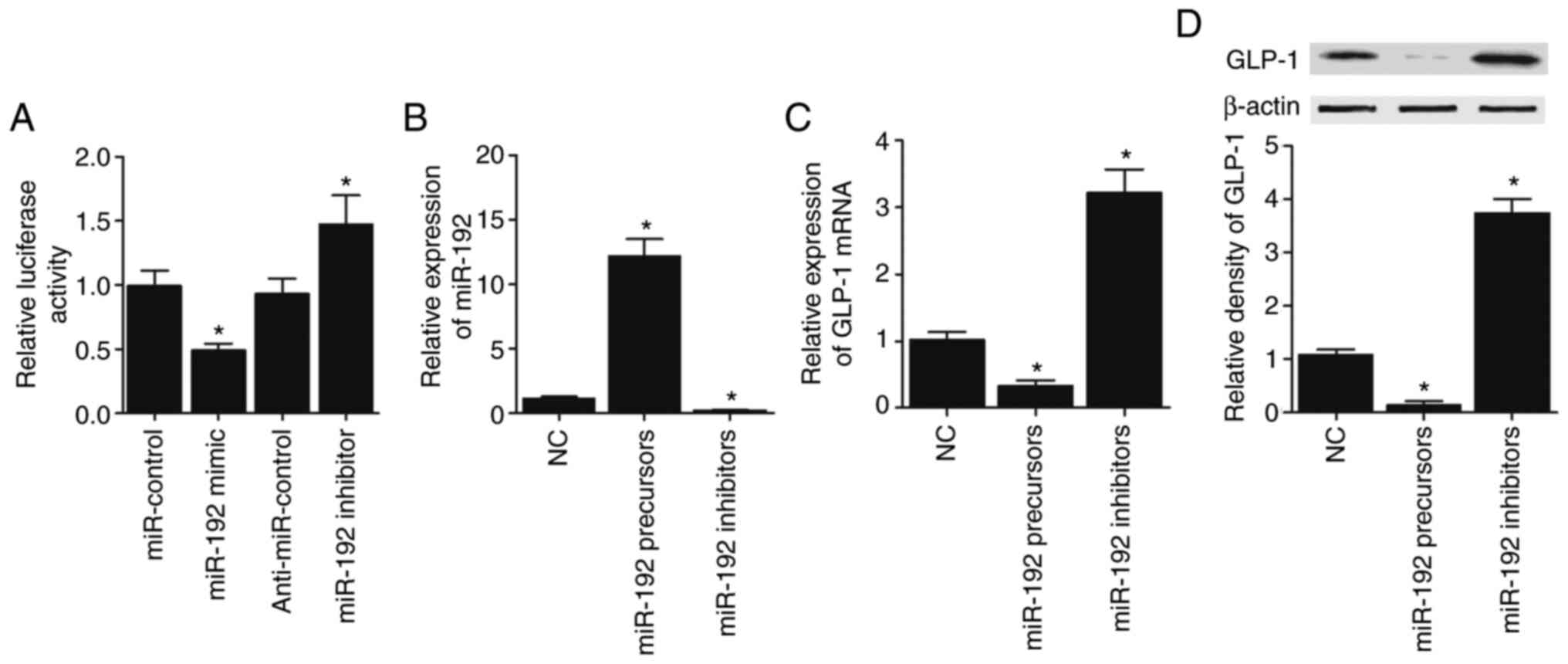
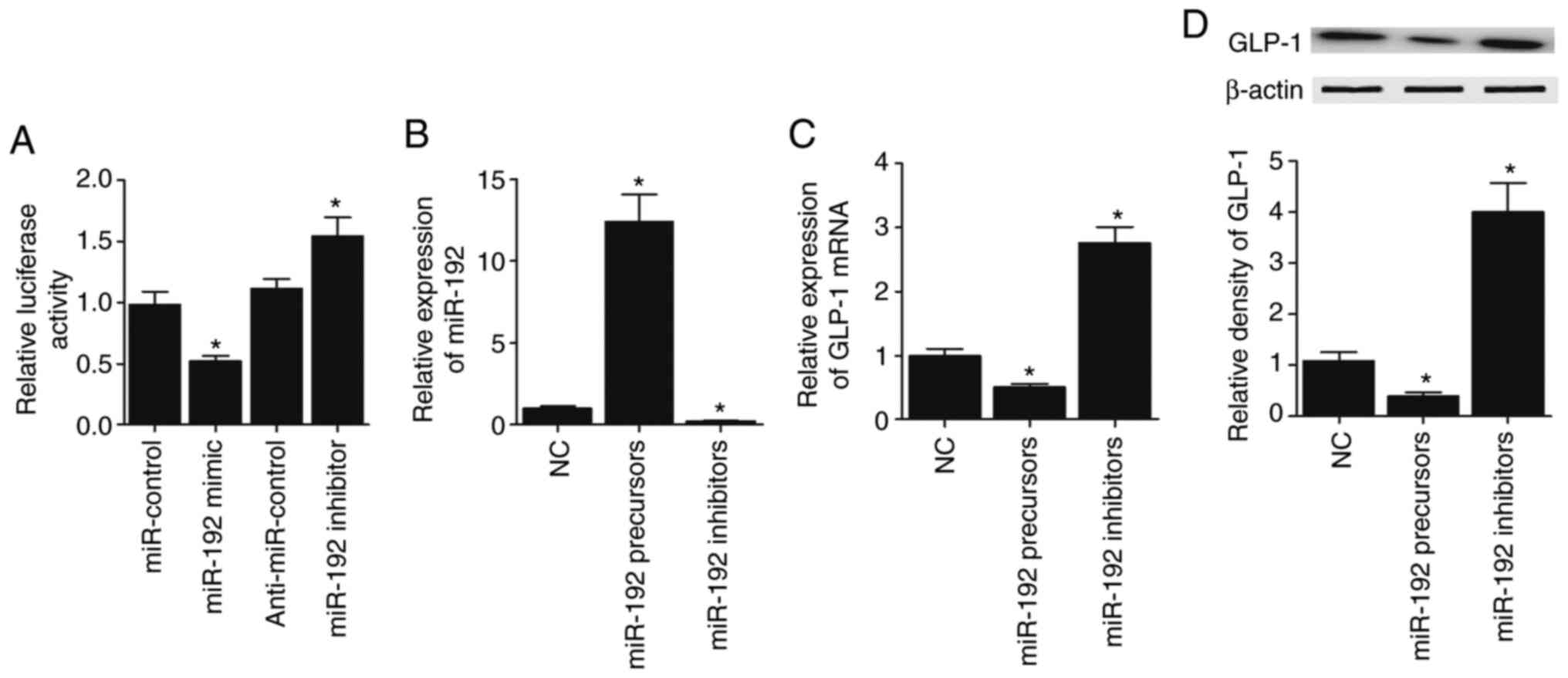
In order to explore the regulatory role of
miR-192-5p in GLP-1 expression, luciferase plasmids of wild-type
GLP-1 3′ UTR were constructed to co-transfect CAPAN-1 and HPAC
cells with miR-192-5p precursors, miR-192-5p inhibitors,
anti-miR-control and miR-control, respectively. miR-192-5p
effectively suppressed the luciferase activity of wild-type GLP-1
3′ UTR in CAPAN-1 (Fig. 5A) cells.
Then, miR-192-5p precursors and miR-192-5p inhibitors were
transfected into CAPAN-1 cells, and miR-192-5p expression was
significantly elevated in CAPAN-1 cells transfected with miR-192-5p
precursors but decreased in CAPAN-1 cells transfected with
miR-192-5p inhibitors (Fig. 5B). On
the contrary, GLP-1 mRNA (Fig. 5C)
and protein (Fig. 5D) expression
was significantly decreased in CAPAN-1 cells transfected with
miR-192-5p precursors and significantly increased by miR-192-5p
inhibitors. Furthermore, all the results described above were
confirmed in HPAC cells (Fig.
6A-D).
Discussion
HBV can induce severe liver disorders, such as
hepatocellular carcinoma, cirrhosis, and chronic and acute
hepatitis (30). In particular, a
number of patients with CHBVI are infected during early childhood,
whereas infection with HBV in adults can be caused by sharing
syringes and sexual intercourse (21,22,31).
In a previous controlled case study, it was found that CHBVI
apparently elevated the risks of pancreatic cancer in patients in
Korea and Taiwan (23). Moreover,
DM patients showed an apparently higher rate of CHBVI than patients
with DM who were not positive for PC. By contrast, it was also
shown that the patients with CHBVI had a lower level of B.
catenulatum. Therefore, it was suggested that B.
catenulatum could help to protect the liver against acute
injuries by restoring the normal status of intestinal microbiotas
to decrease the levels of endotoxin and cytokines in the stomach
(24,25). In the present study, three groups of
patients were recruited, Group A (HBsAg-negative control), group B
(HBsAg-positive with normal ALT levels) and group C (HBsAg-positive
with high ALT levels), to analyze the profiles of intestinal
microbiota in different groups. Total lactobacillus count was
obviously elevated in Group C patients. ALT levels are a signal of
liver lesions, and in patients with high or low ALT, the microbiota
was different, which interfered with miR-192/GLP-1 signaling. In
addition, RT-qPCR was performed to evaluate the expression of
miR-192-5p in the feces, peripheral blood and intestinal mucosal
tissue samples of patients in different groups, and the miR-192-5p
expression was notably increased in the patients in Group C.
miR-192-5p-5p expression is reduced in various types of cancer,
such as colorectal, renal and lung cancer. In addition, the
activation of miR-192-5p-5p has been shown to promote renal cell
apoptosis (10,32,33).
Moreover, Boni et al (26)
demonstrated that increased miR-192-5p expression also elevated the
efficacy of 5-fluoruracil in killing tumor cells of colon cancer by
inducing cell cycle arrest. Moreover, Hu et al (27) reported that miR-192-5p expression
was reduced in breast cancer. The present study compared the
expression of GLP-1 as a target of miR-192-5p-5p in the intestinal
mucosal tissue samples of patients in various groups, and GLP-1
expression was found to be remarkably decreased in patients in
Group C.
It has been demonstrated that fecal miRNAs are
essential components in the lumen of the stomach (34). Moreover, fecal miRNAs are mainly
produced by epithelial cells in the intestine (35). In particular, fecal miRNAs help to
regulate stomach microbiota by targeting the genes of various
bacteria (34). The aforementioned
results indicate the important role of stomach microbiota in
maintaining the homeostasis of its host (36). Moreover, miR-192-5p expression is
increased in DM to suppress the secretion of insulin and the
proliferation of β-cells (37).
Moreover, miR-192-5p can suppress the proliferation of NIT-1 cells,
while promoting their apoptosis (16). In the present study, luciferase
assays were performed to explore the regulatory relationship
between miR-192-5p and GLP-1, and the results showed that
miR-192-5p suppressed the luciferase activity of GLP-1 vector. In
addition, CAPAN-1 and HPAC cells were transfected with miR-192-5p
precursors and inhibitors. The transfection of miR-192-5p
precursors effectively upregulated the expression of miR-192-5p,
but suppressed the expression of GLP-1, while the transfection of
miR-192-5p inhibitors inhibited the expression of miR-192-5p but
promoted the expression of GLP-1. In line with this, a previous
study showed suppressed expression of GLP-1 in the presence of
miR-192-5p (37), suggesting that
miR-192-5p can promote DM development by negatively regulating
GLP-1 expression.
GLP-1 is released by L-cells living in small
intestines and can increase the secretion of insulin from β-cells
while reducing the production of glucose in the liver (38,39).
GLP-1 can also participate in the physiological process of weight
loss (40,41). The dosing of GLP-1 in mice was found
to reduce sucrose responses, while the suppression of GLP-1
expression increased food intake (42,43).
Increased GLP-1 expression can promote insulin
secretion to accelerate the metabolism of glucose, while the
release of GLP-1 is elevated by adding fatty acids, glucose,
dietary fibers and amino acids into the diet (41). In addition, using a diet containing
fatty acids, proteins and glucose rather than glucose by itself,
the prognosis of patients with DM can be improved (41).
In conclusion, the present results demonstrated the
association between HBV infection and the composition of gut
microbiota in patients with DM. HBV infection can affect the
composition of the gut microbiota, leading to an imbalanced
proportion of Lactobacillus. Therefore, the gut microbiota
composition was altered in patients with different HBV-infection
statuses and serum ALT levels, indicating the presence of a
potential link between the severity of DM and the expression of
miR-192-5p and GLP-1.
Acknowledgements
Not applicable.
Funding
This study was funded by The Medical Application
Technology Tracking Project of Hebei Province (grant no.
G201734).
Availability of data and materials
The datasets used and/or analyzed during the current
study are available from the corresponding author upon reasonable
request.
Author's contributions
YL designed the study, collected the experimental
data, analyzed the data, confirmed the authenticity of all the raw
data and wrote the manuscript.
Ethics approval and consent to
participate
All procedures were approved by the ethics committee
of The Third Hospital of Hebei Medical University (approval no.
CTHHMU2015037) and written informed consent was obtained from
participants before the initiation of this study.
Patient consent for publication
Not applicable.
Competing interests
The author declares that he has no competing
interests.
References
|
1
|
Beasley RP: Rocks along the road to the
control of HBV and HCC. Ann Epidemiol. 19:231–234. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Cho EJ, Kim SE, Suk KT, An J, Jeong SW,
Chung WJ and Kim YJ: Current status and strategies for hepatitis B
control in Korea. Clin Mol Hepatol. 23:205–211. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Younossi Z, Kochems K, de Ridder M, Curran
D, Bunge EM and de Moerlooze L: Should adults with diabetes
mellitus be vaccinated against hepatitis B virus? A systematic
review of diabetes mellitus and the progression of hepatitis B
disease. Hum Vaccin Immunother. 13:2695–2706. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Szabo G and Bala S: Alcoholic liver
disease and the gut-liver axis. World J Gastroenterol.
16:1321–1329. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Zhang D, Hao X, Xu L, Cui J, Xue L and
Tian Z: Intestinal flora imbalance promotes alcohol-induced liver
fibrosis by the TGFβ/smad signaling pathway in mice. Oncol Lett.
14:4511–4516. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Mou H, Yang F, Zhou J and Bao C:
Correlation of liver function with intestinal flora, vitamin
deficiency and IL-17A in patients with liver cirrhosis. Exp Ther
Med. 16:4082–4088. 2018.PubMed/NCBI
|
|
7
|
Macfarlane LA and Murphy PR: MicroRNA:
Biogenesis, function and role in cancer. Curr Genomics. 11:537–561.
2010. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Fisher K and Lin J: MicroRNA in
inflammatory bowel disease: Translational research and clinical
implication. World J Gastroenterol. 21:12274–12282. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Peck BC, Weiser M, Lee SE, Gipson GR, Iyer
VB, Sartor RB, Herfarth HH, Long MD, Hansen JJ, Isaacs KL, et al:
MicroRNAs classify different disease behavior phenotypes of crohn's
disease and may have prognostic utility. Inflamm Bowel Dis.
21:2178–2187. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Chiang Y, Song Y, Wang Z, Liu Z, Gao P,
Liang J, Zhu J, Xing C and Xu H: microRNA-192, −194 and −215 are
frequently downregulated in colorectal cancer. Exp Ther Med.
3:560–566. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Jones MF, Hara T, Francis P, Li XL, Bilke
S, Zhu Y, Pineda M, Subramanian M, Bodmer WF and Lal A: The
CDX1-microRNA-215 axis regulates colorectal cancer stem cell
differentiation. Proc Natl Acad Sci USA. 112:E1550–E1558. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Fischer J, Walker LC, Robinson BA,
Frizelle FA, Church JM and Eglinton TW: Clinical implications of
the genetics of sporadic colorectal cancer. ANZ J Surg.
89:1224–1229. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Moloney GM, Viola MF, Hoban AE, Dinan TG
and Cryan JF: Faecal microRNAs: Indicators of imbalance at the
host-microbe interface? Benef Microbes. 9:175–183. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Dalmasso G, Nguyen HT, Yan Y, Laroui H,
Charania MA, Ayyadurai S, Sitaraman SV and Merlin D: Microbiota
modulate host gene expression via microRNAs. PLoS One.
6:e192932011. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Liu S, da Cunha AP, Rezende RM, Cialic R,
Wei Z, Bry L, Comstock LE, Gandhi R and Weiner HL: The host shapes
the gut Microbiota via fecal MicroRNA. Cell Host Microbe. 19:32–43.
2016. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Pan W, Zhang Y, Zeng C, Xu F, Yan J and
Weng J: miR-192 is upregulated in T1DM, regulates pancreatic β-cell
development and inhibits insulin secretion through suppressing
GLP-1 expression. Exp Ther Med. 16:2717–2724. 2018.PubMed/NCBI
|
|
17
|
Yun Y, Chang Y, Kim HN, Ryu S, Kwon MJ,
Cho YK, Kim HL, Cheong HS and Joo EJ: Alterations of the gut
microbiome in chronic hepatitis B virus infection associated with
alanine aminotransferase level. J Clin Med. 8:1732019. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Lin PY, Chen SC, Lo TC and Kuo HW: Dual
infection with hepatitis B virus and hepatitis C virus correlated
with type 2 diabetes mellitus. Exp Clin Endocrinol Diabetes.
128:38–42. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Xie QH, He XX, Chang Y, Jiang X and Lin
JS: HBx gene down-regulates miR-192 expression and inhibits
apoptosis of human hepatoma cell line HepG2. Zhonghua Gan Zang Bing
Za Zhi. 19:857–860. 2011.(In Chinese). PubMed/NCBI
|
|
20
|
Yuan X, Ni H, Chen X, Feng X, Wu Q and
Chen J: Identification of therapeutic effect of glucagon-like
peptide 1 in the treatment of STZ-induced diabetes mellitus in rats
by restoring the balance of intestinal flora. J Cell Biochem.
119:10067–10074. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Shin EC, Sung PS and Park SH: Immune
responses and immunopathology in acute and chronic viral hepatitis.
Nat Rev Immunol. 16:509–523. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Ou JH: Molecular biology of hepatitis B
virus e antigen. J Gastroenterol Hepatol. 12:S178–S187. 1997.
View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Ben Q, Li Z, Liu C, Cai Q, Yuan Y, Wang K,
Xiao L, Gao J and Zhang H: Hepatitis B virus status and risk of
pancreatic ductal adenocarcinoma: A case-control study from China.
Pancreas. 41:435–440. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Li YT, Wang L, Chen Y, Chen YB, Wang HY,
Wu ZW and Li LJ: Effects of gut microflora on hepatic damage after
acute liver injury in rats. J Trauma. 68:76–83. 2010.PubMed/NCBI
|
|
25
|
Neelam K, Goenadi CJ, Lun K, Yip CC and Au
Eong KG: Putative protective role of lutein and zeaxanthin in
diabetic retinopathy. Br J Ophthalmol. 101:551–558. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Boni V, Bitarte N, Cristobal I, Zarate R,
Rodriguez J, Maiello E, Garcia-Foncillas J and Bandres E:
miR-192/miR-215 influence 5-fluorouracil resistance through cell
cycle-mediated mechanisms complementary to its post-transcriptional
thymidilate synthase regulation. Mol Cancer Ther. 9:2265–2275.
2010. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Hu F, Meng X, Tong Q, Liang L, Xiang R,
Zhu T and Yang S: BMP-6 inhibits cell proliferation by targeting
microRNA-192 in breast cancer. Biochim Biophys Acta.
1832:2379–2390. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) method. Methods. 25:402–408. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Agarwal V, Bell GW, Nam JW and Bartel DP:
Predicting effective microRNA target sites in mammalian mRNAs.
Elife. 4:e050052015. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Hutin YJ, Bulterys M and Hirnschall GO:
How far are we from viral hepatitis elimination service coverage
targets? J Int AIDS Soc. 21 (Suppl 2):e250502018. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Milich D and Liang TJ: Exploring the
biological basis of hepatitis B e antigen in hepatitis B virus
infection. Hepatology. 38:1075–1086. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Khella HW, Bakhet M, Allo G, Jewett MA,
Girgis AH, Latif A, Girgis H, Von Both I, Bjarnason GA and Yousef
GM: miR-192, miR-194 and miR-215: A convergent microRNA network
suppressing tumor progression in renal cell carcinoma.
Carcinogenesis. 34:2231–2239. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Zhang L, Zhou W, Velculescu VE, Kern SE,
Hruban RH, Hamilton SR, Vogelstein B and Kinzler KW: Gene
expression profiles in normal and cancer cells. Science.
276:1268–1272. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Rashid H, Hossain B, Siddiqua T, Kabir M,
Noor Z, Ahmed M and Haque R: Fecal microRNAs as potential
biomarkers for screening and diagnosis of intestinal diseases.
Front Mol Biosci. 7:1812020. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Sarshar M, Scribano D, Ambrosi C, Palamara
AT and Masotti A: Fecal microRNAs as innovative biomarkers of
intestinal diseases and effective players in host-microbiome
interactions. Cancers (Basel). 12:21742020. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Honda K and Littman DR: The microbiome in
infectious disease and inflammation. Annu Rev Immunol. 30:759–795.
2012. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Kim M and Zhang X: The profiling and role
of miRNAs in diabetes mellitus. J Diabetes Clin Res. 1:5–23.
2019.PubMed/NCBI
|
|
38
|
Holst JJ, Vilsboll T and Deacon CF: The
incretin system and its role in type 2 diabetes mellitus. Mol Cell
Endocrinol. 297:127–136. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Kieffer TJ and Habener JF: The
glucagon-like peptides. Endocr Rev. 20:876–913. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Flint A, Raben A, Astrup A and Holst JJ:
Glucagon-like peptide 1 promotes satiety and suppresses energy
intake in humans. J Clin Invest. 101:515–520. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Verdich C, Flint A, Gutzwiller JP, Näslund
E, Beglinger C, Hellström PM, Long SJ, Morgan LM, Holst JJ and
Astrup A: A meta-analysis of the effect of glucagon-like peptide-1
(7–36) amide on ad libitum energy intake in humans. J Clin
Endocrinol Metab. 86:4382–4389. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Dickson SL, Shirazi RH, Hansson C,
Bergquist F, Nissbrandt H and Skibicka KP: The glucagon-like
peptide 1 (GLP-1) analogue, exendin-4, decreases the rewarding
value of food: A new role for mesolimbic GLP-1 receptors. J
Neurosci. 32:4812–4820. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Meeran K, O'Shea D, Edwards CM, Turton MD,
Heath MM, Gunn I, Abusnana S, Rossi M, Small CJ, Goldstone AP, et
al: Repeated intracerebroventricular administration of
glucagon-like peptide-1-(7–36) amide or exendin-(9–39) alters body
weight in the rat. Endocrinology. 140:244–250. 1999. View Article : Google Scholar : PubMed/NCBI
|